Natural Contamination of Rice with Ustiloxins and the Connection with Climate Conditions in Southern China
Abstract
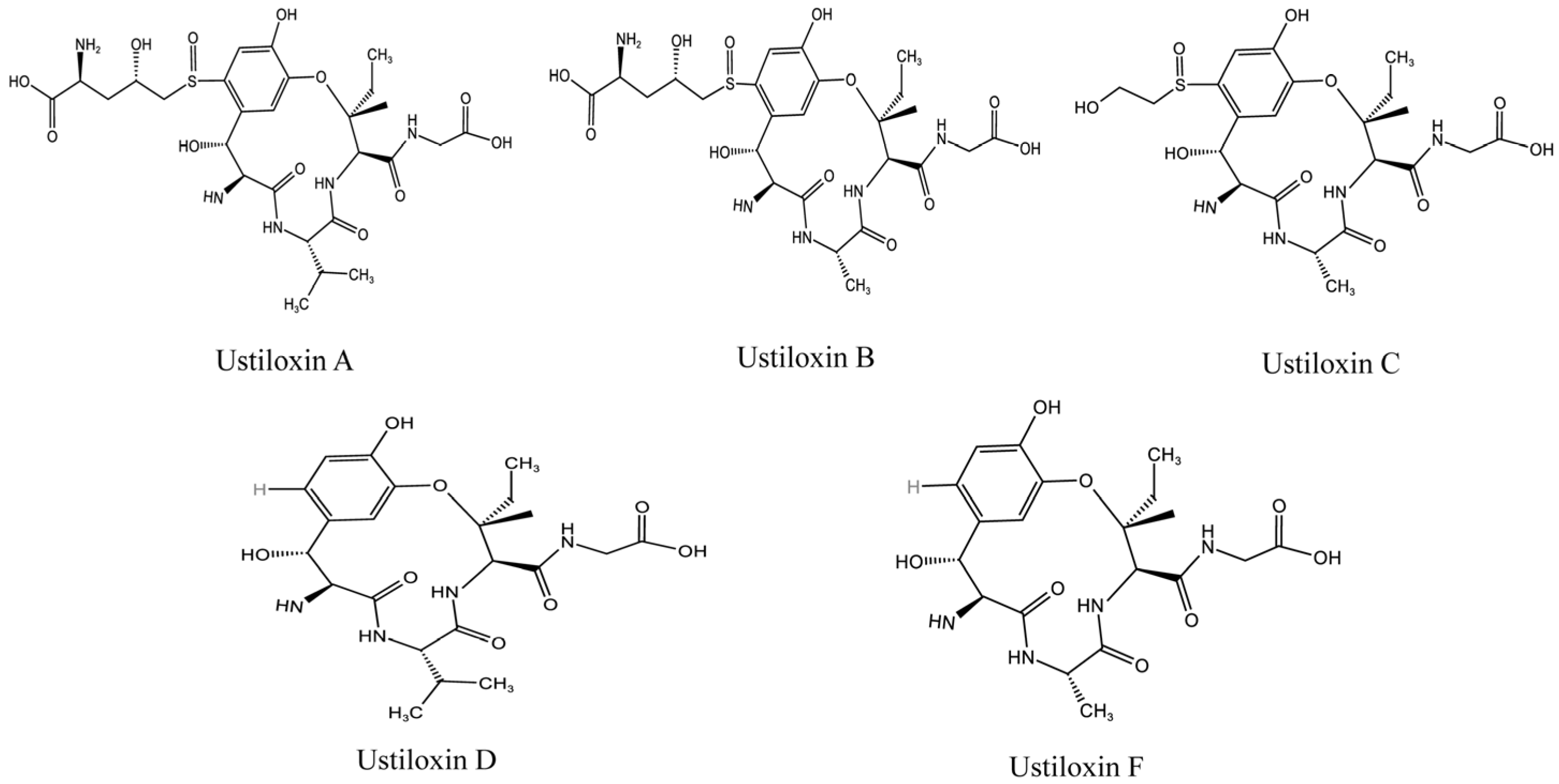
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
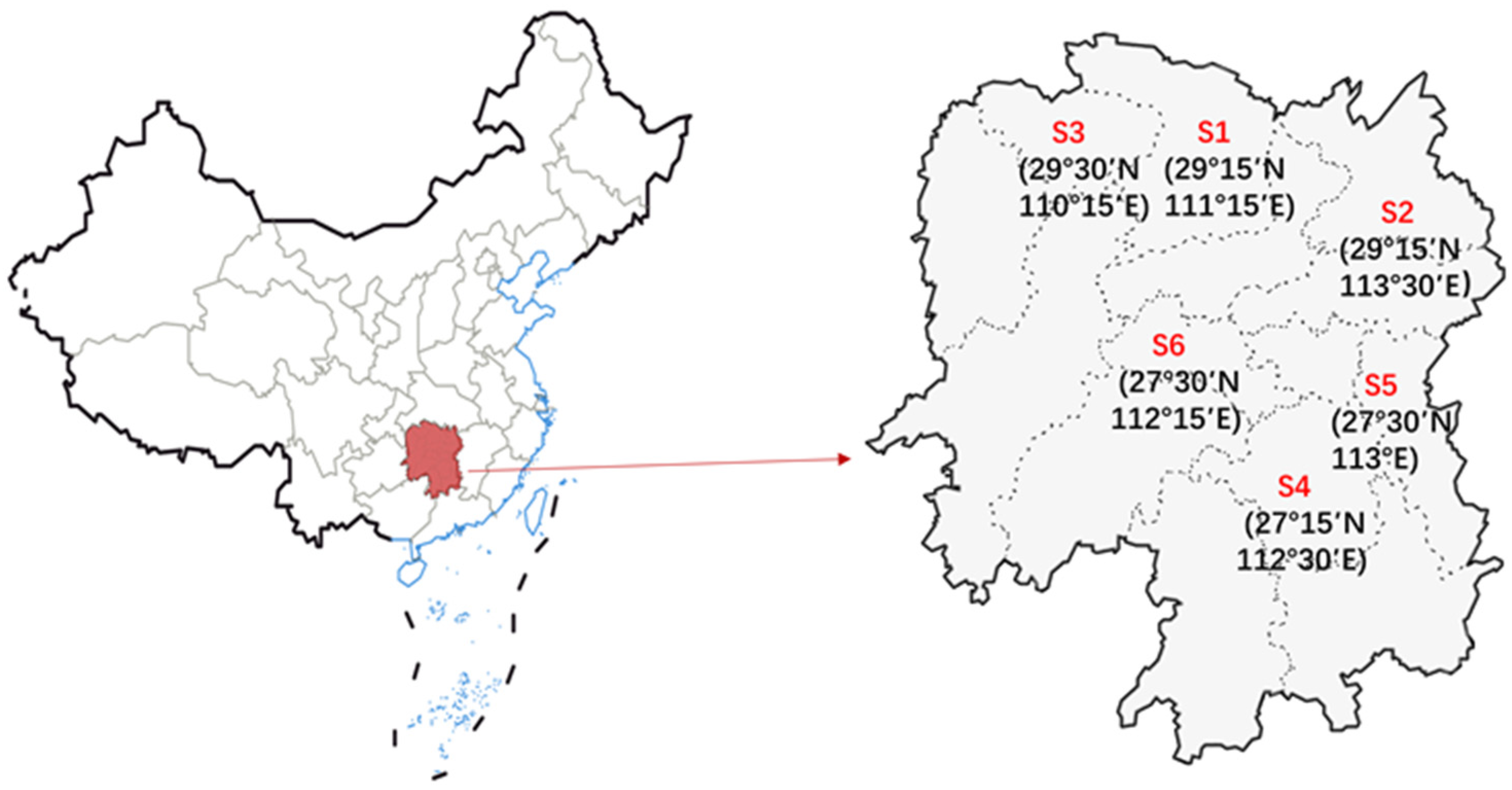
2.2. Samples
2.3. Analytical Method
2.3.1. Extraction
2.3.2. LC-MS/MS Analysis
2.4. Method Performance
2.5. Data Analysis
3. Results and Discussion
3.1. Validation of the Analytical Method
3.2. The Occurrence of Ustiloxins in Paddy Samples
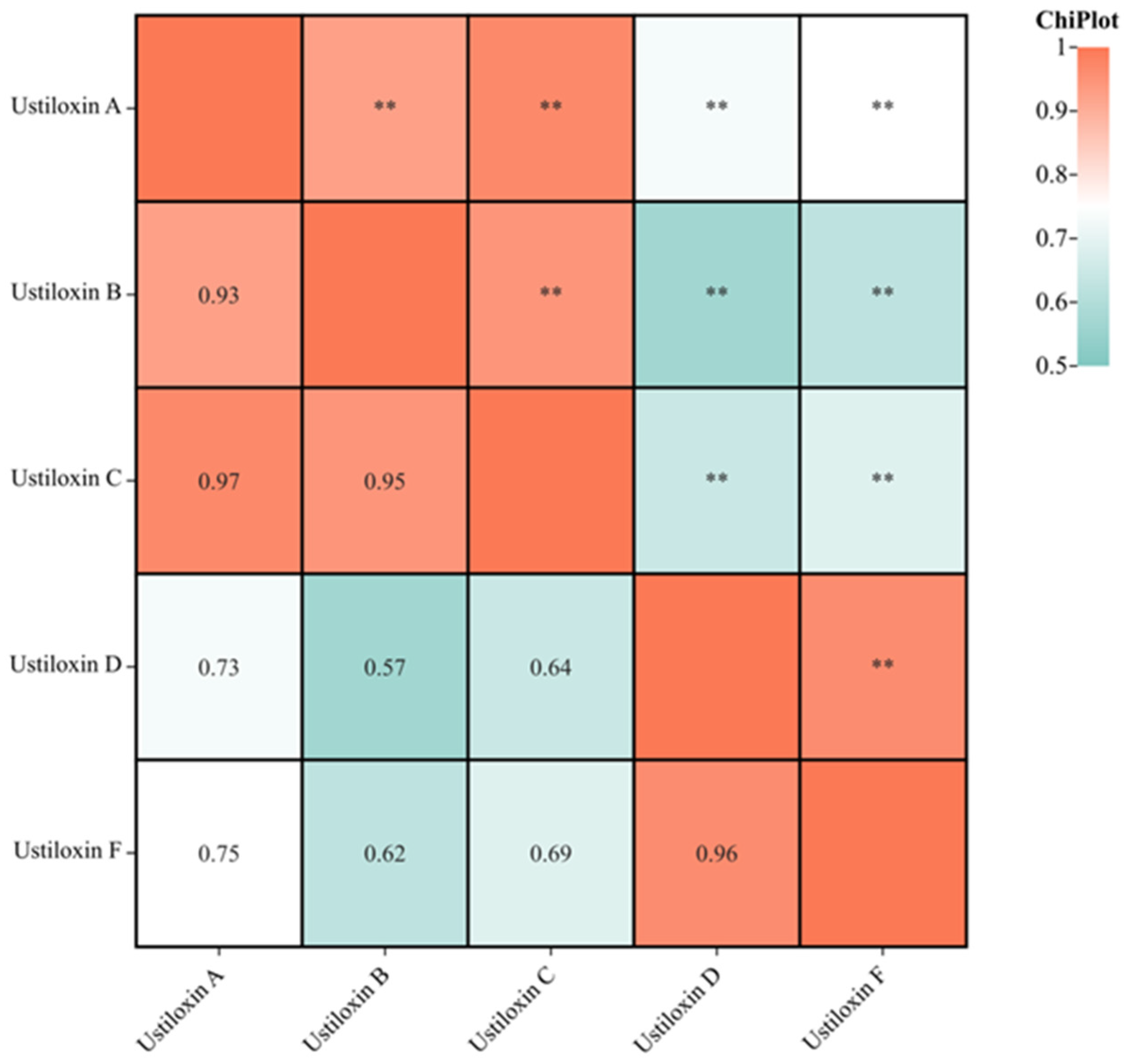
3.3. Correlation among the Contents of Five Ustiloxins in Paddy Samples
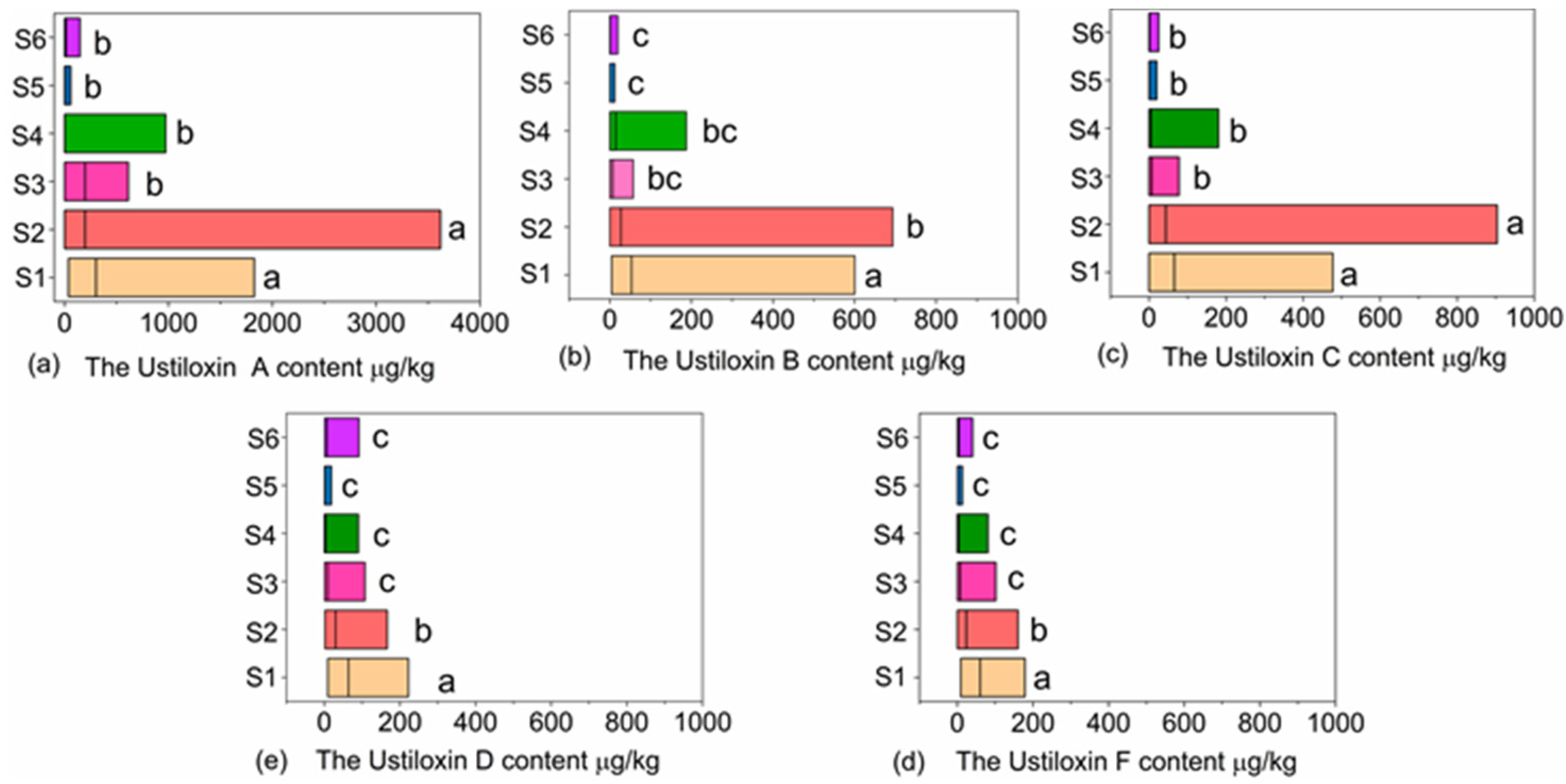
3.4. The Ustiloxins Content in Paddy Samples Varied across Geographic Regions
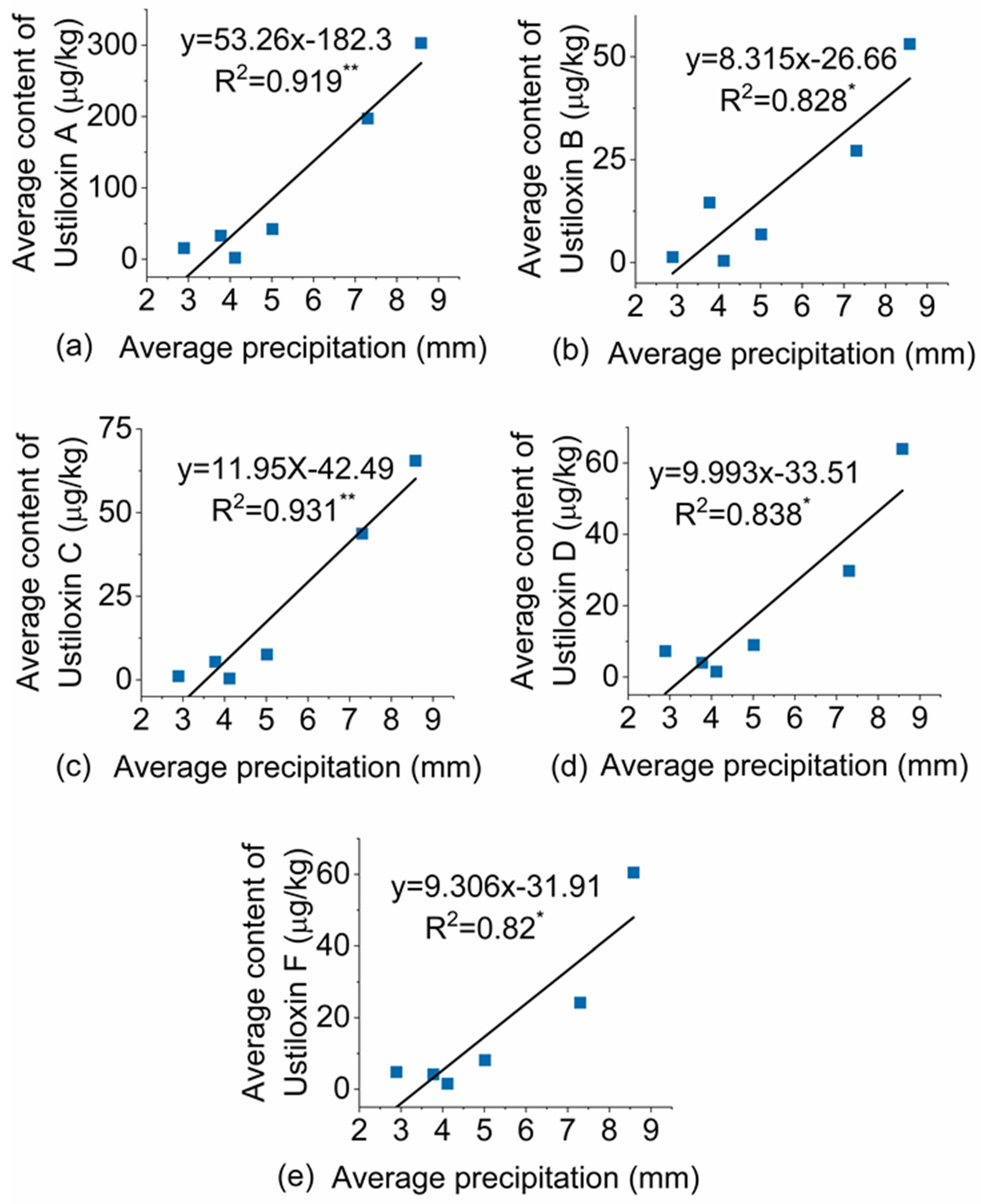
3.5. The Effect of Weather Conditions on Ustiloxin Contamination of Paddy Samples
3.6. Comparison of Our Study with Other Studies
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Bao, J.; Ying, Y.; Xu, Y.; Zhou, X.; Yan, S.; Pang, Y. Variation in physicochemical properties and nutritional quality in chalky mutants derived from an indica rice. J. Cereal Sci. 2020, 91, 102899. [Google Scholar] [CrossRef]
- Chandravarnan, P.; Agyei, D.; Ali, A. Green and sustainable technologies for the decontamination of fungi and mycotoxins in rice: A review. Trends Food Sci. Technol. 2022, 124, 278–295. [Google Scholar] [CrossRef]
- Reddy, K.R.N.; Reddy, C.S.; Abbas, H.K.; Abel, C.A.; Muralidharan, K. Mycotoxigenic fungi, mycotoxins, and management of rice grains. Toxin Rev. 2008, 27, 287–317. [Google Scholar] [CrossRef]
- Mazaheri, M. Investigating the possibility of the occurrence of mycotoxins in rice and rice flour imported to Iran. J. Food Compos. Anal. 2023, 122, 105464. [Google Scholar] [CrossRef]
- Laganà, A. Introduction to the toxins special issue on LC-MS/MS methods for mycotoxin analysis. Toxin 2017, 9, 325. [Google Scholar] [CrossRef] [PubMed]
- Ferre, F.S. Worldwide occurrence of mycotoxins in rice. Food Control 2016, 62, 291–298. [Google Scholar] [CrossRef]
- Reinhold, L.; Reinhardt, K. Mycotoxins in foods in lower Saxony (Germany): Results of official control analyses performed in 2009. Mycotoxin Res. 2011, 27, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Aydin, A.; Aksu, H.; Gunsen, U. Mycotoxin levels and incidence of mould in Turkish rice. Environ. Monit. Assess. 2011, 178, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Lai, X.W.; Liu, R.C.; Ruan, C.Q.; Zhang, H.; Liu, C.L. Occurrence of aflatoxins and ochratoxin A in rice samples from six provinces in China. Food Control 2015, 50, 401–404. [Google Scholar] [CrossRef]
- Li, R.; Wang, X.; Zhou, T.; Yang, D.G.; Wang, Q.; Zhou, Y. Occurrence of four mycotoxins in cereal and oil products in Yangtze Delta region of China and their food safety risks. Food Control 2014, 35, 117–122. [Google Scholar] [CrossRef]
- Mesterhazy, A.; Olah, J.; Popp, J. Losses in the grain supply chain: Causes and solutions. Sustainability 2020, 12, 2342. [Google Scholar] [CrossRef]
- Commission Regulation (EU). 2023/915 of 25 April 2023 on maximum levels for certain contaminants in food and repealing Regulation (EC) No 1881/2006. Off. J. Eur. Union 2023, 119, 103–157. [Google Scholar]
- Taghizadeh, S.F.; Rezaee, R.; Badiebostan, H.; Giesy, J.P.; Karimi, G. Occurrence of mycotoxins in rice consumed by Iranians: A probabilistic assessment of risk to health. Food Addit. Contam. Part A 2020, 37, 342–354. [Google Scholar] [CrossRef] [PubMed]
- Khanal, S.; Gaire, S.; Zhou, X.G. Kernel Smut and False Smut: The Old-Emerging Diseases of Rice—A Review. Phytopathology 2023, 113, 931–944. [Google Scholar] [CrossRef] [PubMed]
- Brooks, S.A.; Anders, M.M.; Yeater, K.M. Effect of cultural management practices on the severity of false smut of rice. Plant Dis. 2009, 93, 1202–1208. [Google Scholar] [CrossRef]
- Koiso, Y.; Li, Y.; Iwasaki, S.; Hanaoka, K.; Kobayashi, T.; Fujita, Y.; Yaegashi, H.; Sato, Z. Ustiloxins, antimitotic cyclic peptides from false smut balls on rice panicles caused by Ustilaginoidea virens. J. Antibiot. 1994, 47, 765–773. [Google Scholar] [CrossRef] [PubMed]
- Meng, J.J.; Sun, W.B.; Mao, Z.L.; Xu, D.; Wang, X.H.; Lu, S.Q.; Lai, D.W.; Liu, Y.; Zhou, L.G.; Zhang, G.Z. Main ustilaginoidins and their distribution in rice false smut balls. Toxins 2015, 7, 4023–4034. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, J.; Lai, D.; Wang, W.; Dai, J.; Zhou, L.; Liu, Y. Ustiloxin G, a new cyclopeptide mycotoxin from rice false smut balls. Toxins 2017, 9, 54. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.Y.; Sun, L.H.; Mou, R.X.; Lin, X.Y.; Zhou, R.; Ma, Y.N.; Chen, M.X. Analysis of ustiloxins in rice using polymer cation exchange cleanup followed by liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2016, 1476, 46–52. [Google Scholar] [CrossRef]
- Koiso, Y.; Natori, M.; Iwasaki, S.; Sato, S.; Sonoda, R.; Fujita, Y.; Yaegashi, H.; Sato, Z. Ustiloxin: Aphytotoxin and a mycotoxin from false smut balls on rice panicles. Tetrahedron Lett. 1992, 33, 4157–4160. [Google Scholar] [CrossRef]
- Cheng, S.Y.; Liu, H.; Sun, Q.; Kong, R.; Letcher, R.J.; Liu, C.S. Occurrence of the fungus mycotoxin, ustiloxin A, in surface waters of paddy fields in Enshi, Hubei, China, and toxicity in Tetrahymena thermophila. Environ. Pollut. 2019, 251, 901–909. [Google Scholar] [CrossRef]
- Nakamura, K.; Izumiyama, N.; Ohtsubo, K.; Koiso, Y.; Iwasaki, S. Apoptosis induced in the liver, kidney and urinary bladder of mice by the fungal toxin produced by Ustilaginoidea virens. Mycotoxins 1993, 38, 25–30. [Google Scholar] [CrossRef][Green Version]
- Fu, X.; Wang, W.; Li, Y.; Wang, X.; Tan, G.; Lai, D.; Wang, M.G.; Zhou, L.G.; Wang, B.M. Development of a monoclonal antibody with equal reactivity to ustiloxins A and B for quantification of main cyclopeptide mycotoxins in rice samples. Food Control 2018, 92, 201–207. [Google Scholar] [CrossRef]
- Shen, F.; Wu, Q.; Shao, X.; Zhang, Q. Non-destructive and rapid evaluation of aflatoxins in brown rice by using near-infrared and mid-infrared spectroscopic techniques. J. Food Sci. Technol. 2018, 55, 1175–1184. [Google Scholar] [CrossRef] [PubMed]
- Shan, T.; Sun, W.; Liu, H.; Gao, S.; Lu, S.; Wang, M.; Sun, W.; Chen, Z.; Wang, S.; Zhou, L. Determination and analysis of ustiloxins A and B by LC-ESI-MS and HPLC in false smut balls of rice. Int. J. Mol. Sci. 2012, 13, 11275–11287. [Google Scholar] [CrossRef]
- Liao, C.; Wong, J.; Zhang, K.; Hayward, D.; Lee, N.; Trucksess, M. Multi-mycotoxin analysis of finished grain and nut products using high-performance liquid chromatography-triple-quadrupole mass spectrometry. J. Agric. Food Chem. 2013, 61, 4771–4782. [Google Scholar] [CrossRef]
- Al-Zoreky, N.S.; Saleh, F.A. Limited survey on aflatoxin contamination in rice. Saudi J. Biol. Sci. 2019, 26, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Chhaya, R.S.; O’Brien, J.; Cummins, E. Feed to fork risk assessment of mycotoxins under climate change influences-recent developments. Trends Food Sci. Technol. 2022, 126, 126–141. [Google Scholar] [CrossRef]
- Moretti, A.; Pascale, M.; Logrieco, A.F. Mycotoxin risks under a climate change scenario in Europe. Trends Food Sci. Technol. 2019, 84, 38–40. [Google Scholar] [CrossRef]
- Mahato, D.K.; Devi, S.; Pandhi, S.; Sharma, B.; Maurya, K.K.; Mishra, S.; Dhawan, K.; Selvakumar, R.; Kamle, M.; Mishra, A.K.; et al. Occurrence, impact on agriculture, human health, and management strategies of zearalenone in food and feed: A review. Toxins 2021, 13, 92. [Google Scholar] [CrossRef]
- Song, J.H.; Yan, W.; Chen, L.R.; Zhang, S.J.; Chuan, N.; Zhu, D.; Liang, Y.; Luo, J.F.; Dou, Z.; Gao, H.; et al. Higher relative humidity and more moderate temperatures increase the severity of rice false smut disease in the rice–crayfish coculture system. Food Energy Secur. 2022, 11, 323. [Google Scholar]
- Sun, X.D.; Su, P.; Shan, H. Mycotoxin contamination of rice in China. J. Food Sci. 2017, 82, 573–584. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.Y.; Matsumoto, H.; Lv, T.X.; Zhan, C.F.; Fang, H.D.; Pan, Q.Q.; Xu, H.R.; Fan, X.Y.; Chu, T.Y.; Chen, S.L.; et al. Phyllosphere microbiome induces host metabolic defence against rice false-smut disease. Nat. Microbiol. 2023, 8, 1419–1433. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, A.; Gkrillas, A.; Dorne, J.L.; Dall’Asta, C.; Palumbo, R.; Lima, N.; Battilani, P.; Venâncio, A.; Giorni, P. Pre- and postharvest strategies to minimize mycotoxin contamination in the rice food chain. Compr. Rev. Food Sci. Food Saf. 2019, 18, 441–454. [Google Scholar] [CrossRef] [PubMed]
- Koiso, Y.; Morisaki, N.; Yamashita, Y.; Mitsui, Y.; Shirai, R.; Hashimoto, Y.; Iwasaki, S. Isolation and structure of an antimitotic cyclic peptide, ustiloxin F: Chemical interrelation with a homologous peptide, ustiloxin B. J. Antibiot. 1998, 51, 418–422. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Qian, S.A.; Fan, K.; Yu, Y.; Liu, X.; Liu, H.; Meng, J.; Zhao, Z.; Han, Z. Natural occurrence of ustiloxins in rice from five provinces in China and the removal efficiencies of different milling steps. J. Sci. Food Agric. 2023, 103, 6272–6279. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; He, D.; Zhao, F.; Hu, J.; Lee, Y.W.; Shi, J.; Xu, J. Extraction and purification of ustiloxin A from rice false smut balls by a combination of macroporous resin and high-speed countercurrent chromatography. Food Prod. Process. Nutr. 2020, 2, 29. [Google Scholar] [CrossRef]
- Ksenija, N. Mycotoxins–climate impact and steps to prevention based on prediction. Acta Vet. 2018, 68, 1–15. [Google Scholar] [CrossRef]
- Cotty, P.J.; Jaime-Garcia, R. Influences of climate on aflatoxin producing fungi and aflatoxin contamination. Int. J. Food Microbiol. 2007, 119, 109–115. [Google Scholar] [CrossRef]
- Franz, E.; Boou, K.; Van Der Fels-Klerx, I. Prediction of deoxynivalenol contentin Dutch winter wheat. J. Food Prot. 2009, 72, 2170–2177. [Google Scholar] [CrossRef]
- Parikka, P.; Hakala, K.; Tiilikkala, K. Expected shifts in Fusarium species’ composition on cereal grain in Northern Europe due to climatic change. Food Addit. Contam. Part A 2012, 29, 1543–1555. [Google Scholar] [CrossRef] [PubMed]
- Jaime-Garcia, R.; Cotty, P.J. Aflatoxin contamination in commercial cottonseed in South Texas. Phytopathology 2003, 93, 1190–1200. [Google Scholar] [CrossRef] [PubMed]
- Gagiu, V.; Mateescu, E.; Armeanu, I.; Dobre, A.A.; Smeu, I.; Cucu, M.E.; Oprea, O.; Iorga, E.; Belc, N. Post-harvest contamination with mycotoxins in the context of the geographic and agroclimatic conditions in Romania. Toxins 2018, 10, 533. [Google Scholar] [CrossRef]
- Reyneri, A. The Role of Climatic Condition on Micotoxin Production in Cereal. Vet. Res. Commun. 2006, 30, 87–92. [Google Scholar] [CrossRef]
| Mycotoxin | Calibration Curve | Correlation Coefficient (r) | LOQ (µg/kg) | LOD (µg/kg) | Matrix Effect (%) | Recovery ± RSD (%) (n = 6) | Intra-Day Precision (%) | Inter-Day Precision (%) | ||
|---|---|---|---|---|---|---|---|---|---|---|
| A1 | A2 | A3 | ||||||||
| Ustiloxin A | y = 429.463x + 269.388 | 0.9999 | 8.12 | 2.68 | 30.09 | 96.4 ± 4.8 | 101.9 ± 6.7 | 105.0 ± 5.3 | 4.1 | 10.6 |
| Ustiloxin B | y = 936.351x + 2949.53 | 0.9998 | 0.82 | 0.27 | 78.30 | 85.1 ± 5.4 | 81.9 ± 7.7 | 91.2 ± 7.8 | 4.3 | 9.9 |
| Ustiloxin C | y = 366.019x + 545.535 | 0.9997 | 16.50 | 4.95 | 46.76 | 88.0 ± 7.4 | 86.2 ± 4.0 | 84.1 ± 6.4 | 3.9 | 8.3 |
| Ustiloxin D | y = 8775.77x − 137.548 | 0.9999 | 0.18 | 0.06 | 8.96 | 81.1 ± 7.3 | 94.5 ± 6.8 | 93.8 ± 7.9 | 1.3 | 2.7 |
| Ustiloxin F | y = 8614.85x + 858.712 | 0.9998 | 0.09 | 0.03 | 32.64 | 90.4 ± 5.9 | 93.0 ± 5.8 | 91.5 ± 3.8 | 3.5 | 4.8 |
| Mycotoxin | No. of Positive/No. of Total Samples | Percentage of Positive Samples (%) | Mean of Positive Samples (µg/kg) | SD (µg/kg) | Min (µg/kg) | Max (µg/kg) |
|---|---|---|---|---|---|---|
| Ustiloxin A | 167/300 | 55.7 | 177.8 | 388.7 | 4.2 | 3620.9 |
| Ustiloxin B | 124/300 | 41.3 | 41.8 | 89.5 | 1.6 | 693.7 |
| Ustiloxin C | 87/300 | 29.0 | 72.0 | 114.5 | 5.4 | 903.9 |
| Ustiloxin D | 281/300 | 93.7 | 20.6 | 39.6 | 0.2 | 222.5 |
| Ustiloxin F | 290/300 | 96.7 | 17.8 | 34.8 | 0.1 | 179.2 |
| Total ustiloxins | 296/300 | 98.7 | 175.8 | 485.7 | 0.27 | 5520.7 |
| July | August | September | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Mean Precipitation/mm | Air Temperature/°C | Air Humidity/% | Mean Precipitation/mm | Air Temperature/°C | Air Humidity/% | Mean Precipitation/mm | Air Temperature/°C | Air Humidity/% | |
| Mean concentrations of ustiloxins/µg/kg | 0.370 * | −0.185 NS | 0.399 * | 0.598 ** | −0.292 NS | 0.361 NS | 0.159 NS | −0.246 NS | 0.394 * |
| The incidence of ustiloxins/% | 0.367 * | −0.363 * | 0.492 ** | 0.507 ** | −0.426 * | 0.435 * | 0.200 NS | −0.406 * | 0.472 ** |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miao, X.; Miao, Y.; Liu, Y.; Tao, S.; Zheng, H.; Kuang, N.; Wang, J.; Tang, Q. Natural Contamination of Rice with Ustiloxins and the Connection with Climate Conditions in Southern China. Agronomy 2024, 14, 976. https://doi.org/10.3390/agronomy14050976
Miao X, Miao Y, Liu Y, Tao S, Zheng H, Kuang N, Wang J, Tang Q. Natural Contamination of Rice with Ustiloxins and the Connection with Climate Conditions in Southern China. Agronomy. 2024; 14(5):976. https://doi.org/10.3390/agronomy14050976
Chicago/Turabian StyleMiao, Xuexue, Ying Miao, Yang Liu, Shuhua Tao, Huabin Zheng, Na Kuang, Jiemin Wang, and Qiyuan Tang. 2024. "Natural Contamination of Rice with Ustiloxins and the Connection with Climate Conditions in Southern China" Agronomy 14, no. 5: 976. https://doi.org/10.3390/agronomy14050976
APA StyleMiao, X., Miao, Y., Liu, Y., Tao, S., Zheng, H., Kuang, N., Wang, J., & Tang, Q. (2024). Natural Contamination of Rice with Ustiloxins and the Connection with Climate Conditions in Southern China. Agronomy, 14(5), 976. https://doi.org/10.3390/agronomy14050976





