Milk Thistle (Silybum Marianum L.) as a Novel Multipurpose Crop for Agriculture in Marginal Environments: A Review
Abstract
1. Introduction
2. Origin and Distribution
3. Genetics and Breeding
4. Description
4.1. Morphology
4.2. Biology and Physiology
5. Management Techniques
5.1. Adaptation
5.2. Cropping System
5.3. Soil Management
5.4. Fertilization
5.5. Irrigation
5.6. Harvest and Seed Yields
5.7. Weed, Pest, and Disease Management
6. Qualitative Characteristics of Seeds
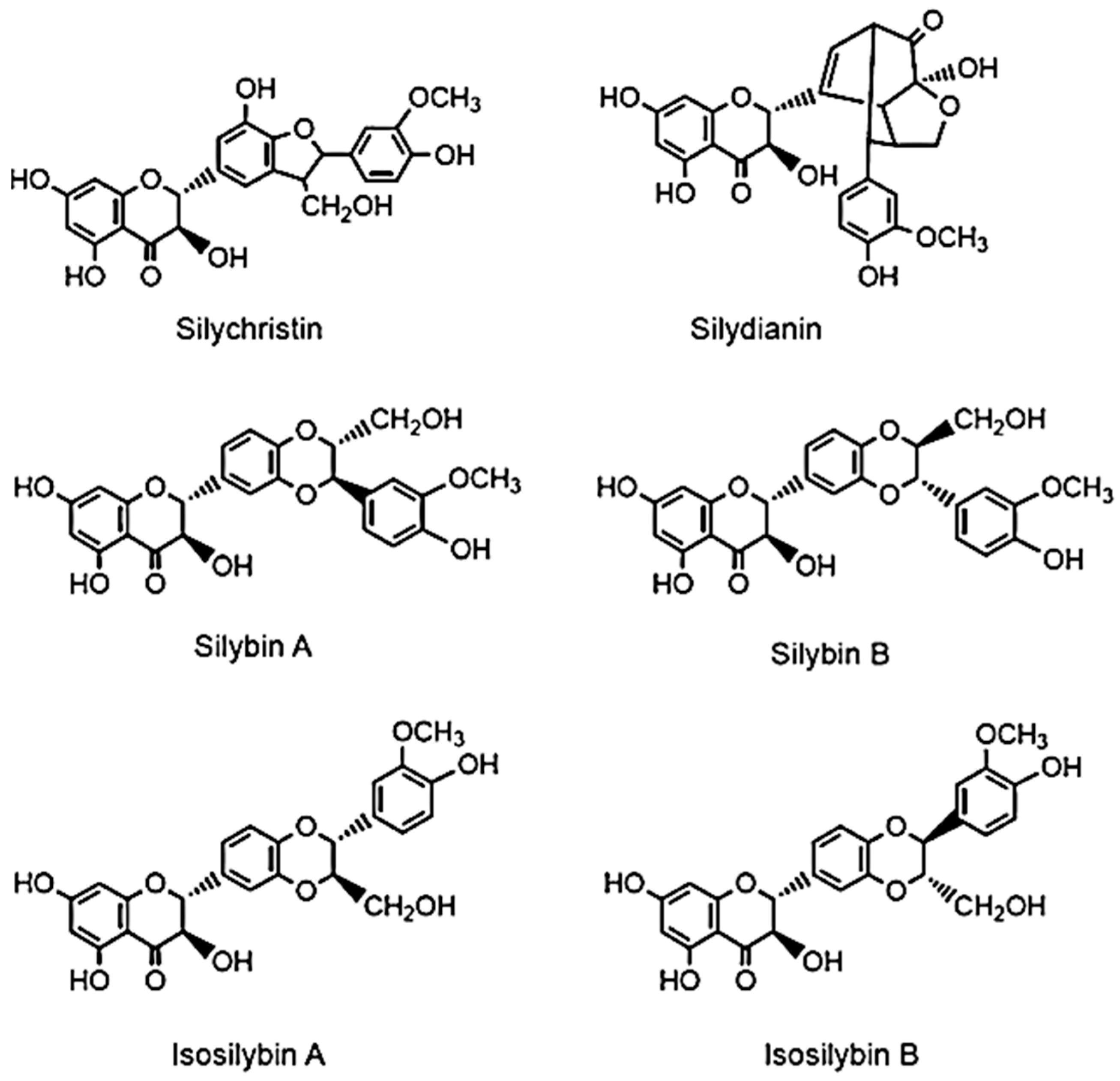
6.1. Silymarin
6.2. Biosynthesis and Accumulation of Silymarin
6.3. Extraction Methods
6.4. Biotechnological Production
7. Utilizations
7.1. Livestock Feeding
7.2. Food Uses
7.3. Industrial and Other Non-Medical Applications
7.3.1. Cosmetic Industry and Bioenergy Production
7.3.2. Biocidal Activity
7.3.3. Phytoremediation
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pignatti, S. Flora d’Italia; Edagricole: Bologna, Italia, 1982; Volume 3, p. 163. [Google Scholar]
- Young, J.A.; Evans, R.A.; Hawkes, R.B. Milk thistle (Silybum marianum) seed germination. Weed Sci. 1978, 26, 395–398. [Google Scholar] [CrossRef]
- Gresta, F.; Avola, G.; Guarnaccia, P. Agronomic characterization of some spontaneous genotypes of milk thistle (Silybum marianum L. Gaertn.) in Mediterranean environment. J. Herbs Spices Med. Plants 2007, 12, 51–60. [Google Scholar] [CrossRef]
- POWO, Plants of the World Online. Facilitated by the Royal Botanic Gardens, Kew. 2022. Available online: http://www.plantsoftheworldonline.org/ (accessed on 16 February 2022).
- Hamilton, W.R.; Stohs, S.J. Hepatic effects of herbal remedies, Chapter 3. In Herbal Medicinals. A Clinician’s Guide; Miller, L.G., Murray, W.J., Eds.; Pharmaceutical Products Press: Binghamton, NY, USA; New York, NY, USA, 1998; pp. 37–63. [Google Scholar]
- Rainone, F. Milk thistle. Am. Fam. Physician 2005, 72, 1285–1288. [Google Scholar] [PubMed]
- Polyak, S.J.; Morishima, C.; Lohmann, V.; Pal, S.; Lee, D.Y.; Liu, Y.; Graf, T.N.; Oberlies, N.H. Identification of hepatoprotective flavonolignans from silymarin. Proc. Natl. Acad. Sci. USA 2010, 107, 5995–5999. [Google Scholar] [CrossRef] [PubMed]
- Saller, R.; Brignoli, R.; Melzer, J.; Meier, R. An updated systematic review with meta-analysis for the clinical evidence of silymarin. Complement. Med. Res. 2008, 15, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.Y.W.; Liu, Y. Molecular Structure and Stereochemistry of Silybin A, Silybin B, Isosilybin A, and Isosilybin B, Isolated from Silybum marianum (Milk Thistle). J. Nat. Prod. 2003, 66, 1171–1174. [Google Scholar] [CrossRef] [PubMed]
- Anthony, K.; Saleh, M.A. Chemical profiling and antioxidant activity of commercial milk thistle food supplements. J. Chem. Pharm. Res. 2012, 4, 4440–4450. [Google Scholar]
- Engelberth, A.S.; Carrier, D.J.; Clausen, E.C. Separation of silymarins from milk thistle (Silybum marianum L.) extracted with pressurized hot water using fast centrifugal partition chromatography. J. Liq. Chromatogr. Relat. Technol. 2008, 31, 3001–3011. [Google Scholar] [CrossRef]
- Flora, K.; Hahn, M.; Rosen, H.; Benner, K. Milk thistle (Silybum marianum) for the therapy of liver disease. Am. J. Gastroenterol. 1998, 93, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Šeršeň, F.; Vencel, T.; Annus, J. Silymarin and its components scavenge phenylglyoxylic ketyl radicals. Fitoterapia 2006, 77, 525–529. [Google Scholar] [CrossRef]
- Morazzoni, P.; Bombardelli, E. Silybum marianum (Carduus marianus). Fitoterapia 1995, 66, 3–42. [Google Scholar]
- Schadewaldt, H. The history of Silymarin. Contribution to the history of liver therapy. Die Med. Welt 1969, 20, 902–914. [Google Scholar]
- Vargas-Mendoza, N.; Madrigal-Santillán, E.; Morales-González, Á.; Esquivel-Soto, J.; Esquivel-Chirino, C.; González-Rubio, M.G.L.; Gayosso-de-Lucio, J.A.; Morales-González, J.A. Hepatoprotective effect of silymarin. World J. Hepatol. 2014, 6, 144. [Google Scholar] [CrossRef] [PubMed]
- Karimi, G.; Vahabzadeh, M.; Lari, P.; Rashedinia, M.; Moshiri, M. “Silymarin”, a promising pharmacological agent for the treatment of diseases. Iran. J. Basic Med. Sci. 2011, 14, 308. [Google Scholar]
- Abenavoli, L.; Capasso, R.; Milic, N.; Capasso, F. Milk thistle in liver diseases: Past, present, future. Phytother. Res. 2010, 24, 1423–1432. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, N.; Zhao, J.; Wolf, D.M.; Agarwal, R. Inhibition of human carcinoma cell growth and DNA synthesis by silibinin, an active constituent of milk thistle: Comparison with silymarin. Cancer Lett. 1999, 147, 77–84. [Google Scholar] [CrossRef]
- Deep, G.; Agarwal, R. Chemopreventive efficacy of silymarin in skin and prostate cancer. Integr. Cancer Ther. 2007, 6, 130–145. [Google Scholar] [CrossRef]
- Andrzejewska, J.; Martinelli, T.; Sadowska, K. Silybum marianum: Non-medical exploitation of the species. Ann. Appl. Biol. 2015, 167, 285–297. [Google Scholar] [CrossRef]
- Bhattacharya, S. Phytotherapeutic properties of milk thistle seeds: An overview. J. Adv. Pharm. Educ. Res. 2011, 1, 69–79. [Google Scholar]
- Andrzejewska, J.; Sadowska, K.; Mielcarek, S. Effect of sowing date and rate on the yield and flavonolignan content of the fruits of milk thistle (Silybum marianum L. Gaertn.) grown on light soil in a moderate climate. Ind. Crops Prod. 2011, 33, 462–468. [Google Scholar] [CrossRef]
- Zheljazkov, V.D.; Zhalnov, I.; Nedkov, N.K. Herbicides for weed control in blessed thistle (Silybum marianum). Weed Technol. 2006, 20, 1030–1034. [Google Scholar] [CrossRef]
- Andrew, R.; Izzo, A.A. Principles of pharmacological research of nutraceuticals. Br. J. Pharmacol. 2017, 174, 1177–1194. [Google Scholar] [CrossRef] [PubMed]
- ISMEA. Piante Officinali in Italia: Un’istantanea della Filiera e dei Rapporti tra i Diversi Attori. Ist. Serv. Merc. Agric. Aliment. 2013. Available online: https://www.politicheagricole.it/flex/cm/pages/ServeBLOB.php/L/IT/IDPagina/6678 (accessed on 2 March 2022).
- Carrubba, A.; la Torre, R.; Matranga, A. Cultivation Trials of Some Aromatic and Medicinal Plants in a Semiarid Mediterranean Environment. Acta Hortic. 2002, 576, 207–213. [Google Scholar] [CrossRef]
- Carrubba, A.; la Torre, R. Cultivation trials of milk thistle (Silybum marianum Gaertn.) into the semiarid Mediterranean environment. Agron. Mediterr. 2003, 133, 14–19. [Google Scholar]
- Khan, M.A.; Blackshaw, R.E.; Marwat, K.B. Biology of milk thistle (Silybum marianum) and the management options for growers in north-western Pakistan. Weed Biol. Manag. 2009, 9, 99–105. [Google Scholar] [CrossRef]
- CABI—Centre for Agriculture and Bioscience International. Invasive Species Compendium. Silybum marianum (Variegated Thistle). 2021. Available online: https://www.cabi.org/isc/datasheet/50304#tosummaryOfInvasiveness (accessed on 29 December 2021).
- Sindel, B.M. A review of the ecology and control of thistles in Australia. Weed Res. 1991, 31, 189–201. [Google Scholar] [CrossRef]
- Groves, R.H.; Kaye, P.E. Germination and phenology of seven introduced thistle species in southern Australia. Aust. J. Bot. 1989, 37, 351–359. [Google Scholar] [CrossRef]
- Carrier, D.J.; Crowe, T.; Sokhansanj, S.; Wahab, J.; Barl, B. Milk thistle, Silybum marianum (L.) Gaertn., flower head development and associated marker compound profile. J. Herbs Spices Med. Plants 2003, 10, 65–74. [Google Scholar] [CrossRef]
- Abd-El-hady, M.A.M.; Arafa, S.G. Morphological, chemical characteristics and antioxidant activity of Egypt grown wild milk thistle (Silybum marianum L.) seeds and evaluates their oil in fast frying process comparing with some vegetable oils. Middle East J. Appl. Sci. 2019, 9, 1198–1214. [Google Scholar] [CrossRef]
- Rottoli, M. Zafferanone Selvatico (Carthamus lanatus) e Cardo della Madonna (Silybum marianum), Piante Raccolte o Coltivate nel Neolitico Antico a “La Marmotta”; Bullettino di Paletnologia Italiana: Roma, Italy, 2001; pp. 91–92. (In Italian) [Google Scholar]
- Křen, V.; Walterová, D. Silybin and silymarin-new effects and applications. Biomedical Pap. 2005, 149, 29–41. [Google Scholar] [CrossRef]
- Karkanis, A.; Bilalis, D.; Efthimiadou, A. Cultivation of milk thistle (Silybum marianum L. Gaertn.), a medicinal weed. Ind. Crops Prod. 2011, 34, 825–830. [Google Scholar] [CrossRef]
- Holm, L.; Pancho, J.V.; Herberger, J.P.; Plucknett, D.L. A Geographical Atlas of World Weeds; John Wiley & Sons: New York, NY, USA, 1979. [Google Scholar]
- Vereš, T.; Týr, Š. Milk thistle (Silybum marianum (L.) Gaertn.) as a weed in sustainable crop rotation. Res. J. Agric. Sci. 2012, 44, 118–122. [Google Scholar]
- Sulas, L.; Porqueddu, C.; Bullitta, P. Persistency of self-reseeding legumes under sheep grazing. In Proceedings of the 14th General Meeting of the European Grassland Federation, Lahti, Finland, 8–11 June 1992; pp. 299–303. [Google Scholar]
- Andrzejewska, J.; Skinder, Z. Yield and quality of raw material of milk thistle [Silybum marianum (L). Gaertn.] grown in monoculture and in crop rotation. Part I. Reaction of milk thistle to the sowing date. Herba Pol. 2006, 52, 11–17. [Google Scholar]
- Tutin, T.G.; Heywood, V.H.; Burges, N.A.; Valentine, D.H. Flora Europaea: Plantaginaceae to Compositae (and Rubiaceae); Cambridge University Press: Cambridge, UK, 1976; Volume 4. [Google Scholar]
- USDA. Plant Hardiness Zone Map for the United States. USDA Misc. Publ. 1960, 814, 1. [Google Scholar]
- Hetz, E.; Liersch, R.; Schieder, O. Genetic investigations on Silybum marianum and S. eburneum with respect to leaf colour, outcrossing ratio, and flavonolignan composition. Planta Med. 1995, 61, 54–57. [Google Scholar] [CrossRef] [PubMed]
- Alemardan, A.; Karkanis, A.; Salehi, R. Breeding objectives and selection criteria for milk thistle [Silybum marianum (L.) Gaertn.] improvement. Not. Bot. Horti Agrobot. Cluj-Napoca 2013, 41, 340–347. [Google Scholar] [CrossRef][Green Version]
- Carrubba, A.; Catalano, C. Essential oil crops for sustainable agriculture—a review. In Climate Change, Intercropping, Pest Control and Beneficial Microorganisms; Lichtfouse, E., Ed.; Springer: Dijon, France, 2009; pp. 137–187. [Google Scholar] [CrossRef]
- Martinelli, T. Identification of Milk Thistle Shatter-Resistant Mutant Lines with Altered Lignocellulosic Profile for the Complete Domestication of the Species. Crop. Sci. 2019, 59, 2119–2127. [Google Scholar] [CrossRef]
- Gonceariuc, M. Some breeding results of Silybum marianum Gaertn. Rom. Biol. Sci. 2007, 5, 52–53. [Google Scholar]
- Sulas, L.; Re, G.A.; Bullitta, S.; Piluzza, G. Chemical and productive properties of two Sardinian milk thistle (Silybum marianum (L.) Gaertn.) populations as sources of nutrients and antioxidants. Genet. Resour. Crop Evol. 2016, 63, 315–326. [Google Scholar] [CrossRef]
- El-Garhy, H.A.; Khattab, S.; Moustafa, M.M.; Abou Ali, R.; Azeiz, A.Z.A.; Elhalwagi, A.; El Sherif, F. Silybin content and overexpression of chalcone synthase genes in Silybum marianum L. plants under abiotic elicitation. Plant Physiol. Biochem. 2016, 108, 191–202. [Google Scholar] [CrossRef]
- ElSayed, A.I.; El-Hamahmy, M.A.; Rafudeen, M.S.; Mohamed, A.H.; Omar, A.A. The impact of drought stress on antioxidant responses and accumulation of flavonolignans in milk thistle (Silybum marianum (L.) Gaertn.). Plants 2019, 8, 611. [Google Scholar] [CrossRef] [PubMed]
- Soliman, S.M.; Sherif, H.S.; Bonfill, M.M.; El-Garhy, H.A. Bioinformatics and differential expression analysis of chalcone synthase genes (CHS1, 2, 3) under gamma rays elicitation in Silybum marianum L. In Proceedings of the 4th International Conference on Biotechnology Applications in Agriculture (ICBAA), Benha University, Hurghada, Egypt, 28–30 March 2018; pp. 1–11. [Google Scholar] [CrossRef]
- Torres, M.; Corchete, P. Gene expression and flavonolignan production in fruits and cell cultures of Silybum marianum. J. Plant Physiol. 2016, 192, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Kazmierczak, K.; Seidler-Lozykowska, K. Silma-polska odmiana ostropestu plamistego (Silybum marianum L. Gaertn.). Herba Pol. 1997, 43, 195–198. [Google Scholar]
- Shokrpour, M.; Mohammadi, S.A.; Moghaddam, M.; Ziai, S.A.; Javanshir, A. Variation in flavonolignan concentration of milk thistle (Silybum marianum) fruits grown in Iran. J. Herbs Spices Med. Plants 2008, 13, 55–69. [Google Scholar] [CrossRef]
- Dušková, E.; Dušek, K.; Smékalová, K. A descriptor list of Silybum marianum (L.) Gaertner-morphological and biological characters. In Proceedings of the 6th International Symposium Breeding Research on Medicinal and Aromatic Plants, Quedlinburg, Germany, 19–23 June 2016. [Google Scholar] [CrossRef]
- Montemurro, P.; Fracchiolla, M.; Lonigro, A. Effects of some environmental factors on seed germination and spreading potentials of Silybum marianum Gaertner. Ital. J. Agron. 2007, 2, 315–320. [Google Scholar] [CrossRef]
- Hetz, F.; Liersch, R.; Schieder, O. The Ratio of Auto- and Xenogamy in Silybum marianum. Planta Med. 1993, 59, A702. [Google Scholar] [CrossRef]
- Nyiredy, S.; Szűcs, Z.; Antus, S.; Samu, Z. New components from Silybum marianum L. fruits: A theory comes true. Chromatographia 2008, 68, 5–11. [Google Scholar] [CrossRef]
- Vaknin, Y.; Hadas, R.; Schafferman, D.; Murkhovsky, L.; Bashan, N. The potential of milk thistle (Silybum marianum L.), an Israeli native, as a source of edible sprouts rich in antioxidants. Int. J. Food Sci. Nutr. 2008, 59, 339–346. [Google Scholar] [CrossRef]
- Evans, W.C. Trease and Evans. In Pharmacognosy, 9th ed.; Saunders Elsevier: Amsterdam, The Netherlands, 2002; p. 553. [Google Scholar]
- Dodd, J. Phenology and seed production of variegated thistle, Silybum marianum (L.) Gaertn., in Australia in relation to mechanical and biological control. Weed Res. 1989, 29, 255–263. [Google Scholar] [CrossRef]
- Martinelli, T.; Andrzejewska, J.; Salis, M.; Sulas, L. Phenological growth stages of Silybum marianum according to the extended BBCH scale. Ann. Appl. Biol. 2015, 166, 53–66. [Google Scholar] [CrossRef]
- Gabay, R.; Plitmann, U.; Danin, A. Factors affecting the dominance of Silybum marianum (Asteraceae) in its specific habitats. Flora 1994, 189, 201–206. [Google Scholar] [CrossRef]
- Valladares, F.; Gianoli, E.; Gómez, J.M. Ecological limits to plant phenotypic plasticity. New Phytol. 2007, 176, 749–763. [Google Scholar] [CrossRef]
- Matesanz, S.; Gianoli, E.; Valladares, F. Global change and the evolution of phenotypic plasticity in plants. Ann. N. Y. Acad. Sci. 2010, 1206, 35–55. [Google Scholar] [CrossRef] [PubMed]
- Kozera, W.; Nowak, K.; Techniczno-Rolnicza, A. The effect of fertilization on milk thistle (Silybum marianum) yield and its chosen features. Ann. Univ. Mariae Curie-Skłodowska Lub. 2004, 59, 369–374. [Google Scholar]
- Purohit, S.M.; Iqbal, M.; Srivastava, P.S. Seed germination studies on an important medicinal plant, Silybum marianum (Linn.). Hamdard Med. 1997, 40, 31–33. [Google Scholar]
- Macák, M.; Demjanová, E.; Hunková, E. Forecrop value of milk thistle (Silybum marianum [L.] Gaertn.) in sustainable crop rotation. In Book of Scientific Papers and Abstracts, Proceeding of the 1st International Scientific Conference on Medicinal, Aromatic and Spice Plants, Nitra, Slovak, 5-6 December 2007; Slovak University of Agriculture: Nitra-Chrenová, Slovak, 2007; pp. 102–104. [Google Scholar]
- Habán, M.; Otepka, P.; Habánová, M. Production and quality of milk thistle (Silybum marianum [L.] Gaertn.) cultivated in cultural conditions of warm agri-climatic macroregion. Hortic. Sci. 2009, 36, 69–74. [Google Scholar] [CrossRef]
- Omidbaigi, R.; Karimzadeh, G.; Koshki, M.H. A study on the influence of sowing date and plant density on the productivity of Silybum marianum and the characteristics correlation. Iran. J. Sci. Technol. 2003, 1, 203–212. [Google Scholar]
- Gupta, M. Effect of nutrient management on yield attributes and yield of milk thistle (Silybum marianum). Environ. Conserv. J. 2020, 21, 163–166. [Google Scholar] [CrossRef]
- Omer, E.A.; Refaat, A.M.; Ahmed, S.S.; Kamel, A.; Hammouda, F.M. Effect of spacing and fertilization on the yield and active constituents of milk thistle, Silybum marianum. J. Herbs Spices Med. Plants 1993, 1, 17–23. [Google Scholar] [CrossRef]
- Belitz, A.R.; Sams, C.E. The effect of water stress on the growth, yield, and flavonolignan content in milk thistle (Silybum marianum). Acta Hortic. 2007, 756, 259–266. [Google Scholar] [CrossRef]
- Carrubba, A. Sustainable Fertilization in Medicinal and Aromatic Plants. In Medicinal and Aromatic Plants of the World; Springer Science+Business Media: Dordrecht, The Netherlands, 2015; Volume 1, pp. 187–203. [Google Scholar] [CrossRef]
- Sivakumar, M.V.K.; Huda, A.K.S. Potential agricultural productivity in summer and winter rainfall areas. In Nutrient Balances and the Need for Fertilizers in Semi-Arid and Arid Regions; Der Bund: Bern, Switzerland, 1983; pp. 23–47. [Google Scholar]
- Bielski, S. Milk thistle (Silybum marianum L. Gaertn.) achene yield had a positive response to nitrogen fertilization, row spacing, sowing date, and weed control methods. Ind. Crops Prod. 2021, 160, 113104. [Google Scholar] [CrossRef]
- Liava, V.; Karkanis, A.; Tsiropoulos, N. Yield and silymarin content in milk thistle (Silybum marianum (L.) Gaertn.) fruits affected by the nitrogen fertilizers. Ind. Crops Prod. 2021, 171, 113955. [Google Scholar] [CrossRef]
- Skolnikova, M.; Skarpa, P.; Ryant, P. Effect of nitrogen fertilization on yield and quality of milk thistle [Silybum marianum L. (Gaertn.)] achenes. J. Elem. 2019, 24, 701–710. [Google Scholar] [CrossRef]
- Andrzejewska, J.; Sadowska, K. Effect of cultivation conditions on the variability and interrelation of yield and raw material quality in milk thistle (Silybum marianum (L.) Gaertn.). Acta Sci. Pol. Agron. 2008, 7, 3–12. [Google Scholar]
- Geneva, M.; Zehirov, G.; Stancheva, I.; Iliev, L.; Georgiev, G. Effect of soil fertilizer, foliar fertilizer, and growth regulator application on milk thistle development, seed yield, and silymarin content. Commun. Soil Sci. Plant Anal. 2007, 39, 17–24. [Google Scholar] [CrossRef]
- Afshar, K.R.; Chaichi, M.R.; Alipour, A.; Ansari Jovini, M.; Dashtaki, M.; Hashemi, M. Potential of Milk Thistle for Biomass Production in Semiarid Regions. Crop Sci. 2015, 55, 1295–1301. [Google Scholar] [CrossRef]
- Hevia, F.; Wilckens, R.L.; Berti, M.T.; Fischer, S.U. Calidad de Cardo Mariano (Silybum marianum (L.) Gaertn.) cosechado en diferentes estados fenológicos. Inf. Tecnol. 2007, 18, 69–74. [Google Scholar] [CrossRef]
- Curioni, A.; Garcia, M.; Alfonso, W.; Arizio, O. Milk thistle harvest prediction through the external characteristics of the heads. Acta Hortic. 2002, 569, 257–261. [Google Scholar] [CrossRef]
- Tsiaousi, A.; Vasilakoglou, I.; Gravalos, I.; Koutroubas, S.D. Comparison of milk thistle (Silybum marianum) and cardoon (Cynara cardunculus) productivity for energy biomass under weedy and weed-free conditions. Eur. J. Agron. 2019, 110, 125924. [Google Scholar] [CrossRef]
- Berner, D.K.; Paxson, L.K.; Bruckart, W.L.; Luster, D.G.; McMahon, M.; Michael, J.L. First report of Silybum marianum as a host of Puccinia punctiformis. Plant Dis. 2002, 86, 1271. [Google Scholar] [CrossRef]
- Basso, F. Cardo mariano (Silybum marianum Gaertn.). In Piante officinali, Aromatiche e Medicinali. Aspetti Bioagronomici Aromatici e Fitoterapeutici; Pitagora Editrice: Bologna, Italy, 2009; pp. 199–201. [Google Scholar]
- Abdel-Moniem, A.S.H. The seed-head weevil, Larinus latus Herbst (Coleoptera: Curculionidae) as a new record in Egypt on the milk thistle, Silybum marianum (L.) (Asteraceae: Compositae). Arch. Phytopathol. Plant Prot. 2002, 35, 157–160. [Google Scholar] [CrossRef]
- Abourashed, E.A.; Mikell, J.R.; Khan, I.A. Bioconversion of silybin to phase I and II microbial metabolites with retained antioxidant activity. Bioorg. Med. Chem. 2012, 20, 2784–2788. [Google Scholar] [CrossRef] [PubMed]
- Malekzadeh, M.; Mirmazloum, S.I.; Mortazavi, S.N.; Panahi, M.; Angorani, H.R. Physicochemical properties and oil constituents of milk thistle (Silybum marianum Gaertn. cv. Budakalaszi) under drought stress. J. Med. Plants Res. 2011, 5, 2886–2889. [Google Scholar] [CrossRef]
- Garaev, E.A.; Movsumov, I.S.; Gazizov, F.Y. Neutral lipids from Silybum marianum seeds. Chem. Nat. Compd. 2010, 46, 629–630. [Google Scholar] [CrossRef]
- Martinelli, T.; Potenza, E.; Moschella, A.; Zaccheria, F.; Benedettelli, S.; Andrzejewska, J. Phenotypic evaluation of a milk thistle germplasm collection: Fruit morphology and chemical composition. Crop Sci. 2016, 56, 3160–3172. [Google Scholar] [CrossRef]
- Keshavarz Afshar, R.K.; Chaichi, M.R.; Rezaei, K.; Asareh, M.H.; Karimi, M.; Hashemi, M. Irrigation regime and organic fertilizers influence on oil content and fatty acid composition of milk thistle seeds. Agron. J. 2015, 107, 187–194. [Google Scholar] [CrossRef]
- Hadolin, M.; Škerget, M.; Knez, Z.; Bauman, D. High pressure extraction of vitamin E-rich oil from Silybum marianum. Food Chem. 2001, 74, 355–364. [Google Scholar] [CrossRef]
- El-Mallah, M.H.; El-Shami, S.M.; Hassanein, M.M. Detailed studies on some lipids of Silybum marianum (L.) seed oil. Grasas Aceites 2003, 54, 397–402. [Google Scholar] [CrossRef]
- Růžičková, G.; Fojtová, J.; Součková, M. The yield and quality of milk thistle [Silybum marianum (L). Gaertn.] seed oil from the perspective of environment and genotype-a pilot study. Sci. J. Phytotech. Zootech. 2011, 14, 9–12. [Google Scholar]
- Wagner, H.; Horhammer, L.; Munster, R. On the chemistry of silymarin (silybin), the active principle of the fruits from Silybum marianum (L.) Gaertn (Carduus marianus L.). Arzneim.-Forsch./Drug Res. 1968, 18, 688–696. [Google Scholar]
- Dvořák, Z.; Kosina, P.; Walterová, D.; Šimánek, V.; Bachleda, P.; Ulrichová, J. Primary cultures of human hepatocytes as a tool in cytotoxicity studies: Cell protection against model toxins by flavonolignans obtained from Silybum marianum. Toxicol. Lett. 2003, 137, 201–212. [Google Scholar] [CrossRef]
- Lee, J.I.; Narayan, M.; Barrett, J.S. Analysis and comparison of active constituents in commercial standardized silymarin extracts by liquid chromatography–electrospray ionization mass spectrometry. J. Chromatogr. B 2007, 845, 95–103. [Google Scholar] [CrossRef]
- Rajabian, T.; Rezazadeh, S.H.; Falah, H.H. Analysis of silymarin components in the seed extracts of some milk thistle ecotypes from Iran by HPLC. Iran. J. Sci. Technol. 2008, 32, 141–146. [Google Scholar] [CrossRef]
- Abenavoli, L.; Milic, N. Silymarin for liver disease. In Liver Pathophysiology; Muriel, P., Ed.; Academic Press: London, UK, 2017; pp. 621–631. [Google Scholar]
- Pelter, A.; Hänsel, R. The structure of silybin (silybum substance E6), the first flavonolignan. Tetrahedron Lett. 1968, 9, 2911–2916. [Google Scholar] [CrossRef]
- Takke, A.; Shende, P. Nanotherapeutic silibinin: An insight of phytomedicine in healthcare reformation. Nanomed-Nanotechnol. Biol. Med. 2019, 21, 102057. [Google Scholar] [CrossRef] [PubMed]
- Martin, R.J.; Lauren, D.R.; Smith, W.A.; Jensen, D.J.; Deo, B.; Douglas, J.A. Factors influencing silymarin content and composition in variegated thistle (Silybum marianum). N. Z. J. Crop Hort. Sci. 2006, 34, 239–245. [Google Scholar] [CrossRef]
- AbouZid, S.F.; Chen, S.N.; Pauli, G.F. Silymarin content in Silybum marianum populations growing in Egypt. Ind. Crops Prod. 2016, 83, 729–737. [Google Scholar] [CrossRef] [PubMed]
- Poppe, L.; Petersen, M. Variation in the flavonolignan composition of fruits from different Silybum marianum chemotypes and suspension cultures derived therefrom. Phytochemistry 2016, 131, 68–75. [Google Scholar] [CrossRef]
- Elateeq, A.A.; Sun, Y.; Nxumalo, W.; Gabr, A.M. Biotechnological production of silymarin in Silybum marianum L.: A review. Biocatal. Agron. Biotechnol. 2020, 29, 101775. [Google Scholar] [CrossRef]
- Adzet, T.; Coll, M.R.; Iglesias, J.; Puigmaciá, M. Selection and improvement of Silybum marianum. 1. Characterization of populations from different origins. Plant Physiol. Biochem. 1987, 25, 129–135. [Google Scholar]
- Martinelli, T.; Fulvio, F.; Pietrella, M.; Focacci, M.; Lauria, M.; Paris, R. In Silybum marianum Italian wild populations the variability of silymarin profiles results from the combination of only two stable chemotypes. Fitoterapia 2021, 148, 104797. [Google Scholar] [CrossRef] [PubMed]
- Qavami, N.; Naghdi Badi, H.; Labbafi, M.R.; Mehrafarin, A. A review on pharmacological, cultivation and biotechnology aspects of milk thistle (Silybum marianum (L.) Gaertn.). J. Med. Plants 2013, 12, 19–37. [Google Scholar]
- Dewick, P.M. The shikimate pathway: Aromatic amino acids and phenylpropanoids. In Medicinal Natural Products. A Biosynthetic Approach; Dewick, P.M., Ed.; John Wiley & Sons: West Sussex, UK, 2002; pp. 121–166. [Google Scholar]
- Althagafy, H.S.; Meza-Aviña, M.E.; Oberlies, N.H.; Croatt, M.P. Mechanistic Study of the Biomimetic Synthesis of Flavonolignan Diastereoisomers in Milk Thistle. J. Org. Chem. 2013, 78, 7594–7600. [Google Scholar] [CrossRef] [PubMed]
- Lv, Y.; Gao, S.; Xu, S.; Du, G.; Zhou, J.; Chen, J. Spatial organization of silybin biosynthesis in milk thistle [Silybum marianum (L.) Gaertn]. Plant J. 2017, 92, 995–1004. [Google Scholar] [CrossRef] [PubMed]
- Giuliani, C.; Tani, C.; Bini, L.M.; Fico, G.; Colombo, R.; Martinelli, T. Localization of phenolic compounds in the fruits of Silybum marianum characterized by different silymarin chemotype and altered colour. Fitoterapia 2018, 130, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Keshavarz, A.R.; Chaichi, M.R.; Ansari Jovini, M.; Jahanzad, E.; Hashemi, M. Accumulation of silymarin in milk thistle seeds under drought stress. Planta 2015, 242, 539–543. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, Z.; Wu, S.C. Health benefits of Silybum marianum: Phytochemistry, pharmacology, and applications. J. Agri. Food Chem. 2020, 68, 11644–11664. [Google Scholar] [CrossRef]
- Wianowska, D.; Wiśniewski, M. Simplified procedure of silymarin extraction from Silybum marianum L. Gaertner. J. Chromatogr. Sci. 2015, 53, 366–372. [Google Scholar] [CrossRef]
- Lorenzo, J.M.; Putnik, P.; Bursać Kovačević, D.; Petrović, M.; Munekata, P.E.; Gómez, B.; Marszałek, K.; Roohinejad, S.; Barba, F.J. Silymarin compounds: Chemistry, innovative extraction techniques and synthesis. In Studies in Natural Products Chemistry; Elsevier: Amsterdam, The Netherlands, 2020; Volume 64, Chapter 4; pp. 111–130. [Google Scholar] [CrossRef]
- Rady, M.R.; Saker, M.M.; Matter, M.A. In vitro culture, transformation and genetic fidelity of Milk Thistle. J. Genet. Eng. Biotechnol. 2018, 16, 563–572. [Google Scholar] [CrossRef]
- Devi, K.P. Milk thistle (Silybum marianum). In Nonvitamin and Nonmineral Nutritional Supplements; Nabavi, S.M., Silva, A.S., Eds.; Academic Press: Oxford, UK, 2019; pp. 321–325. [Google Scholar] [CrossRef]
- Bekheet, S.A.; El-Bahr, M.K.; Ali, S.A.; Hamed, M.A. Callus production of globe artichoke and milk thistle: In vitro hypolipidemic and antioxidant activities. World J. Pharma Res. 2014, 3, 1–71. [Google Scholar]
- Hassanen, E.I.; Khalaf, A.A.A.; Zaki, A.R.; Ibrahim, M.A.; Galal, M.K.; Farroh, K.Y.; Azouz, R.A. Ameliorative effect of ZnO-NPs against bioaggregation and systemic toxicity of lead oxide in some organs of albino rats. Environ. Sci. Pollut. Res. 2021, 28, 37940–37952. [Google Scholar] [CrossRef] [PubMed]
- Das, S.K.; Mukherjee, S.; Vasudevan, D.M. Medicinal properties of milk thistle with special reference to silymarin—an overview. Nat. Prod. Rad. 2008, 7, 182–192. [Google Scholar]
- Verpoorte, R.; van der Heijden, R.T.J.M.; Ten Hoopen, H.J.G.; Memelink, J. Metabolic engineering of plant secondary metabolite pathways for the production of fine chemicals. Biotechnol. Lett. 1999, 21, 467–479. [Google Scholar] [CrossRef]
- Liu, S.Q.; Cai, Q.G. Callus formation from protoplasts and plant regeneration from tissue culture of Silybum marianum Gaertn. Acta Bot. Sin. 1990, 32, 19–25. [Google Scholar]
- Alikaridis, F.; Papadakis, D.; Pantelia, K.; Kephalas, T. Flavonolignan production from Silybum marianum transformed and untransformed root cultures. Fitoterapia 2000, 71, 379–384. [Google Scholar] [CrossRef]
- Sánchez-Sampedro, M.A.; Fernández-Tárrago, J.; Corchete, P. Yeast extract and methyl jasmonate-induced silymarin production in cell cultures of Silybum marianum (L.) Gaertn. J. Biotech. 2005, 119, 60–69. [Google Scholar] [CrossRef] [PubMed]
- Hasanloo, T.; Khavari-Nejad, R.A.; Majidi, E.; Ardakani, M.S. Flavonolignan production in cell suspension culture of Silybum marianum. Pharm. Biol. 2008, 46, 876–882. [Google Scholar] [CrossRef]
- Rahimi, S.; Hasanloo, T.; Najafi, F.; Khavari-Nejad, R.A. Methyl jasmonate influence on silymarin production and plant stress responses in Silybum marianum hairy root cultures in a bioreactor. Nat. Prod. Res. 2012, 26, 1662–1667. [Google Scholar] [CrossRef] [PubMed]
- Fallah, M.; Davoodvandi, A.; Nikmanzar, S.; Aghili, S.; Mirazimi, S.M.A.; Aschner, M.; Rashidian, A.; Hamblin, M.R.; Chamanara, M.; Naghsh, N.; et al. Silymarin (milk thistle extract) as a therapeutic agent in gastrointestinal cancer. Biomed. Pharmacother. 2021, 142, 112024. [Google Scholar] [CrossRef] [PubMed]
- Le, Q.U.; Lay, H.L.; Wu, M.C.; Joshi, R.K. Phytoconstituents and pharmacological activities of Silybum marianum (Milk Thistle): A critical review. Am. J. Essent. Oils Nat. Prod. 2018, 6, 41–47. [Google Scholar]
- Marmouzi, I.; Bouyahya, A.; Ezzat, S.M.; El Jemli, M.; Kharbach, M. The food plant Silybum marianum (L.) Gaertn.: Phytochemistry, Ethnopharmacology and clinical evidence. J. Ethnopharmacol. 2021, 265, 113303. [Google Scholar] [CrossRef] [PubMed]
- Porwal, O.; Ameen, M.S.M.; Anwer, E.T.; Uthirapathy, S.; Ahamad, J.; Tahsin, A. Silybum marianum (Milk Thistle): Review on its chemistry, morphology, ethno medical uses, phytochemistry and pharmacological activities. J. Drug Deliv. Ther. 2019, 9, 199–206. [Google Scholar] [CrossRef]
- Ravari, S.S.; Talaei, B.; Gharib, Z. The effects of silymarin on type 2 diabetes mellitus: A systematic review and meta-analysis. Obes. Med. 2021, 26, 100368. [Google Scholar] [CrossRef]
- Vidimce, J.; Pennell, E.N.; Foo, M.; Shiels, R.G.; Shibeeb, S.; Watson, M.; Bulmer, A.C. Effect of Silymarin Treatment on Circulating Bilirubin and Cardiovascular Disease Risk Factors in Healthy Men: A Single-Blind, Randomized Crossover Trial. Clin. Pharm. Drug Dev. 2021, 10, 1156–1165. [Google Scholar] [CrossRef] [PubMed]
- Radko, L.; Cybulski, W. Application of silymarin in human and animal medicine. J. Pre-Clin. Clin. Res. 2007, 1, 22–26. [Google Scholar]
- Ladas, E.J.; Kelly, K.M. Milk thistle: Is there a role for its use as an adjunct therapy in patients with cancer? J. Alter. Complement. Med. 2003, 9, 411–416. [Google Scholar] [CrossRef] [PubMed]
- Mojaddam, A.; Chaji, M.; Mohammadabadi, T.; Tabatabaei, V.S. Feeding Value of Silybum marianum for Sheep and its Effect on Fiber and Protein Digestion. Iran. J. Anim. Sci. Res. 2015, 7, 267–277. [Google Scholar]
- Szumacher-Strabel, M.; Cieslak, A.; Nowakowska, A. Effect of oils rich in linoleic acid on in vitro rumen fermentation parameters of sheep, goats and dairy cows. J. Anim. Feed Sci. 2009, 18, 440–452. [Google Scholar] [CrossRef]
- Karaiskou, C.; Kasapidou, E.; Michailidis, G.; Markantonatos, X.; Basdagianni, Z. Effect of dietary milk thistle (Silybum marianum L.) oil supplementation on animal performance and milk fatty acid composition in dairy ewes. Small Rumin. Res. 2021, 203, 106493. [Google Scholar] [CrossRef]
- Stringi, L.; Tedesco, D.; Di Miceli, G.; Di Grigoli, A.; Bonanno, A.; Galletti, S.; Giambalvo, D.; Tava, A. Effects of supplement of milk thistle seeds on milk production in Comisana ewes. In Proceedings of the 16th SIPAOC, Siena, Italy, 29 September–2 October 2004; Kalb Publisher: Cagliari, Italy, 2004. [Google Scholar]
- Piłat, J.; Mikołajczak, J.; Grabowicz, M.; Kaczmarek, I. Application of whole crop Silybum marianum (L.) Gaertn. in feeding of cattle. Zeszyty Naukowe AR We Wrocławiu (Konferencje XXII) 1999, 361, 105–110. [Google Scholar]
- Grabowicz, M.; Piłat, J.; Mikołajczak, J. Effect of silage from Silybum marianum (L.) Gaertn. on the dairy cow production. Ann. Warsaw Agric. Univ. Anim. Sci. 2001, 313–317. [Google Scholar]
- Potkanski, A.; Kowalczyk, J.; Nowak, W.; Czauderna, M.; Michalak, S. Effect of milk thistle (Silybum marianum L.) endosperm in the diet for cows on milk yield and fatty acid profiles. J. Anim. Feed Sci. 2001, 10, 83–90. [Google Scholar] [CrossRef]
- Potkanski, A.; Nowak, W.; Kujawa, A. Blessed milk thistle (Silybum marianum) endosperm and maize byproduct in diet for late rearing calves. Rocz. Akad. Rol. Poznaniu. Zootech. 1991, 229, 85–93. [Google Scholar]
- Li, F.; Li, F.; Zhao, T.; Mao, G.; Zou, Y.; Zheng, D.; Takase, M.; Feng, W.; Wu, X.; Yang, L. Solid-state fermentation of industrial solid wastes from the fruits of milk thistle Silybum marianum for feed quality improvement. Appl. Microbiol. Biotechnol. 2013, 97, 6725–6737. [Google Scholar] [CrossRef] [PubMed]
- Tedesco, D.; Domeneghini, C.; Sciannimanico, D.; Tameni, M.; Steidler, S.; Galletti, S. Silymarin, a possible hepatoprotector in dairy cows: Biochemical and histological observations. J. Vet. Med. 2004, 51, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Onmaz, A.; Ulger, I.; Ayaşan, T. Effects of silymarin (Silybum marianum) supplementation on milk and blood parameters of dairy cattle. S. Afr. J. Anim. Sci. 2017, 47, 758–765. [Google Scholar]
- Nikzad, Z.; Chaji, M.; Mirzadeh, K.; Mohammadabadi, T.; Sari, M. Effect of different levels of milk thistle (Silybum marianum) in diets containing cereal grains with different ruminal degradation rate on rumen bacteria of Khuzestan buffalo. Iran. J. Appl. Anim. Sci. 2017, 7, 401–409. [Google Scholar]
- Janocha, A.; Milczarek, A.; Pietrusiak, D. Impact of Milk Thistle (Silybum marianum [L.] Gaertn.) Seeds in Broiler Chicken Diets on Rearing Results, Carcass Composition, and Meat Quality. Animals 2021, 11, 1550. [Google Scholar] [CrossRef] [PubMed]
- Dumari, A.M.; Sarir, H.; Fani Makki, O.; Afzali, N. Effect of milk thistle (Silybum marianum L.) on biochemical parameters and immunity of broiler chicks fed aflatoxin B1 after three weeks. Iran. J. Toxicol. 2014, 8, 1098–1103. [Google Scholar]
- Chand, N.; Muhammad, D.; Durrani, F.R.; Qureshi, M.S.; Ullah, S.S. Protective effects of milk thistle (Silybum marianum) against aflatoxin B1 in broiler chicks. Asian-Austrlsn J. Anim. Sci. 2011, 24, 1011–1018. [Google Scholar] [CrossRef]
- Alhidary, I.A.; Rehman, Z.; Khan, R.U.; Tahir, M. Anti-aflatoxin activities of milk thistle (Silybum marianum) in broiler. World. Poult. Sci. J. 2017, 73, 559–566. [Google Scholar] [CrossRef]
- Muhammad, D.; Chand, N.; Khan, S.; Sultan, A.; Mushtaq, M. Hepatoprotective Role of Milk Thistle (Silybum marianum) in Meat Type Chicken Fed Aflatoxin B 1 Contaminated Feed. Pak. Vet. J. 2012, 32, 443–446. [Google Scholar]
- Schiavone, A.; Righi, F.; Quarantelli, A.; Bruni, R.; Serventi, P.; Fusari, A. Use of Silybum marianum fruit extract in broiler chicken nutrition: Influence on performance and meat quality. J. Anim. Physiol. Anim. Nutr. 2007, 91, 256–262. [Google Scholar] [CrossRef]
- Tedesco, D.; Steidler, S.; Galletti, S.; Tameni, M.; Sonzogni, O.; Ravarotto, L. Efficacy of silymarin-phospholipid complex in reducing the toxicity of aflatoxin B1 in broiler chicks. Poult. Sci. 2004, 83, 1839–1843. [Google Scholar] [CrossRef]
- Beskow, W.B.; Harrington, K.C.; Hodgson, J. Dispersal of weed seeds in goat dung. In Proceedings of the 15th Australian Weeds Conference, Adelaide, South Australia, 24–28 September 2006; Preston, C., Watts, J.H., Crossman, N.D., Eds.; Weed Management Society of South Australia: Adelaide, Australia, 2006; p. 894. [Google Scholar]
- Harrington, K.C.; Beskow, W.B.; Hodgson, J. Recovery and viability of seeds ingested by goats. N. Z. Plant Prot. 2011, 64, 75–80. [Google Scholar] [CrossRef][Green Version]
- Greenfield, R.; Tozer, K.; Zobel, G.; Cameron, C.; North, E. The impact of cutting prior to goat grazing on variegated thistle (Silybum marianum). N. Z. Plant Prot. 2019, 72, 158–165. [Google Scholar] [CrossRef]
- Arviv, A.; Muklada, H.; Kigel, J.; Voet, H.; Glasser, T.; Dvash, L.; Landau, S.Y. Targeted grazing of milk thistle (Silybum marianum) and Syrian thistle (Notobasis syriaca) by goats: Preference following preconditioning, generational transfer, and toxicity. Appl. Anim. Behav. Sci. 2016, 179, 53–59. [Google Scholar] [CrossRef]
- Stanley, D.F.; Holst, P.J.; Allan, C.J. The effect of sheep and goat grazing on variegated thistle (Silybum marianum) populations in annual pastures. Plant Prot. Quart. 2000, 15, 116–118. [Google Scholar]
- Grela, E.R.; Świątkiewicz, M.; Florek, M.; Wojtaszewska, I. Impact of milk thistle (Silybum marianum L.) seeds in fattener diets on pig performance and carcass traits and fatty acid profile and cholesterol of meat, backfat and liver. Livest. Sci. 2020, 239, 104180. [Google Scholar] [CrossRef]
- Kosina, P.; Dokoupilová, A.; Janda, K.; Sládková, K.; Silberová, P.; Pivodová, V.; Ulrichová, J. Effect of Silybum marianum fruit constituents on the health status of rabbits in repeated 42-day fattening experiment. Anim. Feed Sci. Techol. 2017, 223, 128–140. [Google Scholar] [CrossRef]
- Cullere, M.; Dalle Zotte, A.; Celia, C.; Renteria-Monterrubio, A.L.; Gerencsér, Z.; Szendrő, Z.; Kovács, M.; Kachlek, M.L.; Matics, Z. Effect of Silybum marianum herb on the productive performance, carcass traits and meat quality of growing rabbits. Livest. Sci. 2016, 194, 31–36. [Google Scholar] [CrossRef]
- Alishahi, M.; Soltani, M.; Mesbah, M.; Rad, A.E. Effects of dietary Silybum marianum extract on immune parameters of the common carp (Cyprinus carpio). J. Vet. Res. 2011, 66, 255–263. [Google Scholar]
- Banaee, M.; Sureda, A.; Mirvaghefi, A.R.; Rafei, G.R. Effects of long-term silymarin oral supplementation on the blood biochemical profile of rainbow trout (Oncorhynchus mykiss). Fish Physiol. Biochem. 2011, 4, 885–896. [Google Scholar] [CrossRef] [PubMed]
- Jadayil, S.A.; Tukan, S.K.; Takruri, H.R. Bioavailability of iron from four different local food plants in Jordan. Plant Foods Hum. Nutr. 1999, 54, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Lancioni, M.C.; Ballero, M.; Mura, L.; Maxia, A. Usi alimentari e terapeutici nella tradizione popolare del Goceano (Sardegna centrale). Atti. Soc. Toscana Sci. Nat. Pisa Ser. B 2007, 114, 45–56. [Google Scholar]
- Morales, P.; Ferreira, I.C.F.R.; Carvalho, A.M.; Sánchez-Mata, M.C.; Cámara, M.; Fernández-Ruiz, V.; Pardo-de-Santayana, M.; Tardío, J. Mediterranean non-cultivated vegetables as dietary sources of compounds with antioxidant and biological activity. LWT-Food Sci. Technol. 2014, 55, 389–396. [Google Scholar] [CrossRef]
- García-Herrera, P.; Sánchez-Mata, M.C.; Cámara, M.; Fernández-Ruiz, V.; Díez-Marqués, C.; Molina, M.; Tardío, J. Nutrient composition of six wild edible Mediterranean Asteraceae plants of dietary interest. J. Food Composit. Anal. 2014, 34, 163–170. [Google Scholar] [CrossRef]
- Köksal, E.; Gülçin, I.; Beyza, S.; Sarikaya, O.; Bursal, E. In vitro antioxidant activity of silymarin. J. Enzym. Inhib. Med. Chem. 2009, 24, 395–405. [Google Scholar] [CrossRef]
- Matusevicius, P.; Juodeikiene, G.; Cizeikiene, D.; Ceskeviciute, V.; Vidmantaite, D.; Basinskiene, L.; Akunceva, I.; Stankevicius, M.; Maruska, A.; Bartkiene, E.; et al. The advantages of solid state fermentation to developed products of Silybum marianum L. seeds with higher nutrition value and safety for wheat bread. Curr. Opin. Biotech. 2013, 24, S58. [Google Scholar] [CrossRef]
- Juodeikiene, G.; Cizeikiene, D.; Ceskeviciute, V.; Vidmantiene, D.; Basinskiene, L.; Akuneca, I.; Stankevicius, M.; Maruska, A.; Bartkiene, E.; Ragazinskiene, O.; et al. Solid-state fermentation of Silybum marianum L. seeds used as additive to increase the nutritional value of wheat bread. Food Technol. Biotech. 2013, 51, 528–538. [Google Scholar]
- Zhu, S.; Dong, Y.; Tu, J.; Zhou, Y.; Dai, C. Amino acid composition and in vitro digestibility of protein isolates from Silybum marianum. J. Food Agric. Environ. 2013, 11, 136–140. [Google Scholar]
- Menasra, A.; Fahloul, D. Quality characteristics of biscuit prepared from wheat and milk thistle seeds (Silybum marianum (L) Gaertn.) flour. Carpath. J. Food Sci. Tech. 2019, 11, 5–19. [Google Scholar] [CrossRef]
- Choe, U.; Li, Y.; Gao, B.; Yu, L.; Wang, T.T.; Sun, J.; Chen, P.; Yu, L.L. The chemical composition of a cold-pressed milk thistle seed flour extract, and its potential health beneficial properties. Food Funct. 2019, 10, 2461–2470. [Google Scholar] [CrossRef] [PubMed]
- Bortlíková, V.; Kolarič, L.; Šimko, P. Application of milk thistle (Silybum marianum) in functional biscuits formulation. Acta Chim. Slovaca 2019, 12, 192–199. [Google Scholar] [CrossRef]
- Ahmad, T.; Zeb, A.; Nagra, S.A.; Perveen, S. Characteristics of Silybum marianum as a potential source of dietary oil and protein. Biol. Sci. 2007, 50, 36–40. [Google Scholar]
- Sulas, L.; Ventura, A.; Murgia, L. Phytomass production from Silybum marianum for bioenergy. Options Méditer Ser. A 2008, 79, 487–490. [Google Scholar]
- Ledda, L.; Deligios, P.A.; Farci, R.; Sulas, L. Biomass supply for energetic purposes from some Cardueae species grown in Mediterranean farming systems. Ind. Crops Prod. 2013, 47, 218–226. [Google Scholar] [CrossRef]
- Kalamaras, S.D.; Kotsopoulos, T.A. Anaerobic co-digestion of cattle manure and alternative crops for the substitution of maize in South Europe. Bioresour. Technol. 2014, 172, 68–75. [Google Scholar] [CrossRef]
- Zheljazkov, V.; Nikolov, S. Accumulation of Cd, Pb, Cu, Mn and Zn by Silybum marianum L. on polluted soils. Acta Hortic. 1996, 426, 297. [Google Scholar] [CrossRef]
- Del Río-Celestino, M.; Font, R.; Moreno-Rojas, R.; De Haro-Bailón, A. Uptake of lead and zinc by wild plants growing on contaminated soils. Ind. Crops Prod. 2006, 24, 230–237. [Google Scholar] [CrossRef]
- Brunetti, G.; Soler-Rovira, P.; Farrag, K.; Senesi, N. Tolerance and accumulation of heavy metals by wild plant species grown in contaminated soils in Apulia region, Southern Italy. Plant Soil 2009, 318, 285–298. [Google Scholar] [CrossRef]
- Tang, S.; Willey, N.J. Uptake of 134 Cs by four species from the Asteraceae and two varieties from the Chenopodiaceae grown in two types of Chinese soil. Plant Soil 2003, 250, 75–81. [Google Scholar] [CrossRef]
- Domínguez, M.T.; Montiel-Rozas, M.M.; Madejón, P.; Diaz, M.J.; Madejón, E. The potential of native species as bioenergy crops on trace-element contaminated Mediterranean lands. Sci. Total Environ. 2017, 590, 29–39. [Google Scholar] [CrossRef]
- Domínguez, M.T.; Madejón, P.; Madejón, E.; Diaz, M.J. Novel energy crops for Mediterranean contaminated lands: Valorization of Dittrichia viscosa and Silybum marianum biomass by pyrolysis. Chemosphere 2017, 186, 968–976. [Google Scholar] [CrossRef]
- Vachleva, N.; Atanassov, N.; Vachlev, V.; Vuchleva, R.; Karadjova, O. Toxic action of plant extracts on some pests of economic importance. Bulg. J. Agric. Sci. 2001, 7, 133–139. [Google Scholar]
- Elhaak, M.A.; Ebrahim, M.K.H.; Elshintinawy, F.; Mehana, H. Allelopathic potential of Silybum marianum and its utilization ability as a bio herbicide. Int. J. Curr. Microbiol. Appl. Sci. 2014, 3, 389–401. [Google Scholar]
- Baranowska, B.; Kurzepa, K.; Marczak, E.; Szczucinska, A.; Lipkowski, A.W. Utylizacja odpadu nasion ostropestu plamistego II. Biologicznie czynne peptydy z odpadu nasion ostropestu plamistego [Utilisation of milk thistle seed waste II. Biologicaly active peptides from milk thistle seed waste]. Oilseed Crops 2003, 24, 725–732. [Google Scholar]
- Semalty, A.; Semalty, M.; Rawat, M.S.M.; Franceschi, F. Supramolecular phospholipids-polyphenolics interactions: The PHYTOSOME® strategy to improve the bioavailability of phytochemicals. Fitoterapia 2010, 81, 306–314. [Google Scholar] [CrossRef]
- Ahmad, M.; Zafar, M.; Sultana, S.; Azam, A.; Khan, M.A. The optimization of biodiesel production from a novel source of wild non-edible oil yielding plant Silybum marianum. Int. J. Green Energy 2014, 11, 589–594. [Google Scholar] [CrossRef]
- Takase, M.; Feng, W.; Wang, W.; Gu, X.; Zhu, Y.; Li, T.; Yang, L.; Wu, X. Silybum marianum oil as a new potential non-edible feedstock for biodiesel: A comparison of its production using conventional and ultrasonic assisted method. Fuel Process. Technol. 2014, 123, 19–26. [Google Scholar] [CrossRef]
- Hahn, H.J.; Jung, H.J.; Schrammek-Drusios, M.C.; Lee, S.N.; Kim, J.-H.; Kwon, S.B.; An, I.-S.; An, S.; Ahn, K.J. Instrumental evaluation of anti-aging effects of cosmetic formulations containing palmitoyl peptides, Silybum marianum seed oil, vitamin E and other functional ingredients on aged human skin. Exp. Ther. Med. 2016, 12, 1171–1176. [Google Scholar] [CrossRef] [PubMed]
- Ligęza, M.; Wyglądacz, D.; Tobiasz, A.; Jaworecka, K.; Reich, A. Natural cold pressed oils as cosmetic products. Fam. Med. Prim. Care Rev. 2016, 4, 443–447. [Google Scholar] [CrossRef]
- Singh, R.P.; Agarwal, R. Cosmeceuticals and silibynin. Clin. Dermatol. 2009, 27, 479–484. [Google Scholar] [CrossRef]
- Jabłonowska, M.; Ciganović, P.; Jablan, J.; Marguí, E.; Tomczyk, M.; Končić, M.Z. Silybum marianum glycerol extraction for the preparation of high-value anti-ageing extracts. Ind. Crops Prod. 2021, 168, 113613. [Google Scholar] [CrossRef]
- Drouet, S.; Leclerc, E.A.; Garros, L.; Tungmunnithum, D.; Kabra, A.; Abbasi, B.H.; Lainé, É.; Hano, C. A green ultrasound-assisted extraction optimization of the natural antioxidant and anti-aging flavonolignans from milk thistle Silybum marianum (L.) Gaertn. fruits for cosmetic applications. Antioxidants 2019, 8, 304. [Google Scholar] [CrossRef] [PubMed]
- Fehér, P.; Vecsernyés, M.; Fenyvesi, F.; Váradi, J.; Kiss, T.; Újhelyi, Z.; Nagy, K.; Bácskay, I. Topical application of Silybum marianum extract. Arad. Med. J. 2011, 14, 5–8. [Google Scholar]
- Křížová, L.; Watzková, J.; Třinaáctý, J.; Richter, M.; Buchta, M. Rumen degradability and whole tract digestibility of flavonolignans from milk thistle (Silybum marianum) fruit expeller in dairy cows. Czech J. Anim. Sci. 2011, 56, 269–278. [Google Scholar] [CrossRef]
- Harradine, A.R. Effect of 2,4-D ester on the nitrate concentration of variegated thistle (Silybum marianum) (L.) Gaertn. Aus. J. Exp. Agron. 1990, 30, 221–226. [Google Scholar] [CrossRef]
- Knott, S.G. Nitrite poisoning in livestock. Qld. Agron. J. 1971, 97, 485–489. [Google Scholar]
- European Food Safety Authority. Compendium of botanicals that have been reported to contain toxic, addictive, psychotropic or other substances of concern on request of EFSA. Eur. Food Saf. Auth. J. 2009. Available online: https://efsa.onlinelibrary.wiley.com/doi/epdf/10.2903/j.efsa.2009.281 (accessed on 10 January 2022).
- Bongiovanni, G.A.; Soria, E.A.; Eynard, A.R. Effects of the plant flavonoids silymarin and quercetin on arsenite-induced oxidative stress in CHO-K1 cells. Food Chem. Toxicol. 2007, 45, 971–976. [Google Scholar] [CrossRef]
- Santamaria, P. Nitrate in vegetables: Toxicity, content, intake and EC regulation. J. Sci. Food Agron. 2005, 86, 10–17. [Google Scholar] [CrossRef]


| Livestock Species | S. Marianum as Fodder or Feed Additive | Present and/or Potential Uses | References |
|---|---|---|---|
| Sheep | Plant (green forage) | Dry matter intake, rumen fermentation parameters, and blood metabolites were positively affected by diets containing S. marianum, proving its suitability to the feeding of small ruminants. | [138] |
| Seed (oil) | Oil from S. marianum proved to influence rumen fermentation, improving protozoa counts in sheep rumen. | [139] | |
| Ruminant diets with oils derived from S. marianum plants enhanced desirable milk fatty acid (FA) profile and milk yield. | [140] | ||
| Seed (silymarin extract) | A higher milk production throughout lactation was detected, with no evidence of toxic effects in ewes. | [141] | |
| Dairy cow | Plant (green forage) | Forage from S. marianum appeared to be worse than green forage from barley. | [142,143] |
| Plant (silage) | S. marianum could be used as a source of flavonolignans, beneficial for health, milk yield, and milk quality in dairy cattle. | [143] | |
| Seed (whole/ground) | Supplement from milk thistle endosperm considerably increased the concentration of monounsaturated fatty acids in milk fat. | [144] | |
| Seed (flour) | S. marianum seed flour proved to be a suitable feed additive for young ruminants. | [145] | |
| Fermented solid wastes from milk thistle seed flour showed good suitability for the improvement of feed quality. | [146] | ||
| Seed (oil) | Oil from S. marianum showed to influence rumen fermentation, improving protozoa counts in cows’ rumen. | [139] | |
| Seed (silymarin extract) | Earlier peak of milk production and higher milk production throughout lactation were detected, with no evidence of toxic effects in dairy cows. | [147] | |
| The hepatoprotective effect on dairy cows was detected. | [147] | ||
| Silymarin supplementation to feed rations speeded up the transition to the metabolic adaptation of dairy cows at the beginning of lactation, with no negative side effects. | [148] | ||
| Buffalo | Plant (dried and powdered) | Milk thistle could be used in up to 20% of the buffalo’s diet without any negative effect on digestion and fermentation characteristics of whole rumen microorganisms and bacteria. | [149] |
| Broiler chicken | Seed (whole/ground) | The introduction of S. marianum in starter and grower rations allowed for the highest body weight at the lowest feed conversion per body weight gain unit without influencing muscle mass and fattening grade, at the same time improving the meat’s nutritional value. | [150] |
| S. marianum seeds showed good hepatoprotective efficacy in counteracting the toxic effects of food contamination with aflatoxin B1. | [151,152,153,154] | ||
| Seed (extract) | The use of S. marianum fruit extract in broiler chicken nutrition enhanced the performance and meat quality. | [155] | |
| Seed (silymarin—phospholipid complex) | Protection against the negative effects of aflatoxin B1. | [156] | |
| Goat | Plant (green forage) | Evidence of the degradability/digestibility of S. marianum seeds and the diffusion of the species in local pasture areas through natural grazing. | [157,158] |
| Goats consumed up to 99% of the cut S. marianum foliage, also underlining the potential to include the grazing of goats in an integrated weed management strategy for the crop. | [159] | ||
| Goats ingest the S. marianum pasture readily. | [160] | ||
| Goats readily included the capitula of S. marianum in their diet, especially before maturity, determining the quick reduction of fodder availability and the potential seed production. | [161] | ||
| Seed (oil) | Oil from S. marianum was observed to influence rumen fermentation, improving bacteria counts in the goats’ rumen. | [139] | |
| Pig | Seed (whole/ground) | S. marianum seeds appeared to be a useful feed admixture for fattened pigs to improve meat quality, oxidative stability, weight gains, feed utilization, polyunsaturated fatty acid content in tissues, and water holding. | [162] |
| Rabbit | Seed (whole/ground) | Flavonolignans and taxifolin showed a mild effect on the growth performance of rabbits, and the diet containing the highest amount of S. marianum constituents was able to attenuate the morbidity and mortality of broiler rabbits. | [163,164] |
| Dietary supplementation with S. marianum changed the sensory characteristics of rabbit loin. | [164] | ||
| Common carp | Seed (silymarin extract) | S. marianum appeared to enhance some nonspecific immune responses. | [165] |
| Rainbow trout | Seed (silymarin extract) | Reduction of plasmatic glucose and cholesterol, with side effects on blood biochemical and clinical parameters only when doses were high, were detected. | [166] |
| Plant Parts, Products, and By-Products | Present and/or Potential Uses | References |
|---|---|---|
| Leaves, young stems, and sprouts | Young fleshy stems and sprouts are traditional food items in several Mediterranean countries. | [167] |
| S. marianum is a good candidate source of healthy edible sprouts. | [64] | |
| Young heads and stems are traditionally eaten in Sardinia. | [168] | |
| In Spain, traditional use as a fresh vegetable in salads or consumed boiled and fried has been reported. | [169] | |
| Whole seed (unprocessed) | High amounts in healthy substances such as fibers, and Ca and K levels, even higher than many conventional vegetables, was detected. | [170] |
| Seed (processed/fermented) | Effective antioxidant and radical-scavenging activity of silymarin from seeds suggests its utilization for minimizing or preventing lipid oxidation in food products. | [171] |
| Fermented S. marianum seeds proved to be a suitable additive for the natural flavoring of baked goods. | [172,173] | |
| S. marianum protein had an excellent balance of all essential amino acids, with potential application as a functional food ingredient. | [174] | |
| Seed (flour) | S. marianum seed flour was successfully tried for the enrichment of biscuits. | [175] |
| S. marianum seed flour could be added to functional foods with potential health benefits due to its phytochemical composition and gut microbiota-modulating, free radical-scavenging, anti-inflammatory, and anti-proliferative properties. | [176] | |
| Partial wheat flour replacement with S. marianum seed flour was suggested for the production of healthy biscuits, providing for a new item in functional bakery products on the market. | [177] | |
| Seed (oil) | S. marianum seed oil was suggested as a substitute for highly unsaturated fatty oils for human consumption. | [178] |
| S. marianum seed oil can be used in food and industrial foods as well as cottonseed, sunflower, and soybean oils. | [31] | |
| Seed (silymarin extract) | The introduction of pure silymarin in food could hamper the formation of toxic oxidation products, helping to maintain nutritional quality and prolonging the shelf life of food. | [175] |
| When used as a food stabilizer, silymarin exhibited a protective effect against arsenic-induced cytotoxicity. | [171] |
| Plant Parts, Products, and By-Products | Present and/or Potential Uses | References |
|---|---|---|
| Whole plant (crop/unprocessed) | Good biomass productivity suggested utilization as a bioenergy source even in Mediterranean environments. | [179,180] |
| S. marianum lignocellulosic biomass was tested for biogas production with positive results. | [181] | |
| Whole plants or residues after seed harvest were submitted to anaerobic fermentation, indicating S. marianum as a suitable raw material for biogas production. | [21] | |
| The plant’s ability to grow in soils with a wide range of heavy metal contamination, where it behaves as a metal excluder or a tolerant plant, suggested its utilization for phytoremediation purposes. | [182,183,184,185,186,187] | |
| Various plant parts (extracts) | Byproducts of the pharmaceutical processing of S. marianum seeds were reported to have insecticidal properties against the green peach aphid and the greenhouse whitefly. | [188] |
| Bioherbicides and biodegradable pesticides, useful in organic farming, were obtained from S. marianum seed, flower, stem, leaf, and root extracts. | [189] | |
| Seed (flour) | A bacteriostatic effect was detected, allowing S. marianum to be adopted in phytosomal formulations used as functional cosmetics. | [190,191] |
| Seed (oil) | Oil extracted from S. marianum seeds proved suitable for biodiesel production. | [192,193] |
| S. marianum seed oil and other mixed ingredients proved effective in the treatment of facial wrinkles, improving skin elasticity, dermal density, and tone. | [194] | |
| Cold-pressed S. marianum seed oil could be applied to the skin as cosmetics, with no irritating effect and rare allergic reactions. | [195] | |
| Seed (silymarin extract) | Antioxidant and UV-protection activity of silybin allowed for the use of S. marianum derivatives in the preparation of cosmeceuticals for skin protection. | [146,196] |
| The excellent toxicological and bioactivity profile of silymarin in a glycerol suspension as well as its anti-inflammatory activity, indicated that S. marianum glycerol extracts can be used in the preparation of high-value anti-aging products in cosmetics. | [197] | |
| Ultrasound-assisted extraction (UAE) of silymarin from mature fruits of S. marianum allowed for the green, efficient extraction of flavonolignans with potent antioxidant and anti-aging activity, to be employed in the development of cosmetic products. | [198] | |
| Topical treatments with a w/o emulsion from S. marianum extracts proved to possess skin whitening properties, promoting a significant decrease in skin melanin level. | [199] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marceddu, R.; Dinolfo, L.; Carrubba, A.; Sarno, M.; Di Miceli, G. Milk Thistle (Silybum Marianum L.) as a Novel Multipurpose Crop for Agriculture in Marginal Environments: A Review. Agronomy 2022, 12, 729. https://doi.org/10.3390/agronomy12030729
Marceddu R, Dinolfo L, Carrubba A, Sarno M, Di Miceli G. Milk Thistle (Silybum Marianum L.) as a Novel Multipurpose Crop for Agriculture in Marginal Environments: A Review. Agronomy. 2022; 12(3):729. https://doi.org/10.3390/agronomy12030729
Chicago/Turabian StyleMarceddu, Roberto, Lucia Dinolfo, Alessandra Carrubba, Mauro Sarno, and Giuseppe Di Miceli. 2022. "Milk Thistle (Silybum Marianum L.) as a Novel Multipurpose Crop for Agriculture in Marginal Environments: A Review" Agronomy 12, no. 3: 729. https://doi.org/10.3390/agronomy12030729
APA StyleMarceddu, R., Dinolfo, L., Carrubba, A., Sarno, M., & Di Miceli, G. (2022). Milk Thistle (Silybum Marianum L.) as a Novel Multipurpose Crop for Agriculture in Marginal Environments: A Review. Agronomy, 12(3), 729. https://doi.org/10.3390/agronomy12030729








