Variation of Bio-Morphometric Traits and Antioxidant Compounds of Brassica oleracea L. Accessions in Relation to Drought Stress
Abstract
:1. Introduction
2. Material and Methods
2.1. Plant Material and Experimental Design
2.2. Bio-Morphometric Traits
2.3. Soluble Solids Content and SPAD Measurements
2.4. Biochemical Traits
2.4.1. Determination of Photosynthetic Pigment
2.4.2. Ascorbic Acid Analysis
2.4.3. Glutathione Determination
2.4.4. Total Phenolic Content (TPC)
2.5. Statistical Analysis
3. Results
3.1. Bio Morphometric Traits
3.2. Biochemical Analysis
3.2.1. Photosynthetic Pigment
3.2.2. Total Phenolic Compound (TPC)
3.2.3. Ascorbic Acid and Glutathione
3.3. Principal Component Analysis and Heat Maps of Correlations
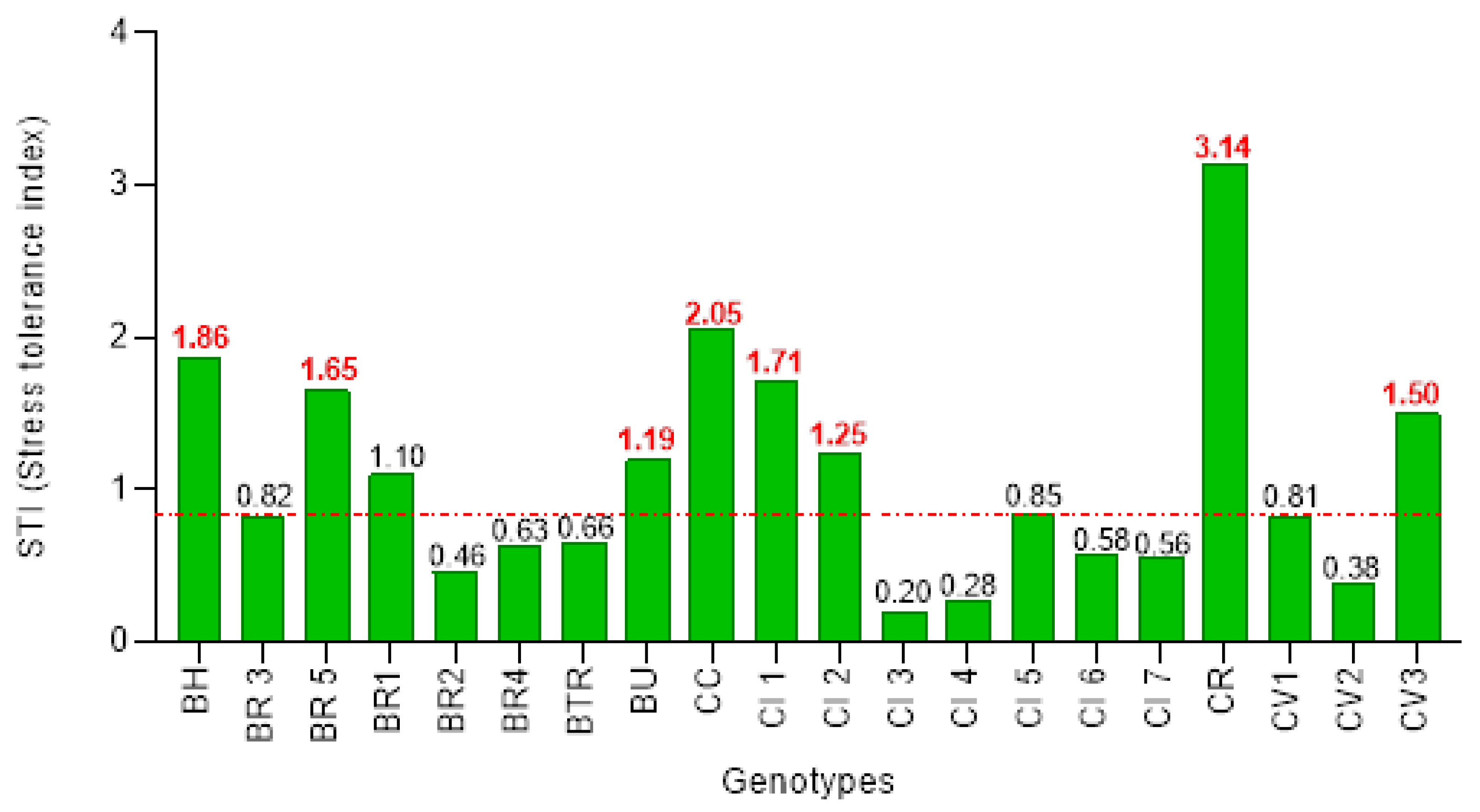
3.4. Screening of Brassica Genotypes using Stress Tolerance Index (STI)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Altieri, M.A.; Nicholls, C.I. The adaptation and mitigation potential of traditional agriculture in a changing climate. Clim. Change 2013, 140, 33–45. [Google Scholar] [CrossRef]
- Jeppesen, E.; Brucet, S.; Naselli-Flores, L.; Papastergiadou, E.; Stefanidis, K.; Nõges, T.; Nõges, P.; Attayde, J.L.; Zohary, T.; Coppens, J.; et al. Ecological impacts of global warming and water abstraction on lakes and reservoirs due to changes in water level and related changes in salinity. Hydrobiologia 2015, 750, 201–227. [Google Scholar] [CrossRef]
- Seleiman, M.F.; Al-Suhaibani, N.; Ali, N.; Akmal, M.; Alotaibi, M.; Refay, Y.; Dindaroglu, T.; Abdul-Wajid, H.H.; Battaglia, M.L. Drought Stress Impacts on Plants and Different Approaches to Alleviate Its Adverse Effects. Plants 2021, 10, 259. [Google Scholar] [CrossRef] [PubMed]
- Saud, S.; Fahad, S.; Yajun, C.; Ihsan, M.Z.; Hammad, H.M.; Nasim, W.; Amanullah, J.; Arif, M.; Alharby, H. Effects of Nitrogen Supply on Water Stress and Recovery Mechanisms in Kentucky Bluegrass Plants. Front. Plant Sci. 2017, 8, 983. [Google Scholar] [CrossRef]
- Fahad, S.; Bajwa, A.A.; Nazir, U.; Anjum, S.A.; Farooq, A.; Zohaib, A.; Sadia, S.; Nasim, W.; Adkins, S.; Saud, S.; et al. Crop Production under Drought and Heat Stress: Plant Responses and Management Options. Front. Plant Sci. 2017, 8, 1147. [Google Scholar] [CrossRef]
- Arve, L.; Torre, S.; Olsen, J.; Tanino, K. Stomatal responses to drought stress and air humidity. In Abiotic Stress in Plants—Mechanisms and Adaptations; Shanker, A., Venkateswarlu, B., Eds.; InTech: London, UK, 2011; pp. 267–280. [Google Scholar]
- Shao, H.-B.; Chu, L.-Y.; Jaleel, C.A.; Zhao, C.-X. Water-deficit stress-induced anatomical changes in higher plants. C. R. Biol. 2008, 331, 215–225. [Google Scholar] [CrossRef]
- Razi, K.; Muneer, S. Drought stress-induced physiological mechanisms, signaling pathways and molecular response of chloroplasts in common vegetable crops. Crit. Rev. Biotechnol. 2021, 41, 669–691. [Google Scholar] [CrossRef]
- Da Silva, E.C.; de Albuquerque, M.B.; Azevedo Neto, A.D.; Da Silva Junior, C.D. Drought and Its Consequences to Plants—From Individual to Ecosystem. In Responses of Organisms to Water Stress; Akıncı, S., Ed.; InTech: London, UK, 2013; pp. 18–47. [Google Scholar]
- Human, J.J.; Du Toit, D.; Bezuidenhout, H.D.; De Bruyn, L.P. The Influence of Plant Water Stress on Net Photosynthesis and Yield of Sunflower (Helianthus annuus L.). J. Agron. Crop Sci. 1990, 164, 231–241. [Google Scholar] [CrossRef]
- Mangena, P. Water Stress: Morphological and Anatomical Changes in Soybean (Glycine max L.) Plants. In Plant, Abiotic Stress and Responses to Climate Change; Andjelkovic, V., Ed.; InTech: London, UK, 2018; pp. 9–31. [Google Scholar]
- Kapoor, D.; Bhardwaj, S.; Landi, M.; Sharma, A.; Ramakrishnan, M.; Sharma, A. The Impact of Drought in Plant Metabolism: How to Exploit Tolerance Mechanisms to Increase Crop Production. Appl. Sci. 2020, 10, 5692. [Google Scholar] [CrossRef]
- Mageney, V.; Baldermann, S.; Albach, D.C. Intraspecific Variation in Carotenoids of Brassica oleracea var. sabellica. J. Agric Food Chem. 2016, 64, 3251–3257. [Google Scholar] [CrossRef]
- Georgieva, K.; Lichtenthaler, H.K. Photosynthetic response of different pea cultivars to low and high temperature treatments. Photosynthetica 2006, 44, 569–578. [Google Scholar] [CrossRef]
- Zhou, X.; Huang, W.; Kong, W.; Ye, H.; Dong, Y.; Casa, R. Assessment of leaf carotenoids content with a new carotenoid index: Development and validation on experimental and model data. Int. J. Appl. Earth Obs. Geoinf. 2017, 57, 24–35. [Google Scholar] [CrossRef]
- Sarker, U.; Oba, S. Drought stress enhances nutritional and bioactive compounds, phenolic acids and antioxidant capacity of Amaranthus leafy vegetable. BMC Plant Biol. 2018, 18, 1258. [Google Scholar] [CrossRef] [PubMed]
- Sewelam, N.; Kazan, K.; Schenk, P.M. Global Plant Stress Signaling: Reactive Oxygen Species at the Cross-Road. Front. Plant Sci. 2016, 7, 187. [Google Scholar] [CrossRef] [PubMed]
- Chaves, M.M.; Oliveira, M.M. Mechanisms underlying plant resilience to water deficits: Prospects for water-saving agriculture. J. Exp. Bot. 2004, 55, 2365–2384. [Google Scholar] [CrossRef]
- Zhang, H.; Zhao, Y.; Zhu, J.-K. Thriving under Stress: How Plants Balance Growth and the Stress Response. Dev. Cell 2020, 55, 529–543. [Google Scholar] [CrossRef]
- Ramegowda, V.; Senthil-Kumar, M. The interactive effects of simultaneous biotic and abiotic stresses on plants: Mechanistic understanding from drought and pathogen combination. J. Plant Physiol. 2015, 176, 47–54. [Google Scholar] [CrossRef]
- Xiao, M.; Zixuan, L.; Li, Z.; Jiayi, W.; Bo, Z.; Fuyu, Z.; Beiping, Z.; Haiwen, Z.; Yujie, W.; Zhijin, Z. The Multiple Roles of Ascorbate in the Abiotic Stress Response of Plants: Antioxidant, Cofactor and Regulator. Front. Plant Sci. 2021, 12, 598173. [Google Scholar] [CrossRef]
- Siddique, Z.; Jan, S.; Imadi, S.R.; Gul, A.; Ahmad, P. Drought stress and photosynthesis in plants. In Water Stress and Crop Plants—A Sustainable Approache; Parvaiz, A., Ed.; Wiley: Hoboken, NJ, USA, 2016; Volume 2, pp. 1–11. [Google Scholar]
- Fàbregas, N.; Fernie, A.R. The metabolic response to drought. J. Exp. Bot. 2019, 70, 1077–1085. [Google Scholar] [CrossRef]
- Nadarajah, K.K. ROS Homeostasis in Abiotic Stress Tolerance in Plants. Int. J. Mol. Sci. 2020, 21, 5208. [Google Scholar] [CrossRef]
- Morales, F.; Ancín, M.; Fakhet, D.; González-Torralba, J.; Gámez, A.L.; Seminario, A.; Soba, D.; Ben Mariem, S.; Garriga, M.; Aranjuelo, I. Photosynthetic Metabolism under Stressful Growth Conditions as a Bases for Crop Breeding and Yield Improvement. Plants 2020, 9, 88. [Google Scholar] [CrossRef] [PubMed]
- Perdomo, J.A.; Capó-Bauçà, S.; Carmo-Silva, E.; Galmés, J. Rubisco and Rubisco Activase Play an Important Role in the Biochemical Limitations of Photosynthesis in Rice, Wheat, and Maize under High Temperature and Water Deficit. Front. Plant Sci. 2017, 8, 490. [Google Scholar] [CrossRef] [PubMed]
- Weber, A.P.M.; Bar-Even, A. Update: Improving the Efficiency of Photosynthetic Carbon Reactions. Plant Physiol. 2019, 179, 803–812. [Google Scholar] [CrossRef] [PubMed]
- Szepesi, Á. Role of metabolites in abiotic stress tolerance. In Plant Life Under Changing Environment—Response and Management; Tripathi, D.K., Singh, V.P., Chauhan, D.K., Sharma, S., Prasad, S.M., Dubey, N.K., Ramawat, N., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 755–774. [Google Scholar]
- Das, K.; Roychoudhury, A. Reactive oxygen species (ROS) and response of antioxidants as ROS-scavengers during environmental stress in plants. Front. Environ. Sci. 2014, 2, 53. [Google Scholar] [CrossRef]
- Arbona, V.; Manzi, M.; De Ollas, C.; Gómez-Cadenas, A. Metabolomics as a Tool to Investigate Abiotic Stress Tolerance in Plants. Int. J. Mol. Sci. 2013, 14, 4885–4911. [Google Scholar] [CrossRef]
- Genga, A.; Mattana, M.; Coraggio, I.; Locatelli, F.; Piffanelli, P.; Consonni, R. Plant Metabolomics: A Characterisation of Plant Responses to Abiotic Stresses. In Abiotic Stress in Plants—Mechanisms and Adaptations; Shanker, A., Venkateswarlu, B., Eds.; InTech: London, UK, 2011; pp. 309–350. [Google Scholar]
- Fraire-Velazquez, S.; Emmanuel, V. Abiotic Stress in Plants and Metabolic Responses. In Abiotic Stress—Plant Responses and Applications in Agriculture; Vahdati, K., Leslie, C., Eds.; InTech: London, UK, 2013; pp. 25–48. [Google Scholar]
- Seminario, A.; Song, L.; Zulet, A.; Nguyen, H.T.; González, E.M.; Larrainzar, E. Drought Stress Causes a Reduction in the Biosynthesis of Ascorbic Acid in Soybean Plants. Front. Plant Sci. 2017, 8, 1042. [Google Scholar] [CrossRef]
- Bilska, K.; Wojciechowska, N.; Alipour, S.; Kalemba, E.M. Ascorbic Acid—The Little-Known Antioxidant in Woody Plants. Antioxidants 2019, 8, 645. [Google Scholar] [CrossRef]
- Sachdev, S.; Ansari, S.A.; Ansari, M.I.; Fujita, M.; Hasanuzzaman, M. Abiotic Stress and Reactive Oxygen Species: Generation, Signaling, and Defense Mechanisms. Antioxidants 2021, 10, 277. [Google Scholar] [CrossRef]
- Fujiwara, A.; Satoko, T.; Takahiro, H.; Hideyuki, M.; Chikara, M.; Tsuyoshi, I. Ascorbic acid accumulates as a defense response to Turnip Mosaic Virus in resistant Brassica rapa cultivars. J. Exp. Bot. 2016, 67, 4391–4402. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Bhuyan, M.H.M.B.; Anee, T.I.; Parvin, K.; Nahar, K.; Mahmud, J.A.; Fujita, M. Regulation of Ascorbate-Glutathione Pathway in Mitigating Oxidative Damage in Plants under Abiotic Stress. Antioxidants 2019, 8, 384. [Google Scholar] [CrossRef] [Green Version]
- Foyer, C.H.; Noctor, G. Ascorbate and Glutathione: The heart of the redox hub. Plant Physiol. 2011, 155, 2–18. [Google Scholar] [CrossRef] [PubMed]
- Jahangir, M.; Abdel-Farid, I.B.; Kim, H.K.; Choi, Y.H.; Verpoorte, R. Healthy and unhealthy plants: The effect of stress on the metabolism of Brassicaceae. Environ. Exp. Bot. 2009, 67, 23–33. [Google Scholar] [CrossRef]
- Pandey, K.B.; Rizvi, S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxid. Med. Cell. Longev. 2009, 2, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Yeshi, K.; Crayn, D.; Ritmejerytė, E.; Wangchuk, P. Plant Secondary Metabolites Produced in Response to Abiotic Stresses Has Potential Application in Pharmaceutical Product Development. Molecules 2022, 27, 313. [Google Scholar] [CrossRef]
- Branca, F.; Ragusa, L.; Tribulato, A.; Bagatta, M.; Scalzo, R.L.; Picchi, V. Evaluation of sicilian wild brassica species (n = 9) for glucosinolate profile and antioxidant compounds. Acta Hortic. 2013, 1005, 181–188. [Google Scholar] [CrossRef]
- Toscano, S.; Trivellini, A.; Cocetta, G.; Bulgari, R.; Francini, A.; Romano, D.; Ferrante, A. Effect of Preharvest Abiotic Stresses on the Accumulation of Bioactive Compounds in Horticultural Produce. Front. Plant Sci. 2019, 10, 1212. [Google Scholar] [CrossRef]
- Branca, F.; Chiarenza, G.L.; Cavallaro, C.; Gu, H.; Zhao, Z.; Tribulato, A. Diversity of Sicilian broccoli (Brassica oleracea var. italica) and cauliflower (Brassica oleracea var. botrytis) landraces and their distinctive bio-morphological, antioxidant, and genetic traits. Genet. Resour. Crop Evol. 2018, 65, 485–502. [Google Scholar] [CrossRef]
- Amec, D.; Piljac-Žegarac, J.; Bogović, M.; Habjanič, K.; Grúz, J. Antioxidant potency of white (Brassica oleracea L. var. capitata) and Chinese (Brassica rapa L. var. pekinensis (Lour.)) cabbage: The influence of development stage, cultivar choice and seed selection. Sci. Hortic. 2011, 128, 78–83. [Google Scholar]
- Cartea, M.E.; Francisco, M.; Soengas, P.; Velasco, P. Phenolic Compounds in Brassica Vegetables. Molecules 2010, 16, 251–280. [Google Scholar] [CrossRef]
- Capra, A.; Consoli, S.; Russo, A.; Scicolone, B. Integrated Agro-Economic Approach to Deficit Irrigation on Lettuce Crops in Sicily (Italy). J. Irrig. Drain. Eng. 2008, 134, 437–445. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K.; Buschmann, C. Chlorophylls and Carotenoids: Measurement and Characterization by UV-VIS Spectroscopy. Curr. Protoc. Food Anal. Chem. 2001, 1, F4.3.1–F4.3.8. [Google Scholar] [CrossRef]
- Picchi, V.; Migliori, C.; Lo Scalzo, R.; Campanelli, G.; Ferrari, V.; Di Cesare, L.F. Phytochemical content in organic and conventionally grown Italian cauliflower. Food Chem. 2012, 130, 501–509. [Google Scholar] [CrossRef]
- Wechtersbach, L.; Cigić, B. Reduction of dehydroascorbic acid at low pH. J. Biochem. Biophys. Methods 2007, 70, 767–772. [Google Scholar] [CrossRef]
- Yap, L.P.; Sancheti, H.; Ybanez, M.D.; Garcia, J.; Cadenas, E.; Han, D. Determination of GSH, GSSG and GSNO using HPLC with electrochemical detection. Methods Enzymol. 2011, 473, 137–147. [Google Scholar]
- Picchi, V.; Gobbi, S.; Fattizzo, M.; Zefelippo, M.; Faoro, F. Chitosan Nanoparticles Loaded with N-Acetyl Cysteine to Mitigate Ozone and Other Possible Oxidative Stresses in Durum Wheat. Plants 2021, 10, 691. [Google Scholar] [CrossRef]
- Di Bella, M.C.; Niklas, A.; Toscano, S.; Picchi, V.; Romano, D.; Lo Scalzo, R.; Branca, F. Morphometric Characteristics, Polyphenols and Ascorbic Acid Variation in Brassica oleracea L. Novel Foods: Sprouts, Microgreens and Baby Leaves. Agronomy 2020, 10, 782. [Google Scholar] [CrossRef]
- Pour-Aboughadareh, A.; Yousefian, M.; Moradkhani, H.; Moghaddam Vahed, M.; Poczai, P.; Siddique, K.H.M. iPASTIC: An online toolkit to estimate plant abiotic stress indices. Appl. Plant Sci. 2019, 7, e11278. [Google Scholar] [CrossRef]
- Barickman, T.C.; Ku, K.M.; Sams, C.E. Differing precision irrigation thresholds for kale (Brassica oleracea L. var. acephala) induces changes in physiological performance, metabolites, and yield. Environ. Exp. Bot. 2020, 180, 104253. [Google Scholar] [CrossRef]
- Channaoui, S.; El Kahkahi, R.; Charafi, J.; Mazouz, H.; El Fechtali, M.; Nabloussi, A. Germination and Seedling Growth of a Set of Rapeseed (Brassica napus) Varieties under Drought Stress Conditions. Int. J. Environ. Agric. Biotechnol. 2017, 2, 487–494. [Google Scholar] [CrossRef]
- EL Sabagh, A.; Hossain, A.; Barutçular, C.; Islam, M.S.; Ratnasekera, D.; Kumar, N.; Meena, R.S.; Gharib, H.S.; Saneoka, H.; da Silva, J.A.T. Drought and salinity stress management for higher and sustainable canola (Brassica napus L.) production: A critical review. Aust. J. Crop Sci. 2019, 13, 88–97. [Google Scholar] [CrossRef]
- Žnidarčič, D.; Ban, D.; Šircelj, H. Carotenoid and chlorophyll composition of commonly consumed leafy vegetables in Mediterranean countries. Food Chem. 2011, 129, 1164–1168. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-E.; Liu, W.-J.; Su, Y.-Q.; Cui, J.-M.; Zhang, Z.-W.; Yuan, M.; Zhang, H.-Y.; Yuan, S. Different response of photosystem II to short and long-term drought stress in Arabidopsis thaliana. Physiol. Plant. 2016, 158, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Farooq, M.; Wahid, A.; Kobayashi, N.; Fujita, D.; Basra, S.M.A. Plant Drought Stress: Effects. Mechanisms and Management. In Agronomy for Sustainable Development; Springer: Berlin/Heidelberg, Germany; EDP Sciences: Les Ulis, France; INRA: Paris, France, 2009; Volume 29, pp. 185–212. [Google Scholar]
- Farnheim, M.W.; Kopsell, D.A. Importance of genotype on carotenoid and chlorophyll levels in broccoli heads. HortScience 2009, 44, 1248–1253. [Google Scholar] [CrossRef]
- Ahmad, Z.; Waraich, E.A.; Akhtar, S.; Anjum, S.; Ahmad, T.; Mahboob, W.; Hafeez, O.B.A.; Tapera, T.; Labuschagne, M.; Rizwan, M. Physiological responses of wheat to drought stress and its mitigation approaches. Acta Physiol. Plant. 2018, 40, 80. [Google Scholar] [CrossRef]
- Lefsrud, M.; Kopsell, D.; Wenzel, A.; Sheehan, J. Changes in kale (Brassica oleracea L. var. acephala) carotenoid and chlorophyll pigment concentrations during leaf ontogeny. Sci. Hortic. 2007, 112, 136–141. [Google Scholar] [CrossRef]
- Wang, Z.; Li, G.; Sun, H.; Ma, L.; Guo, Y.; Zhao, Z.; Gao, H.; Mei, L. Effects of drought stress on photosynthesis and photosynthetic electron transport chain in young apple tree leaves. Biol. Open 2018, 7, bio035279. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, S.; Murata, N. How do environmental stresses accelerate photoinhibition? Trends Plant Sci. 2008, 13, 178–182. [Google Scholar] [CrossRef]
- Issarakraisila, M.; Ma, Q.; Turner, D.W. Photosynthetic and growth responses of juvenile Chinese kale (Brassica oleracea var. alboglabra) and Caisin (Brassica rapa subsp. parachinensis) to waterlogging and water deficit. Sci. Hortic. 2007, 111, 107–113. [Google Scholar] [CrossRef]
- Kalaji, H.M.; Dąbrowski, P.; Cetner, M.D.; Samborska, I.A.; Łukasik, I.; Brestic, M.; Zivcak, M.; Tomasz, H.; Mojski, J.; Kociel, H.; et al. A comparison between different chlorophyll content meters under nutrient deficiency conditions. J. Plant Nutr. 2016, 40, 1024–1034. [Google Scholar] [CrossRef]
- Jaleel, C.A.; Manivannan, P.; Wahid, A.; Farooq, M.; Somasundaram, R.; Panneerselvam, R. Drought stress in plants: A review on morphological characteristics and pigments composition. Int. J. Agric. Biol. 2009, 11, 100–105. [Google Scholar]
- Van den Berg, H.; Faulks, R.; Granado, H.F.; Hirschberg, J.; Olmedilla, B.; Sandmann, G.; Southon, S.; Stahl, W. The potential for the improvement of carotenoid levels in foods and the likely systemic effects. J. Sci. Food Agric. 2000, 80, 880–912. [Google Scholar] [CrossRef]
- Manavalan, L.P.; Guttikonda, S.K.; Phan Tran, L.S.; Nguyen, H.T. Physiological and Molecular Approaches to Improve Drought Resistance in Soybean. Plant. Cell. Physiol. 2009, 50, 1260–1276. [Google Scholar] [CrossRef] [PubMed]
- Raza, A.; Razzaq, A.; Mehmood, S.S.; Zou, X.; Zhang, X.; Lv, Y.; Xu, J. Impact of Climate Change on Crops Adaptation and Strategies to Tackle Its Outcome: A Review. Plants 2019, 8, 34. [Google Scholar] [CrossRef] [Green Version]
- Ferreres, F.; Valentão, P.; Pereira, J.A.; Bento, A.; Noites, A.; Seabra, R.M.; Andrade, P.B. HPLC-DAD-MS/MS-ESI Screening of Phenolic Compounds in Pieris brassicae L. Reared on Brassica rapa var. rapa L. J. Agric. Food Chem. 2008, 56, 844–853. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Xiao, Z.; Lin, L.-Z.; Lester, G.E.; Wang, Q.; Harnly, J.M.; Chen, P. Profiling Polyphenols in Five Brassica Species Microgreens by UHPLC-PDA-ESI/HRMSn. J. Agric. Food Chem. 2013, 61, 10960–10970. [Google Scholar] [CrossRef]
- Heimler, D.; Vignolini, P.; Dini, M.G.; Vincieri, F.F.; Romani, A. Antiradical activity and polyphenol composition of local Brassicaceae edible varieties. Food Chem. 2006, 99, 464–469. [Google Scholar] [CrossRef]
- Sousa, C.; Valentão, P.; Rangel, J.; Lopes, G.; Pereira, J.A.; Ferreres, F.; Seabra, R.M.; Andrade, P.B. Influence of Two Fertilization Regimens on the Amounts of Organic Acids and Phenolic Compounds of Tronchuda Cabbage (Brassica oleracea L. Var. costata DC). J. Agric. Food Chem. 2005, 53, 9128–9132. [Google Scholar] [CrossRef]
- Kaulmann, A.; Jonville, M.-C.; Schneider, Y.-J.; Hoffmann, L.; Bohn, T. Carotenoids, polyphenols and micronutrient profiles of Brassica oleraceae and plum varieties and their contribution to measures of total antioxidant capacity. Food Chem. 2014, 155, 240–250. [Google Scholar] [CrossRef]
- Shawon, R.A.; Kang, B.S.; Lee, S.G.; Kim, S.K.; Lee, H.J.; Katrich, E.; Gorinstein, S.; Ku, Y.G. Influence of drought stress on bioactive compounds, antioxidant enzymes and glucosinolate contents of Chinese cabbage (Brassica rapa). Food Chem. 2020, 308, 125657. [Google Scholar] [CrossRef]
- Latif, M.; Akram, N.A.; Ashraf, M. Regulation of some biochemical attributes in drought-stressed cauliflower (Brassica oleracea L.) by seed pre-treatment with ascorbic acid. J. Hortic. Sci. Biotechnol. 2016, 91, 129–137. [Google Scholar] [CrossRef]
- Castro, B.; Citterico, M.; Kimura, S.; Stevens, D.M.; Wrzaczek, M.; Coaker, G. Stress-induced reactive oxygen species compartmentalization, perception and signalling. Nat. Plants 2021, 7, 403–412. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.; Upadhyay, A.K.; Prasad, K.; Bahadur, A.; Rai, M. Variability of carotenes, vitamin C, E and phenolics in Brassica vegetables. J. Food Compos. Anal. 2007, 20, 106–112. [Google Scholar] [CrossRef]
- Sikora, E.; Cieślik, E.; Leszczyńska, T.; Filipiak-Florkiewicz, A.; Pisulewski, P.M. The antioxidant activity of selected cruciferous vegetables subjected to aquathermal processing. Food Chem. 2008, 107, 55–59. [Google Scholar] [CrossRef]
- Podse˛ dek, A.; Sosnowska, D.; Redzynia, M.; Anders, B. Antioxidant capacity and content of Brassica oleracea dietary antioxidants. Int. J. Food Sci. Technol. 2006, 41, 49–58. [Google Scholar] [CrossRef]
- Sarker, U.; Oba, S. Catalase, superoxide dismutase and ascorbate-glutathione cycle enzymes confer drought tolerance of Amaranthus tricolor. Sci. Rep. 2018, 8, 16496. [Google Scholar] [CrossRef]
- Khazaei, Z.; Esmaielpour, B.; Estaji, A. Ameliorative effects of ascorbic acid on tolerance to drought stress on pepper (Capsicum annuum L.) plants. Physiol. Mol. Biol. Plants 2020, 26, 1649–1662. [Google Scholar] [CrossRef]
- Biondi, F.; Balducci, F.; Capocasa, F.; Visciglio, M.; Mei, E.; Vagnoni, M.; Mezzetti, B.; Mazzoni, L. Environmental Conditions and Agronomical Factors Influencing the Levels of Phytochemicals in Brassica Vegetables Responsible for Nutritional and Sensorial Properties. Appl. Sci. 2021, 11, 1927. [Google Scholar] [CrossRef]
- Koh, E.; Wimalasiri, K.M.S.; Chassy, A.W.; Mitchell, A.E. Content of ascorbic acid, quercetin, kaempferol and total phenolics in commercial broccoli. J. Food Compos. Anal. 2009, 22, 637–643. [Google Scholar] [CrossRef]
- Raseetha, S.; Leong, S.Y.; Burritt, D.J.; Oey, I. Understanding the degradation of ascorbic acid and glutathione in relation to the levels of oxidative stress biomarkers in broccoli (Brassica oleracea L. italica cv. Bellstar) during storage and mechanical processing. Food Chem. 2013, 138, 1360–1369. [Google Scholar] [CrossRef]
- Picchi, V.; Scalzo, R.L.; Tava, A.; Doria, F.; Argento, S.; Toscano, S.; Treccarichi, S.; Branca, F. Phytochemical Characterization and In Vitro Antioxidant Properties of Four Brassica Wild Species from Italy. Molecules 2020, 25, 3495. [Google Scholar] [CrossRef]
- Tůmová, L.; Tarkowská, D.; Řehořová, K.; Marková, H.; Kočová, M.; Rothová, O.; Čečetka, P.; Holá, D. Drought-tolerant and drought-sensitive genotypes of maize (Zea mays L.) differ in contents of endogenous brassinosteroids and their drought-induced changes. PLoS ONE 2018, 13, e0197870. [Google Scholar] [CrossRef] [PubMed]
- Belay, G.A.; Zhang, Z.; Xu, P. Physio-Morphological and Biochemical Trait-Based Evaluation of Ethiopian and Chinese Wheat Germplasm for Drought Tolerance at the Seedling Stage. Sustainability 2021, 13, 4605. [Google Scholar] [CrossRef]
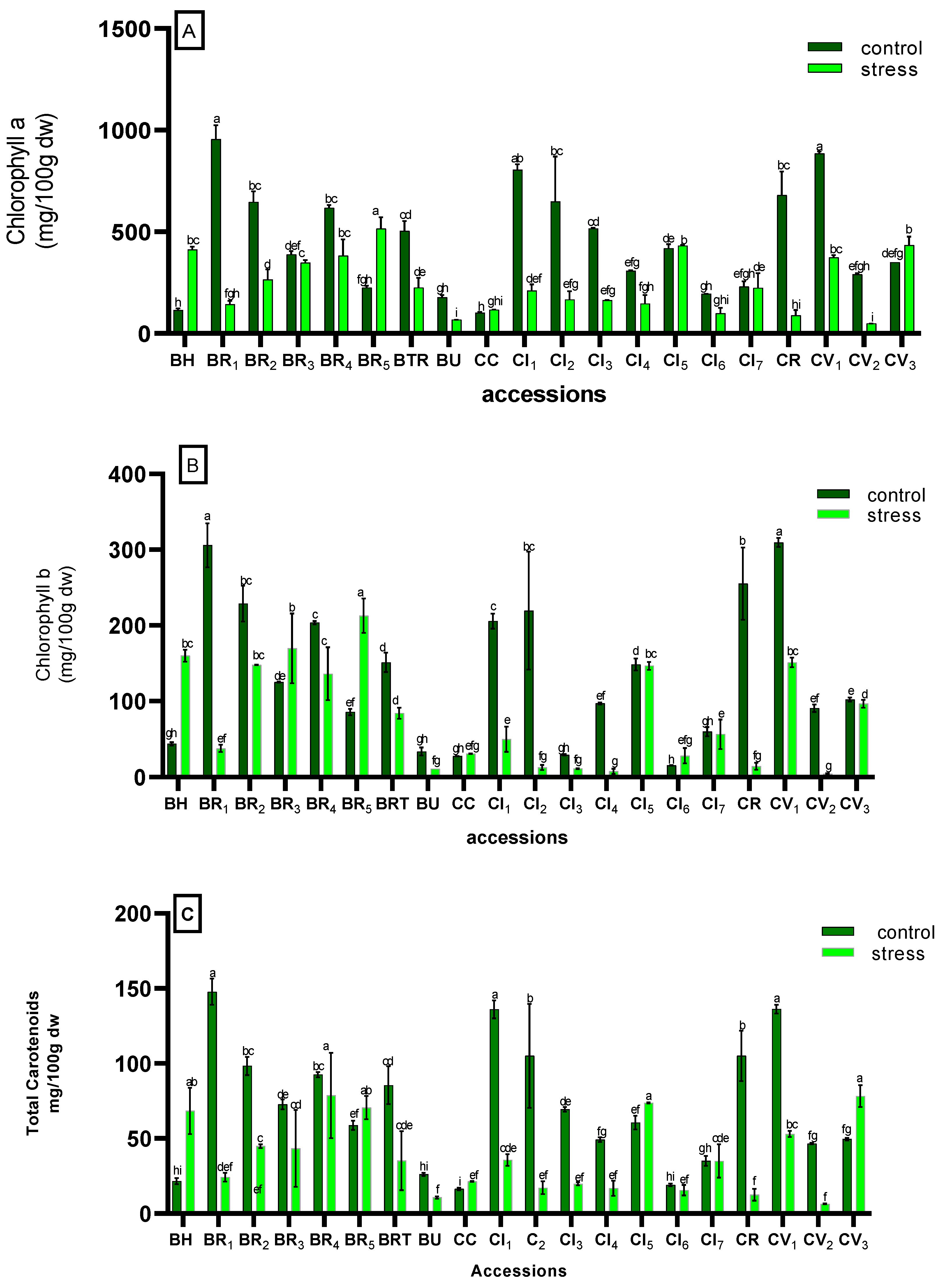

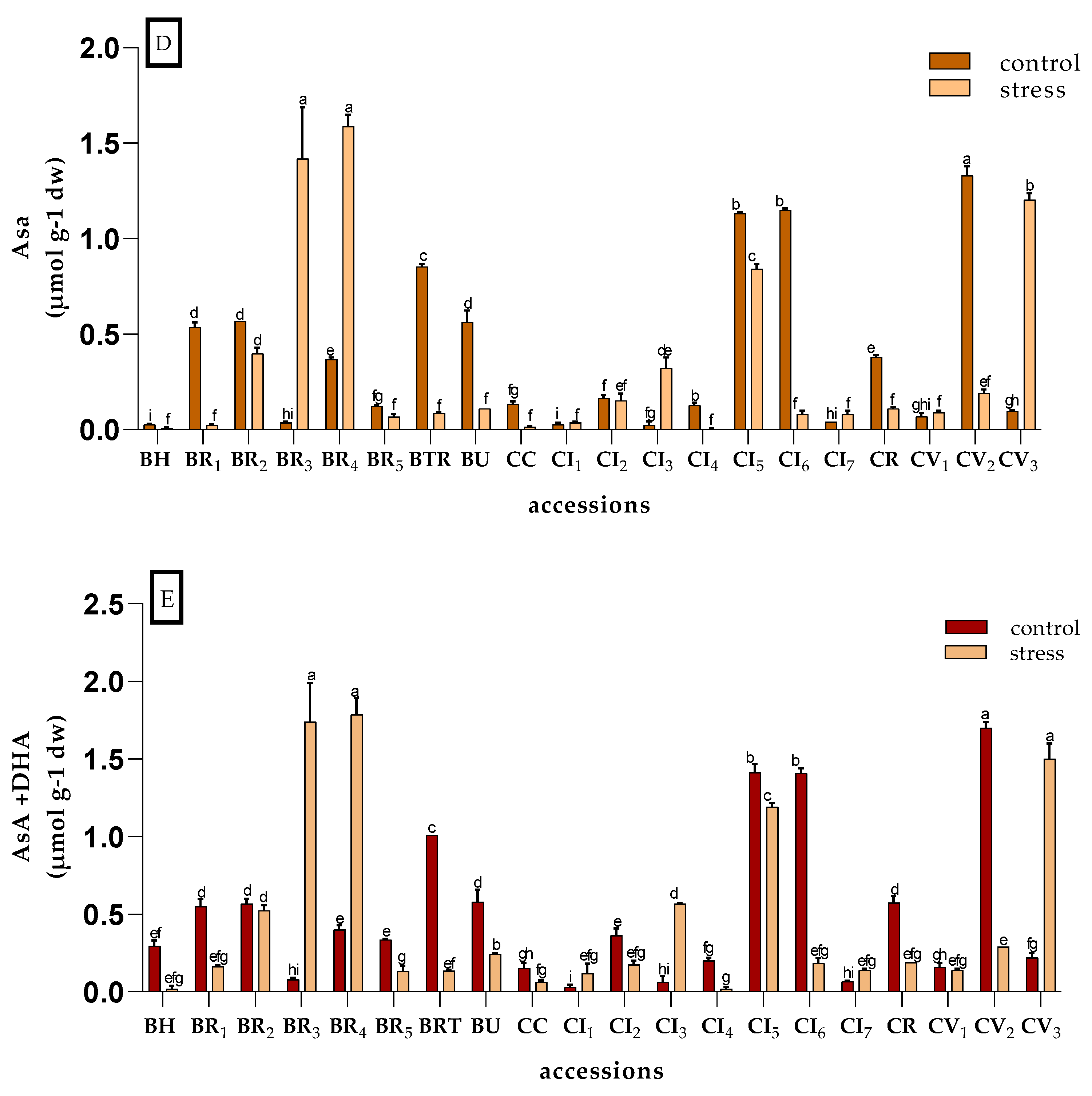


| Accessions | Conditions | PW(g) | PH (cm) | SD (cm) | N°L | FW (mg) | %DM | SPAD | SSC |
|---|---|---|---|---|---|---|---|---|---|
| MEAN ± SD | MEAN ± SD | MEAN ± SD | MEAN ± SD | MEAN ± SD | MEAN ± SD | MEAN ± SD | MEAN ± SD | ||
| BH | Control | 1367.4 ± 642.6 f–i | 114.0 ± 64.0 a | 18.8 ± 0.3cde | 17.0 ± 4.0 c–f | 85.9 ± 24.1 e | 26.5 ± 0.9 abc | 60.4 ± 18.9 abc | 5.7 ± 0.8 a–e |
| Stress | 673.0 ± 194.9 hi | 97.5 ± 12.5 bcd | 19.7 ± 1.6 c–f | 14.0 ± 2.0 i | 30.1 ± 2.5 n | 17.0 ± 3.4 c–f | 50.2 ± 7.4 c | 5.8 ± 0.4 b–g | |
| BR1 | Control | 1758 ± 33.4 e–i | 75.6 ± 13.4 abc | 26.5 ± 4.7 a–d | 23.0 ± 2.0 a–e | 60.6 ± 90.5 e | 17.7 ± 2.3 efg | 67.9 ± 0.9 abc | 6.8 ± 0.3 abc |
| Stress | 1012 ± 226.6 f–i | 66.1 ± 19.1 efg | 20.4 ± 5.4 b–f | 18.0 ± 3.0 ghi | 50.6 ± 4.8 lm | 15.1 ± 0.9 c–f | 65.2 ± 7.9 abc | 6.2 ± 1.0 a–g | |
| BR2 | Control | 2586.4 ± 176.2 c–h | 131.8 ± 0.3 a | 32.8 ± 4.1 abc | 22.0 ± 1.0 a–e | 909.5 ± 2.5 abc | 10.32 ± 4.2 g | 53.2 ± 9.9 abc | 5.8 ± 0.3 a–e |
| Stress | 990.0 ± 6.0 f–i | 78.0 ± 4.0 d–g | 18.1 ± 0.7 def | 26.0 ± 2.0 b–g | 319.6 ± 2.6 a | 10.9 ± 0.7 def | 65.0 ± 3.9 abc | 6.5 ± 1.0 a–f | |
| BR3 | Control | 2732.5 ± 187.3 b–g | 95.0 ± 10.4 abc | 34.4 ± 5.1 ab | 27.0 ± 8.5 abc | 1505.1 ± 5.0 a | 11.9 ± 1.7 d–h | 70.4 ± 10.5 abc | 7.3 ± 0.8 a |
| Stress | 1330 ± 223.9 e–i | 81.3 ± 13.4 c–f | 22.4 ± 2.4 b–e | 19.0 ± 1.0 e–i | 157.6 ± 57.9 f | 16.6 ± 4.7 c–f | 76.0 ± 13.1 ab | 8.2 ± 1.0 a | |
| BR4 | Control | 1208.1 ± 470.0 f–i | 83.0 ± 15.1 abc | 15.3 ± 1.7 de | 19.0 ± 4 b–e | 483.1 ± 4.4 b–e | 11.6 ± 0.7 cd | 75.8 ± 7.5 ab | 7.3 ± 1.3 a |
| Stress | 1421.8 ± 109.5 d–i | 59.5 ± 0.5 fg | 21.8 ± 0.1 b–e | 27 ± 3.2 b–e | 305.2 ± 1.4 b | 13.1 ± 0.0 c–f | 65.7 ± 7.9 abc | 6.2 ± 0.8 a–g | |
| BR5 | Control | 1052.4 ± 280.4 g–i | 85.0 ± 11.0 abc | 28.8 ± 16.3 a–d | 16.0 ± 2.0 ef | 197.1 ± 0.0 cde | 26.1 ± 0.7 abc | 67.4 ± 9.5 abc | 6.2 ± 0.8 a–d |
| Stress | 1603.7 ± 184.1 d–h | 112 ± 15.0 ab | 22.4 ± 1.6 b–e | 35.0 ± 3.0 a | 57.7 ± 0.2 kl | 15.3 ± 2.4 c–f | 77.6 ± 4.2 a | 7.9 ± 1.1 ab | |
| BTR | Control | 2134.3 ± 384.3 d–i | 115.0 ± 10.0 ab | 27.9 ± 0.1 a–d | 21.0 ± 1.0 a–e | 255 ± 0.0 b–e | 18.2 ± 1.9 c–f | 47.8 ± 4.6 c | 3.5 ± 0.5 e |
| Stress | 918.5 ± 1.6 f–i | 64.5 ± 1.5 fg | 29.7 ± 6.5 a–d | 14.0 ± 2.0 i | 59.6 ± 0.1 kl | 8.7 ± 0.7 f | 58.5 ± 2.4 abc | 3.0 ± 0.0 i | |
| BU | Control | 738.6 ± 4.45 hi | 70.0 ± 25.0 bc | 22.7 ± 5.2 b–e | 17.0 ± 1.0 c–f | 123.6 ± 0.0 de | 19.9 ± 3.7 cd | 52.5 ± 6.7 bc | 5.7 ± 0.8 a–e |
| Stress | 810.4 ± 400.7 g–i | 95.0 ± 5.0 bcd | 30.3 ± 5.9 abc | 17 ±2.0 hi | 150.7 ± 30.7 f | 14.5 ± 2.6 bcd | 67 ± 13.0 abc | 3.5 ± 0.5 hi | |
| CC | Control | 2970.7 ± 221.3 b–f | 90.3 ± 1.3 abc | 31.8 ± 7.9 abc | 26.0 ± 3.0 a–d | 118.3 ± 0.0 de | 32.2 ± 5.8 a | 49.2 ± 1.3 bc | 4.7 ± 0.3 cde |
| Stress | 2155 ± 192.5 c–e | 81.8 ± 0.5 c–f | 25.5 ± 0.5 a–d | 25.0 ± 3.0 b–g | 64.6 ± 0.2 jk | 15.4 ± 1.2 a | 66.2 ± 3.8 abc | 4.2 ± 0.3 ghi | |
| CI1 | Control | 5226 ± 707.4 a | 105.7 ± 12.7 ab | 22.3 ± 2.9 b–e | 21.0 ± 7.0 a–e | 302.3 ± 0.0 b–e | 20.9 ± 1.3 bcd | 50.7 ± 7.6 bc | 5.3 ± 0.6 a–e |
| Stress | 3790.2 ± 240.2 a | 108.5 ± 3.5 abc | 28.2 ± 0.1 a–d | 30.0 ± 1.0 abc | 85.45 ± 5.15 hi | 19.8 ± 1.4 bc | 51.8 ± 4.5 bc | 6.5 ± 0.5 a–f | |
| CI2 | Control | 2930.2 ± 990.0 b–g | 92.0 ± 27.9 abc | 30.6 ± 5.4a–d | 22.0 ± 1.0 a–e | 215 ± 28.0 a–d | 14.8 ± 4.0 d–h | 58.6 ± 1.9 abc | 4.8 ± 0.3 b–e |
| Stress | 2593.1 ± 874.9 bc | 93.8 ± 16.8 b–e | 24.5 ± 8.1 a–d | 24.0 ± 2.0 b–g | 77.2 ± 7.9 i | 20.34 ± 1.9 c–f | 48.2 ± 13.9 c | 7.1 ± 1.6 a–e | |
| CI3 | Control | 1928 ± 196.4 e–i | 118.5 ± 5.5 ab | 30.6 ± 1.0 a–d | 31.0 ± 3.0 a | 316.3 ± 18.3 b–e | 14.4 ± 1.8 d–h | 66.2 ± 0.7abc | 5.3 ± 0.6 a–e |
| Stress | 1660.2 ± 391.0 c–g | 94.0 ± 5.3 bcd | 21.8 ± 3.3 b–e | 28.0 ± 4.0 a–d | 40.9 ± 7.9 mn | 3.4 ± 0.8 c–f | 61.9 ± 7.5 abc | 5.3 ± 0.5 e–h | |
| CI4 | Control | 4544.3 ± 1271.9 ab | 124 ± 5.3 ab | 30.1 ± 5.4 a–d | 29.0 ± 3.0 ab | 427.8 ± 0.3 b–e | 17.7 ± 3.1 c–g | 57.5 ± 7.4 abc | 6.8 ± 1.6 abc |
| Stress | 1817.9 ± 423.6 c–f | 101.7 ± 12.6 bcd | 23.9 ± 3.2 bcd | 26.0 ± 2.0 b–f | 74.4 ± 24.4 ij | 3.8 ± 1.8 c–f | 55.2 ± 5.1 abc | 6.6 ± 0.4 a–f | |
| CI5 | Control | 3858.2 ± 52.5 a–d | 118.5 ± 8.5 ab | 27.5 ± 1.9 a–d | 23.0 ± 4.0 a–e | 393.6 ± 193.4 b–e | 9.4 ± 1.8 fgh | 80.6 ± 8.3a | 5.8 ± 0.75 a–e |
| Stress | 2197.8 ± 66.2 c–e | 99.5 ± 10.5 bcd | 24.9 ± 2.5 a–d | 31.0 ± 1.0 ab | 96 ± 0.0 h | 21.8 ± 1.9 ef | 71.5 ± 3.2 abc | 7.7 ± 0.3a–d | |
| CI6 | Control | 4463.2 ± 413.1 a–c | 118.5 ± 4.5 ab | 31.5 ± 2.8 abc | 26.0 ± 3.0 a–e | 257.3 ± 24.9 b–e | 17.1 ± 3.8 h | 71.6 ± 15.5 abc | 6.1 ± 0.5 a–d |
| Stress | 3686.3 ± 53.7 a | 130.9 ± 2.7 a | 36.2 ± 7.9 a | 27.0 ± 3.0 b–f | 135.6 ± 15.6 g | 8.3 ± 3.0 f | 58.6 ± 3.6 abc | 5.5 ± 0.5 d–h | |
| CI7 | Control | 3539 ± 1439 a–e | 127.6 ± 26.7 ab | 40.8 ± 1.0 a | 28.0 ± 2.0 ab | 324.9 ± 24.9 b–e | 11.9 ± 2.8 d–h | 67.5 ± 3.9 abc | 4.3 ± 0.8 de |
| Stress | 3498.4 ± 2.0 ab | 133.5 ± 1.5 a | 32.3 ± 3.9 ab | 22.0 ± 2.0 d–h | 169.8 ± 51.8 e | 11.4 ± 0.0 c–f | 58.7 ± 16.4 abc | 5.7 ± 0.3 c–h | |
| CR | Control | 651.6 ± 100.5 i | 36.5 ± 5.5 c | 10.43 ± 0.7 e | 7.0 ± 2.0 f | 78.15 ± 7.6 e | 29.5 ± 4.5 ab | 54.8 ± 6.1 abc | 4.0 ± 0.5 de |
| Stress | 541.0 ± 191.0 i | 28 ± 1.0 h | 8.3 ± 0.3 f | 6.0 ± 2.0 j | 74.9 ± 15.1 ij | 25.8 ± 4.2ab | 49.6 ± 8.2 c | 4.5 ± 0.0 f–i | |
| CV1 | Control | 5010.2 ± 866 a | 107.3 ± 7.5 ab | 29.23 ± 0.3 a–d | 29.0 ± 1.0 ab | 923.6 ± 16.5 ab | 16.4 ± 1.8 d–h | 58.6 ± 3.8 abc | 7.2 ± 1.3 ab |
| Stress | 2360.5 ± 266.1 cd | 94.25 ± 5.8 bcd | 18.1 ± 4.6 def | 19.0 ± 5.0 f–i | 183.45 ± 1.7 d | 12.0 ± 3.8 c–f | 59.0 ± 8.9 abc | 7.8 ± 0.3 abc | |
| CV2 | Control | 5281.1 ± 592.2 a | 93.0 ± 7.0 abc | 32.0 ± 0.9 abc | 29.0 ± 2.0 ab | 538.1 ± 11.9 b–e | 8.6 ± 2.7 gh | 54.4 ± 6.6 abc | 5.7 ± 0.8 a–e |
| Stress | 3957.1 ± 440.8 a | 92.75 ± 2.75 b–e | 28.2 ± 0.06 a–d | 23.0 ± 3.0 c–h | 231.7 ± 11.9 c | 10.7 ± 0.0 f | 54 ± 5.9 abc | 7.7 ± 0.8 a–d | |
| CV3 | Control | 775.5 ± 276.3 hi | 76.5 ± 23.5 abc | 26.1 ± 2.6 a–d | 16.0 ± 3 def | 259.1 ± 69.1 b–e | 18.8 ± 4.3 cde | 61.3 ± 16.8 abc | 7.0 ± 0.5 abc |
| Stress | 606.2 ± 157.2 i | 51.5 ± 0.5 gh | 11.3 ± 0.1 ef | 16.0 ± 1.0 hi | 51.0 ± 1.3 lm | 19.3 ± 3.0 cde | 55.7 ± 6.3 abc | 6.3 ± 0.8 a–g |
| Accessions | PW(g) | PH (cm) | SD (cm) | N°L | FW (mg) | %DM | SPAD | SSC (°Bx) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| % Var | p Value | % Var | p Value | % Var | p Value | % Var | p Value | % Var | p Value | % Var | p Value | % Var | p Value | % Var | p Value | |
| BH | 50.8 | ns | 14.5 | ns | −4.79 | ns | 17.6 | ns | 50.86 | ns | 35.80 | ** | 16.80 | ns | −2.60 | ns |
| BR1 | 42.4 | ns | 12.6 | ns | 23.3 | ns | 20.3 | ns | 91.70 | *** | 14.60 | ns | 3.90 | ns | 8.60 | ns |
| BR2 | 61.7 | *** | 40.8 | **** | 44.9 | ** | −16.7 | ns | 64.90 | *** | −5.30 | ns | 22.30 | ns | 11.40 | ns |
| BR3 | 51.3 | *** | 14.4 | ns | 35.1 | * | 29.3 | * | 89.50 | *** | 39.10 | ns | −8.10 | ns | 11.40 | ns |
| BR4 | 17.7 | ns | 28.3 | ns | −42.5 | ns | −37.9 | ns | 36.80 | ns | 13.00 | ns | 13.30 | ns | 15.90 | ns |
| BR5 | −52.3 | ns | −31.8 | * | 22.3 | ns | −54.3 | **** | 70.70 | ns | 41.30 | *** | 15.20 | ns | 28.10 | ns |
| BTR | 56.9 | ** | 43.9 | *** | −6.3 | ns | 31.7 | ns | 76.60 | ns | 51.90 | ** | 22.40 | ns | 14.30 | ns |
| BU | −9.7 | ns | −35.7 | ns | −33.4 | ns | 0 | ns | −21.93 | ns | 27.14 | ns | 27.62 | ns | 38.60 | * |
| CC | 27.5 | * | 9.4 | ns | 19.6 | ns | 6.3 | ns | 45.60 | ns | 52.00 | *** | 34.60 | ns | 10.00 | ns |
| CI1 | 27.5 | *** | −2.6 | ns | −26.5 | ns | −42.9 | ** | 71.30 | * | 5.10 | ns | −2.20 | ns | 21.90 | ns |
| CI2 | 11.5 | ns | −2 | ns | 19.8 | ns | −10.6 | ns | 64.20 | ns | 37.10 | ns | 17.80 | ns | 46.20 | ** |
| CI3 | 13.9 | ns | 20.7 | ns | 28.6 | ns | 9.7 | ns | 87.10 | ** | 76.50 | *** | 6.40 | ns | 0.00 | ns |
| CI4 | 60 | *** | 18 | ns | 20.6 | ns | 10.2 | ns | 82.60 | *** | 78.60 | *** | 3.90 | ns | 3.90 | ns |
| CI5 | 43 | *** | 16 | ns | 9.2 | ns | −36.2 | * | 82.50 | *** | 56.80 | *** | 11.30 | ns | 33.30 | * |
| CI6 | 17.4 | ns | −10.5 | ns | −14.8 | ns | −1.9 | ns | 47.30 | *** | 51.60 | ** | 18.20 | ns | 9.80 | ns |
| CI7 | 1.1 | ns | −4.7 | ns | 21 | ns | 21.2 | ns | 47.70 | *** | 4.90 | ns | 13.00 | ns | 30.80 | ns |
| CR | 17 | ns | 23.3 | ns | 20.6 | ns | 21.7 | ns | 4.20 | ns | 12.60 | ns | 9.50 | ns | 12.50 | ns |
| CV1 | 52.9 | *** | 12.2 | ns | 38.2 | ns | 34.5 | ** | 80.10 | *** | 26.40 | ns | −0.60 | ns | −8.10 | ns |
| CV2 | 25.1 | ** | 0.3 | ns | 11.9 | ns | 20.7 | ns | 57.00 | *** | 24.10 | ns | 0.80 | ns | 35.30 | * |
| CV3 | 21.8 | ns | 32.7 | ns | 56.7 | ** | 2.1 | ns | 80.30 | ns | −2.60 | ns | 9.20 | ns | 9.50 | ns |
| Traits | G | IR | G X IR | Control | Stress | RC (%) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Min | Max | MEAN ± SD | CV (%) | Min | Max | MEAN ± SD | CV (%) | |||||
| PW | <0.0001 *** | <0.0001 *** | <0.0001 *** | 651.60 | 5281.00 | 2738 ± 1578 | 59.58 | 540.80 | 3957.00 | 1881 ± 1121 | 60.10 | 31.30 |
| PH | 0.001 ** | <0.0001 *** | 0.0003 *** | 36.50 | 131.80 | 98.89± 23.7 | 23.97 | 28.00 | 133.50 | 88.21 ± 25.6 | 29.03 | 10.80 |
| SD | <0.0001 *** | <0.0001 *** | <0.0001 *** | 10.40 | 40.80 | 27.5 ± 6,92 | 25.20 | 8.28 | 36.20 | 23.4 ± 6.67 | 28.50 | 14.91 |
| N°L | <0.0001 *** | 0.72 | <0.0001 *** | 7.67 | 31.00 | 22.6 ± 5.93 | 26.30 | 6.00 | 35.00 | 22.4 ± 6.94 | 31.00 | 0.88 |
| DM | <0.0001 *** | <0.0001 *** | 0.72 | 8.25 | 32.20 | 15.6 ± 6.53 | 42.00 | 3.40 | 26.50 | 16.3 ± 6.44 | 39.50 | −4.49 |
| SPAD | <0.0001 *** | 0.74 | 0.05 | 47.80 | 80.60 | 61.3 ± 9.22 | 15.00 | 48.20 | 77.60 | 60.8 ± 8.46 | 13.90 | 0.82 |
| SSC | <0.0001 *** | 0.016 * | <0.0001 *** | 3.50 | 7.33 | 5.76 ± 1.11 | 19.30 | 3.00 | 8.17 | 6.1 ± 1.46 | 23.90 | −5.90 |
| Chla | <0.0001 *** | <0.0001 *** | <0.0001 *** | 101.50 | 955.50 | 453.3 ± 258.6 | 57.06 | 48.91 | 515.30 | 243.4 ± 142.5 | 58.53 | 46.30 |
| Chlb | <0.0001 *** | <0.0001 *** | <0.0001 *** | 15.60 | 310.00 | 137 ± 94.1 | 68.70 | 4.44 | 213.00 | 78.6 ± 67.7 | 86.20 | 42.63 |
| Tchl | <0.0001 *** | <0.0001 *** | <0.0001 *** | 129.00 | 1261.00 | 590 ± 347 | 58.80 | 53.40 | 728.00 | 322 ± 206 | 64.00 | 45.42 |
| Caro | <0.0001 *** | 0.0122 * | <0.0001 *** | 12.80 | 148.00 | 66.7± 40.4 | 60.60 | 6.49 | 439.00 | 56± 93.7 | 167.00 | 16.04 |
| Tchl/caro | <0.0001 *** | 0.31 | 0.0036 ** | 1.50 | 11.10 | 7.34 ± 2.44 | 33.30 | 0.13 | 17.10 | 7.78 ± 3.58 | 46.00 | −5.99 |
| TPC | <0.0001 *** | <0.0001 *** | <0.0001 *** | 177.00 | 710.00 | 403 ± 152 | 37.80 | 259.00 | 1594.00 | 656 ± 310 | 47.30 | −62.78 |
| GSH | <0.0001 *** | 0.88 | <0.0001 *** | 0.02 | 0.75 | 0.207 ± 0.25 | 121.00 | 0.02 | 1.53 | 0.205 ± 0.327 | 160.00 | 0.97 |
| GSSG | <0.0001 *** | 0.0403 * | <0.0001 *** | 0.32 | 1.56 | 0.691 ± 0.323 | 46.70 | 0.33 | 1.26 | 0.72 ± 0.271 | 37.70 | −4.20 |
| AsA | <0.0001 *** | <0.0001 *** | <0.0001 *** | 0.02 | 1.33 | 0.388 ± 0.423 | 109.00 | 0.00 | 1.59 | 0.342 ± 0.5 | 146.00 | 11.86 |
| Tot AsA | <0.0001 *** | <0.0001 *** | <0.0001 *** | 0.03 | 1.70 | 0.509 ± 0.495 | 97.30 | 0.02 | 1.79 | 0.467 ± 0.585 | 125.00 | 8.25 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ben Ammar, H.; Picchi, V.; Arena, D.; Treccarichi, S.; Bianchi, G.; Lo Scalzo, R.; Marghali, S.; Branca, F. Variation of Bio-Morphometric Traits and Antioxidant Compounds of Brassica oleracea L. Accessions in Relation to Drought Stress. Agronomy 2022, 12, 2016. https://doi.org/10.3390/agronomy12092016
Ben Ammar H, Picchi V, Arena D, Treccarichi S, Bianchi G, Lo Scalzo R, Marghali S, Branca F. Variation of Bio-Morphometric Traits and Antioxidant Compounds of Brassica oleracea L. Accessions in Relation to Drought Stress. Agronomy. 2022; 12(9):2016. https://doi.org/10.3390/agronomy12092016
Chicago/Turabian StyleBen Ammar, Hajer, Valentina Picchi, Donata Arena, Simone Treccarichi, Giulia Bianchi, Roberto Lo Scalzo, Sonia Marghali, and Ferdinando Branca. 2022. "Variation of Bio-Morphometric Traits and Antioxidant Compounds of Brassica oleracea L. Accessions in Relation to Drought Stress" Agronomy 12, no. 9: 2016. https://doi.org/10.3390/agronomy12092016
APA StyleBen Ammar, H., Picchi, V., Arena, D., Treccarichi, S., Bianchi, G., Lo Scalzo, R., Marghali, S., & Branca, F. (2022). Variation of Bio-Morphometric Traits and Antioxidant Compounds of Brassica oleracea L. Accessions in Relation to Drought Stress. Agronomy, 12(9), 2016. https://doi.org/10.3390/agronomy12092016










