Effects of Closing Cut Date and Nitrogen Fertilization on Seed Yield and Seed Quality in Two Novel Cultivars of Urochloa spp.
Abstract
1. Introduction
2. Materials and Methods
2.1. Location of the Experiment
2.2. Meteorological Data
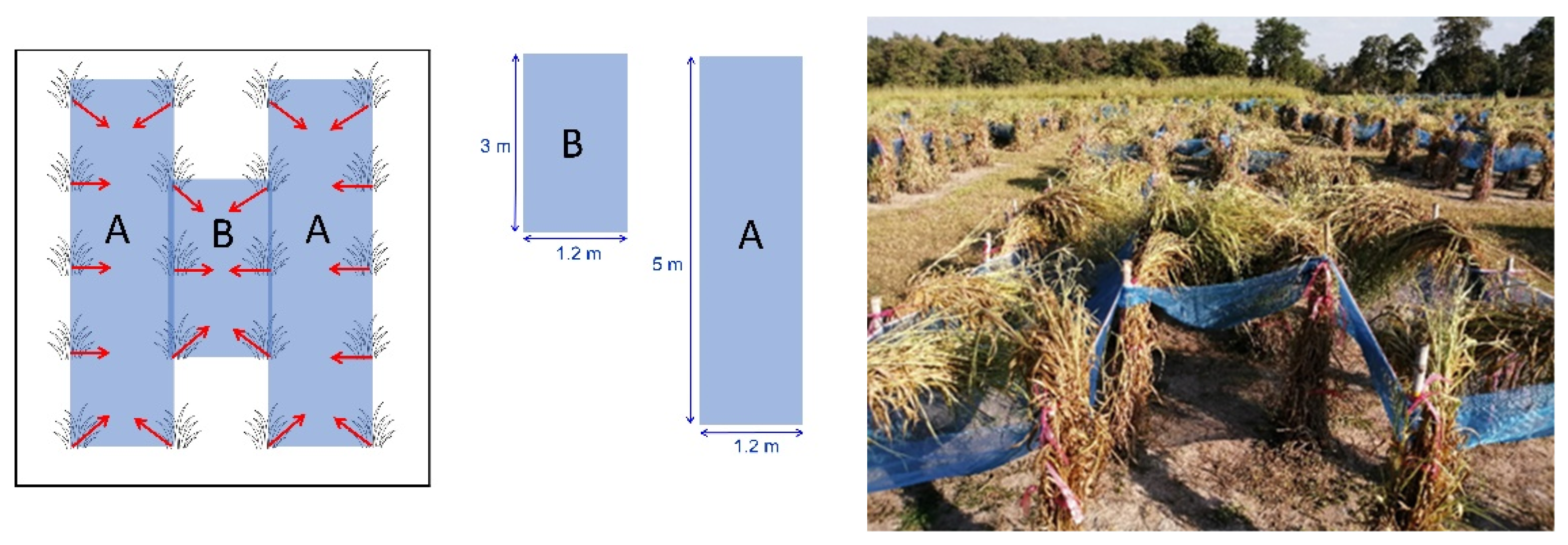
2.3. Experimental Design and Treatments
2.4. Data Collection and Seed Harvesting
2.5. Seed Processing and Calculation of Secondary Attributes
2.6. Statistical Analysis
3. Results and Discussion
3.1. Soil and Weather Conditions
3.2. Effects of CCD and N-Rate on Seed Yield, Seed Yield Components, and Seed Quality in the Two Novel Cultivars of Urochloa spp.
3.2.1. Trial 1: Effect of CCD and N-Rate on Seed Yield, Seed Yield Components, and Seed Quality in Cultivar ‘OKI-1’
3.2.2. Trial 2: Effect of CCD and N-Rate on Seed Yield, Seed Yield Components and Seed Quality in Cultivar ‘Br-203’
4. Conclusions
- No significant mutual interactions were observed between the CCD and N-rate on seed yield, seed yield components, or seed quality in both cultivars.
- The highest seed yields in cv. ‘OKI-1’ and cv. ‘Br-203’ were obtained when the CCD was 1 July and 15 July, respectively.
- The CCD affected most seed quality attributes, except seed germination percentage (GP) in cv. ‘OKI-1’.
- The CCD in late-June to early-July and early-July to mid-July are recommended for cv. ‘OKI-1’ and cv. ‘Br-203’, respectively.
- The seed yields in cv. ‘OKI-1’ and cv. ‘Br-203’ were non-significant in response to the different N-rates in the fertilization timing in our experiment.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fisher, M.J.; Kerridge, P.C. The agronomy and physiology of Brachiaria species. In Brachiaria: Biology, Agronomy, and Improvement; Miles, J.W., Maass, B.L., do Valle, C.B., Eds.; CIAT: Cali, Colombia, 1996; pp. 43–52. [Google Scholar]
- Rao, I.; Peters, M.; Castro, A.; Schultze-Kraft, R.; White, D.; Fisher, M.; Miles, J.; Lascano, C.; Blümmel, M.; Bungenstab, D.; et al. LivestockPlus—The sustainable intensification of forage-based agricultural systems to improve livelihoods and ecosystem ser-vices in the tropics. Trop. Grassl.-Forrajes Trop. 2015, 3, 59–82. [Google Scholar] [CrossRef]
- Cheruiyot, D.; Midega, C.A.O.; Pittchar, J.O.; Pickett, J.A.; Khan, Z.R. Farmers’ perception and evaluation of Brachiaria grass (Brachiaria spp.) genotypes for smallholder cereal-livestock production in East Africa. Agriculture 2020, 10, 268. [Google Scholar] [CrossRef]
- Kouki, K.; Ishigaki, G.; Akashi, R.; Shimabukuro, H. The newly-bred cultivar production in Brachiaria grass. In (1) Production of the High Seed Yield Strains Using Sexual Tetraploid Ruzigrass ‘Miyaokikoku-Ichigou’; Annual Report; Okinawa Prefectural Livestock and Grassland Research Center: Okinawa, Japan, 2014; Volume 52, pp. 73–75. [Google Scholar]
- Ishigaki, G.; Gondo, T.; Suenaga, K.; Akashi, R. Induction of tetraploid ruzigrass (Brachiaria ruziziensis) plants by colchicine treatment of in vitro multiple-shoot clumps and seedlings. Grassl. Sci. 2009, 55, 164–170. [Google Scholar] [CrossRef]
- Thaikua, S.; Chanpeng, P.; Juntasin, W.; Donsawai, S.; Chuchuay, P.; Kouki, K.; Kawamoto, Y. Effect of ploidy on digestibility and fiber fraction of ruzigrass (Brachiaria ruziziensis). In Proceedings of the 18th Asian–Australasian Animal Production (AAAP) Congress, Sarawak, Malaysia, 1–5 August 2018; p. 434. [Google Scholar]
- Nakmanee, G.; Ebina, M.; Shimoda, K.; Thaikua, S.; Srisomporn, W.; Patipan, C.; Suenaga, K.; Ando, S.; Kouki, K.; Ishigaki, G.; et al. A new candidate cultivar of Brachiaria grass ‘Br-203’ developed with apomixis marker assisted selection, through a collaborative breeding activity of Thailand and Japan. In Proceedings of the 23rd International Grassland Congress, New Dehli, India, 20–24 November 2015; Roy, M.M., Malaviya, D.R., Yadav, V.K., Singh, T., Sah, R.P., Vijay, D., Radhakrishna, A., Eds.; Range Management Society of India: New Dehli, India, 2015. [Google Scholar]
- Loch, D.S.; Aviles, R.; Harvey, G.L. Crop management: Grasses. In Forage Seed Production Vol. 2: Tropical and Subtropical Species; Loch, D.S., Ferguson, J.E., Eds.; CABI Publishing: Wallingford, UK, 1999; pp. 159–176. [Google Scholar]
- Hare, M.D.; Tatsapong, S.; Saipraset, K. Seed production of two brachiaria hybrid cultivars in north-east Thailand. 2. Closing date defoliation. Trop. Grassl. 2007, 41, 35–42. [Google Scholar]
- Cropping Strategies, a Summary of Recent Research Findings: Closing Date in Perennial Ryegrass, Closing Date in Italian Ryegrass. Foundation of Arable Research PO Box 23133, Templeton, Christchurch 8445, New Zealand. Available online: https://www.far.org.nz/assets/files/blog/files/2c6f1d23-3c63-4bf7-8ed2-bb7b5543083f.pdf (accessed on 27 May 2021).
- Hare, M.D.; Wongpichet, K.; Tatsapong, S.; Narksombat, S.; Saengkhum, M. Method of seed harvest, closing date and height of closing cut affect seed yield and seed yield components in Paspalum atratum in Thailand. Trop. Grassl. 1999, 33, 82–90. [Google Scholar]
- Gobius, N.R.; Phaikaew, C.; Pholsen, P.; Rodchompoo, O.; Susena, W. Effects of date of closing cut on seed yield and its components of Andropogon gayanus cv. Kent. Trop. Grassl. 1998, 32, 230–234. [Google Scholar]
- Phaikaew, C.; Pholsen, P. Ruzigrass (Brachiaria ruziziensis) seed production and research in Thailand. In Proceedings of the Third Meeting of Regional Working Group on Grazing and Feed Resources of Southeast Asia, Khon Kaen, Thailand, 31 January–6 February 1993; pp. 165–173. [Google Scholar]
- Nakmanee, G.; Phaikaew, C.; Thinnakorn, S. Effect of closing cut date and nitrogen rates on seed yield and seed quality of Mulato grass (Brachiaria ruziziensis × Brachiaria brizantha). In Annual Report; Bureau of Animal Nutrition Development, Department of Livestock Development: Bangkok, Thailand, 2007; pp. 199–210. [Google Scholar]
- Phaikaew, C.; Intaramanee, S.; Tewahudee, P.; Boonpakdee, W.; Suriyajantrathong, W.; Senakulp, U.; Nakmanee, G.; Pholboon, P. Effect of cutting periods on seed yields of ruzi grass (Brachiaria ruziziensis). In Annual Report; Bureau of Animal Nutrition Development, Department of Livestock Development: Bangkok, Thailand, 1985; pp. 106–116. [Google Scholar]
- Bouathong, C.; Hare, M.; Losirikul, M.; Wongpichet, K. Effect of nitrogen-rates on plant growth, seed yield and seed quality of three lines of Brachiaria hybrid grass. Khon Kaen Agr. J. 2011, 39, 295–306. [Google Scholar]
- Satjipanon, C.; Nakmanee, K.; Maneedool, C.; Boonrat, J. Effect of defoliation management and rate of nitrogen on seed yield and seed quality of Ruzi grass (Brachiaria ruziziensis). In Annual Report; Bureau of Animal Nutrition Development, Department of Livestock Development: Bangkok, Thailand, 1987; pp. 195–215. [Google Scholar]
- Wongsuwan, N. Effects of Cutting, Nitrogen, Closing Date and Water on Herbage and Seed Production in Ruzi Grass (Brachiaria ruziziensis Germain and Everard). Ph.D. Thesis, Massey University, Palmerston North, New Zealand, 1999. [Google Scholar]
- Phaikaew, C.; Intarit, S.; Tudsri, S.; Tsuzuki, E.; Numaguchi, H.; Ishii, Y. Effects of time of final closing cut on seed yield and seed quality of Paspalum atratum in Thailand. Trop. Grassl. 2002, 36, 150–158. [Google Scholar]
- De Lima, B.G.; Verzignassi, J.R.; Fernandes, C.D.; Do Valle, C.B.; Macedo, M.C.M.; De Liborio, C.B.; Monterio, L.C. Productivity and quality of Brachiaria brizantha B4 seeds in function of nitrogen doses. Ciênc. Rural. 2016, 46, 1566–1571. [Google Scholar]
- Canto, M.W.; Pancera, E.J., Jr.; Barth Neto, A.; Bremm, C.; Vier, P.U.; Costa, A.C.S. Effects of nitrogen fertilization and irrigation on seed yield and yield components of signal grass (Urochloa decumbens). Crop Pasture Sci. 2020, 71, 294–303. [Google Scholar]
- Adjolohoun, S.; Bindelle, J.; Adandedjan, C.; Toléba, S.S.; Houinato, M.; Kindomihou, V.; Nonfon, W.R.V.; Sinsin, B. Influence de l’écartement et de la fertilisation azotée sur le rendement et la qualité des semences de Brachiaria ruziziensis en climat tropical sub-humide. Fourrages 2013, 216, 339–345. [Google Scholar]
- Group of Soil Series. Available online: http://oss101.ldd.go.th/osr_data&service/OSR_PDF/TB_SSK_Distribute/P_SSK529.pdf (accessed on 14 November 2021).
- ISTA (International Seed Testing Association). International Rules for Seed Testing Edition 2011; The International Seed Testing Association: Bassersdorf, Switzerland, 2011. [Google Scholar]
- The R Project for Statistical Computing. Available online: https://www.R-project.org/ (accessed on 14 November 2021).
- Handbook of Soil Chemical Analysis. Available online: http://www.ldd.go.th/PMQA/2553/Manual/OSD-03.pdf (accessed on 14 November 2021).
- Rao, I.M.; Kerridge, P.C.; Macedo, M. Nutritional requirements of Brachiaria and adaptation to acid soils. In Brachiaria: Biology, Agronomy, and Improvement; Miles, J.W., Maass, B.L., do Valle, C.B., Eds.; Centro International de Agricultura Tropical (CIAT): Cali, Colombia, 1996; pp. 53–71. [Google Scholar]
- Boonman, J.G. Determinants of seed yield. In On the Seed Production of Tropical Grasses in Kenya; Leniger, H.A., Ed.; Centre for Agricultural Publishing and Documentation: Wageningen, The Netherlands, 1973; pp. 2–9. [Google Scholar]
- Stür, W.W.; Hopkinson, J.M.; Chen, C.P. Regional experience with Brachiaria: Asia, the South Pacific, and Australia. In Brachiaria: Biology, Agronomy, and Improvement; Miles, J.W., Maass, B.L., do Valle, C.B., Eds.; Centro International de Agricultura Tropical (CIAT): Cali, Colombia; CNPGC/EMBRAPA: Grande MS, Brazil, 1996; pp. 258–271. [Google Scholar]
- Brachiaria Hybrid. In: Pastures Australia. Available online: https://keys.lucidcentral.org/keys/v3/pastures/Html/Brachiaria_hybrid.htm (accessed on 14 November 2021).
- Nadew, B.B. Effects of climatic and agronomic factors on yield and quality of bread wheat (Triticum aestivum L.) seed: A review on selected factors. Adv. Crop Sci. Technol. 2018, 6, 356. [Google Scholar]
- Wongsuwan, N. Seed Production Studies in Ruzi Grass (Brachiaria ruziziensis Germain and Everard). Master’s Thesis, Massey University, Palmerston North, New Zealand, 1994. [Google Scholar]
- Deinum, B.; Dirven, J.G.P. Climate, nitrogen and grass. 5. Influence of age, light intensity and temperature on the production and chemical composition of Congo grass (Brachiaria ruziziensis Germain et Everard). Neth. J. Agri. Sci. 1972, 20, 125–132. [Google Scholar] [CrossRef]
- Galdos, M.V.; Brown, E.; Rosolem, C.A.; Pires, L.F.; Hallett, P.D.; Mooney, S.J. Brachiaria species influence nitrate transport in soil by modifying soil structure with their root sytem. Sci. Rep. 2020, 10, 5072. [Google Scholar] [CrossRef]
- Jones, C.A.; Koenig, R.T.; Ellsworth, J.W.; Brown, B.D.; Jackson, G.D. Management of Urea Fertilizer to Minimize Volatilization; The U.S. Department of Agriculture (USDA), Steele, D.L., Eds.; Extension Service; Montana State University: Bozeman, MT, USA, 2007; pp. 1–12.
- Sun, X.; Luo, N.; Longhurst, B.; Luo, J. Fertiliser nitrogen and factors affecting pasture responses. Open Agric. J. 2008, 2, 35–42. [Google Scholar] [CrossRef]
- Gbenou, B.; Adjolohoun, S.; Houndjo, D.B.M.; Ahoton, L.; Aliou Saidou, A.; Houinato, M.; Seibou Toleba, S.; Sinsin, B.A. Practical aspects of grass forage seed production and quality with particular reference to planting row spacing and nitrogen fertilization in tropical regions: A review. Int. J. Biol. Chem. Sci. 2018, 12, 508–518. [Google Scholar] [CrossRef]
- Brunse, H.; Watkin, B.R. Tropical pasture management for grass seed production. In Proceedings of the Lectures and Findings of Symposium and Workshop on Forage Grass Seed Production, Muaklek, Thailand, 28 October–8 November 1991; Brunse, H., Sukpituksakul, P., Eds.; Bangkok, Thailand, 1992; pp. 9–16. [Google Scholar]
- Catuchi, T.A.; Soratto, R.P.; Francisquini Júnior, A.; Guidorizzi, F.V.C.; Tiritan, C.S. Nitrogen management of forage grasses for nutrition, seed production, and nutrients in residual straw. Pesqui. Agropecu. Bras. 2019, 54, e00114. [Google Scholar] [CrossRef]
- Dida, M.F.; Lemore, A.A.; Seid, K.A. Effect of nitrogen level on herbage and seed yield of Rhodes grass (Chloris gayana). Int. J. Agron. 2021, 2021, 5540596. [Google Scholar]
- Canto, M.W.; Almeida, G.M.; Costa, A.C.S.; Neto, A.B.; Scaliante, J.R., Jr.; Orlandini, C.F. Seed production of ‘Mombasa’ grass subjected to different closing cut dates and nitrogen rates. Pesq. Agropec. Bras. 2016, 51, 766–775. [Google Scholar] [CrossRef][Green Version]
- Joaquín, T.M.B.; Gómez, V.A.; Ramos, J.J.A.; Aranda, I.E.M.; Pérez, A.J.A.; Joaquín, C.S. Yield and quality of guinea grass seed cv. Mombaza at different pre-cut dates. Rev. Mex. Cienc. Agric. 2020, 24, 191–200. [Google Scholar]
- Lopes, R.R.; Ost, H.J.; Souza, C.H.L.; Franke, L.B. Management of consecutive cuts in the production and quality of wintergreen paspalum seeds. Rev. Bras. Zootec. 2016, 45, 587–595. [Google Scholar] [CrossRef]
- Boonman, J.G. Experimental studies on seed production of tropical grasses in Kenya. 2. Tillering and heading in seed crops of eight grasses. Neth. J. Agri. Sci. 1971, 19, 237–249. [Google Scholar] [CrossRef]
- Humphreys, L.R.; Riveros, F. Tropical Pasture Seed Production; FAO Plant Production and Protection Paper; Publications Division, Food and Agriculture Organization of the United Nations: Rome, Italy, 1986; 216p. [Google Scholar]
- Hill, M.J.; Loch, D.S. Achieving potential herbage seed yields in tropical regions. In Proceedings of the XVII International Grassland Congress, Palmerston North, New Zealand, 8–21 February 1993; pp. 1629–1635. [Google Scholar]
- Martiniello, P.; Silva, J.A.T. Physiological and bioagronomical aspects involved in growth and yield components. of cultivated forage species in mediterranean environments: A review. Eur. J. Plant Sci. Biotechnol. 2011, 5, 64–68. [Google Scholar]
- Awad, A.; Hafiz, S.; Hammada, M.S.; El-Nouby, A.; El-Hendawy, S. Grain yield production of Sudan grass (Sorghum sudanense (Piper) Stapf) as influenced by cutting numbers, potassium rates, and intrarow spacing in a semiarid environment. Turk. J. Agric. For. 2013, 37, 657–664. [Google Scholar] [CrossRef]
- Mishra, M.L.; Chatterjee, B.N. Seed production in the forage grasses Pennisetum polystachyon and Andropogon gayanus in the India tropics. Trop. Grassl. 1968, 2, 51–56. [Google Scholar]
- Kumar, D.; Dwivedi, G.K.; Singh, S.N. Seed yield and quality of buffel grass (Cenchrus ciliaris) as influenced by row spacing and fertilizer level. Trop. Grassl. 2005, 39, 107–111. [Google Scholar]
- Langer, R.H.M. Growth of the grass plant in relation to seed production. In Herbage Seed Production: Proceedings of a Conference at Lincoln College, Canterbury, New Zealand, 13–15 November 1979; Lancashire, J.A., Ed.; Grassland Research and Practice Series No.1; New Zealand Grassland Association (Inc.): Palmerston North, New Zealand, 1980; pp. 6–11. Available online: https://www.grassland.org.nz/publications/nzgrassland_publication_539.pdf (accessed on 15 November 2021).
- Meijer, W.J.M.; Vreeke, S. Nitrogen fertilization of grass seed crops as related to soil mineral nutrition. Neth. J. Agri. Sci. 1988, 36, 375–385. [Google Scholar] [CrossRef]
- Hare, M.D.; Pizarro, E.A.; Phengphet, S.; Songsiri, T.; Sutin, N. Evaluation of new hybrid Brachiaria lines in Thailand. 2. Seed production. Trop. Grassl.-Forrajes Trop. 2015, 3, 94–103. [Google Scholar] [CrossRef]
- Miles, J.W.; Hare, M.D. Plant breeding and seed production of apomictic tropical forage grasses. In Seed Production in the Northern Light, Proceedings of the Sixth International Herbage Seed Conference, Grimstad, Norway, 18–20 June 2007; pp. 74–81.
- Risso-Pascotto, C.; Pagliarini, M.S.; Do Valle, C.B.; Jank, L. Asynchronous meiosis in an interspecific hybrid of Brachiaria ruziziensis and B. brizantha. Plant Cell Rep. 2004, 23, 304–310. [Google Scholar] [CrossRef]
- Risso-Pascotto, C.; Pagliarini, M.S.; Do Valle, C.B. Meiotic behavior in interspecific hybrids between Brachiaria ruziziensis and Brachiaria brizantha (Poaceae). Euphytica 2005, 145, 155–159. [Google Scholar] [CrossRef]



| Trial | pH | OM (%) | Total N (%) | Extractable (mg/kg) | EC (dS/m) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| P | K | Ca | Mg | Na | S | Mn | Fe | Cu | Zn | |||||
| 1 | 5.39 | 0.67 | 0.03 | 0.89 | 39.84 | 614.38 | 36.66 | 12.44 | 3.03 | 14.15 | 55.70 | 0.24 | 0.07 | 0.83 |
| 2 | 5.68 | 0.73 | 0.04 | 0.87 | 85.23 | 225.66 | 23.10 | 9.03 | 2.43 | 11.76 | 26.45 | 0.02 | 0.04 | 0.70 |
| N-Rate (kg N/ha) | pH | Organic Matter (%) | Total N (%) | Extractable (mg/kg) | |
|---|---|---|---|---|---|
| P | K | ||||
| 0 | 5.15 | 1.53 | 0.08 | 0.71 | 24.74 |
| 50 | 4.76 | 0.94 | 0.05 | 0.86 | 25.24 |
| 100 | 5.16 | 0.85 | 0.04 | 0.67 | 27.99 |
| 150 | 5.05 | 1.24 | 0.06 | 0.68 | 25.76 |
| N-Rate (kg N/ha) | pH | Organic Matter (%) | Total N (%) | Extractable (mg/kg) | |
|---|---|---|---|---|---|
| P | K | ||||
| 0 | 5.12 | 1.16 | 0.06 | 0.71 | 39.15 |
| 50 | 5.23 | 1.23 | 0.06 | 0.89 | 44.04 |
| 100 | 5.15 | 1.19 | 0.06 | 0.84 | 45.46 |
| 150 | 5.18 | 1.16 | 0.06 | 0.82 | 34.52 |
| Treatment | TSY (kg/ha) | TN (No./m2) | FTN (No./m2) | IN (No./m2) | FTP (%) | IN/T (No.) | RN/I (No.) | SN/R (No.) |
|---|---|---|---|---|---|---|---|---|
| Closing cut date | ||||||||
| Uncut | 484.96 b | 94.62 b | 83.31 | 173.60 | 88.37 ab | 2.07 ab | 3.54 | 37.05 ab |
| 15 June | 510.23 ab | 105.25 a | 89.31 | 171.67 | 86.30 ab | 1.87 b | 3.26 | 38.34 a |
| 1 July | 660.44 a | 92.88 b | 85.90 | 182.65 | 92.81 a | 2.14 a | 3.61 | 34.79 b |
| 15 July | 479.29 b | 99.69 ab | 80.85 | 157.39 | 82.01 b | 1.90 ab | 3.61 | 34.58 b |
| CV (%) | 40.20 | 12.20 | 19.70 | 25.50 | 13.10 | 16.70 | 14.50 | 12.30 |
| N-rate (kg N/ha) | ||||||||
| 0 | 587.25 | 87.42 b | 80.96 | 174.03 | 93.01 a | 2.13 | 3.73 a | 36.05 |
| 50 | 562.57 | 100.94 a | 89.04 | 175.19 | 88.45 ab | 1.95 | 3.40 ab | 35.24 |
| 100 | 443.62 | 100.15 a | 86.38 | 159.54 | 86.62 ab | 1.85 | 3.66 a | 37.01 |
| 150 | 541.47 | 103.94 a | 83.00 | 176.54 | 81.41 b | 2.05 | 3.23 b | 36.44 |
| CV (%) | 45.70 | 14.90 | 23.20 | 31.50 | 17.20 | 21.00 | 15.20 | 12.20 |
| Mean | 533.73 | 98.11 | 84.84 | 171.33 | 87.37 | 2.00 | 3.51 | 36.19 |
| CCD × N-rate | ns | ns | ns | ns | ns | ns | ns | ns |
| Traits | CCD | TSY | TN | FTN | IN | FTP | IN/T | RN/I |
|---|---|---|---|---|---|---|---|---|
| TSY | −0.3600 | |||||||
| TN | −0.3668 | −0.5223 | ||||||
| FTN | −0.1675 | 0.3454 | 0.4199 | |||||
| IN | 0.2100 | 0.7733 | −0.4966 | 0.5782 | ||||
| FTP | 0.1931 | 0.8311 | −0.6407 | 0.4271 | 0.9817 ** | |||
| IN/T | 0.3686 | 0.6802 | −0.9443 * | −0.0990 | 0.7543 | 0.8555 | ||
| RN/I | 0.0378 | 0.2722 | −0.8100 | −0.8042 | −0.0536 | 0.1361 | 0.5890 | |
| SN/R | 0.4129 | −0.3906 | 0.5640 | 0.6699 | 0.1631 | −0.0174 | −0.3566 | −0.8943 * |
| Treatment | FSP (%) | TSW (g) | PP (%) | PSY (kg/ha) | GP (%) | PGSY (kg/ha) |
|---|---|---|---|---|---|---|
| Closing cut date | ||||||
| Uncut | 24.56 b | 8.42 ab | 98.73 b | 478.82 b | 91.62 | 439.71 b |
| 15 June | 28.55 ab | 8.51 ab | 99.00 ab | 504.87 ab | 93.02 | 474.05 ab |
| 1 July | 35.08 a | 8.58 a | 99.22 a | 654.97 a | 93.28 | 611.31 a |
| 15 July | 28.70 ab | 8.32 b | 98.72 b | 473.39 b | 91.38 | 432.55 b |
| CV (%) | 28.40 | 3.40 | 0.50 | 40.40 | 3.50 | 41.40 |
| N-rate (kg N/ha) | ||||||
| 0 | 33.52 a | 8.72 a | 99.30 a | 582.89 | 92.55 | 539.26 |
| 50 | 30.03 ab | 8.49 ab | 98.47 b | 553.68 | 92.97 | 514.97 |
| 100 | 27.52 b | 8.34 b | 98.99 ab | 439.62 | 92.31 | 408.71 |
| 150 | 25.82 b | 8.29 b | 98.91 ab | 535.86 | 91.47 | 494.68 |
| CV (%) | 21.50 | 4.30 | 0.80 | 45.50 | 5.10 | 46.30 |
| Mean | 29.22 | 8.46 | 98.92 | 528.01 | 92.32 | 489.40 |
| CCD × N-rate | ns | ns | ns | ns | ns | ns |
| Traits | CCD | FSP | TSW | PP | PSY | GP |
|---|---|---|---|---|---|---|
| FSP | −0.7112 | |||||
| TSW | −0.1391 | 0.6318 | ||||
| PP | −0.4614 | 0.8609 | 0.9326 * | |||
| PSY | −0.3621 | 0.9089 * | 0.8176 | 0.9161 * | ||
| GP | −0.4023 | 0.7084 | 0.9515 * | 0.9596 ** | 0.7725 | |
| PGSY | −0.3711 | 0.9072 * | 0.8460 | 0.9376 * | 0.9983 ** | 0.8082 |
| Traits | N-Rate | TSY | TN | FTN | IN | FTP | IN/T | RN/I |
|---|---|---|---|---|---|---|---|---|
| TSY | −0.5259 | |||||||
| TN | 0.8610 | −0.4558 | ||||||
| FTN | 0.1248 | −0.3545 | 0.5828 | |||||
| IN | −0.1323 | 0.9084 * | −0.0668 | −0.2586 | ||||
| FTP | −0.9856 ** | 0.3914 | −0.8884 * | −0.1462 | −0.0247 | |||
| IN/T | −0.3612 | 0.8466 | −0.5798 | −0.7948 | 0.7571 | 0.2823 | ||
| RN/I | −0.6904 | −0.1618 | −0.7842 | −0.2286 | −0.5548 | 0.8025 | −0.0296 | |
| SN/R | 0.5155 | −0.7649 | 0.1253 | −0.3261 | −0.7035 | −0.3715 | −0.3048 | 0.2403 |
| Traits | N-Rate | FSP | TSW | PP | PSY | GP |
|---|---|---|---|---|---|---|
| FSP | −0.9880 ** | |||||
| TSW | −0.9630 ** | 0.9929 ** | ||||
| PP | −0.2450 | 0.3445 | 0.3963 | |||
| PSY | −0.5310 | 0.6107 | 0.6767 | 0.0477 | ||
| GP | −0.7968 | 0.6971 | 0.6148 | −0.3038 | 0.2000 | |
| PGSY | −0.5455 | 0.6214 | 0.6845 | 0.0298 | 0.9996 ** | 0.2267 |
| Treatment | TSY (kg/ha) | TN (no./m2) | FTN (no./m2) | IN (no./m2) | FTP (%) | IN/T (no.) | RN/I (no.) | SN/R (no.) |
|---|---|---|---|---|---|---|---|---|
| Closing cut date | ||||||||
| Uncut | 303.17 | 163.60 | 127.46 | 315.00 | 76.86 | 2.30 | 3.42 | 36.33 a |
| 15 June | 298.85 | 173.81 | 126.25 | 293.93 | 73.73 | 2.28 | 3.39 | 34.62 ab |
| 1 July | 339.76 | 163.12 | 119.58 | 308.38 | 71.93 | 2.29 | 3.31 | 34.45 ab |
| 15 July | 361.02 | 162.75 | 115.42 | 327.30 | 69.56 | 2.36 | 3.05 | 30.77 b |
| CV (%) | 45.30 | 12.80 | 18.80 | 32.20 | 21.40 | 19.30 | 21.50 | 14.60 |
| N-rate (kg N/ha) | ||||||||
| 0 50 100 150 | 411.91 387.20 302.22 374.70 | 158.92 161.73 167.85 174.79 | 120.12 118.71 114.58 135.29 | 293.32 293.64 310.15 347.50 | 75.83 72.16 67.00 77.09 | 2.17 2.27 2.38 2.41 | 3.29 3.07 3.30 3.51 | 32.21 35.38 32.83 35.75 |
| CV (%) | 63.90 | 17.50 | 42.70 | 69.40 | 36.80 | 49.20 | 23.90 | 16.40 |
| Mean | 325.70 | 165.82 | 122.18 | 311.15 | 73.02 | 2.31 | 3.29 | 34.04 |
| CCD × N-rate | ns | ns | ns | ns | ns | ns | ns | ns |
| Traits | CCD | TSY | TN | FTN | IN | FTP | IN/T | RN/I |
|---|---|---|---|---|---|---|---|---|
| TSY | −0.6000 | |||||||
| TN | −0.1754 | −0.6503 | ||||||
| FTN | 0.7072 | −0.9893 ** | 0.5337 | |||||
| IN | 0.0675 | 0.7285 | −0.8455 | −0.6401 | ||||
| FTP | 0.8914 * | −0.8821 * | 0.2174 | 0.9413 * | −0.3919 | |||
| IN/T | −0.2454 | 0.7815 | −0.5508 | −0.7522 | 0.8946 * | −0.6356 | ||
| RN/I | 0.5993 | −0.9167 * | 0.4430 | 0.9289 * | −0.7345 | 0.8879 * | −0.9195 * | |
| SN/R | 0.7261 | −0.8437 | 0.2263 | 0.8892 * | −0.5645 | 0.9288 * | −0.8398 | 0.9724 |
| Treatment | FSP (%) | TSW (g) | PP (%) | PSY (kg/ha) | GP (%) | PGSY (kg/ha) |
|---|---|---|---|---|---|---|
| Closing cut date | ||||||
| Uncut | 18.25 | 5.72 | 95.93 | 290.90 | 93.03 | 269.56 |
| 15 June | 12.38 | 5.79 | 95.21 | 287.54 | 91.72 | 264.05 |
| 1 July | 14.61 | 5.72 | 95.31 | 325.12 | 91.06 | 299.10 |
| 15 July | 12.12 | 5.90 | 87.68 | 340.75 | 93.17 | 318.26 |
| CV (%) | 61.90 | 6.60 | 13.60 | 46.80 | 6.30 | 48.30 |
| N-rate (kg N/ha) | ||||||
| 0 50 100 150 | 16.40 12.17 14.43 14.33 | 5.90 ab 5.98 a 5.73 ab 5.55 b | 90.08 97.36 92.62 94.07 | 301.62 331.58 289.13 321.96 | 93.90 92.28 90.72 92.12 | 282.18 304.43 266.32 298.04 |
| CV (%) | 64.50 | 8.60 | 13.60 | 64.80 | 5.50 | 65.20 |
| Mean | 14.37 | 5.78 | 93.53 | 311.07 | 92.23 | 287.74 |
| CCD × N-rate | ns | ns | ns | ns | ns | ns |
| Traits | CCD | FSP | TSW | PP | PSY | GP |
|---|---|---|---|---|---|---|
| FSP | 0.9171 * | |||||
| TSW | −0.5491 | −0.7422 | ||||
| PP | 0.4982 | 0.5872 | −0.9377 * | |||
| PSY | −0.6109 | −0.4277 | 0.5556 | −0.7727 | ||
| GP | 0.4388 | 0.1978 | 0.4964 | −0.5415 | 0.0913 | |
| PGSY | −0.5743 | −0.4116 | 0.5895 | −0.8070 | 0.9972 ** | 0.1649 |
| Traits | N-Rate | TSY | TN | FTN | IN | FTP | IN/T | RN/I |
|---|---|---|---|---|---|---|---|---|
| TSY | −0.5385 | |||||||
| TN | 0.9845 ** | −0.4567 | ||||||
| FTN | 0.5900 | 0.3364 | 0.6823 | |||||
| IN | 0.9074 * | −0.2248 | 0.9633 ** | 0.8406 | ||||
| FTP | −0.0394 | 0.8285 | 0.0880 | 0.7835 | 0.3438 | |||
| IN/T | 0.9771 ** | −0.6991 | 0.9357 * | 0.4050 | 0.8068 | −0.2508 | ||
| RN/I | 0.6394 | −0.1345 | 0.7618 | 0.7318 | 0.8621 | 0.4199 | 0.5333 | |
| SN/R | 0.5845 | 0.1445 | 0.5316 | 0.6348 | 0.5324 | 0.3285 | 0.4927 | 0.0721 |
| Traits | N-Rate | FSP | TSW | PP | PSY | GP |
|---|---|---|---|---|---|---|
| FSP | −0.2951 | |||||
| TSW | −0.8789 * | −0.1906 | ||||
| PP | −0.6613 | −0.2844 | 0.7748 | |||
| PSY | 0.1246 | −0.6624 | 0.1416 | 0.6618 | ||
| GP | −0.6845 | 0.4739 | 0.4161 | 0.6840 | 0.2287 | |
| PGSY | 0.0716 | −0.5615 | 0.1404 | 0.6945 | 0.9917 ** | 0.3415 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Juntasin, W.; Imura, Y.; Nakamura, I.; Hossain, M.A.; Thaikua, S.; Poungkaew, R.; Kawamoto, Y. Effects of Closing Cut Date and Nitrogen Fertilization on Seed Yield and Seed Quality in Two Novel Cultivars of Urochloa spp. Agronomy 2022, 12, 513. https://doi.org/10.3390/agronomy12020513
Juntasin W, Imura Y, Nakamura I, Hossain MA, Thaikua S, Poungkaew R, Kawamoto Y. Effects of Closing Cut Date and Nitrogen Fertilization on Seed Yield and Seed Quality in Two Novel Cultivars of Urochloa spp. Agronomy. 2022; 12(2):513. https://doi.org/10.3390/agronomy12020513
Chicago/Turabian StyleJuntasin, Weenaporn, Yoshimi Imura, Ichiro Nakamura, Mohammad Amzad Hossain, Sarayut Thaikua, Rattikan Poungkaew, and Yasuhiro Kawamoto. 2022. "Effects of Closing Cut Date and Nitrogen Fertilization on Seed Yield and Seed Quality in Two Novel Cultivars of Urochloa spp." Agronomy 12, no. 2: 513. https://doi.org/10.3390/agronomy12020513
APA StyleJuntasin, W., Imura, Y., Nakamura, I., Hossain, M. A., Thaikua, S., Poungkaew, R., & Kawamoto, Y. (2022). Effects of Closing Cut Date and Nitrogen Fertilization on Seed Yield and Seed Quality in Two Novel Cultivars of Urochloa spp. Agronomy, 12(2), 513. https://doi.org/10.3390/agronomy12020513






