Catch the Best: Novel Screening Strategy to Select Stress Protecting Agents for Crop Plants
Abstract
:1. Introduction
2. Results and Discussion
2.1. Bait Plant Strategy
2.2. Insight into Rhizosphere Communities Using Molecular Fingerprints
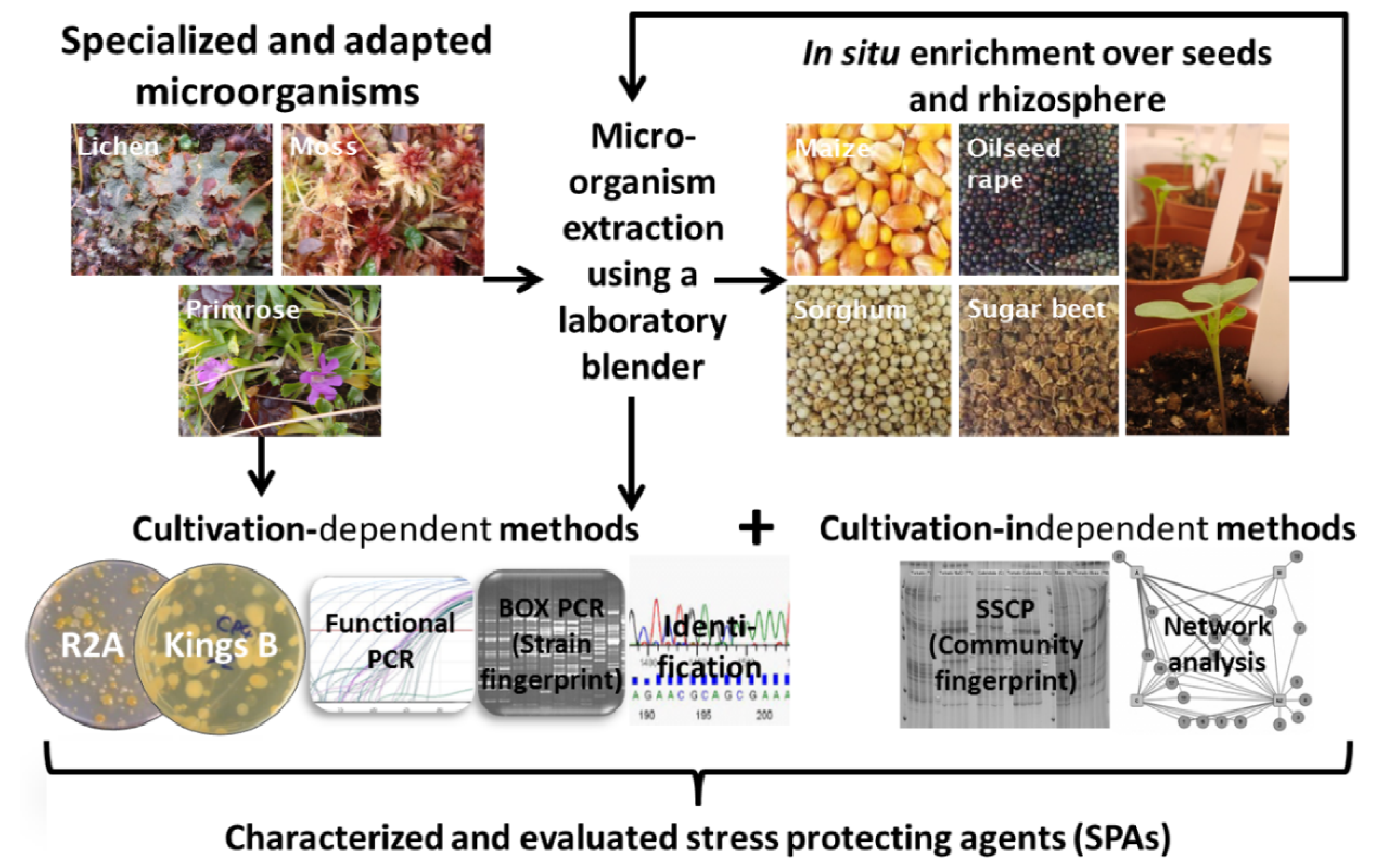
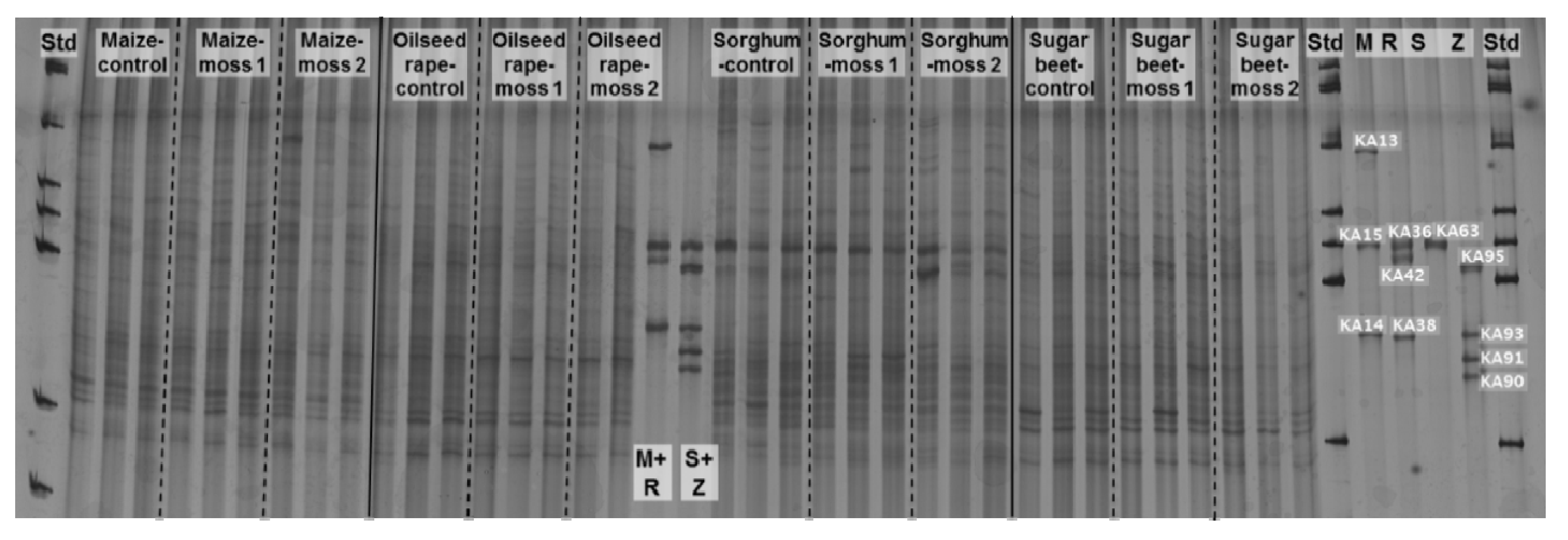
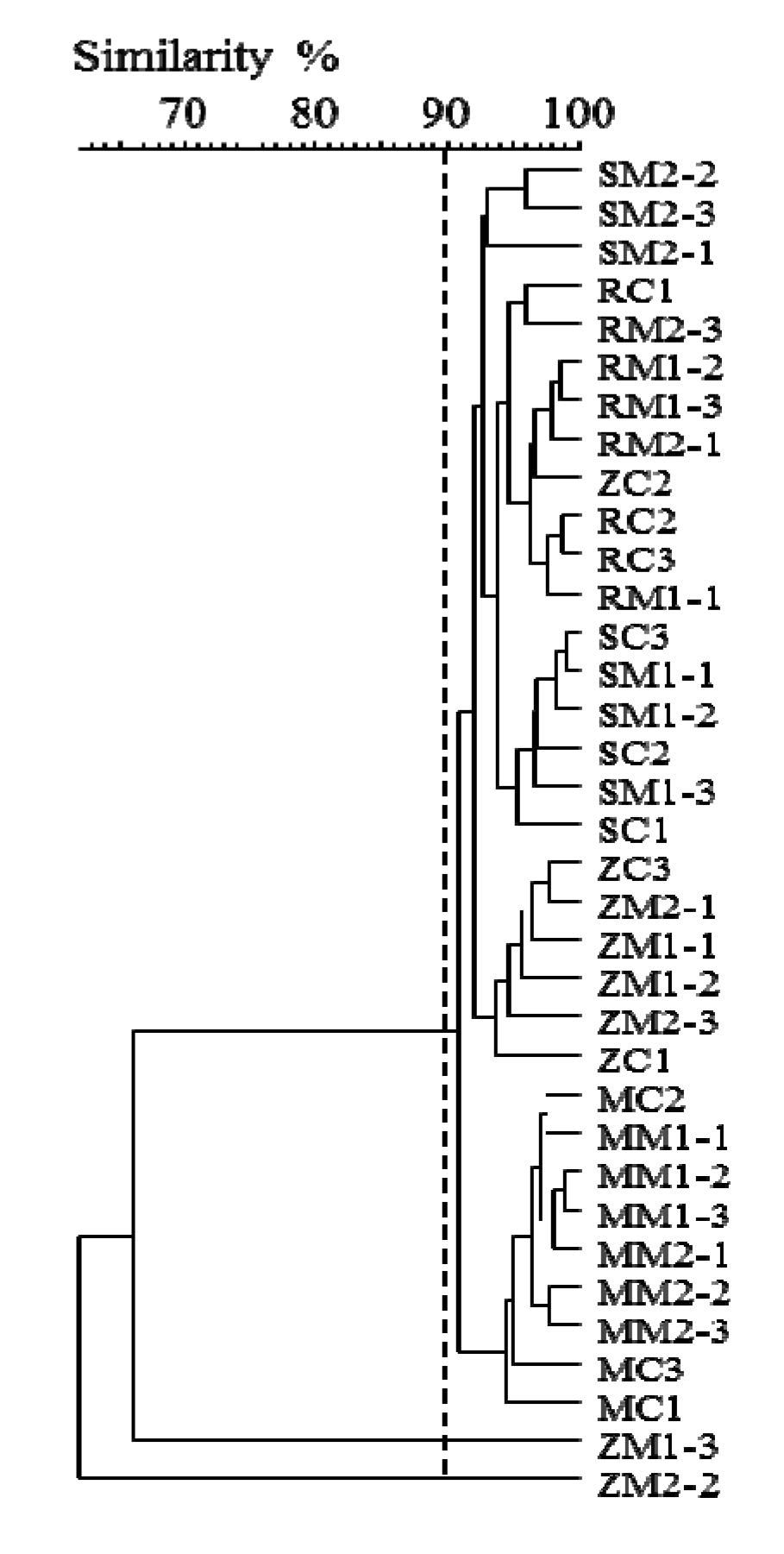
2.3. Characterization of Selected Bacteria and Score Evaluation for SPAs
| Screening Target | Result a | Evaluation Weight | ||
|---|---|---|---|---|
| (1) Antagonism | Ascomycota | Alternaria alternata Nees | yes/no | 3 |
| Botrytis cinerea Pers. | yes/no | 0 | ||
| Sclerotium sclerotiorum Sacc. | yes/no | 3 | ||
| Verticillium dahliae Kleb. V25 | yes/no | 3 | ||
| Basidiomycota | Rhizoctonia solani Kühn AG2-2IIIB | yes/no | 3 | |
| Rhizoctonia solani Kühn AG4 | yes/no | 3 | ||
| Sclerotium rolfsii (Curzi) C.C.Tu and Kimbr. | yes/no | 3 | ||
| Oomycetes | Phytophthora infestans (Mont.) De Bary | yes/no | 3 | |
| (2) Nutrient allocation | Nitrogen fixation | yes/no | 1 | |
| Phosphate solubilizing | yes/no | 3 | ||
| Siderophore production | yes/no | 3 | ||
| (3) Exoenzyme activity | Chitinase | yes/no | 3 | |
| Glucanase | yes/no | 3 | ||
| Protease | yes/no | 1 | ||
| (4) AB gene detection | Pyrrolnitrin (prnD) | yes/no | 0 | |
| Diacetylphloroglucinol (phlD) | yes/no | 0 | ||
| (5) Growth in presence of stress | ROS detection | TMAMQ | Range | 5 |
| Tellurite | Range | 3 | ||
| H2O2 | Range | 3 | ||
| Drought | 24 h Desiccation | yes/no | 5 | |
| Salt | In presence of various NaCl concentrations | Range | 3 | |
| Temperatures | NA, 24 h, 13 °C | yes/no | 4 | |
| NA, 24 h, 37 °C | yes/no | 1 | ||
| R2A, 24 h, 13 °C | yes/no | 4 | ||
| R2A, 24 h, 37 °C | yes/no | 1 | ||
| (6) Plant assay | Germination after 3 days | Maize | Range | 3 |
| Oilseed rape | Range | 1 | ||
| Sorghum | Range | 1 | ||
| Sugar beet | Range | 3 | ||
| Germination after 6 days | Maize | Range | 1 | |
| Oilseed rape | Range | 1 | ||
| Sorghum | Range | 3 | ||
| Sugar beet | Range | 3 | ||
| Indole-3-acetic acid | Range | 3 | ||
| ACC deaminase | Range | 3 | ||
| (7) Surface activity | Rhamnolipid | yes/no | 1 | |
| Toluol | yes/no | 1 | ||
| Paraffin (ONC) | yes/no | 1 | ||
| Engine oil (ONC) | yes/no | 1 | ||
| Drop collapse assay | yes/no | 1 | ||
| Strain information | Antagonism | Nutrient allocation | Exoenzyme activity | AB gene detection | Stress | Plant assay | Surface activity | Score values | |||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Abb. | Species (risk group) | Strain number a | ARDRA/BOX group | ||||||||
| KA001 | Pseudomonas kilonensis (1) | MF1-1-4 | 10/13 | 6 | 2 | 1 | 0 | 5 | 4 | 0 | 43 |
| KA002 | MF1-2-3 | n.d./18 | 2 | 2 | 0 | 0 | 5 | 5 | 1 | 32 | |
| KA003 | Arthrobacter nitroguaiacolicus (1) | MF1-2-4 | 13/7 | 2 | 2 | 2 | 1 | 6 | 2 | 1 | 37 |
| KA004 | MF1-3-1 | 10/13 | 3 | 2 | 1 | 0 | 6 | 3 | 0 | 36 | |
| KA005 | MF2-1-1 | 13/3 | 3 | 0 | 1 | 0 | 6 | 3 | 1 | 35 | |
| KA006 | MF2-1-2 | 10/13 | 5 | 2 | 1 | 0 | 5 | 4 | 0 | 40 | |
| KA007 | MF2-1-3 | 10/13 | 5 | 2 | 1 | 0 | 5 | 3 | 0 | 39 | |
| KA008 | Pseudomonas corrugata (1) | MF2-3-1 | 10/13 | 2 | 2 | 0 | 0 | 5 | 4 | 0 | 30 |
| KA009 | Pseudomonas kilonensis (1) | MM1-2-2 | 10/13 | 6 | 2 | 1 | 0 | 5 | 3 | 0 | 40 |
| KA010 | MM1-2-3 | 10/13 | 6 | 2 | 1 | 0 | 5 | 2 | 0 | 41 | |
| KA011 | MM1-2-4 | 10/13 | 5 | 2 | 1 | 0 | 5 | 4 | 0 | 40 | |
| KA012 | MM1-3-1 | 10/13 | 5 | 2 | 1 | 0 | 5 | 3 | 0 | 39 | |
| KA013 | Pseudomonas kilonensis (1) | MM1-3-2 | 10/16 | 6 | 2 | 1 | 0 | 5 | 3 | 0 | 40 |
| KA014 | Arthrobacter nicotinovorans (1) | MM1-3-4 | 13/3 | 2 | 0 | 1 | 0 | 5 | 2 | 5 | 27 |
| KA015 | Pseudomonas brassicacearum subsp. brassicacearum (1) | MM2-1-2 | 10/23 | 2 | 1 | 1 | 1 | 5 | 2 | 1 | 27 |
| KA016 | MM2-1-4 | 5/1 | 4 | 1 | 1 | 1 | 6 | 1 | 3 | 37 | |
| KA017 | Pseudomonas kilonensis (1) | MM2-2-2 | 9/13 | 4 | 1 | 0 | 0 | 2 | 1 | 3 | 24 |
| KA018 | Pseudomonas otitidis (2) | MM2-3-1 | 4/11 | 5 | 2 | 1 | 0 | 6 | 3 | 3 | 45 |
| KA019 | MP1-1-4 | 4/21 | 4 | 0 | 1 | 1 | 4 | 2 | 0 | 26 | |
| KA020 | MP1-2-1 | 10/30 | 1 | 1 | 1 | 0 | 5 | 2 | 1 | 22 | |
| KA021 | Pseudomonas corrugata (1) | MP1-2-2 | 10/13 | 2 | 2 | 1 | 0 | 4 | 2 | 0 | 28 |
| KA022 | MP1-3-1 | 10/13 | 6 | 2 | 1 | 0 | 6 | 2 | 0 | 44 | |
| KA023 | MP1-3-2 | 10/13 | 6 | 2 | 1 | 0 | 6 | 2 | 0 | 46 | |
| KA024 | MP1-3-3 | 12/23 | 6 | 2 | 1 | 0 | 6 | 1 | 0 | 43 | |
| KA025 | MP1-3-4 | 10/14 | 4 | 1 | 1 | 1 | 5 | 3 | 0 | 33 | |
| KA026 | MP2-1-2 | 10/13 | 3 | 1 | 1 | 0 | 4 | 2 | 1 | 30 | |
| KA027 | Pseudomonas kilonensis (1) | MP2-1-4 | 10/13 | 3 | 1 | 1 | 0 | 6 | 2 | 0 | 34 |
| KA028 | MP2-2-2 | 10/13 | 4 | 2 | 1 | 0 | 7 | 3 | 0 | 44 | |
| KA029 | RF1-1-1 | 10/4 | 5 | 2 | 1 | 0 | 5 | 3 | 0 | 37 | |
| KA030 | Pseudomonas asplenii (1p) | RF1-2-2 | 3/24 | 6 | 1 | 1 | 1 | 3 | 1 | 0 | 33 |
| KA031 | RF1-3-3 | 1/38 | 2 | 0 | 1 | 0 | 1 | 1 | 1 | 14 | |
| KA032 | Pseudomonas asplenii (1p) | RF2-1-5 | 10/4 | 2 | 1 | 2 | 0 | 1 | 2 | 0 | 19 |
| KA033 | RF2-3-3 | 10/13 | 6 | 1 | 0 | 1 | 4 | 1 | 0 | 33 | |
| KA034 | Pseudomonas brenneri (1) | RF2-3-4 | 10/37 | 5 | 1 | 1 | 0 | 5 | 1 | 0 | 32 |
| KA035 | RF2-3-5 | 10/4 | 2 | 2 | 1 | 0 | 4 | 3 | 1 | 30 | |
| KA036 | Pseudomonas brassicacearum subsp. brassicacearum (1) | RM1-1-3 | 10/9 | 4 | 1 | 0 | 1 | 3 | 1 | 0 | 26 |
| KA037 | Pseudomonas kilonensis (1) | RM1-1-4 | 10/13 | 4 | 1 | 0 | 0 | 5 | 3 | 0 | 32 |
| KA038 | Pseudomonas otitidis (1) | RM1-3-1 | 4/11 | 6 | 1 | 1 | 0 | 7 | 3 | 0 | 47 |
| KA039 | RM2-1-2 | 5/1 | 5 | 0 | 0 | 1 | 5 | 3 | 0 | 30 | |
| KA040 | RM2-1-5 | 10/19 | 6 | 0 | 0 | 1 | 5 | 2 | 1 | 33 | |
| KA041 | RM2-2-2 | 5/1 | 1 | 1 | 0 | 0 | 4 | 3 | 1 | 26 | |
| KA042 | Pantoea sp. | RM2-2-4 | 5/40 | 5 | 0 | 0 | 0 | 6 | 3 | 1 | 36 |
| KA043 | RM2-3-1 | 10/9 | 4 | 0 | 0 | 0 | 6 | 2 | 1 | 30 | |
| KA044 | RM2-3-3 | 10/9 | 5 | 1 | 0 | 0 | 5 | 3 | 0 | 35 | |
| KA045 | RP1-1-1 | 5/33 | 5 | 1 | 0 | 0 | 5 | 2 | 0 | 32 | |
| KA046 | RP1-1-3 | 10/14 | 1 | 1 | 0 | 0 | 6 | 3 | 1 | 32 | |
| KA047 | RP1-2-1 | 10/14 | 3 | 2 | 1 | 0 | 5 | 3 | 0 | 32 | |
| KA048 | Pseudomonas rhodesiae (1) | RP1-2-2 | 10/14 | 2 | 3 | 1 | 0 | 5 | 2 | 0 | 31 |
| KA049 | RP1-3-1 | 10/29 | 4 | 3 | 1 | 0 | 5 | 4 | 0 | 38 | |
| KA050 | RP1-3-3 | 5/12 | 4 | 2 | 0 | 0 | 3 | 3 | 1 | 32 | |
| KA051 | Pseudomonas asplenii (1p) | RP1-3-4 | 10/4 | 1 | 2 | 1 | 0 | 5 | 2 | 1 | 28 |
| KA052 | RP2-1-3 | 10/14 | 6 | 0 | 1 | 1 | 3 | 2 | 0 | 33 | |
| KA053 | RP2-2-2 | 14/39 | 3 | 2 | 1 | 0 | 5 | 3 | 0 | 31 | |
| KA054 | RP2-2-4 | 10/14 | 1 | 0 | 2 | 0 | 4 | 3 | 0 | 24 | |
| KA055 | SF1-1-1 | 10/14 | 4 | 3 | 1 | 0 | 5 | 3 | 0 | 37 | |
| KA056 | SF1-1-4 | 5/8 | 2 | 2 | 1 | 0 | 5 | 4 | 0 | 32 | |
| KA057 | Serratia proteamaculans (2) | SF1-3-1 | 11/17 | 1 | 2 | 0 | 0 | 5 | 3 | 1 | 28 |
| KA058 | Yersinia kristensenii (2) | SF2-1-2 | 9/35 | 2 | 2 | 1 | 0 | 6 | 4 | 1 | 40 |
| KA059 | SF2-2-1 | 9/35 | 3 | 2 | 1 | 0 | 5 | 2 | 1 | 34 | |
| KA060 | Pseudomonas poae (1) | SF2-2-2 | 10/36 | 2 | 1 | 1 | 0 | 5 | 3 | 0 | 30 |
| KA061 | SF2-2-4 | 10/37 | 3 | 2 | 1 | 0 | 6 | 2 | 0 | 38 | |
| KA062 | SF2-3-1 | 10/32 | 1 | 1 | 1 | 0 | 5 | 3 | 0 | 25 | |
| KA063 | Pseudomonas veronii (1) | SM1-1-2 | 10/30 | 2 | 1 | 0 | 0 | 5 | 3 | 0 | 24 |
| KA064 | Pseudomonas koreensis (1) | SM1-1-4 | 10/20 | 3 | 2 | 1 | 0 | 2 | 3 | 1 | 26 |
| KA065 | Pseudomonas kilonensis (1) | SM1-2-4 | 10/13 | 5 | 1 | 0 | 1 | 5 | 3 | 1 | 37 |
| KA066 | Pseudomonas brenneri (1) | SM1-3-1 | 10/34 | 4 | 2 | 1 | 0 | 5 | 2 | 0 | 38 |
| KA067 | Pseudomonas otitidis (2) | SM1-3-2 | 10/34 | 3 | 2 | 1 | 0 | 5 | 3 | 1 | 35 |
| KA068 | SM1-3-4 | 10/34 | 6 | 2 | 1 | 0 | 5 | 4 | 1 | 44 | |
| KA069 | Enterobacter cowanii (2) | SM2-1-2 | 4/31 | 3 | 2 | 1 | 0 | 5 | 1 | 0 | 29 |
| KA070 | Pseudomonas veronii (1) | SM2-2-4 | 4/30 | 3 | 0 | 0 | 0 | 5 | 4 | 1 | 33 |
| KA071 | SM2-3-1 | 7/31 | 4 | 1 | 1 | 0 | 5 | 2 | 1 | 31 | |
| KA072 | SM2-3-2 | 12/42 | 4 | 0 | 1 | 0 | 5 | 3 | 0 | 28 | |
| KA073 | SP1-1-1 | 2/41 | 4 | 2 | 1 | 0 | 6 | 2 | 0 | 36 | |
| KA074 | Pseudomonas tremae (1) | SP1-1-3 | 18/27 | 3 | 2 | 0 | 0 | 3 | 3 | 1 | 32 |
| KA075 | SP1-1-4 | 6/28 | 2 | 2 | 1 | 1 | 4 | 6 | 0 | 37 | |
| KA076 | Serratia proteamaculans (2) | SP1-3-1 | 8/22 | 3 | 0 | 1 | 0 | 5 | 2 | 1 | 27 |
| KA077 | SP2-1-2 | 16/14 | 4 | 3 | 1 | 0 | 5 | 6 | 0 | 47 | |
| KA078 | Pseudomonas putida (1) | SP2-2-2 | 15/13 | 3 | 1 | 1 | 0 | 5 | 5 | 1 | 33 |
| KA079 | SP2-2-3 | 16/13 | 3 | 2 | 1 | 0 | 5 | 7 | 0 | 42 | |
| KA080 | Pseudomonas rhodesiae (1) | SP2-3-3 | 16/14 | 5 | 3 | 1 | 0 | 4 | 4 | 1 | 41 |
| KA081 | Pseudomonas rhodesiae (1) | SP2-3-4 | 16/14 | 3 | 3 | 1 | 0 | 2 | 4 | 1 | 29 |
| KA082 | Pseudomonas poae (1) | ZF1-2-4 | 16/26 | 0 | 2 | 1 | 0 | 4 | 2 | 1 | 26 |
| KA083 | ZF2-1-1 | 15/15 | 1 | 0 | 0 | 0 | 5 | 2 | 0 | 20 | |
| KA084 | Pseudomonas corrugata (1) | ZF2-1-2 | 16/13 | 5 | 2 | 1 | 0 | 5 | 1 | 1 | 38 |
| KA085 | ZF2-1-4 | 16/2 | 2 | 3 | 0 | 0 | 5 | 1 | 1 | 28 | |
| KA086 | ZF2-2-1 | 16/30 | 1 | 2 | 1 | 0 | 3 | 2 | 1 | 26 | |
| KA087 | ZF2-2-3 | 15/24 | 1 | 1 | 0 | 0 | 0 | 3 | 1 | 10 | |
| KA088 | ZF2-3-2 | 16/5 | 1 | 0 | 2 | 0 | 5 | 4 | 1 | 29 | |
| KA089 | ZF2-3-4 | 16/32 | 1 | 1 | 1 | 0 | 5 | 2 | 1 | 27 | |
| KA090 | Microbacterium xylanilyticum (1) | ZM1-1-3 | 15/25 | 1 | 0 | 1 | 0 | 4 | 4 | 1 | 27 |
| KA091 | Chryseobacterium wanjuense (1) | ZM2-1-1 | 17/18 | 2 | 1 | 1 | 0 | 7 | 1 | 0 | 32 |
| KA092 | Pseudomonas kilonensis (1) | ZM2-1-2 | 16/13 | 5 | 1 | 2 | 0 | 5 | 3 | 0 | 37 |
| KA093 | Pseudomonas otitidis (1) | ZM2-2-3 | 16/11 | 2 | 1 | 1 | 0 | 4 | 4 | 0 | 29 |
| KA094 | Pseudomonas corrugata (1) | ZM2-3-1 | 16/13 | 7 | 3 | 1 | 0 | 4 | 4 | 1 | 45 |
| KA095 | Rhodanobacter fulvus (1) | ZM2-3-2 | 17/43 | 2 | 1 | 0 | 0 | 3 | 4 | 1 | 26 |
| KA096 | Pseudomonas kilonensis (1) | ZM2-3-4 | 16/13 | 5 | 2 | 1 | 0 | 5 | 2 | 0 | 34 |
| KA097 | ZP1-1-3 | 16/26 | 2 | 1 | 1 | 1 | 5 | 3 | 1 | 29 | |
| KA098 | ZP1-1-4 | 16/6 | 1 | 2 | 0 | 0 | 5 | 4 | 1 | 29 | |
| KA099 | Pseudomonas kilonensis (1) | ZP1-2-2 | 16/13 | 5 | 2 | 1 | 0 | 4 | 1 | 1 | 33 |
| KA100 | ZP1-2-4 | 16/18 | 2 | 1 | 0 | 0 | 6 | 3 | 1 | 28 | |
| KA101 | Pseudomonas kilonensis (1) | ZP1-3-1 | 16/13 | 5 | 3 | 1 | 0 | 4 | 3 | 0 | 37 |
| KA102 | Pseudomonas kilonensis (1) | ZP1-3-2 | 16/13 | 6 | 3 | 1 | 0 | 4 | 4 | 0 | 39 |
| KA103 | Pseudomonas kilonensis (1) | ZP1-3-3 | 16/13 | 6 | 3 | 1 | 0 | 4 | 1 | 0 | 34 |
| KA104 | ZP1-3-4 | 15/10 | 3 | 2 | 0 | 1 | 6 | 3 | 0 | 35 | |
| KA105 | Bacillus anthracis (3) | ZP2-1-3 | n.d./44 | 2 | 1 | 0 | 0 | 7 | 2 | 1 | 30 |

3. Experimental Section
3.1. Experimental Design for Isolation and Characterization of Stress Protecting Bacteria
3.2. Molecular Analysis
3.3. Stress Confrontation Assays
3.4. Plant Pathogen Confrontation Assays
3.5. Exoenzyme Activity, Biosurfactants and Nutrient Allocation
3.6. Bioassays
3.7. Statistics
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Shaw, M.W. Preparing for changes in plant disease due to climate change. Plant Prot. Sci. 2009, 45, S3–S10. [Google Scholar]
- Newton, A.C.; Johnson, S.N.; Gregory, P.J. Implications of climate change for diseases, crop yields and food security. Euphytica 2011, 179, 3–18. [Google Scholar] [CrossRef]
- Maheswari, M.; Yadav, S.K.; Shanker, A.K.; Kumar, M.A.; Venkateswarlu, B. Overview of Plant Stresses: Mechanisms, Adaptations and Research Pursuit. In Crop Stress and its Management: Perspectives and Strategies; Venkateswarlu, B., Shanker, A.K., Shanker, C., Maheswari, M., Eds.; Springer: New York, NY, USA, 2012; pp. 1–18. [Google Scholar]
- Berendsen, R.L.; Pieterse, C.M.; Bakker, P.A. The rhizosphere microbiome and plant health. Trends Plant Sci. 2012, 17, 478–486. [Google Scholar] [CrossRef]
- Alavi, P.; Starcher, M.R.; Zachow, C.; Müller, H.; Berg, G. Root-microbe systems: the effect and mode of interaction of Stress Protecting Agent (SPA) Stenotrophomonas rhizophila DSM14405T. Front. Plant Sci. 2013, 4, 141. [Google Scholar] [CrossRef]
- Berg, G.; Smalla, K. Plant species and soil type cooperatively shape the structure and function of microbial communities in the rhizosphere. FEMS Microbiol. Ecol. 2009, 68, 1–13. [Google Scholar] [CrossRef]
- Köberl, M.; Müller, H.; Ramadan, E.M.; Berg, G. Desert farming benefits from microbial potential in arid soils and promotes diversity and plant health. PLoS One 2011, 6, e24452. [Google Scholar]
- Köberl, M.; Ramadan, E.M.; Roßmann, B.; Staver, C.; Fürnkranz, M.; Lukesch, B.; Grube, M.; Berg, G. Using Ecological Knowledge and Molecular Tools to Develop Effective and Safe Biocontrol Strategies. In Pesticides in the Modern World—Pests Control and Pesticides Exposure and Toxicity Assessment; Stoytcheva, M., Ed.; InTech e-book: Rijeka, Croatia, 2011; pp. 3–26. [Google Scholar]
- Yang, J.; Kloepper, J.W.; Ryu, C.M. Rhizosphere bacteria help plants tolerate abiotic stress. Trends Plant Sci. 2009, 14, 1–4. [Google Scholar] [CrossRef]
- Opelt, K.; Chobot, V.; Hadacek, F.; Schönmann, S.; Eberl, L.; Berg, G. Investigations of the structure and function of bacterial communities associated with Sphagnum mosses. Environ. Microbiol. 2007, 91, 2795–2809. [Google Scholar]
- Opelt, K.; Berg, C.; Berg, G. The bryophyte genus Sphagnum is a reservoir for powerful and extraordinary antagonists and potentially facultative human pathogens. FEMS Microbiol. Ecol. 2007, 61, 38–53. [Google Scholar] [CrossRef]
- Grube, M.; Cardinale, M.; de Castro, J.V., Jr.; Müller, H.; Berg, G. Species-specific structural and functional diversity of bacterial communities in lichen symbioses. ISME J. 2009, 3, 1105–1115. [Google Scholar] [CrossRef]
- Berg, G.; Hartenberger, K.; Liebminger, S.; Zachow, C. Antagonistic Endophytes from Mistletoes as Bio-resource to Control Plant as well as Clean Room Pathogens. In IOBC/WPRS Bulletin, Working Group “Biological Control of Fungal and Bacterial Plant Pathogens”; Pertot, I., Elad, Y., Gessler, C., Cini, A., Eds.; International Organisation for Biological Control (IOBC): Zurich, Switzerland, 2012; Volume 78, pp. 29–32. [Google Scholar]
- Zachow, C.; Grosch, R.; Berg, G. Impact of biotic and a-biotic parameters on structure and function of microbial communities living on sclerotia of the soil-borne pathogenic fungus Rhizoctonia solani. Appl. Soil Ecol. 2011, 48, 193–200. [Google Scholar]
- Amann, R.I.; Ludwig, W.; Schleifer, K.H. Phylogenetic identification and in situ detection of individual microbial cells without cultivation. Microbiol. Rev. 1995, 59, 143–169. [Google Scholar]
- Schwieger, F.; Tebbe, C.C. A new approach to utilize PCR-single-strand conformation polymorphism for 16S rRNA gene-based microbial community analysis. Appl. Environ. Microbiol. 1998, 64, 4870–4876. [Google Scholar]
- Smalla, K.; Oros-Sichler, M.; Milling, A.; Heuer, H.; Baumgarte, S.; Becker, R.; Neuber, G.; Kropf, S.; Ulrich, A.; Tebbe, C.C. Bacterial diversity of soils assessed by DGGE, T-RFLP and SSCP fingerprints of PCR-amplified 16S rRNA gene fragments: Do the different methods provide similar results? J. Microbiol. Methods 2007, 69, 470–479. [Google Scholar] [CrossRef]
- Bragina, A.; Oberauner-Wappis, L.; Halwachs, B.; Thallinger, G.G.; Müller, H.; Berg, G. The Sphagnum microbiome support greatly ecosystem functioning in bog ecosystems under extreme conditions. 2013. submitted. [Google Scholar]
- Grosch, R.; Dealtry, S.; Schreiter, S.; Berg, G.; Mendonça-Hagler, L.; Smalla, K. Biocontrol of Rhizoctonia solani: Complex interaction of biocontrol strains, pathogen and indigenous microbial community in the rhizosphere of lettuce shown by molecular methods. Plant Soil 2012, 361, 343–357. [Google Scholar] [CrossRef]
- Hartmann, A.; Schmid, M.; van Tuinen, D.; Berg, G. Plant-driven selection of microbes. Plant Soil 2009, 321, 235–257. [Google Scholar] [CrossRef]
- Bulgarelli, D.; Rott, M.; Schlaeppi, K.; Ver Loren van Themaat, E.; Ahmadinejad, N.; Assenza, F.; Rauf, P.; Huettel, B.; Reinhardt, R.; Schmelzer, E.; et al. Revealing structure and assembly cues for Arabidopsis root-inhabiting bacterial microbiota. Nature 2012, 488, 91–95. [Google Scholar] [CrossRef]
- Lundberg, D.S.; Lebeis, S.L.; Paredes, S.H.; Yourstone, S.; Gehring, J.; Malfatti, S.; Tremblay, J.; Engelbrektson, A.; Kunin, V.; del Rio, T.G.; et al. Defining the core Arabidopsis thaliana root microbiome. Nature 2012, A488(7409), 86–90. [Google Scholar]
- Hartmann, A.; Rothballer, M.; Schmid, M. Lorenz Hiltner, a pioneer in rhizosphere microbial ecology and soil bacteriology research. Plant Soil 2008, 312, 7–14. [Google Scholar] [CrossRef]
- Ehlers, R.U. Regulation of Biological Control Agents; Springer: Dordrecht, The Netherlands, 2011. [Google Scholar]
- Zachow, C.; Pirker, H.; Westendorf, C.; Tilcher, R.; Berg, G. The Caenorhabditis elegans assay: a tool to evaluate the pathogenic potential of bacterial biocontrol agents. Eur. J. Plant Pathol. 2009, 125, 367–376. [Google Scholar]
- Loper, J.E.; Hassan, K.A.; Mavrodi, D.V.; Davis, E.W., II; Lim, C.K.; Shaffer, B.T.; Elbourne, L.D.H.; Stockwell, V.O.; Hartney, S.L.; Breakwell, K.; et al. Comparative genomics of plant-associated Pseudomonas spp.: Insights into diversity and inheritance of traits involved in multitrophic interactions. PLoS Genet. 2012, 8, e1002784. [Google Scholar] [CrossRef]
- Zachow, C.; Tilcher, R.; Berg, G. Sugar beet-associated bacterial and fungal communities show a high indigenous antagonistic potential against plant pathogens. Microbial Ecol. 2008, 55, 119–129. [Google Scholar] [CrossRef]
- Yeager, C.M.; Kornosky, L.; Housman, C.; Grote, E.E.; Belnap, J.; Kuske, C.R. Diazotrophic community structure and function in two successional stages of biological soil crusts from the Colorado Plateau and Chihuahuan Desert. Appl. Environ. Microbiol. 2004, 70, 973–983. [Google Scholar] [CrossRef]
- Raaijmakers, J.M.; Weller, D.M.; Thomashow, L.S. Frequency of antibiotic-producing Pseudomonas spp. in natural environments. Appl. Environ. Microbiol. 1997, 63, 881–887. [Google Scholar]
- De Souza, J.T.; Raaijmakers, J.M. Polymorphisms within the prnD and pltC genes from pyrrolnitrin and pyoluteorin-producing Pseudomonas and Burkholderia spp. FEMS Microbiol. Ecol. 2003, 43, 21–34. [Google Scholar]
- Martin, B.; Humbert, O.; Camara, M.; Guenzi, E.; Walker, J.; Mitchell, T.; Andrew, P.; Prudhomme, M.; Alloing, G.; Hakenbeck, R.; Morrison, D.A.; Boulnois, G.J.; Claverys, J.P. A highly conserved repeated DNA element located in the chromosome of Streptococcus pneumonia. Nucleic Acids Res. 1992, 20, 3479–3483. [Google Scholar] [CrossRef]
- Berg, G.; Roskot, N.; Steidle, A.; Eberl, L.; Zock, A.; Smalla, K. Plant-dependent genotypic and phenotypic diversity of antagonistic rhizobacteria isolated from different Verticillium host plants. Appl. Environ. Microbiol. 2002, 68, 3328–3338. [Google Scholar] [CrossRef]
- Tamura, K.; Peterson, D.; Peterson, N.; Stecher, G.; Nei, M.; Kumar, S. MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 2011, 28, 2731–2739. [Google Scholar] [CrossRef]
- Prasetyo, E.N.; Kudanga, T.; Steiner, W.; Murkovic, M.; Wonisch, W.; Nyanhongo, G.S.; Gübitz, G.M. Cellular and plasma antioxidant activity assay using tetramethoxy azobismethylene quinone. Free Radic. Biol. Med. 2010, 49, 1205–1211. [Google Scholar] [CrossRef]
- De Bruijn, I.; de Kock, M.J.D.; Yang, M.; de Waard, P.; van Beek, T.A.; Raaijmakers, J.M. Genome-based discovery, structure prediction and functional analysis of cyclic lipopeptide antibiotics in Pseudomonas species. Mol. Microbiol. 2007, 63, 417–428. [Google Scholar] [CrossRef]
- Jain, D.K.; Lee, H.; Trevors, J.T. Effect of addition of Pseudomonas aeruginosa UG2 inocula or biosurfactants on biodegradation of selected hydrocarbons in soil. J. Ind. Microbiol. 1992, 10, 87–93. [Google Scholar] [CrossRef]
- Fürnkranz, M.; Müller, H.; Berg, G. Characterization of plant growth promoting bacteria from crops in Bolivia. J. Plant Dis. Prot. 2009, 116, 149–155. [Google Scholar]
- Sawar, M.; Kremer, R.J. Determination of bacterially derived auxins using a microplate method. Lett. Appl. Microbiol. 1995, 20, 282–285. [Google Scholar] [CrossRef]
- Kropf, S.; Heuer, H.; Grüning, M.; Smalla, K. Significance test for comparing complex microbial community fingerprints using pairwise similarity measures. J. Microbiol. Methods 2004, 57, 187–195. [Google Scholar] [CrossRef]
Supplementary
| Sampling material (Abbreviation) | Original isolation − weight of material (g) + volume of sodium chloride (mL) | Sampling coordinates | Sampling altitude | Sampling date | |
|---|---|---|---|---|---|
| Moss Sphagnum magellanicum (M) | 1. | 20.0 + 200 | 47.091017–14.564606 | 1850 m | October 10, 2010 |
| 2. | 20.0 + 200 | ||||
| 3. | 20.0 + 200 | ||||
| 4. | 20.0 + 200 | ||||
| Lichen Solorina crocea (F) | 1. | 10.0 + 200 | 47.083157–14.562011 | 1970 m | October 10, 2010 |
| 2. | 6.0 + 200 | ||||
| 3. | 8.3 + 200 | ||||
| 4. | 10.0 + 200 | ||||
| Alpine Primula vulgaris (P) | 1. | 100.0 + 200 | 47.083157–14.562011 | 1970 m | October 10, 2010 |
| 2. | 3.5 + 18 | ||||
| 3. | 3.5 + 18 | ||||
| Sampling material (Abbreviation) | Treatment | Bait plant isolation 10.0 mL sodium chloride | |||||
|---|---|---|---|---|---|---|---|
| Repetition 1 | Repetition 2 | Repetition 3 | |||||
| # plants | m in g | # plants | m in g | # plants | m in g | ||
| Maize (M) | C | 4 | 8.27 | 4 | 17.78 | 3 | 15.71 |
| F1 | 4 | 15.95 | 3 | 11.21 | 4 | 15.53 | |
| F2 | 4 | 16.41 | 4 | 11.33 | 3 | 11.87 | |
| M1 | 4 | 19.09 | 4 | 23.94 | 3 | 14.62 | |
| M2 | 4 | 14.19 | 3 | 13.76 | 3 | 14.85 | |
| P1 | 4 | 19.58 | 4 | 13.96 | 4 | 17.90 | |
| P2 | 3 | 11.56 | 4 | 18.45 | 3 | 12.58 | |
| Oilseed rape (R) | C | 5 | 1.56 | 5 | 0.92 | 3 | 0.56 |
| F1 | 5 | 1.27 | 5 | 1.27 | 5 | 1.84 | |
| F2 | 5 | 2.29 | 5 | 1.70 | 5 | 2.09 | |
| M1 | 5 | 1.55 | 5 | 1.66 | 5 | 1.04 | |
| M2 | 5 | 1.63 | 5 | 2.01 | 5 | 2.16 | |
| P1 | 5 | 0.64 | 5 | 1.78 | 5 | 1.81 | |
| P2 | 5 | 0.84 | 5 | 1.27 | 5 | 1.05 | |
| Sorghum (S) | C | 2 | 0.57 | 5 | 1.75 | 3 | 1.08 |
| F1 | 4 | 1.59 | 3 | 0.86 | 3 | 1.12 | |
| F2 | 2 | 1.63 | 3 | 1.77 | 2 | 0.89 | |
| M1 | 3 | 1.60 | 4 | 2.05 | 3 | 2.50 | |
| M2 | 3 | 1.53 | 4 | 1.29 | 4 | 1.74 | |
| P1 | 2 | 2.11 | 3 | 0.82 | 4 | 1.68 | |
| P2 | 4 | 1.68 | 5 | 1.75 | 4 | 1.67 | |
| Sugar beet (Z) | C | 5 | 0.91 | 5 | 0.82 | 5 | 0.71 |
| F1 | 5 | 1.74 | 4 | 0.93 | 5 | 0.53 | |
| F2 | 5 | 0.66 | 3 | 0.41 | 5 | 1.15 | |
| M1 | 5 | 0.84 | 4 | 1.08 | 5 | 1.95 | |
| M2 | 4 | 0.46 | 4 | 0.80 | 5 | 1.08 | |
| P1 | 2 | 0.36 | 4 | 0.62 | 3 | 0.29 | |
| P2 | 5 | 0.60 | 4 | 0.40 | 5 | 1.02 | |

© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zachow, C.; Müller, H.; Tilcher, R.; Donat, C.; Berg, G. Catch the Best: Novel Screening Strategy to Select Stress Protecting Agents for Crop Plants. Agronomy 2013, 3, 794-815. https://doi.org/10.3390/agronomy3040794
Zachow C, Müller H, Tilcher R, Donat C, Berg G. Catch the Best: Novel Screening Strategy to Select Stress Protecting Agents for Crop Plants. Agronomy. 2013; 3(4):794-815. https://doi.org/10.3390/agronomy3040794
Chicago/Turabian StyleZachow, Christin, Henry Müller, Ralf Tilcher, Christina Donat, and Gabriele Berg. 2013. "Catch the Best: Novel Screening Strategy to Select Stress Protecting Agents for Crop Plants" Agronomy 3, no. 4: 794-815. https://doi.org/10.3390/agronomy3040794
APA StyleZachow, C., Müller, H., Tilcher, R., Donat, C., & Berg, G. (2013). Catch the Best: Novel Screening Strategy to Select Stress Protecting Agents for Crop Plants. Agronomy, 3(4), 794-815. https://doi.org/10.3390/agronomy3040794




