Assessment of Injury by Four Major Pests in Soybean Plants Using Hyperspectral Proximal Imaging
Abstract
1. Introduction
2. Material and Methods
2.1. Insects
2.2. Bioassay
2.3. Data Collection
2.4. Data Analysis
3. Results
3.1. Defoliation
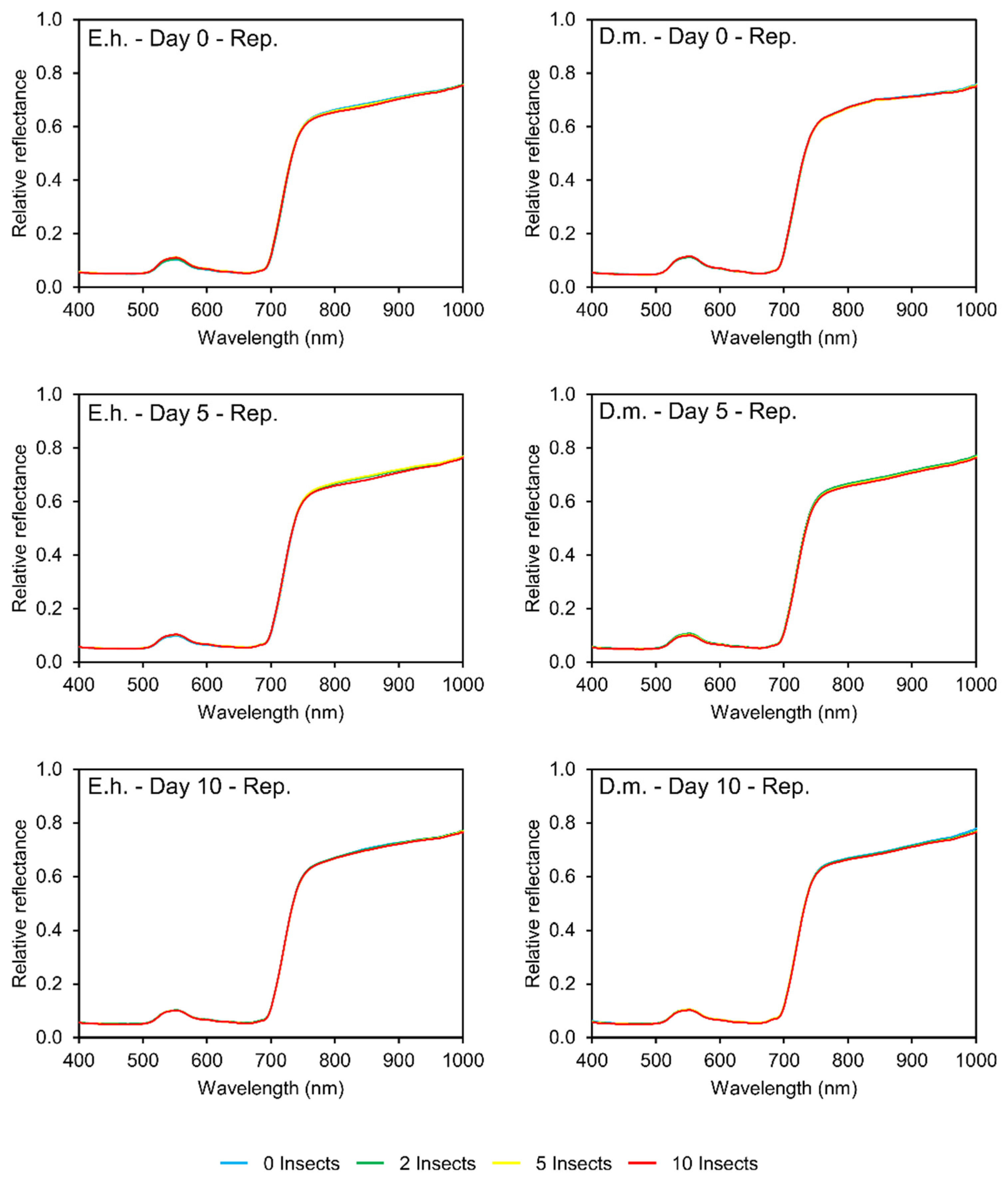
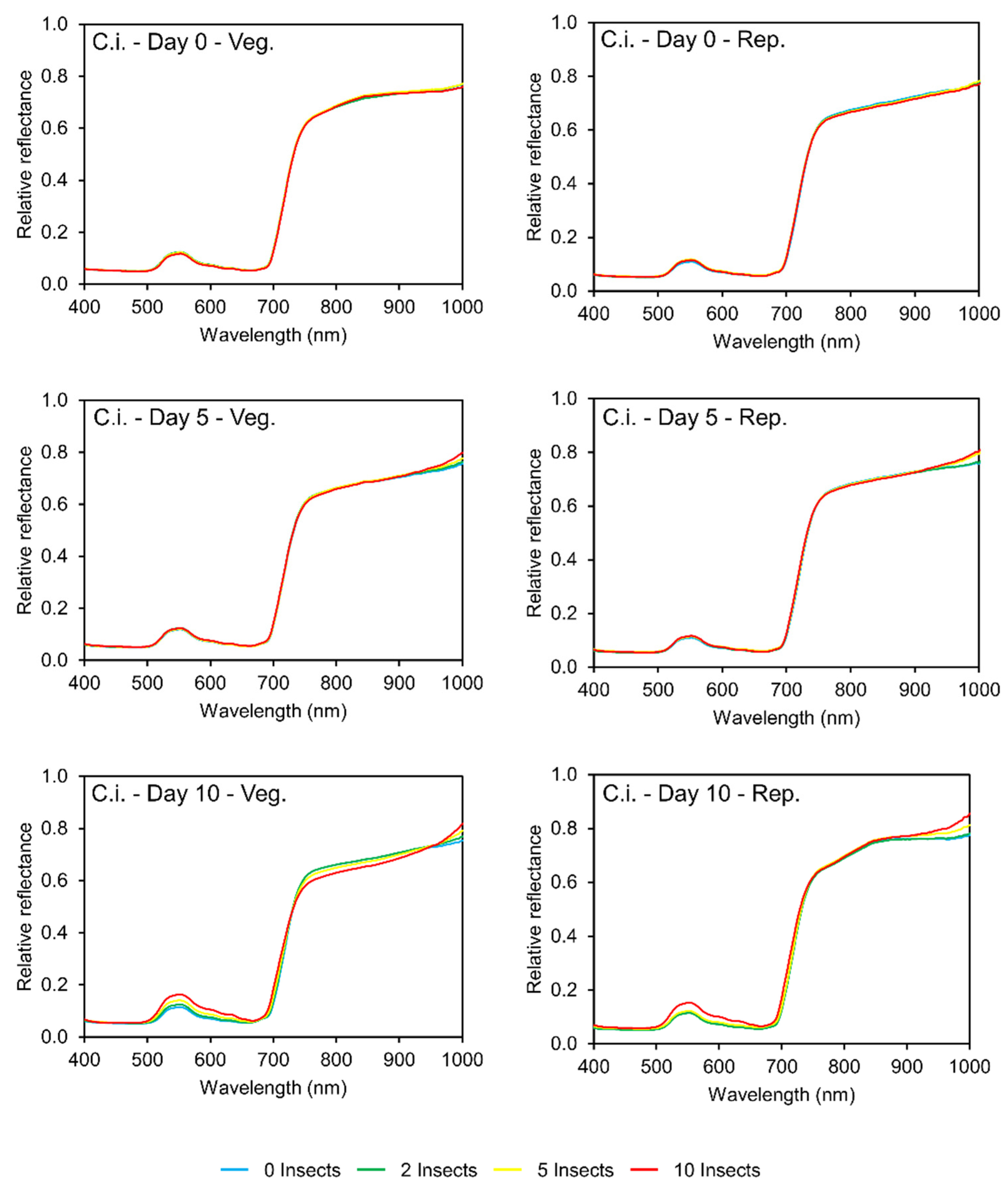
3.2. Reflectance Patterns
3.2.1. Stinkbugs
3.2.2. Caterpillars
3.3. Principal Component Analysis and Analysis of Variance
3.3.1. Stinkbugs
3.3.2. Caterpillars
3.4. Results of Multilayer Perceptron Artificial Neural Network
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hartman, G.L.; West, E.D.; Herman, T.K. Crops that feed the World 2. Soybean worldwide production, use, and constraints caused by pathogens and pests. Food Secur. 2011, 3, 5–17. [Google Scholar] [CrossRef]
- Companhia Nacional De Abastecimento—CONAB. Acompanhamento de Safra Brasileira: Grãos, Safra 2020/21, Oitavo Levantamento. Available online: https://www.conab.gov.br/info-agro/safras/graos/boletim-da-safra-de-graos/item/download/37061_9f96937bef1ba89e8b223cc73dbb2475 (accessed on 7 February 2022).
- United States Department of Agriculture—USDA. World Agricultural Production, Circular Series, WAP 2-22, February 2022. Available online: https://apps.fas.usda.gov/psdonline/circulars/production.pdf (accessed on 20 February 2022).
- Oliveira, C.; Auad, A.; Mendes, S.; Frizzas, M. Crop losses and the economic impact of insect pests on Brazilian agriculture. Crop Prot. 2014, 56, 50–54. [Google Scholar] [CrossRef]
- Rangel, L.E.P. Perdas e danos para o agronegócio. Agroanalysis 2015, 35, 30–31. [Google Scholar]
- Bueno, A.F.; Panizzi, A.R.; Corrêa-Ferreira, B.S.; Hoffmann-Campo, C.B.; Sosa-Gómez, D.R.; Gazzoni, D.L.; Hirose, E.; Moscardi, F.; Corso, I.C.; Oliveira, L.J.; et al. Histórico e evolução do Manejo Integrado de Pragas da soja no Brasil. In Soja—Manejo Integrado de Insetos e Outros Artrópodes Praga; Hoffmann-Campo, C.B., Corrêa-Ferreira, S.B., Moscardi, F., Eds.; Embrapa: Brasília, Brazil, 2012; pp. 37–74. [Google Scholar]
- Denez, M.D.; Bueno, A.F.; Pasini, A.; Bortolotto, O.C.; Stecca, C.S. Biological parameters of Podisus nigrispinus (Hemiptera: Pentatomidae) fed with different soybean insect pests. Ann. Entomol. Soc. Am. 2014, 107, 967–997. [Google Scholar] [CrossRef]
- Horikoshi, R.J.; Dourado, P.M.; Berger, G.U.; Fernandes, D.S.; Omoto, C.; Willse, A.; Martinelli, S.; Head, G.P.; Corrêa, A.S. Large-scale assessment of lepidopteran soybean pests and efficacy of Cry1Ac soybean in Brazil. Sci. Rep. 2021, 11, 15956. [Google Scholar] [CrossRef]
- Justus, C.M.; Paula-Moraes, S.V.; Pasini, A.; Hoback, W.W.; Hayashida, R.; Bueno, A.F. Simulated soybean pod and flower injuries and economic thresholds for Spodoptera eridania (Lepidoptera: Noctuidae) management decisions. Crop Prot. 2022, 155, 105936. [Google Scholar] [CrossRef]
- Panizzi, A.R.; Lucini, T.; Aldrich, J.R. Dynamics in pest status of phytophagous stink bugs in the Neotropics. Neotrop. Entomol. 2022, 51, 18–31. [Google Scholar] [CrossRef]
- Gomes, E.C.; Hayashida, R.; Bueno, A.F. Dichelops melacanthus and Euschistus heros injury on maize: Basis for re-evaluating stink bug thresholds for IPM decisions. Crop Prot. 2020, 130, 105050. [Google Scholar] [CrossRef]
- Bueno, A.F.; Panizzi, A.R.; Hunt, T.E.; Dourado, P.M.; Pitta, R.M.; Gonçalves, J. Challenges for adoption of integrated pest management (IPM): The soybean example. Neotrop. Entomol. 2021, 50, 5–20. [Google Scholar] [CrossRef]
- Sosa-Gómez, D.R.; Silva, J.J. Neotropical brown stink bug (Euschistus heros) resistance to methamidophos in Paraná, Brazil. Pesqui. Agropecuária Bras. 2010, 45, 767–769. [Google Scholar] [CrossRef]
- Guedes, R.N.C.; Cutler, G.C. Insecticide-induced hormesis and arthropod pest management. Pest Manag. Sci. 2014, 70, 690–697. [Google Scholar] [CrossRef] [PubMed]
- Pazini, J.D.B.; Grützmacher, A.D.; Martins, J.F.D.S.; Pasini, R.A.; Rakes, M. Selectivity of pesticides used in rice crop on Telenomus podisi and Trichogramma pretiosum. Pesqui. Agropecuária Trop. 2016, 46, 327–335. [Google Scholar] [CrossRef]
- Gazzoni, D.L. Perspectivas do manejo de pragas. In Soja—Manejo Integrado de Insetos e Outros Artrópodes Praga; Hoffmann-Campo, C.B., Corrêa-Ferreira, S.B., Moscardi, F., Eds.; Embrapa: Brasília, Brazil, 2012; pp. 789–830. [Google Scholar]
- Liu, Z.Y.; Qi, J.G.; Wang, N.N.; Zhu, Z.R.; Luo, J.; Liu, L.J.; Tang, J.; Cheng, J.A. Hyperspectral discrimination of foliar biotic damages in rice using principal component analysis and probabilistic neural network. Precis. Agric. 2018, 19, 973–991. [Google Scholar] [CrossRef]
- Nansen, C.; Elliot, N. Remote Sensing and reflectance profiling in Entomology. Annu. Rev. Entomol. 2016, 61, 139–158. [Google Scholar] [CrossRef] [PubMed]
- Iost Filho, F.H.; Heldens, W.B.; Kong, Z.; Lange, E.S. Drones: Innovative technology for use in precision pest management. J. Econ. Entomol. 2020, 113, 1–25. [Google Scholar] [CrossRef]
- Greene, A.D.; Reay-Jones, F.P.F.; Kirk, K.R.; Peoples, B.K.; Greene, J.K. Spatial Associations of key lepidopteran pests with defoliation, NDVI, and plant height in soybean. Environ. Entomol. 2021, 50, 1378–1392. [Google Scholar] [CrossRef] [PubMed]
- Moran, M.S.; Inoue, Y.; Barnes, E.M. Opportunities and limitations for imagebased remote sensing in precision crop management. Remote Sens. Environ. 1997, 61, 319–346. [Google Scholar] [CrossRef]
- Pinto, J.; Powell, S.; Peterson, R.; Rosalen, D.; Fernandes, O. Detection of defoliation injury in peanut with hyperspectral proximal remote Sensing. Remote Sens. 2021, 12, 3828. [Google Scholar] [CrossRef]
- Terentev, A.; Dolzhenko, V.; Fedotov, A.; Eremenko, D. Current state of hyperspectral remote sensing for early plant disease detection: A review. Sensors 2022, 22, 757. [Google Scholar] [CrossRef]
- Ang, K.L.M.; Seng, J.K.P. Big data and machine learning with hyperspectral information in agriculture. IEEE Access 2021, 9, 36699–36718. [Google Scholar] [CrossRef]
- Bendel, N.; Backhaus, A.; Kicherer, A.; Köckerling, J.; Maixner, M.; Jarausch, B.; Biancu, S.; Klück, H.-C.; Seiffert, U.; Voegele, R.T.; et al. Detection of two different grapevine yellows in Vitis vinifera using hyperspectral imaging. Remote Sens. 2020, 24, 4151. [Google Scholar] [CrossRef]
- Kasinathan, T.; Uyyala, R.S. Machine learning ensemble with image processing for pest identification and classification in field crops. Neural Comput. Appl. 2021, 33, 7491–7504. [Google Scholar] [CrossRef]
- Silva, C.B.; Silva, A.A.N.; Barroso, G.; Yamamoto, P.T.; Arthur, V.; Toledo, C.F.M.; Mastrangelo, T.D.A. Convolutional neural networks using enhanced radiographs for real-time detection of Sitophilus zeamais in maize grain. Foods 2021, 10, 879. [Google Scholar] [CrossRef] [PubMed]
- Moshou, D.; Bravo, C.; West, J.; Wahlen, S.; McCartney, A.; Ramon, H. Automatic detection of ‘yellow rust’ in wheat using reflectance measurements and neural networks. Comput. Electron. Agric. 2004, 44, 173–188. [Google Scholar] [CrossRef]
- Zhao, X.; Zhang, J.; Huang, Y.; Tian, Y.; Yuan, L. Detection and discrimination of disease and insect stress of tea plants using hyperspectral imaging combined with wavelet analysis. Comput. Electron. Agric. 2022, 193, 106717. [Google Scholar] [CrossRef]
- Golhani, K.; Balasundram, S.K.; Vadamalai, G.; Pradhan, B. A review of neural networks in plant disease detection using hyperspectral data. Inf. Process. Agric. 2018, 5, 354–371. [Google Scholar] [CrossRef]
- Bueno, R.C.O.F.; Parra, J.R.P.; Bueno, A.F.; Haddad, M.L. Desempenho de tricogramatídeos como potenciais agentes de controle de Pseudoplusia includens Walker (Lepidoptera: Noctuidae). Neotrop. Entomol. 2009, 38, 389–394. [Google Scholar] [CrossRef]
- Greene, G.L.; Leppla, N.C.; Dickerson, W.A. Velvetbean caterpillar: A rearing procedure and artificial medium. J. Econ. Entomol. 1976, 69, 487–488. [Google Scholar] [CrossRef]
- Fehr, W.R.; Caviness, C.E. Stages of Soybean Development; Special Report 80; Iowa State University of Science and Technology: Ames, IA, USA, 1977; 11p. [Google Scholar]
- Liu, Z.Y.; Wu, H.F.; Huang, J.F. Application of neural networks to discriminate fungal infection levels in rice panicles using hyperspectral refectance and principal components analysis. Comput. Electron. Agr. 2010, 72, 99–106. [Google Scholar] [CrossRef]
- Paoletti, M.E.; Haut, J.M.; Plaza, J.; Plaza, A. Deep learning classifiers for hyperspectral imaging: A review. ISPRS J. Photogramm. 2019, 158, 279–317. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. 2020. Available online: https://www.R-project.org/ (accessed on 20 February 2022).
- Riffel, C.T.; Garcia, M.S.; Santi, A.L.; Basso, C.J.; Della Flora, L.P.; Cherubin, M.R.; Eitelwein, M.T. Sample density applied to the georeferenced monitoring of defoliating caterpillars in soybean crop. Ciência Rural 2012, 43, 2112–2119. [Google Scholar] [CrossRef]
- Da Silva Stefanelo, L.; Filho, A.C.; Guedes, J.V.C.; Sturmer, G.R.; Facco, G.; de Bem, C.M. Sequential sampling for evaluation of caterpillars, small and large in soybean. Afr. J. Agric. Res. 2017, 12, 932–943. [Google Scholar]
- Fernandes, M.G.; Costa, E.N.; Cavada, L.H.; Mota, T.A.; Fonseca, P.R.B. Spatial distribution and sampling plan of the phytophagous stink bug complex in different soybean production systems. J. Appl. Entomol. 2019, 143, 236–249. [Google Scholar] [CrossRef]
- Pezzini, D.T.; DiFonzo, C.D.; Finke, D.L.; Hunt, T.E.; Knodel, J.J.; Krupke, C.H.; McCornack, B.; Michel, A.P.; Moon, R.D.; Philips, C.R.; et al. Spatial patterns and sequential sampling plans for estimating densities of stink bugs (Hemiptera: Pentatomidae) in soybean in the north central region of the United States. J. Econ. Entomol. 2019, 112, 1732–1740. [Google Scholar] [CrossRef] [PubMed]
- Lillesand, T.M.; Kiefer, R.W.; Chipman, J.W. Remote Sensing and Image Interpretation; Wiley: Hoboken, NJ, USA, 2015; p. 736. [Google Scholar]
- Pazini, J.D.B.; Botta, R.A.; Seidel, E.J.; Silva, F.F.; Martins, J.F.S.; Barrigossi, J.A.F.; Rübenich, R. Geostatistics applied to the study of the spatial distribution of Tibraca limbativentris in flooded rice fields. Ciência Rural 2012, 45, 1006–1012. [Google Scholar] [CrossRef]
- Liang, G.C.; Ouyang, Y.C.; Dai, S.M. Detection and classification of rice infestation with rice leaf folder (Cnaphalocrocis medinalis) using hyperspectral imaging techniques. Remote Sens. 2021, 13, 4587. [Google Scholar] [CrossRef]
- Xiao, Z.; Yin, K.; Geng, L.; Wu, J.; Zhang, F.; Liu, Y. Pest identification via hyperspectral image and deep learning. Signal Image Video Process. 2022, 16, 873–880. [Google Scholar] [CrossRef]
- Ranjitha, G.; Srinivasan, M.R. Hyperspectral radiometry for the detection and discrimination of damage caused by sucking pests of cotton. Curr. Biotica 2014, 8, 5–12. [Google Scholar]
- Liu, X.D.; Sun, Q.H. Early assessment of the yield loss in rice due to the brown planthopper using a hyperspectral remote sensing method. Int. J. Pest Manag. 2016, 62, 205–213. [Google Scholar] [CrossRef]
- Chen, T.; Zeng, R.; Guo, W.; Hou, X.; Lan, Y.; Zhang, L. Detection of stress in cotton (Gossypium hirsutum L.) caused by aphids using leaf level hyperspectral measurements. Sensors 2018, 18, 2798. [Google Scholar] [CrossRef]
- Liu, T.; Shi, T.; Zhang, H.; Wu, C. Detection of rise damage by leaf folder (Cnaphalocrocis medinalis) using unmanned aerial vehicle based hyperspectral data. Sustainability 2020, 12, 9343. [Google Scholar] [CrossRef]
- Barros, P.P.; Schutze, I.X.; Iost Filho, F.H.; Yamamoto, P.T.; Fiorio, P.R.; Demattê, J.A. Monitoring Bemisia tabaci (Gennadius) (Hemiptera: Aleyrodidae) infestation in Soybean by proximal sensing. Insects 2021, 12, 47. [Google Scholar] [CrossRef] [PubMed]
- Furuya, D.E.G.; Ma, L.; Pinheiro, M.M.F.; Gomes, F.D.G.; Gonçalvez, W.N.; Marcato Junior, J.; Rodrigues, D.C.; Blassioli-Moraes, M.C.; Michereff, M.F.F.; Borges, M.; et al. Prediction of insect-herbivory-damage and insect-type attack in maize plants using hyperspectral data. Int. J. Appl. Earth Obs. Geoinf. 2021, 105, 102608. [Google Scholar]
- Bonaventure, G. Perception of insect feeding by plants. Plant Biol. 2012, 14, 872–880. [Google Scholar] [CrossRef]
- Ongaratto, S.; Baldin, E.L.; Hunt, T.E.; Montezano, D.G.; Robinson, E.A.; Santos, M.C. Effects of intraguild interactions on Anticarsia gemmatalis and Chrysodeixis includens larval fitness and behavior in soybean. Pest Manag. Sci. 2021, 77, 2939–2947. [Google Scholar] [CrossRef]
- Haase, S.; Mccarthy, C.; Ferrelli, M.; Pidre, M.; Sciocco-Cap, A.; Romanowski, V. Development of a Recombination System for the generation of occlusion positive genetically modified Anticarsia Gemmatalis multiple nucleopolyhedrovirus. Viruses 2015, 7, 1599–1612. [Google Scholar] [CrossRef]
- Bortolotto, O.C.; Pomari-Fernandes, A.; Bueno, R.C.O.F.; Bueno, A.F.; Cruz, Y.K.S.D.; Sanzovo, A.; Ferreira, R.B. The use of soybean integrated pest management in Brazil: A review. ASB 2015, 1, 25–32. [Google Scholar] [CrossRef]
- Bernardi, O.; Sorgatto, R.J.; Barbosa, A.D.; Domingues, F.A.; Dourado, P.M.; Carvalho, R.A.; Martinelli, S.; Head, G.P.; Omoto, C. Low susceptibility of Spodoptera cosmioides, Spodoptera eridania and Spodoptera frugiperda (Lepidoptera: Noctuidae) to genetically-modified soybean expressing Cry1Ac protein. Crop Prot. 2014, 58, 33–40. [Google Scholar] [CrossRef]
- Sosa-Gómez, D.R.; Delpin, K.E.; Moscardi, F.; Nozaki, M.D.H. The impact of fungicides on Nomuraea rileyi (Farlow) Samson epizootics and on populations of Anticarsia gemmatalis Hübner (Lepidoptera: Noctuidae), on soybean. Neotrop. Entomol. 2003, 32, 287–291. [Google Scholar] [CrossRef]
- Souza, B.H.S.; Boiça Júnior, A.L.; Janini, J.C.; Silva, A.G.; Lobato Rodrigues, N.E. Feeding of Spodoptera eridania (Lepidoptera: Noctuidae) on soybean genotypes. Rev. Colomb. Entomol. 2012, 38, 215–223. [Google Scholar] [CrossRef]
- Wille, P.E.; Pereira, B.A.; Wille, C.L.; Restelatto, S.S.; Boff, M.I.C.; Franco, C.R. Natural resistance of soybean cultivars to the soybean looper larva Chrysodeixis includens (Lepidoptera: Noctuidae). Pesqui. Agropecuária Bras. 2017, 52, 18–25. [Google Scholar] [CrossRef]
- Silva, D.M.; Bueno, A.F.; Stecca, C.S.; Andrade, K.; Neves, P.M.O.J.; Oliveira, M.C.N. Biology of Spodoptera eridania and Spodoptera cosmioides (Lepidoptera: Noctuidae) on different host plants. Fla. Entomol. 2017, 100, 752–760. [Google Scholar] [CrossRef]
- Hoffer, R.M. Biological and physical considerations in applying computer-aided analysis techniques to remote sensor data. In Remote Sensing: The Quantitative Approach; Swain, P.H., Davis, S.M., Eds.; McGraw-Hill Book Company: New York, NY, USA, 1978; pp. 227–289. [Google Scholar]
- Slaton, M.R.; Raymond Hunt, E.; Smith, W.K. Estimating near-infrared leaf reflectance from leaf structural characteristics. Am. J. Bot. 2001, 88, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Sudbrink, D.L.; Harris, F.A.; Robbins, J.T.; English, P.J.; Willers, J.L. Evaluation of remote sensing to identify variability in cotton plant growth and correlation with larval densities of beet armyworm and cabbage looper (Lepidoptera: Noctuidae). Fla. Entomol. 2003, 86, 290–294. [Google Scholar] [CrossRef]
- Huang, J.; Liao, H.; Zhu, Y.; Sun, J.; Sun, Q.; Liu, X. Hyperspectral detection of rice damaged by rice leaf folder (Cnaphalocrocis medinalis). Comput. Electron. Agr. 2012, 82, 100–107. [Google Scholar] [CrossRef]
- Tetila, E.C.; Machado, B.B.; Astolfi, G.; de Souza Belete, N.A.; Amorim, W.P.; Roel, A.R.; Pistori, H. Detection and classification of soybean pests using deep learning with UAV images. Comput. Electron. Agr. 2020, 179, 105836. [Google Scholar] [CrossRef]
- Gui, J.; Fei, J.; Wu, Z.; Fu, X.; Diakite, A. Grading method of soybean mosaic disease based on hyperspectral imaging technology. Inf. Process. Agric. 2021, 8, 380–385. [Google Scholar] [CrossRef]
- Cao, X.; Zhou, F.; Xu, L.; Meng, D.; Xu, Z. Hyperspectral image classification with Markov random fields and a convolutional neural network. IEEE Trans. Image Process. 2018, 27, 2354–2367. [Google Scholar] [CrossRef]
- Tran, T.T.; Choi, J.W.; Le, T.T.H.; Kim, J.W. A comparative study of deep CNN in forecasting and classifying the macronutrient deficiencies on development of tomato plant. Appl. Sci. 2019, 9, 1601. [Google Scholar] [CrossRef]
- Sabzi, S.; Pourdarbani, R.; Rohban, M.H.; García-Mateos, G.; Paliwal, J.; Molina-Martínez, J.M. Early detection of excess nitrogen consumption in cucumber plants using hyperspectral imaging based on hybrid neural networks and the imperialist competitive algorithm. Agronomy 2021, 11, 575. [Google Scholar] [CrossRef]
- Virnodkar, S.S.; Pachghare, V.K.; Patil, V.C.; Jha, S.K. Remote sensing and machine learning for crop water stress determination in various crops: A critical review. Precis. Agric. 2020, 21, 1121–1155. [Google Scholar] [CrossRef]
- Yan, T.; Xu, W.; Lin, J.; Duan, L.; Gao, P.; Zhang, C.; Lv, X. Combining multi-dimensional convolutional neural network (CNN) with visualization method for detection of aphis gossypii glover infection in cotton leaves using hyperspectral imaging. Front. Plant Sci. 2021, 12, 74. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.; Zhang, W.; Wei, X. A review on weed detection using ground-based machine vision and image processing techniques. Comput. Electron. Agr. 2019, 158, 226–240. [Google Scholar] [CrossRef]
- Gao, B.; Yu, L.; Ren, L.; Zhan, Z.; Luo, Y. Early detection of Dendroctonus valens infestation with machine learning algorithms based on hyperspectral reflectance. Remote Sens. 2022, 14, 1373. [Google Scholar] [CrossRef]
- Johari, S.N.A.M.; Khairunniza-Bejo, S.; Shariff, A.R.M.; Husin, N.A.; Basri, M.M.M.; Kamarudin, N. Identification of bagworm (Metisa plana) instar stages using hyperspectral imaging and machine learning techniques. Comput. Electron. Agric. 2022, 194, 106739. [Google Scholar] [CrossRef]
- Ramos, A.P.M.; Gomes, F.D.G.; Pinheiro, M.M.F.; Furuya, D.E.G.; Gonçalvez, W.N.; Marcato Junior, J.; Michereff, M.F.F.; Blassioli-Moraes, M.C.; Borges, M.; Alaumann, R.A.; et al. Detecting the attack of the fall armyworm (Spodoptera frugiperda) in cotton plants with machine learning and spectral measurements. Precis. Agric. 2022, 23, 470–491. [Google Scholar] [CrossRef]



| State | Hyperparameters | Values |
|---|---|---|
| Optimized | Activation functions | {Tanh, Relu} |
| Optimizer | {Adam, SGD} | |
| Learning rate | Adaptative | |
| Number of hidden layer | {1, 2} | |
| Hidden layer size | {50, 100} |
| Caterpillars (n Five Plants−1) | Leaf Area in cm2 (Leaf Area Index) | ||
|---|---|---|---|
| C.i. | S.e. | df; F; p; CV% # | |
| Soybean vegetative stage | |||
| 0 | 34.7 ± 2.0 † a,A (6.14) | 34.6 ± 1.9 a,A (6.12) | 1; 0.0002; 0.98; 22.16 |
| 2 | 24.7 ± 2.2 b,A (4.38) | 29.5 ± 1.9 b,A (5.23) | 1; 2.64; 0.11; 29.84 |
| 5 | 15.4 ± 2.6 c,B (2.74) | 25.1 ± 1.2 b,A (4.45) | 1; 11.38; 0.002; 38.63 |
| 10 | 4.2 ± 1.9 d,B (0.75) | 19.1 ± 0.9 c,A (3.39) | 1; 47.41; <0.001; 50.63 |
| df; F; p; CV% * | 3; 34.62; <0.001; 43.22 | 3; 17.35; <0.001; 22.58 | -- |
| Soybean reproductive stage | |||
| 0 | 35.2 ± 3.8 a,A (6.23) | 34.4 ± 2.1 a,A (6.09) | 1; 0.03; 0.86; 34.22 |
| 2 | 23.00 ± 2.1 b,A (4.07) | 27.5 ± 1.6 b,A (4.87) | 1; 2.89; 0.09; 29.05 |
| 5 | 10.2 ± 2.1 c,B (1.81) | 22.2 ± 1.2 b,A (3.93) | 1; 22.81; <0.001; 42.43 |
| 10 | 1.4 ± 0.9 d,B (0.25) | 12.4 ± 0.5 c,A (2.19) | 1; 108.16; <0.001; 42.01 |
| df; F; p; CV% * | 3; 35.83; <0.001; 34.83 | 3; 37.96; <0.001; 24.25 | -- |
| Situation of Stress d | Accuracy (%) | Kappa Coefficient | Precision | Recall (Sensitivity) | F1 Score | ||
|---|---|---|---|---|---|---|---|
| Phenology a | DAI b | Caterpillars (n) c | |||||
| Testing dataset | |||||||
| 1- V; R | 5; 10 | 0; 2; 5; 10 | 47.0 | 0.46 | 0.48 | 0.48 | 0.48 |
| 2- V; R | 5; 10 | 0; 2; 5 + 10 | 59.0 | 0.58 | 0.59 | 0.56 | 0.56 |
| 3- V; R | 5; 10 | 0 + 2; 5 + 10 | 70.0 | 0.67 | 0.69 | 0.71 | 0.70 |
| 4- V; R | 5; 10 | 0; 2 + 5 + 10 | 74.0 | 0.73 | 0.70 | 0.72 | 0.71 |
| 5- V + R | 5; 10 | 0; 2; 5; 10 | 50.0 | 0.48 | 0.50 | 0.51 | 0.51 |
| 6- V + R | 5; 10 | 0; 2; 5 + 10 | 56.0 | 0.53 | 0.51 | 0.50 | 0.50 |
| 7- V + R | 5; 10 | 0 + 2; 5 + 10 | 66.0 | 0.63 | 0.66 | 0.66 | 0.66 |
| 8- V + R | 5; 10 | 0; 2 + 5 + 10 | 71.0 | 0.67 | 0.65 | 0.65 | 0.65 |
| Training dataset | |||||||
| 1- V; R | 5; 10 | 0; 2; 5; 10 | 49.8 | 0.49 | 0.51 | 0.50 | 0.49 |
| 2- V; R | 5; 10 | 0; 2; 5 + 10 | 61.4 | 0.60 | 0.59 | 0.57 | 0.57 |
| 3- V; R | 5; 10 | 0 + 2; 5 + 10 | 69.3 | 0.68 | 0.70 | 0.69 | 0.69 |
| 4- V; R | 5; 10 | 0; 2 + 5 + 10 | 76.9 | 0.76 | 0.73 | 0.69 | 0.70 |
| 5- V + R | 5; 10 | 0; 2; 5; 10 | 49.5 | 0.47 | 0.50 | 0.50 | 0.49 |
| 6- V + R | 5; 10 | 0; 2; 5 + 10 | 59.0 | 0.56 | 0.56 | 0.55 | 0.55 |
| 7- V + R | 5; 10 | 0 + 2; 5 + 10 | 68.2 | 0.65 | 0.68 | 0.68 | 0.68 |
| 8- V + R | 5; 10 | 0; 2 + 5 + 10 | 75.1 | 0.72 | 0.71 | 0.66 | 0.67 |
| Situation of Stress d | Accuracy (%) | Kappa Coefficient | Precision | Recall (Sensitivity) | F1 Score | ||
|---|---|---|---|---|---|---|---|
| Phenology a | DAI b | Caterpillars (n) c | |||||
| Testing dataset | |||||||
| 1- V; R | 5; 10 | 0; 2; 5; 10 | 50.0 | 0.47 | 0.47 | 0.52 | 0.52 |
| 2- V; R | 5; 10 | 0; 2; 5 + 10 | 64.0 | 0.60 | 0.59 | 0.58 | 0.58 |
| 3- V; R | 5; 10 | 0 + 2; 5 + 10 | 72.0 | 0.70 | 0.73 | 0.72 | 0.72 |
| 4- V; R | 5; 10 | 0; 2 + 5 + 10 | 75.0 | 0.70 | 0.70 | 0.73 | 0.72 |
| 5- V + R | 5; 10 | 0; 2; 5; 10 | 33.0 | 0.23 | 0.32 | 0.33 | 0.33 |
| 6- V + R | 5; 10 | 0; 2; 5 + 10 | 63.0 | 0.54 | 0.59 | 0.58 | 0.58 |
| 7- V + R | 5; 10 | 0 + 2; 5 + 10 | 73.0 | 0.71 | 0.70 | 0.71 | 0.71 |
| 8- V + R | 5; 10 | 0; 2 + 5 + 10 | 75.0 | 0.70 | 0.72 | 0.71 | 0.72 |
| Training dataset | |||||||
| 1- V; R | 5; 10 | 0; 2; 5; 10 | 67.9 | 0.66 | 0.69 | 0.68 | 0.67 |
| 2- V; R | 5; 10 | 0; 2; 5 + 10 | 76.4 | 0.74 | 0.76 | 0.74 | 0.73 |
| 3- V; R | 5; 10 | 0 + 2; 5 + 10 | 82.8 | 0.80 | 0.84 | 0.83 | 0.83 |
| 4- V; R | 5; 10 | 0; 2 + 5 + 10 | 86.1 | 0.84 | 0.83 | 0.82 | 0.82 |
| 5- V + R | 5; 10 | 0; 2; 5; 10 | 67.8 | 0.63 | 0.69 | 0.68 | 0.68 |
| 6- V + R | 5; 10 | 0; 2; 5 + 10 | 73.1 | 0.67 | 0.71 | 0.70 | 0.70 |
| 7- V + R | 5; 10 | 0 + 2; 5 + 10 | 82.1 | 0.76 | 0.83 | 0.82 | 0.82 |
| 8- V + R | 5; 10 | 0; 2 + 5 + 10 | 85.7 | 0.75 | 0.78 | 0.76 | 0.77 |
| Situation of Stress d | Accuracy (%) | Kappa Coefficient | Precision | Recall (Sensitivity) | F1 Score | ||
|---|---|---|---|---|---|---|---|
| Phenology a | DAI b | Caterpillars (n) c | |||||
| Testing dataset | |||||||
| 1- V; R | 5; 10 | 0; 2; 5; 10 | 54.0 | 0.51 | 0.55 | 0.54 | 0.54 |
| 2- V; R | 5; 10 | 0; 2; 5 + 10 | 61.0 | 0.57 | 0.62 | 0.58 | 0.58 |
| 3- V; R | 5; 10 | 0 + 2; 5 + 10 | 66.0 | 0.57 | 0.62 | 0.65 | 0.64 |
| 4- V; R | 5; 10 | 0; 2 + 5 + 10 | 74.0 | 0.69 | 0.70 | 0.70 | 0.70 |
| 5- V + R | 5; 10 | 0; 2; 5; 10 | 40.0 | 0.31 | 0.38 | 0.40 | 0.39 |
| 6- V + R | 5; 10 | 0; 2; 5 + 10 | 57.0 | 0.47 | 0.57 | 0.53 | 0.53 |
| 7- V + R | 5; 10 | 0 + 2; 5 + 10 | 63.0 | 0.51 | 0.63 | 0.65 | 0.64 |
| 8- V + R | 5; 10 | 0; 2 + 5 + 10 | 74.0 | 0.62 | 0.70 | 0.67 | 0.70 |
| Training dataset | |||||||
| 1- V; R | 5; 10 | 0; 2; 5; 10 | 57.0 | 0.54 | 0.59 | 0.57 | 0.56 |
| 2- V; R | 5; 10 | 0; 2; 5 + 10 | 70.1 | 0.67 | 0.68 | 0.68 | 0.67 |
| 3- V; R | 5; 10 | 0 + 2; 5 + 10 | 77.3 | 0.74 | 0.78 | 0.77 | 0.77 |
| 4- V; R | 5; 10 | 0; 2 + 5 + 10 | 82.0 | 0.79 | 0.78 | 0.76 | 0.76 |
| 5- V + R | 5; 10 | 0; 2; 5; 10 | 57.8 | 0.52 | 0.59 | 0.58 | 0.58 |
| 6- V + R | 5; 10 | 0; 2; 5 + 10 | 65.5 | 0.58 | 0.64 | 0.63 | 0.63 |
| 7- V + R | 5; 10 | 0 + 2; 5 + 10 | 75.0 | 0.67 | 0.76 | 0.75 | 0.75 |
| 8- V + R | 5; 10 | 0; 2 + 5 + 10 | 82.1 | 0.73 | 0.77 | 0.75 | 0.75 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iost Filho, F.H.; de Bastos Pazini, J.; de Medeiros, A.D.; Rosalen, D.L.; Yamamoto, P.T. Assessment of Injury by Four Major Pests in Soybean Plants Using Hyperspectral Proximal Imaging. Agronomy 2022, 12, 1516. https://doi.org/10.3390/agronomy12071516
Iost Filho FH, de Bastos Pazini J, de Medeiros AD, Rosalen DL, Yamamoto PT. Assessment of Injury by Four Major Pests in Soybean Plants Using Hyperspectral Proximal Imaging. Agronomy. 2022; 12(7):1516. https://doi.org/10.3390/agronomy12071516
Chicago/Turabian StyleIost Filho, Fernando Henrique, Juliano de Bastos Pazini, André Dantas de Medeiros, David Luciano Rosalen, and Pedro Takao Yamamoto. 2022. "Assessment of Injury by Four Major Pests in Soybean Plants Using Hyperspectral Proximal Imaging" Agronomy 12, no. 7: 1516. https://doi.org/10.3390/agronomy12071516
APA StyleIost Filho, F. H., de Bastos Pazini, J., de Medeiros, A. D., Rosalen, D. L., & Yamamoto, P. T. (2022). Assessment of Injury by Four Major Pests in Soybean Plants Using Hyperspectral Proximal Imaging. Agronomy, 12(7), 1516. https://doi.org/10.3390/agronomy12071516








