Hormone Signals Involved in the Regulation of Cucumber Seedling Lateral Branch Elongation by Far-Red Light
Abstract
1. Introduction
2. Materials and Methods
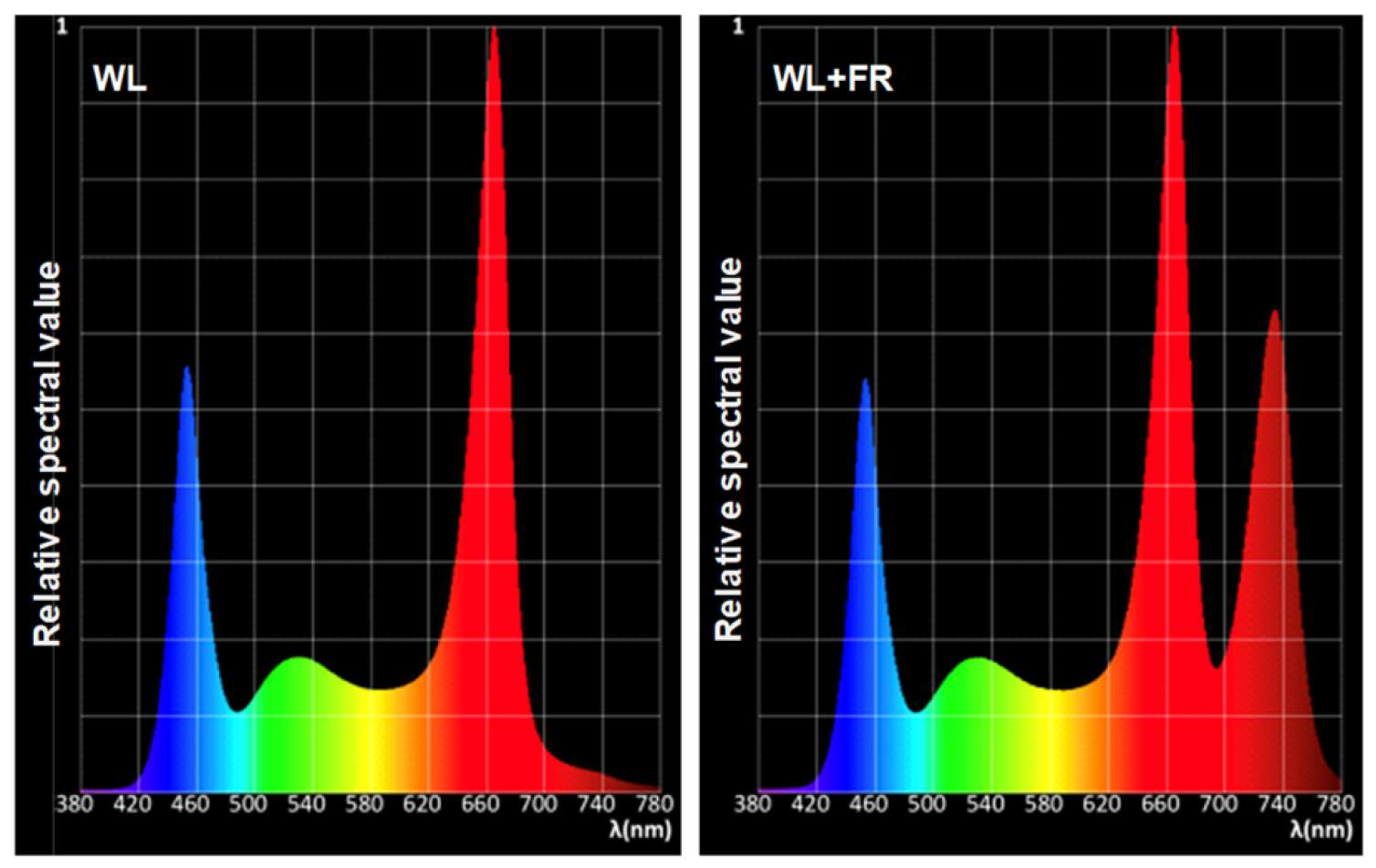
2.1. Plant Materials and Treatments
2.2. Measurement of Endogenous Hormone Contents
2.3. Real-Time Quantitative PCR (RT–qPCR) Analysis
2.4. Statistical Analyses
3. Results
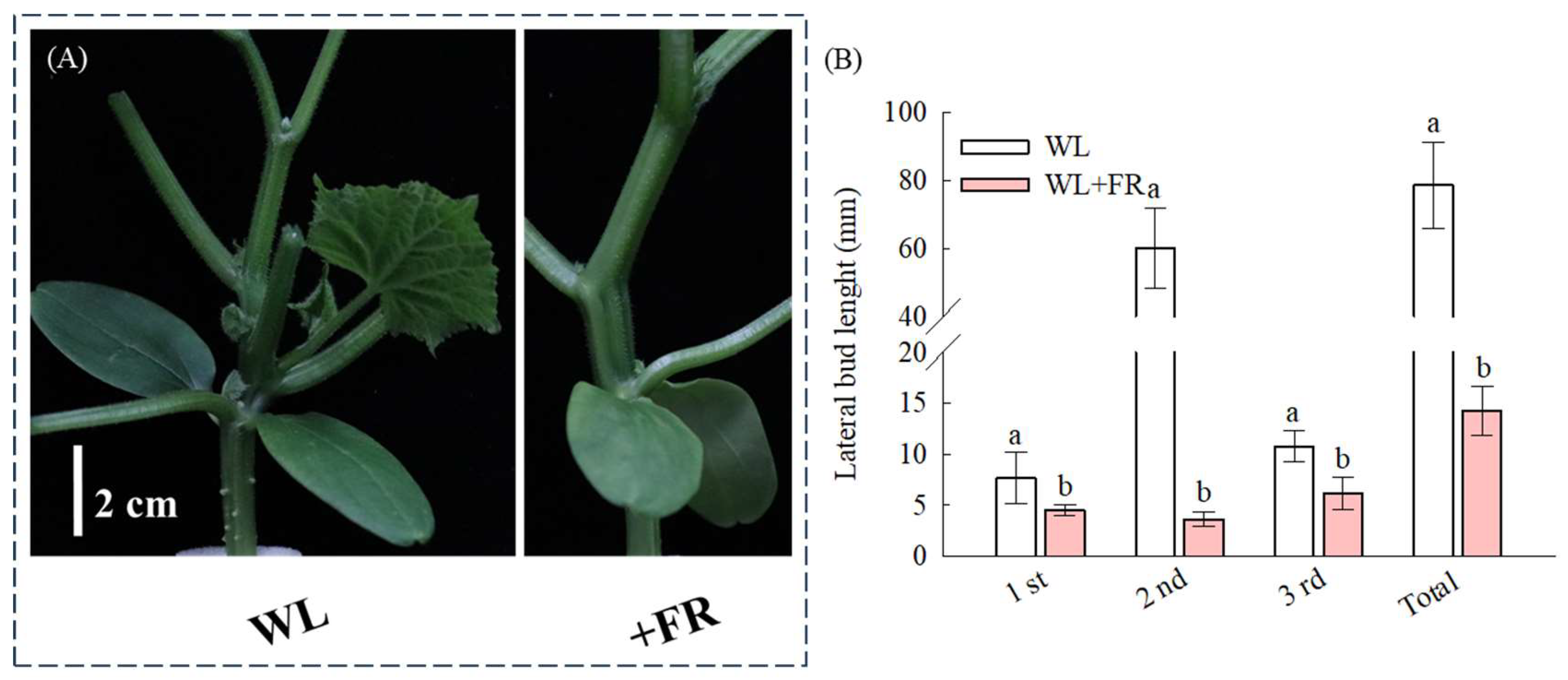
3.1. FR Light Inhibits Lateral Branch Elongation in Cucumber Seedlings
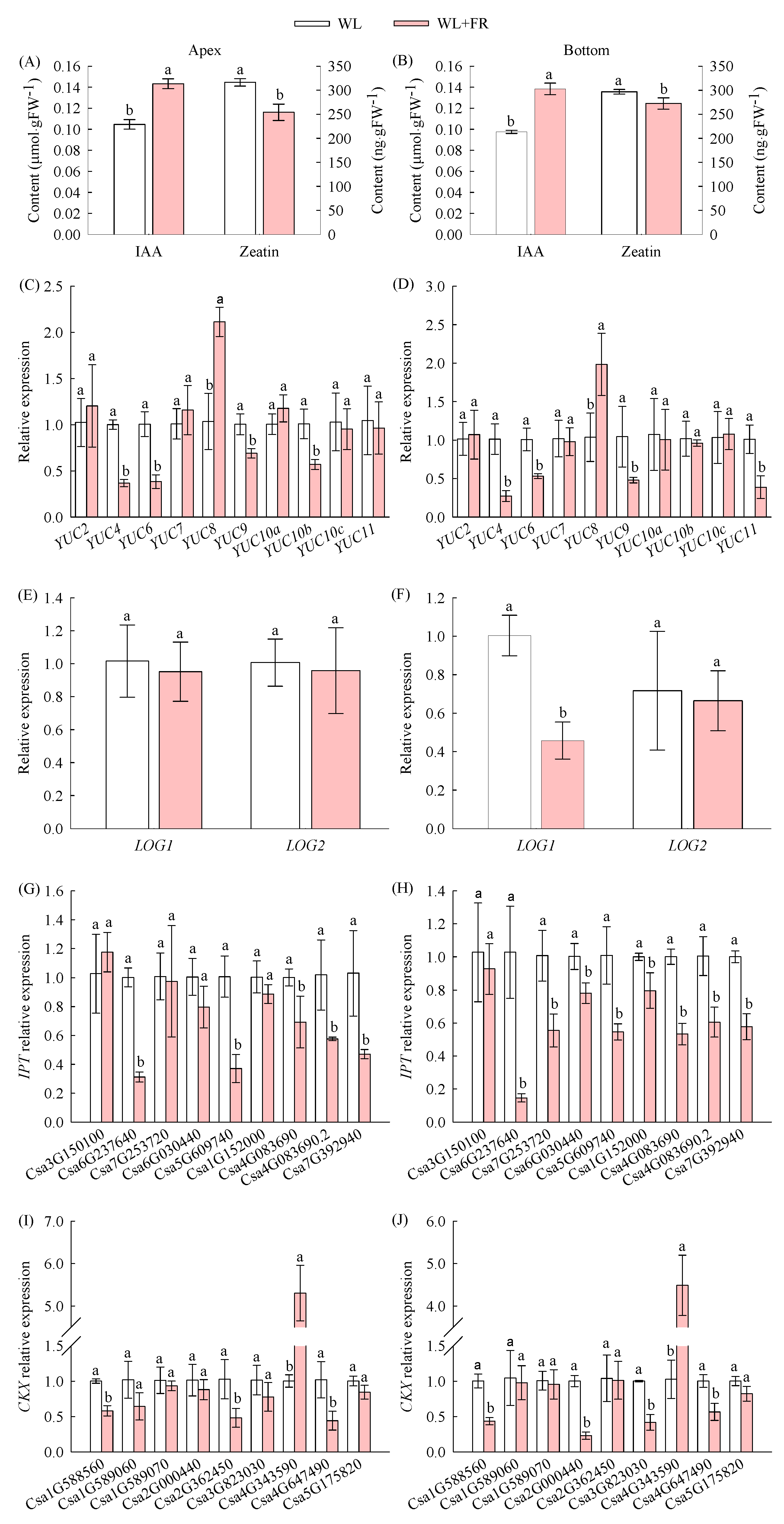
3.2. Effect of FR Light on the Endogenous Hormone Contents and Related Gene Expression in Cucumber Seedlings
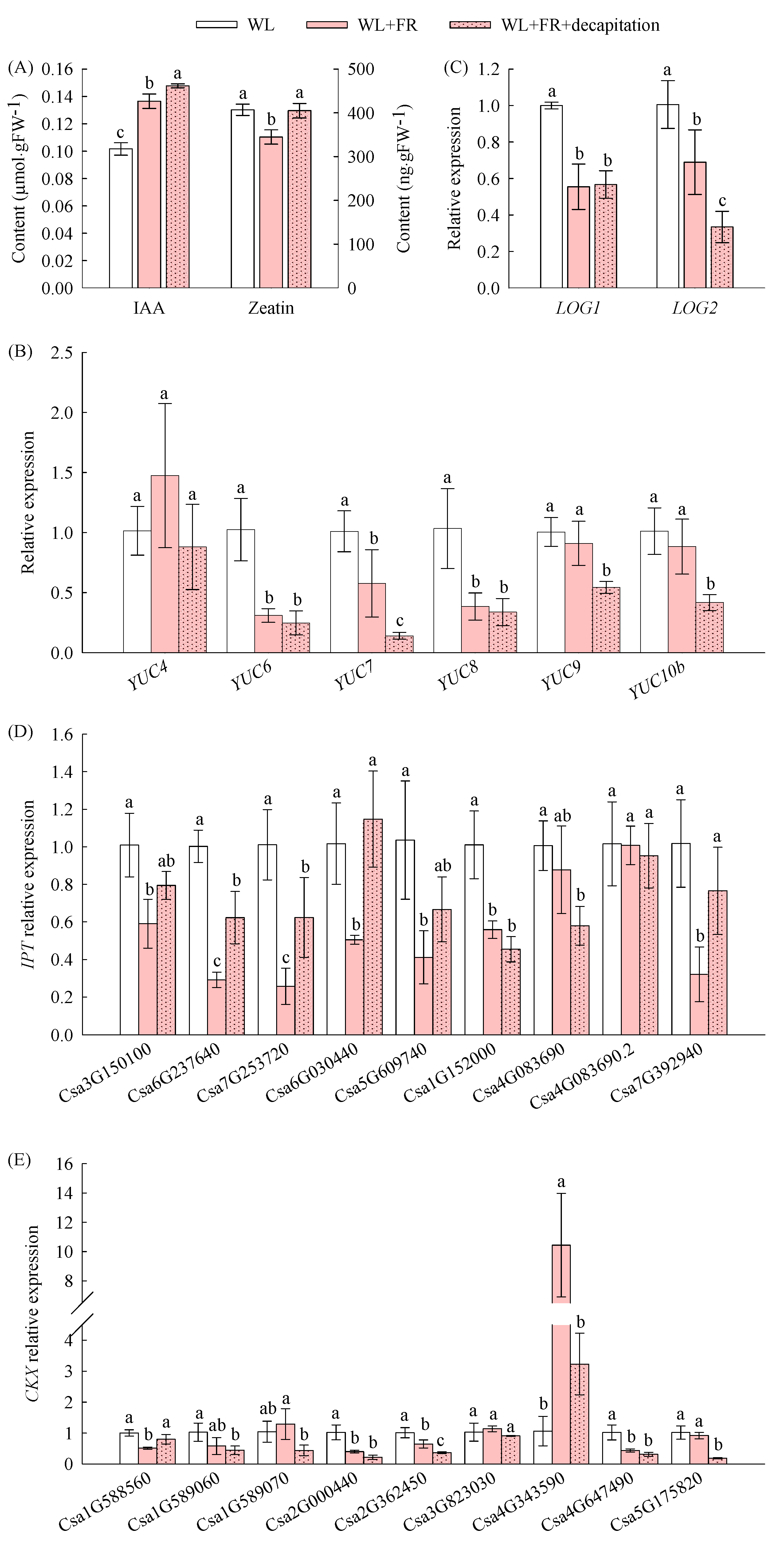
3.3. The Inhibitory Effect of FR Light on the Branch Elongation of Cucumber Seedlings Was Related to IAA
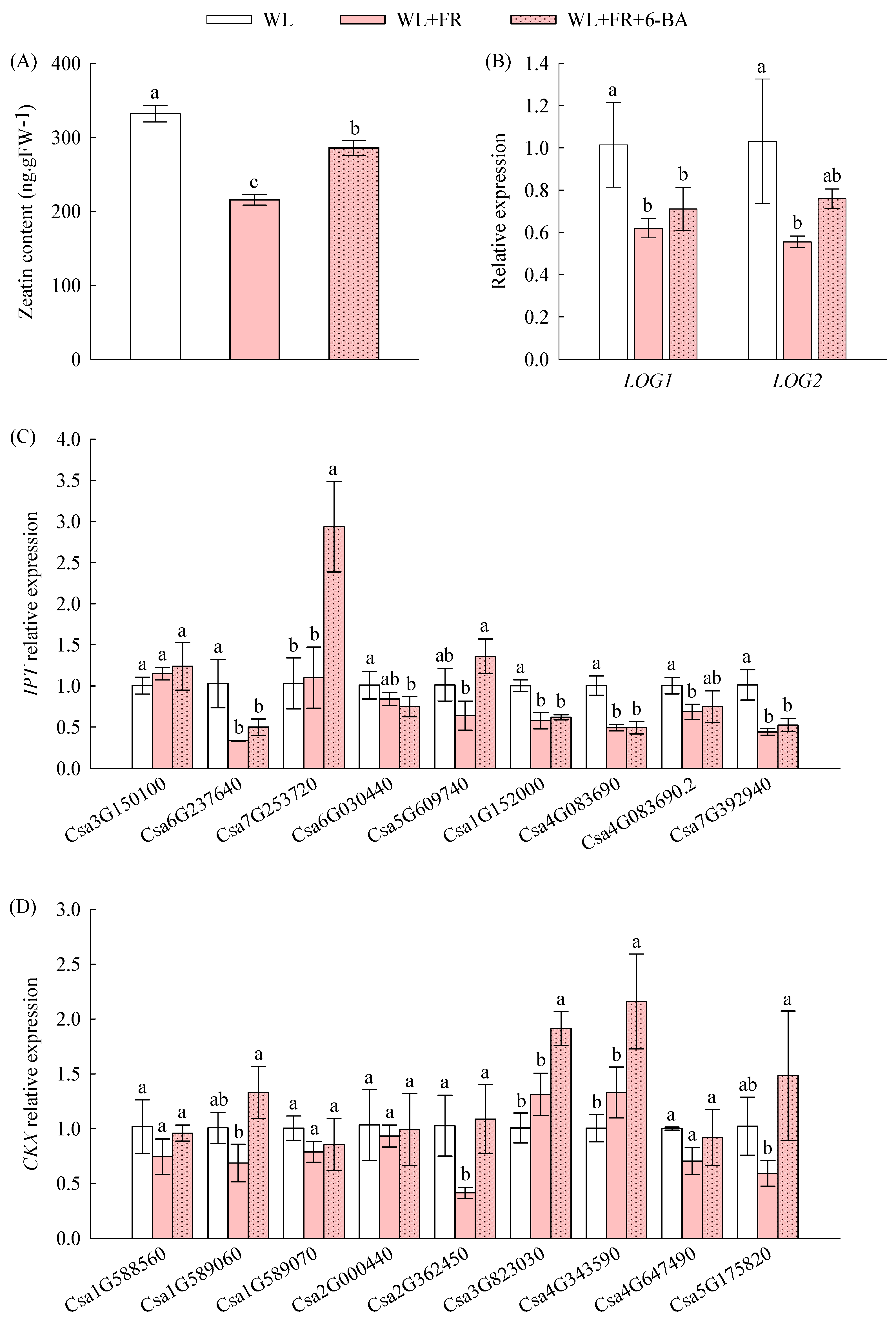
3.4. The Inhibitory Effect of FR Light on the Branch Elongation of Cucumber Seedlings Was Related to CTK
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Wang, B.; Smith, S.M.; Li, J. Genetic Regulation of Shoot Architecture. Annu. Rev. Plant Biol. 2018, 69, 437–468. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, T.; Morinaka, Y.; Ohnishi, T.; Sunohara, H.; Fujioka, S.; Ueguchi-Tanaka, M.; Mizutani, M.; Sakata, K.; Takatsuto, S.; Yoshida, S.; et al. Erect Leaves Caused by Brassinosteroid Deficiency Increase Biomass Production and Grain Yield in Rice. Nat. Biotechnol. 2006, 24, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Zhang, Y.; Ge, D.; Wang, Z.; Song, W.; Gu, R.; Che, G.; Cheng, Z.; Liu, R.; Zhang, X. CsBRC1 Inhibits Axillary Bud Outgrowth by Directly Repressing the Auxin Efflux Carrier CsPIN3 in Cucumber. Proc. Natl. Acad. Sci. USA 2019, 116, 17105–17114. [Google Scholar] [CrossRef] [PubMed]
- Mathan, J.; Bhattacharya, J.; Ranjan, A. Enhancing Crop Yield by Optimizing Plant Developmental Features. Development 2016, 143, 3283–3294. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Shi, Q.; Gong, B. S-Nitrosoglutathione Reductase-Mediated Nitric Oxide Affects Axillary Buds Outgrowth of Solanum lycopersicum L. by Regulating Auxin and Cytokinin Signaling. Plant Cell Physiol. 2021, 62, 458–471. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.; Wang, J.; Song, X.; Hu, C.; Zhu, C.; Sun, T.; Zhou, Z.; Hu, Z.; Xia, X.; Zhou, J.; et al. HY5 Functions as a Systemic Signal by Integrating BRC1-Dependent Hormone Signaling in Tomato Bud Outgrowth. Proc. Natl. Acad. Sci. USA 2023, 120, e2301879120. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Song, X.; Zheng, Q.; Liu, Y.; Yu, J.; Zhou, Y.; Xia, X. The Transcription Factor SPL13 Mediates Strigolactone Suppression of Shoot Branching by Inhibiting Cytokinin Synthesis in Solanum Lycopersicum. J. Exp. Bot. 2023, 74, 5722–5735. [Google Scholar] [CrossRef]
- Liu, X.; Chen, J.; Zhang, X. Genetic Regulation of Shoot Architecture in Cucumber. Hortic. Res. 2021, 8, 143. [Google Scholar] [CrossRef]
- Barbier, F.F.; Dun, E.A.; Kerr, S.C.; Chabikwa, T.G.; Beveridge, C.A. An Update on the Signals Controlling Shoot Branching. Trends Plant Sci. 2019, 24, 220–236. [Google Scholar] [CrossRef]
- Leduc, N.; Roman, H.; Barbier, F.; Péron, T.; Huché-Thélier, L.; Lothier, J.; Demotes-Mainard, S.; Sakr, S. Light Signaling in Bud Outgrowth and Branching in Plants. Plants 2014, 3, 223–250. [Google Scholar] [CrossRef]
- Girault, T.; Bergougnoux, V.; Combes, D.; Viemont, J.; Leduc, N. Light Controls Shoot Meristem Organogenic Activity and Leaf Primordia Growth during Bud Burst in Rosa Sp. Plant Cell Environ. 2008, 31, 1534–1544. [Google Scholar] [CrossRef]
- Fustec, J.; Beaujard, F. Effect of Photoperiod and Nitrogen Supply on Basal Shoots Development in Rhododendron Catawbiense. Biol. Plant. 2000, 43, 511–515. [Google Scholar] [CrossRef]
- Demotes-Mainard, S.; Péron, T.; Corot, A.; Bertheloot, J.; Le Gourrierec, J.; Pelleschi-Travier, S.; Crespel, L.; Morel, P.; Huché-Thélier, L.; Boumaza, R.; et al. Plant Responses to Red and Far-Red Lights, Applications in Horticulture. Environ. Exp. Bot. 2016, 121, 4–21. [Google Scholar] [CrossRef]
- Franklin, K.A.; Whitelam, G.C. Phytochromes and Shade-Avoidance Responses in Plants. Ann. Bot. 2005, 96, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.-J.; Park, S.-Y.; Oh, M.-M. Growth and Cell Division of Lettuce Plants under Various Ratios of Red to Far-Red Light-Emitting Diodes. Hortic. Environ. Biotechnol. 2015, 56, 186–194. [Google Scholar] [CrossRef]
- Lee, M.-J.; Son, K.-H.; Oh, M.-M. Increase in Biomass and Bioactive Compounds in Lettuce under Various Ratios of Red to Far-Red LED Light Supplemented with Blue LED Light. Hortic. Environ. Biotechnol. 2016, 57, 139–147. [Google Scholar] [CrossRef]
- Fernández-Milmanda, G.L.; Ballaré, C.L. Shade Avoidance: Expanding the Color and Hormone Palette. Trends Plant Sci. 2021, 26, 509–523. [Google Scholar] [CrossRef] [PubMed]
- Su, H.; Abernathy, S.D.; White, R.H.; Finlayson, S.A. Photosynthetic Photon Flux Density and Phytochrome B Interact to Regulate Branching in Arabidopsis. Plant Cell Environ. 2011, 34, 1986–1998. [Google Scholar] [CrossRef] [PubMed]
- Tucker, D.J. The Effects of Far-Red Light on Lateral Bud Outgrowth in Decapitated Tomato Plants and the Associated Changes in the Levels of Auxin and Abscisic Acid. Plant Sci. Lett. 1977, 8, 339–344. [Google Scholar] [CrossRef]
- Tucker, D.J.; Mansfield, T.A. Effects of Light Quality on Apical Dominance in Xanthium Strumarium and the Associated Changes in Endogenous Levels of Abscisic Acid and Cytokinins. Planta 1971, 102, 140–151. [Google Scholar] [CrossRef]
- Finlayson, S.A.; Krishnareddy, S.R.; Kebrom, T.H.; Casal, J.J. Phytochrome Regulation of Branching in Arabidopsis. Plant Physiol. 2010, 152, 1914–1927. [Google Scholar] [CrossRef] [PubMed]
- Leivar, P.; Monte, E. PIFs: Systems Integrators in Plant Development. Plant Cell 2014, 26, 56–78. [Google Scholar] [CrossRef] [PubMed]
- Cao, D.; Chabikwa, T.; Barbier, F.; Dun, E.A.; Fichtner, F.; Dong, L.; Kerr, S.C.; Beveridge, C.A. Auxin-Independent Effects of Apical Dominance Induce Changes in Phytohormones Correlated with Bud Outgrowth. Plant Physiol. 2023, 192, 1420–1434. [Google Scholar] [CrossRef]
- Domagalska, M.A.; Leyser, O. Signal Integration in the Control of Shoot Branching. Nat. Rev. Mol. Cell Biol. 2011, 12, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Reddy, S.K.; Holalu, S.V.; Casal, J.J.; Finlayson, S.A. Abscisic Acid Regulates Axillary Bud Outgrowth Responses to the Ratio of Red to Far-Red Light. Plant Physiol. 2013, 163, 1047–1058. [Google Scholar] [CrossRef] [PubMed]
- Yao, C.; Finlayson, S.A. Abscisic Acid Is a General Negative Regulator of Arabidopsis Axillary Bud Growth. Plant Physiol. 2015, 169, 611–626. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Ran, S.; Yuan, S.; Chang, K.; Han, M.; Zhong, F. Gibberellin-Mediated Far-Red Light-Induced Leaf Expansion in Cucumber Seedlings. Protoplasma 2024. [Google Scholar] [CrossRef] [PubMed]
- Xia, X.; Dong, H.; Yin, Y.; Song, X.; Gu, X.; Sang, K.; Zhou, J.; Shi, K.; Zhou, Y.; Foyer, C.H.; et al. Brassinosteroid Signaling Integrates Multiple Pathways to Release Apical Dominance in Tomato. Proc. Natl. Acad. Sci. USA 2021, 118, e2004384118. [Google Scholar] [CrossRef]
- He, Z.; Zhan, Y.; Zeng, F.; Zhao, X.; Wang, X. Drought Physiology and Gene Expression Characteristics of Fraxinus Interspecific Hybrids. Plant Growth Regul. 2016, 78, 179–193. [Google Scholar] [CrossRef]
- Tsago, Y.; Chen, Z.; Cao, H.; Sunusi, M.; Khan, A.U.; Shi, C.; Jin, X. Rice Gene, OsCKX2-2, Regulates Inflorescence and Grain Size by Increasing Endogenous Cytokinin Content. Plant Growth Regul. 2020, 92, 283–294. [Google Scholar] [CrossRef]
- Yan, S.; Che, G.; Ding, L.; Chen, Z.; Liu, X.; Wang, H.; Zhao, W.; Ning, K.; Zhao, J.; Tesfamichael, K.; et al. Different Cucumber CsYUC Genes Regulate Response to Abiotic Stresses and Flower Development. Sci. Rep. 2016, 6, 20760. [Google Scholar] [CrossRef] [PubMed]
- Mandal, N.K.; Kumari, K.; Kundu, A.; Arora, A.; Bhowmick, P.K.; Iquebal, M.A.; Jaiswal, S.; Behera, T.K.; Munshi, A.D.; Dey, S.S. Cross-Talk between the Cytokinin, Auxin, and Gibberellin Regulatory Networks in Determining Parthenocarpy in Cucumber. Front. Genet. 2022, 13, 957360. [Google Scholar] [CrossRef]
- Zhu, Y.; Jiang, X.; Zhang, J.; He, Y.; Zhu, X.; Zhou, X.; Gong, H.; Yin, J.; Liu, Y. Silicon Confers Cucumber Resistance to Salinity Stress through Regulation of Proline and Cytokinins. Plant Physiol. Bioch. 2020, 156, 209–220. [Google Scholar] [CrossRef] [PubMed]
- Zlotorynski, E. Blue Light Gives CRY the Blues. Nat. Rev. Mol. Cell Biol. 2016, 17, 740. [Google Scholar] [CrossRef] [PubMed]
- Holalu, S.V.; Reddy, S.K.; Finlayson, S.A. Low Red Light:Far Red Light Inhibits Branching by Promoting Auxin Signaling. J. Plant Growth Regul. 2021, 40, 2028–2036. [Google Scholar] [CrossRef]
- Song, X.; Gu, X.; Chen, S.; Qi, Z.; Yu, J.; Zhou, Y.; Xia, X. Far-red Light Inhibits Lateral Bud Growth Mainly through Enhancing Apical Dominance Independently of Strigolactone Synthesis in Tomato. Plant Cell Environ. 2023, 47, 429–441. [Google Scholar] [CrossRef] [PubMed]
- Brewer, P.B.; Dun, E.A.; Gui, R.; Mason, M.G.; Beveridge, C.A. Strigolactone Inhibition of Branching Independent of Polar Auxin Transport. Plant Physiol. 2015, 168, 1820–1829. [Google Scholar] [CrossRef]
- Drummond, R.S.M.; Janssen, B.J.; Luo, Z.; Oplaat, C.; Ledger, S.E.; Wohlers, M.W.; Snowden, K.C. Environmental Control of Branching in Petunia. Plant Physiol. 2015, 168, 735–751. [Google Scholar] [CrossRef]
- De Smet, I.; Jürgens, G. Patterning the Axis in Plants—Auxin in Control. Curr. Opin. Genet. Dev. 2007, 17, 337–343. [Google Scholar] [CrossRef]
- Barbier, F.F.; Dun, E.A.; Beveridge, C.A. Apical Dominance. Curr. Biol. 2017, 27, R864–R865. [Google Scholar] [CrossRef]
- Schneider, A.; Godin, C.; Boudon, F.; Demotes-Mainard, S.; Sakr, S.; Bertheloot, J. Light Regulation of Axillary Bud Outgrowth along Plant Axes: An Overview of the Roles of Sugars and Hormones. Front. Plant Sci. 2019, 10, 1296. [Google Scholar] [CrossRef]
- Krishna Reddy, S.; Finlayson, S.A. Phytochrome B Promotes Branching in Arabidopsis by Suppressing Auxin Signaling. Plant Physiol. 2014, 164, 1542–1550. [Google Scholar] [CrossRef] [PubMed]
- Franklin, K.A.; Lee, S.H.; Patel, D.; Kumar, S.V.; Spartz, A.K.; Gu, C.; Ye, S.; Yu, P.; Breen, G.; Cohen, J.D.; et al. PHYTOCHROME-INTERACTING FACTOR 4 (PIF4) Regulates Auxin Biosynthesis at High Temperature. Proc. Natl. Acad. Sci. USA 2011, 108, 20231–20235. [Google Scholar] [CrossRef] [PubMed]
- Hornitschek, P.; Kohnen, M.V.; Lorrain, S.; Rougemont, J.; Ljung, K.; López-Vidriero, I.; Franco-Zorrilla, J.M.; Solano, R.; Trevisan, M.; Pradervand, S.; et al. Phytochrome Interacting Factors 4 and 5 Control Seedling Growth in Changing Light Conditions by Directly Controlling Auxin Signaling. Plant J. 2012, 71, 699–711. [Google Scholar] [CrossRef] [PubMed]
- Leivar, P.; Tepperman, J.M.; Cohn, M.M.; Monte, E.; Al-Sady, B.; Erickson, E.; Quail, P.H. Dynamic Antagonism between Phytochromes and PIF Family Basic Helix-Loop-Helix Factors Induces Selective Reciprocal Responses to Light and Shade in a Rapidly Responsive Transcriptional Network in Arabidopsis. Plant Cell 2012, 24, 1398–1419. [Google Scholar] [CrossRef] [PubMed]
- Holalu, S.V.; Finlayson, S.A. The Ratio of Red Light to Far Red Light Alters Arabidopsis Axillary Bud Growth and Abscisic Acid Signalling before Stem Auxin Changes. J. Exp. Bot. 2017, 68, 943–952. [Google Scholar] [CrossRef] [PubMed]
- Dun, E.A.; De Saint Germain, A.; Rameau, C.; Beveridge, C.A. Antagonistic Action of Strigolactone and Cytokinin in Bud Outgrowth Control. Plant Physiol. 2012, 158, 487–498. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Zhou, X.; Xi, L.; Li, J.; Zhao, R.; Ma, N.; Zhao, L. Roles of DgBRC1 in Regulation of Lateral Branching in Chrysanthemum (Dendranthema × grandiflora Cv. Jinba). PLoS ONE 2013, 8, e61717. [Google Scholar] [CrossRef]
- Chen, L.; Zhao, J.; Song, J.; Jameson, P.E. Cytokinin Glucosyl Transferases, Key Regulators of Cytokinin Homeostasis, Have Potential Value for Wheat Improvement. Plant Biotechnol. J. 2021, 19, 878–896. [Google Scholar] [CrossRef]
- Kieber, J.J.; Schaller, G.E. Cytokinins. Arab. Book 2014, 12, e0168. [Google Scholar] [CrossRef]
- Galuszka, P.; Frébort, I.; Šebela, M.; Sauer, P.; Jacobsen, S.; Peč, P. Cytokinin Oxidase or Dehydrogenase? Mechanism of Cytokinin Degradation in Cereals. Eur. J. Biochem. 2001, 268, 450–461. [Google Scholar] [CrossRef]
- Shimizu-Sato, S.; Tanaka, M.; Mori, H. Auxin–Cytokinin Interactions in the Control of Shoot Branching. Plant Mol. Biol. 2009, 69, 429–435. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Takei, K.; Kojima, M.; Sakakibara, H.; Mori, H. Auxin Controls Local Cytokinin Biosynthesis in the Nodal Stem in Apical Dominance. Plant J. 2006, 45, 1028–1036. [Google Scholar] [CrossRef] [PubMed]
- He, G.; Yang, P.; Tang, Y.; Cao, Y.; Qi, X.; Xu, L.; Ming, J. Mechanism of Exogenous Cytokinins Inducing Bulbil Formation in Lilium Lancifolium in Vitro. Plant Cell Rep. 2020, 39, 861–872. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Miwa, K.; Ishikawa, K.; Yamada, H.; Aiba, H.; Mizuno, T. The Arabidopsis Sensor His-Kinase, AHK4, Can Respond to Cytokinins. Plant Cell Physiol. 2001, 42, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Salam, B.B.; Barbier, F.; Danieli, R.; Teper-Bamnolker, P.; Ziv, C.; Spíchal, L.; Aruchamy, K.; Shnaider, Y.; Leibman, D.; Shaya, F.; et al. Sucrose Promotes Stem Branching through Cytokinin. Plant Physiol. 2021, 185, 1708–1721. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Hao, Q.; Tian, F.; Li, Q.; Wang, W. Cytokinin-Regulated Sucrose Metabolism in Stay-Green Wheat Phenotype. PLoS ONE 2016, 11, e0161351. [Google Scholar] [CrossRef] [PubMed]
- Martín-Trillo, M.; Grandío, E.G.; Serra, F.; Marcel, F.; Rodríguez-Buey, M.L.; Schmitz, G.; Theres, K.; Bendahmane, A.; Dopazo, H.; Cubas, P. Role of Tomato BRANCHED1 -like Genes in the Control of Shoot Branching. Plant J. 2011, 67, 701–714. [Google Scholar] [CrossRef]
- Wang, M.; Pérez-Garcia, M.-D.; Davière, J.-M.; Barbier, F.; Ogé, L.; Gentilhomme, J.; Voisine, L.; Péron, T.; Launay-Avon, A.; Clément, G.; et al. Outgrowth of the Axillary Bud in Rose Is Controlled by Sugar Metabolism and Signalling. J. Exp. Bot. 2021, 72, 3044–3060. [Google Scholar] [CrossRef]
- Liu, W.; Peng, B.; Song, A.; Zhang, Y.; Jiang, J.; Chen, F. Sucrose-Induced Bud Outgrowth in Chrysanthemum Morifolium Involves Changes of Auxin Transport and Gene Expression. Sci. Hortic. 2022, 296, 110904. [Google Scholar] [CrossRef]


| Genes | Accession Number | Primer Sequences (Forward/Reverse) |
|---|---|---|
| YUC2 | Csa020745 | F 5′-TGCAAGGCAAAAGACTTCACG-3′ R 5′-GCTATACATTCGGCTCTTTCAAGGA-3′ |
| YUC4 | Csa006121 | F 5′-TGGCTAAAGCAGGGGTGTGA-3′ R 5′-CGTTGGCGATTTTCATAGCG-3′ |
| YUC6 | Csa021077 | F 5′-GAAGTATTTGGAGGATTACGCTG-3′ R 5′-TGTTTCCTCAGAACGACCGC -3′ |
| YUC7 | Csa000192 | F 5′-GGTGAGGCTTACCGTGGGAAAC-3′ R 5′-CTTGGCATCATGGTTACAAAGA-3′ |
| YUC8 | Csa008610 | F 5′-CATACGCCAAGCATTTTGAGAT-3′ R 5′-ATGTATTCGACCTCGTTACGGG-3′ |
| YUC9 | Csa001400 | F 5′-CCGAGTCTTCCGTTTCTGGT-3′ R 5′-GGCATGACACACTCTGCATTTTC-3′ |
| YUC10a | Csa017301 | F 5′-GTCCTTCTGGCTTGGCTACCT-3′ R 5′-TGGCTAAGTGAAGGCATAAACG-3′ |
| YUC10b | Csa008786 | F 5′-CCTTCTGGTCTTGCCACTGC-3′ R 5′-CAAAACCGATTGGGGAGG-3′ |
| YUC10c | Csa019194 | F 5′-CTATCCACCGCCGCATGTTTA-3′ R 5′-CGCCAGCTCCGATGATTTCTT-3′ |
| YUC11 | Csa016452 | F 5′-CAATGCACCGACGTATATTTCG-3′ R 5′-CCTCTCTTGCTCACCACTACTTGT-3′ |
| LOG1 | Csa6G127300 | F 5′-CAACTCGGGTCATCGAAAAG-3′ R 5′-CCATCAACCCAACACTTCCT-3′ |
| LOG2 | Csa4G646190 | F 5′-CGGAGGAGGTAGTGTTGGA-3′ R 5′-ACTGCCTTCACTTCTCCCACT-3′ |
| Genes | Accession Number | Primer Sequences (Forward/Reverse) |
|---|---|---|
| IPTs | Csa3M150100 | F 5′-CGGCGGAAAGATAAAGTGG-3′ R 5′- CAACATCAAGTCCTTCATAAACCTG-3′ |
| Csa6M237640 | F 5′-AAACGATGTCTCCCTATTCTGGC-3′ R 5′-CCGACACGAACGAGTTGAGG-3′ | |
| Csa7M253720 | F 5′-CGATGTTGCTCTGCCTGTTC-3′ R 5′-CGAACTTCTTCCACTAACCCTAAC-3′ | |
| Csa6M030440 | F 5′- GCGGGTGGATGAGATGTTG-3′ R 5′-CTTCAAATGCTCTTGCCTGC-3′ | |
| Csa5M609740 | F 5′-GGTCCAACTCCTTCATTCACG-3′ R 5′-AATGGGAAACTCAACGTCTAACC-3′ | |
| Csa1M152000 | F 5′-TTGATGGCATAATTTCTCGTGG-3′ R 5′-CAGAAGAAACGGACTAACAAGAGC-3′ | |
| Csa4M083690 | F 5′-TGTTCCGATAGTTTGTGGTGG-3′ R 5′-CTTCATTTCTCCGCAAGTCTG-3′ | |
| Csa4M083690.2 | F 5′-TTCCGATAGTTTGTGGTGGG-3′ R 5′-AACTTCAGCAGCAATGTCTGG-3′ | |
| Csa7M392940 | F 5′-AGCAAGGGAAAGAAGCAGATG-3′ R 5′-TTCGTTGGGAACTTTGTGGC-3′ | |
| CKXs | Csa1M588560 | F 5′-GGATTGAACTATTTAGAGGGTTTGC-3′ R 5′-GATGATTGTAGAGTGATGAGAAGGTG-3′ |
| Csa1M589060 | F 5′-GATCGAGTTCATACCGACGAGG-3′ R 5′-GACGCCAGAAGTGAGATTACGC-3′ | |
| Csa1M589070 | F 5′- GTCTCGTGGGTGGATTATTTGC-3′ R 5′-TGAGGACCGAACCGAAACG-3′ | |
| Csa2M000440 | F 5′-CCGTAAACCAACAACCAAACA-3′ R 5′- CGTAATCCGACGAGGCTATG-3′ | |
| Csa2M362450 | F 5′-TCTTATGACCCGAGACCCGC-3′ R 5′-CCACTCATTTCAATCACCACCC-3′ | |
| Csa3M823030 | F 5′-GGCATAGTTATCAACATGGAGTCC-3′ R 5′-AGATTCCCACTGTGAACCTGC-3′ | |
| Csa4M343590 | F 5′-CCATCAACCCTCTTCACATCAG-3′ R 5′-CCATAAACCTTTCGCTCGG-3′ | |
| Csa4M647490 | F 5′-GCGGAAGAGGAAGCAGTTG-3′ R 5′-CTTGGAACGCAGAAGAGGG-3′ | |
| Csa5M175820 | F 5′-TGCCTCAGCACAGTTCTTTCTA-3′ R 5′-CAAGCAGTGGATTTCCCTACAA-3′ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, S.; Tian, J.; Ran, S.; Zhou, Y.; Gao, H.; Zhong, F. Hormone Signals Involved in the Regulation of Cucumber Seedling Lateral Branch Elongation by Far-Red Light. Agronomy 2024, 14, 366. https://doi.org/10.3390/agronomy14020366
Li S, Tian J, Ran S, Zhou Y, Gao H, Zhong F. Hormone Signals Involved in the Regulation of Cucumber Seedling Lateral Branch Elongation by Far-Red Light. Agronomy. 2024; 14(2):366. https://doi.org/10.3390/agronomy14020366
Chicago/Turabian StyleLi, Shuhao, Jun Tian, Shengxiang Ran, Yuqi Zhou, Hongdou Gao, and Fenglin Zhong. 2024. "Hormone Signals Involved in the Regulation of Cucumber Seedling Lateral Branch Elongation by Far-Red Light" Agronomy 14, no. 2: 366. https://doi.org/10.3390/agronomy14020366
APA StyleLi, S., Tian, J., Ran, S., Zhou, Y., Gao, H., & Zhong, F. (2024). Hormone Signals Involved in the Regulation of Cucumber Seedling Lateral Branch Elongation by Far-Red Light. Agronomy, 14(2), 366. https://doi.org/10.3390/agronomy14020366





