Lignin–Chitosan Nanocarriers for the Delivery of Bioactive Natural Products against Wood-Decay Phytopathogens
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material and Reagents
2.2. Fungal and Bacterial Isolates
2.3. Preparation of Plant Extracts
2.4. Procedure for the Synthesis of the Biodegradable Nanocarriers
2.5. Characterization of the Nanocarriers
2.6. In Vitro Antimicrobial Activity Assessment
2.7. In Planta Application of ML–COS NCs-Based Treatments
2.8. Statistical Analysis
3. Results
3.1. Characterization by ATR-FTIR
3.2. Characterization by Thermal Analysis
3.3. Characterization by Transmission Electron Microscopy, Dynamic Light Scattering, and Zeta Potential
3.4. In Vitro Antimicrobial Activity
3.4.1. Antifungal Activity
3.4.2. Antibacterial Activity
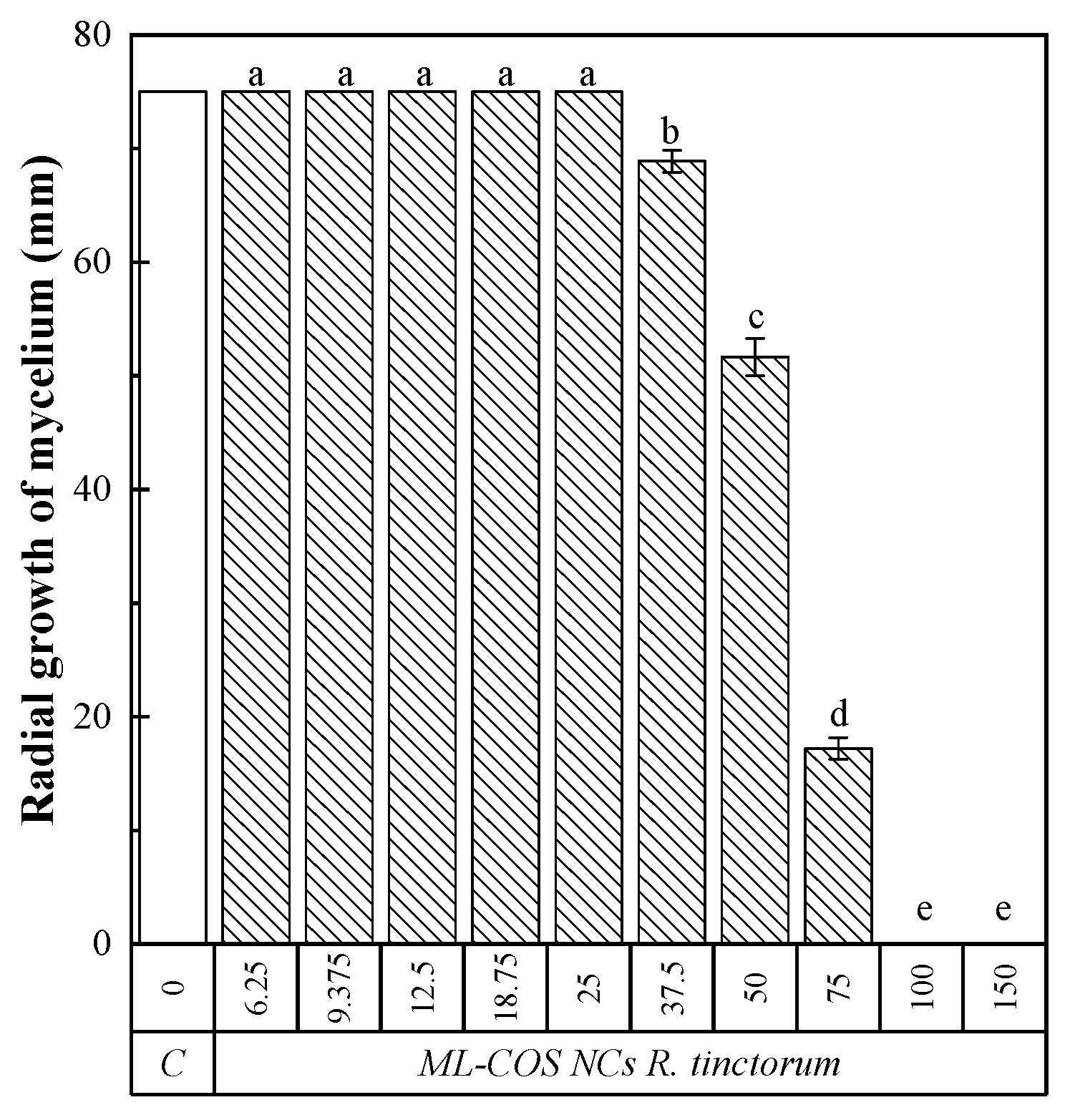
3.5. In Planta Application of ML–COS NCs Loaded with R. tinctorum Extract Treatment
4. Discussion
4.1. Comparison with Other NCs-Based Treatments
4.2. Limitations of the Study
5. Conclusions
6. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mondello, V.; Songy, A.; Battiston, E.; Pinto, C.; Coppin, C.; Trotel-Aziz, P.; Clément, C.; Mugnai, L.; Fontaine, F. Grapevine trunk diseases: A review of fifteen years of trials for their control with chemicals and biocontrol agents. Plant Dis. 2018, 102, 1189–1217. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wagschal, I.; Abou-Mansour, E.; Petit, A.-N. Wood diseases of grapevine: A review on eutypa dieback and esca. In Plant-Microbe Interactions; Ait Barka, E., Clément, C., Eds.; Research Signpost: Trivandrum, India, 2008; pp. 1–25. [Google Scholar]
- Bertsch, C.; Ramírez-Suero, M.; Magnin-Robert, M.; Larignon, P.; Chong, J.; Abou-Mansour, E.; Spagnolo, A.; Clément, C.; Fontaine, F. Grapevine trunk diseases: Complex and still poorly understood. Plant Pathol. 2013, 62, 243–265. [Google Scholar] [CrossRef] [Green Version]
- Gramaje, D.; Úrbez-Torres, J.R.; Sosnowski, M.R. Managing grapevine trunk diseases with respect to etiology and epidemiology: Current strategies and future prospects. Plant Dis. 2018, 102, 12–39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hofstetter, V.; Buyck, B.; Croll, D.; Viret, O.; Couloux, A.; Gindro, K. What if esca disease of grapevine were not a fungal disease? Fungal Divers. 2012, 54, 51–67. [Google Scholar] [CrossRef] [Green Version]
- Broda, M. Natural Compounds for Wood Protection against Fungi—A Review. Molecules 2020, 25, 3538. [Google Scholar] [CrossRef]
- Yiamsawas, D.; Beckers, S.J.; Lu, H.; Landfester, K.; Wurm, F.R. Morphology-controlled synthesis of lignin nanocarriers for drug delivery and carbon materials. ACS Biomater. Sci. Eng. 2017, 3, 2375–2383. [Google Scholar] [CrossRef]
- Yiamsawas, D.; Baier, G.; Thines, E.; Landfester, K.; Wurm, F.R. Biodegradable lignin nanocontainers. RSC Adv. 2014, 4, 11661–11663. [Google Scholar] [CrossRef] [Green Version]
- Fischer, J.; Beckers, S.J.; Yiamsawas, D.; Thines, E.; Landfester, K.; Wurm, F.R. Targeted drug delivery in plants: Enzyme-responsive lignin nanocarriers for the curative treatment of the worldwide grapevine trunk disease Esca. Adv. Sci. 2019, 6, 1802315. [Google Scholar] [CrossRef] [Green Version]
- Wurm, F.R.; Weiss, C.K. Nanoparticles from renewable polymers. Front. Chem. 2014, 2, 49. [Google Scholar] [CrossRef] [Green Version]
- Pathania, D.; Gupta, D.; Agarwal, S.; Asif, M.; Gupta, V.K. Fabrication of chitosan-g-poly(acrylamide)/CuS nanocomposite for controlled drug delivery and antibacterial activity. Mater. Sci. Eng. C 2016, 64, 428–435. [Google Scholar] [CrossRef]
- Beckers, S.J.; Wetherbee, L.; Fischer, J.; Wurm, F.R. Fungicide-loaded and biodegradable xylan-based nanocarriers. Biopolymers 2020, 111, e23413. [Google Scholar] [CrossRef] [PubMed]
- Ciftci, N.; Sargin, I.; Arslan, G.; Arslan, U.; Okudan, A. Ascorbic acid adsorption-release performance and antibacterial activity of chitosan-ter(GMA-MA-NTBA) polymer microcapsules. J. Polym. Environ. 2020, 28, 2277–2288. [Google Scholar] [CrossRef]
- Zou, T.; Sipponen, M.H.; Österberg, M. Natural shape-retaining microcapsules with shells made of chitosan-coated colloidal lignin particles. Front. Chem. 2019, 7, 370. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rosova, E.; Smirnova, N.; Dresvyanina, E.; Smirnova, V.; Vlasova, E.; Ivan’kova, E.; Sokolova, M.; Maslennikova, T.; Malafeev, K.; Kolbe, K.; et al. Biocomposite materials based on chitosan and lignin: Preparation and characterization. Cosmetics 2021, 8, 24. [Google Scholar] [CrossRef]
- Aradmehr, A.; Javanbakht, V. A novel biofilm based on lignocellulosic compounds and chitosan modified with silver nanoparticles with multifunctional properties: Synthesis and characterization. Colloids Surf. A Physicochem. Eng. Asp. 2020, 600, 124952. [Google Scholar] [CrossRef]
- Machado, T.O.; Beckers, S.J.; Fischer, J.; Müller, B.; Sayer, C.; de Araújo, P.H.H.; Landfester, K.; Wurm, F.R. Bio-based lignin nanocarriers loaded with fungicides as a versatile platform for drug delivery in plants. Biomacromolecules 2020, 21, 2755–2763. [Google Scholar] [CrossRef]
- Peil, S.; Beckers, S.J.; Fischer, J.; Wurm, F.R. Biodegradable, lignin-based encapsulation enables delivery of Trichoderma reesei with programmed enzymatic release against grapevine trunk diseases. Mater. Today Bio 2020, 7, 100061. [Google Scholar] [CrossRef]
- Langa-Lomba, N.; Buzón-Durán, L.; Martín-Ramos, P.; Casanova-Gascón, J.; Martín-Gil, J.; Sánchez-Hernández, E.; González-García, V. Assessment of conjugate complexes of chitosan and Urtica dioica or Equisetum arvense extracts for the control of grapevine trunk pathogens. Agronomy 2021, 11, 976. [Google Scholar] [CrossRef]
- Langa-Lomba, N.; Sánchez-Hernández, E.; Buzón-Durán, L.; González-García, V.; Casanova-Gascón, J.; Martín-Gil, J.; Martín-Ramos, P. Activity of anthracenediones and flavoring phenols in hydromethanolic extracts of Rubia tinctorum against grapevine phytopathogenic fungi. Plants 2021, 10, 1527. [Google Scholar] [CrossRef]
- Langa-Lomba, N.; Buzón-Durán, L.; Sánchez-Hernández, E.; Martín-Ramos, P.; Casanova-Gascón, J.; Martín-Gil, J.; González-García, V. Antifungal activity against Botryosphaeriaceae fungi of the hydro-methanolic extract of Silybum marianum capitula conjugated with stevioside. Plants 2021, 10, 1363. [Google Scholar] [CrossRef]
- Leonardo, D.-C. Botryosphariaceae species associated with stem canker, die-back and fruit rot on apple in Uruguay. Eur. J. Plant Pathol. 2016, 146, 637–655. [Google Scholar] [CrossRef]
- Szegedi, E.; Civerolo, E.L. Bacterial diseases of grapevine. Int. J. Hortic. Sci. 2011, 17, 45–49. [Google Scholar] [CrossRef] [Green Version]
- Stevens, N.E. Two apple black rot fungi in the United States. Mycologia 2018, 25, 536–548. [Google Scholar] [CrossRef]
- Brown-Rytlewski, D.E.; McManus, P.S. Virulence of Botryosphaeria dothidea and Botryosphaeria obtusa on apple and management of stem cankers with fungicides. Plant Dis. 2000, 84, 1031–1037. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Willems, A.; Gillis, M.; Kersters, K.; Van Den Broecke, L.; De Ley, J. Transfer of Xanthomonas ampelina Panagopoulos 1969 to a new genus, Xylophilus gen. nov., as Xylophilus ampelinus (Panagopoulos 1969) comb. nov. Int. J. Syst. Bacteriol. 1987, 37, 422–430. [Google Scholar] [CrossRef]
- Gerin, D.; Cariddi, C.; de Miccolis Angelini, R.M.; Rotolo, C.; Dongiovanni, C.; Faretra, F.; Pollastro, S. First report of Pseudomonas grapevine bunch rot caused by Pseudomonas syringae pv. syringae. Plant Dis. 2019, 103, 1954–1960. [Google Scholar] [CrossRef]
- Santos-Moriano, P.; Fernandez-Arrojo, L.; Mengibar, M.; Belmonte-Reche, E.; Peñalver, P.; Acosta, F.N.; Ballesteros, A.O.; Morales, J.C.; Kidibule, P.; Fernandez-Lobato, M.; et al. Enzymatic production of fully deacetylated chitooligosaccharides and their neuroprotective and anti-inflammatory properties. Biocatal. Biotransform. 2017, 36, 57–67. [Google Scholar] [CrossRef] [Green Version]
- Buzón-Durán, L.; Martín-Gil, J.; Pérez-Lebeña, E.; Ruano-Rosa, D.; Revuelta, J.L.; Casanova-Gascón, J.; Ramos-Sánchez, M.C.; Martín-Ramos, P. Antifungal agents based on chitosan oligomers, ε-polylysine and Streptomyces spp. secondary metabolites against three Botryosphaeriaceae species. Antibiotics 2019, 8, 99. [Google Scholar] [CrossRef] [Green Version]
- Sannan, T.; Kurita, K.; Iwakura, Y. Studies on chitin, 2. Effect of deacetylation on solubility. Makromol. Chem. 1976, 177, 3589–3600. [Google Scholar] [CrossRef]
- Yang, Y.; Shu, R.; Shao, J.; Xu, G.; Gu, X. Radical scavenging activity of chitooligosaccharide with different molecular weights. Eur. Food Res. Technol. 2005, 222, 36–40. [Google Scholar] [CrossRef]
- Maghami, G.G.; Roberts, G.A.F. Evaluation of the viscometric constants for chitosan. Makromol. Chem. 1988, 189, 195–200. [Google Scholar] [CrossRef]
- Tian, M.; Tan, H.; Li, H.; You, C. Molecular weight dependence of structure and properties of chitosan oligomers. RSC Adv. 2015, 5, 69445–69452. [Google Scholar] [CrossRef]
- Krizsán, K.; Szókán, G.; Toth, Z.A.; Hollósy, F.; László, M.; Khlafulla, A. HPLC analysis of anthraquinone derivatives in madder root (Rubia tinctorum) and its cell cultures. J. Liq. Chromatogr. Relat. Technol. 2006, 19, 2295–2314. [Google Scholar] [CrossRef]
- Cruz, G.; Crnkovic, P.M. Investigation into the kinetic behavior of biomass combustion under N2/O2 and CO2/O2 atmospheres. J. Therm. Anal. Calorim. 2015, 123, 1003–1011. [Google Scholar] [CrossRef]
- Arendrup, M.C.; Cuenca-Estrella, M.; Lass-Flörl, C.; Hope, W. EUCAST technical note on the EUCAST definitive document EDef 7.2: Method for the determination of broth dilution minimum inhibitory concentrations of antifungal agents for yeasts EDef 7.2 (EUCAST-AFST). Clin. Microbiol. Infect. 2012, 18, E246–E247. [Google Scholar] [CrossRef] [Green Version]
- CLSI. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically, 11th ed.; CLSI standard M07; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2018. [Google Scholar]
- Choudhary, R.C.; Kumari, S.; Kumaraswamy, R.V.; Pal, A.; Raliya, R.; Biswas, P.; Saharan, V. Characterization methods for chitosan-based nanomaterials. In Plant Nanobionics; Springer: Cham, Switzerland, 2019; pp. 103–116. [Google Scholar] [CrossRef]
- Kumaraswamy, R.V.; Kumari, S.; Choudhary, R.C.; Pal, A.; Raliya, R.; Biswas, P.; Saharan, V. Engineered chitosan based nanomaterials: Bioactivities, mechanisms and perspectives in plant protection and growth. Int. J. Biol. Macromol. 2018, 113, 494–506. [Google Scholar] [CrossRef]
- Bhattacharjee, S. DLS and zeta potential—What they are and what they are not? J. Controll. Release 2016, 235, 337–351. [Google Scholar] [CrossRef]
- Machado, T.O.; Beckers, S.J.; Fischer, J.; Sayer, C.; de Araújo, P.H.H.; Landfester, K.; Wurm, F.R. Cellulose nanocarriers via miniemulsion allow pathogen-specific agrochemical delivery. J. Colloid Interface Sci. 2021, 601, 678–688. [Google Scholar] [CrossRef]
- Wurm, F.; Landfester, K.; Yiamsawas, D.; Thines, E.; Fischer, J. Lignin Biomaterial as Agricultural Drug Carrier. U.S. Patent 2019/0037837 A1, 7 February 2019. [Google Scholar]
- Tang, G.; Tian, Y.; Niu, J.; Tang, J.; Yang, J.; Gao, Y.; Chen, X.; Li, X.; Wang, H.; Cao, Y. Development of carrier-free self-assembled nanoparticles based on fenhexamid and polyhexamethylene biguanide for sustainable plant disease management. Green Chem. 2021, 23, 2531–2540. [Google Scholar] [CrossRef]


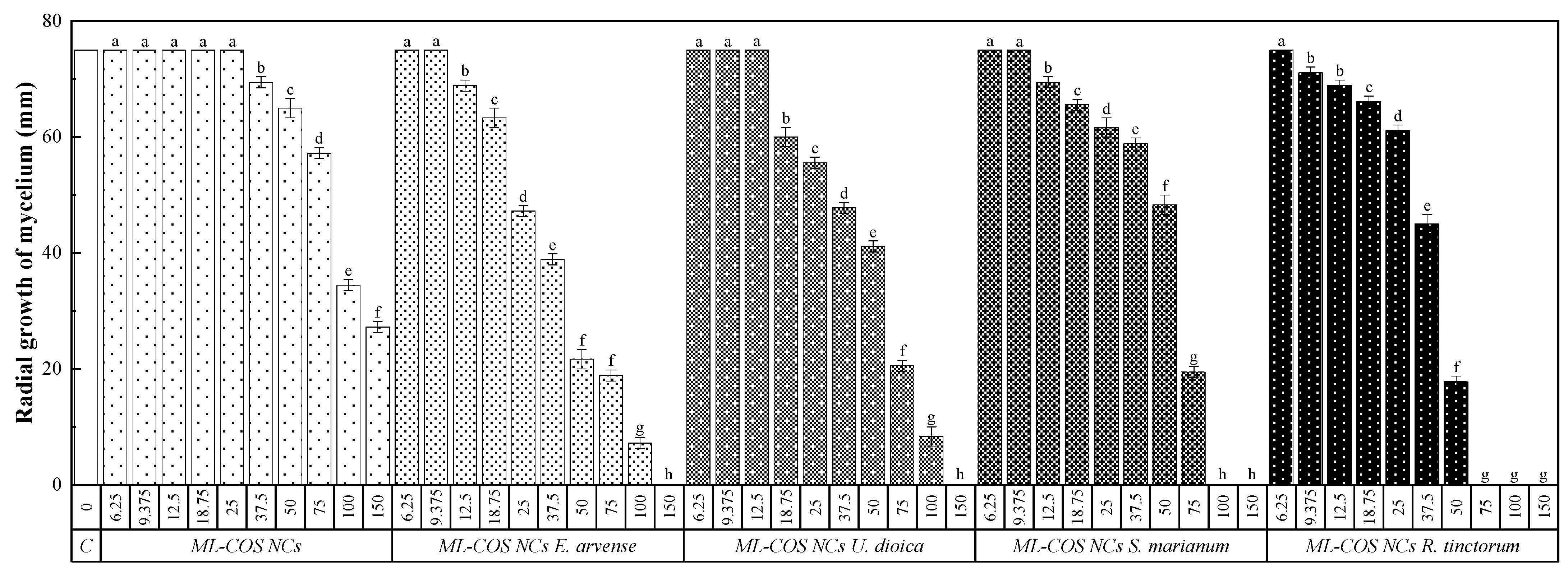

| Effective Concentration | NC-Based Treatments | Non-Encapsulated Products [19,20,21] | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| ML–COS | ML–COS– E. arvense | ML–COS– U. dioica | ML–COS– S. marianum | ML–COS– R. tinctorum | COS | E. arvense | U. dioica | S. marianum | R. tinctorum | |
| EC50 | 82.7 | 66.5 | 50.2 | 60.9 | 41.2 | 680.2 | * | * | 557 | 92.3 |
| EC90 | 243.2 | 105.2 | 113.0 | 90.6 | 65.8 | 1326.6 | * | * | 2938 | 184.0 |
| Effective Concentration | Encapsulated Product | Non-Encapsulated Product |
|---|---|---|
| ML–COS–R. tinctorum | R. tinctorum | |
| EC50 | 59.3 | 78.0 |
| EC90 | 91.0 | 87.8 |
| Pathogen | Concentration | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 6.25 | 9.75 | 12.5 | 18.75 | 25 | 37.5 | 50 | 75 | 100 | 150 | |
| P. syringae pv. syringae | + | + | + | + | + | − | − | − | − | − |
| X. ampelinus | + | + | + | + | + | − | − | − | − | − |
| Composition | Pathogen | Assays | Ref. |
|---|---|---|---|
| Kraft cationic lignin NCs loaded with Trichoderma reesei spores | Fungi associated with GTDs (esca) | Dual in vitro culture against Phaeomoniella chlamydospora and Phaeoacremonium minimum + in vitro NC degradation assays with culture filtrate of the two esca pathogens | [18] |
| Methacrylated Kraft lignin NCs loaded with pyraclostrobin | Fungi associated with GTDs (esca) | Field study with trunk injections in V. vinifera plants | [9] |
| Methacrylated Kraft lignin NCs loaded with different synthetic fungicides (pyraclostrobin, azoxystrobin, tebuconazole, boscalid) | Ligninase-producing microorganisms | In vitro antifungal activity against P. chlamydospora, Neonectria ditissima, Phytophthora infestans, Magnoporthe oryzae, Botrytis cinerea, N. parvum + in planta study through trunk injections in V. vinifera plants | [17] |
| Pyraclostrobin-loaded xylan NCs | Xylanase-producing fungi in viticulture and horticulture | In vitro assays against Pyricularia oryzae, B. cinerea, P. chlamydospora, P. minimum, and N. ditissima | [12] |
| Cellulose modified with undec-10-enoic acid NCs loaded with pyraclostrobin and captan | Cellulase segregating fungi in viticulture and apple trees | In vitro assays against P. chlamydospora, N. ditissima, P. infestans, M. oryzae, B. cinerea, and N. parvum | [41] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez-Hernández, E.; Langa-Lomba, N.; González-García, V.; Casanova-Gascón, J.; Martín-Gil, J.; Santiago-Aliste, A.; Torres-Sánchez, S.; Martín-Ramos, P. Lignin–Chitosan Nanocarriers for the Delivery of Bioactive Natural Products against Wood-Decay Phytopathogens. Agronomy 2022, 12, 461. https://doi.org/10.3390/agronomy12020461
Sánchez-Hernández E, Langa-Lomba N, González-García V, Casanova-Gascón J, Martín-Gil J, Santiago-Aliste A, Torres-Sánchez S, Martín-Ramos P. Lignin–Chitosan Nanocarriers for the Delivery of Bioactive Natural Products against Wood-Decay Phytopathogens. Agronomy. 2022; 12(2):461. https://doi.org/10.3390/agronomy12020461
Chicago/Turabian StyleSánchez-Hernández, Eva, Natalia Langa-Lomba, Vicente González-García, José Casanova-Gascón, Jesús Martín-Gil, Alberto Santiago-Aliste, Sergio Torres-Sánchez, and Pablo Martín-Ramos. 2022. "Lignin–Chitosan Nanocarriers for the Delivery of Bioactive Natural Products against Wood-Decay Phytopathogens" Agronomy 12, no. 2: 461. https://doi.org/10.3390/agronomy12020461
APA StyleSánchez-Hernández, E., Langa-Lomba, N., González-García, V., Casanova-Gascón, J., Martín-Gil, J., Santiago-Aliste, A., Torres-Sánchez, S., & Martín-Ramos, P. (2022). Lignin–Chitosan Nanocarriers for the Delivery of Bioactive Natural Products against Wood-Decay Phytopathogens. Agronomy, 12(2), 461. https://doi.org/10.3390/agronomy12020461











