Biosurfactants: Potential and Eco-Friendly Material for Sustainable Agriculture and Environmental Safety—A Review
Abstract
1. Introduction
- Biosurfactants have emerged as potential molecules for drug delivery vehicles, medicinal applications, agricultural applications, and environmental safety, all of which provide economically appealing and scientifically novel applications.
- The current study discusses biosurfactants and their production by bacteria, with an emphasis on their involvement in oil cleanup.
- To explore novel biosurfactants those are commonly used for soil remediation.
- To identify the notable biomolecules that potentially replace harsh surfactants now employed in pesticide manufacturing.
- To determining the significance of environmental biosurfactants in plant growth promotion and other agricultural uses requires detailed investigation.
- Emphasize the use of biosurfactants as eco-friendly and alternatives to synthetic surfactants.
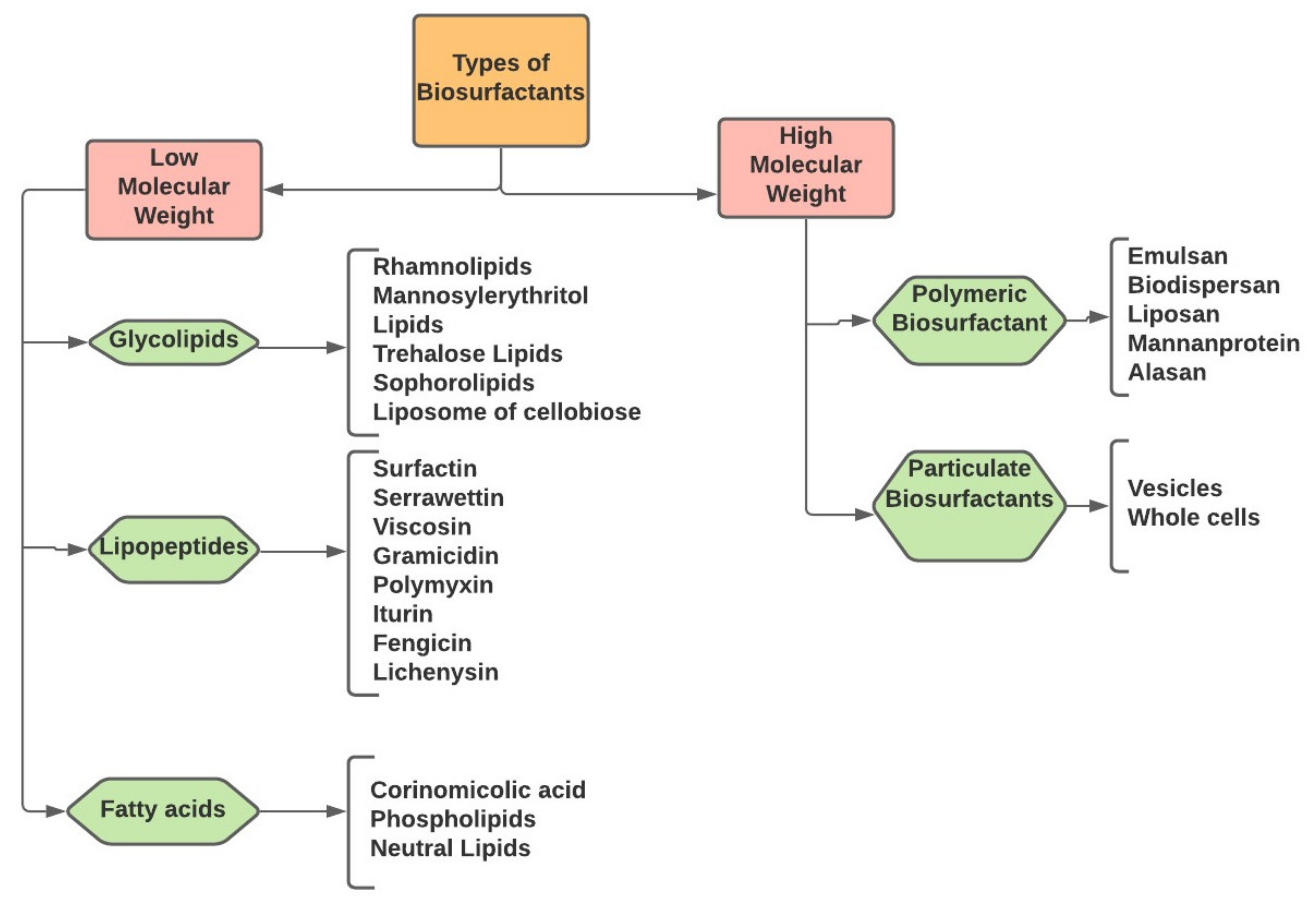
2. Classification of Biosurfactants
2.1. Glycolipid
2.1.1. Rhamnolipid
2.1.2. Sophorolipids
2.2. Lipoproteins and Lipopeptides
Surfactin
2.3. Fatty Acids, Phospholipids, and Neutral Lipids
2.4. Polymeric Biosurfactants
2.4.1. Emulsan
2.4.2. Liposan
2.4.3. Mannoprotein
2.5. Particulate Biosurfactant
3. Biosurfactant Producing Mechanisms
Mechanism of Action of Surfactants
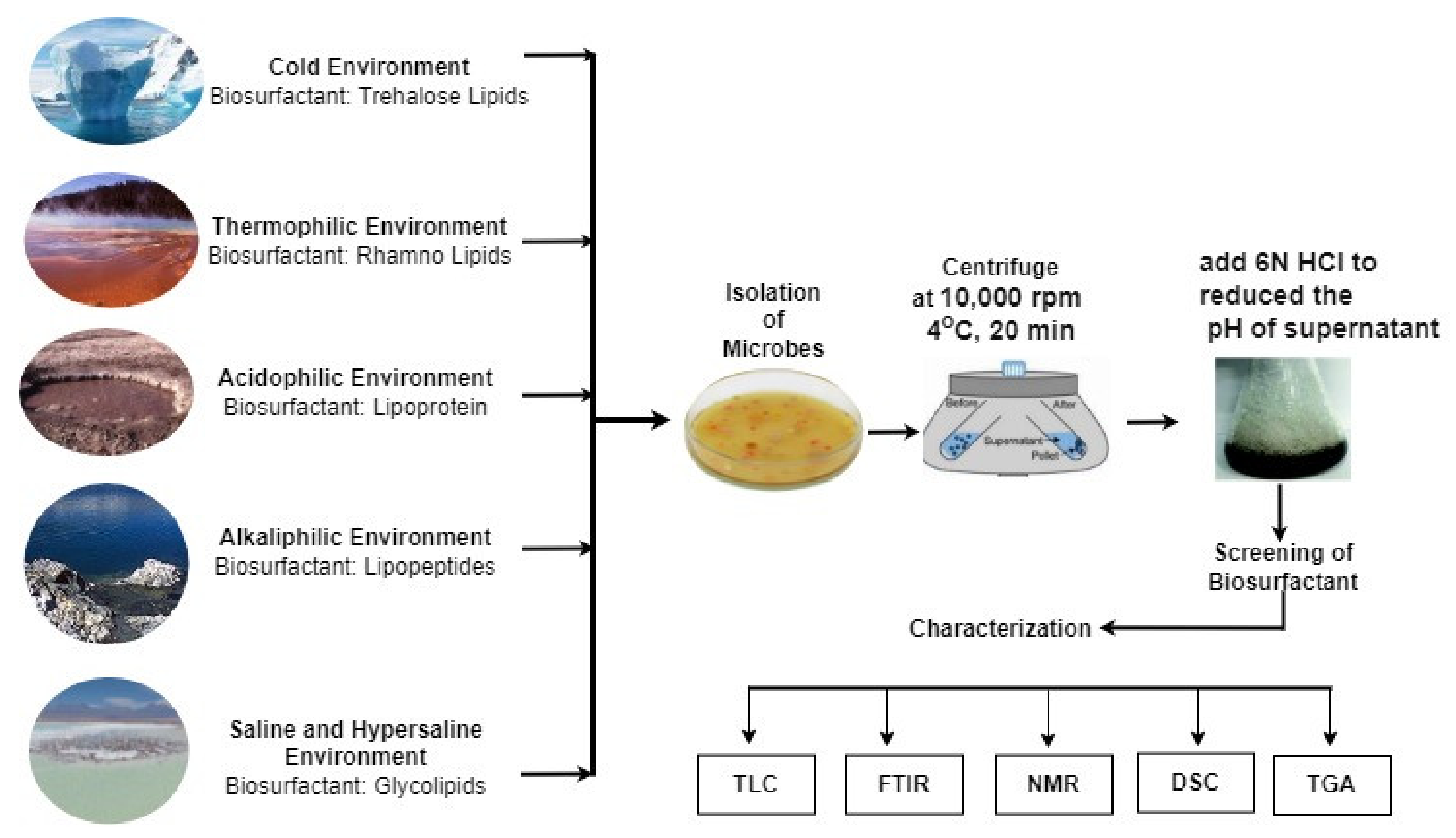
4. Isolation of Biosurfactant
5. Biosurfactant Produced by Microorganisms
| Microorganism | Biosurfactant | Property | Technology/Application | References |
|---|---|---|---|---|
| A. calcoaceticus RAG-1 (Arthrobacter RAG) | Heteropolysaccharides | A very good bioemulsifier, which makes heavy crude oil less viscous. | Biostimulation | [95] |
| Arthrobacter. sp., Rhodococcus aurantiacus | Trehalose, sucrose, and fructose, lipids | Lower the interfacial tension and make hydrophobic compounds more “pseudosoluble.” | Biodegradation and Bioaugmentation | [96] |
| Trehalose dimycolates | Mycobacterium sp., Nocardia sp. | Used more in cosmetic industries because of its lower irritability. | Bioaugmentation | [97] |
| Arthrobacter MIS 38 Bacillus atrophaeus 5-2a Pseudomonas fluorescence | Lipopeptide | Low interphase surface tension due to emulsifying action. | Bioaugmentation and biostimulation | [51] |
| Bacillus subtilis ATCC 21332 | Surfactin | Used in enhancement of the iron-remediation, anti-inflammatory activity. | Bioaugmentation and biodegradation | [49,50] |
| Pseudomonas aeruginosa L2-1, Bacillus sp. AB-2 | Rhamnolipids | Playing a crucial role in the field of pharmaceuticals. | Biodegradation | [16] |
| Candida bombicola ATCC 22214 | Sophorose lipids | Substantial % E24 against diverse hydrocarbons, including light and heavy crude oils, and high stability under salinity, pH, and extreme heat. | Bioaugmentation | [98] |
| Candida tropicalis | Mannan-fatty acid | Recognized as key antigenic determinants. | Biostimulation | [99] |
| Candida lipolytica Y-917, Torulopsis bombicola | Sophoros lipid | Produces hydrocarbon and oil emulsions in a liquid like water. | Biostimulation | [100] |
| Clostridium pasteurianum; Nocardia erythropolis | Neutral lipids | Have novel organic pollutant catabolism pathways and potential soil bioremediation capabilities for hydrocarbons and aromatic chemicals. | Biodegradation | [101] |
| Corynebacterium hydrocarbolastus, Corynebacterium lepus Strain MM1, Phaffta rhodozyma | Protein-lipid -carbohydrate | Fluids viscosified with viscoelastic surfactants (VESs) used in hydrocarbon recovery procedures. | Biodegradation | [102] |
| Myroides sp., Pseudomonas sp., Thibacillus sp., Agrobacterium sp., Cluconobacter sp. | Ornithine lipids | A potential biorestoration technique for contaminated sediments. | Bioaccumulation and biodegradation | [103] |
| Corynebacterium insidiosum | Phospholipids | Multiantibiotic resistant. | Biostimulation | [104] |
| Ochrobactrum anthropiHM-1 and Citrobacter freundiiHM-2 | Proteins | Reduces the viscosity of heavy oil, cleans oil storage tanks, and increases the flow of oil through pipelines. | Biostimulation | [104] |
| Penicillium spiculisporum | Spiculosporic acid | Used as a bioactive compound to remove heavy metal cations from water. | Biostimulation and bioaugmentation | [105] |
| Rhodococcus erythropolis | Trehalose-dicarynomycolate | A potential biorestoration technique for contaminated sediments. | Bioremediation and biotransformation | [106] |
| Rhodococcus sp. ST-5 Rhodococcus sp. H13-A Rhodococcus sp. 33 | Glycolipid | Responsiveness to hazardous and refractory chemicals such chlorinated aliphatic and aromatic hydrocarbons, N- and S-heterocyclic compounds, and synthetic polymers. | Bioremediation and biotransformation | [106] |
6. Properties of Biosurfactants
6.1. Surface and Interface Activity
6.2. Temperature, PH, Ionic Strength Tolerance
6.3. Biodegradability and Low Toxicity
6.4. Emulsion Forming and Emulsion Breaking
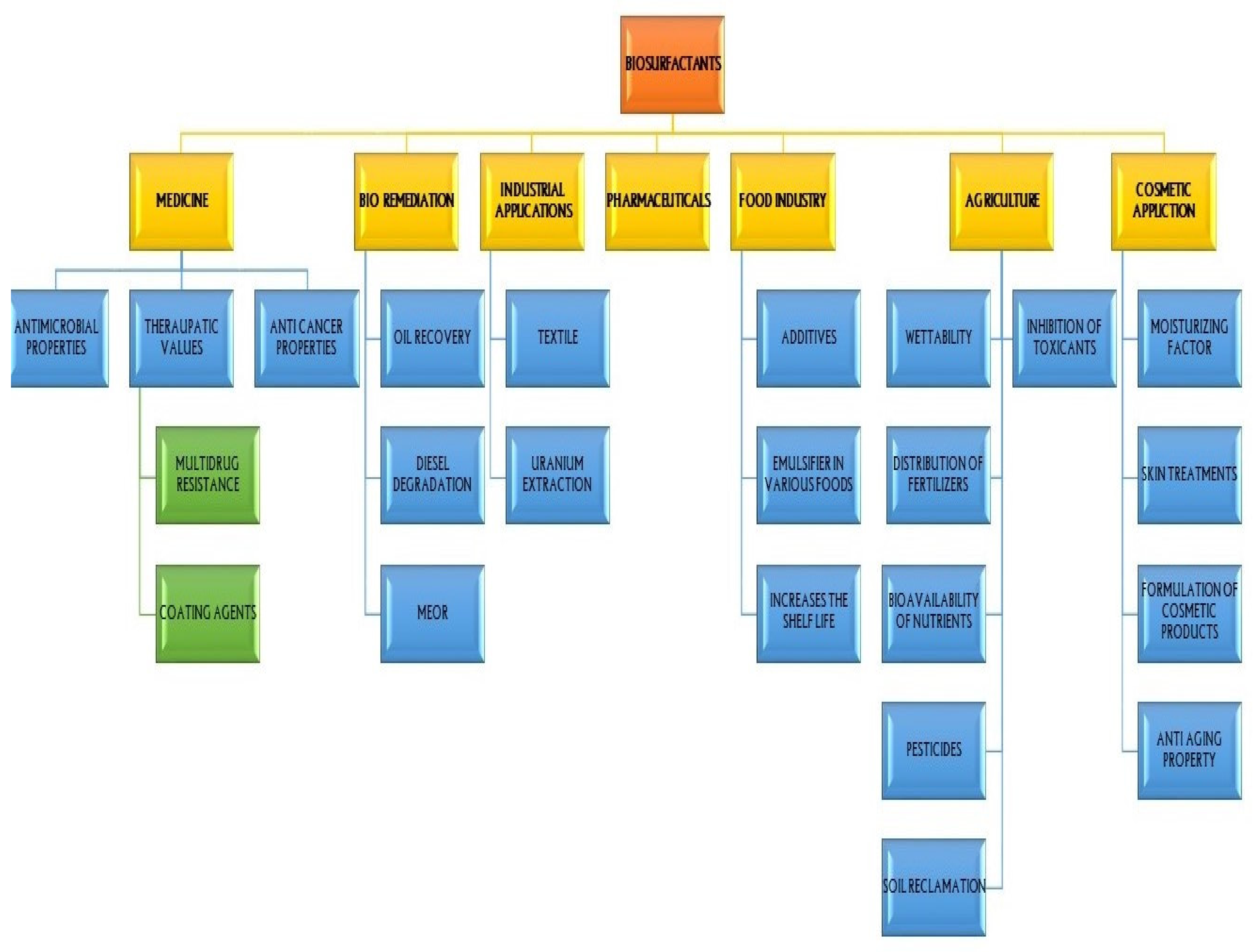
7. Application of Biosurfactants
7.1. Agriculture
7.2. Biosurfactants in Agriculture and Agrochemicals
7.3. Biosurfactant Mediated Plant Growth Promotion in Soils
7.4. Food and Agricultural Waste as Substrates for Biosurfactant Production
7.5. Biosurfactant for Cosmetic Application
7.6. Environment and Bioremediation
7.7. Industries
7.8. Pharmaceuticals
8. Critical Micelle Concentration of Various Biosurfactants
9. Recent Advances
9.1. Influence of Biosurfactant-Producing Bacteria on Compost
9.2. Effects of Biosurfactant on Green Waste Vermicomposting
9.3. Biosurfactants in Bionanotechnology
10. Future of Biosurfactants
11. Prospective and Challenges of Biosurfactant Production and Application
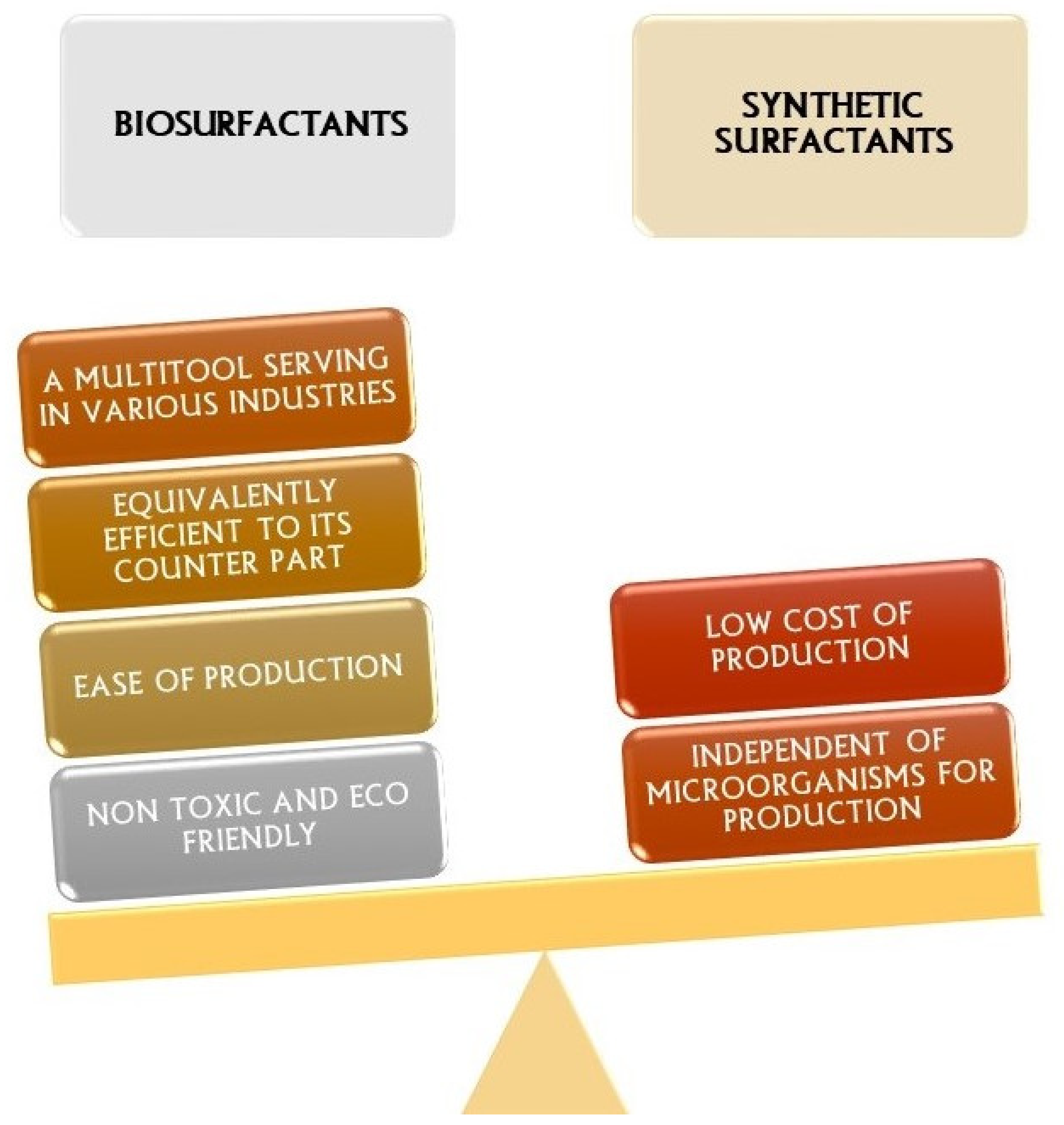
- After many years of mediocre interest, biosurfactants have recently risen to the top of many corporations’ agendas due to sustainability initiatives and green agendas.
- Biosurfactants may be customized for particular purposes and generated at a cost competitive with chemical surfactants.
- Several concerns must be addressed before large-scale exploitation may occur. The two issues facing rhamnolipids are safety and yield.
- Despite their immune system impacts and virulence factors, rhamnolipids are likely safe to use in many items, especially cleaning and laundry products.
- P. aeruginosa, being pathogenic is more challenging, although some companies have clearly overcome it, and the identification of potential new nonpathogenic producer organisms offers a potential solution, provided the products are suitable and yields are acceptable.
- The quorum sensing system controls rhamnolipid synthesis in P. aeruginosa, preventing hyperproducing strains from being produced through mutagenesis and selection or genetic manipulation. Inability to get large yields may prevent rhamnolipids from being used in many applications.
- Thus, these do not appear to be any significant barriers to the widespread use of biosurfactants in a variety of goods and applications over the next several years, and we may anticipate a growing range of home items containing at least sophorolipids and MELs on supermarket shelves.
12. Conclusions
Author Contributions
Funding
Conflicts of Interest
Nomenclature
| EI24 | Emulsification Index (%) |
| CMC | Critical Micelle Concentration |
| Bs | Biosurfactant |
| BSs | Biosurfactants |
References
- Georgiou, G.; Lin, S.C.; Sharma, M.M. Surface-active compounds from microorganisms. Nat. Biotechnol. 1992, 10, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Guerra-Santos, L.H.; Kappelli, O.; Fiechter, A. Dependence of Pseudomonas aeruginosa Continuous Culture Biosurfactant Production on Nutritional and Environmental Factors. Appl. Microbiol. Biotechnol. 1986, 24, 443–448. [Google Scholar] [CrossRef]
- Cooper, D.G.; Goldenberg, B.G. Surface Active Agents from Two Bacillus Species. Am. Soc. Microbiol. 1987, 53, 224–227. [Google Scholar] [CrossRef] [PubMed]
- Palejwala, S.; Desai, J.D. Production of an extracellular emulsifier by a gram-negative bacterium. Biotechnol. Lett. 1989, 11, 115–118. [Google Scholar] [CrossRef]
- Passeri, A.; Schmidt, M.; Haffner, T.; Wray, V.; Lang, S.; Wagner, F. Marine biosurfactants. IV. Production, characterization and biosynthesis of an anionic glucose lipid from the marine bacterial strain MM1. Appl. Microbiol. Biotechnol. 1992, 37, 281–286. [Google Scholar] [CrossRef]
- Hommel, R.K.; Ratledge, C. Biosynthetic Mechanisms of Low Molecular Weight Surfactants and Their Precursor Molecules. In Biosurfactants; Kosaric, N., Ed.; Marcel Dekker, Inc.: New York, NY, USA, 1993; pp. 3–63. [Google Scholar]
- Zuckerberg, A.; Diver, A.; Peeri, Z.; Gutnick, D.L.; Rosenberg, E. Emulsifier of Arthrobacter RAG-1: Chemical and Physical Properties. Appl. Environ. Microbiol. 1979, 37, 414–420. [Google Scholar] [CrossRef]
- Rosas-Galván, N.S.; Martínez-Morales, F.; Marquina-Bahena, S.; Tinoco-Valencia, R.; Serrano-Carreón, L.; Bertrand, B.; León-Rodríguez, R.; Guzmán-Aparicio, J.; Alvaréz-Berber, L.; Trejo-Hernández, M.D.R. Improved production, purification, and characterization of biosurfactants produced bySerratia marcescensSM3 and its isogenic SMRG-5 strain. Biotechnol. Appl. Biochem. 2018, 65, 690–700. [Google Scholar] [CrossRef]
- Desai, J.D.; Banat, I.M. Microbial production of Surfactant and Their Commercial Potential. Microbiol. Mol. Biol. Rev. 1997, 61, 47–64. [Google Scholar]
- Perfumo, A.; Smyth, T.J.P.; Marchant, R.; Banat, I.M. Production and roles of biosurfactant and bioemulsifiers in accessing hydrophobic substrates. In Microbiology of Hydrocarbons, Oils, Lipids and Derived Compounds; Kenneth, N.T., Ed.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 1502–1512. [Google Scholar]
- Sarubbo, L.A.; Sobrinho, H.B.d.S.; Luna, J.M.d.; Rufino, R.D.; Porto, A.L.F. Assessment of toxicity of a biosurfactant from Candida sphaerica UCP 0995 cultivated with industrial residues in a bioreactor. Electron. J. Biotechnol. 2013, 16, 4. [Google Scholar] [CrossRef]
- Singh, B.R.; Dwivedi, S.; Al-Khedhairy, A.A.; Musarrat, J. Synthesis of stable cadmium sulfide nanoparticles using surfactin produced by Bacillus amyloliquifaciens strain KSU-109. Colloids Surf. B Biointerfaces 2011, 85, 207–213. [Google Scholar] [CrossRef]
- Abalos, A.; Pinazo, A.; Infante, M.R.; Casals, M.; García, F.; Manresa, A. Physicochemical and Antimicrobial Properties of New Rhamnolipids Produced by Pseudomonas aeruginosa AT10 from Soybean Oil Refinery Wastes. ACS Publ. 2001, 17, 1367–1371. [Google Scholar] [CrossRef]
- Mulligan, C.N.; Mahmourides, G.; Gibbs, B.F. The influence of phosphate metabolism on biosurfactant production by Pseudomonas aeruginosa. J. Biotechnol. 1989, 12, 199–209. [Google Scholar] [CrossRef]
- Costa, S.G.V.A.O.; Nitschke, M.; Lépine, F.; Déziel, E.; Contiero, J. Structure, properties and applications of rhamnolipids produced by Pseudomonas aeruginosa L2-1 from cassava wastewater. Process Biochem. 2010, 45, 1511–1516. [Google Scholar] [CrossRef]
- Magalhães, L.; Nitschke, M. Antimicrobial activity of rhamnolipids against Listeria monocytogenes and their synergistic interaction with nisin. Food Control 2013, 29, 138–142. [Google Scholar] [CrossRef]
- Pornsunthorntawee, O.; Wongpanit, P.; Rujiravanit, R. Rhamnolipid Biosurfactants: Production and their Potential in Environmental Biotechnology. Biosurfactants 2010, 672, 211–221. [Google Scholar] [CrossRef]
- Toledo, F.L.; Gonzalez-Lopez, J.; Calvo, C. Production of bioemulsifier by Bacillus subtilis, Alcaligenes faecalis and Enterobacter species in liquid culture. Bioresour. Technol. 2008, 99, 8470–8475. [Google Scholar] [CrossRef]
- Zawawi, R.B.M. Production of Biosurfactant by Locally Isolated Bacteria from Petrochemical Waste. Master’s Thesis, Universiti Teknologi Malaysia, Skudai, Malaysia, 2005. [Google Scholar]
- Parry, A.J.; Parry, N.J.; Peilow, C.; Stevenson, P.S. Combinations of Rhamnolipids and Enzymes for Improved Cleaning. EP 2596087 A1, 16 December 2015. [Google Scholar]
- Takenaka, S.; Tonoki, T.; Taira, K.; Murakami, S.; Aoki, K. Adaptation of Pseudomonas sp. strain 7–6 to quaternary ammonium compounds and their degradation via dual pathways. Appl. Environ. Microbiol. 2007, 73, 1797–1802. [Google Scholar] [CrossRef]
- Shen, H.-H.; Lin, T.-W.; Thomas, R.K.; Taylor, D.J.F.; Penfold, J. Surfactin Structures at Interfaces and in Solution: The Effect of pH and Cations. J. Phys. Chem. B 2011, 115, 4427–4435. [Google Scholar] [CrossRef]
- Wu, Y.-S.; Ngai, S.-C.; Goh, B.-H.; Chan, K.-G.; Lee, L.-H.; Chuah, L.-H. Anticancer Activities of Surfactin and Potential Application of Nanotechnology Assisted Surfactin Delivery. Front. Pharmacol. 2017, 8, 761. [Google Scholar] [CrossRef]
- Morita, T.; Fukuoka, T.; Imura, T.; Kitamoto, D. Glycolipid Biosurfactants. Ref. Modul. Chem. Mol. Sci. Chem. Eng. 2016. [Google Scholar] [CrossRef]
- Gorin, P.A.J.; Spencer, J.F.T.; Tulloch, A.P. Tulloch Hydroxy Fatty Acid Glycosides of Sophorose From Torulopsis Magnoliae Can. J. Chem. 1961, 39, 846–855. [Google Scholar] [CrossRef]
- Castelein, M.; Verbruggen, F.; Van Renterghem, L.; Spooren, J.; Yurramendi, L.; Du Laing, G.; Boon, N.; Soetaert, W.; Hennebel, T.; Roelants, S.; et al. Bioleaching of metals from secondary materials using glycolipid biosurfactants. Miner. Engineering 2021, 163, 106665. [Google Scholar] [CrossRef]
- Price, N.P.J.; Ray, K.J.; Vermillion, K.E.; Dunlap, C.A.; Kurtzman, C.P. Structural characterization of novel sophorolipid biosurfactants from a newly identified species of Candida yeast. Carbohydr. Res. 2012, 348, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Van Renterghem, L.; Roelants, S.L.K.W.; Baccile, N.; Uyttersprot, K.; Taelman, M.C.; Everaert, B.; Mincke, S.; Ledegen, S.; Debrouwer, S.; Scholtens, K.; et al. From lab to market: An integrated bioprocess design approach for new-to-nature biosurfactants produced by Starmerella bombicola. Biotechnol. Bioeng. 2018, 115, 1195–1206. [Google Scholar] [CrossRef] [PubMed]
- Van Bogaert, I.N.A.; Saerens, K.; De Muynck, C.; Develter, D.; Soetaert, W.; Vandamme, E.J. Microbial production and application of sophorolipids. Appl. Microbiol. Biotechnol. 2007, 76, 23–34. [Google Scholar] [CrossRef]
- Global Market Insights, Inc. Sophorolipids Market—Global Industry Analysis, Size, Share, Growth, Trends and Forecast 2016–2023. J. Clean. Product. 2017, 100, 149. [Google Scholar]
- Cooper, D.G.; Paddock, D.A. Production of a Biosurfactant from Torulopsis bombicola. Appl. Environ. Microbiol. 1984, 47, 173–176. [Google Scholar] [CrossRef]
- Kitamoto, D. Functions and Potential Applications of Glycolipid Biosurfactants—From Energy-Saving Materials to Gene Delivery Carriers. J. Biosci. Bioeng. 2002, 94, 187–201. [Google Scholar] [CrossRef]
- Joshi-Navare, K.; Prabhune, A. A Biosurfactant-Sophorolipid Acts in Synergy with Antibiotics to Enhance Their Efficiency. BioMed Res. Int. 2013, 2013, 512495. [Google Scholar] [CrossRef]
- Kim, K.; Dalsoo, Y.; Youngbum, K.; Baekseok, L.; Doonhoon, S.; Eun-Ki, K.I.M. Characteristics of Sophorolipid as an Antimicrobial Agent. J. Microbiol. Biotechnol. 2002, 12, 235–241. [Google Scholar]
- Kurtzman, C.P.; Price, N.P.J.; Ray, K.J.; Kuo, T.-M. Production of Sophorolipid Biosurfactants by Multiple Species of The Starmerella (Candida) Bombicola Yeast Clade. FEMS Microbiol. Lett. 2010, 311, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Raza, Z.A.; Khan, M.S.; Khalid, Z.M. Evaluation of distant carbon sources in biosurfactant production by a gamma ray-induced Pseudomonas putida mutant. Process Biochem. 2006, 42, 686–692. [Google Scholar] [CrossRef]
- Lydon, H.L.; Baccile, N.; Callaghan, B.; Marchant, R.; Mitchell, C.A.; Banat, I.M. Adjuvant antibiotic activity of acidic sophorolipids with potential for facilitating wound healing. Antimicrob. Agents Chemother. 2017, 61, e02547-16. [Google Scholar] [CrossRef] [PubMed]
- Scott, M.J.; Jones, M.N. The biodegradation of surfactants in the environment. Biochim. Biophys. Acta 2000, 1508, 235–251. [Google Scholar] [CrossRef]
- Pekin, G.; Vardar-Sukan, P.; Kosaric, N. Production of sophorolipids from Candida bombicola ATCC 22214 using Turkish corn oil and honey. Eng. Life Sci. 2005, 5, 357–362. [Google Scholar] [CrossRef]
- Sanchita, K.; Pritisnigdha, P. Production and functional characterization of food compatible biosurfactants. Appl. Food Sci. J. 2019, 3, 1–4. [Google Scholar]
- Ramani, K.; Jain, S.C.; Mandal, A.B.; Sekaran, G. Microbial induced lipoprotein biosurfactant from slaughterhouse lipid waste and its application to the removal of metal ions from aqueous solution. Colloids Surf. B Biointerfaces 2012, 97, 254–263. [Google Scholar] [CrossRef]
- Zeraik, A.E.; Nitschke, M. Biosurfactants as agents to reduce adhesion of pathogenic bacteria to polystyrene surfaces: Effect of temperature and hydrophobicity. Curr. Microbiol. 2010, 61, 554. [Google Scholar] [CrossRef]
- Kim, P.I.; Ryu, J.; Kim, Y.H.; Chi, Y.T. Production of biosurfactant lipopeptides Iturin A, fengycin and surfactin A from Bacillus subtilis CMB32 for control of Colletotrichum gloeosporioides. J. Microbiol. Biotechnol. 2010, 20, 138–145. [Google Scholar] [CrossRef]
- Arima, K.; Kakinuma, A.; Tamura, G. Surfactin, a crystalline peptide lipid surfactant produced by Bacillus subtilis: Isolation, characterization and its inhibition of fibrin clot formation. Biochem. Biophys. Res. Commun. 1968, 3, 488–494. [Google Scholar] [CrossRef]
- Mulligan, C.N.; Gibbs, B.F. Types, Production and Applications of Biosurfactants. Proc. Indian Natl. Sci. Acad. 2004, B70, 31–55. [Google Scholar]
- Cameotra, S.S.; Makkar, R.S. Recent applications of biosurfactants as biological and immunological molecules. Curr. Opin. Microbiol. 2004, 7, 262–266. [Google Scholar] [CrossRef] [PubMed]
- Sen, S.; Borah, S.N.; Bora, A.; Deka, S. Production, characterization, and antifungal activity of a biosurfactant produced by Rhodotorula babjevae YS3. Microb. Cell Factories 2017, 16, 95. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.-C.; Juang, R.-S.; Wei, Y.-H. Applications of a lipopeptide biosurfactant, surfactin, produced by microorganisms. Biochem. Eng. J. 2015, 103, 158–169. [Google Scholar] [CrossRef]
- Wang, X.; Gonga, L.; Liang, S.; Han, X.; Zhua, C.; Li, Y. Algicidal activity of rhamnolipid biosurfactants produced by Pseudomonas aeruginosa. Harmful Algae 2005, 4, 433–443. [Google Scholar] [CrossRef]
- Yea, D.; Jo, S.; Lim, J. Synthesis of Eco-friendly Nano-Structured Biosurfactants from Vegetable Oil Sources and Characterization of Their Interfacial Properties for Cosmetic Applications. MRS Adv. 2019, 4, 377–384. [Google Scholar] [CrossRef]
- Zhang, J.; Xue, Q.; Gao, H. Production of lipopeptide biosurfactants by Bacillus atrophaeus 5-2a and their potential use in microbial enhanced oil recovery. Microb. Cell Fact. 2016, 15, 168. [Google Scholar] [CrossRef]
- Williams, W.; Kunorozva, L.; Klaiber, I.; Henkel, M.; Pfannstiel, J.; Van Zyl, L.J.; Hausmann, R.; Burger, A.; Trindade, M. Novel metagenome-derived ornithine lipids identified by functional screening for biosurfactants. Appl. Microbiol. Biotechnol. 2019, 103, 4429–4441. [Google Scholar] [CrossRef]
- Vyast, T.K.; Dave, B.P. Production of biosurfactant by Nocardia otitidiscaviarum and its role in biodegradation of crude oil. Int. J. Environ. Sci. Technol. 2011, 8, 425–432. [Google Scholar] [CrossRef]
- Singh, A.; Hamme, J.D.V.; Ward, O.P. Surfactants in microbiology and biotechnology: Part 2: Application aspects. Biotechnol. Adv. 2007, 25, 99–121. [Google Scholar] [CrossRef]
- Bo-Zhong, M.; Fei, D.; Zhou, G.-W.; Yu, Z.-Q.; Gang, H.-G.; Liu, J.-F.; Yang, S.-Z.; Ye, R.-Q. Low-Toxic and Nonirritant Biosurfactant Surfactin and its Performances in Detergent Formulations. J. Surfactants Deterg. 2020, 23, 109–118. [Google Scholar]
- Desai, J.D.; Desai, A.J. Production of Biosurfactant. In Biosurfactants Production, Properties and Applications; Kosaric, N., Ed.; Marcel Dekker, Inc.: New York, NY, USA, 1993; pp. 65–97. [Google Scholar]
- Thanomsub, B.; Pumeechockchai, W.; Limtrakul, A.; Panarat, A.; Petchleelaha, W.; Nitoda, T.; Kanzaki, H. Chemical structures and biological activities of rhamnolipids produced by Pseudomonas aeruginosa B189 isolated from milk factory waste. Bioresour. Technol. 2006, 98, 1149–1153. [Google Scholar] [CrossRef] [PubMed]
- Whanga, L.-M.; Liu, P.-W.G.; Maa, C.-C.; Cheng, S.-S. Application of biosurfactants, rhamnolipid, and surfactin, for enhanced biodegradation of diesel-contaminated water and soil. J. Hazard. Mater. 2008, 151, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Beeba, J.L.; Umbreit, W.W. Extracellular lipid of Thiobacillus thiooxidans. J. Bacteriol. 1971, 108, 612–615. [Google Scholar] [CrossRef]
- Cooper, D.G.; Zajic, J.E.; Gerson, D.F. Production of surface-active lipids by Corynebacterium lepus. Appl. Environ. Microbiol. 1979, 37, 4–10. [Google Scholar] [CrossRef]
- Kretschmer, A.; Bock, H.; Wagner, F. Chemical and Physical Characterization of Interfacial-Active Lipids from Rhodococcus erythropolis grown on N-alkanes. Appl. Environ. Microbiol. 1982, 44, 864–870. [Google Scholar] [CrossRef]
- Janek, T.; Lukaszewicz, M.; Krasowska, A. Identification and characterization of biosurfactants produced by the Arctic bacterium Pseudomonas putida BD2. Colloids Surf. B Biointerfaces 2013, 110, 379–386. [Google Scholar] [CrossRef]
- Adamu, A.; Ijah, U.J.; Riskuwa, M.L.; Ismail, H.Y.; Ibrahim, U.B. Study on Biosurfactant Production by Two Bacillus Species. Int. J. Sci. Res. Knowl. 2015, 3, 013–020. [Google Scholar] [CrossRef]
- Janek, T.; Krasowska, A.; Czyżnikowska, Z.; Łukaszewicz, M. Trehalose Lipid Biosurfactant Reduces Adhesion of Microbial Pathogens to Polystyrene and Silicone Surfaces: An Experimental and Computational Approach. Front. Microbiol. 2018, 9, 2441. [Google Scholar] [CrossRef]
- Vecino, X.; Ferreira, A.; Ferreira, D.; Cruz, J.M.; Moldes, A.B.; Rodrigues, L.R. Novel cosmetic formulations containing a biosurfactant from Lactobacillus paracasei. Colloids Surf. B Biointerfaces 2017, 155, 522–529. [Google Scholar]
- Zokaei, E.; Badoei-dalfrad, A.; Ansari, M.; Karami, Z.; Eslaminejad, T.; Nematollahi-Mahani, S.N. Therapeutic potential of DNAzyme loaded on chitosan/cyclodextrin nanoparticle to recovery of chemosensitivity in the mcf-7 cell line. Appl. Biochem. Biotechnol. 2018, 183, 126–136. [Google Scholar] [CrossRef]
- Lang, S. Biological amphiphiles (microbial biosurfactants). Curr. Opin. Colloid Interface Sci. 2002, 7, 12–20. [Google Scholar] [CrossRef]
- Hatha, A.A.M.; Edward, G.; Rahman, K.S.M.P. Microbial biosurfactants-review. J. Mar. Atmos. Res. 2007, 3, 1–17. [Google Scholar]
- Amani, H.; Kariminezhad, H. Study on emulsification of crude oil in water using emulsan biosurfactant for pipeline transportation. Pet. Sci. Technol. 2016, 34, 216–222. [Google Scholar] [CrossRef]
- Choi, J.W.; Choi, H.G.; Lee, W.H. Effects of ethanol and phosphate on emulsan production by Acinetobacter calcoaceticus RAG-1. J. Biotechnol. 1996, 45, 217–225. [Google Scholar] [CrossRef]
- Rodriguez-López, L.; Vecino, X.; Barbosa-Pereira, L.; Moldes, A.B.; Cruz, J.M. A multifunctional extract from corn steep liquor: Antioxidant and surfactant activities. Food Funct. 2016, 7, 3724–3732. [Google Scholar] [CrossRef]
- Kaplan, N.; Zosim, Z.; Rosenberg, E. Reconstitution of emulsifying activity of Acinetobacter calcoaceticus BD5 emulsan by using pure polysaccharide and proteins. Appl. Environ. Microbiol. 1987, 53, 440–446. [Google Scholar] [CrossRef]
- Chakrabarti, S. Bacterial Biosurfactant: Characterization, Antimicrobial and Metal Remediation Properties. Master’s Thesis, NIT Rourkela, Rourkela, India, 2012. [Google Scholar]
- Campos, J.M.; Stamford, T.L.; Sarubbo, L.A.; de Luna, J.M.; Rufino, R.D.; Banat, I.M. Microbial biosurfactants as additives for food industries. Biotechnol. Prog. 2013, 29, 1097–1108. [Google Scholar] [CrossRef]
- Cameron, D.R.; Cooper, D.G.; Neufeld, R.J. The mannoprotein of Saccharomyces cerevisiae is an effective bioemulsifier. Appl. Environ. Microbiol. 1988, 54, 1420–1425. [Google Scholar] [CrossRef]
- Casanova, M.; Ribot, J.L.L.; Martínez, J.P.; Sentandreu, R. Characterization of cell wall proteins from yeast and mycelial cells of Candida albicans by labelling with biotin: Comparison with other techniques. Infect. Immun. 1992, 60, 4898–4906. [Google Scholar] [CrossRef]
- Oliveira, M.C.; Figueiredo-Lima, D.F.; Faria Filho, D.E.; Marques, R.H.; Moraes, V.M.B. Effect of mannanoligosaccharides and/or enzymes on antibody titers against infectious bursal and Newcastle disease viruses. Arq. Bras. Med. Vet. 2009, 61, 6–11. [Google Scholar] [CrossRef]
- Lukondeh, T.; Ashbolt, N.; Rogers, P. Evaluation of Kluyveromyces marxianus F11 510700 grown on a lactose-based medium as a source of natural bioemulsifier. J. Ind. Microbiol. Biotechnol. 2003, 30, 715–730. [Google Scholar] [CrossRef] [PubMed]
- Alcantara, V.A.; Pajares, I.G.; Simbahan, J.F.; Edding, S.N. Downstream recovery and purification of a bioemulsifier from Saccharomyces cerevisiae 2031. Phil. Agric. Sci. 2014, 96, 349–359. [Google Scholar]
- Jagtap, S.; Yavankar, S.; Pardesi, K.; Chopade, B. Production of bioemulsifier by Acinetobacter sp. from healthy human skin of tribal population. Ind. J. Expt. Biol. 2010, 48, 70–76. [Google Scholar]
- Kappeli, O.; Finnerty, W.R. Partition of alkane by an extracellular vesicle derived from hexadecane-grow Acinetobacter. J. Bacteriol. 1979, 140, 707–712. [Google Scholar] [CrossRef]
- Fiechter, A. Biosurfactants: Moving towards industrial application. Trends Biotechnol. 1992, 10, 208–218. [Google Scholar] [CrossRef]
- Banat, I.M. Biosurfactants Production and Possible Uses in Microbial Enhanced Oil Recovery and Oil Pollution Remediation: A Review. Bioresour. Technol. 1995, 51, 1–12. [Google Scholar] [CrossRef]
- Hommel, R.K. Formation and Physiology Role of Biosurfactants Produced by Hydrocarbon-utilizing Microorganisms. Biodegradation 1990, 1, 107–119. [Google Scholar] [CrossRef]
- Pornsunthorntawee, O.; Chavadej, S.; Rujiravanit, R. Solution properties and vesicle formation of rhamnolipid biosurfactants produced by Pseudomonas aeruginosa SP4. Colloids Surf. B Biointerfaces 2009, 72, 6–15. [Google Scholar] [CrossRef]
- Gibson, D.; White, A.; Snyder, S.; Martin, S.; Heiss, C.; Azadi, P. Salmonella produces an O-antigen capsule regulated by AgfD and important for environmental persistence. J. Bacteriol. 2006, 188, 7722–7730. [Google Scholar] [CrossRef]
- Araujo, D. Rhamnolipid and surfactin inhibit Listeria monocytogenes adhesion. Food Res. Int. 2011, 44, 481–488. [Google Scholar] [CrossRef]
- Mukherjee, A.K.; Das, K. Microbial Surfactants and Their Potential Applications: An Overview. In Biosurfactants; Sen, R., Ed.; Advances in Experimental Medicine and Biology; Springer: New York, NY, USA, 2010; Volume 672. [Google Scholar] [CrossRef]
- Fenibo, E.O.; Douglas, S.I.; Stanley, H.O. A Review on Microbial Surfactants: Production, Classifications, Properties and Characterization. J. Adv. Microbiol. 2019, 18, 1–22. [Google Scholar] [CrossRef]
- Itrich, N.R.; McDonough, K.M.; van Ginkel, C.G.; Bisinger, E.C.; LePage, J.N.; Schaefer, E.C.; Menzies, J.Z.; Casteel, K.D.; Federle, T.W. Widespread microbial adaptation to l-Glutamate-N, N-diacetate (L-GLDA) following its market introduction in a consumer cleaning product. Environ. Sci. Technol. 2015, 49, 13314–13321. [Google Scholar] [CrossRef]
- Ni’matuzahroh; Sari, S.K.; Trikurniadewi, N.; Pusfita, A.D.; INingrum, P.; Ibrahim, S.N.M.M.; Fatimah; Surtiningsih, T. Utilization of Rice Straw Hydrolysis Product of Penicillium sp. H9 as A Substrate of Biosurfactant Production by LII61 Hydrocarbonoclastic Bacteria. IOP Conf. Ser. Earth Environ. Sci. 2018, 217, 012028. [Google Scholar] [CrossRef]
- Walter, V.; Syldatk, C.; Hausmann, R. Screening Concepts for the Isolation of Bio-surfactant Producing Microorganisms. In Madame Curie Bioscience Database [Internet]; Landes Bioscience: Austin, TX, USA, 2000. [Google Scholar]
- Rahman, K.; Rahman, T.; McClean, S.; Marchant, R.; Banat, I.M. Rhamnolipid biosurfactant production by strains of Pseudomonas aeruginosa using low-cost raw materials. Biotechnol. Prog. 2002, 18, 1277–1281. [Google Scholar] [CrossRef]
- Patowary, K.; Patowary, R.; Kalita, M.C.; Deka, S. Characterization of Biosurfactant Produced during Degradation of Hydrocarbons Using Crude Oil As Sole Source of Carbon. Front. Microbiol. 2017, 8, 279. [Google Scholar] [CrossRef]
- Abdeli, F.; Rigane, G.; Ben Salem, R.; El Arbi, M.; Aifa, S.; Cherif, S. Use of Surfactants and Biosurfactants in Oil Recovery Processing and Cellulose Hydrolysis. J. Bacteriol. Mycol. 2019, 6, 1114. [Google Scholar]
- Franzetti, A.; Gandolfi, I.; Bestetti, G.; Smyth, T.J.P.; Banat, I.M. Production and applications of trehalose lipid biosurfactants. Eur. J. Lipid Sci. Technol. 2010, 112, 617–627. [Google Scholar] [CrossRef]
- Shekhar, S.; Sundaramanickam, A.; Balasubramanian, T. Biosurfactant Producing Microbes and their Potential Applications: A Review. Crit. Rev. Environ. Sci. Technol. 2014, 45, 1522–1554. [Google Scholar] [CrossRef]
- Elshafie, A.E.; Joshi, S.; Al-Wahaibi, Y.; Al-Bemani, A.; Al-Bahry, S.N.; Al-Maqbali, D.; Banat, I.M. Sophorolipids Production by Candida bombicola ATCC 22214 and its Potential Application in Microbial Enhanced Oil Recovery. Front. Microbiol. 2015, 6, 1324. [Google Scholar] [CrossRef]
- Kuraoka, T.; Yamada, T.; Ishiyama, A.; Oyamada, H.; Ogawa, Y.; Kobayashi, H. Determination of α-1,3-Linked Mannose Residue in the Cell Wall Mannan of Candida tropicalis NBRC 1400 Strain. Adv. Microbiol. 2020, 10, 14–26. [Google Scholar] [CrossRef][Green Version]
- Alizadeh-Sani, M.; Hamishehkar, H.; Khezerlou, A.; Azizi-Lalabadi, M.; Azadi, Y.; Nattagh-Eshtivani, E.; Fasihi, M.; Ghavami, A.; Aynehchi, A.; Ehsani, A. Bioemulsifiers Derived from Microorganisms: Applications in the Drug and Food Industry. Adv. Pharm. Bull. 2018, 8, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Rang, R.; Liu, G.; Chen, T.; Li, S.; An, L.; Zhang, G.; Li, G.; Chang, S.; Zhang, W.; Chen, X.; et al. Characterization of the genome of a Nocardia strain isolated from soils in the Qinghai-Tibetan Plateau that specifically degrades crude oil and of this biodegradation. Genomics 2019, 111, 356–366. [Google Scholar] [CrossRef]
- Santos, E.F.; Teixeira, M.F.S.; Converti, A.; Porto, A.L.F.; Sarubbo, L.A. Production of a new lipoprotein biosurfactant by Streptomyces sp. DPUA1566 isolated from lichens collected in the Brazilian Amazon using agroindustry wastes. Biocatal. Agric. Biotechnol. 2019, 17, 142–150. [Google Scholar] [CrossRef]
- Cecchi, G.; Cutroneo, L.; Di Piazza, S.; Besio, G.; Capello, M.; Zotti, M. Port Sediments: Problem or Resource? A Review Concerning the Treatment and Decontamination of Port Sediments by Fungi and Bacteria. Microorganisms 2021, 9, 1279. [Google Scholar] [CrossRef]
- Yagüe, G.; Segovia, M.; Valero-Guillen, P.L. Phospholipid composition of several clinically relevant Corynebacterium species as determined by mass spectrometry: An unusual fatty acyl moiety is present in inositol-containing phospholipids of Corynebacterium urealyticum. Microbiology 2003, 149, 1675–1685. [Google Scholar] [CrossRef]
- Wang, C.L.; Liu, C.; Niu, L.L.; Wang, L.R.; Hou, L.H.; Cao, X.H. Surfactin-induced apoptosis through ROS-ERS-Ca2+-ERK pathways in HepG2 cells. Cell Biochem. Biophys. 2013, 67, 1443–1449. [Google Scholar] [CrossRef]
- Cappelletti, M.; Presentato, A.; Piacenza, E.; Firrincieli, A.; Turner, R.J.; Zannoni, D. Biotechnology of Rhodococcus for the production of valuable compounds. Appl. Microbiol. Biotechnol. 2020, 104, 8567–8594. [Google Scholar] [CrossRef]
- Cooper, D.G. Biosurfactants. Microbiol. Sci. 1986, 3, 145–149. [Google Scholar]
- Smyth, T.J.P.; Perfumo, A.; Marchant, R.; Banat, I.M. Isolation and Analysis of Lipopeptide and high molecular weight biosurfactant. In Handbook of Hydrocarbon and Lipid Microbiology; Timmis, K.N., Ed.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 3687–3704. [Google Scholar]
- Butt, H.J.; Graf, K.; Kappl, M. Surfactants, micelles, emulsions, and foams. In Physics and Chemistry of Interfaces; Butt, H.-J., Graf, K., Kappl, M., Eds.; WEILY-VCH Verlag GmbH & Co. KgaA: Weinheim, Germany, 2004; pp. 246–279. [Google Scholar]
- Andersen, K.K.; Otzen, D.E. Folding of outer membrane proteinA in the anionic biosurfactant rhamnolipid. FEBS Lett. 2014, 588, 1955–1960. [Google Scholar] [CrossRef]
- Rodrigues, L.R. Microbial surfactants: Fundamentals and applicability in the formulation of nano-sized drug delivery vectors. J. Colloid Interface Sci. 2015, 449, 304–316. [Google Scholar] [CrossRef] [PubMed]
- Urum, K.; Grigson, S.; Pekdemir, T.; McMenamy, S. A comparison of the efficiency of different surfactants for removal of crude oil from contaminated soils. Chemosphere 2006, 62, 1403–1410. [Google Scholar] [CrossRef] [PubMed]
- Yeh, M.S.; Wei, Y.H.; Chang, J.S. Enhanced production of surfactin from Bacillus subtilis by addition of solid carriers. Biotechnol. Prog. 2005, 21, 1329–1334. [Google Scholar] [CrossRef] [PubMed]
- McInerney, M.J.; Maudgalya, S.K.; Knapp, R.; Folmsbee, M. Development of Biosurfactant-Mediated Oil Recovery in Model Porous Systems and Computer Simulations of Biosurfactant-Mediated Oil Recovery; University of Oklahoma: Norman, OK, USA.
- Purwasena, I.A.; Astuti, D.I.; Syukron, M.; Amaniyah, M.; Sugai, Y. Stability test of biosurfactant produced by Bacillus licheniformis DS1 using experimental design and its application for MEOR. J. Pet. Sci. Eng. 2019, 183, 106383. [Google Scholar] [CrossRef]
- Zahir, Z.A.; Arshad, M.; William, T.; Frankenberger, W.T., Jr. Plant growth promoting rhizobacteria: Applications and perspectives in agriculture. Adv. Agron. 2004, 81, 97–168. [Google Scholar]
- Rosenberg, E.; Ron, E.Z. High- and low-molecular-mass microbial surfactants. Appl. Microbiol. Biotechnol. 1999, 52, 154–162. [Google Scholar] [CrossRef]
- Luna, J.M.; Rufino, R.D.; Sarubbo, L.A.; Campos-Takaki, G.M.M. Characterisation, surface properties and biological activity of a biosurfactant produced from industrial waste by Candida sphaerica UCP0995 for application in the petroleum industry. Colloids Surf. B Biointerfaces 2013, 102, 202–209. [Google Scholar] [CrossRef]
- Amodu, O.S.; Ntwampe, S.K.; Ojumu, T.V. Emulsification of hydrocarbons by biosurfactant: Exclusive use of agrowaste. BioResources 2014, 9, 3508–3525. [Google Scholar] [CrossRef]
- Rufino, R.D.; Sarubbo, L.A.; Neto, B.B.; Campos-Takaki, G.M. Experimental design for the production of tensio-active agent by Candida lipolytica. J. Ind. Microbiol. Biotechnol. 2008, 35, 907–914. [Google Scholar] [CrossRef]
- Hu, X.; Wang, C.; Wang, P. Optimization and characterization of biosurfactant production from marine Vibrio sp. strain 3B-2. Front. Microbiology 2015, 6, 976. [Google Scholar]
- Muthusamy, K.; Gopalakrishnan, S.; Ravi, T.K.; Sivachidambaram, P. Biosurfactants: Properties, commercial production and application. Curr. Sci. 2008, 94, 736–747. [Google Scholar]
- Dasgupta, D.; Ghosh, R.; Sengupta, T.K. Biofilm-mediated enhanced crude oil degradation by newly isolated Pseudomonas species. ISRN Biotechnol. 2013, 2013, 250749. [Google Scholar] [CrossRef] [PubMed]
- Rahman, P.K.S.M.; Uzoigwe, C.; Burgess, J.G.; Ennis, C.J. Bioemulsifiers are not biosurfactants and require different screening approaches. Front. Microbiol. 2015, 6, 245. [Google Scholar]
- Vijayakumar, S.; Saravanan, V. In vitro Cytotoxicity and Antimicrobial Activity of Biosurfactant Produced by Pseudomonas aeruginosa Strain PB3A. Asian J. Sci. Res. 2015, 8, 510–518. [Google Scholar] [CrossRef][Green Version]
- Amaral, P.F.F.; da Silva, J.M.; Lehocky, M.; Barros-Timmons, A.M.V.; Coelho, M.A.Z.; Marrucho, I.M. Production and characterization of a bioemulsifier from Yarrowia lipolytica. Proc. Biochem. 2006, 41, 1894–1898. [Google Scholar] [CrossRef]
- Monteiro, A.S.; Bonfim, M.R.; Domingues, V.S.; Correa, A., Jr.; Siqueira, E.P.; Zani, C.L. Identification and characterization of bioemulsifier-producing yeasts isolated from effluents of a diary industry. Bioresour. Technol. 2010, 101, 5186–5193. [Google Scholar] [CrossRef]
- Thies, S.; Rausch, S.C.; Kovacic, F.; Schmidt-Thaler, A.; Wilhelm, S.; Rosenau, F.; Daniel, R.; Streit, W.; Pietruszka, J.; Jaeger, K.-E. Metagenomic discovery of novel enzymes and biosurfactants in a slaughterhouse biofilm microbial community. Sci. Rep. 2016, 6, 27035. [Google Scholar] [CrossRef]
- Jain, R.M.; Mody, K.; Joshi, N.; Mishra, A.; Jha, B. Production and structural characterization of biosurfactant produced by an alkaliphilic bacterium, Klebsiella sp.: Evaluation of different carbon sources. Colloids Surf. B Biointerf. 2013, 108, 199–204. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, C.; Dong, B. Anti-inflammatory Activity and Mechanism of Surfactin in Lipopolysaccharide-Activated Macrophages. Inflammation 2015, 38, 756–764. [Google Scholar] [CrossRef]
- Panjiar, N.; Sachan, S.G.; Sachan, A. Screening of bioemulsifier-producing microorganisms isolated from oil-contaminated sites. Ann. Microbiol. 2014, 65, 753–764. [Google Scholar] [CrossRef]
- Campos, J.M.; Stamford, T.L.M.; Sarubbo, L.A. Production of a bioemulsifier with potential applications in the food industry. Appl. Biochem. Biotech. 2014, 172, 3234–3252. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, E.F.; Hassan, S.S. Antimicrobial activity of a bioemulsifier produced by Serratia marcescens S10. J. AL-Nahrain Univ. 2013, 16, 147–155. [Google Scholar] [CrossRef]
- Nalini, S.; Parthasarathi, R. Optimization of rhamnolipid biosurfactant production from Serratia rubidaea SNAU02 under solid-state fermentation and its biocontrol efficacy against Fusarium wilt of eggplant. Ann. Agrar. Sci. 2018, 16, 108–113. [Google Scholar] [CrossRef]
- Kachholz, T.T.; Schlingmann, M.M. Possible food and agriculture application of microbial surfactants: An assessment. In Biosurfactants and Biotechnology; Kosaric, N., Cairns, W.L., Gray, N.C.C., Eds.; Surfactant Sciences Series; Dekker: Basel, Switzerland, 1987; Volume 25, pp. 183–210. [Google Scholar]
- Gamalero, E.; Glick, B.R. Mechanisms Used by Plant Growth- promoting Bacteria. Chapter 2. In Bacteria in Agrobiology: Plant Nutrient Management; Maheshwari, D.K., Ed.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 17–46. [Google Scholar]
- Yuliani, H.; Perdani, M.S.; Savitri, I.; Manurung, M.; Sahlan, M.; Wijanarko, A.; Hermansyah, H. Antimicrobial activity of biosurfactant derived from Bacillus subtilis C19. Energy Procedia 2018, 153, 274–278. [Google Scholar] [CrossRef]
- Shreve, G.S.; Makula, R. Characterization of a New Rhamnolipid Biosurfactant Complex from Pseudomonas Isolate DYNA270. Biomolecules 2019, 9, 885. [Google Scholar] [CrossRef] [PubMed]
- Schenk, T.; Schuphan, I.; Schmidt, B. High-performance liquid chromatographic determination of therhamnolipids produced by Pseudomonas aeruginosa. J. Chromatogr. A 1994, 693, 7–13. [Google Scholar] [CrossRef]
- Takahashi, M.; Morita, T.; Fukuoka, T.; Imura, T.; Kitamoto, D. Glycolipid biosurfactants, mannosylerythritol lipids, show antioxidant and protective effects against H2O2-induced oxidative stress in cultured human skin fibroblasts. J. Oleo Sci. 2012, 61, 457–464. [Google Scholar] [CrossRef]
- Lima, T.M.; Procópio, L.C.; Brandão, F.D.; Carvalho, A.M.; Tótola, M.R.; Borges, A.C. Biodegradability of bacterial surfactants. Biodegradation 2011, 22, 585–592. [Google Scholar] [CrossRef]
- Moldes, A.B.; Paradelo, R.; Rubinos, D.; Devesa-Rey, R.; Cruz, J.M.; Barral, M.T. Ex situ treatment of hydrocarbon-contaminated soil using biosurfactants from Lactobacillus pentosus. J. Agric. Food Chem. 2011, 59, 9443–9447. [Google Scholar] [CrossRef]
- Benincasa, M. Rhamnolipid produced from agroindustrial wastes enhances hydrocarbon biodegradation in contaminated soil. Curr. Microbiol. 2007, 54, 445–449. [Google Scholar] [CrossRef]
- Mnif, I. Improvement of Bread dough quality by Bacillus subtilis SPB1 biosurfactant addition: Optimized extraction using response surface methodology. J. Sci. Food Agric. 2012, 21, 3055. [Google Scholar]
- Nair, A.M.; Rebello, S.; Rishad, K.S.; Asok, A.K.; Jisha, M.S. Biosurfactant Facilitated Biodegradation of Quinalphos at High Concentrations by Pseudomonas aeruginosa Q10. Soil Sediment Contam. Int. J. 2015, 24, 542–553. [Google Scholar] [CrossRef]
- Sachdev, D.P.; Cameotra, S.S. Biosurfactants in agriculture. Appl. Microbiol. Biotechnol. 2013, 97, 1005–1016. [Google Scholar] [CrossRef] [PubMed]
- García-Reyes, S.; Yáñez-Ocampo, G.; Wong-Villarreal, A.; Rajaretinam, R.K.; Thavasimuthu, C.; Patiño, R. Partial characterization of a biosurfactant extracted from Pseudomonas sp. B0406 that enhances the solubility of pesticides. Environ. Technol. 2018, 39, 2622–2631. [Google Scholar] [CrossRef] [PubMed]
- Chopra, A.; Bobate, S.; Rahi, P.; Banpurkar, A.; Mazumder, P.B.; Satpute, S. Pseudomonas aeruginosa RTE4: A Tea Rhizobacterium With Potential for Plant Growth Promotion and Biosurfactant Production. Front. Bioeng. Biotechnol. 2020, 8, 861. [Google Scholar] [CrossRef]
- Rawat, G.; Dhasmana, A.; Kumar, V. Biosurfactants: The next generation biomolecules for diverse applications. Environ. Sustain. 2020, 3, 353–369. [Google Scholar] [CrossRef]
- Gudiña, E.J.; Teixeira, J.A.; Rodrigues, L.R. Biosurfactant-Producing Lactobacilli: Screening, Production Profiles, and Effect of Medium Composition. Appl. Environ. Soil Sci. 2011, 2011, 201254. [Google Scholar] [CrossRef]
- Das, A.J.; Ambust, S.; Kumar, R. Development of Biosurfactant Based Cosmetic Formulation of Toothpaste and Exploring its Efficacy. Adv. Ind. Biotechnol. 2018, 1, 5. [Google Scholar]
- Panadare, D.C.; Rathod, V.K. Applications of waste cooking oil other than biodiesel: A review. Iran. J. Chem. Eng. 2015, 12, 55–76. [Google Scholar]
- Singh, P.; Patil, Y.; Rale, V. Biosurfactant production: Emerging trends and promising strategies. J. Appl. Microbiol. 2019, 126, 2–13. [Google Scholar] [CrossRef]
- Manga, E.B.; Celik, P.A.; Cabuk, A.; Banat, I.M. Biosurfactants: Opportunities for the development of a sustainable future. Curr. Opin. Colloid Interface Sci. 2021, 56, 101514. [Google Scholar] [CrossRef]
- Mardawati, E.; Andoyo, R.; Syukra, K.A.; Kresnowati, M.T.A.P.; Bindar, Y. Production of xylitol from corn cob hydrolysate through acid and enzymatic hydrolysis by yeas. In Proceedings of the 2nd International Conference on Biomass: Toward Sustainable Biomass Utilization for Industrial and Energy Applications, Bogor, Indonesia, 24–25 July 2017; Volume 141, p. 012019. [Google Scholar]
- Ni’Matuzahroh; Sari, S.K.; Trikurniadewi, N.; Ibrahim, S.N.M.M.; Khiftiyah, A.M.; Abidin, A.Z.; Nurhariyati, T.; Fatimah. Bioconversion of agricultural waste hydrolysate from lignocellulolytic mold into biosurfactant by Achromobacter sp. BP(1)5. Biocatal. Agric. Biotechnol. 2020, 24, 101534. [Google Scholar] [CrossRef]
- Mouafo, T.H.; Mbawala, A.; Ndjouenkeu, R. Effect of Different Carbon Sources on Biosurfactants’ Production by Three Strains of Lactobacillus spp. BioMed Res. Int. 2018, 2018, 5034783. [Google Scholar] [CrossRef] [PubMed]
- Kaur, H.P.; Prasad, B.; Kaur, S. A review on applications of biosurfactants produced from unconventional inexpensive wastes in food and agriculture industry. World J. Pharm. Res. 2015, 4, 827–842. [Google Scholar]
- Fracchia, L.; Ceresa, C.; Banat, I.M. Biosurfactants in Cosmetic, Biomedical and Pharmaceutical Industry. In Microbial Biosurfactants and their Environmental and Industrial Applications; Thavasi, R., Banat., I.M., Eds.; CRC Press: Boca Raton, FL, USA, 2018; p. 258. [Google Scholar]
- Satpute, S.K.; Płaza, G.A.; Banpurkar, A.G. Biosurfactants’ production from renewable natural resources: Example of innovativeand smart technology in circular bioeconomy. Manag. Syst. Prod. Eng. 2017, 25, 46–54. [Google Scholar] [CrossRef]
- Muhammad, I.-M.; Mahsa, S.-S. Rhamnolipids: Well-characterized glycolipids with potential broad applicability as biosurfactants. Ind. Biotechnol. 2014, 10, 285–291. [Google Scholar]
- Morita, T.; Fukuoka, T.; Imura, T.; Kitamoto, D. Production of mannosylerythritol lipids and their application in cosmetics. Appl. Microbiol. Biotechnol. 2013, 97, 4691–4700. [Google Scholar] [CrossRef]
- Meena, K.R.; Kanwar, S.S. Lipopeptides as antifungal and antibacterial agents: Applications in food safety and therapeutics. Biomed Res. Int. 2015, 2015, 473050. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Huang, G.H.; An, C.J.; Cheng, G.H.; Wei, J. Effects of freeze-thawing cycles on desorption behaviors of PAH-contaminated soil in the presence of a biosurfactant: A case study in western Canada. Environ. Sci. Processes Impacts 2017, 19, 874–882. [Google Scholar] [CrossRef]
- Banat, I.M.; Samarah, N.; Murad, M.; Horne, R.; Banerjee, S. Biosurfactant production and use in oil tank clean-up. World J. Microbiol. Biotechnol. 1991, 7, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.N.; Li, Q. Microbial production of rhamnolipids using sugars as carbon sources. Microb. Cell Fact. 2018, 17, 89. [Google Scholar] [CrossRef] [PubMed]
- Krepsky, N.; Da Silva, F.S.; Fontana, L.F.; Crapez, M.A. Alternative methodology for isolation of biosurfactant-producing bacteria. Braz. J. Biol. 2007, 67, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Ganesh, A.; Lin, J. Diesel degradation and biosurfactant production by Gram-positive isolates. Afr. J. Biotechnol. 2009, 8, 5847–5854. [Google Scholar]
- Daud, F.N.; Yahya, A.; Salleh, M.M.; Suhaimi, N.; Xuan, C.H. Production of Biosurfactant by locally isolated thermophilic facultatively anaerobic bacteria strain B160. Environmental Applications for Biosurfactants. Environ. Pollut. 1987, 133, 183–198. [Google Scholar]
- Amiriyan, A.; Assadi, M.; Saggadian, V.V.; Noohi, A. Bio emulsion production by Iranian oil reservoirs microorganisms. Iran. J. Health Sci. Eng. 2004, 1, 28–35. [Google Scholar]
- Makkar, R.S.; Rockne, K.J. Comparison of synthetic surfactants and biosurfactants in enhancing biodegradation of polycyclic aromatic hydrocarbons. Environ. Toxicol. Chem. 2009, 22, 2280–2292. [Google Scholar] [CrossRef]
- Deziel, E.; Paquette, G.; Villemur, R.; Lepine, F.; Bisaillon, J. Biosurfactant production by a soil Pseudomonas strain growing on polycyclic aromatic hydrocarbons. Appl. Environ. Microbiol. 1996, 62, 1908–1912. [Google Scholar] [CrossRef] [PubMed]
- Salihu, A.; Abdulkadir, I.; Almustapha, M.N. An investigation for potential development on biosurfactants. Biotechnol. Mol. Biol. Rev. 2009, 3, 111–117. [Google Scholar]
- Wei, Y.-H.; Cheng, C.-L.; Chien, C.-C.; Wan, H.-M. Enhanced di-rhamnolipid production with an indigenous isolate Pseudomonas aeruginosa J16. Process Biochem. 2008, 43, 769–774. [Google Scholar] [CrossRef]
- Batista, S.B.; Mounteer, A.H.; Amorim, F.R.; Totola, M.R. Isolation and characterization of biosurfactant/bio emulsifier-producing bacteriafrom petroleum contaminated sites. Bioresour. Technol. 2005, 97, 868–875. [Google Scholar] [CrossRef]
- Techaoei, S.; Leelapornpisid, P.; Santiarwarn, D.; Lumyong, S. Preliminary Screening of Biosurfactant-Producing Microorganism Isolated from Hot Spring and Garages in Northern Thailand. KMITL Sci. Tech. J. 2007, 7, 38–43. [Google Scholar]
- Bordas, F.O.; Lafrance, P.; Villemur, R. Conditions for effective removal of pyrene from an artificially contaminated soil using Pseudomonas aeruginosa 57SJ rhamnolipids. Environ. Pollut. 2005, 138, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Calvo, C.; Manzanera, M.; Silva-Castro, G.A.; Uad, I.; González-López, J. Application of bioemulsifiers in soil oil bioremediation processes. Future prospects. Sci. Total Environ. 2009, 407, 3634–3640. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, S.; Mukherji, S.; Mukherji, S. Surface hydrophobicity of petroleum hydrocarbon-degrading Burkholderia strains and their interactions with NAPLs and surfaces. Colloids Surf. B Biointerfaces 2010, 78, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Eddouaouda, K.; Mnif, S.; Badis, A.; BenYounes, S.; Cherif, S.; Ferhat, S.; Mhiri, N.; Chamkha, M.; Sayadi, S. Characterization of a novel biosurfactant produced by Staphylococcus sp. strain 1E with potential application on hydrocarbon bioremediation. J. Basic Microbiol. 2012, 52, 408–418. [Google Scholar] [CrossRef]
- Vollenbroich, D.; Pauli, G.; Ozel, M. Antimycoplasma properties and applications in cell culture of surfactin, a lipopeptide antibiotic from Bacillus subtilis. Appl. Environ. Microbiol. 1997, 63, 44–49. [Google Scholar] [CrossRef]
- Manickam, N.; Bajaj, A.; Saini, H.S.; Shanker, R. Surfactant mediated enhanced biodegradation of hexachlorocyclohexane (HCH) isomers by Sphingomonas sp. NM05. Biodegradation 2012, 23, 673–682. [Google Scholar] [CrossRef]
- Chaprao, C.M.J.; Ferreira, I.N.; Correa, P.F.; Rufino, R.D.; Luna, J.M.; Silva, E.J.; Sarubbo, L.A. Application of bacterial and yeast biosurfactants for enhanced removal and biodegradation of motor oil from contaminated sand. Electron. J. Biotechnol. 2015, 18, 471–479. [Google Scholar] [CrossRef]
- Araújo, H.W.; Andrade, R.F.; Montero-Rodríguez, D.; Rubio-Ribeaux, D.; Alves da Silva, C.A.; Campos-Takaki, G.M. Sustainable biosurfactant produced by Serratia marcescens UCP 1549 and its suitability for agricultural and marine bioremediation applications. Microb. Cell Fact. 2019, 18, 2. [Google Scholar] [CrossRef]
- Nitschke, M.; Ferraz, C.; Pastore, G.M. Selection of microorganisms for biosurfactant production using agro-industrial wastes. Braz. J. Microbiol. 2004, 35, 81–85. [Google Scholar] [CrossRef]
- Bodour, A.A.; Miller-Maier, R.M. Application of a Modified drop collapsingtechnique for Surfactant Quantitation and Screening of Biosurfactant-producing Microorganisms. J. Microbiol. 1998, 32, 273–280. [Google Scholar]
- Prieto, L.M.; Michelon, M.; Burkert, J.F.; Kalil, S.J.; Burkert, C.A. The production of rhamnolipid by a Pseudomonas aeruginosa strain isolated from a southern coastal zone in Brazil. Chemosphere 2008, 71, 1781–1785. [Google Scholar] [CrossRef] [PubMed]
- Câmara, J.M.D.A.; Sousa, M.A.S.B.; Barros Neto, E.L. Application of rhamnolipid biosurfactant produced by Pseudomonas aeruginosa in microbial-enhanced oil recovery (MEOR). J. Petrol. Explor. Prod. Technol. 2019, 9, 2333–2341. [Google Scholar] [CrossRef]
- Bai, G.; Brusseau, M.L.; Miller, R.M. Biosurfactant enhanced removal of residual hydrocarbon from soil. J. Contam. Hydrol. 1996, 25, 157–170. [Google Scholar] [CrossRef]
- Wei, Y.H.; Chu, I.M. Enhancement of surfactin production in iron-enriched media by Bacillus subtilis ATCC 21332. Enzym. Microb. Technol. 1998, 22, 724–728. [Google Scholar] [CrossRef]
- Guan, R.; Yuan, X.; Wu, Z.; Wang, H.; Jiang, L.; Li, Y.; Zeng, G. Functionality of surfactants in waste-activated sludge treatment: A review. Sci. Total Environ. 2017, 609, 1433–1442. [Google Scholar] [CrossRef]
- Camargo, F.P.; doPrado, P.F.; Tonello, P.S.; Santos, A.C.A.D.; Duarte, I.C.S. Bioleaching of toxic metals from sewage sludge by co-inoculation of Acidithiobacillus and the biosurfactant-producing yeast Meyerozyma guilliermondii. J. Environ. Manag. 2018, 211, 28–35. [Google Scholar] [CrossRef]
- Hsueh, Y.-H.; Somers, E.B.; Lereclus, D.; Ghelardi, E.; Lee, W.; Amy, C. Biosurfactant Production and Surface Translocation Are Regulated by PlcR in Bacillus cereus ATCC 14579 under Low-Nutrient Conditions. Appl. Environ. Microbiol. 2007, 73, 7225–7231. [Google Scholar] [CrossRef]
- Huang, W.; Lang, Y.; Hakeem, A.; Lei, Y.; Gan, L.; Yang, X. Surfactin-based nanoparticles loaded with doxorubicin to overcome multidrug resistance in cancers. Int. J. Nanomed. 2018, 13, 1723–1736. [Google Scholar] [CrossRef]
- Ohadi, M.; Shahravan, A.; Dehghannoudeh, N.; Eslaminejad, T.; Banat, I.M.; Dehghannoudeh, G. Potential Use of Microbial Surfactant in Microemulsion Drug Delivery System: A Systematic Review. Drug Des. Devel. Ther. 2020, 14, 541–550. [Google Scholar] [CrossRef]
- Rienzo, M.A.D.D.; Banat, I.M.; Dolman, B.; Winterburn, J.; Martin, P.J. Sophorolipid biosurfactants: Possible uses as antibacterial and antibiofilm agent. New Biotechnol. 2015, 32, 720–726. [Google Scholar] [CrossRef] [PubMed]
- Eslaminejad, T.; Nematollahi-Mahani, S.N.; Ansari, M. Cationic β-cyclodextrin–Chitosan conjugates as potential carrier for pmcherry-c1 gene delivery. Mol. Biotechnol. 2016, 58, 287–298. [Google Scholar] [CrossRef] [PubMed]
- Eslaminejad, T.; Nematollahi-Mahani, S.N.; Ansari, M. Synthesis, characterization, and cytotoxicity of the plasmid EGFP-p53 loaded on pullulan–spermine magnetic nanoparticles. J. Magn. Magn. Mater. 2016, 402, 34–43. [Google Scholar] [CrossRef]
- Sharma, P.; Sharma, N. Microbial Biosurfactants-an Ecofriendly Boon to Industries for Green Revolution. Recent Pat. Biotechnol. 2020, 14, 169–183. [Google Scholar] [CrossRef] [PubMed]
- Araujo, S.C.d.S.; Silva-Portela, R.C.B.; de Lima, D.C. MBSP1: A biosurfactant protein derived from a metagenomic library with activity in oil degradation. Sci. Rep. 2020, 10, 1340. [Google Scholar] [CrossRef] [PubMed]
- Delbeke, E.I.P.; Van Geem, K.M.; Stevens, C.V.; Van Bogaert, I.N.A. Sophorolipid Modification: The Power of Yeasts and Enzymes. Lipid Modif. Enzym. Eng. Microbes 2018, 315–341. [Google Scholar] [CrossRef]
- Jahan, R.; Bodratti, A.M.; Tsianou, M.; Alexandridis, P. Biosurfactants, natural alternatives to synthetic surfactants: Physicochemical properties and applications. Adv. Colloid Interface Sci. 2020, 275, 102061. [Google Scholar] [CrossRef]
- Li, H.; Hu, D.; Liang, F.; Huang, X.; Zhu, Q. Influence factors on the critical micelle concentration determination using pyreneas a probe and a simple method of preparingsamples. R. Soc. Open Sci. 2020, 7, 192092. [Google Scholar] [CrossRef]
- Wong, F.W.F.; Ariff, A.B.; Stuckey, D.C. Downstream protein separation by surfactant precipitation: A review. Crit. Rev. Biotechnol. 2018, 38, 31–46. [Google Scholar] [CrossRef]
- Garcia-Junco, M.; Gomez-Lahoz, C.; Niqui-Arroyo, J.L.; Ortega-Calvo, J.J. Biodegradation and biosurfactant-enhanced partitioning of polycyclic aromatic hydrocarbons from nonaqueous-phase liquids. Environ. Sci. Technol. 2003, 37, 2988–2996. [Google Scholar] [CrossRef]
- Su, H.; Wang, F.; Ran, W.; Zhang, W.; Dai, W.; Wang, H.; Anderson, C.F.; Wang, Z.; Zheng, C.; Zhang, P.; et al. The role of critical micellization concentration in efficacy and toxicity of supramolecular polymers. Proc. Natl. Acad. Sci. USA 2020, 117, 4518–4526. [Google Scholar] [CrossRef] [PubMed]
- Esmaeili, H.; Mousavi, S.M.; Hashemi, S.A.; Lai, C.W.; Chiang, W.; Bahrani, S. Chapter 7—Application of biosurfactants in the removal of oil from emulsion. In Green Sustainable Process for Chemical and Environmental Engineering and Science; Elsevier: Amsterdam, The Netherlands, 2021; pp. 107–127. [Google Scholar]
- Guo, J.; Wen, X. Performance and kinetics of benzo(a)pyrene biodegradation in contaminated water and soil and improvement of soil properties by biosurfactant amendment. Ecotoxicol. Environ. Saf. 2021, 207, 111292. [Google Scholar] [CrossRef] [PubMed]
- Santos, M.S. Molecular Thermodynamics of Micellization: Micelle Size Distributions and Geometry Transitions. Braz. J. Chem. Eng. 2016, 33, 515–523. [Google Scholar] [CrossRef]
- Santos, D.K.F.; Rufino, R.D.; Luna, J.M.; Santos, V.A.; Sarubbo, L.A. Biosurfactants: Multifunctional Biomolecules of the 21st Century. Int. J. Mol. Sci. 2016, 17, 401. [Google Scholar] [CrossRef] [PubMed]
- Adetunji, A.I.; Olaniran, A.O. Production and potential biotechnological applications of microbial surfactants: An overview. Saudi J. Biol. Sci. 2021, 28, 669–679. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Pandey, L.M. Hydrophobic Surface Induced Biosorption and Microbial Ex Situ Remediation of Oil-Contaminated Sites. Ind. Eng. Chem. Res. 2021, 60, 9378–9388. [Google Scholar] [CrossRef]
- De Giani, A.; Zampolli, J.; Di Gennaro, P. Recent Trends on Biosurfactants With Antimicrobial Activity Produced by Bacteria Associated With Human Health: Different Perspectives on Their Properties, Challenges, and Potential Applications. Front. Microbiol. 2021, 12, 678. [Google Scholar] [CrossRef]
- Shi, J.G.; Zeng, G.M.; Yuan, X.Z. The stimulatory effects of surfactants on composting of waste rich in cellulose. World J. MicrobBiot. 2006, 22, 1121–1127. [Google Scholar] [CrossRef]
- Jahanshah, G.; Nahvi, I.; Zarkesh-Esfahani, S.H. Enhancing compost quality by using whey-grown biosurfactant-producing bacteria as inocula. Ann. Microbiol. 2013, 63, 91–100. [Google Scholar] [CrossRef]
- Zhang, Q.; Weimin, C.A.I.; Juan, W. Stimulatory effects of biosurfactant produced by Pseudomonas aeruginosa BSZ-07 on rice straw decomposing. J. Environ. Sci. 2008, 20, 975–980. [Google Scholar] [CrossRef]
- Molina, M.J.; Soriano, M.D.; Ingelmo, F.; Llinares, J. Stabilisation of sewage sludge and vinasse bio-wastes by vermicomposting with rabbit manure using Eisenia fetida. Bioresour. Technol. 2013, 137, 88–97. [Google Scholar] [CrossRef]
- Subramanian, S.; Sivarajan, M.; Saravanapriya, S. Chemical changes during vermicomposting of sago industry solid wastes. J. Hazard. Mater. 2010, 179, 318–322. [Google Scholar] [CrossRef] [PubMed]
- Ni’matuzahroh; Sari, S.K.; Ningrum, I.P.; Pusfita, A.D.; Marjayandari, L.; Trikurniadewi, N.; Fatimah, S.N.M.M.I.; Nurhariyati, T.; Surtiningsih, T.; Yuliani, H. The potential of indigenous bacteria from oil sludge for biosurfactant production using hydrolysate of agricultural waste. Biodiversitas 2019, 20, 1374–1379. [Google Scholar] [CrossRef][Green Version]
- Dadrasnia, A.; Ismail, S. Biosurfactant Production by Bacillus salmalaya for Lubricating Oil Solubilization and Biodegradation. Int. J. Environ. Res. Public Health 2015, 12, 9848–9863. [Google Scholar] [CrossRef] [PubMed]
- Fenibo, E.O.; Ijoma, G.N.; Selvarajan, R.; Chikere, C.B. Microbial Surfactants: The Next Generation Multifunctional Biomolecules for Applications in the Petroleum Industry and Its Associated Environmental Remediation. Microorganisms 2019, 7, 581. [Google Scholar] [CrossRef] [PubMed]
- Mullan, C.N.; Gibbs, B.F. Correlation of Nitrogen Metabolism with Biosurfactant Productionby Pseudomonas aeruginosa. Appl. Environ. Microbiol. 1989, 55, 3016–3019. [Google Scholar] [CrossRef]
- Malla, M.A.; Dubey, A.; Kumar, A.; Yadav, S.; Hashem, A.; Abd-Allah, E.F. Exploring the human microbiome: The potential future role of next-generation sequencing in disease diagnosis and treatment. Front. Immunol. 2019, 9, 2868. [Google Scholar] [CrossRef]
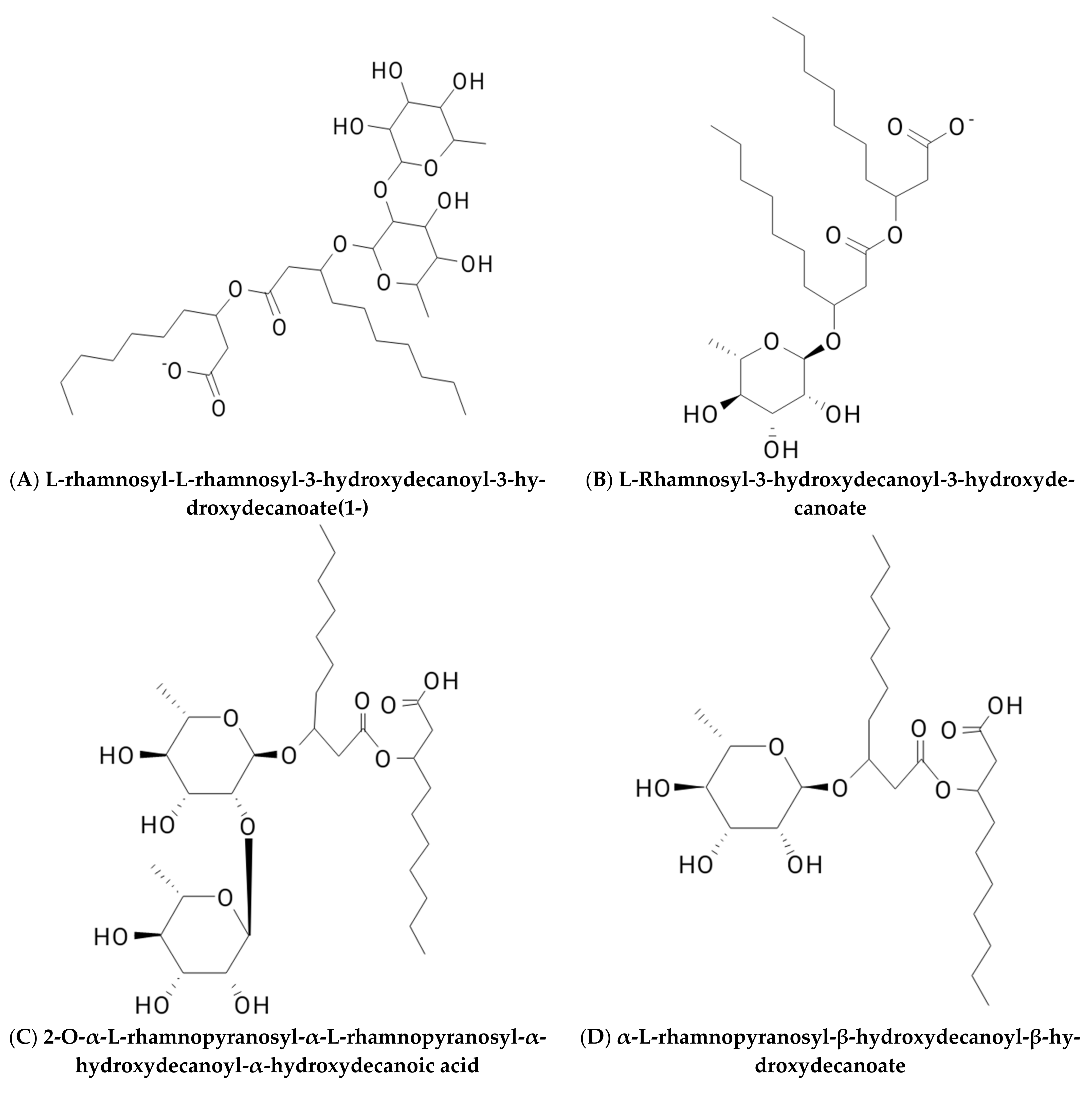
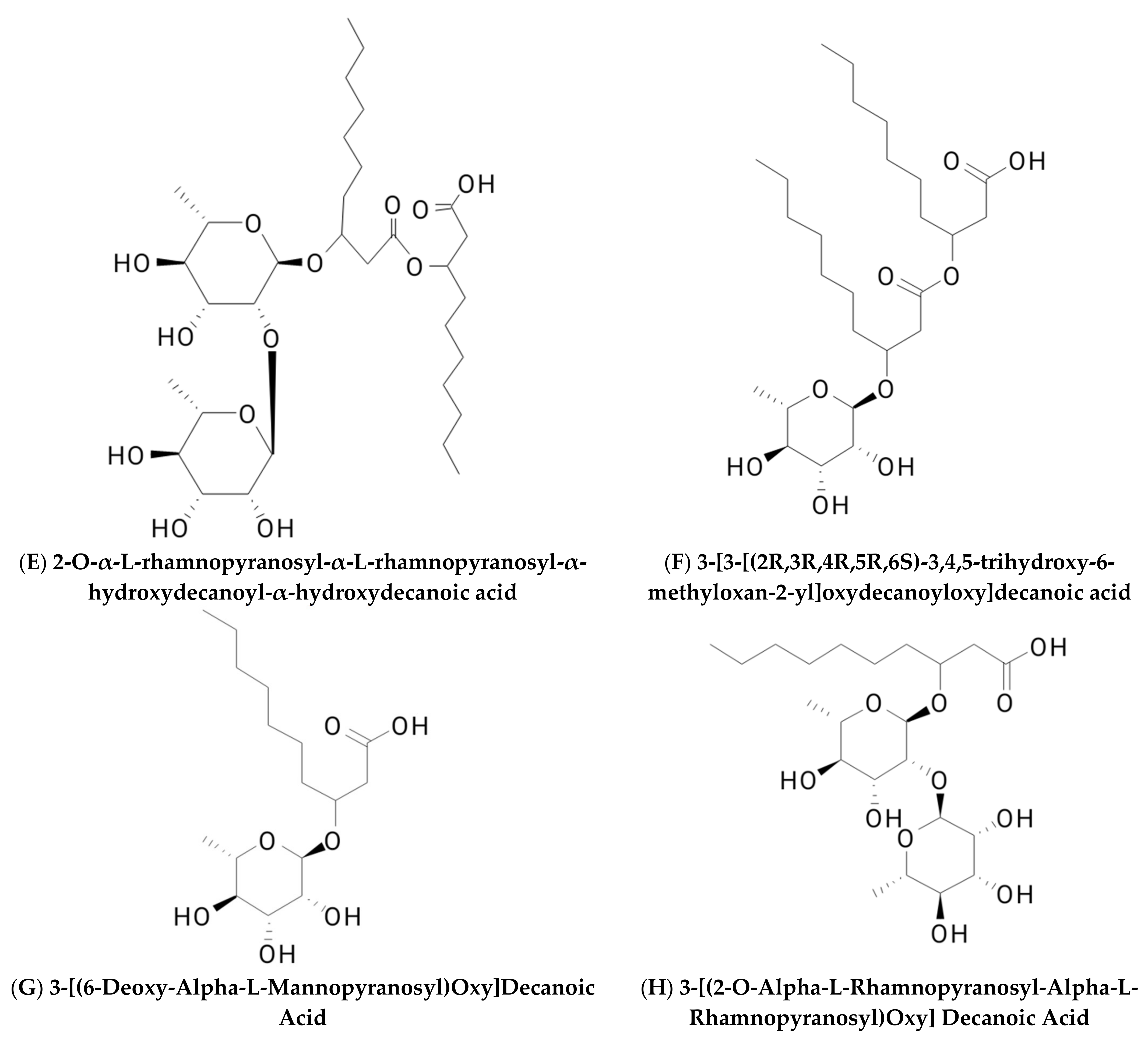
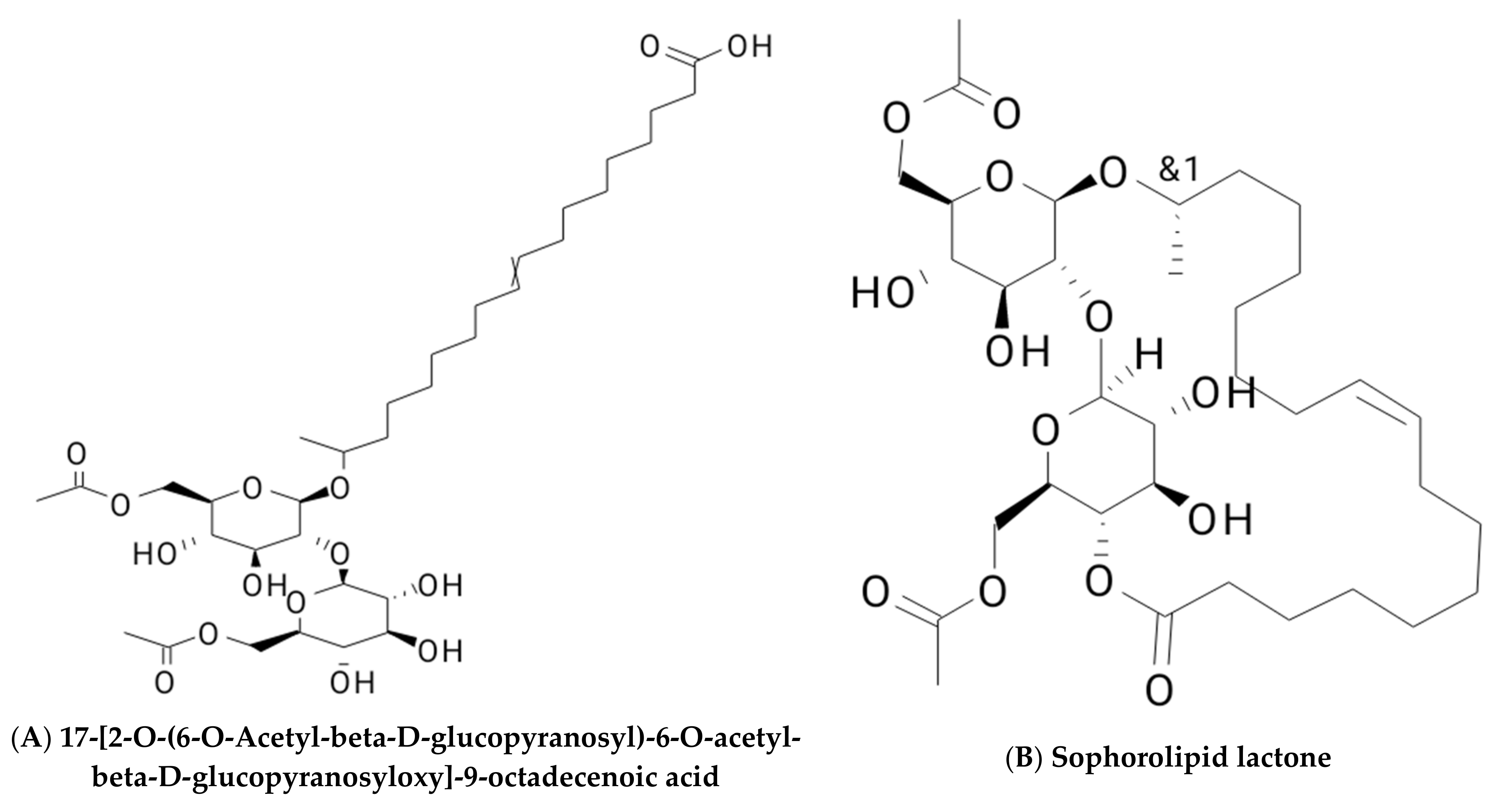
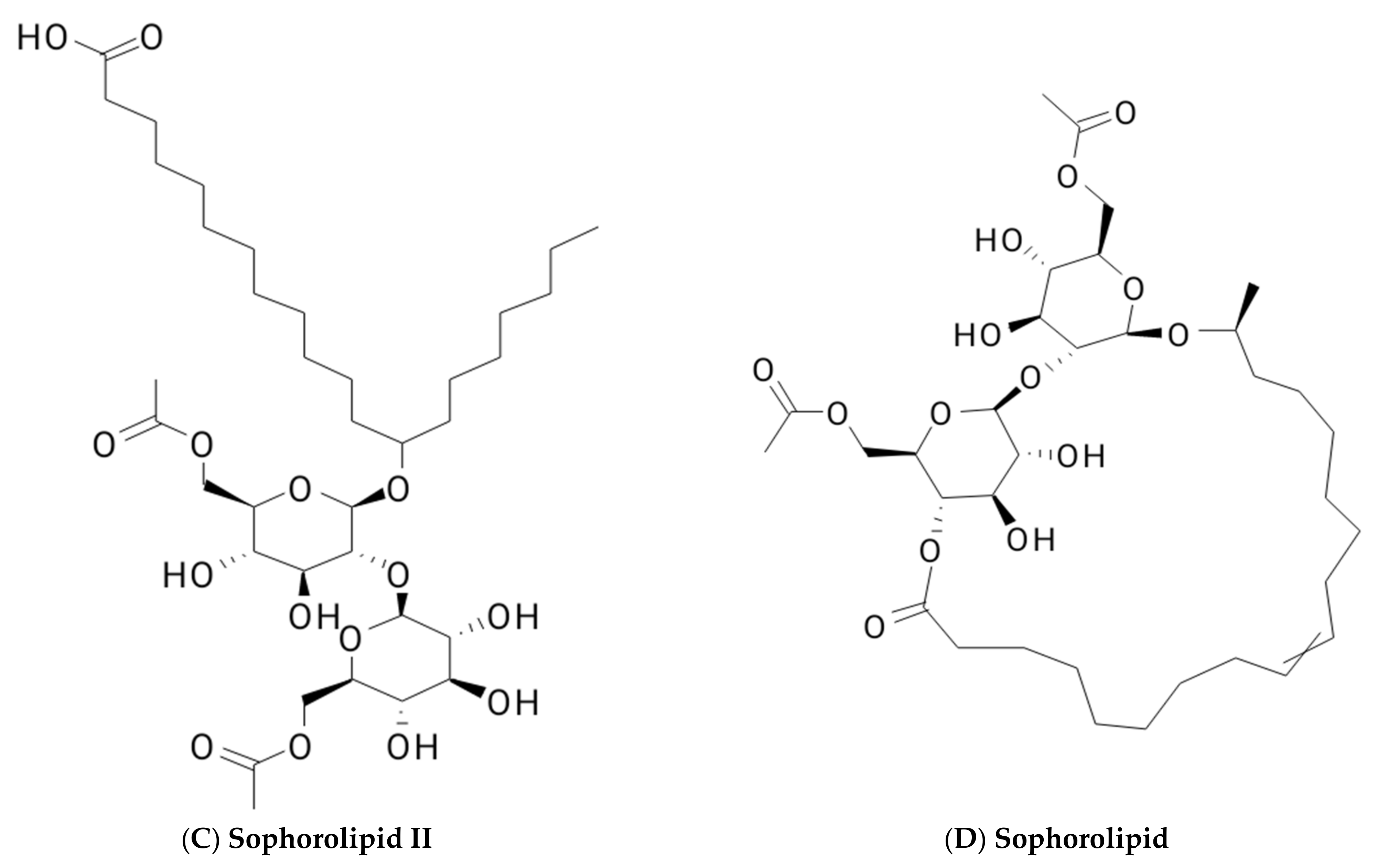
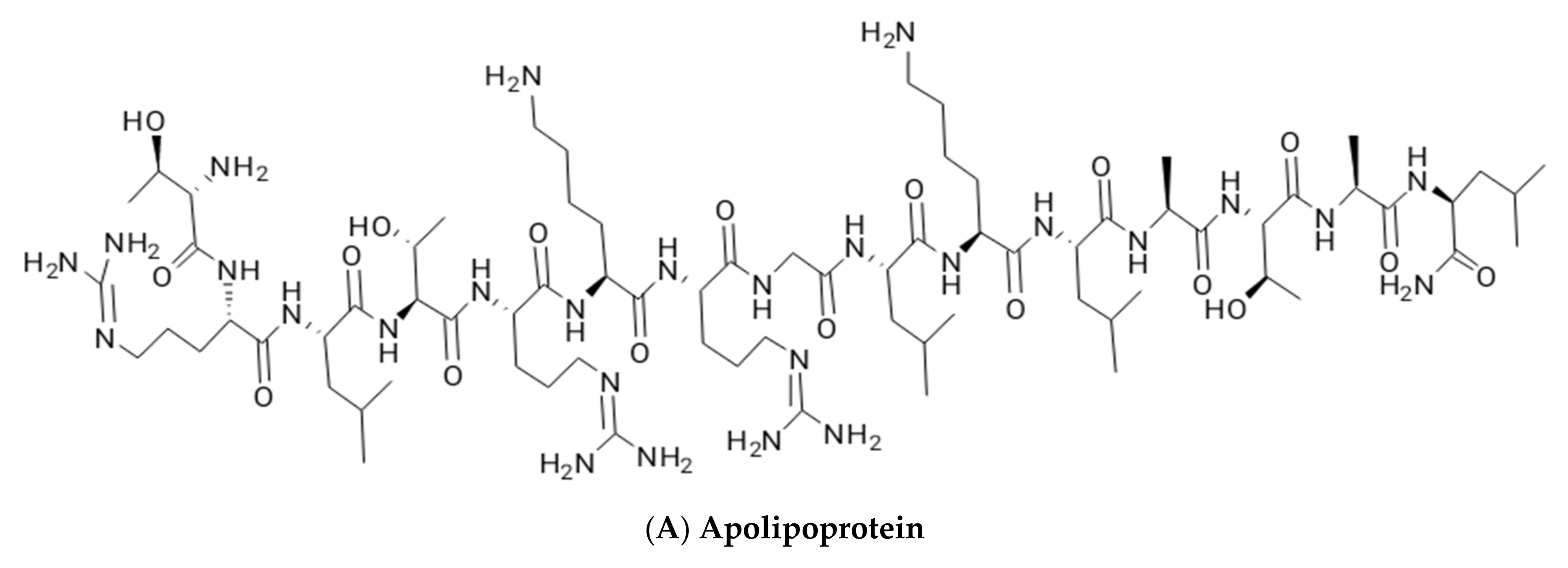
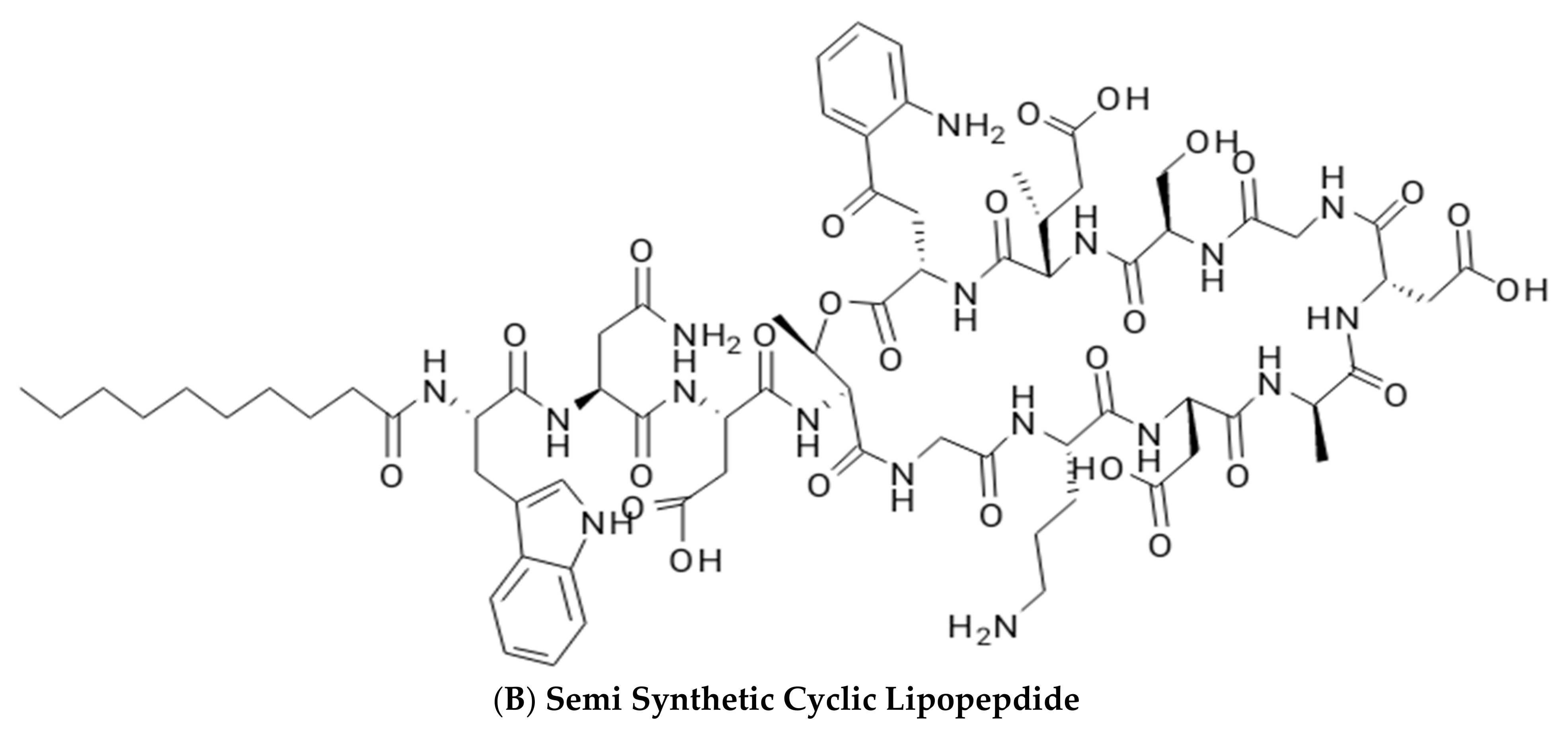

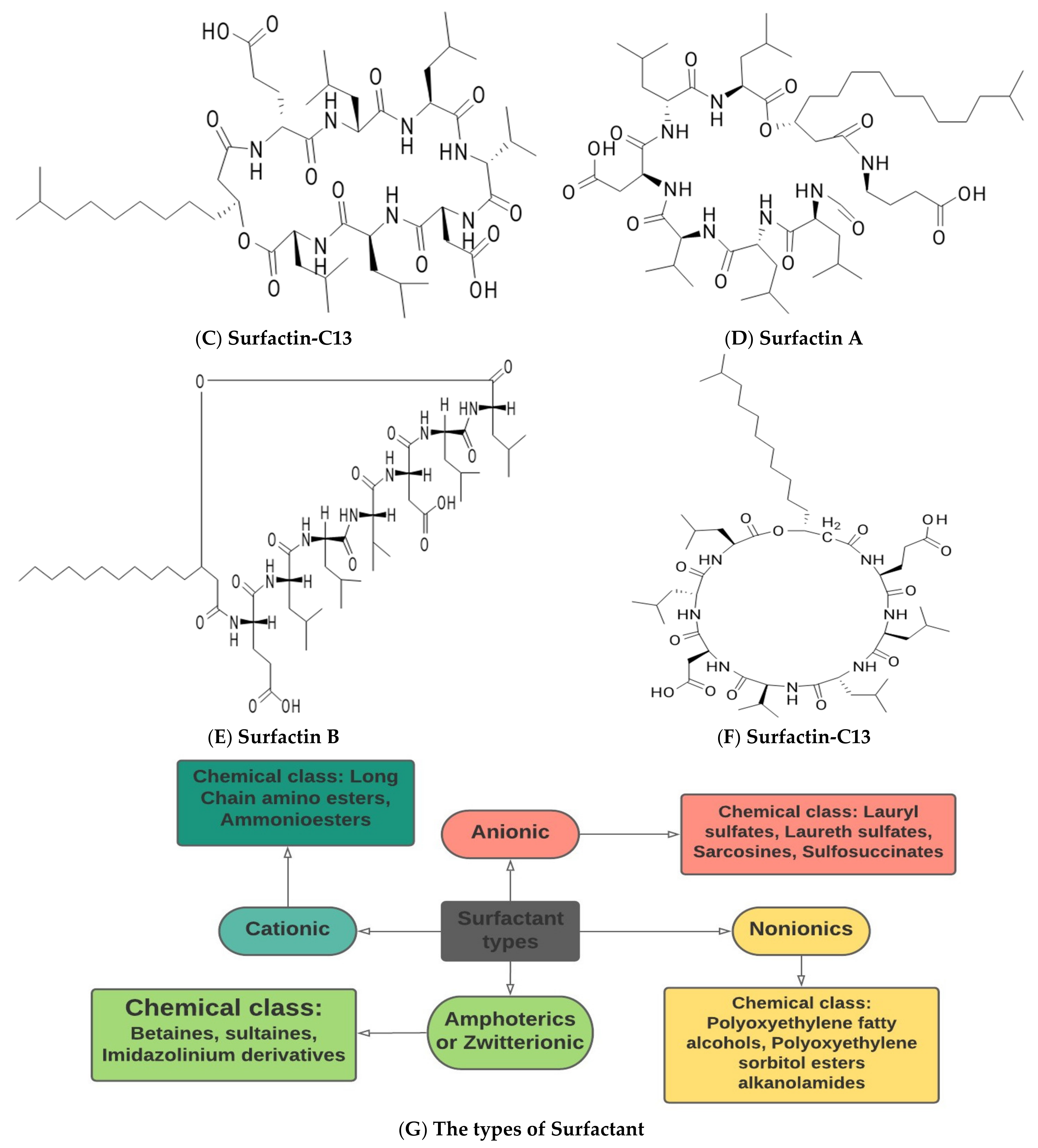
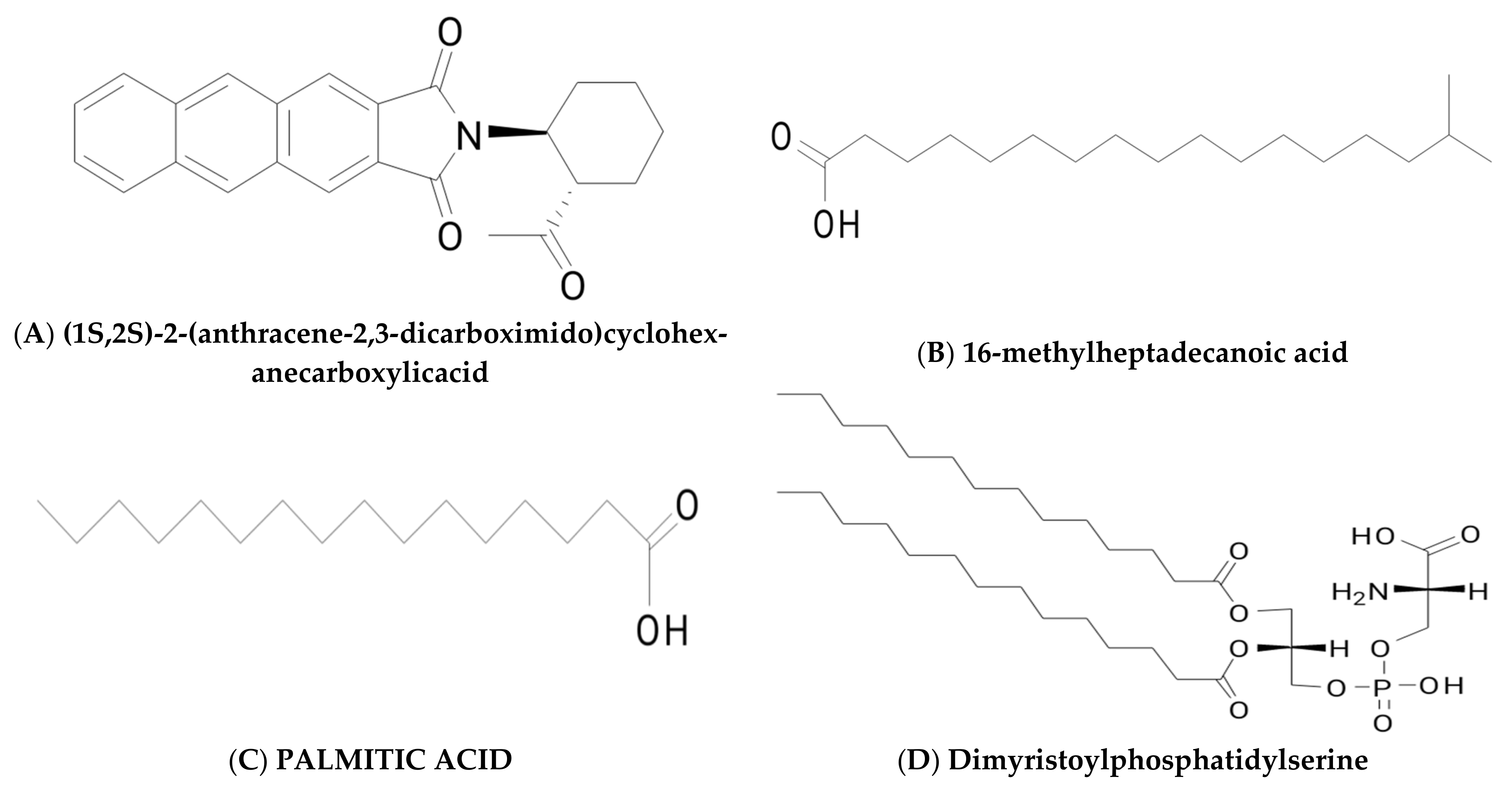

| Glycolipids | Lipo–Peptides | Surface-Active Antibiotics | Fatty Acids/Neutral Lipids | Polymeric Surfactants | Particulate Biosurfactants |
|---|---|---|---|---|---|
|
|
|
|
|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gayathiri, E.; Prakash, P.; Karmegam, N.; Varjani, S.; Awasthi, M.K.; Ravindran, B. Biosurfactants: Potential and Eco-Friendly Material for Sustainable Agriculture and Environmental Safety—A Review. Agronomy 2022, 12, 662. https://doi.org/10.3390/agronomy12030662
Gayathiri E, Prakash P, Karmegam N, Varjani S, Awasthi MK, Ravindran B. Biosurfactants: Potential and Eco-Friendly Material for Sustainable Agriculture and Environmental Safety—A Review. Agronomy. 2022; 12(3):662. https://doi.org/10.3390/agronomy12030662
Chicago/Turabian StyleGayathiri, Ekambaram, Palanisamy Prakash, Natchimuthu Karmegam, Sunita Varjani, Mukesh Kumar Awasthi, and Balasubramani Ravindran. 2022. "Biosurfactants: Potential and Eco-Friendly Material for Sustainable Agriculture and Environmental Safety—A Review" Agronomy 12, no. 3: 662. https://doi.org/10.3390/agronomy12030662
APA StyleGayathiri, E., Prakash, P., Karmegam, N., Varjani, S., Awasthi, M. K., & Ravindran, B. (2022). Biosurfactants: Potential and Eco-Friendly Material for Sustainable Agriculture and Environmental Safety—A Review. Agronomy, 12(3), 662. https://doi.org/10.3390/agronomy12030662








