Early-Season Mapping of Johnsongrass (Sorghum halepense), Common Cocklebur (Xanthium strumarium) and Velvetleaf (Abutilon theophrasti) in Corn Fields Using Airborne Hyperspectral Imagery
Abstract
:1. Introduction
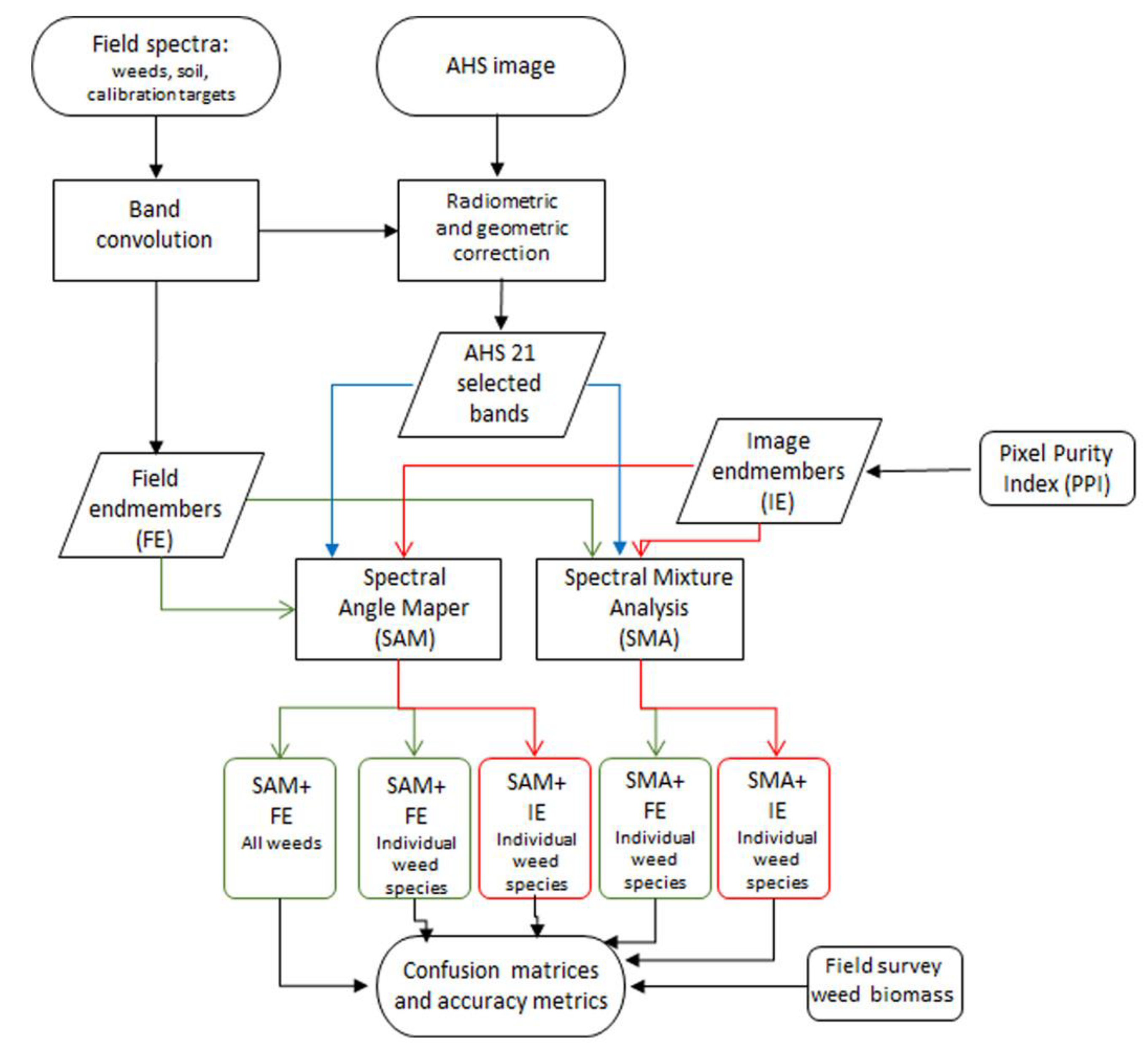
2. Materials and Methods
2.1. Study Area
2.2. Data Acquisition: Hyperspectral Images and Ancillary Ground Information
2.3. Hyperspectral Image Pre-Processing
2.4. Image Classification: Weed Maps
3. Results
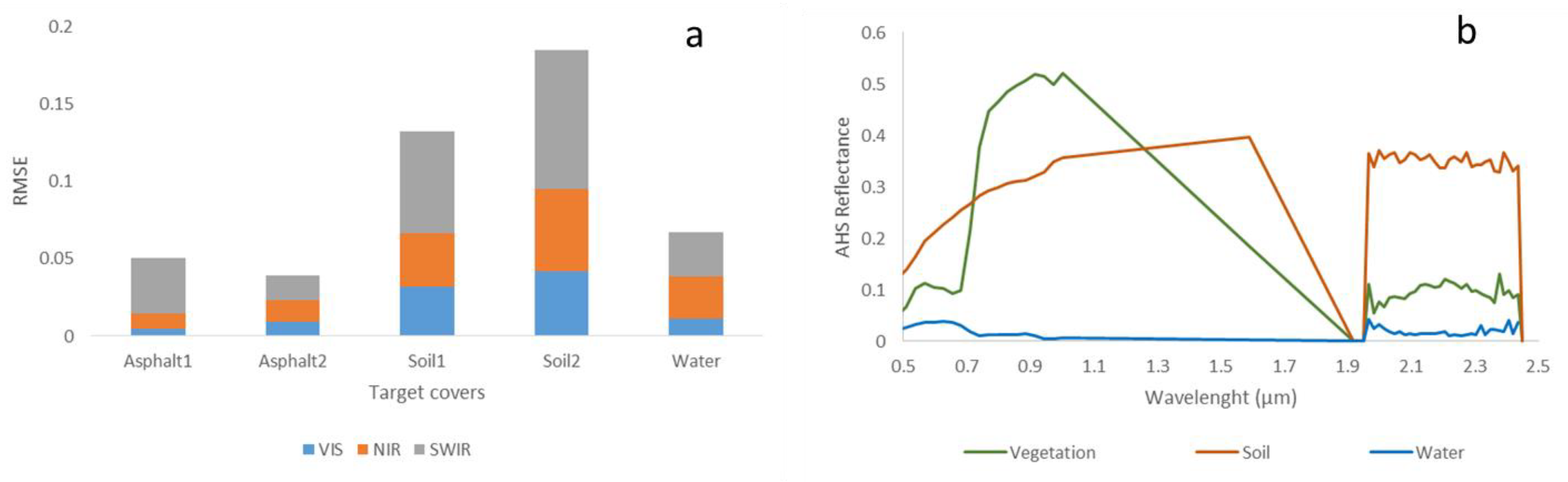
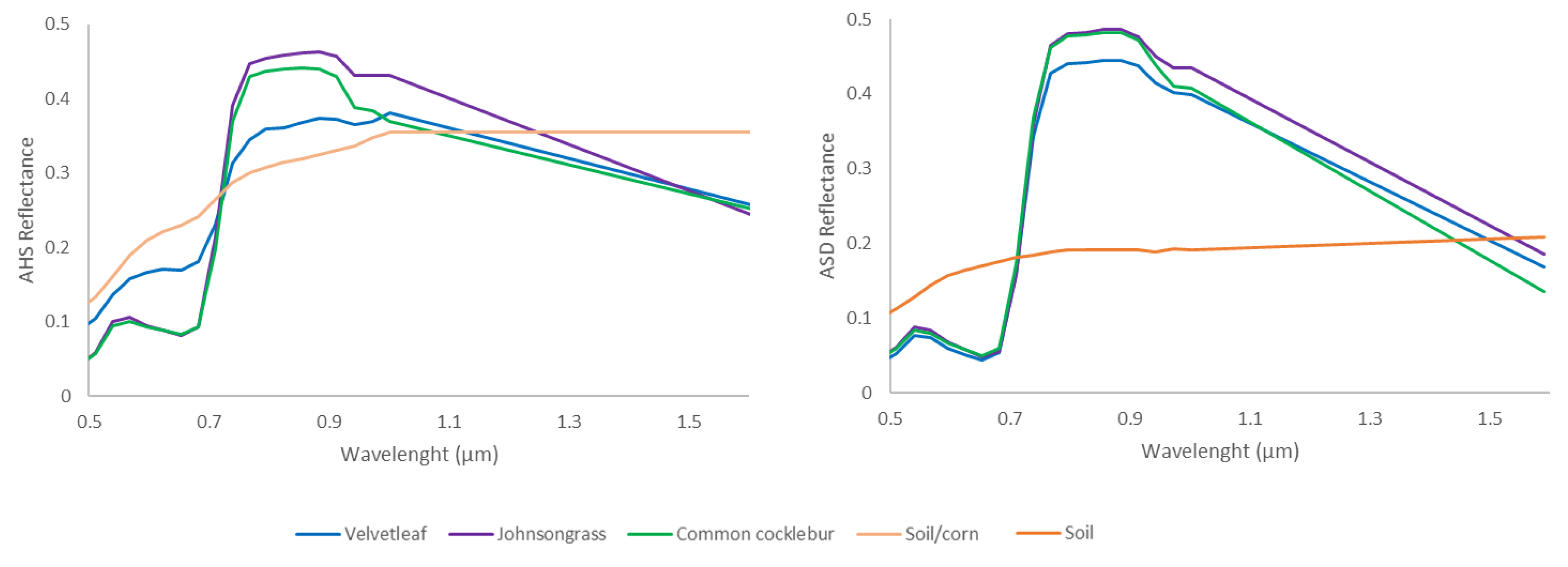
3.1. Hyperspectral Image Pre-Processing
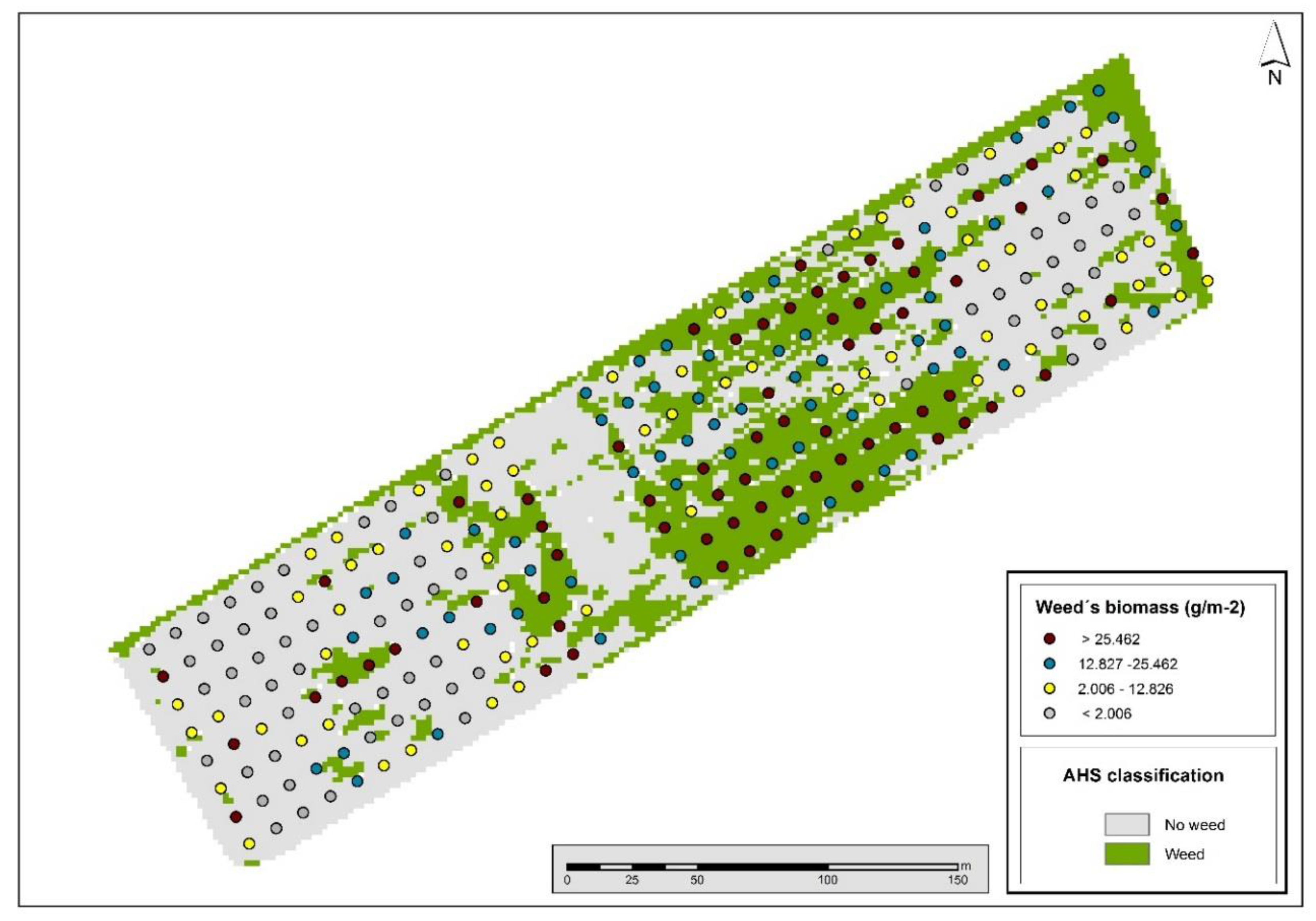
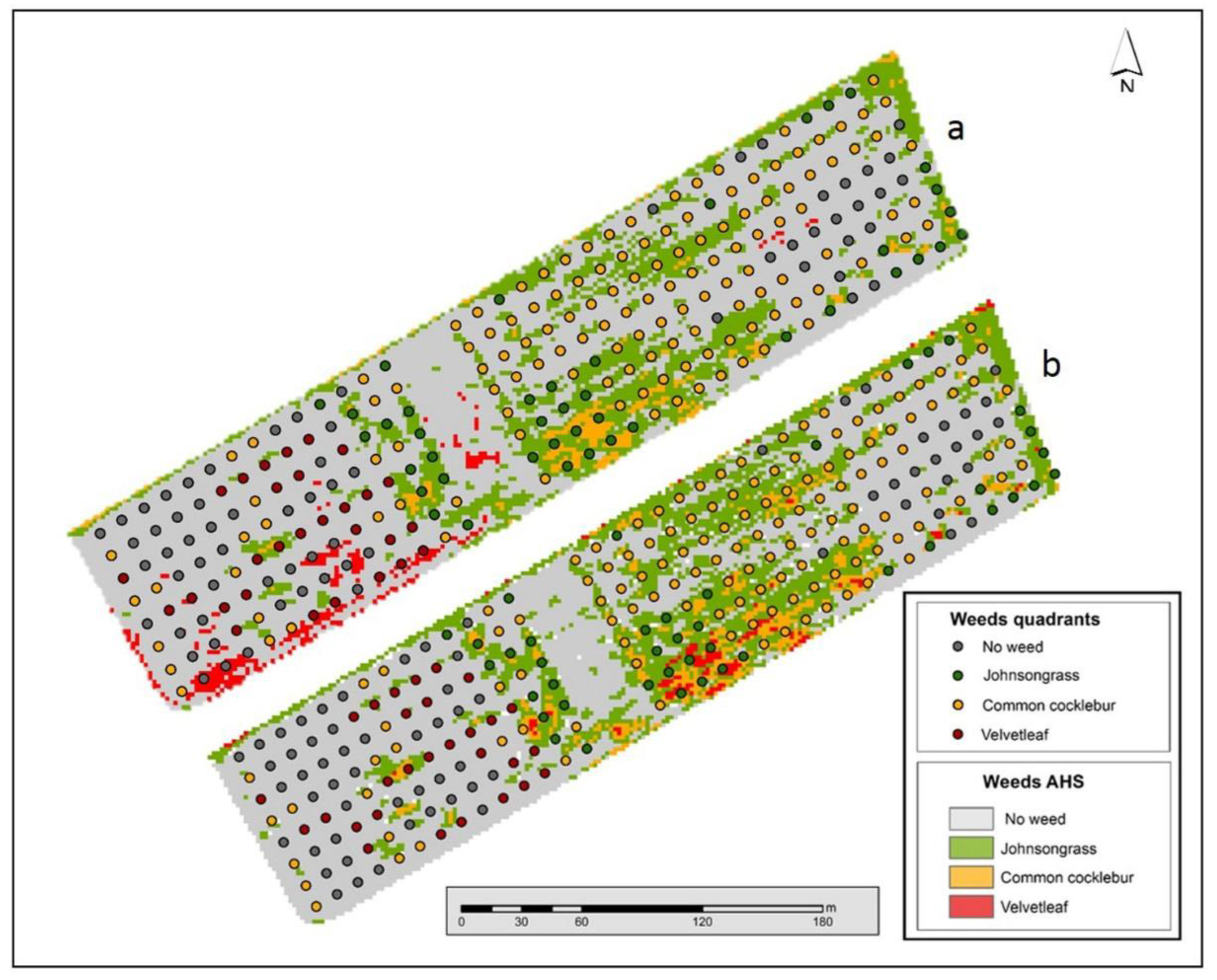
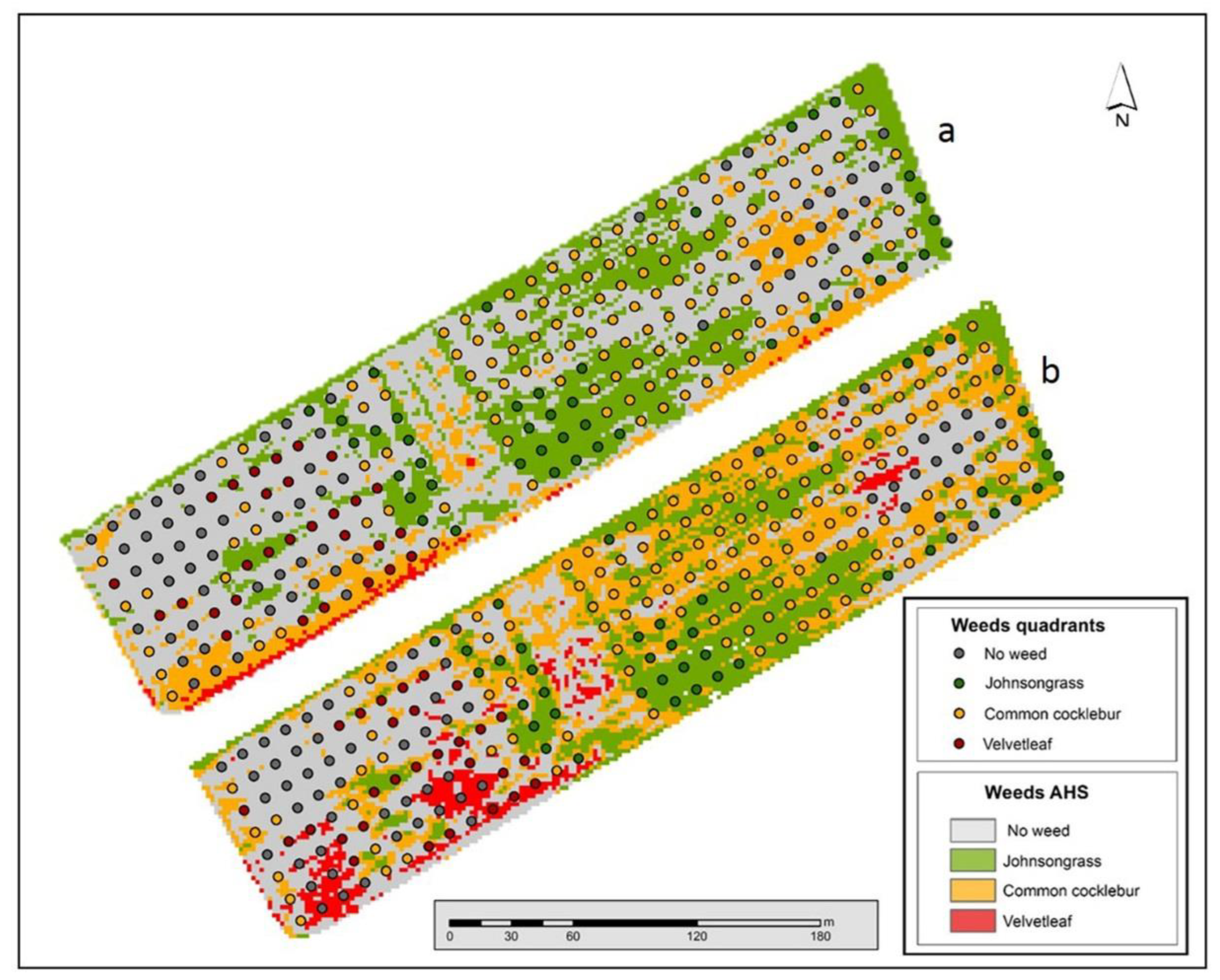
3.2. Image Classification: Weed Maps
3.2.1. Endmember Selection
3.2.2. Spectral Angle Mapper Classification
3.2.3. Spectral Mixture Analysis Classification
4. Discussion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Christensen, S.; Søgaard, H.T.; Kudsk, P.; Nørremark, M.; Lund, I.; Nadimi, E.S.; Jørgensen, R. Site-specific weed control technologies. Weed Res. 2009, 49, 233–241. [Google Scholar] [CrossRef]
- Fernández-Quintanilla, C.; Dorado, J.; Andújar, D.; Peña, J.M. Site-Specific Based Models. In Decision Support Systems for Weed Management; Chantre, G.R., González-Andújar, J.L., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 143–157. [Google Scholar]
- Fernandez-Quintanilla, C.; Dorado, J.; Andújar, D.; Peña-Barragán, J.M. Advanced detection technologies for weed scouting. In Advances in Integrated Weed Management; Kudsk, P., Ed.; Burleigh Dodds Science Publishing: Cambridge, UK, 2022; pp. 205–227. [Google Scholar]
- Fernández-Quintanilla, C.; Peña, J.M.; Andújar, D.; Dorado, J.; Ribeiro, A.; López-Granados, F. Is the current state of the art of weed monitoring suitable for site-specific weed management in arable crops? Weed Res. 2018, 58, 259–272. [Google Scholar] [CrossRef]
- Gerhards, R.; Andújar, D.; Hamouz, P.; Peteinatos, G.G.; Christensen, S.; Fernandez-Quintanilla, C. Advances in site-specific weed management in agriculture—A review. Weed Res. 2022, 62, 123–133. [Google Scholar] [CrossRef]
- Lati, R.N.; Rasmussen, J.; Andujar, D.; Dorado, J.; Berge, T.W.; Wellhausen, C.; Pflanz, M.; Nordmeyer, H.; Schirrmann, M.; Eizenberg, H.; et al. Site-specific weed management—Constraints and opportunities for the weed research community: Insights from a workshop. Weed Res. 2021, 61, 147–153. [Google Scholar] [CrossRef]
- López-Granados, F. Weed detection for site-specific weed management: Mapping and real-time approaches. Weed Res. 2011, 51, 1–11. [Google Scholar] [CrossRef]
- Andújar, D.; Calle, M.; Fernández-Quintanilla, C.; Ribeiro, A.; Dorado, J. Three-Dimensional Modeling of Weed Plants Using Low-Cost Photogrammetry. Sensors 2018, 18, 1077. [Google Scholar] [CrossRef]
- Longchamps, L.; Panneton, B.; Simard, M.-J.; Leroux, G.D. A Technique for High-Accuracy Ground-Based Continuous Weed Mapping at Field Scale. Trans. ASABE 2013, 56, 1523–1533. [Google Scholar] [CrossRef]
- Weis, M.; Sökefeld, M. Detection and Identification of Weeds. In Precision Crop Protection—The Challenge and Use of Heterogeneity; Oerke, E.-C., Gerhards, R., Menz, G., Sikora, R.A., Eds.; Springer: Dordrecht, The Netherlands, 2010; pp. 119–134. [Google Scholar]
- Martín, M.P.; Barreto, L.; Fernández-Quintanilla, C. Discrimination of sterile oat (Avena sterilis) in winter barley (Hordeum vulgare) using QuickBird satellite images. Crop Prot. 2011, 30, 1363–1369. [Google Scholar] [CrossRef]
- Zhang, Y.; Slaughter, D.C.; Staab, E.S. Robust hyperspectral vision-based classification for multi-season weed mapping. ISPRS J. Photogramm. Remote Sens. 2012, 69, 65–73. [Google Scholar] [CrossRef]
- Lamb, D.W.; Brown, R.B. PA—Precision Agriculture: Remote-Sensing and Mapping of Weeds in Crops. J. Agric. Eng. Res. 2001, 78, 117–125. [Google Scholar] [CrossRef]
- Lamb, D.W.; Weedon, M. Evaluating the accuracy of mapping weeds in fallow fields using airborne digital imaging: Pancium effusum in oilseed rape stubble. Weed Res. 1998, 38, 443–451. [Google Scholar] [CrossRef]
- Peña-Barragán, J.M.; Torres-Sánchez, J.; de Castro, A.; Kelly, M.; López-Granados, F. Weed Mapping in Early-Season Maize Fields Using Object-Based Analysis of Unmanned Aerial Vehicle (UAV) Images. PLoS ONE 2013, 8, e77151. [Google Scholar] [CrossRef]
- de Castro, A.I.; Torres-Sánchez, J.; Peña, J.M.; Jiménez-Brenes, F.M.; Csillik, O.; López-Granados, F. An Automatic Random Forest-OBIA Algorithm for Early Weed Mapping between and within Crop Rows Using UAV Imagery. Remote Sens. 2018, 10, 285. [Google Scholar] [CrossRef]
- Che’Ya, N.N.; Ernest, D.; Madan, G. Assessment of Weed Classification Using Hyperspectral Reflectance and Optimal Multispectral UAV Imagery. Agronomy 2021, 11, 1435. [Google Scholar] [CrossRef]
- de Castro, A.I.; Jurado-Expósito, M.; Gómez-Casero, M.-T.; López-Granados, F. Applying Neural Networks to Hyperspectral and Multispectral Field Data for Discrimination of Cruciferous Weeds in Winter Crops. Sci. World J. 2012, 2012, 630390. [Google Scholar] [CrossRef] [PubMed]
- Martín, M.P.; Barreto, L.; Riaño, D.; Fernandez-Quintanilla, C.; Vaughan, P. Assessing the potential of hyperspectral remote sensing for the discrimination of grassweeds in winter cereal crops. Int. J. Remote Sens. 2011, 32, 49–67. [Google Scholar] [CrossRef]
- Gao, J.; Nuyttens, D.; Lootens, P.; He, Y.; Pieters, J.G. Recognising weeds in a maize crop using a random forest machine-learning algorithm and near-infrared snapshot mosaic hyperspectral imagery. Biosyst. Eng. 2018, 170, 39–50. [Google Scholar] [CrossRef]
- Li, Y.; Al-Sarayreh, M.; Irie, K.; Hackell, D.; Bourdot, G.; Reis, M.M.; Ghamkhar, K. Identification of Weeds Based on Hyperspectral Imaging and Machine Learning. Front. Plant Sci. 2021, 11, 611622. [Google Scholar] [CrossRef]
- Okamoto, H.; Murata, T.; Kataoka, T.; Hata, S.-I. Plant classification for weed detection using hyperspectral imaging with wavelet analysis. Weed Biol. Manag. 2007, 7, 31–37. [Google Scholar] [CrossRef]
- Scherrer, B.; Sheppard, J.; Jha, P.; Shaw, J.A. Hyperspectral imaging and neural networks to classify herbicide-resistant weeds. J. Appl. Remote Sens. 2019, 13, 044516. [Google Scholar] [CrossRef]
- Andújar, D.; Barroso, J.; Fernández-Quintanilla, C.; Dorado, J. Spatial and temporal dynamics of Sorghum halepense patches in maize crops. Weed Res. 2012, 52, 411–420. [Google Scholar] [CrossRef]
- Andújar, D.; Ruiz, D.; Ribeiro, A.; Fernández-Quintanilla, C.; Dorado, J. Spatial Distribution Patterns of Johnsongrass (Sorghum halepense) in Corn Fields in Spain. Weed Sci. 2011, 59, 82–89. [Google Scholar] [CrossRef]
- Gray, C.J.; Shaw, D.R.; Gerard, P.D.; Bruce, L.M. Utility of Multispectral Imagery for Soybean and Weed Species Differentiation. Weed Technol. 2008, 22, 713–718. [Google Scholar] [CrossRef]
- Medlin, C.R.; Shaw, D.R.; Gerard, P.D.; LaMastus, F.E. Using remote sensing to detect weed infestations in Glycine max. Weed Sci. 2000, 48, 393–398. [Google Scholar] [CrossRef]
- AVIRIS. Available online: https://aviris.jpl.nasa.gov (accessed on 3 January 2023).
- APEX. Available online: https://apex-esa.org (accessed on 10 January 2023).
- Goel, P.K.; Prasher, S.O.; Landry, J.A.; Patel, R.M.; Bonnell, R.B.; Viau, A.A.; Miller, J.R. Potential of airborne hyperspectral remote sensing to detect nitrogen deficiency and weed infestation in corn. Comput. Electron. Agric. 2003, 38, 99–124. [Google Scholar] [CrossRef]
- Karimi, Y.; Prasher, S.O.; McNairn, H.; B. Bonnell, R.; Dutilleul, P.; K. Goel, P. Classification accuracy of discriminant analysis, artificial neural networks, and decision trees for weed and nitrogen stress detection in corn. Trans. ASAE 2005, 48, 1261–1268. [Google Scholar] [CrossRef]
- Yang, C.; Everitt, J.H. Mapping three invasive weeds using airborne hyperspectral imagery. Ecol. Inform. 2010, 5, 429–439. [Google Scholar] [CrossRef]
- Singh, V.; Rana, A.; Bishop, M.; Filippi, A.M.; Cope, D.; Rajan, N.; Bagavathiannan, M. Chapter Three—Unmanned aircraft systems for precision weed detection and management: Prospects and challenges. In Advances in Agronomy; Sparks, D.L., Ed.; Academic Press: Cambridge, MA, USA, 2020; Volume 159, pp. 93–134. [Google Scholar]
- Gogineni, R.; Chaturvedi, A. Hyperspectral Image Classification. In Processing and Analysis of Hyperspectral Data; Jie, C., Yingying, S., Hengchao, L., Eds.; IntechOpen: Rijeka, Croatia, 2019; Chapter 2. [Google Scholar]
- Vyas, D.; Krishnayya, N.S.R.; Manjunath, K.R.; Ray, S.S.; Panigrahy, S. Evaluation of classifiers for processing Hyperion (EO-1) data of tropical vegetation. Int. J. Appl. Earth Obs. Geoinf. 2011, 13, 228–235. [Google Scholar] [CrossRef]
- Kumar, M.N.; Seshasai, M.V.R.; Vara Prasad, K.S.; Kamala, V.; Ramana, K.V.; Dwivedi, R.S.; Roy, P.S. A new hybrid spectral similarity measure for discrimination among Vigna species. Int. J. Remote Sens. 2011, 32, 4041–4053. [Google Scholar] [CrossRef]
- South, S.; Qi, J.; Lusch, D.P. Optimal classification methods for mapping agricultural tillage practices. Remote Sens. Environ. 2004, 91, 90–97. [Google Scholar] [CrossRef]
- Plaza, A.; Benediktsson, J.A.; Boardman, J.W.; Brazile, J.; Bruzzone, L.; Camps-Valls, G.; Chanussot, J.; Fauvel, M.; Gamba, P.; Gualtieri, A.; et al. Recent advances in techniques for hyperspectral image processing. Remote Sens. Environ. 2009, 113, S110–S122. [Google Scholar] [CrossRef]
- Miao, X.; Gong, P.; Swope, S.; Pu, R.; Carruthers, R.; Anderson, G.L.; Heaton, J.S.; Tracy, C.R. Estimation of yellow starthistle abundance through CASI-2 hyperspectral imagery using linear spectral mixture models. Remote Sens. Environ. 2006, 101, 329–341. [Google Scholar] [CrossRef]
- San Martín, C.; Andújar, D.; Fernández-Quintanilla, C.; Dorado, J. Spatial Distribution Patterns of Weed Communities in Corn Fields of Central Spain. Weed Sci. 2015, 63, 936–945. [Google Scholar] [CrossRef]
- Jhala, A.J.; Knezevic, S.Z.; Ganie, Z.A.; Singh, M. Integrated weed management in maize. In Recent Advances in Weed Management; Springer: New York, NY, USA, 2014; pp. 177–196. [Google Scholar]
- Dieleman, J.A.; Mortensen, D.A. Characterizing the spatial pattern of Abutilon theophrasti seedling patches. Weed Res. 1999, 39, 455–467. [Google Scholar] [CrossRef]
- Miguel, E.d.; Jimenez, M.; Pérez, I.; Cámara, O.G.d.l.; Muñoz, F.; Gomez-Sanchez, J.A. AHS and CASI Processing for the REFLEX Remote Sensing Campaign: Methods and Results. Acta Geophys. 2015, 63, 1485–1498. [Google Scholar] [CrossRef]
- Van Cleemput, E.; Roberts, D.A.; Honnay, O.; Somers, B. A novel procedure for measuring functional traits of herbaceous species through field spectroscopy. Methods Ecol. Evol. 2019, 10, 1332–1338. [Google Scholar] [CrossRef]
- Ben-Dor, E.; Kindel, B.; Goetz, A.F.H. Quality assessment of several methods to recover surface reflectance using synthetic imaging spectroscopy data. Remote Sens. Environ. 2004, 90, 389–404. [Google Scholar] [CrossRef]
- Chi, M.; Feng, R.; Bruzzone, L. Classification of hyperspectral remote-sensing data with primal SVM for small-sized training dataset problem. Adv. Space Res. 2008, 41, 1793–1799. [Google Scholar] [CrossRef]
- Alajlan, N.; Bazi, Y.; Melgani, F.; Yager, R.R. Fusion of supervised and unsupervised learning for improved classification of hyperspectral images. Inf. Sci. 2012, 217, 39–55. [Google Scholar] [CrossRef]
- Debba, P.; van Ruitenbeek, F.J.A.; van der Meer, F.D.; Carranza, E.J.M.; Stein, A. Optimal field sampling for targeting minerals using hyperspectral data. Remote Sens. Environ. 2005, 99, 373–386. [Google Scholar] [CrossRef]
- Roberts, D.A.; Numata, I.; Holmes, K.; Batista, G.; Krug, T.; Monteiro, A.; Powell, B.; Chadwick, O.A. Large area mapping of land-cover change in Rondônia using multitemporal spectral mixture analysis and decision tree classifiers. J. Geophys. Res. Atmos. 2002, 107, LBA 40-41-LBA 40-18. [Google Scholar] [CrossRef]
- Jiménez-Muñoz, J.C.; Sobrino, J.A.; Plaza, A.; Guanter, L.; Moreno, J.; Martínez, P. Comparison Between Fractional Vegetation Cover Retrievals from Vegetation Indices and Spectral Mixture Analysis: Case Study of PROBA/CHRIS Data Over an Agricultural Area. Sensors 2009, 9, 768–793. [Google Scholar] [CrossRef] [PubMed]
- Kruse, F.A.; Lefkoff, A.B.; Boardman, J.W.; Heidebrecht, K.B.; Shapiro, A.T.; Barloon, P.J.; Goetz, A.F.H. The spectral image processing system (SIPS)—Interactive visualization and analysis of imaging spectrometer data. Remote Sens. Environ. 1993, 44, 145–163. [Google Scholar] [CrossRef]
- Shimabukuro, Y.E.; Ponzoni, F.J. The Linear Spectral Mixture Model. In Spectral Mixture for Remote Sensing: Linear Model and Applications; Springer International Publishing: Cham, Switzerland, 2019; pp. 23–41. [Google Scholar]
- Chein, I.C.; Plaza, A. A fast iterative algorithm for implementation of pixel purity index. IEEE Geosci. Remote Sens. Lett. 2006, 3, 63–67. [Google Scholar] [CrossRef]
- Andújar, D.; Ribeiro, A.; Carmona, R.; Fernández-Quintanilla, C.; Dorado, J. An assessment of the accuracy and consistency of human perception of weed cover. Weed Res. 2010, 50, 638–647. [Google Scholar] [CrossRef]
- Hudson, W.D.; Ramm, C.W. Correct formulation of the Kappa coefficient of agreement. Photogramm. Eng. Remote Sens. 1987, 53, 421–422. [Google Scholar]
- Martín, M.P.; Barreto, L.; Riaño, D.; Fernández-Quintanilla, C.; Vaughan, P.; De Santis, A. Cartografía de malas hierbas en cultivos de maíz mediante imágenes hiperespectrales aeroportadas (AHS). In Proceedings of the XIII Congreso de la Asociación Española de Teledetección Agua y Desarrollo Sostenible, Calatayud, Spain, 23–26 September 2009; pp. 41–44. [Google Scholar]
- Esposito, M.; Crimaldi, M.; Cirillo, V.; Sarghini, F.; Maggio, A. Drone and sensor technology for sustainable weed management: A review. Chem. Biol. Technol. Agric. 2021, 8, 18. [Google Scholar] [CrossRef]
- Lass, L.W.; Thill, D.C.; Shafii, B.; Timothy, S.P. Detecting Spotted Knapweed (Centaurea maculosa) with Hyperspectral Remote Sensing Technology. Weed Technol. 2002, 16, 426–432. [Google Scholar] [CrossRef]
- Lass, L.W.; Prather, T.S.; Glenn, N.F.; Weber, K.T.; Mundt, J.T.; Pettingill, J. A Review of Remote Sensing of Invasive Weeds and Example of the Early Detection of Spotted Knapweed (Centaurea maculosa) and Babysbreath (Gypsophila paniculata) with a Hyperspectral Sensor. Weed Sci. 2005, 53, 242–251. [Google Scholar] [CrossRef]
- Gibson, K.D.; Richard, D.; Case, R.M.; Loree, J. Detection of Weed Species in Soybean Using Multispectral Digital Images. Weed Technol. 2004, 18, 742–749. [Google Scholar] [CrossRef]
- Thorp, K.R.; Tian, L.F. A Review on Remote Sensing of Weeds in Agriculture. Precis. Agric. 2004, 5, 477–508. [Google Scholar] [CrossRef]
- Gibson, D.J.; Young, B.G.; Wood, A.J. Can weeds enhance profitability? Integrating ecological concepts to address crop-weed competition and yield quality. J. Ecol. 2017, 105, 900–904. [Google Scholar] [CrossRef]
- Torra, J.; Royo-Esnal, A.; Romano, Y.; Osuna, M.D.; León, R.G.; Recasens, J. Amaranthus palmeri a New Invasive Weed in Spain with Herbicide Resistant Biotypes. Agronomy 2020, 10, 993. [Google Scholar] [CrossRef]
- Karnieli, A.; Bayarjargal, Y.; Bayasgalan, M.; Mandakh, B.; Dugarjav, C.; Burgheimer, J.; Khudulmur, S.; Bazha, S.N.; Gunin, P.D. Do vegetation indices provide a reliable indication of vegetation degradation? A case study in the Mongolian pastures. Int. J. Remote Sens. 2013, 34, 6243–6262. [Google Scholar] [CrossRef]
- Matongera, T.N.; Mutanga, O.; Dube, T.; Lottering, R.T. Detection and mapping of bracken fern weeds using multispectral remotely sensed data: A review of progress and challenges. Geocarto Int. 2018, 33, 209–224. [Google Scholar] [CrossRef]

| Port 1 VIS/NIR | Port 2A SWIR | Port 2 SWIR | Port 3 MIR | Port 4 TIR | |
|---|---|---|---|---|---|
| Spectral range (nm) | 440–1020 | 1490–1650 | 1900–2600 | 3000–5500 | 8000–13,000 |
| Bandwidth (nm) | 28 | 160 | 18 | 30–40 | 400–550 |
| Number of bands | 20 | 1 | 42 | 7 | 10 |
| Weed Biomass Threshold/ Quartiles * (g m−2) | Cover | Commission Error | Omission Error | Overall Accuracy | Kappa Coefficient |
|---|---|---|---|---|---|
| Q1 = 2.006 | Weeds | 15.8 | 75.4 | 60.3 | 0.20 |
| Soil/crop | 43.7 | 4.5 | |||
| Q2 = 12.826 | Weeds | 7.9 | 49.2 | 72.6 | 0.46 |
| Soil/crop | 35 | 4.5 | |||
| Q3 = 25.462 | Weeds | 5.5 | 25 | 85.1 | 0.72 |
| Soil/crop | 21.2 | 4.5 |
| Ground Truth | |||||||
|---|---|---|---|---|---|---|---|
| AHS | Soil/Crop | Johnsongrass | Common Cocklebur | Velvetleaf | Total | Commission Error (%) | |
| Soil/Crop | 66 | 17 | 91 | 27 | 201 | 67.2 | |
| Johnsongrass | 0 | 20 | 41 | 1 | 62 | 67.7 | |
| Common Cocklebur | 0 | 5 | 0 | 0 | 5 | 100 | |
| Velvetleaf | 2 | 0 | 1 | 1 | 4 | 75 | |
| Total | 68 | 42 | 133 | 29 | 272 | ||
| Omission Error (%) | 2.9 | 52.4 | 100 | 96.5 | |||
| Ground Truth | |||||||
|---|---|---|---|---|---|---|---|
| AHS | Soil/Crop | Johnsongrass | Common Cocklebur | Velvetleaf | Total | Commission Error (%) | |
| Soil/Crop | 65 | 1 | 67 | 24 | 167 | 61 | |
| Johnsongrass | 3 | 22 | 56 | 4 | 85 | 74.1 | |
| Common Cocklebur | 0 | 5 | 8 | 1 | 14 | 42.8 | |
| Velvetleaf | 0 | 4 | 0 | 0 | 4 | 100 | |
| Total | 68 | 42 | 131 | 29 | 270 | ||
| Omission Error (%) | 4.4 | 47.6 | 93.9 | 100 | |||
| Ground Truth | |||||||
|---|---|---|---|---|---|---|---|
| AHS | Soil/Crop | Johnsongrass | Common Cocklebur | Velvetleaf | Total | Commission Error (%) | |
| Soil/Crop | 58 | 16 | 81 | 19 | 174 | 66.6 | |
| Johnsongrass | 2 | 25 | 47 | 7 | 81 | 69.1 | |
| Common Cocklebur | 8 | 1 | 5 | 3 | 17 | 7.6 | |
| Velvetleaf | 0 | 0 | 0 | 0 | 0 | - | |
| Total | 68 | 42 | 133 | 29 | 272 | ||
| Omission Error (%) | 17.7 | 40.5 | 96.2 | 100 | |||
| Ground Truth | |||||||
|---|---|---|---|---|---|---|---|
| AHS | Soil/Crop | Johnsongrass | Common Cocklebur | Velvetleaf | Total | Commission Error (%) | |
| Soil/Crop | 53 | 4 | 31 | 16 | 104 | 49 | |
| Johnsongrass | 0 | 28 | 43 | 2 | 73 | 61.6 | |
| Common Cocklebur | 10 | 10 | 59 | 10 | 89 | 33.7 | |
| Velvetleaf | 5 | 0 | 0 | 1 | 6 | 83.3 | |
| Total | 68 | 42 | 133 | 29 | 272 | ||
| Omission Error (%) | 22 | 33.3 | 55.6 | 96.5 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martín, M.P.; Ponce, B.; Echavarría, P.; Dorado, J.; Fernández-Quintanilla, C. Early-Season Mapping of Johnsongrass (Sorghum halepense), Common Cocklebur (Xanthium strumarium) and Velvetleaf (Abutilon theophrasti) in Corn Fields Using Airborne Hyperspectral Imagery. Agronomy 2023, 13, 528. https://doi.org/10.3390/agronomy13020528
Martín MP, Ponce B, Echavarría P, Dorado J, Fernández-Quintanilla C. Early-Season Mapping of Johnsongrass (Sorghum halepense), Common Cocklebur (Xanthium strumarium) and Velvetleaf (Abutilon theophrasti) in Corn Fields Using Airborne Hyperspectral Imagery. Agronomy. 2023; 13(2):528. https://doi.org/10.3390/agronomy13020528
Chicago/Turabian StyleMartín, María Pilar, Bernarda Ponce, Pilar Echavarría, José Dorado, and Cesar Fernández-Quintanilla. 2023. "Early-Season Mapping of Johnsongrass (Sorghum halepense), Common Cocklebur (Xanthium strumarium) and Velvetleaf (Abutilon theophrasti) in Corn Fields Using Airborne Hyperspectral Imagery" Agronomy 13, no. 2: 528. https://doi.org/10.3390/agronomy13020528
APA StyleMartín, M. P., Ponce, B., Echavarría, P., Dorado, J., & Fernández-Quintanilla, C. (2023). Early-Season Mapping of Johnsongrass (Sorghum halepense), Common Cocklebur (Xanthium strumarium) and Velvetleaf (Abutilon theophrasti) in Corn Fields Using Airborne Hyperspectral Imagery. Agronomy, 13(2), 528. https://doi.org/10.3390/agronomy13020528





