Effect of Chitosan Nanoparticles (CS-NPs) on In Vitro Regeneration Response and Production of Potato virus Y (PVY)-Free Plants of Potato
Abstract
1. Introduction
2. Materials and Methods
2.1. Selection of the PVY-Infected Plants
2.2. PVY Indexing by DAS-ELISA
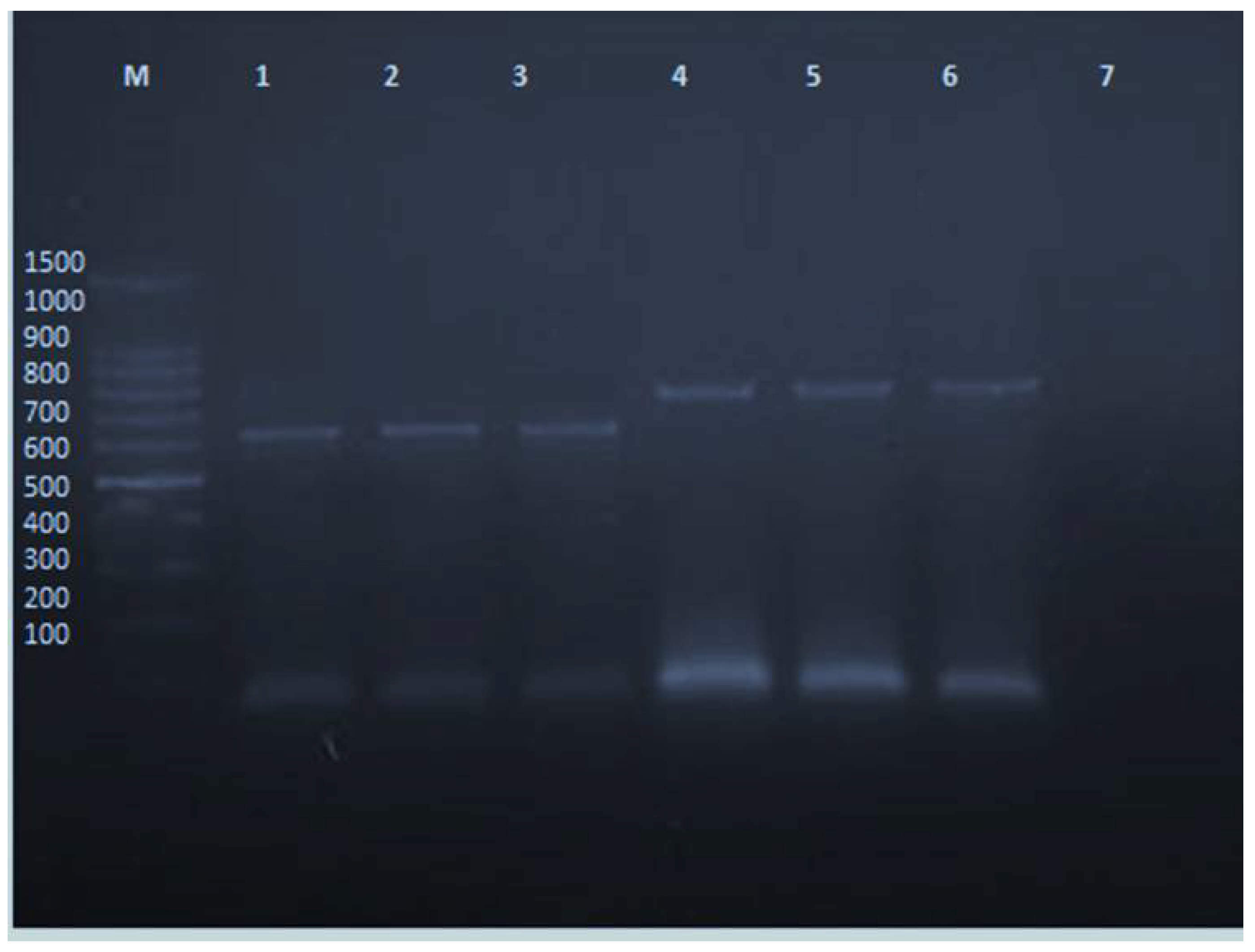
2.3. PVY Indexing by RT-PCR
2.4. Chitosan (CS-NPs) Preparation
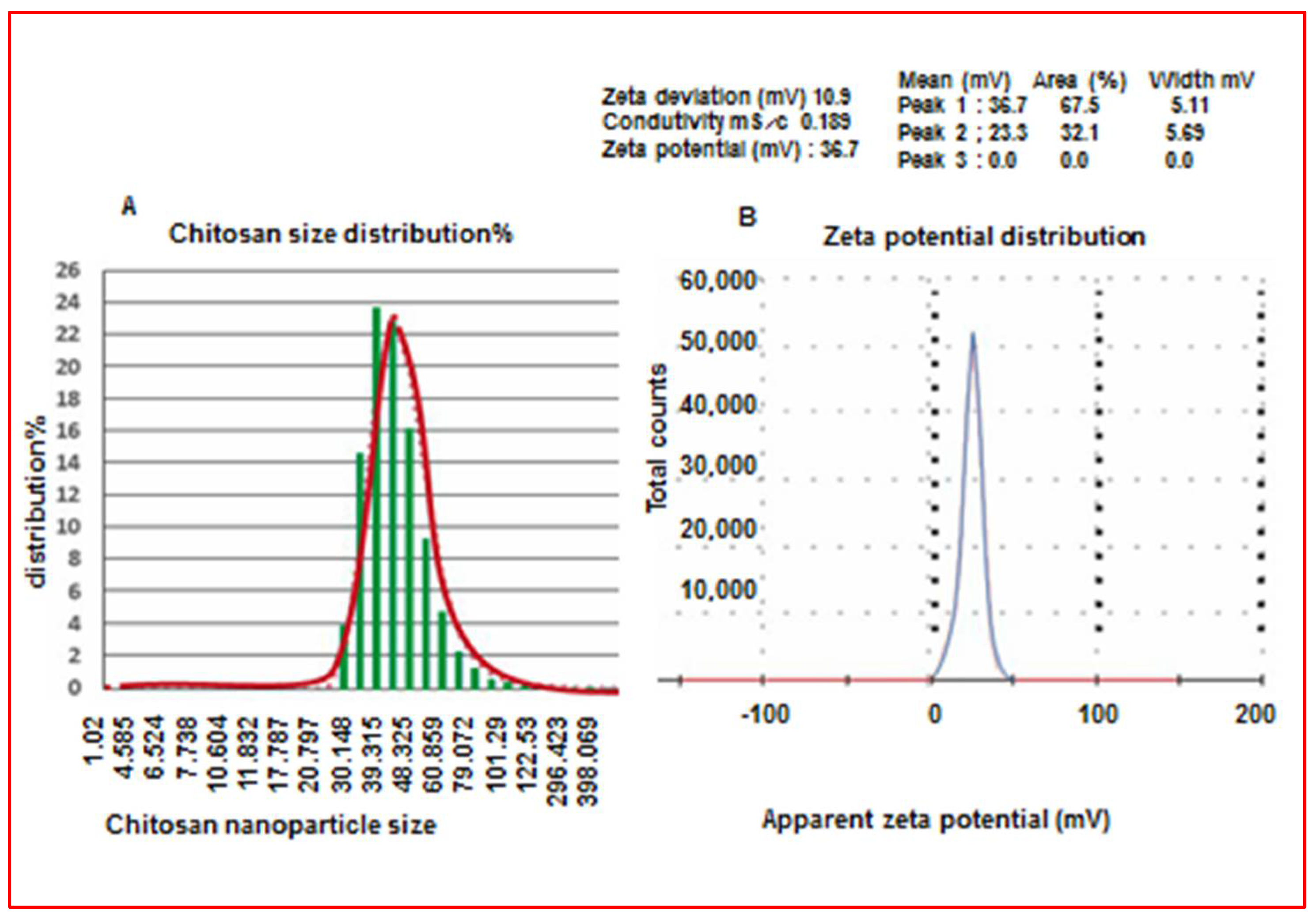
2.4.1. Characterization of Chitosan Nanoparticles
2.4.2. Determination of Nanoparticle Size
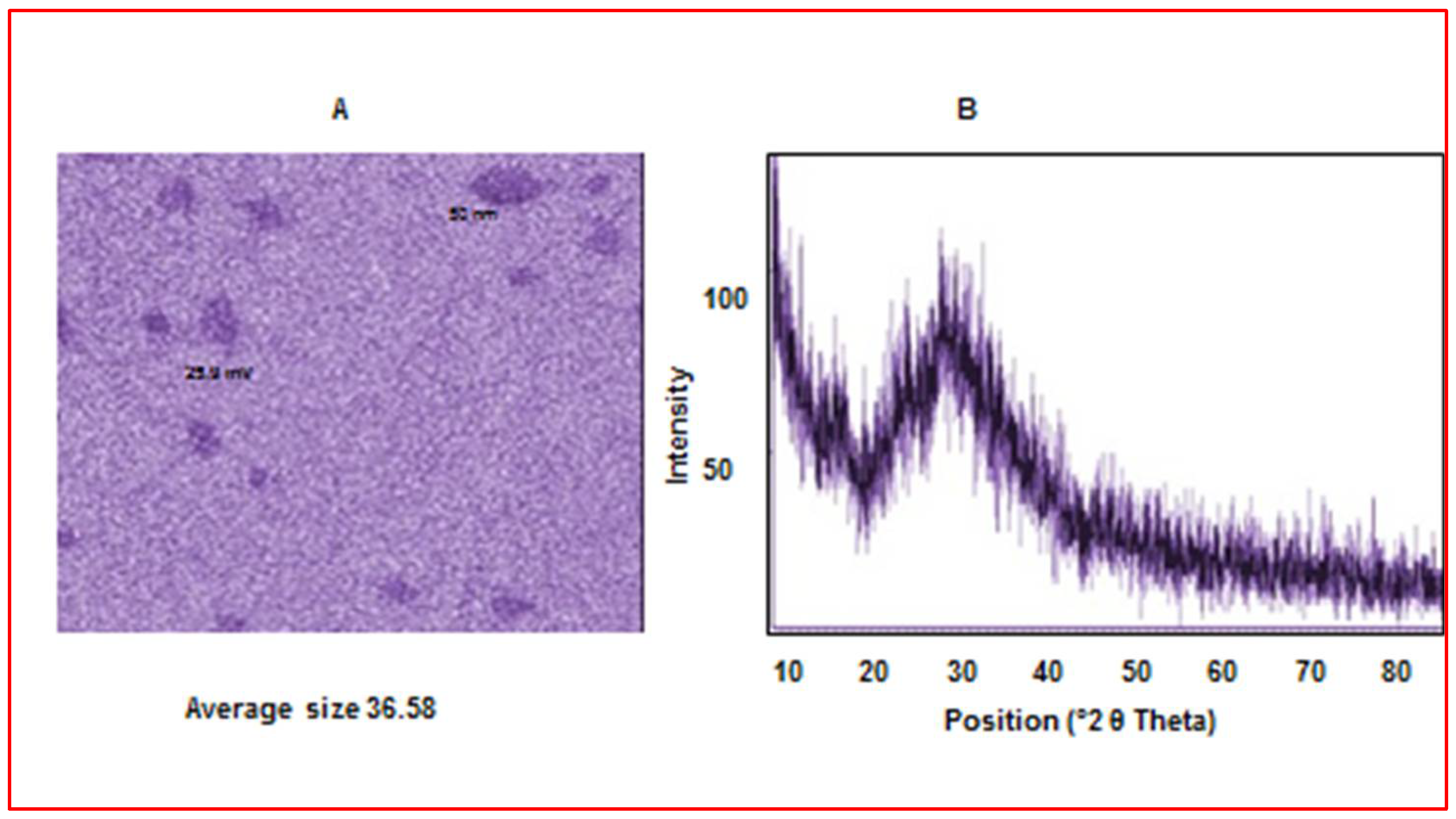
2.4.3. X-ray Diffraction (XRD) Measurement
2.4.4. The Morphology of the Prepared Nanoparticles
2.5. In Vitro Initiation and Stock Cultures
2.6. CS-NPs Treatments for PVY Eradication
2.7. In Vitro Rooting Induction, Hardening and Acclimation
2.8. PVY Detection after CS-NPs Treatment
2.9. Statistical Analysis
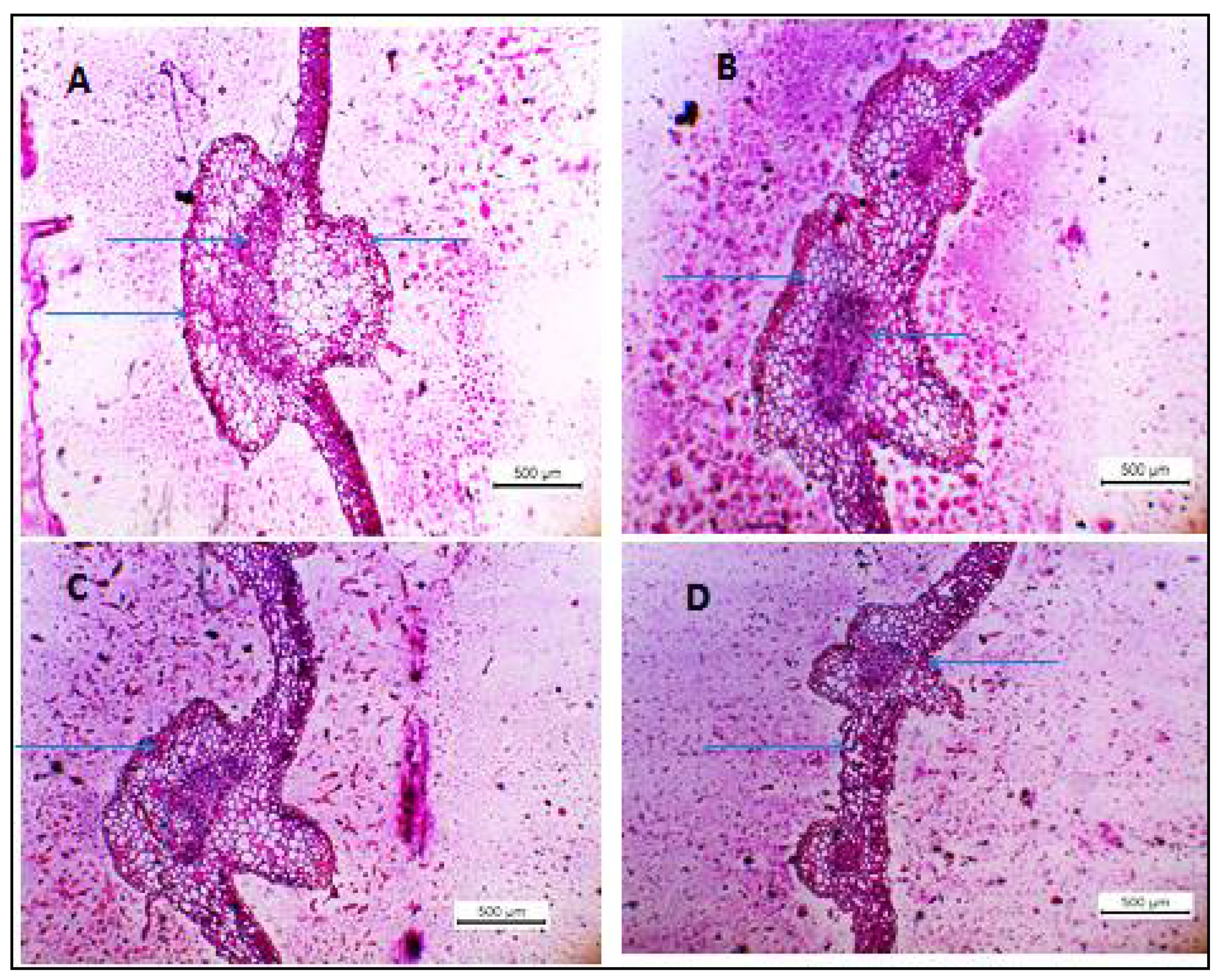
2.10. Histopathological Studies
3. Results
3.1. Effect of Chitosan (CS-NPs) Antiviral on Regeneration Response and Virus Elimination
Characterization of Chitosan Nanoparticles (CS-NPs)
3.2. Histopathological Changes
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Spooner, M.; Mclean, K.; Ramsay, G.; Waugh, R.; Bryan, J. A single Domestication for Potato Based on Multilocus Amplified Fragment Length Polymorphism Genotyping. Proc. Natl. Acad. Sci. USA 2011, 41, 14694–14699. [Google Scholar] [CrossRef] [PubMed]
- Hawkes, G.; Javier, F. The Potato in Spain During the late 16th century. Econ. Bot. 1992, 46, 86–97. [Google Scholar] [CrossRef]
- Russ, A.M. Causes of Flowering of Long-day Potato Species Under Short-day and Cold-night Conditions. J. Plant Physiol. 2002, 49, 465–469. [Google Scholar]
- Myers, S.S.; Zanobetti, A.; Kloog, I.; Huybers, P.; Leakey, A.D.; Bloom, A.J.; Carlisle, E.; Dietterich, L.H.; Fitzgerald, G.; Hasegawa, T.; et al. Increasing CO2 Threatens Human Nutrition. Nat. Hasegawa 2014, 510, 139–142. [Google Scholar] [CrossRef] [PubMed]
- Central Agency for Public Mobilization and Statistics. Estimates by Governorate 1/1/2018. Available online: www.capmas.gov.eg (accessed on 2 November 2018).
- Food and Agriculture Organization. Crop Water Information; Food and Agriculture Organization of the United Nations. Available online: https://www.fao.org/land-water/databases-and-software/crop-information/en/ (accessed on 7 November 2021).
- Saberi Riseh, R.; Gholizadeh Vazvani, M.; Ebrahimi-Zarandi, M.; Skorik, Y.A. Alginate-Induced Disease Resistance in Plants. Polymers 2022, 14, 661. [Google Scholar] [CrossRef]
- Tamisier, L.; Szadkowski, M.; Girardot, G.; Djian-Caporalino, C.; Palloix, A.; Hirsch, J. Concurrent Evolution of Resistance and Tolerance to Potato virus Y in Capsicum annuum Revealed by genome-wide association. Mol. Plant Pathol. 2022, 23, 254–264. [Google Scholar] [CrossRef]
- Sergio, P.; Piero, R.; Maurizio, C. Yield Losses in Virus-infected Crops. Arch. Phytopathol. Plant Prot. 1996, 30, 283–296. [Google Scholar]
- Kreuze, J.; Souza-Dias, J.; Jeevalatha, A.; Figueira, A.; Valkonen, J.; Jones, R. Viral Diseases in Potato. In The Potato Crop; Springer: Berlin, Germany, 2020; pp. 389–430. [Google Scholar]
- Abbas, M.F.; Hameed, S.; Rauf, A.; Nosheen, Q.; Ghani, A.; Qadir, A.; Zakia, S. Incidence of Six Viruses in Potato Growing Areas of Pakistan. Pak. J. Phytopathol. Zakia 2012, 24, 44–47. [Google Scholar]
- Liang, Z.; Dickison, V.; Singh, M.; Xiong, X.; Nie, X. Studies of Tomato Plants in Response to Infections with PVX and Different PVY Isolates Reveal a Remarkable PVX-PVYNTN Synergism and Diverse Expression Profiles of Genes Involved in Different Pathways. Eur. J. Plant Pathol. 2016, 144, 55–71. [Google Scholar] [CrossRef]
- Lacomme, C.; Pickup, J.; Fox, A.; Glais, L.; Dupuis, B.; Steinger, T.; Rolot, J.L.; Valkonen, J.; Kruger, K.; Nie, X.; et al. Transmission and Epidemiology of Potato virus Y; Springer International Publishing: New York, NY, USA, 2017; pp. 141–176. [Google Scholar]
- Dupuis, B.; Bragard, C.; Carnegie, S.; Kerr, J.; Glais, L.; Singh, M.; Lacomme, C. Potato virus Y: Control, Management and Seed Certification Programmers. In Potato virus Y: Biodiversity, Pathogenicity, Epidemiology and Management; Springer: Berlin, Germany, 2017; pp. 177–220. [Google Scholar]
- Harahagazwe, D.; Condori, B.; Barreda, C.; Bararyenya, A.; Byarugaba, A.A.; Kude, D.A.; Lung’aho, C.; Martinho, C.; Mbiri, D.; Nasona, B.; et al. How Big is the Potato (Solanum tuberosum L.) Yield Gap in Sub-Saharan Africa and why? A participatory approach. Open Agric. 2018, 3, 180–189. [Google Scholar] [CrossRef]
- Karasev, A.V.; Gray, S. Continuous and Emerging Challenges of Potato virus Y in potato. Annu. Rev. Phytopathol. 2013, 51, 571–586. [Google Scholar] [CrossRef]
- Kehoe, M.; Jones, R. Improving Potato virus Y Strain Nomenclature: Lessons from comparing isolates obtained over a 73-year period. Plant Pathol. 2016, 65, 322–333. [Google Scholar] [CrossRef]
- Fuentes, S.; Gibbs, A.; Adams, I.P.; Wilson, C.; Botermans, M.; Fox, A. Potato virus A isolates from Three Continents: Their bio-logical properties, phylogenetics, and prehistory. Am. Phytopathol. Soc. 2021, 111, 217–226. [Google Scholar] [CrossRef]
- Houben, S.; Braber, H.; Blom-Zandstra, M.; Anten, N. Current Potato Production in Algeria; Stichting Wageningen Research, Wageningen Plant Research, Business Unit Plant: Wageningen, The Netherlands, 2017; 51p. [Google Scholar]
- Gong, H.; Clement, I.; Leonce, D. Major In Vitro Techniques for Potato Virus Elimination and Post Eradication Detection Methods. A Review. Am. J. Potato Res. 2019, 96, 379–389. [Google Scholar] [CrossRef]
- Bettoni, J.C.; Mathew, L.; Pathirana, R.; Wiedow, C.; Hunter, D.A.; McLachlan, A.; Khan, S.; Tang, J.; Nadarajan, J. Eradication of Potato Virus S, Potato Virus A, and Potato Virus M From Infected in vitro-Grown Potato Shoots Using in vitro Therapies. Front. Plant Sci. 2022, 13, 1431. [Google Scholar] [CrossRef] [PubMed]
- Pandit, M.A.; Kumar, J.; Gulati, S.; Bhandari, N.; Mehta, P.; Katyal, R.; Rawat, C.D.; Mishra, V.; Kaur, J. Major Biological Control Strategies for Plant Pathogens. Pathogens 2022, 11, 273. [Google Scholar] [CrossRef]
- Saqib, S.; Faryad, S.; Afridi, M.I.; Arshad, B.; Younas, M.; Naeem, M.; Zaman, W.; Ullah, F.; Nisar, M.; Ali, S.; et al. Bimetallic Assembled Silver Nanoparticles Impregnated in Aspergillus fumigatus Extract Damage the Bacterial Membrane Surface and Release Cellular Contents. Coatings 2022, 12, 1505. [Google Scholar] [CrossRef]
- Hoang, N.H.; Thanh, T.; Sangpueak, R.; Treekoon, J.; Saengchan, C.; Thepbandit, W.; Papathoti, N.K.; Kamkaew, A.; Buensanteai, N. Chitosan Nanoparticles-Based Ionic Gelation Method: A Promising Candidate for Plant Disease Management. Polymers 2022, 14, 662. [Google Scholar] [CrossRef] [PubMed]
- Behl, T.; Kaur, I.; Sehgal, A.; Singh, S.; Sharma, N.; Bhatia, S.; Al-Harrasi, A.; Bungau, S. The Dichotomy of Nanotechnology as The Cutting Edge of Agriculture: Nano-farming as an asset versus nanotoxicity. Chemosphere 2022, 288, 132–533. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Li, Z. Nano-enabled Agriculture: How Do Nanoparticles Cross Barriers in Plants. Plant Commun. 2022, 3, 1100346. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Yada, R.Y. International Conference on Food and Agriculture Applications of Nanotechnologies, NanoAgri, São Pedro, SP, Brazil. Trends Food Sci. Technol. 2011, 22, 583–584. [Google Scholar] [CrossRef]
- Parisi, C.; Vigani, M.; Rodríguez-Cerezo, E. Agricultural Nanotechnologies: What Are The Current Possibilities. Nano Today 2015, 10, 124–127. [Google Scholar] [CrossRef]
- Su, S.; Kang, P.M. Systemic Review of Biodegradable Nanomaterials in Nanomedicine. Nanomaterials 2020, 10, 656. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.; Wang, C.; Wagner, D.C.; Jorge, L.; Torresdey, G.; He, F.; Cyren, M. Foliar Application of Nanoparticles: Mechanisms of Absorption, Transfer, and Multiple Impacts. Environ. Sci. Nano Rico 2021, 8, 1196–1210. [Google Scholar] [CrossRef]
- Hegazi, S.; Yousef, A.; Mohamed, A.; Allatif, A.; Shaban, T.; Kamal, M. Effect of Silver Nanoparticles, Medium Composition and Growth Regulators on in vitro propagation of Picual Olive Cultivar. Egypt. J. Chem. 2021, 64, 6961–6969. [Google Scholar]
- El-Kosary, S.; Allatif, A.; Stino, R.; Hassan, M.; Kinawy, A. Effect Of Silver Nanoparticles On Micropropagation Of Date Palm (Phoenix Dactylifera L, Cv. Sewi and Medjool). Plant Arch. 2020, 2, 9701. [Google Scholar]
- Badoni, A.; Chauhan, J. Potato Seed Production Of Cultivar Kufri Himalini, in vitro. Health Environ. Res. Online 2010, 1, 7–10. [Google Scholar]
- Singh, C.R. Review On Problems And its Remedy In Plant Tissue Culture. Asian J. Biol. Sci. 2018, 11, 165–172. [Google Scholar] [CrossRef]
- Yasmine, R.; Ahmad, J.; Qamar, S.; Qureshi, M.I. Engineered Nanomaterials for Sustainable Agricultural Production, Soil Improvement and Stress Management. In Plant Biology, Sustainability and Climate Change; Elsevier: Amsterdam, The Netherlands, 2023; pp. 495–512. [Google Scholar] [CrossRef]
- Clark, M.F.; Adams, A.N. Characteristics of The Microplate Method of Enzyme-Linked Immunosorbent Assay for The Detection of Plant Viruses. J. Gen. Virol. 1977, 34, 475–483. [Google Scholar] [CrossRef]
- Dominguez, S.; Aparicio, F.; Sánchez-Navarro, J.A.; Pallás, V.; Garcia-Brunton, J.; Cano, A. Studies on the Incidence of Ilarviruses and Apple Chlorotic Leafspot Trichovirus in Apricot Trees in the Murcia Region (Spain) using serological and molecular hybridization methods. Acta Hortic. 1997, 472, 203–210. [Google Scholar] [CrossRef]
- Shalaby, A.A.; Nakhla, M.K.; Soliman, A.M.; Mazyad, H.M.; Hadidi, A.; Maxwell, D.P. Development of a Highly Sensitive Multiplex Reverstrans-cription-Polymerase Chain Reaction (m-RT-PCR) Method for Detection of Three Potato Viruses in a Single Reaction and nested PCR. Arab J. Biotech. 2002, 5, 275–286. [Google Scholar]
- Sambrook, J.; Fritsch, E.F.; Maniatis, T. Molecular Cloning: A Laboratory Manual; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 1987; p. 1546. [Google Scholar]
- Murdock, R.C.; Braydich-Stolle, L.; Schrand, A.M.; Schlager, J.J.; Hussain, S.M. Characterization of Nanomaterial Dispersion in Solution Prior to In Vitro Exposure Using Dynamic Light Scattering Technique. Toxicol. Sci. 2008, 101, 239–253. [Google Scholar] [CrossRef] [PubMed]
- Phanjom, P.; Zoremi, E.; Mazumder, J.; Saha, M.; Baruah, S.B. Green Synthesis of Silver Nanoparticles Using Leaf Extract of Myrica esculenta. Int. J. NanoSci. Nanotechnol. 2012, 3, 73–79. [Google Scholar]
- Masarudin, M.J.; Cutts, S.M.; Evison, B.J.; Phillips, D.R.; Pigram, P.J. Factors Determining the Stability, Size Distribution, and Cellular Accumulation of Small, Monodisperse Chitosan Nanoparticlies as Candidate Vectors for Anticancer Drug Delivery: Application to the passive encapsulation of [14C]- doxorubicin. Nanotechnol. Sci. Appl. 2015, 8, 67–80. [Google Scholar] [CrossRef]
- Safavi, K. Effect of Titanium Dioxide Nanoparticles in Plant Tissue Culture Media for Enhance Resistance to Bacterial Activity. Bull. Environ. Pharmacol. Life Sci. 2014, 3, 163–166. [Google Scholar]
- Murashige, T.; Skoog, F. A Revised Medium for Rapid Growth and Bio Assays with Tobacco Tissue Cultures. Physiol. Plantarum. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Meyers, L.; Grossen, N.; Theory, P.; Design, W.H.; Freeman, S.F. Analysis of independent group designs. Am. Psychol. Assoc. 1974, 237–252. [Google Scholar]
- Ruzin, S.E. Staining Techniques. In Plant Microtechnique and Microscopy; Oxford University Press: Oxford, UK, 1999; pp. 87–116. [Google Scholar]
- Hansen, A.; Lane, W. Elimination of Apple Chlorotic Leafspot Virus from Apple Shoot Cultures by Ribavirin. Plant Dis. 1985, 69, 134–135. [Google Scholar]
- Sharma, S.; Rani, G.; Zaidi, A.A.; Hallan, V.; Nagpal, A.; Virk, G.S. Production of Indian Citrus Ringspot Virus-free Plants of Kinnow mandarin (Citrus nobilis Lour × C. deliciosa Tenora) employing chemotherapy coupled with shoot tip grafting. J. Plant Biotechnol. 2007, 8, 1–8. [Google Scholar]
- Simpkins, I.; Walkey, D.; Neely, H. Chemical Suppression of Virus in Cultured Plant Tissues. Ann. Appl. Biol. 1981, 9, 161–169. [Google Scholar] [CrossRef]
- Klein, R.; Livingston, C. Eradication of potato viruses X and S from potato shoot-tip cultures with ribavirin. Phytopathology 1983, 73, 1049–1050. [Google Scholar] [CrossRef]
- Wambugu, F.; Secor, G.; Gudmestad, N.J. Eradication of Potato virus Y and S from Potato by Chemotherapy of Cultured Axillary Bud Tips. Am. Potato Springer 1985, 62, 667–672. [Google Scholar] [CrossRef]
- Toussaint, A.; Kummert, J.; Maroquin, C.; Lebrun, A.; Roggemans, J. Tissue Culture o. Use of Virazole to Eradicate odontoglossum Ringspot Virus from In Vitro Cultures of Cymbidium Sw. In Plant Cell, Tissue and Organ Culture; Springer: Berlin, Germany, 1993; Volume 32, pp. 303–309. [Google Scholar]
- Ram, R.; Verma, N.; Singh, A.; Singh, L.; Hallan, V.; Zaidi, A. Indexing and Production of Virus-Rree Chrysanthemums. Biol. Plant Springer 2005, 49, 149–152. [Google Scholar] [CrossRef]
- Ahmed, G.; Mia, Z.; Mahmoud, A.; Tarek, S. Silver Nanoparticles as A Highly Viricidal Agent to Deter Plant-Infecting Viruses and Disrupt their Acquisition and Transmissibility by Vector Aphid. Res. Sq. 2021, 1–20. [Google Scholar]
- Fraser, R.; Gerwitz, A. Effects of 2-α-hydroxybenzylbenzimidazole on Tobacco Mosaic Virus and Host RNA Synthesis in Tobacco Leaf Discs and plants. Plant Sci. Lett. 1984, 34, 111–117. [Google Scholar] [CrossRef]
- Chirkov, S. The antiviral activity of chitosan. J. Appl. Biochem. Microbiol. 2002, 38, 1–8. [Google Scholar] [CrossRef]
- Abdelkhalek, A.; Qari, S.H.; Abu-Saied, M.A.A.-R.; Khalil, A.M.; Younes, H.A.; Nehela, Y.; Behiry, S.I. Chitosan Nanoparticles Inactivate Alfalfa Mosaic Virus Replication and Boost Innate Immunity in Nicotiana glutinosa Plants. Plants 2021, 10, 2701. [Google Scholar] [CrossRef]
- Davydova, V.N.; Nagorskaya, V.I.; Gorbach, A.A.; Kalitnik, A.V.; Reunov, T.F.; Solov’Eva, I.M. Applied Biochemistry Ermak, and Microbiology’Chitosan antiviral activity: Dependence on structure and depolymerization method. Appl. Biochem. 2011, 47, 103–108. [Google Scholar] [CrossRef]




| Concentrations of (CS-NPs) in Growth Medium mg L−1 | Proliferation | Percent Shoot Regeneration | Plant Length, cm ± SD | DAS-ELISA | RT-PCR |
|---|---|---|---|---|---|
| Untreated (Control) | 1.78 ± 0.30 * | 90.96 ± 1.42 | 3.99 ± 0.40 cm | 0.654 | + |
| 300 | 0.00 | 10.41 ± 1.20 c | - | 0.236 a | − |
| 250 | 6.11 ± 0.26 | 90.97 ± 1.41 a | 12.40 ± 0.38 * | 0.256 a | − |
| 200 | 4.69 ± 0.24 | 87.49 ± 1.22 a | 11.96 ± 0.18 * | 0.564 b | − |
| 100 | 3.69 ± 0.30 | 43.05 ± 1.85 b | 6.6 ± 0.01 * | 0.595 b | + |
| Treatment | Hair | Cuticle Thickness | Epidermal Thickness | Palisade Tissue | Collenchyma Tissue | Spongy Tissue | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Healthy control | 145.6 a * | 3 b | 1 c | 7.0 b | 5.5 c | 4.5 b | 9.7 c | 45.8 d | 1 f | 6 d | 110.2 f | 2 d | 58.8 f | 72.3 d | 4 f |
| Chitosan (CS-NPs) | 250.7 a | 6 b | 4 c | 9.0 b | 6.3 c | 17.4 b | 11.3 c | 76.5 d | 1 f | 6 d | 190.6 f | 3 d | 70.0 f | 95.2 d | 5 f |
| Treatment | Mid. Vein Thickness | Vasc. Bundle Length | Vasc. Bundle Width | Ex. Phlo. Thickness | Intr. Phlo. Thickness | Xylem Thickness | No. Xylem Vessel | Thickness of Major Xylem |
|---|---|---|---|---|---|---|---|---|
| Healthy control | 7745.2 | 215.2 | 460.6 | 35.3 | 39.4 | 120.4 | 12 | 35.2 |
| Chitosan (CS-NPs) | 980.6 | 225.6 | 560.4 | 55.5 | 48.3 | 132.6 | 20 | 45.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elsahhar, S.; Shahba, M.; Elsayed, T.; Mikhail, M.; Galal, A. Effect of Chitosan Nanoparticles (CS-NPs) on In Vitro Regeneration Response and Production of Potato virus Y (PVY)-Free Plants of Potato. Agronomy 2022, 12, 2901. https://doi.org/10.3390/agronomy12112901
Elsahhar S, Shahba M, Elsayed T, Mikhail M, Galal A. Effect of Chitosan Nanoparticles (CS-NPs) on In Vitro Regeneration Response and Production of Potato virus Y (PVY)-Free Plants of Potato. Agronomy. 2022; 12(11):2901. https://doi.org/10.3390/agronomy12112901
Chicago/Turabian StyleElsahhar, Sary, Mohamed Shahba, Tarek Elsayed, Maurice Mikhail, and Azza Galal. 2022. "Effect of Chitosan Nanoparticles (CS-NPs) on In Vitro Regeneration Response and Production of Potato virus Y (PVY)-Free Plants of Potato" Agronomy 12, no. 11: 2901. https://doi.org/10.3390/agronomy12112901
APA StyleElsahhar, S., Shahba, M., Elsayed, T., Mikhail, M., & Galal, A. (2022). Effect of Chitosan Nanoparticles (CS-NPs) on In Vitro Regeneration Response and Production of Potato virus Y (PVY)-Free Plants of Potato. Agronomy, 12(11), 2901. https://doi.org/10.3390/agronomy12112901


_Asaduzzaman.jpg)




