Development of a Molecular Breeding Strategy for the Integration of Transgenic Traits in Outcrossing Perennial Grasses
Abstract
:1. Introduction
- screening agronomically superior populations for tissue culture responsive (TCR) genotypes to be used in genetic transformation;
- screening and selecting both T0 events and recipient genotypes concurrently;
- ensuring agronomically fit, genetically-diverse, recipient genotypes were used in crossing,
- introducing endophytes in the T1 generation via the recipient parent; and
- attempting to complete two cycles of crossing within one year.
- selection methods of tissue culture responsive genotypes;
- development, evaluation, and selection of primary T0 transgenic events in Lolium;
- integration of the gene technologies into the wider breeding population; and
- evaluation of progeny for trait stability and agronomic performance.
2. Selection Methods for Tissue Culture Responsive Genotypes
3. Development, Evaluation and Selection of Primary T0 Transgenic Events in Lolium Grasses
4. Introgression of the Transgene into the Wider Breeding Population
5. Evaluation of Progeny for Transgenic Trait Stability and Agronomic Performance
6. An Optimum Transgenic Breeding Strategy in Lolium Grasses that Is Compatible with Genomic Selection
- Year 1:
- (i)
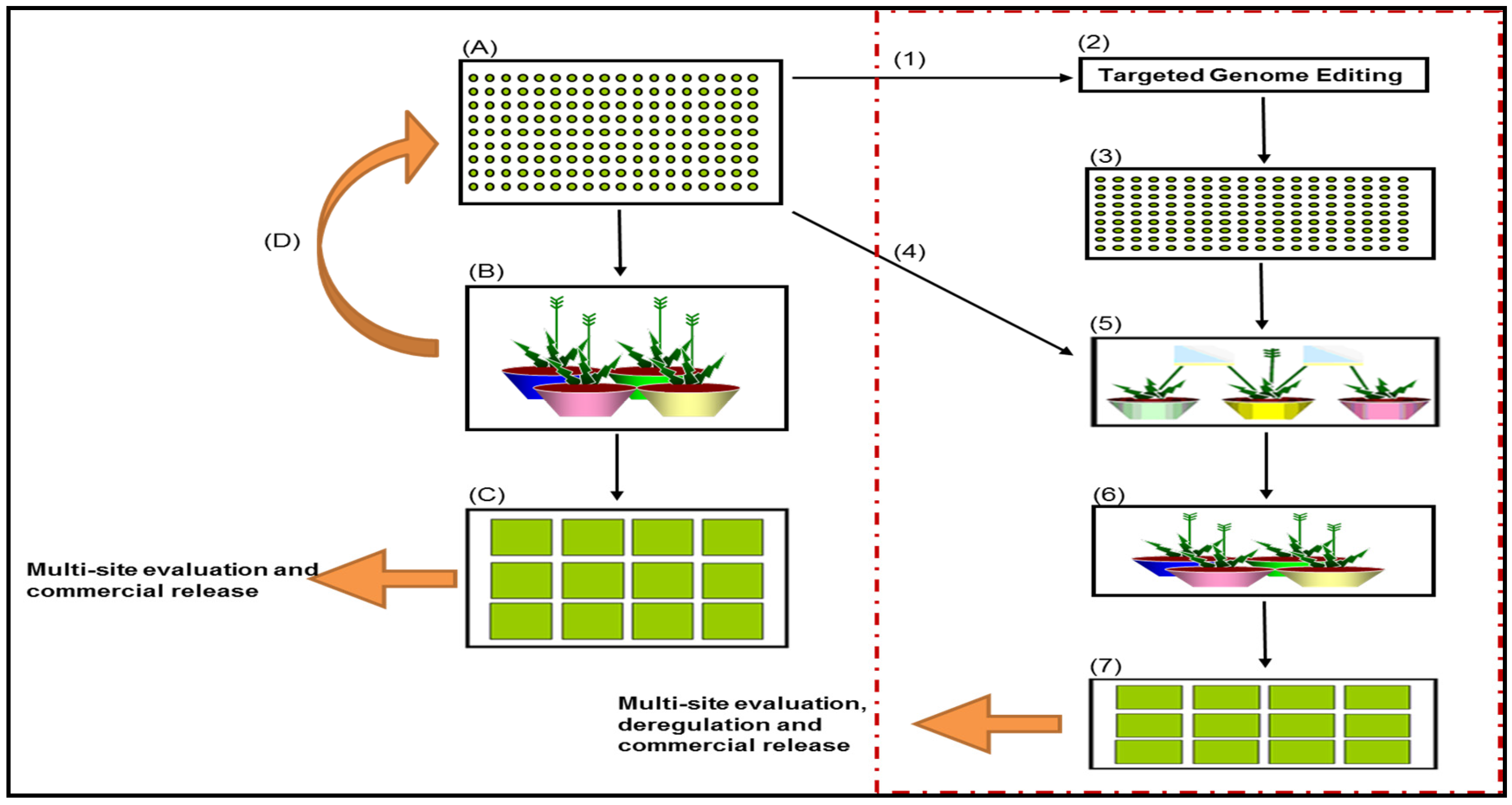
- Selection of TCR and agronomically superior genotypes for transformation—Figure 1, number 1.Selection of superior genotypes from the genomic breeding nursery (Figure 1A) for transformation, based on genomic predictions of performance for a range of agronomic traits, as well as the TCR trait that is required both for callus induction and callus regeneration during transformation.
- (ii)
- Creation of primary T0 transgenic events in Lolium grasses through targeted genome editing—Figure 1, number 2.Generation of primary T0 events through targeted genome editing technology. Genome editing can accurately delete, substitute, or add nucleotides at a specific site within the genome [35]. This technology has distinct advantages over Agrobacterium-mediated transformation and biolistic transformation in that the transgene integration is site specific, which has the potential to reduce insertional mutagenesis, complex integration patterns, and multiple copy insertions [35]. It will also reduce the burden of identifying and characterising insertion sites for deregulation purposes, as the integration site is known and targeted for [35].
- (iii)
- Application to Regulator for the limited release of transgenic Lolium grasses for evaluation.Transgenic technology is governed by the Gene Technology Act 2000 and regulated by the Office of the Gene Technology Regulator in Australia. A licence by the Regulator is required for the intentional release of a transgenic event into the environment.
- Year 2–3:
- (i)
- Evaluation and selection of primary transgenic T0 events in Lolium grasses—Figure 1, number 3.Phase A: Pre-screening of T0 events for transgene expression and qualitative traits under containment conditions, if possible.Phase B: Establishment and evaluation of pre-screened T0 events in a clonally-replicated, space-planted field trial for agronomic performance and performance of the targeted trait.Phase C: Select 5–10 primary T0 events for progression, based on “transgene traits” and transgene expression levels under field conditions. Data is based on phenotypic evaluation.
- (ii)
- Selection of recipient genotypes to cross with the primary T0 transgenic events—Figure 1, number 4.Selection of genetically-diverse recipient genotypes from the genomic breeding nursery (Figure 1, box A) based on genomic predictions of diversity and performance for a range of traits and endophyte presence.
- Year 4:
- (i)
- Pair-crossing of recipient genotypes and primary T0 transgenic events—Figure 1, number 5.Four to eight genetically-diverse recipient genotypes (with endophyte) are to be pair-crossed to each of the selected T0 events (5–10 events). Harvest seed from each recipient parent (endophyte present) as a half-sib family.
- (ii)
- Selection of T1 progeny for trait stability and agronomic performance.Phase A: Screen all half-sib families for transgene presence. Transgenic events that exhibit a monogenic Mendelian segregation ratio of 1:1 for the transgene can be progressed. Using targeted genome editing, trait inheritance and trait expression should be more predictable in the progeny compared to alternative transformation methods.Phase B: T1 plants from selected events can be screened using genomic predictions for a range of traits/all traits. 4–6 T1 plants that are genetically-diverse and show high transgene expression, can be selected for synthetic cultivar development through poly-crossing.
- (iii)
- Poly-crossing of selected T1 events—Figure 1, number 6.Phase A: Poly-crosses are made for each selected transgenic event (3–5 events). All crosses are 4–6 parent synthetics. Seed should be harvested from each parent as a half-sib family. Molecular analysis for each half-sib family should indicate a segregation ratio of 1:2:1 for the transgene in the T2/F2 progeny.Phase B: T2 progeny homozygous for the gene of interest GOI can be used as Syn0 parents in a synthetic breeding program. The Syn0 seed from the 3–5 transgenic events can progress to mini-sward evaluations.
- Year 5–6:
- (i)
- Mini-sward field evaluation of Syn0 progeny for cultivar development—Figure 1, number 7.All Phases: Mini swards are sown as Syn0 half-sib populations. Each of the potential 4–6 parent synthetic populations, (i.e., the 4–6 mini-swards) will need to be observed together for assessment of flowering time uniformity. Phenotypic evaluation of the transgenic trait is measured on a population basis. Choices in regards to which event to deregulate can be made at this point. If the trait is deregulated, the progeny of the Syn0 parents could be used in a recurrent breeding nursery or genomic selection nursery, where sub-selections can be made for varietal development.
7. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| TCR | Tissue Culture Responsive |
| T0 | Primary Transformant |
| T1 | First Generation Transformant |
| GEBV | Genomic Estimated Breeding Value |
| GOI | Gene of Interest |
References
- Wang, Z.Y.; Brummer, E.C. Is genetic engineering ever going to take off in forage, turf and bioenergy crop breeding? Ann. Bot. 2012, 110, 1317–1325. [Google Scholar] [CrossRef] [PubMed]
- OGTR. The Biology of Lolium multiflorum Lam. (Italian ryegrass), Lolium perenne L. (perennial ryegrass) and Lolium arundinaceum (Schreb.) Darbysh (Tall Fescue); Office of the Gene Technology Regulator: Canberra, Australia, 2008.
- Spangenberg, G.; Lidget, R.; Lidgett, A.; Sawbridge, T.; Ong, E.K.; John, U. Transgenesis and genomics in molecular breeding of forage plants. In Proceedings of the Australian Agronomy Conference, Hobart, Tasmania, 2001; Available online: http://www.regional.org.au/au/asa/2001 (accessed on 1 November 2016).
- Smith, K.F.; Forster, J.W.; Spangenberg, G.C. Converting genomic discoveries into genetic solutions for diary pastures. Aust. J. Exp. Agric. 2007, 47, 1032–1038. [Google Scholar] [CrossRef]
- Wang, Z.Y.; Ge, Y. Agrobacterium-mediated high efficiency transformation of tall fescue (Festuca arundinacea). J. Plant Physiol. 2005, 162, 103–114. [Google Scholar] [CrossRef] [PubMed]
- Woodfield, D.R.; White, D.W.R. Breeding strategies for developing transgenic white clover cultivars. Agron. Soc. N. Z. Spec. Publ. 1996, 11, 125–130. [Google Scholar]
- Smith, K.F.; Lewis, C.; Ludemann, C.I.; Malcolm, B.; Banks, R.G.; Jacobs, J.L.; Fennessy, P.F.; Spangenberg, G. Estimating the value of genetic gain in perennial pastures with emphasis on temperate species. Crop Pasture Sci. 2014, 65, 1238–1247. [Google Scholar] [CrossRef]
- Ludemann, C.I.; Eckard, R.J.; Cullen, B.C.; Jacobs, J.L.; Malcolm, B.; Smith, K.F. Higher energy concentration traits in perennial ryegrass (Lolium perenne L.) may increase farm profits and improve energy conversion on dairy farms. Agric. Syst. 2015, 137, 89–100. [Google Scholar] [CrossRef]
- Ludemann, C.I.; Smith, K.F. A comparison of methods to assess the likely on-farm value for meat production systems of pasture traits and genetic gain through plant breeding using phalaris (Phalaris aquatica L.) as an example. Grass Forage Sci. 2016, 71, 66–78. [Google Scholar] [CrossRef]
- Zhong, G.Y. Genetic issues and pitfalls in transgenic plant breeding. Euphytica 2001, 118, 137–144. [Google Scholar] [CrossRef]
- Visarada, K.; Meena, K.; Aruna, C.; Srujana, S.; Saikishore, N.; Seetharama, N. Transgenic breeding: Perspectives and prospects. Crop Sci. 2009, 49, 1555–1563. [Google Scholar] [CrossRef]
- Kalla, R.; Chu, P.G.; Spangenberg, G. Molecular breeding of forage legumes for virus resistance. In Molecular Breeding of Forages and Turf; Kluwer Academin Publishers: Dordrecht, The Netherlands; Boston, MA, USA; London, UK, 2001; Volume 10, pp. 219–237. [Google Scholar]
- Lin, Z.; Cogan, N.O.I.; Pembleton, L.W.; Spangenberg, G.C.; Forster, J.W.; Hayes, B.J.; Daetwyler, H.D. Genetic Gain and Inbreeding from Genomic Selection in a Simulated Commercial Breeding Program for Perennial Ryegrass. Plant Genome 2016. [Google Scholar] [CrossRef]
- Pembleton, L.W.; Shinozukam, H.; Wang, J.; Spangenberg, G.C.; Forster, J.W.; Cogan, N.O.I. Design of an F1 hybrid breeding strategy for ryegrass based on selection of self-incompatibility locus-specific alleles. Front. Plant Sci. 2015, 6, 764. [Google Scholar] [CrossRef] [PubMed]
- Belhaj, K.; Chaparro-Garcia, A.; Kamoun, S.; Nekrasov, V. Plant genome editing made easy: Targeted mutagenesis in model and crop plants using the CRISPR/Cas system. Plant Methods 2013, 9, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Hayes, B.J.; Cogan, N.O.I.; Pembleton, L.W.; Goddard, M.E.; Wang, J.; Spangenberg, G.C.; Forster, J.W. Prospects for genomic selection in forage plant species. Plant Breed. 2013, 132, 133–143. [Google Scholar] [CrossRef]
- Simeão Resende, R.M.; Casler, M.D.; Vilela de Resende, M.D. Genomic selection in forage breeding: Accuracy and methods. Crop Sci. 2014, 54, 143–156. [Google Scholar] [CrossRef]
- Wang, J.; Pembleton, L.W.; Baillie, R.C.; Drayton, M.C.; Hand, M.L.; Bain, M.; Sawbridge, T.; Spangenberg, G.C.; Forster, J.W.; Cogan, N.O. Development and implementation of a multiplexed single nucleotide polymorphism genotyping tool for differentiation of ryegrass species and cultivars. Mol. Breed. 2014, 33, 435–451. [Google Scholar] [CrossRef]
- Badenhorst, P.E.; Panter, S.; Palanisamy, R.; Georges, S.; Smith, K.F.; Mouradov, A.; Mason, J.; Spangenberg, G. Molecualr breeding of transgenic perennial ryegrass (Lolium perenne L.) with altered fructan biosynthesis through the expression of fructoyltransferases. Mol. Breed. (submitted) 2017. [Google Scholar]
- Bradley, D.E.; Bruneau, A.H.; Qu, R. Effects of cultivar, explant treatment, and medium suppliments on callus induction and plantlet regeneration in perennial ryegrass. Int. Turfgrass Soc. Res. J. 2001, 9, 152–155. [Google Scholar]
- Heide, O.M. Control of flowering and reproduction in temperate grasses. New Phytol. 1994, 128, 347–362. [Google Scholar] [CrossRef]
- Langer, R.H.M. How Grasses Grow; Edward Arnold: London, UK, 1972. [Google Scholar]
- Sharman, B.C. Leaf and bud initiation in the gramineae. Bot. Gaz. 1945, 106, 269–289. [Google Scholar] [CrossRef]
- Aamlid, T.S.; Heide, O.M.; Boelt, B. Primary and secondary induction requirements for flowering of contrasting European varieties of Lolium perenne. Ann. Bot. 2001, 86, 1087–1095. [Google Scholar] [CrossRef]
- Meyer, W.A.; Watkins, E. Tall Fescue (Festuca arundinacea). In Turfgrass Biology Genetics and Breeding; Casler, M.D., Duncan, R.R., Eds.; John Wiley & Sons: Hobken, New Jersey, USA, 2003; pp. 107–127. [Google Scholar]
- Cooper, J.P. Short-day and low-temperature induction in Lolium. Ann. Bot. 1960, 24, 232–247. [Google Scholar]
- Spangenberg, G.S.; Wang, Z.Y.; Potrykus, I. Biotechnology in Forage and Turf Grass Improvement; Springer: Berlin, Germany, 1998. [Google Scholar]
- Wang, Z.Y.; Scott, M.; Bell, J.; Hopkins, A.; Lehmann, D. Field performance of transgenic tall fescue (Festuca arundinacea Schreb.) plants and their progenies. Theor. Appl. Genet. 2003, 107, 406–412. [Google Scholar] [CrossRef] [PubMed]
- Barampuram, S.; Zhang, Z.J. Recent advances in plant transformation. Methods Mol. Biol. 2011, 701, 1–35. [Google Scholar] [PubMed]
- Birch, R.G. Plant transformation: Problems and strategies for practical application. Ann. Rev. Plant Physiol. Plant Mol. Biol. 1997, 48, 297–326. [Google Scholar] [CrossRef] [PubMed]
- Hooykaas, P.J.J. Plant transformation. In Encyclopedia of Life Sciences; John Wiley and Sons, Ltd.: Hoboken, NJ, USA, 2010; Available online: http://onlinelibrary.wiley.com/doi/10.1002/9780470015902.a0003070.pub2/full (accessed on 1 November 2016).
- Newell, C.A. Plant transformation technology. Mol. Biotechnol. 2000, 16, 53–65. [Google Scholar] [CrossRef]
- Rivera, A.L.; Gomez-Lim, M.; Fernandez, F.; Loske, A.M. Physical methods for genetic plant transformation. Phys. Life Rev. 2012, 9, 308–345. [Google Scholar] [CrossRef] [PubMed]
- Wilson, A.K.; Latham, J.R.; Steinbrecher, R.A. Trnasformation-induced mutations in transgenic plants: Analysis and biosafety implications. Biotechnol. Genet. Eng. Rev. 2006, 23, 209–234. [Google Scholar] [CrossRef] [PubMed]
- Urnov, F.D.; Rebar, E.J.; Holmes, M.C.; Zhang, H.S.; Gregory, P.D. Genome editing with engineered zinc finger nucleases. Nat. Rev. Genet. 2010, 11, 636–646. [Google Scholar] [CrossRef] [PubMed]
- Wozniak, C.A.; McHughen, A. Regulation of Agricultural Biotechnology: The United States and Canada; Springer: Dordrecht, The Netherlands; Heidelberg, Germany; New York, NY, USA; London, UK, 2013. [Google Scholar]
- Shukla, V.K.; Doyon, Y.; Miller, J.C.; DeKelver, R.C.; Moehle, E.A.; Worden, S.E.; Mitchell, J.C.; Arnold, N.L.; Gopalan, S.; Meng, X.; et al. Precise genome modification in the crop species Zea mays using zinc-finger nucleases. Nature 2009, 459, 437–441. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Liu, B.; Spalding, M.H.; Weeks, D.P.; Yang, B. High-efficiency TALEN-based gene editing produces disease-resistant rice. Nat. Biotechnol. 2012, 30, 390–392. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, S.K.; Kumar, J.; Alok, A.; Tuli, R. RNA-guided genome editing for target gene mutations in wheat. G3 2013, 3, 2233–2238. [Google Scholar] [CrossRef] [PubMed]
- Wendt, T.; Holm, P.B.; Starker, C.G.; Christian, M.; Voytas, D.F.; Brinch-Pedersen, H.; Holme, I.B. TAL effector nucleases induce mutations at a pre-selected location in the genome of primary barley transformants. Plant Mol. Biol. 2013, 83, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Curtin, S.J.; Anderson, J.E.; Starker, C.G.; Baltes, N.J.; Mani, D.; Voytas, D.F.; Stupar, R.M. Targeted mutagenesis for functional analysis of gene duplication in legumes. Methods Mol. Biol. 2013, 1069, 25–42. [Google Scholar] [PubMed]
- Bradford, K.J.; Van Deynze, A.; Gutterson, N.; Parrott, W.; Strauss, S.H. Reguilating transgenic crops sensibly: Lessons from plant breeding, biotechnology and genomics. Nat. Biotechnol. 2005, 23, 439–444. [Google Scholar] [CrossRef] [PubMed]
- De Greef, W. GM Crops: The Cruching cost of Regulation. 2011. Available online: http://www.agbioworld.org/biotech-info/articles/biotech-art/crushingcost.html (accessed on 1 November 2016).
- Wilkins, P.W.; Humphreys, M.O. Progress in breeding perennial forage grasses for temperate agriculture. J. Agric. Sci. 2003, 140, 129–150. [Google Scholar] [CrossRef]
- Huyghe, C. Sustainable Use of Genetic Diversity in Forage and Turf Breeding; INRA—Poitou Charentes: Lusignan, France, 2010. [Google Scholar]
- Ladizinsky, G. Founder effect in crop-plant evolution. Econ. Bot. 1985, 39, 191–200. [Google Scholar] [CrossRef]
- Kidwell, K.K.; Hartweck, L.M.; Yandell, B.S.; Crump, P.M.; Brummer, J.E.; Moutray, J.; Osborn, T.C. Forage yields of alfalfa populations derived from parents selected on the basis of molecular marker dfiversity. Crop Sci. 1999, 39, 223–227. [Google Scholar] [CrossRef]
- Fehr, W.R. Principles of Cultivar Development; MacMillan: New York, NY, USA, 1987. [Google Scholar]
- Fejer, S.O. Genetic and environmental components of the productivity of perennial ryegrass (Lolium perenne L.). N. Z. J. Agric. Res. 1958, 1, 86–104. [Google Scholar]
- Van Zijl de Jong, E.; Dobrowolski, M.P.; Bannan, N.R.; Stewart, A.; Smith, K.F.; Spangenberg, G.; Forster, J. Global genetic diversity of the perennial ryegrass fungal endophyte Neotyphodium lolii. Crop Sci. 2008, 48, 1487–1501. [Google Scholar] [CrossRef]
- Pawlowski, W.P.; Somers, D.A. Transgene inheritance in plants genetically engineered by microprojectile bombardment. Mol. Biotechnol. 1996, 6, 17–30. [Google Scholar] [CrossRef] [PubMed]
- Yin, Z.; Plader, W.; Malepszy, S. Transgene inheritance in plants. J. Appl. Genet. 2004, 45, 127–144. [Google Scholar] [PubMed]
- Daetwyler, H.D.; Pong-Wong, R.; Villanueva, B.; Woolliams, J.A. The impact of genetic architechture on genome-wide evaluation methods. Genetics 2010, 185, 1021–1031. [Google Scholar] [CrossRef] [PubMed]
- Hayes, B.J.; Bowman, P.J.; Chamberlain, A.J.; Goddard, M.E. Invited review: Genomic selection in dairy cattle: Progress and challenges. J. Dairy Sci. 2009, 92, 433–443. [Google Scholar] [CrossRef] [PubMed]
- Fe, D.; Cericola, F.; Byrne, S.; Lenk, I.; Asraf, B.H.; Pedersen, M.G.; Roulund, N.; Asp, T.; Janss, L.; Jensen, C.S.; et al. Genomic dissection and prediction of heading date in perennial ryegrass. BMC Genom. 2015, 16, 921. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Badenhorst, P.E.; Smith, K.F.; Spangenberg, G. Development of a Molecular Breeding Strategy for the Integration of Transgenic Traits in Outcrossing Perennial Grasses. Agronomy 2016, 6, 56. https://doi.org/10.3390/agronomy6040056
Badenhorst PE, Smith KF, Spangenberg G. Development of a Molecular Breeding Strategy for the Integration of Transgenic Traits in Outcrossing Perennial Grasses. Agronomy. 2016; 6(4):56. https://doi.org/10.3390/agronomy6040056
Chicago/Turabian StyleBadenhorst, Pieter E., Kevin F. Smith, and German Spangenberg. 2016. "Development of a Molecular Breeding Strategy for the Integration of Transgenic Traits in Outcrossing Perennial Grasses" Agronomy 6, no. 4: 56. https://doi.org/10.3390/agronomy6040056
APA StyleBadenhorst, P. E., Smith, K. F., & Spangenberg, G. (2016). Development of a Molecular Breeding Strategy for the Integration of Transgenic Traits in Outcrossing Perennial Grasses. Agronomy, 6(4), 56. https://doi.org/10.3390/agronomy6040056





