Flux of Root-Derived Carbon into the Nematode Micro-Food Web: A Comparison of Grassland and Agroforest
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Site
2.2. Experimental Design and Soil Properties
2.3. Pulse Labeling with 13CO2
2.4. Plant, Soil and Nematode Sampling
2.5. Stable Isotope Analysis
2.5.1. Plant and Soil
2.5.2. Nematodes
2.6. Statistical Analysis
3. Results
3.1. Nematode Density and Trophic Structure
3.2. The Nematode Micro-Food Web
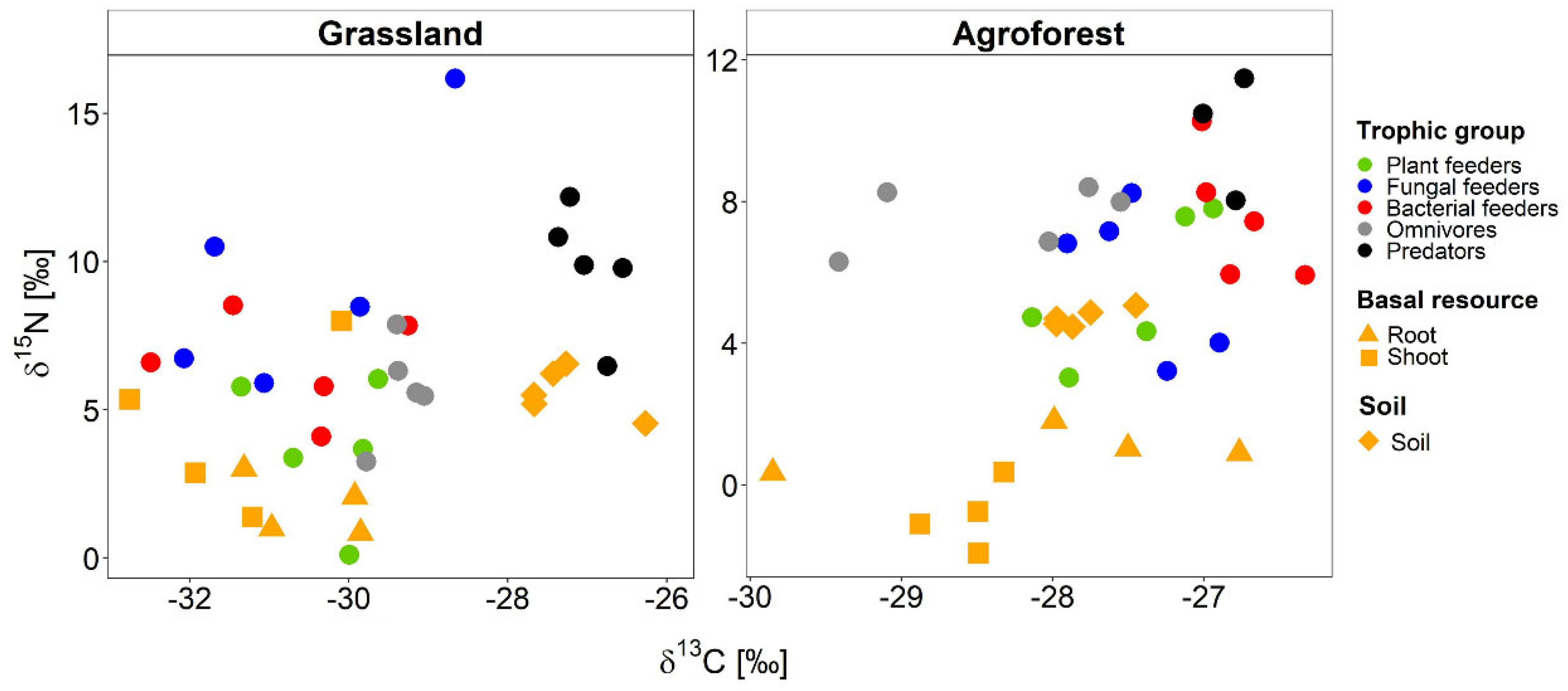
3.2.1. Natural Stable Isotope Ratios of Soil and Basal Food Web Resources
3.2.2. Natural Stable Isotope Ratios of Nematode Trophic Groups
3.2.3. Isotopic Fractionation across Trophic Levels
3.3. Pulse-Labeling with 13CO2
3.3.1. Incorporation of 13C into Soil and Basal Food Web Resources
3.3.2. Incorporation of 13C into Nematode Trophic Groups
4. Discussion
4.1. Land Use Type Affects The Dominant Soil Carbon and Energy Channel
4.2. The Plant Type Affects Predatory Feeding in Higher Trophic Levels
4.3. Plant C Flux Is Strongest and Fastest into Basal Trophic Levels
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Scharlemann, J.P.; Tanner, E.V.; Hiederer, R.; Kapos, V. Global soil carbon: Understanding and managing the largest terrestrial carbon pool. Carbon Manag. 2014, 5, 81–91. [Google Scholar] [CrossRef]
- Pausch, J.; Kuzyakov, Y. Carbon input by roots into the soil: Quantification of rhizodeposition from root to ecosystem scale. Glob. Chang. Biol. 2018, 24, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Neumann, G.; Romheld, V. The release of root exudates as affected by the plant’s physiological status. In The Rhizosphere; Pinton, R., Varanini, Z., Nannipieri, P., Eds.; CRC Press: Boca Raton, FL, USA, 2007; pp. 23–72. [Google Scholar]
- Dennis, P.G.; Miller, A.J.; Hirsch, P.R. Are root exudates more important than other sources of rhizodeposits in structuring rhizosphere bacterial communities? FEMS Microbiol. Ecol. 2010, 72, 313–327. [Google Scholar] [CrossRef] [PubMed]
- Kurganova, I.N.; Lopes de Gerenyu, V.O.; Shvidenko, A.Z.; Sapozhnikov, P.M. Changes in the organic carbon pool of abandoned soils in Russia (1990–2004). Eurasian Soil Sci. 2010, 43, 333–340. [Google Scholar] [CrossRef]
- Mao, R.; Zeng, D.H.; Hu, Y.L.; Li, L.J.; Yang, D. Soil organic carbon and nitrogen stocks in an age-sequence of poplar stands planted on marginal agricultural land in Northeast China. Plant Soil 2010, 332, 277–287. [Google Scholar] [CrossRef]
- Nave, L.E.; Swanston, C.W.; Mishra, U.; Nadelhoffer, K.J. Afforestation effects on soil carbon storage in the United States: A synthesis. Soil Sci. Soc. Am. J. 2013, 77, 1035–1047. [Google Scholar] [CrossRef]
- Houghton, R.A.; Goodale, C.L. Effects of land-use change on the carbon balance of terrestrial ecosystems. Ecosyst. Land Use Chang. 2004, 153, 85–98. [Google Scholar]
- DuPont, S.T.; Culman, S.W.; Ferris, H.; Buckley, D.H.; Glover, J.D. No-tillage conversion of harvested perennial grassland to annual cropland reduces root biomass, decreases active carbon stocks, and impacts soil biota. Agric. Ecosyst. Environ. 2010, 137, 25–32. [Google Scholar] [CrossRef]
- Kuzyakov, Y.; Domanski, G. Carbon input by plants into the soil. J. Plant Nutr. Soil Sci. 2000, 163, 421–431. [Google Scholar] [CrossRef]
- Keel, S.G.; Schädel, C. Expanding leaves of mature deciduous forest trees rapidly become autotrophic. Tree Physiol. 2010, 30, 1253–1259. [Google Scholar] [CrossRef]
- Barbaroux, C.; Bréda, N.; Dufrêne, E. Distribution of above-ground and below-ground carbohydrate reserves in adult trees of two contrasting broad-leaved species (Quercus petraea and Fagus sylvatica). New Phytol. 2013, 157, 605–615. [Google Scholar] [CrossRef] [PubMed]
- Canarini, A.; Kaiser, C.; Merchant, A.; Richter, A.; Wanek, W. Root exudation of primary metabolites: Mechanisms and their roles in plant responses to environmental stimuli. Front. Plant Sci. 2019, 10, 157. [Google Scholar] [CrossRef] [PubMed]
- Markwitz, C.; Knohl, A.; Siebicke, L. Evapotranspiration over agroforestry sites in Germany. Biogeosciences 2020, 17, 5183–5208. [Google Scholar] [CrossRef]
- Hauk, S.; Wittkopf, S.; Knoke, T. Analysis of commercial short rotation coppices in Bavaria, southern Germany. Biomass Bioenergy 2014, 67, 401–412. [Google Scholar] [CrossRef]
- Aylott, M.J.; Casella, E.; Tubby, I.; Street, N.R.; Smith, P.; Taylor, G. Yield and spatial supply of bioenergy poplar and willow short-rotation coppice in the UK. New Phytol. 2008, 178, 358–370. [Google Scholar] [CrossRef]
- Kahle, P.; Baum, C.; Boelcke, B.; Kohl, J.; Ulrich, R. Vertical distribution of soil properties under short-rotation forestry in Northern Germany. J. Plant Nutr. Soil Sci. 2010, 173, 737–746. [Google Scholar] [CrossRef]
- Börjesson, P.; Tufvesson, L.M. Agricultural crop-based biofuels–resource efficiency and environmental performance including direct land use changes. J. Clean. Prod. 2011, 19, 108–120. [Google Scholar] [CrossRef]
- Gissén, C.; Prade, T.; Kreuger, E.; Nges, I.A.; Rosenqvist, H.; Svensson, S.E.; Björnsson, L. Comparing energy crops for biogas production–Yields, energy input and costs in cultivation using digestate and mineral fertilisation. Biomass Bioenergy 2014, 64, 199–210. [Google Scholar] [CrossRef]
- Tsonkova, P.; Böhm, C.; Quinkenstein, A.; Freese, D. Ecological benefits provided by alley cropping systems for production of woody biomass in the temperate region: A review. Agrofor. Syst. 2012, 85, 133–152. [Google Scholar] [CrossRef]
- Zhao, J.; Neher, D.A. Soil energy pathways of different ecosystems using nematode trophic group analysis: A meta analysis. Nematology 2014, 16, 379–385. [Google Scholar] [CrossRef]
- Van Den Hoogen, J.; Geisen, S.; Routh, D.; Ferris, H.; Traunspurger, W.; Wardle, D.A.; Crowther, T.W. Soil nematode abundance and functional group composition at a global scale. Nature 2019, 572, 194–198. [Google Scholar] [CrossRef] [PubMed]
- Čerevková, A. Nematode communities in three types of grassland in the Slovak Republic. Helminthologia 2006, 43, 171–176. [Google Scholar] [CrossRef]
- Háněl, L. Succession of soil nematodes in pine forests on coal-mining sands near Cottbus, Germany. Appl. Soil Ecol. 2001, 16, 23–34. [Google Scholar] [CrossRef]
- Crotty, F.; Blackshaw, R.; Murray, P. Tracking the flow of bacterially derived 13C and 15N through soil faunal feeding channels. Rapid Commun. Mass Spectrom. 2011, 25, 1503–1513. [Google Scholar] [CrossRef]
- Moore, J.C.; McCann, K.; Setälä, H.; De Ruiter, P.C. Top-down is bottom-up: Does predation in the rhizosphere regulate aboveground dynamics? Ecology 2003, 84, 846–857. [Google Scholar] [CrossRef]
- Ferris, H.; Matute, M. Structural and functional succession in the nematode fauna of a soil food web. Appl. Soil Ecol. 2003, 23, 93–110. [Google Scholar] [CrossRef]
- Scheu, S.; Ruess, L.; Bonkowski, M. Interactions between microorganisms and soil micro-and mesofauna. In Microorganisms in Soils: Roles in Genesis and Functions; Buscot, F., Varma, A., Eds.; Springer: Berlin/Heidelberg, Germany, 2005; pp. 253–275. [Google Scholar]
- Yeates, G.W.; Bongers, T. Nematode diversity in agroecosystems. Agric. Ecosyst. Environ. 1999, 74, 113–135. [Google Scholar] [CrossRef]
- Griffiths, B. A comparison of microbial-feeding nematodes and protozoa in the rhizosphere of different plants. Biol. Fertil. Soils 1990, 9, 83–88. [Google Scholar] [CrossRef]
- Gebremikael, M.T.; Steel, H.; Buchan, D.; Bert, W.; De Neve, S. Nematodes enhance plant growth and nutrient uptake under C and N-rich conditions. Sci. Rep. 2016, 6, 1032862. [Google Scholar] [CrossRef]
- Bongers, T.; Bongers, M. Functional diversity of nematodes. Appl. Soil Ecol. 1998, 10, 239–251. [Google Scholar] [CrossRef]
- Ferris, H.; Bongers, T.; De Goede, R. A framework for soil food web diagnostics: Extension of the nematode faunal analysis concept. Appl. Soil Ecol. 2001, 18, 13–29. [Google Scholar] [CrossRef]
- Hu, N.; Li, H.; Tang, Z.; Li, Z.; Tian, J.; Lou, Y.; Li, J.; Li, G.; Hu, X. Community diversity, structure and carbon footprint of nematode food web following reforestation on degraded Karst soil. Sci. Rep. 2016, 6, 28138. [Google Scholar] [CrossRef]
- Hartmann, L.; Richter, F.; Busch, G.; Ehret, M.; Jansen, M.; Lamersdorf, N. Establishment of short rotation coppices in the South of Lower Saxony and in Central Thuringia in the context of the BEST-research framework-site characteristics and initial biomass production. Forstarchiv 2014, 85, 134–150. [Google Scholar]
- Hartmann, L.; Lamersdorf, N. Site conditions, initial growth and nutrient and litter cycling of newly installed short rotation coppice and agroforestry systems. In Bioenergy from Dendromass for the Sustainable Development of Rural Areas; Manning, D.B., Bemmann, A., Bredemeier, M., Lamersdorf, N., Ammer, C., Eds.; Wiley-VHC Verlag GmbH & Co. KGaA: Weinheim, Germany, 2015; pp. 121–138. [Google Scholar]
- Tariq, A.; Gunina, A.; Lamersdorf, N. Initial changes in soil properties and carbon sequestration potential under monocultures and short-rotation alley coppices with poplar and willow after three years of plantation. Sci. Total Environ. 2018, 634, 963–973. [Google Scholar] [CrossRef] [PubMed]
- Ehret, M.; Bühle, L.; Graß, R.; Lamersdorf, N.; Wachendorf, M. Bioenergy provision by an alley cropping system of grassland and shrub willow hybrids: Biomass, fuel characteristics and net energy yields. Agrofor. Syst. 2015, 89, 365–381. [Google Scholar] [CrossRef]
- Walter, K.; Don, A.; Flessa, H. Net N2O and CH4 soil fluxes of annual and perennial bioenergy crops in two central German regions. Biomass Bioenergy 2015, 81, 556–567. [Google Scholar] [CrossRef]
- Ruess, L. Studies on the nematode fauna of an acid forest soil: Spatial distribution and extraction. Nematologica 1995, 41, 229–239. [Google Scholar] [CrossRef]
- Bongers, T. De Nematoden van Nederland; Stichting Uitgeverij van de Koninklijke Natuurhistorische Vereniging: Utrecht, The Netherlands, 1994; pp. 1–408. [Google Scholar]
- Yeates, G.W.; Bongers, T.D.; De Goede, R.G.M.; Freckman, D.; Georgieva, S. Feeding habits in soil nematode families and genera–an outline for soil ecologists. J. Nematol. 1993, 25, 315–331. [Google Scholar]
- Langel, R.; Dyckmans, J. Combined 13C and 15N isotope analysis on small samples using a near-conventional elemental analyzer/isotope ratio mass spectrometer setup. Rapid Commun. Mass Spectrom. 2014, 28, 1019–1022. [Google Scholar] [CrossRef]
- Bardgett, R.D.; Cook, R. Functional aspects of soil animal diversity in agricultural grasslands. Appl. Soil Ecol. 1998, 10, 263–276. [Google Scholar] [CrossRef]
- Renčo, M. The soil nematodes in natural and semi-natural grasslands and their use as bioindicators. In Grasslands: Types, Biodiversity and Impacts; Zhang, W., Ed.; Nova Science Publishers, Inc.: New York, NY, USA, 2012; pp. 119–145. [Google Scholar]
- Viketoft, M.; Sohlenius, B. Soil nematode populations in a grassland plant diversity experiment run for seven years. Appl. Soil Ecol. 2011, 48, 174–184. [Google Scholar] [CrossRef]
- Chen, D.; Pan, Q.; Bai, Y.; Hu, S.; Huang, J.; Wang, Q.; Han, X. Effects of plant functional group loss on soil biota and net ecosystem exchange: A plant removal experiment in the Mongolian grassland. J. Ecol. 2016, 104, 734–743. [Google Scholar] [CrossRef]
- McSorley, R. Effect of disturbances on trophic groups in soil nematode assemblages. Nematology 2011, 13, 553–559. [Google Scholar] [CrossRef]
- Viketoft, M.; Sohlenius, B.; Boström, S.; Palmborg, C.; Bengtsson, J.; Berg, M.P.; Huss Danell, K. Temporal dynamics of soil nematode communities in a grassland plant diversity experiment. Soil Biol. Biochem. 2011, 43, 1063–1070. [Google Scholar] [CrossRef]
- Wasilewska, L. The effect of age of meadows on succession and diversity in soil nematode communities. Pedobiologia 1994, 38, 1–11. [Google Scholar]
- Ruess, L. Nematode soil faunal analysis of decomposition pathways in different ecosystems. Nematology 2003, 5, 179–181. [Google Scholar] [CrossRef]
- Van Der Heijden, M.G.; Bardgett, R.D.; Van Straalen, N.M. The unseen majority: Soil microbes as drivers of plant diversity and productivity in terrestrial ecosystems. Ecol. Lett. 2008, 11, 296–310. [Google Scholar] [CrossRef]
- Stauffer, M.; Leyval, C.; Brun, J.-J.; Leportier, P.; Berthelin, J. Effect of willow short rotation coppice on soil properties after three years of growth as compared to forest, grassland and arable land uses. Plant Soil 2014, 377, 423–438. [Google Scholar] [CrossRef][Green Version]
- Háněl, L. An outline of soil nematode succession on abandoned fields in South Bohemia. Appl. Soil Ecol. 2010, 46, 355–371. [Google Scholar] [CrossRef]
- Kitagami, Y.; Torii, M.; Matsuda, Y. Characterizations of community and trophic structures of soil nematodes in a coastal Japanese black pine forest. Nematol. Res. Jpn. J. Nematol. 2016, 46, 71–78. [Google Scholar] [CrossRef]
- Yeates, G.W. Abundance, diversity, and resilience of nematode assemblages in forest soils. Can. J. For. Res. 2007, 37, 216–225. [Google Scholar] [CrossRef]
- Zhang, X.; Guan, P.; Wang, Y.; Li, Q.; Zhang, S.; Zhang, Z.; Liang, W. Community composition, diversity and metabolic footprints of soil nematodes in differently-aged temperate forests. Soil Biol. Biochem. 2015, 80, 118–126. [Google Scholar] [CrossRef]
- Ruess, L.; Ferris, H. Decomposition pathways and successional changes. Nematol. Monogr. Perspect. 2004, 2, 547–556. [Google Scholar]
- Klarner, B.; Ehnes, R.B.; Erdmann, G.; Eitzinger, B.; Pollierer, M.M.; Maraun, M.; Scheu, S. Trophic shift of soil animal species with forest type as indicated by stable isotope analysis. Oikos 2014, 123, 1173–1181. [Google Scholar] [CrossRef]
- Scheunemann, N.; Digel, C.; Scheu, S.; Butenschoen, O. Roots rather than shoot residues drive soil arthropod communities of arable fields. Oecologia 2015, 179, 1135–1145. [Google Scholar] [CrossRef] [PubMed]
- Furze, M.E.; Huggett, B.A.; Aubrecht, D.M.; Stolz, C.D.; Carbone, M.S.; Richardson, A.D. Whole-tree nonstructural carbohydrate storage and seasonal dynamics in five temperate species. New Phytol. 2019, 221, 1466–1477. [Google Scholar] [CrossRef]
- Li, Z.; Scheunemann, N.; Potapov, A.M.; Shi, L.; Pausch, J.; Scheu, S.; Pollierer, M.M. Incorporation of root-derived carbon into soil microarthropods varies between cropping systems. Biol. Fertil. Soils 2020, 56, 839–851. [Google Scholar] [CrossRef]
- Albers, D.; Schaefer, M.; Scheu, S. Incorporation of plant carbon into the soil animal food web of an arable system. Ecology 2006, 87, 235–245. [Google Scholar] [CrossRef]
- Middelburg, J. Stable isotopes dissect aquatic food webs from the top to the bottom. Biogeosciences 2014, 11, 2357–2371. [Google Scholar] [CrossRef]
- Wood, F.H. Nematode feeding relationships: Feeding relationships of soil-dwelling nematodes. Soil Biol. Biochem. 1973, 5, 593–601. [Google Scholar] [CrossRef]
- Curry, J.P. The invertebrate fauna of grassland and its influence on productivity. 1. The composition of the fauna. Grass Forage Sci. 1987, 42, 103–120. [Google Scholar] [CrossRef]
- Murray, P.J.; Cook, R.; Currie, A.F.; Dawson, L.A.; Gange, A.C.; Grayston, S.J.; Treonis, M. Interactions between fertilizer addition, plants and the soil environment: Implications for soil faunal structure and diversity. Appl. Soil Ecol. 2006, 33, 199–207. [Google Scholar] [CrossRef]
- Kudrin, A.A.; Tsurikov, S.M.; Tiunov, A.V. Trophic position of microbivorous and predatory soil nematodes in a boreal forest as indicated by stable isotope analysis. Soil Biol. Biochem. 2015, 86, 193–200. [Google Scholar] [CrossRef]
- McSorley, R. Ecology of the dorylaimid omnivore genera Aporcelaimellus, Eudorylaimus and Mesodorylaimus. Nematology 2012, 14, 645–663. [Google Scholar] [CrossRef]
- Ngosong, C.; Buse, T.; Ewald, M.; Richter, A.; Glaser, K.; Schöning, I.; Ruess, L. Influence of management intensity and environmental conditions on microbiota in biological soil crust and crust-free soil habitats of temperate forests. Soil Biol. Biochem. 2020, 144, 107761. [Google Scholar] [CrossRef]
- France, R. Carbon-13 enrichment in benthic compared to planktonic algae: Foodweb implications. Mar. Ecol. Prog. Ser. 1995, 124, 307–312. [Google Scholar] [CrossRef]
- Croll, N.A.; de Soyza, K. Comparative calorie values of nematodes. J. Nematol. 1980, 12, 132–135. [Google Scholar]
- Yeates, G. Nematoda of a Danish beech forest. I. Methods and general analysis. Oikos 1972, 23, 178–189. [Google Scholar] [CrossRef]
- Pausch, J.; Kramer, S.; Scharroba, A.; Scheunemann, N.; Butenschoen, O.; Kandeler, E.; Marhan, S.; Riederer, M.; Scheu, S.; Kuzyakov, Y.; et al. Small but active–pool size does not matter for carbon incorporation in below-ground food webs. Funct. Ecol. 2016, 30, 479–489. [Google Scholar] [CrossRef]
- Pausch, J.; Hofmann, S.; Scharroba, A.; Kuzyakov, Y.; Ruess, L. Fluxes of root-derived carbon into the nematode micro-food web of an arable soil. Food Webs 2016, 9, 32–38. [Google Scholar] [CrossRef]
- Glavatska, O.; Müller, K.; Butenschoen, O.; Schmalwasser, A.; Kandeler, E.; Scheu, S.; Totsche, K.U.; Ruess, L. Disentangling the root-and detritus-based food chain in the micro-food web of an arable soil by plant removal. PLoS ONE 2017, 12, e0180264. [Google Scholar] [CrossRef] [PubMed]
- Maboreke, H.R.; Feldhahn, L.; Bönn, M.; Tarkka, M.T.; Buscot, F.; Herrmann, S.; Ruess, L. Transcriptome analysis in oak uncovers a strong impact of endogenous rhythmic growth on the interaction with plant-parasitic nematodes. BMC Genom. 2016, 17, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Háněl, L. Soil nematodes in five spruce forests on the Beskydy mountains, Czech Republic. Fundam. Appl. Nematol. 1996, 19, 15–24. [Google Scholar]
- Kuzyakov, Y. Priming effects: Interactions between living and dead organic matter. Soil Biol. Biochem. 2010, 42, 1363–1371. [Google Scholar] [CrossRef]
- Kuzyakov, Y.; Gavrichkova, O. Time lag between photosynthesis and carbon dioxide efflux from soil: A review of mechanisms and controls. Glob. Chang. Biol. 2010, 16, 3386–3406. [Google Scholar] [CrossRef]
- Culman, S.W.; DuPont, S.T.; Glover, J.D.; Buckley, D.H.; Fick, G.W.; Ferris, H.; Crews, T.E. Long-term impacts of high-input annual cropping and unfertilized perennial grass production on soil properties and belowground food webs in Kansas, USA. Agric. Ecosyst. Environ. 2010, 137, 13–24. [Google Scholar] [CrossRef]
- Ostle, N.; Whiteley, A.S.; Bailey, M.J.; Sleep, D.; Ineson, P.; Manefield, M. Active microbial RNA turnover in a grassland soil estimated using a 13CO2 spike. Soil Biol. Biochem. 2003, 35, 877–885. [Google Scholar] [CrossRef]
- Bakhtiar, Y.; Miller, D.; Cavagnaro, T.; Smith, S. Interactions between two arbuscular mycorrhizal fungi and fungivorous nematodes and control of the nematode with fenamifos. Appl. Soil Ecol. 2001, 17, 107–117. [Google Scholar] [CrossRef]
- Hua, J.; Jiang, Q.; Bai, J.; Ding, F.; Lin, X.; Yin, Y. Interactions between arbuscular mycorrhizal fungi and fungivorous nematodes on the growth and arsenic uptake of tobacco in arsenic-contaminated soils. Appl. Soil Ecol. 2014, 84, 176–184. [Google Scholar] [CrossRef]
- Strickland, M.S.; Wickings, K.; Bradford, M.A. The fate of glucose, a low molecular weight compound of root exudates, in the belowground foodweb of forests and pastures. Soil Biol. Biochem. 2012, 49, 23–29. [Google Scholar] [CrossRef]
- Grigulis, K.; Lavorel, S.; Krainer, U.; Legay, N.; Baxendale, C.; Dumont, M.; Clément, J. Relative contributions of plant traits and soil microbial properties to mountain grassland ecosystem services. J. Ecol. 2013, 101, 47–57. [Google Scholar] [CrossRef]


| Grassland | Agroforest | |
|---|---|---|
| Population density (Ind. g−1) | 21.3 ± 8.0 | 19.5 ± 8.8 |
| Trophic groups (%) | ||
| Plant feeders | 47.7 ± 11.8 | 40.0 ± 16.0 |
| Fungal feeders | 12.4 ± 4.7 | 26.1 ± 14.7 |
| Bacterial feeders | 30.6 ± 9.7 | 26.8 ± 10.3 |
| Omnivores | 6.5 ± 4.4 | 5.4 ± 3.9 |
| Predators | 2.8 ± 2.6 | 1.6 ± 1.5 |
| f/b ratio | 0.4 ± 0.2 | 1.3 ± 1.0 |
| Land Use Type | Resource | Plant Feeders | Fungal Feeders | Bacterial Feeders | Omnivores | Predators |
|---|---|---|---|---|---|---|
| ∆13C | ||||||
| Grassland | Plant material | |||||
| root | 0.2 ± 0.7 | −0.1 ± 1.4 | −0.3 ± 1.2 | 1.2 ± 0.3 | 3.5 ± 0.3 | |
| Nematodes | ||||||
| Plant feeders | 0.9 ± 0.5 | 3.3 ± 0.9 | ||||
| Fungal feeders | 1.3 ± 1.3 | 3.7 ± 1.6 | ||||
| Bacterial feeders | 1.4 ± 1.3 | 3.8 ± 0.9 | ||||
| Agroforest | Plant material | |||||
| root | 0.5 ± 0.5 | 0.6 ± 0.4 | 1.3 ± 0.3 | −0.3 ± 0.8 | 1.1 ± 0.3 | |
| Nematodes | ||||||
| Plant feeders | −0.9 ± 1.0 | 0.5 ± 0.4 | ||||
| Fungal feeders | −0.9 ± 0.5 | 0.5 ± 0.3 | ||||
| Bacterial feeders | −1.6 ± 0.6 | −0.2 ± 0.3 | ||||
| ∆15N | ||||||
| Grassland | Plant material | |||||
| root | 2.1 ± 2.4 | 7.8 ± 4.1 | 4.8 ± 1.7 | 4.0 ± 1.7 | 8.1 ± 2.1 | |
| Nematodes | ||||||
| Plant feeders | 1.9 ± 3.4 | 6.0 ± 3.8 | ||||
| Fungal feeders | 3.2 ± 3.2 | 0.3 ± 4.5 | ||||
| Bacterial feeders | −0.9 ± 2.5 | 3.3 ± 1.0 | ||||
| Agroforest | Plant material | |||||
| root | 4.5 ± 2.1 | 4.9 ± 2.2 | 6.5 ± 1.6 | 6.5 ± 0.9 | 9.0 ± 1.8 | |
| Nematodes | ||||||
| Plant feeders | 2.1 ± 2.6 | 4.4 ± 1.1 | ||||
| Fungal feeders | 1.7 ± 2.8 | 4.8 ± 3.1 | ||||
| Bacterial feeders | 0.0 ± 2.2 | 3.6 ± 1.8 | ||||
| Land Use Type | Factor | Sum of Square | Mean of Square | df1 | df2 | F Value | p Value |
|---|---|---|---|---|---|---|---|
| Grassland | res | 175.04 | 87.70 | 2 | 36 | 106.89 | <0.001 |
| Day | 2.78 | 0.93 | 3 | 36 | 1.13 | 0.35 | |
| res × day | 17.41 | 2.90 | 6 | 36 | 3.54 | 0.01 | |
| Agroforest | res | 161.63 | 80.81 | 2 | 29.20 | 214.55 | <0.001 |
| Day | 0.53 | 0.18 | 3 | 29.26 | 0.47 | 0.71 | |
| res × day | 8.78 | 1.46 | 6 | 29.22 | 3.88 | 0.01 |
| Day | Grass | Willow | |
|---|---|---|---|
| Soil | 3 | 1.1 ± 0.3 | 5.6 ± 0.2 |
| 7 | 1.8 ± 0.9 | 1.4 ± 0.7 | |
| 14 | 1.3 ± 0.5 | 1.2 ± 0.2 | |
| 28 | 1.2 ± 0.2 | 1.3 ± 0.1 | |
| Root | 3 | 5.0 ± 0.6 | 1.0 ± 1.4 |
| 7 | 5.0 ± 0.6 | 0.7 ± 1.3 | |
| 14 | 4.9 ± 1.0 | 2.5 ± 0.6 | |
| 28 | 5.7 ± 0.6 | 2.1 ± 1.1 | |
| Shoot | 3 | 6.6 ± 0.3 | 5.9 ± 0.5 a |
| 7 | 4.0 ± 2.5 | 5.8 ± 0.1 ab | |
| 14 | 6.4 ± 0.3 | 5.3 ± 0.3 ab | |
| 28 | 5.6 ± 0.2 | 5.0 ± 0.5 b |
| Land Use Type | Factor | Sum of Square | Mean of Square | df1 | df2 | F Value | p Value |
|---|---|---|---|---|---|---|---|
| Grassland | tg | 69.08 | 17.27 | 4 | 75.10 | 22.51 | <0.001 |
| Day | 16.39 | 5.46 | 3 | 75.10 | 7.12 | <0.001 | |
| tg × day | 5.04 | 0.42 | 12 | 75.09 | 0.55 | 0.08 | |
| Agroforest | tg | 3.01 | 0.75 | 4 | 69.01 | 1.15 | 0.34 |
| Day | 0.93 | 0.31 | 3 | 69.10 | 0.48 | 0.70 | |
| tg × day | 6.67 | 0.56 | 12 | 69.04 | 0.85 | 0.60 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hemmerling, C.; Li, Z.; Shi, L.; Pausch, J.; Ruess, L. Flux of Root-Derived Carbon into the Nematode Micro-Food Web: A Comparison of Grassland and Agroforest. Agronomy 2022, 12, 976. https://doi.org/10.3390/agronomy12040976
Hemmerling C, Li Z, Shi L, Pausch J, Ruess L. Flux of Root-Derived Carbon into the Nematode Micro-Food Web: A Comparison of Grassland and Agroforest. Agronomy. 2022; 12(4):976. https://doi.org/10.3390/agronomy12040976
Chicago/Turabian StyleHemmerling, Christin, Zhipeng Li, Lingling Shi, Johanna Pausch, and Liliane Ruess. 2022. "Flux of Root-Derived Carbon into the Nematode Micro-Food Web: A Comparison of Grassland and Agroforest" Agronomy 12, no. 4: 976. https://doi.org/10.3390/agronomy12040976
APA StyleHemmerling, C., Li, Z., Shi, L., Pausch, J., & Ruess, L. (2022). Flux of Root-Derived Carbon into the Nematode Micro-Food Web: A Comparison of Grassland and Agroforest. Agronomy, 12(4), 976. https://doi.org/10.3390/agronomy12040976






