Novel Perspective of Medicinal Mushroom Cultivations: A Review Case for ‘Magic’ Mushrooms
Abstract
1. Introduction
2. Medicinal Mushrooms
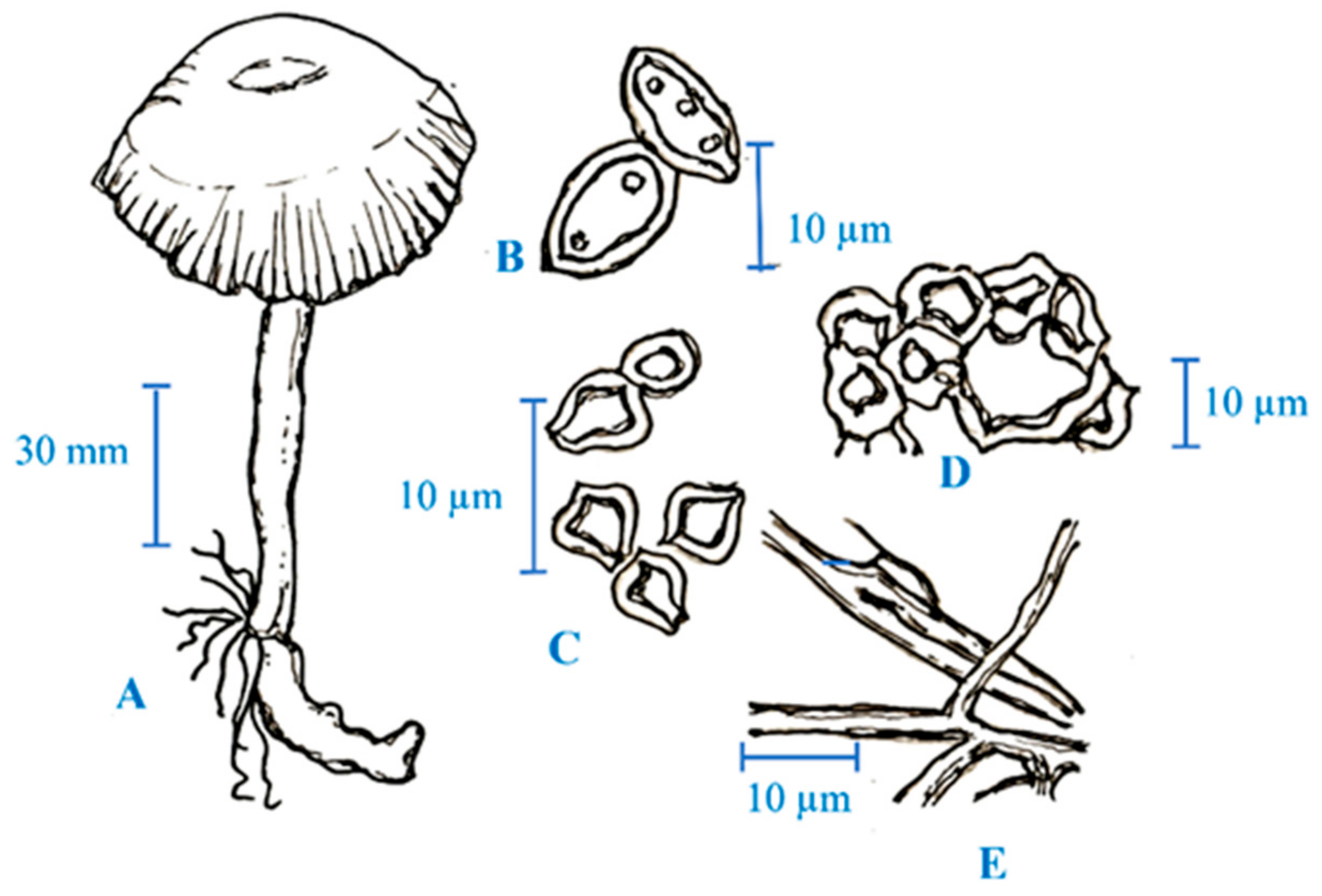
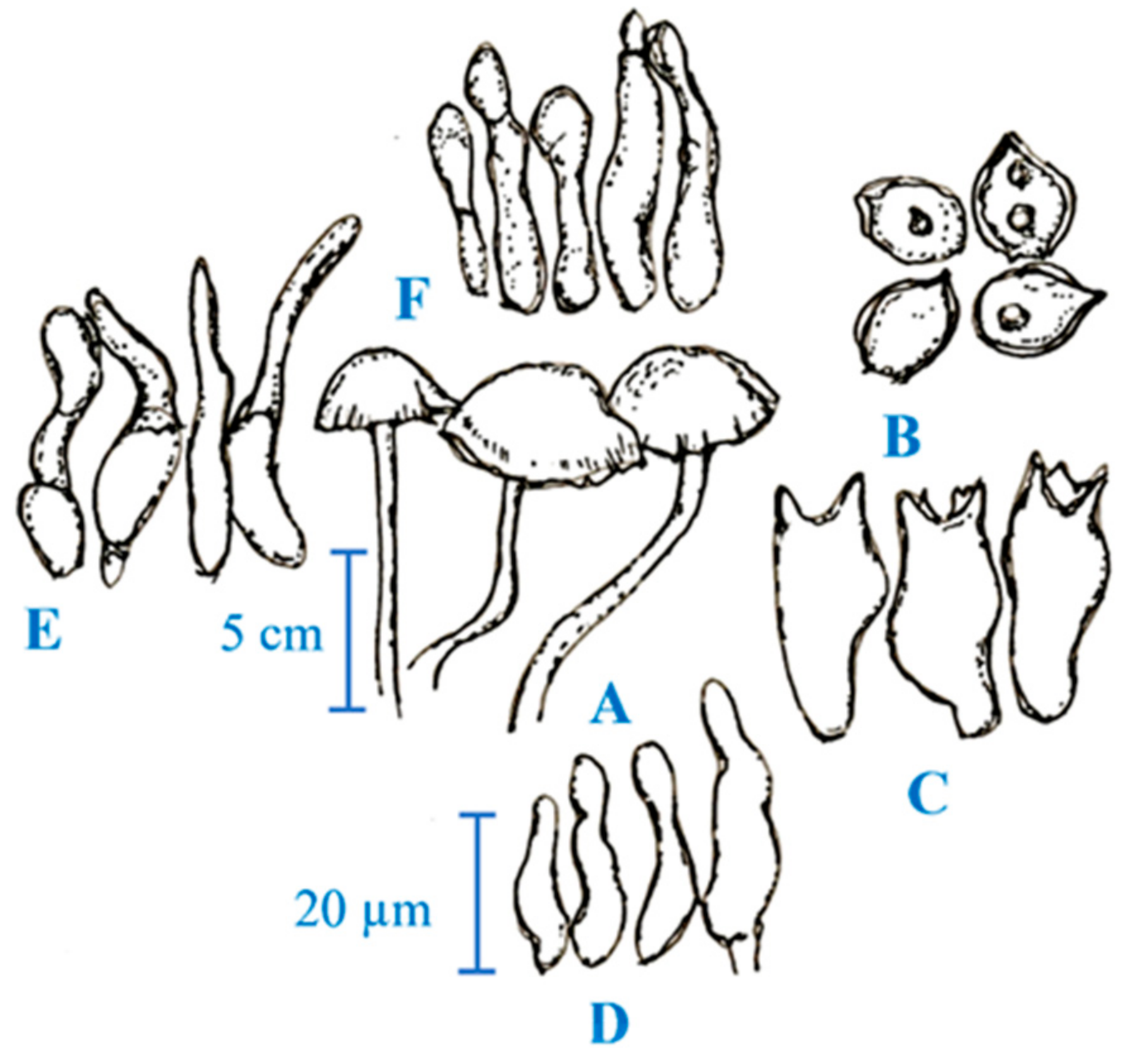
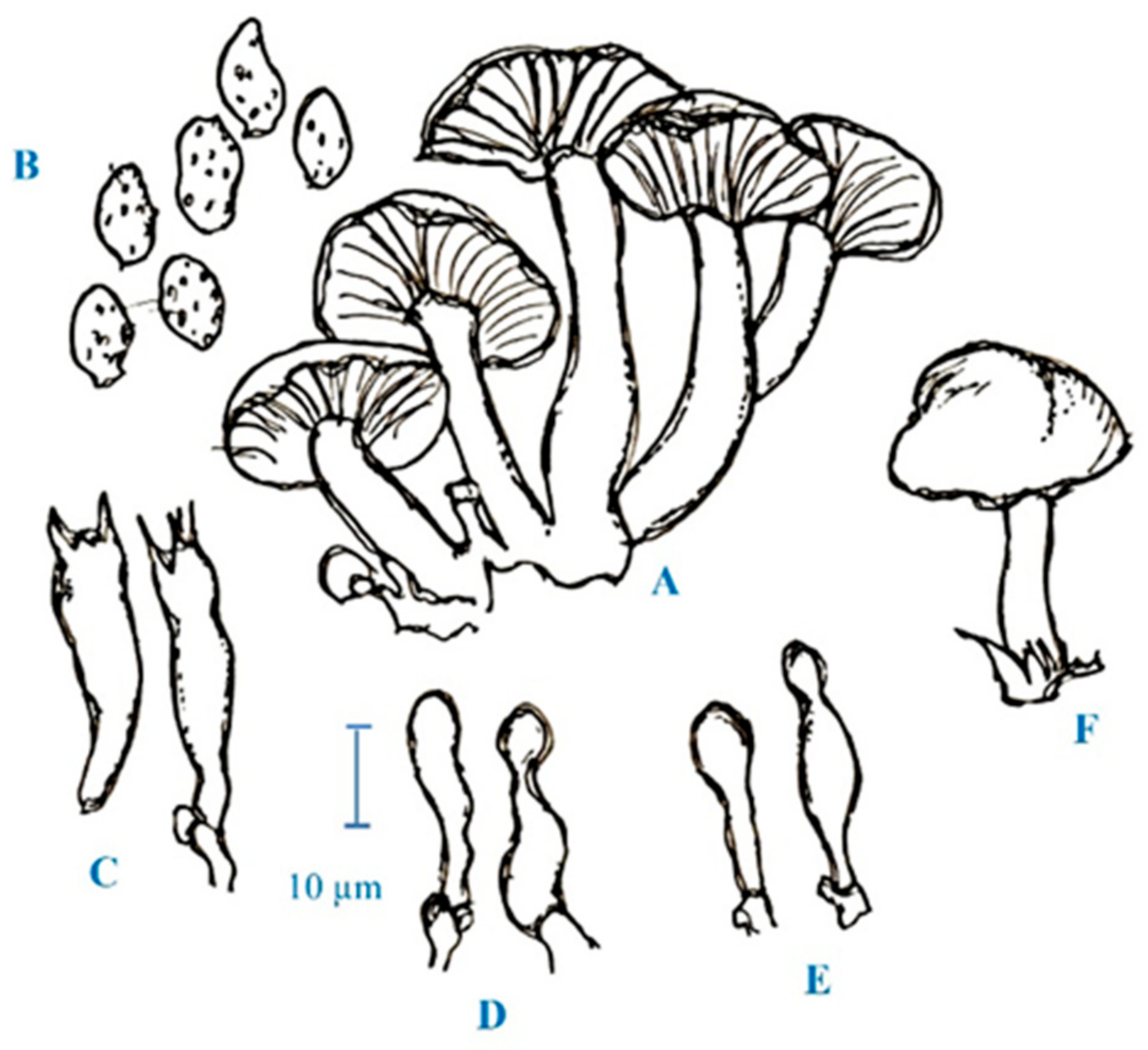
3. Medicinal Mushroom Diversity and Taxonomy Characteristics of Magic Mushrooms
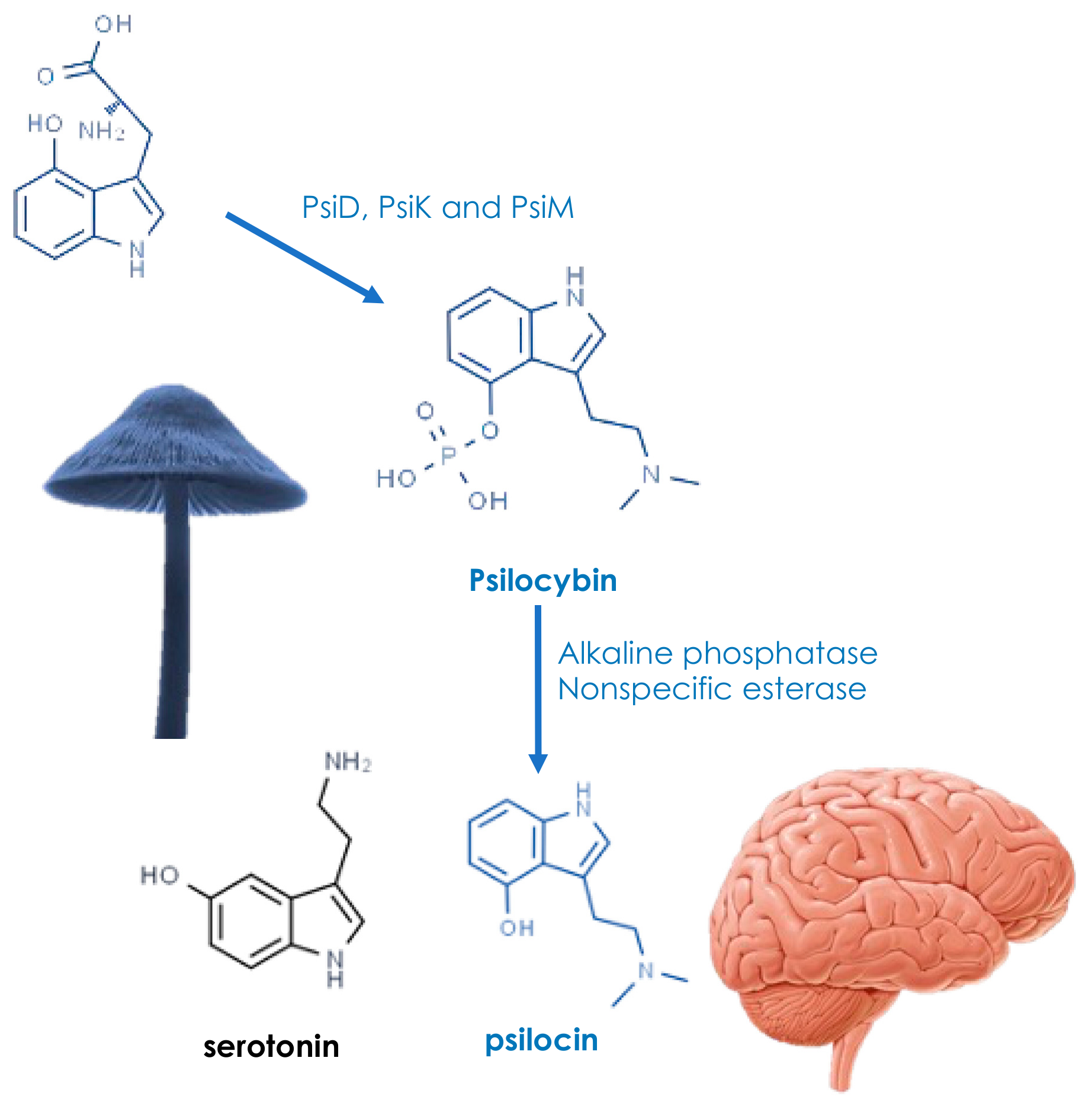
4. Active Ingredients and Medicinal Properties
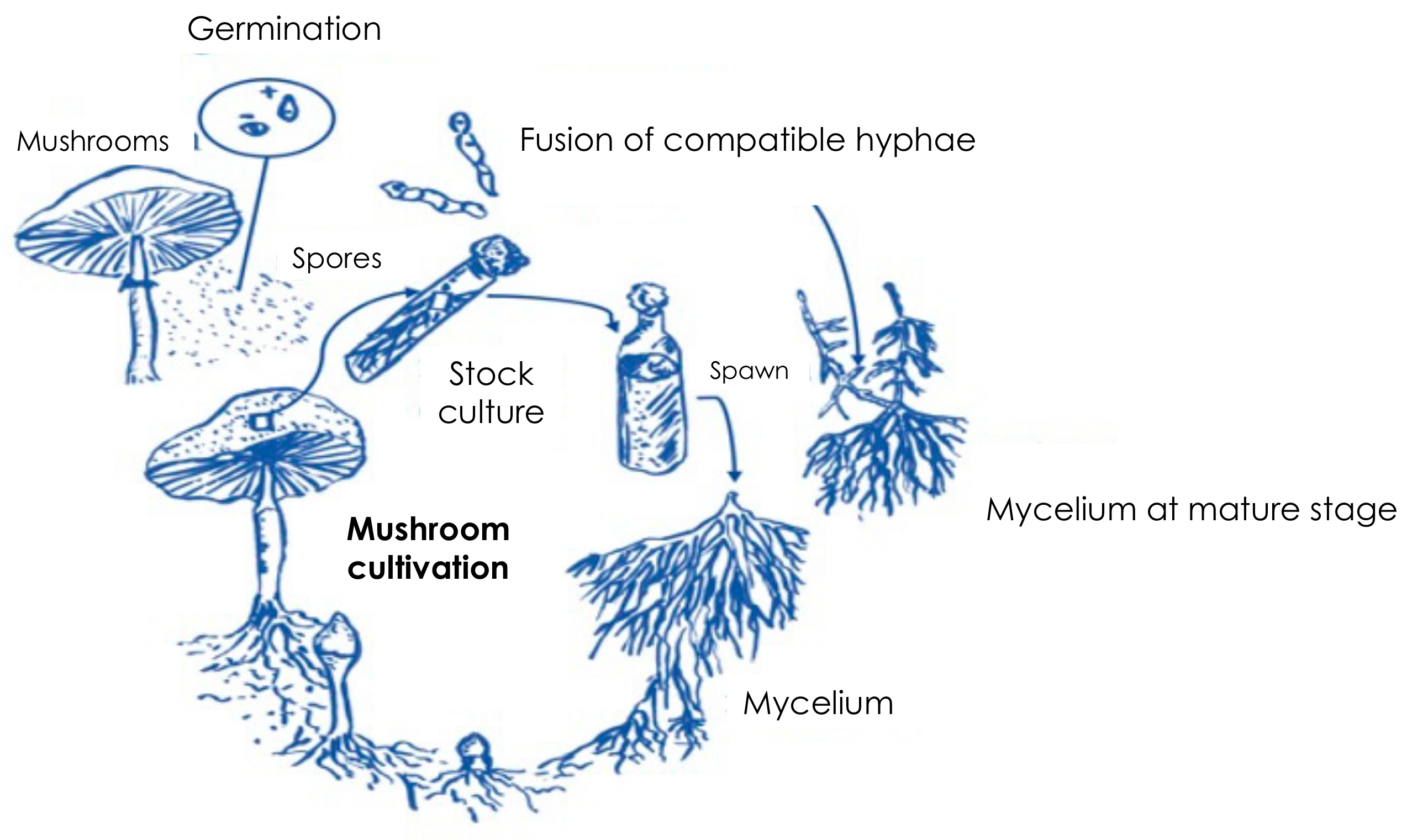
5. Traditional Cultivation
6. In Vitro Cultivation and Synthesis of the Active Metabolites from Medicinal Mushrooms
7. Future Perspective of Medicinal Mushroom Cultivation
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chaturvedi, V.K.; Agarwal, S.; Gupta, K.K.; Ramteke, P.W.; Singh, M.P. Medicinal mushroom: Boon for therapeutic applications. 3 Biotech 2018, 8, 334. [Google Scholar] [CrossRef]
- Elkhateeb, W.A. What medicinal mushroom can do. Chem. Res. J. 2020, 5, 106–118. [Google Scholar]
- Wasser, S. Medicinal mushroom science: Current perspectives, advances, evidences, and challenges. Biomed. J. 2014, 37, 345–356. [Google Scholar] [CrossRef]
- Mau, J.-L.; Lin, H.-C.; Chen, C.-C. Antioxidant Properties of Several Medicinal Mushrooms. J. Agric. Food Chem. 2002, 50, 6072–6077. [Google Scholar] [CrossRef]
- Gartz, J.; Wiedemann, G. Discovery of a new caerulescent Psilocybe mushroom in Germany: Psilocybe germanica sp.nov. Drug Test Anal. 2015, 7, 853–857. [Google Scholar] [CrossRef]
- Amsterdam, J.v.; Opperhuizen, A.; Brink, W.v.d. Harm potential of magic mushroom use: A review. Regul. Toxicol. Pharmacol. 2011, 59, 423–429. [Google Scholar] [CrossRef]
- Nkadimeng, S.M.; Nabatanzi, A.; Steinmann, C.M.L.; Eloff, J.N. Phytochemical, Cytotoxicity, Antioxidant and Anti-Inflammatory Effects of Psilocybe Natalensis Magic Mushroom. Plants 2020, 9, 1127. [Google Scholar] [CrossRef]
- Van Court, R.C.; Wiseman, M.S.; Meyer, K.W.; Ballhorn, D.J.; Amses, K.R.; Slot, J.C.; Dentinger, B.T.M.; Garibay-Orijel, R.; Uehling, J.K. Diversity, biology, and history of psilocybin-containing fungi: Suggestions for research and technological development. Fungal Biol. 2022, 126, 308–319. [Google Scholar] [CrossRef]
- Wasser, S.P. Current findings, future trends, and unsolved problems in studies of medicinal mushrooms. Appl. Microbiol. Biotechnol. 2011, 89, 1323–1332. [Google Scholar] [CrossRef]
- Kirk, P.; Cannon, P.; David, J.; Stalpers, J. Ainsworth and Brisby’s Dictionary of the Fungi; CAB International: Wallingford, UK, 2008. [Google Scholar]
- Ganeshpurkar, A.; Rai, G.; Jain, A.P. Medicinal mushrooms: Towards a new horizon. Pharm. Rev. 2010, 4, 127–135. [Google Scholar] [CrossRef]
- Guzmán, G.; Ramírez Guillén, F.; Hyde, K.D.; Karunarathna, S.C. Psilocybe sp. in Thailand: Four new species and a review of previously recorded species. Mycotaxon 2012, 119, 65–81. [Google Scholar] [CrossRef]
- Grimm, D.; Wösten, H.A.B. Mushroom cultivation in the circular economy. Appl. Microbiol. Biotechnol. 2018, 102, 7795–7803. [Google Scholar] [CrossRef]
- Gargano, M.L.; van Griensven, L.J.L.D.; Isikhuemhen, O.S.; Lindequist, U.; Venturella, G.; Wasser, S.P.; Zervakis, G.I. Medicinal mushrooms: Valuable biological resources of high exploitation potential. Plant Biosyst.-Int. J. Deal. All Asp. Plant Biol. 2017, 151, 548–565. [Google Scholar] [CrossRef]
- Chang, S.T.; Wasser, S.P. The cultivation and environmental impact of mushrooms. In Oxford Research Encyclopedia of Environmental Science; Oxford University Press: Oxford, UK, 2017. [Google Scholar]
- De Silva, D.D.; Rapior, S.; Hyde, K.D.; Bahkali, A.H. Medicinal mushrooms in prevention and control of diabetes mellitus. Fungal Divers. 2012, 56, 1–29. [Google Scholar] [CrossRef]
- Jeong, S.C.; Jeong, Y.T.; Yang, B.K.; Islam, R.; Koyyalamudi, S.R.; Pang, G.; Cho, K.Y.; Song, C.H. White button mushroom (Agaricus bisporus) lowers blood glucose and cholesterol levels in diabetic and hypercholesterolemic rats. Nutr. Res. 2010, 30, 49–56. [Google Scholar] [CrossRef]
- Volman, J.; Mensink, R.; Van Griensven, L.; Plat, J. Effects of α-glucans from Agaricus bisporus on ex vivo cytokine production by LPS and PHA-stimulated PBMCs; a placebo-controlled study in slightly hypercholesterolemic subjects. Eur. J. Clin. Nutr. 2010, 64, 720–726. [Google Scholar] [CrossRef]
- Kim, Y.-W.; Kim, K.-H.; Choi, H.-J.; Lee, D.-S. Anti-diabetic activity of β-glucans and their enzymatically hydrolyzed oligosaccharides from Agaricus blazei. Biotechnol. Lett. 2005, 27, 483–487. [Google Scholar] [CrossRef]
- Ishii, P.L.; Prado, C.K.; Mauro, M.d.O.; Carreira, C.M.; Mantovani, M.S.; Ribeiro, L.R.; Dichi, J.B.; Oliveira, R.J. Evaluation of Agaricus blazei in vivo for antigenotoxic, anticarcinogenic, phagocytic and immunomodulatory activities. Regul. Toxicol. Pharmacol. 2011, 59, 412–422. [Google Scholar] [CrossRef]
- Rony, K.; Mathew, J.; Neenu, P.; Janardhanan, K. Ganoderma lucidum (Fr.) P. Karst occurring in South India attenuates gastric ulceration in rats. Indian J. Nat. Prod. Resour. 2011, 2, 19–27. [Google Scholar]
- Reis, F.S.; Lima, R.T.; Morales, P.; Ferreira, I.C.; Vasconcelos, M.H. Methanolic extract of Ganoderma lucidum induces autophagy of AGS human gastric tumor cells. Molecules 2015, 20, 17872–17882. [Google Scholar] [CrossRef]
- Li, F.; Wen, H.; Zhang, Y.; Aa, M.; Liu, X. Purification and characterization of a novel immunomodulatory protein from the medicinal mushroom Trametes versicolor. Sci. China Life Sci. 2011, 54, 379–385. [Google Scholar] [CrossRef]
- She, Q.-B.; Ng, T.-B.; Liu, W.-K. A Novel Lectin with Potent Immunomodulatory Activity Isolated from Both Fruiting Bodies and Cultured Mycelia of the Edible Mushroom Volvariella volvacea. Biochem. Biophys. Res. Commun. 1998, 247, 106–111. [Google Scholar] [CrossRef]
- Sze, S.; Ho, J.; Liu, W. Volvariella volvacea lectin activates mouse T lymphocytes by a calcium dependent pathway. J. Cell. Biochem. 2004, 92, 1193–1202. [Google Scholar] [CrossRef]
- Zhang, G.; Sun, J.; Wang, H.; Ng, T. A novel lectin with antiproliferative activity from the medicinal mushroom Pholiota adiposa. Acta Biochim. Pol. 2009, 56, 415–421. [Google Scholar] [CrossRef]
- Hyun, K.W.; Jeong, S.C.; Lee, D.H.; Park, J.S.; Lee, J.S. Isolation and characterization of a novel platelet aggregation inhibitory peptide from the medicinal mushroom, Inonotus obliquus. Peptides 2006, 27, 1173–1178. [Google Scholar] [CrossRef]
- Cui, L.; Dong, M.S.; Chen, X.H.; Jiang, M.; Lv, X.; Yan, G. A novel fibrinolytic enzyme from Cordyceps militaris, a Chinese traditional medicinal mushroom. World J. Microbiol. Biotechnol. 2008, 24, 483–489. [Google Scholar] [CrossRef]
- Zhou, X.; Meyer, C.U.; Schmidtke, P.; Zepp, F. Effect of cordycepin on interleukin-10 production of human peripheral blood mononuclear cells. Eur. J. Pharmacol. 2002, 453, 309–317. [Google Scholar] [CrossRef]
- Shaw, L.; Rea, K.; Lachowsky, N.J.; Roth, E.A. Magic Mushroom Use: A Qualitative Interview Study of Post-Trip Impacts and Strategies for Optimizing Experiences. J. Psychoact. Drugs 2022, 1–8. [Google Scholar] [CrossRef]
- Stierle, A.; Strobel, G.; Stierle, D. Taxol and Taxane Production by Taxomyces andreanae, an Endophytic Fungus of Pacific Yew. Science 1993, 260, 214–216. [Google Scholar] [CrossRef]
- Rahi, D.K.; Rahi, S.; Pandey, A.; Rajak, R. Enzymes from mushrooms and their industrial application. In Advances in Fungal Biotechnology; I. K. International: New Delhi, India, 2009; pp. 136–184. [Google Scholar] [CrossRef]
- Egli, S. Mycorrhizal mushroom diversity and productivity—An indicator of forest health? Ann. For. Sci. 2011, 68, 81–88. [Google Scholar] [CrossRef]
- Le Tacon, F.; Rubini, A.; Murat, C.; Riccioni, C.; Robin, C.; Belfiori, B.; Zeller, B.; De la Varga, H.; Akroume, E.; Deveau, A.; et al. Certainties and uncertainties about the life cycle of the Périgord black truffle (Tuber melanosporum Vittad.). Ann. For. Sci. 2016, 73, 105–117. [Google Scholar] [CrossRef]
- Yang, X.; Luedeling, E.; Chen, G.; Hyde, K.D.; Yang, Y.; Zhou, D.; Xu, J.; Yang, Y. Climate change effects fruiting of the prize matsutake mushroom in China. Fungal Divers. 2012, 56, 189–198. [Google Scholar] [CrossRef]
- Stamets, P. Growing Gourmet and Medicinal Mushrooms; Ten Speed Press: Berkeley, CA, USA, 2011. [Google Scholar]
- Strauss, D.; Ghosh, S.; Murray, Z.; Gryzenhout, M. An Overview on the Taxonomy, Phylogenetics and Ecology of the Psychedelic Genera Psilocybe, Panaeolus, Pluteus and Gymnopilus. Front. For. Glob. Chang. 2022, 5, 813998. [Google Scholar] [CrossRef]
- Lenz, C.; Wick, J.; Braga, D.; García-Altares, M.; Lackner, G.; Hertweck, C.; Gressler, M.; Hoffmeister, D. Injury-Triggered Blueing Reactions of Psilocybe “Magic” Mushrooms. Angew. Chem. Int. Ed. 2020, 59, 1450–1454. [Google Scholar] [CrossRef]
- Melgarejo-Estrada, E.; Suarez, M.E.; Rocabado, D.; Maillard, O.; Lechner, B.E. Checklist of Bolivian Agaricales: 1. Species with dark and pink spore prints. Mycotaxon 2020, 134, 739. [Google Scholar] [CrossRef]
- Gerhardt, E. Taxonomische Revision der Gattungen Panaeolus and-Panaeolina (Fungi, Agaricales, Coprinaceae); Schweizerbart and Borntraeger Science Publishers: Stuttgart, Germany, 1996. [Google Scholar]
- He, M.-Q.; Zhao, R.-L.; Hyde, K.D.; Begerow, D.; Kemler, M.; Yurkov, A.; McKenzie, E.H.; Raspe, O.; Kakishima, M.; Sanchez-Ramirez, S. Notes, outline and divergence times of Basidiomycota. Fungal Divers. 2019, 99, 105–367. [Google Scholar]
- Ediriweera, S.; Wijesundera, R.; Nanayakkara, C.; Weerasena, J. First report of Panaeolus sphinctrinus and Panaeolus foenisecii (Psathyrellaceae, Agaricales) on elephant dung from Sri Lanka. Front. Environ. Microbiol. 2015, 1, 19–23. [Google Scholar] [CrossRef]
- Amandeep, K.; Atri, N.; Munruchi, K. Two new coprophilous varieties of Panaeolus (Psathyrellaceae, Agaricales) from Punjab, India. Mycosphere 2013, 4, 616–625. [Google Scholar] [CrossRef]
- Amandeep, K.; Atri, N.; Munruchi, K. Two new species of Panaeolus (Psathyrellaceae, Agaricales) from coprophilous habitats of Punjab, India. J. New Biol. Rep. 2014, 3, 125–132. [Google Scholar]
- Nkadimeng, S.M.; Steinmann, C.M.L.; Eloff, J.N. Effects and safety of Psilocybe cubensis and Panaeolus cyanescens magic mushroom extracts on endothelin-1-induced hypertrophy and cell injury in cardiomyocytes. Sci. Rep. 2020, 10, 22314. [Google Scholar] [CrossRef]
- Iliffe, R. Getting to grips with pluteus. Field Mycol. 2010, 11, 78–92. [Google Scholar] [CrossRef]
- Hosen, M.I.; Liang, X.; Xu, J.; Li, T.H. Pluteus squarrosus sp. nov.(Pluteus sect. Celluloderma, Pluteaceae) from northeast China. Nord. J. Bot. 2019, 37. [Google Scholar] [CrossRef]
- Stijve, T.; Bonnard, J. Psilocybine et urée dans le genre Pluteus. Mycol. Helv. 1986, 2, 123–130. [Google Scholar]
- Justo, A.; Vizzini, A.; Minnis, A.M.; Menolli, N.; Capelari, M.; Rodríguez, O.; Malysheva, E.; Contu, M.; Ghignone, S.; Hibbett, D.S. Phylogeny of the Pluteaceae (Agaricales, Basidiomycota): Taxonomy and character evolution. Fungal Biol. 2011, 115, 1–20. [Google Scholar] [CrossRef]
- Gartz, J. Magic Mushrooms Around the World: A Scientific Journey Across Cultures and Time; LIS Publications: Los Angeles, CA, USA, 1996. [Google Scholar]
- Stamets, P. Psilocybin Mushrooms of the World; Ten Speed Press: Berkeley, CA, USA, 1996. [Google Scholar]
- Holec, J. The Genus Gymnopilus (Fungi, Agaricales) in the Czech Republic with Respect to Collections from Other European Countries, Acta Musei Natl. Pragae 2005, 61, 1–52. [Google Scholar]
- Mau, J.-L.; Lin, H.-C.; Chen, C.-C. Non-volatile components of several medicinal mushrooms. Food Res. Int. 2001, 34, 521–526. [Google Scholar] [CrossRef]
- Friedman, M. Mushroom polysaccharides: Chemistry and antiobesity, antidiabetes, anticancer, and antibiotic properties in cells, rodents, and humans. Foods 2016, 5, 80. [Google Scholar] [CrossRef]
- Muszyńska, B.; Grzywacz-Kisielewska, A.; Kała, K.; Gdula-Argasińska, J. Anti-inflammatory properties of edible mushrooms: A review. Food Chem. 2018, 243, 373–381. [Google Scholar] [CrossRef]
- Ganesan, K.; Xu, B. Anti-obesity effects of medicinal and edible mushrooms. Molecules 2018, 23, 2880. [Google Scholar] [CrossRef]
- Palacios, I.; Lozano, M.; Moro, C.; D’Arrigo, M.; Rostagno, M.A.; Martínez, J.A.; García-Lafuente, A.; Guillamón, E.; Villares, A. Antioxidant properties of phenolic compounds occurring in edible mushrooms. Food Chem. 2011, 128, 674–678. [Google Scholar] [CrossRef]
- Nowacka, N.; Nowak, R.; Drozd, M.; Olech, M.; Los, R.; Malm, A. Analysis of phenolic constituents, antiradical and antimicrobial activity of edible mushrooms growing wild in Poland. LWT-Food Sci. Technol. 2014, 59, 689–694. [Google Scholar] [CrossRef]
- Zhao, S.; Gao, Q.; Rong, C.; Wang, S.; Zhao, Z.; Liu, Y.; Xu, J. Immunomodulatory Effects of Edible and Medicinal Mushrooms and Their Bioactive Immunoregulatory Products. J. Fungi 2020, 6, 269. [Google Scholar] [CrossRef]
- Johnson, M.W.; Griffiths, R.R. Potential Therapeutic Effects of Psilocybin. Neurotherapeutics 2017, 14, 734–740. [Google Scholar] [CrossRef]
- Fricke, J.; Blei, F.; Hoffmeister, D. Enzymatic Synthesis of Psilocybin. Angew. Chem. Int. Ed. 2017, 56, 12352–12355. [Google Scholar] [CrossRef]
- Kalberer, F.; Kreis, W.; Rutschmann, J. The fate of psilocin in the rat. Biochem. Pharmacol. 1962, 11, 261–269. [Google Scholar] [CrossRef]
- Wolbach, A.; Miner, E.; Isbell, H. Comparison of psilocin with psilocybin, mescaline and LSD-25. Psychopharmacologia 1962, 3, 219–223. [Google Scholar] [CrossRef]
- Tylš, F.; Páleníček, T.; Horáček, J. Psilocybin–summary of knowledge and new perspectives. Eur. Neuropsychopharmacol. 2014, 24, 342–356. [Google Scholar] [CrossRef]
- Carhart-Harris, R.L.; Erritzoe, D.; Williams, T.; Stone, J.M.; Reed, L.J.; Colasanti, A.; Tyacke, R.J.; Leech, R.; Malizia, A.L.; Murphy, K. Neural correlates of the psychedelic state as determined by fMRI studies with psilocybin. Proc. Natl. Acad. Sci. USA 2012, 109, 2138–2143. [Google Scholar] [CrossRef]
- Winter, J.; Rice, K.; Amorosi, D.; Rabin, R. Psilocybin-induced stimulus control in the rat. Pharmacol. Biochem. Behav. 2007, 87, 472–480. [Google Scholar] [CrossRef]
- Fantegrossi, W.; Woods, J.; Winger, G. Transient reinforcing effects of phenylisopropylamine and indolealkylamine hallucinogens in rhesus monkeys. Behav. Pharmacol. 2004, 15, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, R.R.; Johnson, M.W.; Carducci, M.A.; Umbricht, A.; Richards, W.A.; Richards, B.D.; Cosimano, M.P.; Klinedinst, M.A. Psilocybin produces substantial and sustained decreases in depression and anxiety in patients with life-threatening cancer: A randomized double-blind trial. J. Psychopharmacol. 2016, 30, 1181–1197. [Google Scholar] [CrossRef] [PubMed]
- Carhart-Harris, R.L.; Bolstridge, M.; Rucker, J.; Day, C.M.; Erritzoe, D.; Kaelen, M.; Bloomfield, M.; Rickard, J.A.; Forbes, B.; Feilding, A. Psilocybin with psychological support for treatment-resistant depression: An open-label feasibility study. Lancet Psychiatry 2016, 3, 619–627. [Google Scholar] [CrossRef] [PubMed]
- Moreno, F.A.; Wiegand, C.B.; Taitano, E.K.; Delgado, P.L. Safety, tolerability, and efficacy of psilocybin in 9 patients with obsessive-compulsive disorder. J. Clin. Psychiatry 2006, 67, 1735–1740. [Google Scholar] [CrossRef] [PubMed]
- Schindler, E.A.; Gottschalk, C.H.; Weil, M.J.; Shapiro, R.E.; Wright, D.A.; Sewell, R.A. Indoleamine hallucinogens in cluster headache: Results of the clusterbusters medication use survey. J. Psychoact. Drugs 2015, 47, 372–381. [Google Scholar] [CrossRef]
- Nichols, D.E.; Frescas, S. Improvements to the synthesis of psilocybin and a facile method for preparing the O-acetyl prodrug of psilocin. Synthesis 1999, 1999, 935–938. [Google Scholar] [CrossRef]
- Nichols, D.E. Psilocybin: From ancient magic to modern medicine. J. Antibiot. 2020, 73, 679–686. [Google Scholar] [CrossRef]
- Fricke, J.; Lenz, C.; Wick, J.; Blei, F.; Hoffmeister, D. Production Options for Psilocybin: Making of the Magic. Chem. Eur. J. 2019, 25, 897–903. [Google Scholar] [CrossRef]
- Sánchez, C. Cultivation of Pleurotus ostreatus and other edible mushrooms. Appl. Microbiol. Biotechnol. 2010, 85, 1321–1337. [Google Scholar] [CrossRef]
- Shah, Z.; Ashraf, M.; Ishtiaq, M. Comparative study on cultivation and yield performance of oyster mushroom (Pleurotus ostreatus) on different substrates (wheat straw, leaves, saw dust). Pak. J. Nutr. 2004, 3, 158–160. [Google Scholar]
- Zhang, R.; Li, X.; Fadel, J. Oyster mushroom cultivation with rice and wheat straw. Bioresour. Technol. 2002, 82, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Oei, P.; Nieuwenhuijzen, B.v. Small-Scale Mushroom Cultivation; Agromisa/CTA: Wageningen, The Netherlands, 2005. [Google Scholar]
- Higgins, C.; Margot, H.; Warnquist, S.; Obeysekare, E.; Mehta, K. Mushroom cultivation in the developing world: A comparison of cultivation technologies. In Proceedings of the 2017 IEEE Global Humanitarian Technology Conference (GHTC), Hilton San Jose, CA, USA, 19–22 October 2017; pp. 1–7. [Google Scholar]
- Smith, J.E.; Rowan, N.J.; Sullivan, R. Medicinal mushrooms: A rapidly developing area of biotechnology for cancer therapy and other bioactivities. Biotechnol. Lett. 2002, 24, 1839–1845. [Google Scholar] [CrossRef]
- Chen, A.W. Natural log cultivation of the medicinal mushroom, Ganoderma lucidum (Reishi). Mushroom Grow. Newsl. 2002, 3, 2–6. [Google Scholar]
- Hur, H. Cultural Characteristics and Log-Mediated Cultivation of the Medicinal Mushroom, Phellinus linteus. Mycobiology 2008, 36, 81–87. [Google Scholar] [CrossRef]
- Wendiro, D.; Wacoo, A.P.; Wise, G. Identifying indigenous practices for cultivation of wild saprophytic mushrooms: Responding to the need for sustainable utilization of natural resources. J. Ethnobiol. Ethnomed. 2019, 15, 64. [Google Scholar] [CrossRef]
- O’Brien, B.J.; Milligan, E.; Carver, J.; Roy, E.D. Integrating anaerobic co-digestion of dairy manure and food waste with cultivation of edible mushrooms for nutrient recovery. Bioresour. Technol. 2019, 285, 121312. [Google Scholar] [CrossRef]
- Mleczek, M.; Gąsecka, M.; Budka, A.; Niedzielski, P.; Siwulski, M.; Kalač, P.; Mleczek, P.; Rzymski, P. Changes in mineral composition of six strains of Pleurotus after substrate modifications with different share of nitrogen forms. Eur. Food Res. Technol. 2021, 247, 245–257. [Google Scholar] [CrossRef]
- Chang, S.-T.; Miles, P.G. Mushrooms: Cultivation, Nutritional Value, Medicinal Effect, and Environmental Impact; CRC Press: Boca Raton, FL, USA, 2004. [Google Scholar]
- Anyakorah, C.; Okafor, N.; Olatunji, O. Carbon And Nitrogen Requirements for the Cultivation of Oyster Mushroom Pleurotus sajor-caju. Niger. Food J. 2004, 22, 127–132. [Google Scholar] [CrossRef]
- Manu-Tawiah, W.; Martin, A. Nitrogen sources and the growth response of Pleurotus ostreatus mushroom mycelium. Can. Inst. Food Sci. Technol. J. 1988, 21, 194–199. [Google Scholar] [CrossRef]
- Gartz, J.; Allen, J.W.; Merlin, M.D. Ethnomycology, biochemistry, and cultivation of Psilocybe samuiensis Guzmán, Bandala and Allen, a new psychoactive fungus from Koh Samui, Thailand. J. Ethnopharmacol. 1994, 43, 73–80. [Google Scholar] [CrossRef]
- Gartz, J. Cultivation and analysis of Psilocybe species and an investigation of Galerina steglichi. Ann. Mus. Civ. Rovereto 1995, 10, 297–306. [Google Scholar]
- Gambaro, V.; Roda, G.; Visconti, G.L.; Arnoldi, S.; Casagni, E.; Dell’Acqua, L.; Farè, F.; Paladino, E.; Rusconi, C.; Arioli, S.; et al. DNA-based taxonomic identification of basidiospores in hallucinogenic mushrooms cultivated in “grow-kits” seized by the police: LC-UV quali-quantitative determination of psilocybin and psilocin. J. Pharm. Biomed. Anal. 2016, 125, 427–432. [Google Scholar] [CrossRef] [PubMed]
- Allen, J.W.; Gartz, J.; Molter, D. The Occurrence, Cultivation, and Chemistry of Psilocybe ovoideocystidiata, a new Bluing Species (Agaricales) from Ohio, Pennsylvania and West Virginia. Ethnomycol. J. Sacred Mushroom Stud. 2009, 8, 70–81. [Google Scholar]
- Rafati, H.; Riahi, H.; Mohammadi, A. Enhancement of Indole Alkaloids Produced by Psilocybe cubensis (Earle) Singer (Agaricomycetideae) in Controlled Harvesting Light Conditions. Int. J. Med. Mushrooms 2009, 11, 419–426. [Google Scholar] [CrossRef]
- Bakratsas, G.; Polydera, A.; Katapodis, P.; Stamatis, H. Recent trends in submerged cultivation of mushrooms and their application as a source of nutraceuticals and food additives. Future Foods 2021, 4, 100086. [Google Scholar] [CrossRef]
- Leung, A.Y.; Smith, A.H.; Paul, A.G. Production of psilocybin in Psilocybe baeocystis saprophytic culture. J. Pharm. Sci. 1965, 54, 1576–1579. [Google Scholar] [CrossRef]
- Catalfomo, P. The production of Psilocybin in Submerged Culture by Psilocybe cubensis; University of Washington: Seattle, WA, USA, 1963. [Google Scholar]
- Adams, A.M.; Kaplan, N.A.; Wei, Z.; Brinton, J.D.; Monnier, C.S.; Enacopol, A.L.; Ramelot, T.A.; Jones, J.A. In vivo production of psilocybin in E. coli. Metab. Eng. 2019, 56, 111–119. [Google Scholar] [CrossRef]
- Milne, N.; Thomsen, P.; Mølgaard Knudsen, N.; Rubaszka, P.; Kristensen, M.; Borodina, I. Metabolic engineering of Saccharomyces cerevisiae for the de novo production of psilocybin and related tryptamine derivatives. Metab. Eng. 2020, 60, 25–36. [Google Scholar] [CrossRef]
- Kargbo, R.B.; Sherwood, A.; Walker, A.; Cozzi, N.V.; Dagger, R.E.; Sable, J.; O’Hern, K.; Kaylo, K.; Patterson, T.; Tarpley, G.; et al. Direct Phosphorylation of Psilocin Enables Optimized cGMP Kilogram-Scale Manufacture of Psilocybin. ACS Omega 2020, 5, 16959–16966. [Google Scholar] [CrossRef]
- Khamsaw, P.; Sangta, J.; Chaiwan, P.; Rachtanapun, P.; Sirilun, S.; Sringarm, K.; Thanakkasaranee, S.; Sommano, S.R. Bio-Circular Perspective of Citrus Fruit Loss Caused by Pathogens: Occurrences, Active Ingredient Recovery and Applications. Horticulturae 2022, 8, 748. [Google Scholar] [CrossRef]
- Sangta, J.; Wongkaew, M.; Tangpao, T.; Withee, P.; Haituk, S.; Arjin, C.; Sringarm, K.; Hongsibsong, S.; Sutan, K.; Pusadee, T.; et al. Recovery of Polyphenolic Fraction from Arabica Coffee Pulp and Its Antifungal Applications. Plants 2021, 10, 1422. [Google Scholar] [CrossRef] [PubMed]
- Moshtaghian, H.; Bolton, K.; Rousta, K. Challenges for Upcycled Foods: Definition, Inclusion in the Food Waste Management Hierarchy and Public Acceptability. Foods 2021, 10, 2874. [Google Scholar] [CrossRef] [PubMed]
- Muhammad, B.L.; Suleiman, B. Global development of mushroom biotechnology. Int. J. Emerg. Trends Sci. Technol. 2015, 2, 2660–2669. [Google Scholar]
- Yildiz, S.; Yildiz, Ü.C.; Gezer, E.D.; Temiz, A. Some lignocellulosic wastes used as raw material in cultivation of the Pleurotus ostreatus culture mushroom. Process Biochem. 2002, 38, 301–306. [Google Scholar] [CrossRef]
- Öztürk, C.; Atila, F. Changes in lignocellulosic fractions of growing substrates during the cultivation of Hypsizygus ulmarius mushroom and its effects on mushroom productivity. Sci. Hortic. 2021, 288, 110403. [Google Scholar] [CrossRef]
- Xie, C.; Yan, L.; Gong, W.; Zhu, Z.; Tan, S.; Chen, D.; Hu, Z.; Peng, Y. Effects of Different Substrates on Lignocellulosic Enzyme Expression, Enzyme Activity, Substrate Utilization and Biological Efficiency of Pleurotus eryngii. Cell. Physiol. Biochem. 2016, 39, 1479–1494. [Google Scholar] [CrossRef]
- Isroi, I.; Millati, R.; Niklasson, C.; Cayanto, C.; Taherzadeh, M.J.; Lundquist, K. Biological treatment of Lignocelluloses with white-rot fungi and its applications. BioResources 2011, 6, 5224–5259. [Google Scholar] [CrossRef]
- Cohen, R.; Persky, L.; Hadar, Y. Biotechnological applications and potential of wood-degrading mushrooms of the genus Pleurotus. Appl. Microbiol. Biotechnol. 2002, 58, 582–594. [Google Scholar] [CrossRef]

| Mushrooms | Uses | Bioactive Constituents | References |
|---|---|---|---|
| White button mushroom (Agaricus bisporus), Almond mushroom (A. subrufescens), Caterpillar fungus (Cordyceps sinensis), Shaggy ink cap (Coprinus comatus), Lingzhi (Ganoderma lucidum), White rot fungus/Chaga (Inonotus obliquus), Phellinus linteus, Oyster mushroom (Pleurotus spp.), Poria cocos, and Sparassis crispa | Possessed hypoglycemic effects on reducing blood glucose levels and antidiabetic effects. | Dietary fiber along with the polyphenols, vitamin C, and ergothioneine, as well as proteins, and polysaccharides (β-Glucans and oligosaccharides). | [16,17,18,19] |
| Almond mushroom | Induced apoptosis of intestinal cancers. | Soluble fibers serve as a desirable food source for bacteria that generate short-chain fatty acids such as butyrate, which may be able to stimulate apoptosis of cancers in human intestinal. | [20] |
| Lingzhi | Effective for treatment of gastric cancer. | Treatment of a gastric cancer cell line with methanolic extract resulted in an increase in cellular autophagy and the production of autophagosomes (AGS). | [21,22] |
| Himematsutake (A. blazei) | Traditional medicine from Japan and used for treatments of diabetes, hyperlipidemia, arteriosclerosis, and chronic hepatitis. In animal study, it increased proliferation of monocyte and promoted destruction of cells with DNA alterations that correlate with the development of cancer. | The immune system is modulated by bioactive β-glucans in supplemental diets and is rendered more effective with regard to phagocytic activity. | [20] |
| Trametes versicolor | The medicinal mushroom frequently used in traditional Chinese medicine for its antiviral, antitumor, and immunomodulatory effects. | The polysaccharide Krestin (PSK) is commercially available for use in cancer immunotherapy. | [23] |
| Flammulina velutipes, Pholiota spp., Lingzhi and straw mushroom (Volvariella volvacea) | The immunomodulatory proteins had been isolated from the fruiting body and cultured mycelia of the mushrooms. | Lectins can bind to cell surface carbohydrates, agglutinate cells, and inhibit cancer cell growth. | [24,25,26] |
| Chaga (Inonotus obliquus) | A fungal parasite, grows on birch trees in colder northern climates with anticancer properties. | Betulin or betulinic acid isolate illustrates platelet adhesion and aggregation plays an important role in the pathogenesis of thrombosis, particularly arteriothrombosis. | [27] |
| Cordyceps spp. | The mushrooms are used in Traditional Chinese medicine due to their various therapeutic properties including immunoregulative, anticancer, antibacterial, and antifungal activities. | Cordycepin, ophicordin, polysaccharides, and L-tryptophan are bioactive constituents isolated from the Cordyceps such as C. militaris, also known as north Cordyceps. | [28,29] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sommano, S.R.; Suksathan, R.; Sombat, T.; Seehanam, P.; Sirilun, S.; Ruksiriwanich, W.; Wangtueai, S.; Leksawasdi, N. Novel Perspective of Medicinal Mushroom Cultivations: A Review Case for ‘Magic’ Mushrooms. Agronomy 2022, 12, 3185. https://doi.org/10.3390/agronomy12123185
Sommano SR, Suksathan R, Sombat T, Seehanam P, Sirilun S, Ruksiriwanich W, Wangtueai S, Leksawasdi N. Novel Perspective of Medicinal Mushroom Cultivations: A Review Case for ‘Magic’ Mushrooms. Agronomy. 2022; 12(12):3185. https://doi.org/10.3390/agronomy12123185
Chicago/Turabian StyleSommano, Sarana Rose, Ratchuporn Suksathan, Thanarat Sombat, Pimjai Seehanam, Sasithorn Sirilun, Warintorn Ruksiriwanich, Sutee Wangtueai, and Noppol Leksawasdi. 2022. "Novel Perspective of Medicinal Mushroom Cultivations: A Review Case for ‘Magic’ Mushrooms" Agronomy 12, no. 12: 3185. https://doi.org/10.3390/agronomy12123185
APA StyleSommano, S. R., Suksathan, R., Sombat, T., Seehanam, P., Sirilun, S., Ruksiriwanich, W., Wangtueai, S., & Leksawasdi, N. (2022). Novel Perspective of Medicinal Mushroom Cultivations: A Review Case for ‘Magic’ Mushrooms. Agronomy, 12(12), 3185. https://doi.org/10.3390/agronomy12123185












