Interactive Effects of Exogenous Hormones, Sucrose, and Environmental Factors on the Growth of Phyllostachys edulis Shoots
Abstract
1. Introduction
2. Materials and Methods
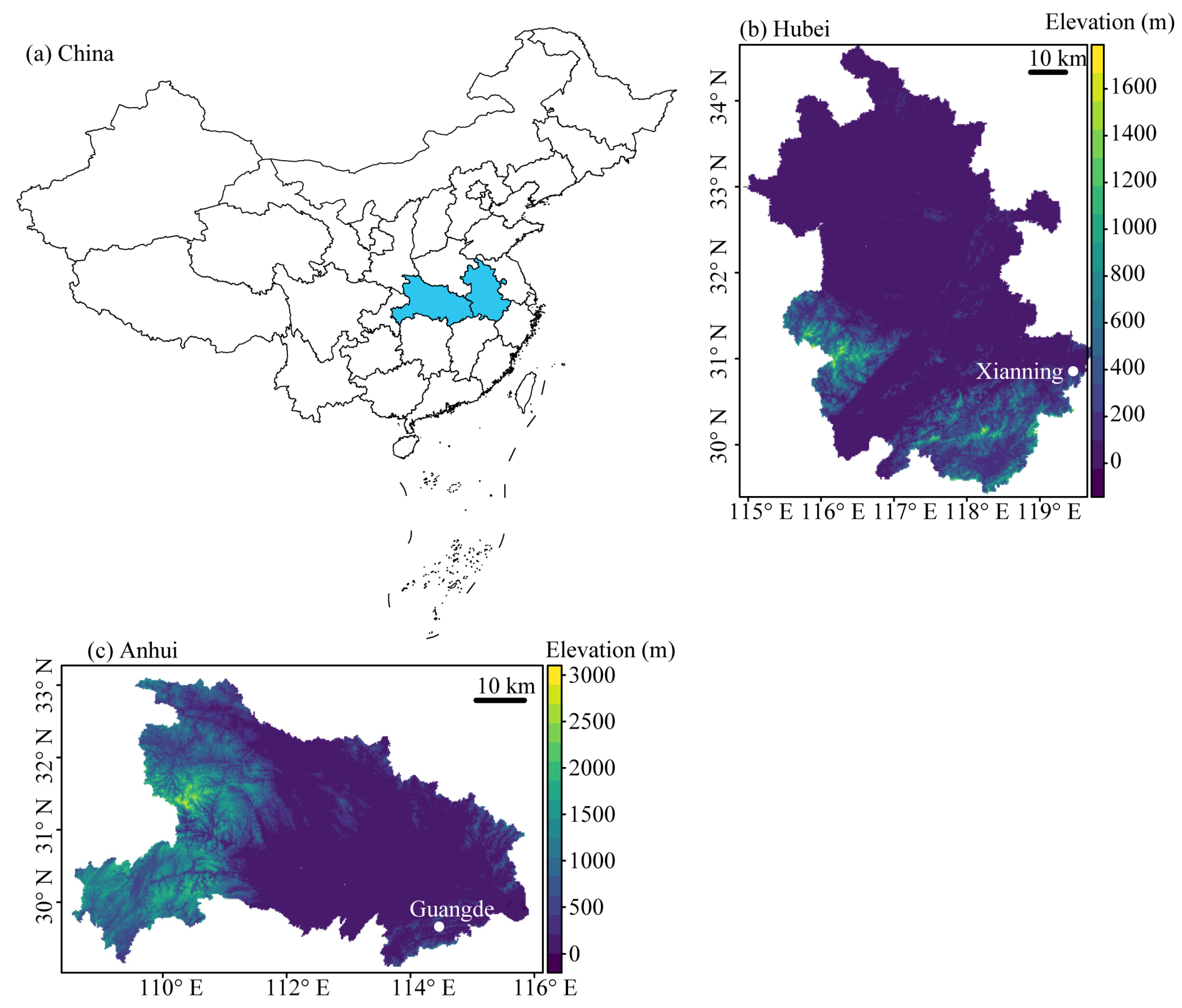
2.1. Study Area
2.2. Field Experiments
2.3. Data Analysis
3. Results
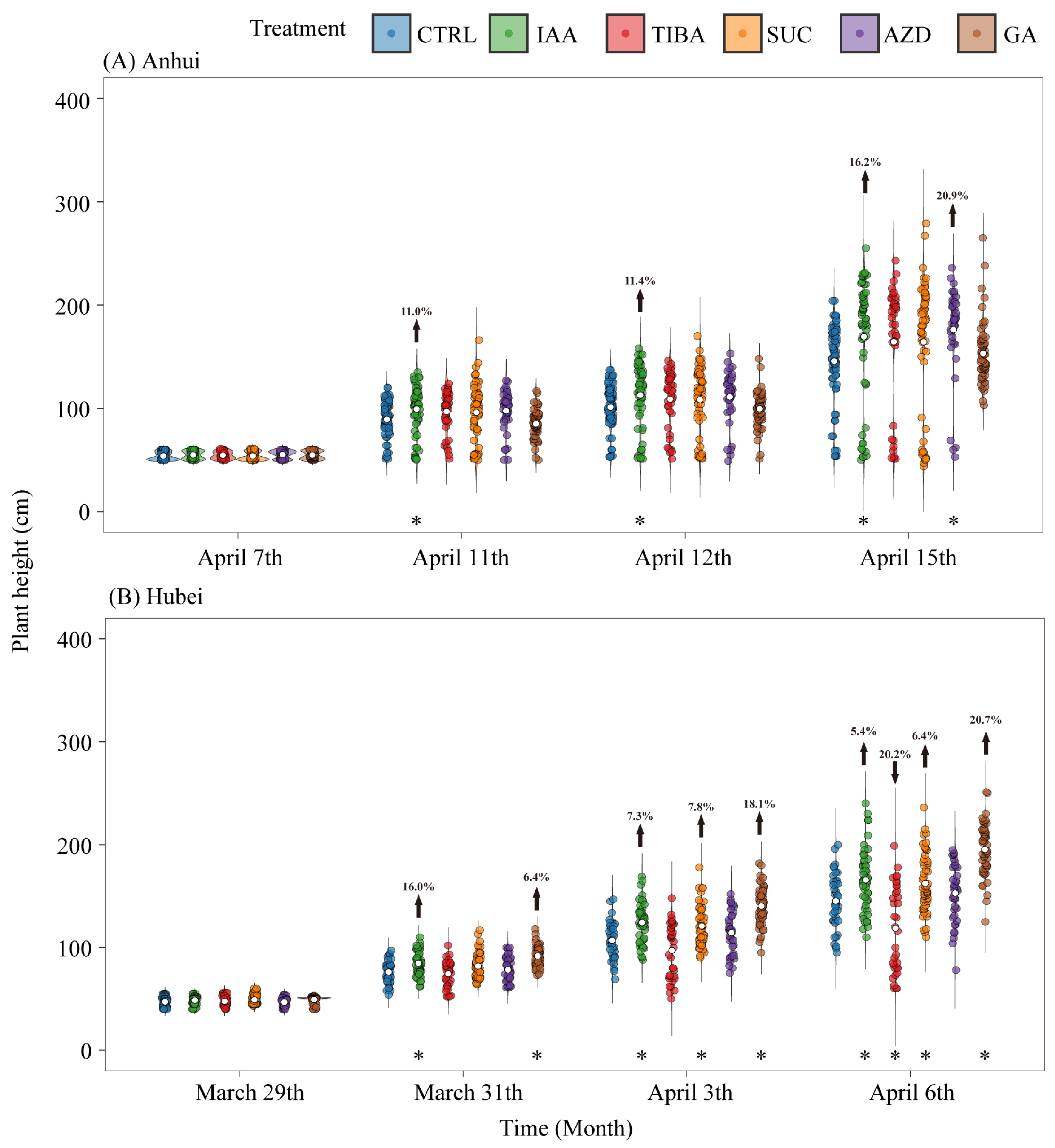
3.1. Effects of Different Exogenous Treatments on Bamboo Shoot Height
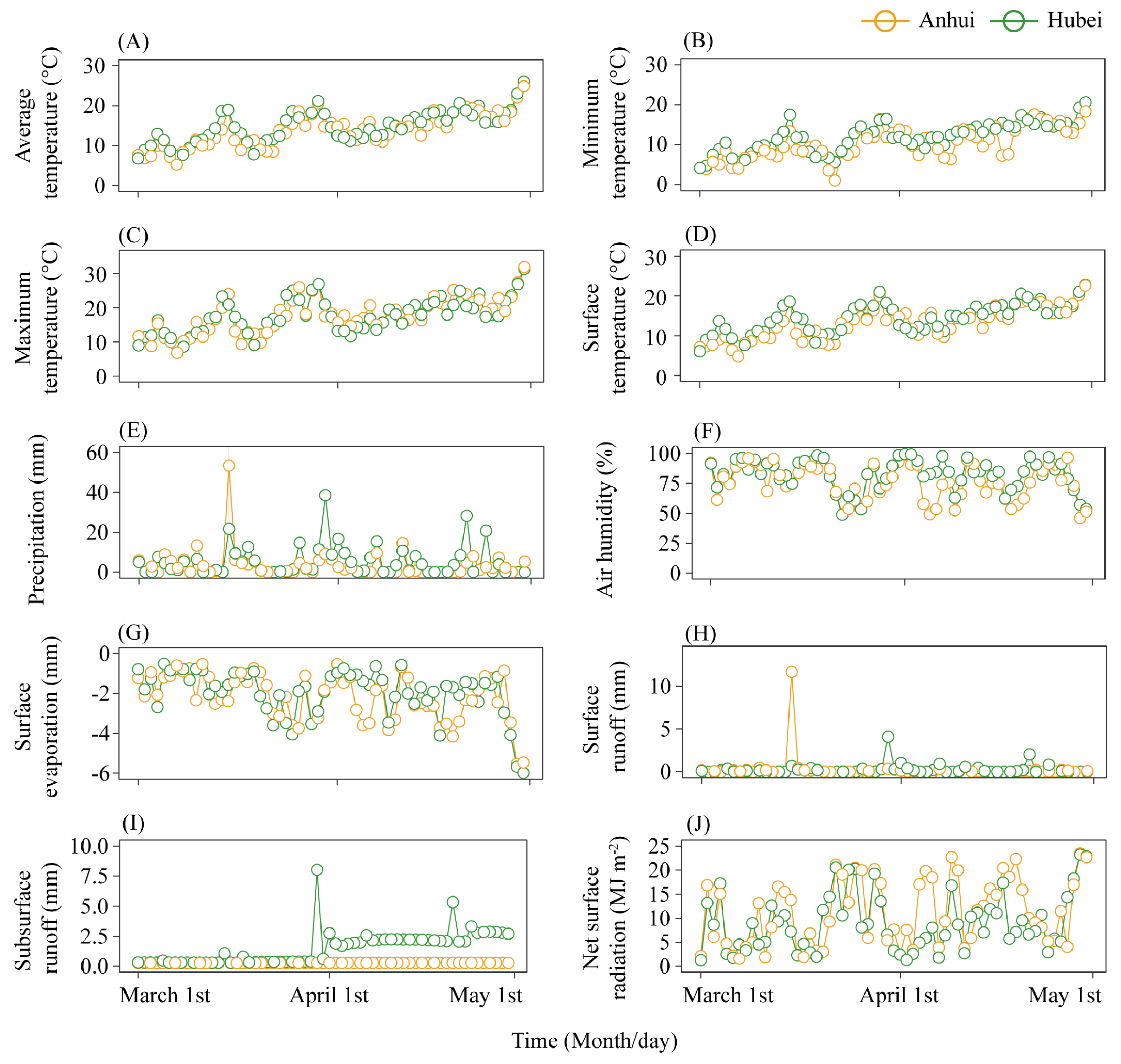
3.2. Environmental Factor Variation Trends During Bamboo Shoot Rapid Growth Periods
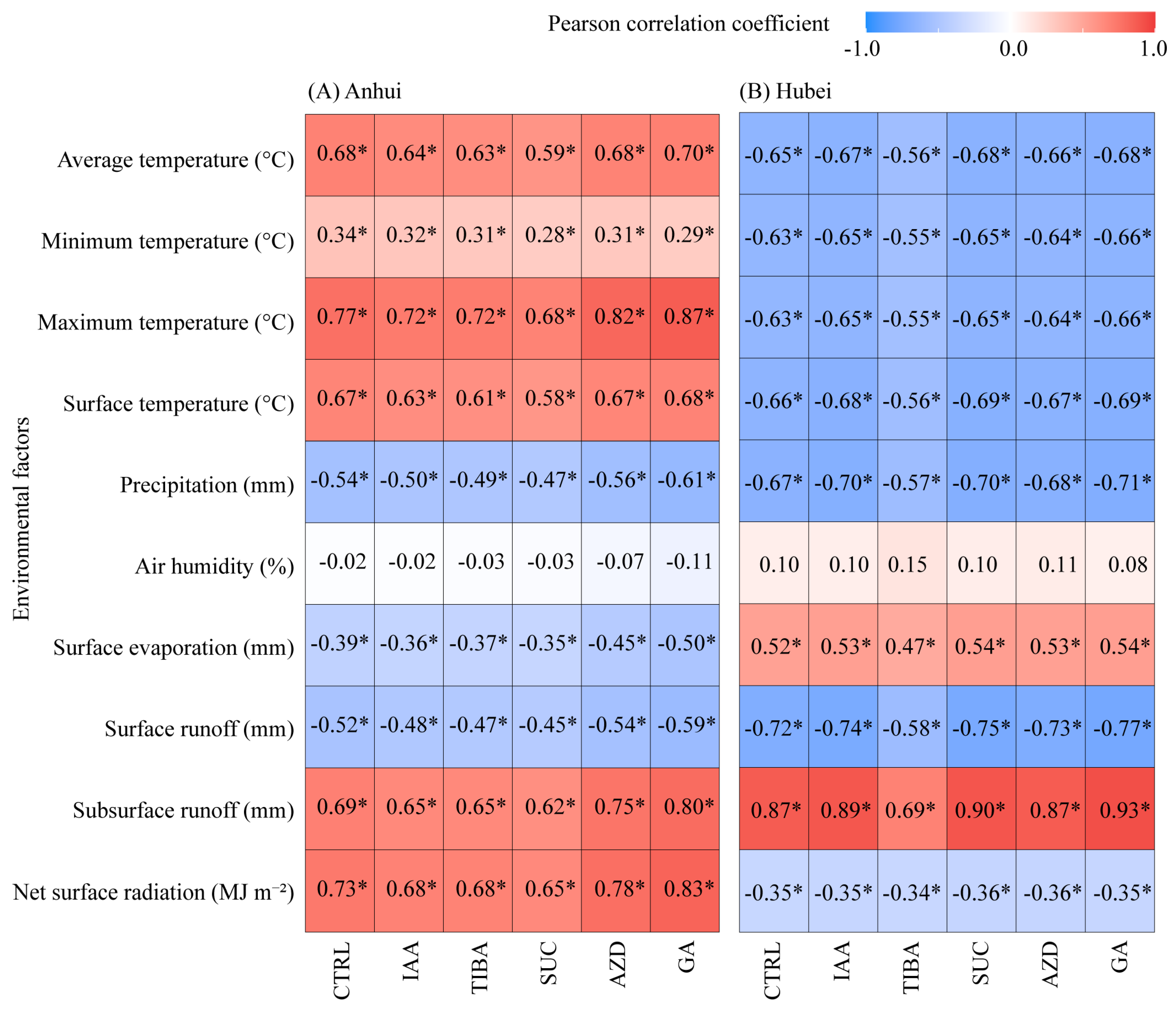
3.3. Correlations Between Bamboo Shoot Height and Environmental Factors Under Different Treatments and Variable Selection
3.4. Interactive Effects of Treatment Methods and Environmental Factors on Bamboo Shoot Height
4. Discussion
4.1. Environmental Factors Governing Bamboo Shoot Growth
4.2. Exogenous Growth Regulators and Their Effects on Shoot Development
4.3. Interactive Mechanisms and Implications for Bamboo Forest Management
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CTRL | Control group |
| IAA | Indole-3-acetic acid |
| GA | Gibberellic acid |
| SUC | Sucrose |
| TIBA | 2,3,5-Triiodobenzoic acid |
| AZD | mTOR kinase inhibitor |
| ANOVA | Analysis of Variance |
| VIF | Variance Inflation Factor |
| MCMC | Markov Chain Monte Carlo |
| GLMM | Generalized Linear Mixed Models |
References
- China’s National Forestry and Grassland Administration. Report of Forest Resources in China (2014–2018); China Forestry Publishing House: Beijing, China, 2019. (In Chinese) [Google Scholar]
- INBAR. China Bamboo and Rattan Commodity International Trade Report; International Bamboo and Rattan Organisation: Beijing, China, 2023. (In Chinese) [Google Scholar]
- Chen, X.W. Variations in patterns of internode and branch lengths for several bamboo species. Plant Biosyst. 2020, 155, 1088–1099. [Google Scholar] [CrossRef]
- Muktar, M.S.; Habte, E.; Teshome, A.; Assefa, Y.; Negawo, A.T.; Lee, K.W.; Zhang, J.; Jones, C.S. Insights into the genetic architecture of complex traits in napier grass (Cenchrus purpureus) and QTL regions governing forage biomass yield, water use efficiency and feed quality traits. Front. Plant Sci. 2021, 12, 678862. [Google Scholar] [CrossRef]
- Yan, Q.; Li, J.; Lu, L.Y.; Gao, L.J.; Lai, D.W.; Yao, N.; Yi, X.F.; Wu, Z.Y.; Lai, Z.Q.; Zhang, J.Y. Integrated analyses of phenotype, phytohormone, and transcriptome to elucidate the mechanism governing internode elongation in two contrasting elephant grass (Cenchrus purpureus) cultivars. Ind. Crops Prod. 2021, 170, 113693. [Google Scholar] [CrossRef]
- Bai, Y.; Cai, M.; Mu, C.; Cheng, W.; Zheng, H.; Cheng, Z.; Li, J.; Mu, S.; Gao, J. New insights into the local auxin biosynthesis and its effects on the rapid growth of moso bamboo (Phyllostachys edulis). Front. Plant Sci. 2022, 13, 858686. [Google Scholar] [CrossRef]
- Hedden, P.; Sponsel, V. A century of gibberellin research. J. Plant Growth Regul. 2015, 34, 740–760. [Google Scholar] [CrossRef] [PubMed]
- Randoux, M.; Jeauffre, J.; Thouroude, T.; Vasseur, F.; Hamama, L.; Juchaux, M.; Sakr, S.; Foucher, F. Gibberellins regulate the transcription of the continuous flowering regulator, RoKSN, a rose TFL1 homologue. J. Exp. Bot. 2012, 63, 6543–6554. [Google Scholar] [CrossRef]
- Chen, Y.; Hou, M.; Liu, L.; Wu, S.; Shen, Y.; Ishiyama, K.; Kobayashi, M.; McCarty, D.R.; Tan, B.C. The maize DWARF1 encodes a gibberellin 3-oxidase and is dual localized to the nucleus and cytosol. Plant Physiol. 2014, 166, 2028–2039. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.; Harberd, N.P. Auxin promotes Arabidopsis root growth by modulating gibberellin response. Nature 2003, 421, 740–743. [Google Scholar] [CrossRef]
- Ishii, Y.; Yamano, A.; Idota, S. Effects of short-day and gibberellic acid treatments on summer vegetative propagation of napier grass (Pennisetum purpureum Schumach). Int. J. Agron. 2016, 2016, 9606914. [Google Scholar] [CrossRef]
- Zhang, X.; He, L.; Zhao, B.; Zhou, S.; Li, Y.; He, H.; Bai, Q.; Zhao, W.; Guo, S.; Liu, Y.; et al. Dwarf and Increased Branching 1 controls plant height and axillary bud outgrowth in Medicago truncatula. J. Exp. Bot. 2020, 71, 6355–6365. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, H.; Zhu, Q.; Gao, Y.; Wang, H.; Zhao, L.; Wang, Y.; Xi, F.; Wang, W.; Yang, Y.; et al. Transcriptome characterization of moso bamboo (Phyllostachys edulis) seedlings in response to exogenous gibberellin applications. BMC Plant Biol. 2018, 18, 125. [Google Scholar] [CrossRef]
- Chen, M.; Guo, L.; Ramakrishnan, M.; Fei, Z.; Vinod, K.K.; Ding, Y.; Jiao, C.; Gao, Z.; Zha, R.; Wang, C.; et al. Rapid growth of Moso bamboo (Phyllostachys edulis): Cellular roadmaps, transcriptome dynamics, and environmental factors. Plant Cell 2022, 34, 3577–3610. [Google Scholar] [CrossRef]
- Wang, X.; Geng, X.; Yang, L.; Chen, Y.; Zhao, Z.; Shi, W.; Kang, L.; Wu, R.; Lu, C.; Gao, J. Total and mitochondrial transcriptomic and proteomic insights into regulation of bioenergetic processes for shoot fast-growth initiation in moso bamboo. Cells 2022, 11, 1240. [Google Scholar] [CrossRef]
- Zheng, H.F.; Cai, M.M.; Bai, Y.C.; Xu, J.L.; Xie, Y.L.; Song, H.J.; Li, J.; Gao, J. The effect of guttation on the growth of bamboo shoots. Forests 2021, 13, 31. [Google Scholar] [CrossRef]
- Wang, S.; Pei, J.; Li, J.; Tang, G.; Zhao, J.; Peng, X.; Nie, S.; Ding, Y.; Wang, C. Sucrose and starch metabolism during Fargesia yunnanensis shoot growth. Physiol. Plant. 2020, 168, 188–204. [Google Scholar] [CrossRef]
- Chongtham, N.; Bisht, M.S.; Haorongbam, S. Nutritional properties of bamboo shoots: Potential and prospects for utilization as a health food. Compr. Rev. Food Sci. Food Saf. 2011, 10, 153–168. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, L.; Li, Y.; Yang, J.; Yang, H.; Zhao, Y.; Chen, G. Bamboo shoot and its food applications in last decade: An undervalued edible resource from forest to feed future people. Trends Food Sci. Technol. 2024, 146, 104399. [Google Scholar] [CrossRef]
- Hersbach, H.; Bell, B.; Berrisford, P.; Hirahara, S.; Horányi, A.; Muñoz-Sabater, J.; Nicolas, J.; Peubey, C.; Radu, R.; Schepers, D.; et al. The ERA5 global reanalysis. Q. J. R. Meteorol. Soc. 2020, 146, 1999–2049. [Google Scholar] [CrossRef]
- Rigatti, S.J. Random forest. J. Insur. Med. 2017, 47, 31–39. [Google Scholar] [CrossRef] [PubMed]
- McGlothlin, A.E.; Viele, K. Bayesian hierarchical models. JAMA 2018, 320, 2365–2366. [Google Scholar] [CrossRef] [PubMed]
- Stephan, K.E.; Penny, W.D.; Daunizeau, J.; Moran, R.J.; Friston, K.J. Bayesian model selection for group studies. NeuroImage 2009, 46, 1004–1017. [Google Scholar] [CrossRef] [PubMed]
- Bauman, D.; Fortunel, C.; Delhaye, G.; Malhi, Y.; Cernusak, L.A.; Bentley, L.P.; Rifai, S.W.; Aguirre-Gutiérrez, J.; Menor, I.O.; Phillips, O.L.; et al. Tropical tree mortality has increased with rising atmospheric water stress. Nature 2022, 608, 528–533. [Google Scholar] [CrossRef]
- Johnson, E.C.B.; Bian, S.; Haque, R.U.; Carter, E.K.; Watson, C.M.; Gordon, B.A.; Ping, L.; Duong, D.M.; Epstein, M.P.; McDade, E.; et al. Cerebrospinal fluid proteomics define the natural history of autosomal dominant Alzheimer’s disease. Nat. Med. 2023, 29, 1979–1988. [Google Scholar] [CrossRef]
- Bürkner, P.C. brms: An R package for Bayesian multilevel models using stan. J. Stat. Softw. 2017, 80, 1–28. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Casal, J.J.; Balasubramanian, S. Thermomorphogenesis. Annu. Rev. Plant Biol. 2019, 70, 321–346. [Google Scholar] [CrossRef]
- Manandhar, A.; Pichaco, J.; McAdam, S.A.M. Abscisic acid increase correlates with the soil water threshold of transpiration decline during drought. Plant Cell Environ. 2024, 47, 5067–5075. [Google Scholar] [CrossRef] [PubMed]
- Kramer, P.J.; Boyer, J.S. Water Relations of Plants and Soils; Academic press: San Diego, CA, USA, 1995. [Google Scholar]
- Zhang, M.; Chen, S.; Jiang, H.; Cao, Q. The water transport profile of Phyllostachys edulis during the explosive growth phase of bamboo shoots. Glob. Ecol. Conserv. 2020, 24, e01251. [Google Scholar] [CrossRef]
- Zhang, F.; Xing, Z.; Rees, H.W.; Dong, Y.; Li, S.; Meng, F. Assessment of effects of two runoff control engineering practices on soil water and plant growth for afforestation in a semi-arid area after 10 years. Ecol. Eng. 2014, 64, 430–442. [Google Scholar] [CrossRef]
- Armstrong, W. Aeration in Higher Plants. Adv. Bot. Res. 1980, 7, 225–332. [Google Scholar] [CrossRef]
- Jackson, M.B.; Colmer, T.D. Response and Adaptation by Plants to Flooding Stress. Ann. Bot. 2005, 96, 501–505. [Google Scholar] [CrossRef]
- Brolsma, R.J.; van Beek, L.P.H.; Bierkens, M.F.P. Vegetation competition model for water and light limitation. II: Spatial dynamics of groundwater and vegetation. Ecol. Model. 2010, 221, 1364–1377. [Google Scholar] [CrossRef]
- Stromberg, J.C.; Tiller, R.; Richter, B. Effects of Groundwater Decline on Riparian Vegetation of Semiarid Regions: The San Pedro, Arizona. Ecol. Appl. 1996, 6, 113–131. [Google Scholar] [CrossRef]
- Zhu, L.; Gong, H.; Dai, Z.; Xu, T.; Su, X. An integrated assessment of the impact of precipitation and groundwater on vegetation growth in arid and semiarid areas. Environ. Earth Sci. 2015, 74, 5009–5021. [Google Scholar] [CrossRef]
- Zheng, H.; Bai, Y.; Li, X.; Song, H.; Cai, M.; Cheng, Z.; Mu, S.; Li, J.; Gao, J. Photosynthesis, phytohormone signaling and sugar catabolism in the culm sheaths of Phyllostachys edulis. Plants 2022, 11, 2866. [Google Scholar] [CrossRef]
- Wooliver, R.; Vtipilthorpe, E.E.; Wiegmann, A.M.; Sheth, S.N. A viewpoint on ecological and evolutionary study of plant thermal performance curves in a warming world. AoB Plants 2022, 14, plac016. [Google Scholar] [CrossRef]
- Lin, H.; Chen, Y.; Zhang, H.; Fu, P.; Fan, Z. Stronger cooling effects of transpiration and leaf physical traits of plants from a hot dry habitat than from a hot wet habitat. Funct. Ecol. 2017, 31, 2202–2211. [Google Scholar] [CrossRef]
- Emenecker, R.J.; Strader, L.C. Auxin-abscisic acid interactions in plant growth and development. Biomolecules 2020, 10, 281. [Google Scholar] [CrossRef]
- Liscum, E.; Reed, J.W. Genetics of Aux/IAA and ARF action in plant growth and development. Plant Mol. Biol. 2002, 49, 387–400. [Google Scholar] [CrossRef]
- Nagai, K.; Mori, Y.; Ishikawa, S.; Furuta, T.; Gamuyao, R.; Niimi, Y.; Hobo, T.; Fukuda, M.; Kojima, M.; Takebayashi, Y.; et al. Antagonistic regulation of the gibberellic acid response during stem growth in rice. Nature 2020, 584, 109–114. [Google Scholar] [CrossRef]
- Sauter, M.; Mekhedov, S.L.; Kende, H. Gibberellin promotes histone H1 kinase activity and the expression of cdc2 and cyclin genes during the induction of rapid growth in deepwater rice internodes. Plant J. 1995, 7, 623–632. [Google Scholar] [CrossRef] [PubMed]
- Rolland, F.; Baena-Gonzalez, E.; Sheen, J. Sugar sensing and signaling in plants: Conserved and novel mechanisms. Annu. Rev. Plant Biol. 2006, 57, 675–709. [Google Scholar] [CrossRef]
- Atkinson, N.J.; Urwin, P.E. The interaction of plant biotic and abiotic stresses: From genes to the field. J. Exp. Bot. 2012, 63, 3523–3543. [Google Scholar] [CrossRef]
- Mittler, R. Abiotic stress, the field environment and stress combination. Trends Plant Sci. 2006, 11, 15–19. [Google Scholar] [CrossRef]
- Peleg, Z.; Blumwald, E. Hormone balance and abiotic stress tolerance in crop plants. Curr. Opin. Plant Biol. 2011, 14, 290–295. [Google Scholar] [CrossRef]
- Ku, Y.-S.; Sintaha, M.; Cheung, M.; Lam, H. Plant hormone signaling crosstalks between biotic and abiotic stress responses. Int. J. Mol. Sci. 2018, 19, 3206. [Google Scholar] [CrossRef]
- Savolainen, O.; Lascoux, M.; Merilä, J. Ecological genomics of local adaptation. Nat. Rev. Genet. 2013, 14, 807–820. [Google Scholar] [CrossRef] [PubMed]
- Kotak, S.; Larkindale, J.; Lee, U.; von Koskull-Döring, P.; Vierling, E.; Scharf, K.-D. Complexity of the heat stress response in plants. Curr. Opin. Plant Biol. 2007, 10, 310–316. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, N.; Mittler, R. Reactive oxygen species and temperature stresses: A delicate balance between signaling and destruction. Physiol. Plant. 2006, 126, 45–51. [Google Scholar] [CrossRef]
- Guy, C.; Kaplan, F.; Kopka, J.; Selbig, J.; Hincha, D.K. Metabolomics of temperature stress. Physiol. Plant. 2008, 132, 220–235. [Google Scholar] [CrossRef]
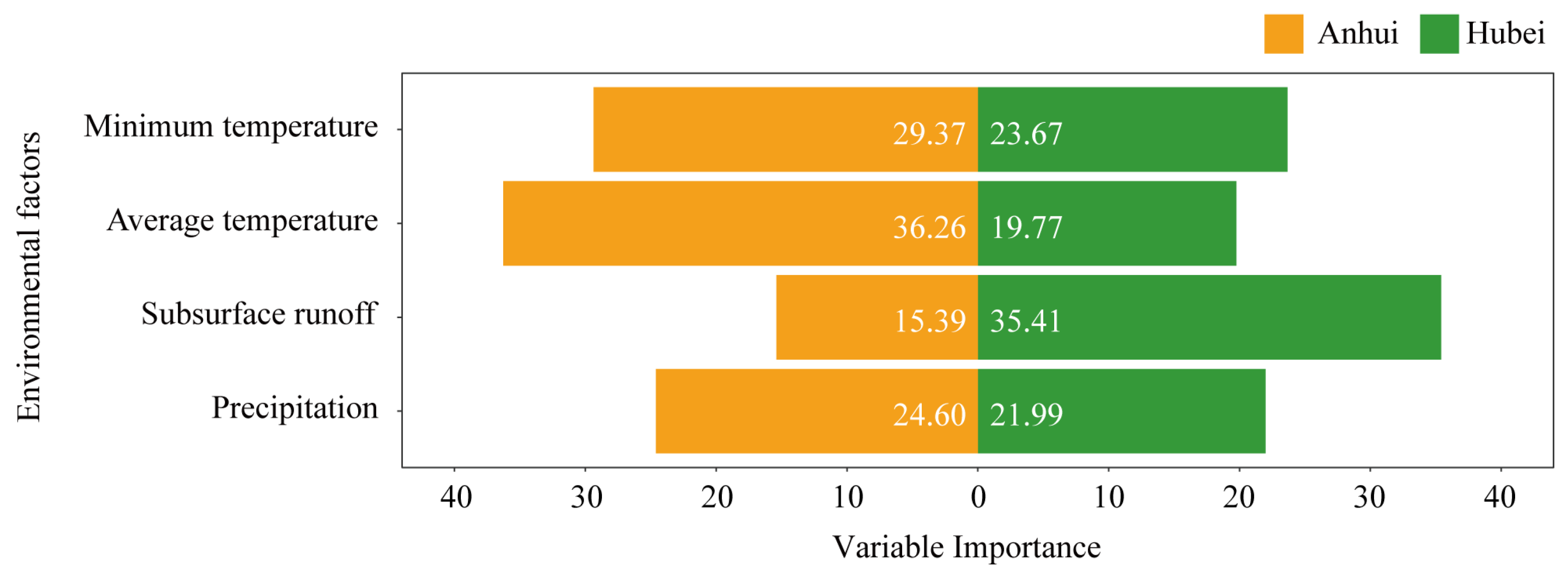
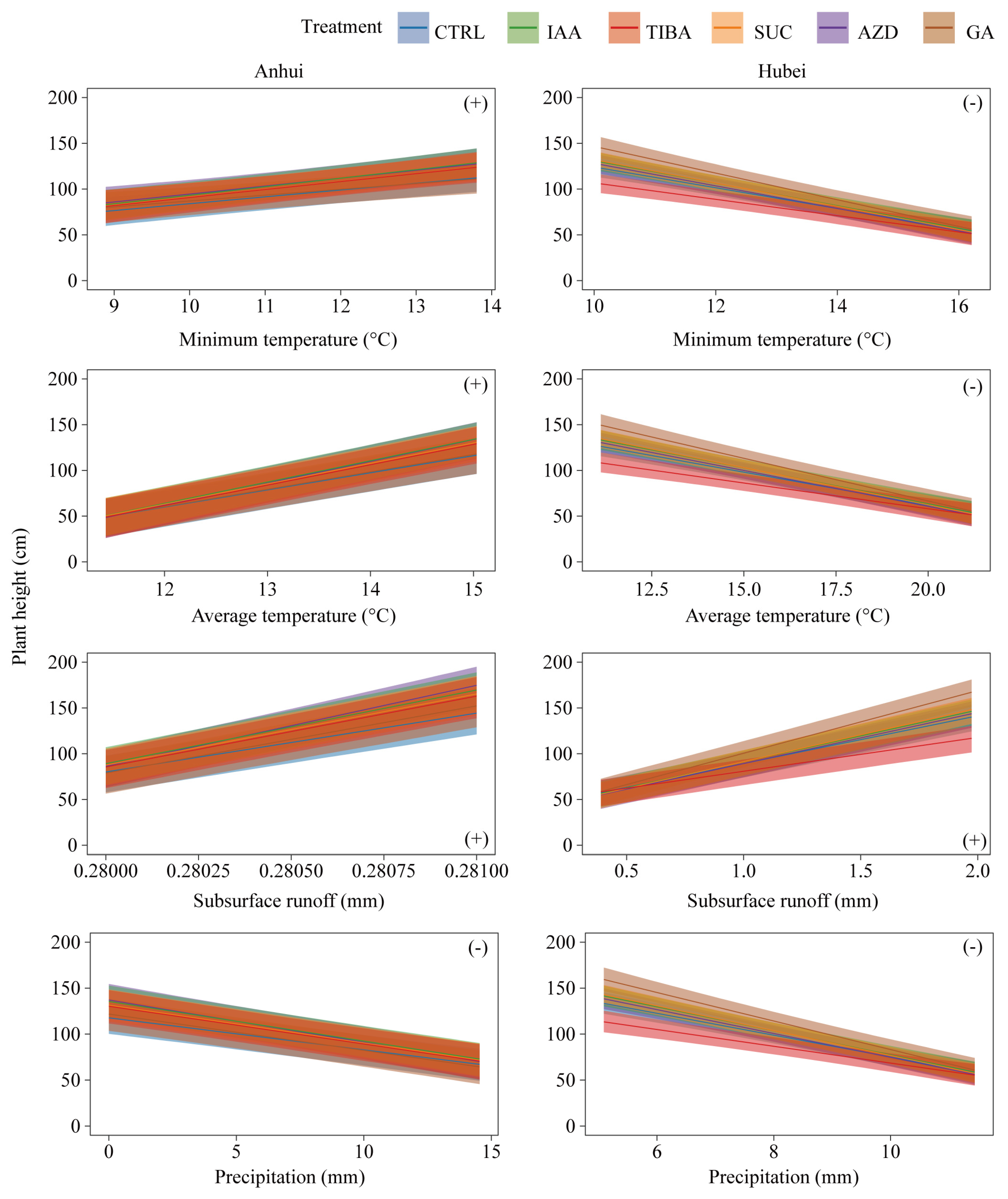
| Model Structure | Anhui | Hubei | ||||||
|---|---|---|---|---|---|---|---|---|
| Estimate | Estimate Error | l-95% CI | u-95% CI | Estimate | Estimate Error | l-95% CI | u-95% CI | |
| Minimum temperature × IAA | 1.87 | 2.19 | −2.43 | 6.18 | −0.97 | 1.72 | −4.36 | 2.42 |
| Minimum temperature × TIBA | 1.23 | 2.46 | −3.55 | 6.06 | 2.39 | 1.76 | −1.09 | 5.83 |
| Minimum temperature × SUC | 1.09 | 2.18 | −3.18 | 5.40 | −1.37 | 1.63 | −4.47 | 1.81 |
| Minimum temperature × AZD | 1.18 | 2.46 | −3.66 | 5.97 | −0.99 | 1.72 | −4.37 | 2.41 |
| Minimum temperature × GA | −1.16 | 2.14 | −5.32 | 3.03 | −3.26 | 1.93 | −7.09 | 0.53 |
| Average temperature × IAA | 4.53 | 2.10 | 0.46 | 8.68 | −0.63 | 1.05 | −2.67 | 1.43 |
| Average temperature × TIBA | 3.45 | 2.35 | −1.18 | 7.99 | 1.60 | 1.07 | −0.47 | 3.66 |
| Average temperature × SUC | 5.00 | 2.39 | 0.30 | 9.65 | −0.88 | 0.98 | −2.82 | 1.04 |
| Average temperature × AZD | 3.48 | 2.12 | −0.64 | 7.68 | −0.61 | 1.06 | −2.70 | 1.45 |
| Average temperature × GA | 0.24 | 2.11 | −3.89 | 4.38 | −2.10 | 1.15 | −4.35 | 0.13 |
| Subsurface runoff × IAA | 3.30 | 3.95 | −4.28 | 11.11 | 3.08 | 2.40 | −1.64 | 7.83 |
| Subsurface runoff × TIBA | 2.57 | 4.46 | −6.31 | 11.38 | −10.35 | 2.50 | −15.19 | −5.35 |
| Subsurface runoff × SUC | 2.34 | 4.02 | −5.49 | 10.12 | 4.22 | 2.28 | −0.24 | 8.81 |
| Subsurface runoff × AZD | 2.85 | 4.56 | −6.00 | 11.56 | 2.56 | 2.43 | −2.18 | 7.36 |
| Subsurface runoff × GA | −1.30 | 3.97 | −9.19 | 6.51 | 11.15 | 2.71 | 5.97 | 16.47 |
| Precipitation × IAA | −0.82 | 0.57 | −1.93 | 0.29 | −1.16 | 1.61 | −4.29 | 2.03 |
| Precipitation × TIBA | −0.60 | 0.64 | −1.86 | 0.68 | 2.75 | 1.65 | −0.50 | 5.96 |
| Precipitation × SUC | −0.62 | 0.57 | −1.74 | 0.50 | −1.57 | 1.53 | −4.54 | 1.45 |
| Precipitation × AZD | −1.10 | 0.64 | −2.34 | 0.15 | −1.08 | 1.64 | −4.25 | 2.07 |
| Precipitation × GA | −0.45 | 0.56 | −1.58 | 0.64 | −3.63 | 1.80 | −7.16 | −0.15 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, C.; Xu, J.; Mu, C.; Xie, Y.; Cheng, W.; Gao, J. Interactive Effects of Exogenous Hormones, Sucrose, and Environmental Factors on the Growth of Phyllostachys edulis Shoots. Agronomy 2025, 15, 2095. https://doi.org/10.3390/agronomy15092095
Wu C, Xu J, Mu C, Xie Y, Cheng W, Gao J. Interactive Effects of Exogenous Hormones, Sucrose, and Environmental Factors on the Growth of Phyllostachys edulis Shoots. Agronomy. 2025; 15(9):2095. https://doi.org/10.3390/agronomy15092095
Chicago/Turabian StyleWu, Chongyang, Junlei Xu, Changhong Mu, Yali Xie, Wenlong Cheng, and Jian Gao. 2025. "Interactive Effects of Exogenous Hormones, Sucrose, and Environmental Factors on the Growth of Phyllostachys edulis Shoots" Agronomy 15, no. 9: 2095. https://doi.org/10.3390/agronomy15092095
APA StyleWu, C., Xu, J., Mu, C., Xie, Y., Cheng, W., & Gao, J. (2025). Interactive Effects of Exogenous Hormones, Sucrose, and Environmental Factors on the Growth of Phyllostachys edulis Shoots. Agronomy, 15(9), 2095. https://doi.org/10.3390/agronomy15092095






