Variability of Bioactive Substances in Potatoes (Solanum Tuberosum L.) Depending on Variety and Maturity
Abstract
:1. Introduction
2. Material and Methodology
2.1. Plant Material
2.2. Extract Preparation
2.3. Total Anthocyanin Content
2.4. Total Polyphenol Content
2.5. HPLC-DAD Determination of Chlorogenic Acid
2.6. Statistical Analysis
3. Results and Discussion
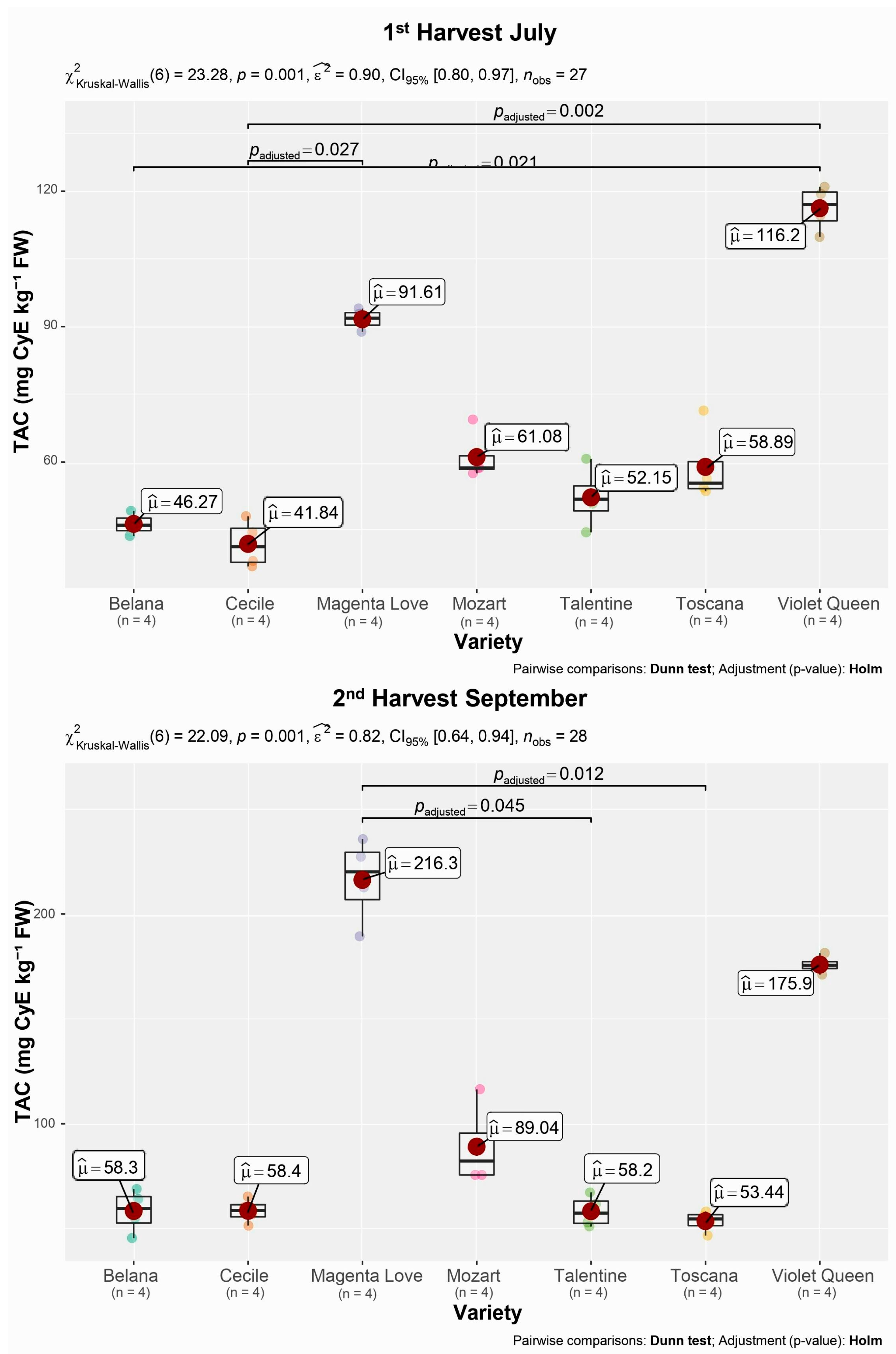
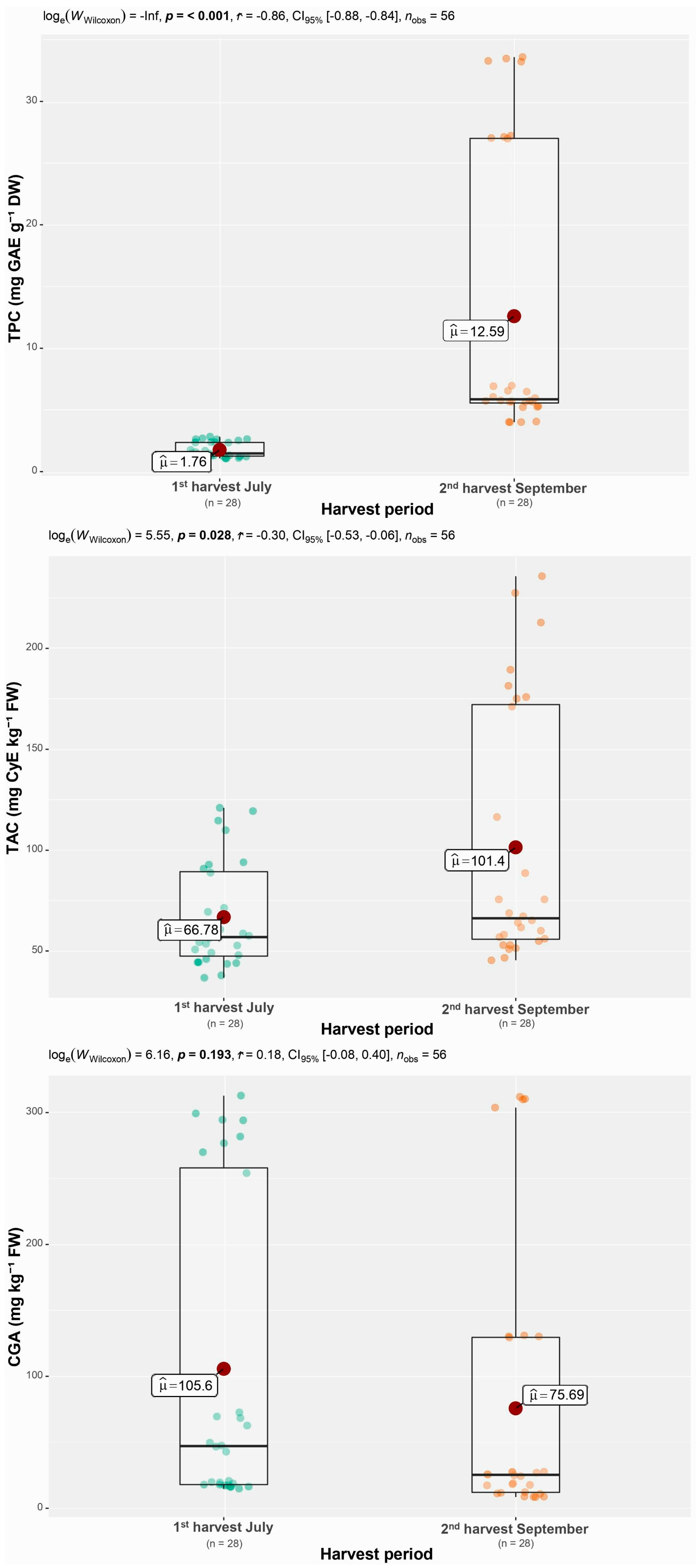
3.1. Total Anthocyanin Content
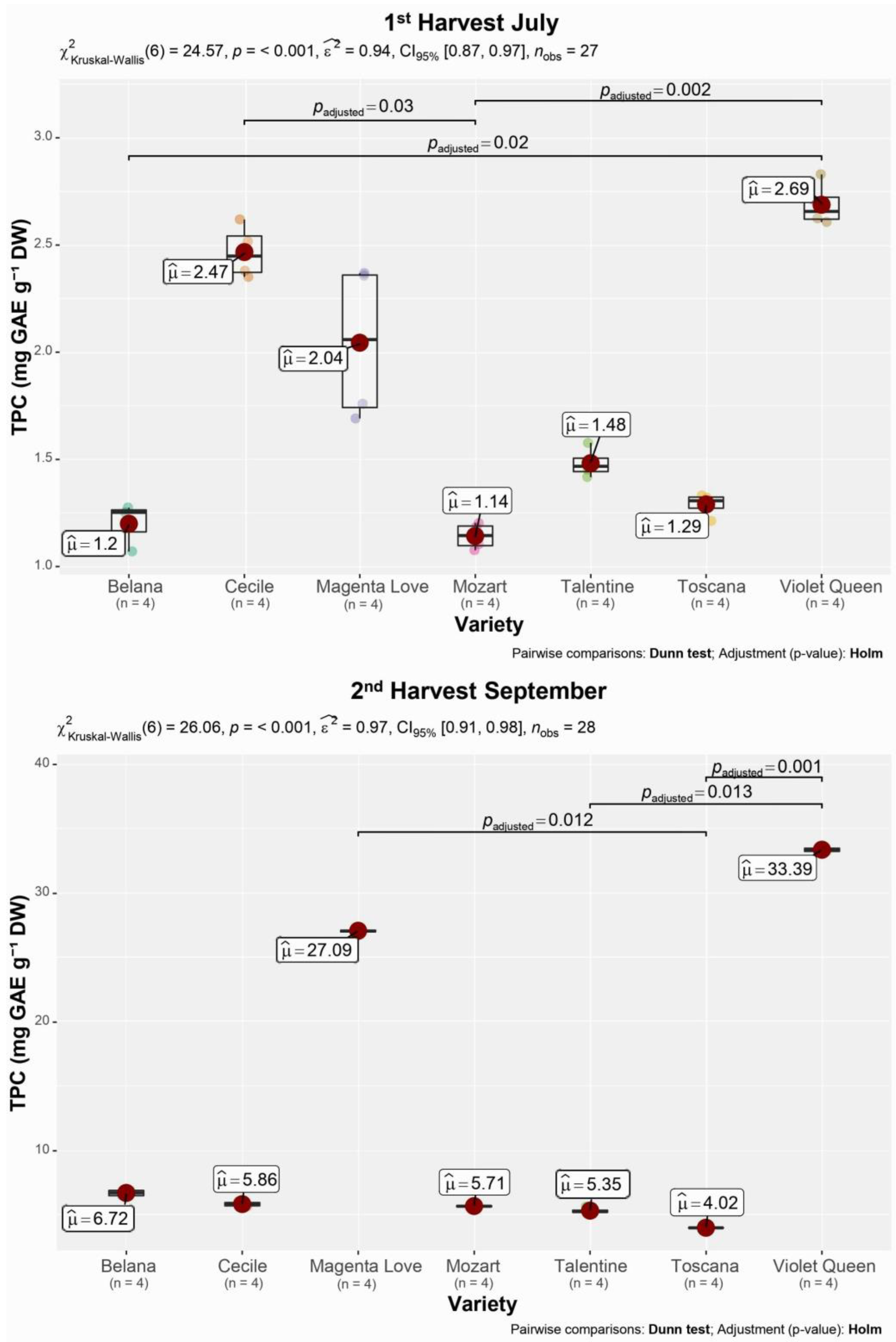
3.2. Total Polyphenol Content
3.3. Chlorogenic Acid
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mishra, T.; Raigond, P.; Thakur, N.; Dutt, S.; Singh, B. Recent Updates on Healthy Phytoconstituents in Potato: A Nutritional Depository. Potato Res. 2020, 63, 323–343. [Google Scholar] [CrossRef]
- Frontuto, D.; Carullo, D.; Harrison, S.M.; Brunton, N.P.; Ferrari, G.; Lyng, J.G.; Pataro, G. Optimization of Pulsed Electric Fields-Assisted Extraction of Polyphenols from Potato Peels Using Response Surface Methodology. Food Bioprocess Technol. 2019, 12, 1708–1720. [Google Scholar] [CrossRef]
- FAOSTAT. Data of Crops Production in the World. Data Division. 2020. Available online: https://www.fao.org/faostat/en/#data/QCL (accessed on 23 March 2022).
- Ngobese, Z.N.; Workneh, T.S.; Alimi, B.A.; Tesfay, S. Nutrient composition and starch characteristics of eight European potato cultivars cultivated in South Africa. J. Food Compos. Anal. 2017, 55, 1–11. [Google Scholar] [CrossRef]
- Nassar, A.M.; Sabally, K.; Kubow, S.; Leclerc, Y.N.; Donnelly, D.J. Some Canadian-grown potato cultivars contribute to a substantial content of essential dietary minerals. J. Agric. Food Chem. 2012, 60, 4688–4696. [Google Scholar] [CrossRef]
- McGill, C.R.; Kurilich, A.C.; Davignon, J. The role of potatoes and potato components in cardiometabolic health: A review. Ann. Med. 2013, 45, 467–473. [Google Scholar] [CrossRef]
- Mäkinen, S. Production, Isolation and Characterization of Bioactive Peptides with Antihypertensive Properties from Rapeseed and Potato Protein. Ph.D. Thesis, Food Chemistry & Food Development, Department of Biochemistry, The University of Turku, Turku, Finland, 2014. Available online: https://www.utupub.fi/bitstream/handle/10024/102101/diss2014M%C3%A4kinen.pdf?sequence=2&isAllowed=y (accessed on 9 June 2022).
- Van Niekerk, C.; Schönfeldt, H.; Hall, N.; Pretorius, B. 2016. The role of biodiversity in food security and nutrition: A potato cultivar case study. Food Nutr. Sci. 2016, 7, 371. [Google Scholar] [CrossRef] [Green Version]
- Burgos, G.; Auqui, S.; Amoros, W.; Salas, E.; Bonierbale, M. Ascorbic acid concentration of native Andean potato varieties as affected by environment, cooking and storage. J. Food Compos. Anal. 2009, 22, 533–538. [Google Scholar] [CrossRef]
- Sun, Q.; Du, M.; Navarre, D.A.; Zhu, M. Effect of Cooking Methods on Bioactivity of Polyphenols in Purple Potatoes. Antioxidants 2021, 10, 1176. [Google Scholar] [CrossRef]
- Schieber, A.; Saldaña, M.D. Potato peels: A source of nutritionally and pharmacologically interesting compounds–A review. Food 2009, 3, 23–29. [Google Scholar] [CrossRef]
- Navarre, D.A.; Brown, C.R.; Sathuvalli, V.R. Potato Vitamins, Minerals and Phytonutrients from a Plant Biology Perspective. Am. J. Potato Res. 2019, 96, 111–126. [Google Scholar] [CrossRef]
- Arts, I.C.; Hollman, P.C. Polyphenols and disease risk in epidemiologic studies. Am. J. Clin. Nutr. 2005, 81, 317S–325S. [Google Scholar] [CrossRef] [Green Version]
- Dai, J.; Mumper, R.J. Plant Phenolics: Extraction, Analysis and Their Antioxidant and Anticancer Properties. Molecules 2010, 15, 7313–7352. [Google Scholar] [CrossRef]
- Singh, A.; Holvoet, S.; Mercenier, A. Dietary polyphenols in the prevention and treatment of allergic diseases. Clin. Exp. Allergy 2011, 41, 1346–1359. [Google Scholar] [CrossRef]
- Zhou, Y.; Zheng, J.; Xu, D.P.; Li, S.; Chen, Y.M.; Li, H.B. Natural Polyphenols for Prevention and Treatment of Cancer. Nutrients 2016, 8, 515. [Google Scholar] [CrossRef]
- Jiang, H.; Wang, J.; Rogers, J.; Xie, J. Brain Iron Metabolism Dysfunction in Parkinson’s Disease. Mol. Neurobiol. 2017, 54, 3078–3101. [Google Scholar] [CrossRef]
- Cory, H.; Passarelli, S.; Szeto, J.; Tamez, M.; Mattei, J. The Role of Polyphenols in Human Health and Food Systems: A Mini-Review. Front. Nutr. 2018, 5, 9. [Google Scholar] [CrossRef] [Green Version]
- Schreiner, M. Vegetable crop management strategies to increase the quantity of phytochemicals. Eur. J. Nutr. 2005, 44, 85–94. [Google Scholar] [CrossRef]
- Tian, J.; Chen, J.; Lv, F.; Chen, S.; Chen, J.; Liu, D.; Ye, X. Domestic cooking methods affect the phytochemical composition and antioxidant activity of purple-fleshed potatoes. Food Chem. 2016, 197, 1264–1270. [Google Scholar] [CrossRef]
- Lachman, J.; Hamouz, K.; Šulc, M.; Orsák, M.; Pivec, V.; Hejtmánková, A.; Dvořák, P.; Čepl, J. Cultivar differences of total anthocyanins and anthocyanidins in red and purple-fleshed potatoes and their relation to antioxidant activity. Food Chem. 2009, 114, 836–843. [Google Scholar] [CrossRef]
- Qiu, G.; Wang, D.; Song, X.; Deng, Y.; Zhao, Y. Degradation kinetics and antioxidant capacity of anthocyanins in air-impingement jet dried purple potato slices. Food Res. Int. 2018, 105, 121–128. [Google Scholar] [CrossRef]
- Gong, S.; Yang, C.; Zhang, J.; Yu, Y.; Gu, X.; Li, W.; Wang, Z. Study on the interaction mechanism of purple potato anthocyanins with casein and whey protein. Food Hydrocoll. 2021, 111, 106223. [Google Scholar] [CrossRef]
- Jiang, Z.; Chen, C.; Wang, J.; Xie, W.; Wang, M.; Li, X.; Zhang, X. Purple potato (Solanum tuberosum L.) anthocyanins attenuate alcohol-induced hepatic injury by enhancing antioxidant defense. J. Nat. Med. 2016, 70, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Bielek, P. Soil Protection: Code of Good Agricultural Practice in the Slovak Republic; MARD SR—SSCRI: Bratislava, Slovakia, 1996; 54p. [Google Scholar]
- Lapornik, B.; Prošek, M.; Wondra, A.G. Comparison of extracts prepared from plant by-products using different solvents and extraction time. J. Food Eng. 2005, 71, 214–222. [Google Scholar] [CrossRef]
- Lachman, J.; Hamouz, K.; Čepl, J.; Pivec, V.; Šulc, M.; Dvořák, P. The Effect of Selected Factors on Polyphenol Content and Antioxidant Activity in Potato Tubers. Chem. Lett. 2006, 100, 522–527. [Google Scholar]
- Vollmannová, A.; Musilová, J.; Lidiková, J.; Árvay, J.; Šnirc, M.; Tóth, T.; Bojňanská, T.; Čičová, I.; Kreft, I.; Germ, M. Concentrations of phenolic acids are differently genetically determined in leaves, flowers, and grain of common buckwheat (Fagopyrum esculentum Moench). Plants 2021, 10, 1142. [Google Scholar] [CrossRef]
- The Jamovi Project. Jamovi (Version 2.2). [Computer Software]. 2021. Available online: https://www.jamovi.org (accessed on 1 April 2022).
- R Core Team 2021. R: A Language and Environment for Statistical Computing. (Version 4.0) [Computer Software]. Available online: https://cran.r-project.org (accessed on 1 April 2021).
- Wickham, H.; Chang, W.; Henry, L.; Pedersen, T.L.; Takahashi, K.; Wilke, C.; Woo, K.; RStudio. ggplot2: Create Elegant Data Visualisations Using the Grammar of Graphics. 2018. [R Package]. Available online: https://CRAN.R-project.org/package=ggplot2 (accessed on 4 April 2022).
- Patil, I. ggstatsplot: ‘ggplot2’ Based Plots with Statistical Details. 2018. [R Package]. Available online: https://CRAN.R-project.org/package=ggstatsplot (accessed on 4 April 2022).
- Brown, C.R. Antioxidants in potato. Am. J. Potato Res. 2005, 82, 163–172. [Google Scholar] [CrossRef]
- Lachman, J.; Hamouz, K.; Orsák, M.; Pivec, V.; Dvořák, P. The influence of flesh colour and growing locality on polyphenolic content and antioxidant activity in potatoes. Sci. Hortic. 2008, 117, 109–114. [Google Scholar] [CrossRef]
- Vaitkevičienė, N.; Kulaitienė, J.; Jarienė, E.; Levickienė, D.; Danillčenko, H.; Średnicka-Tober, D.; Rembiałkowska, E.; Hallmann, E. Characterization of Bioactive Compounds in Colored Potato (Solanum Tuberosum L.) Cultivars Grown with Conventional, Organic, and Biodynamic Methods. Sustainability 2020, 12, 2701. [Google Scholar] [CrossRef] [Green Version]
- Andre, C.M.; Ghislain, M.; Bertin, P.; Oufir, M.; Del Rosario Herrera, M.; Hoffmann, L.; Hausman, J.F.; Larondelle, Y.; Evers, D. Andean potato cultivars (Solanum tuberosum L.) as a source of antioxidant and mineral micronutrients. J. Agric. Food Chem. 2007, 55, 366–378. [Google Scholar] [CrossRef]
- Tierno, R.; Hornero-Méndez, D.; Gallardo-Guerrero, L.; López-Pardo, R.; De Galarreta, J.I.R. Effect of boiling on the total phenolic, anthocyanin and carotenoid concentrations of potato tubers from selected cultivars and introgressed breeding lines from native potato species. J. Food Compos. Anal. 2015, 41, 58–65. [Google Scholar] [CrossRef] [Green Version]
- Hamouz, K.; Lachman, J.; Pazderů, K.; Tomášek, J.; Hejtmánková, K.; Pivec, V. Differences in Anthocyanin Content and Antioxidant Activity of Potato Tubers with Different Flesh Colour. Plant Soil Environ. 2011, 57, 478–485. Available online: https://www.agriculturejournals.cz/publicFiles/265_2011-PSE.pdf (accessed on 11 April 2022). [CrossRef] [Green Version]
- Dalamu, S.B.; Shivali, B.; Pinky, R.; Reena, S.; Alka, J. Assessment of phytochemical diversity in Indian potato cultivars. Indian J. Hortic. 2015, 72, 447–450. [Google Scholar] [CrossRef]
- Lemos, M.A.; Aliyu, M.M.; Hungerford, G. Influence of cooking on the levels of bioactive compounds in Purple Majesty potato observed via chemical and spectroscopic means. Food Chem. 2015, 173, 462–467. [Google Scholar] [CrossRef]
- André, C.M.; Schafleitner, R.; Legay, S.; Lefèvre, I.; Aliaga, C.A.A.; Nomberto, G.; Hoffmann, L.; Hausman, J.F.; Larondelle, Y.; Evers, D. Gene expression changes related to the production of phenolic compounds in potato tubers grown under drought stress. Phytochemistry 2009, 70, 1107–1116. [Google Scholar] [CrossRef] [PubMed]
- Andre, C.M.; Oufir, M.; Hoffmann, L.; Hausman, J.F.; Rogez, H.; Larondelle, Y.; Eevers, D. Influence of environment and genotype on polyphenol compounds and in vitro antioxidant capacity of native Andean potatoes (Solanum tuberosum L.). J. Food Compos. Anal. 2009, 22, 517–524. [Google Scholar] [CrossRef]
- Šulc, M.; Kotíková, Z.; Paznocht, L.; Pivec, V.; Hamouz, K.; Lachman, J. Changes in anthocyanidin levels during the maturation of color-fleshed potato (Solanum tuberosum L.) tubers. Food Chem. 2017, 237, 981–988. [Google Scholar] [CrossRef]
- Payyavula, R.S.; Navarre, D.A.; Kuhl, J.; Pantoja, A. Developmental effects on phenolic, flavonol, anthocyanin, and carotenoid metabolites and gene expression in potatoes. J. Agric. Food Chem. 2013, 61, 7357–7365. [Google Scholar] [CrossRef]
- Reyes, L.F.; Miller, J.C.; Cisneros-Zevallos, L. Antioxidant capacity, anthocyanins and total phenolics in purple-and red-fleshed potato (Solanum tuberosum L.) genotypes. Am. J. Potato Res. 2005, 82, 271–277. [Google Scholar] [CrossRef]
- Minatel, I.O.; Borges, C.V.; Ferreira, M.I.; Gomez, H.A.G.; Lima, C.O.C.A.P.P. Phenolic Compounds: Functional Properties, Impact of Processing and Bioavailability. In Phenolic Compounds—Biological Activity, 1st ed.; Soto-Hernandez, M., Palma-Tenango, M., Garcia-Mateos, M.d.R., Eds.; IntechOpen: Rijeka, Croatia, 2017; pp. 1–24. [Google Scholar]
- Bertelli, A.; Biagi, M.; Corsini, M.; Baini, G.; Cappellucci, G.; Miraldi, E. Polyphenols: From Theory to Practice. Foods 2021, 10, 2595. [Google Scholar] [CrossRef]
- Kita, A.; Bąkowska-Barczak, A.; Hamouz, K.; Kułakowska, K.; Lisińska, G. The effect of frying on anthocyanin stability and antioxidant activity of crisps from red-and purple-fleshed potatoes (Solanum tuberosum L.). J. Food Compos. Anal. 2013, 32, 169–175. [Google Scholar] [CrossRef]
- Hamouz, K.; Lachman, J.; Hejtmánková, K.; Pazderů, K.; Čížek, M.; Dvořák, P. Effect of natural and growing conditions on the content of phenolics in potatoes with different flesh colour. Plant Soil Environ. 2010, 56, 368–374. Available online: https://www.agriculturejournals.cz/publicFiles/49_2010-PSE.pdf (accessed on 20 April 2022). [CrossRef]
- Ševčík, R.; Vasnička, F.; Jirušková, M.; Vacek, J.; Hamouz, K.; Voldřich, M.; Čížková, H.; Kondrashov, A.; Holasová, M.; Fiedlerová, V. Influence of potato variety and culinary treatment on their antioxidant capacity. Nutr. Food 2009, 6, 161–164. [Google Scholar]
- Ezekiel, R.; Singh, N.; Sharma, S.; Kaur, A. Beneficial phytochemicals in potato—A review. Food Res. Int. 2013, 50, 487–496. [Google Scholar] [CrossRef]
- Vokál, B.; Bárta, J.; Bártová, V.; Čepl, J.; Čížek, M.; Doležal, P.; Domkářová, J.; Dohanyos, M.; Faltus, M.; Greplová, M.; et al. Potatoes-Breeding-Cultivation-Use-Economy, 1st ed.; Kavka Print: Praha, Czech Republic, 2013; 160p. [Google Scholar]
- Ru, W.; Pang, Y.; Gan, Y.; Liu, Q.; Bao, J. Phenolic Compounds and Antioxidant Activities of Potato Cultivars with White, Yellow, Red and Purple Flesh. Antioxidants 2019, 8, 419. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deußer, H.; Guignard, C.; Hoffmann, L.; Evers, D. Polyphenol and glycoalkaloid contents in potato cultivars grown in Luxembourg. Food Chem. 2012, 135, 2814–2824. [Google Scholar] [CrossRef] [PubMed]
- Giusti, M.M.; Polit, M.F.; Ayvaz, H.; Tay, D.; Manrique, I. Characterization and Quantitation of Anthocyanins and Other Phenolics in Native Andean Potatoes. J. Agric. Food Chem. 2014, 62, 4408–4416. [Google Scholar] [CrossRef]
- Zarzecka, K.; Gugała, M.; Sikorska, A.; Mystkowska, I.; Baranowska, A.; Niewęgłowski, M.; Dołęga, H. The effect of herbicides and biostimulants on polyphenol content of potato (Solanum tuberosum L.) tubers and leaves. J. Saudi Soc. Agric. Sci. 2017, 18, 102–106. [Google Scholar] [CrossRef]
- Navarre, D.A.; Shakya, R.; Holden, J.; Kumar, S. The Effect of Different Cooking Methods on Phenolics and Vitamin C in Developmentally Young Potato Tubers. Am. J. Potato Res. 2010, 87, 350–359. [Google Scholar] [CrossRef]
- Brazinskiene, V.; Asakaviciute, R.; Razukas, A.; Ivanauskas, L. Quantification of biologically active compounds in the tubers of potato varieties of different maturity. Zemdirb. Agric. 2017, 104, 41–46. [Google Scholar] [CrossRef] [Green Version]
- Lachman, J.; Hamouz, K.; Musilová, J.; Hejtmánková, K.; Kotíková, Z.; Pazderů, K.; Domkářová, J.; Pivec, V.; Cimr, J. Effect of peeling and three cooking methods on the content of selected phytochemicals in potato tubers with various colour of flesh. Food Chem. 2013, 138, 1189–1197. [Google Scholar] [CrossRef]
- Hamouz, K.; Lachman, J.; Pazderů, K.; Hejtmánková, K.; Cimr, J.; Musilová, J.; Pivec, V.; Orsák, M.; Svobodová, A. Effect of cultivar, location and method of cultivation on the content of chlorogenic acid in potatoes with different flesh colour. Plant Soil Environ. 2013, 59, 465–471. [Google Scholar] [CrossRef] [Green Version]
- Moser, S.; Aragon, I.; Furrer, A.; Van Klinken, J.W.; Kaczmarczyk, M.; Lee, B.H.; Hamaker, B.; Ferruzzi, M. Potato Phenolics Modulate Rate of Glucose Transport in a Caco-2 Human Intestinal Cell Model. FASEB J. 2015, 29, 606.6. [Google Scholar] [CrossRef]

| Variety | Description | ||
|---|---|---|---|
| Maturity | Skin | Flesh | |
| Belana | Early to medium early | Pure yellow | Yellow |
| Cecile | Medium early to late | Red | Yellow |
| Magenta Love | Early | Pink | Pink |
| Mozart | Medium early | Red | Pale yellow |
| Talentine | Early | Light yellow | Yellow |
| Toscana | Medium early | Yellow | Deep yellow |
| Violet Queen | Early | Purple | Deep purple |
| Variety | Anthocyanins (mg CyE kg−1 FW ± SD) | |
|---|---|---|
| 1st Harvest July | 2nd Harvest September | |
| Belana | 45.7 ± 2.54 | 58.3 ± 10.35 |
| Cecile | 41.8 ± 5.27 | 58.4 ± 5.81 |
| Magenta Love | 91.6 ± 2.27 | 216 ± 20.3 |
| Mozart | 61.1 ± 5.58 | 89.0 ± 19.26 |
| Talentine | 52.2 ± 6.70 | 58.2 ± 7.60 |
| Toscana | 58.9 ± 8.41 | 53.4 ± 5.03 |
| Violet Queen | 116 ± 5.02 | 176 ± 4.25 |
| Variety | Polyphenols (mg GAE g−1 DW ± SD) | |
|---|---|---|
| 1st Harvest July | 2nd Harvest September | |
| Belana | 1.21 ± 0.10 | 6.72 ± 0.25 |
| Cecile | 2.47 ± 0.13 | 5.86 ± 0.17 |
| Magenta Love | 2.04 ± 0.37 | 27.1 ± 0.10 |
| Mozart | 1.14 ± 0.06 | 5.71 ± 0.05 |
| Talentine | 1.48 ± 0.21 | 5.35 ± 0.21 |
| Toscana | 1.29 ± 0.05 | 4.02 ± 0.02 |
| Violet Queen | 2.69 ± 0.10 | 33.4 ± 0.17 |
| Variety | CGA (mg kg−1 FW ± SD) | |
|---|---|---|
| 1st Harvest July | 2nd Harvest September | |
| Belana | 17.6 ± 1.07 | 18.0 ± 0.71 |
| Cecile | 46.7 ± 2.84 | 25.1 ± 0.65 |
| Magenta Love | 294 ± 17.92 | 309 ± 3.60 |
| Mozart | 16.2 ± 0.99 | 11.5 ± 0.61 |
| Talentine | 68.3 ± 4.16 | 27.4 ± 0.31 |
| Toscana | 19.5 ± 1.19 | 8.70 ± 0.20 |
| Violet Queen | 277 ± 19.87 | 130 ± 0.69 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Franková, H.; Musilová, J.; Árvay, J.; Harangozo, Ľ.; Šnirc, M.; Vollmannová, A.; Lidiková, J.; Hegedűsová, A.; Jaško, E. Variability of Bioactive Substances in Potatoes (Solanum Tuberosum L.) Depending on Variety and Maturity. Agronomy 2022, 12, 1454. https://doi.org/10.3390/agronomy12061454
Franková H, Musilová J, Árvay J, Harangozo Ľ, Šnirc M, Vollmannová A, Lidiková J, Hegedűsová A, Jaško E. Variability of Bioactive Substances in Potatoes (Solanum Tuberosum L.) Depending on Variety and Maturity. Agronomy. 2022; 12(6):1454. https://doi.org/10.3390/agronomy12061454
Chicago/Turabian StyleFranková, Hana, Janette Musilová, Július Árvay, Ľuboš Harangozo, Marek Šnirc, Alena Vollmannová, Judita Lidiková, Alžbeta Hegedűsová, and Edina Jaško. 2022. "Variability of Bioactive Substances in Potatoes (Solanum Tuberosum L.) Depending on Variety and Maturity" Agronomy 12, no. 6: 1454. https://doi.org/10.3390/agronomy12061454
APA StyleFranková, H., Musilová, J., Árvay, J., Harangozo, Ľ., Šnirc, M., Vollmannová, A., Lidiková, J., Hegedűsová, A., & Jaško, E. (2022). Variability of Bioactive Substances in Potatoes (Solanum Tuberosum L.) Depending on Variety and Maturity. Agronomy, 12(6), 1454. https://doi.org/10.3390/agronomy12061454








