Effects of Different Deproteinization Methods on the Antioxidant Activity of Polysaccharides from Flos Sophorae Immaturus Obtained by Ultrasonic Microwave Synergistic Extraction
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
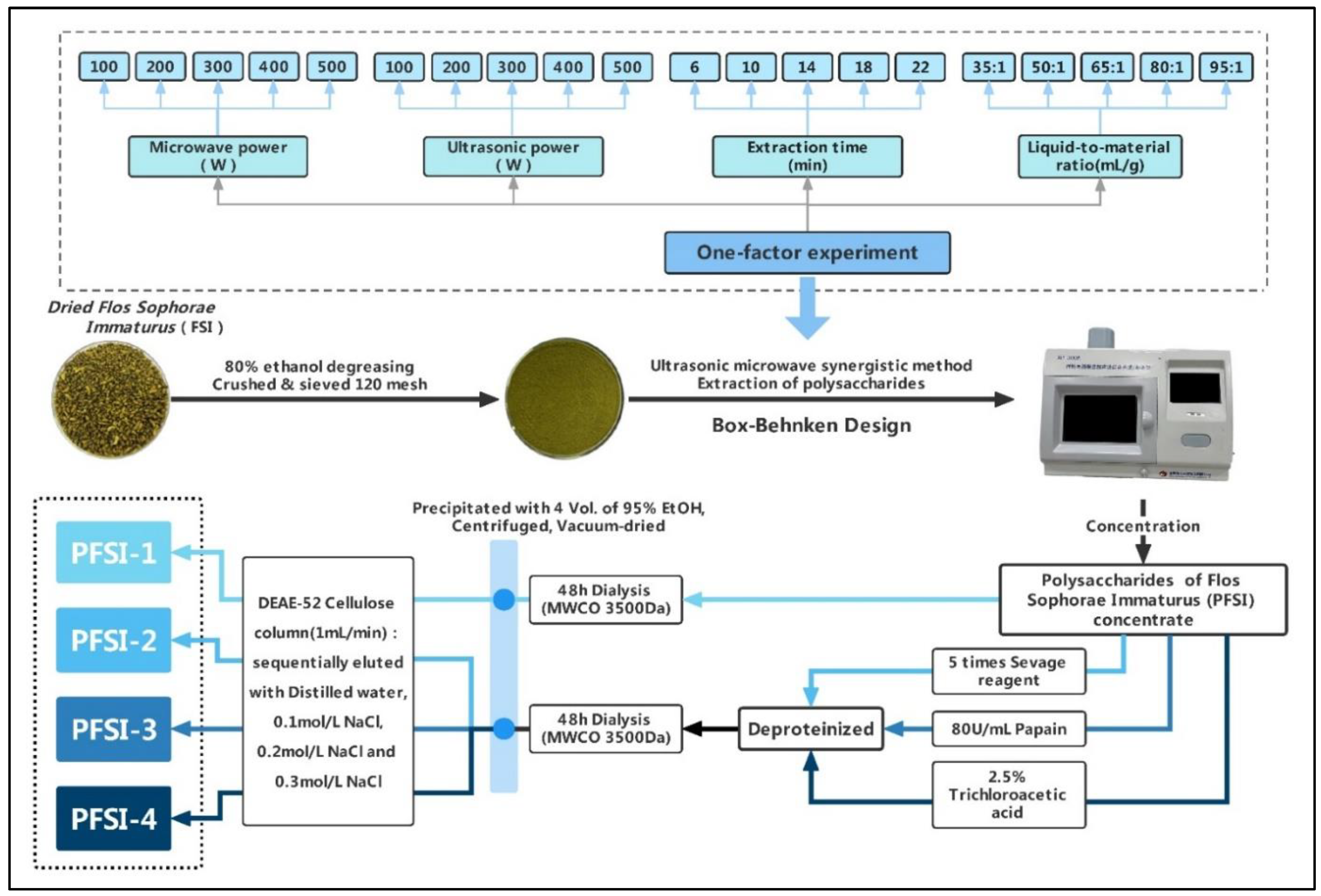
2.2. The Process Investigation of the UMSE Method to Obtain PFSI
2.3. One-Factor Experiment for Extraction
2.3.1. Effect of Microwave Power on Extraction
2.3.2. Effect of Ultrasonic Power on Extraction
2.3.3. Effect of Particle Diameter on Extraction
2.3.4. Effect of Liquid-to-Material Ratio on Extraction
2.3.5. Effect of Time on Extraction
2.4. Design of RSM
2.5. In Vitro Antioxidant Effect of PFSI
2.5.1. Measurement of DPPH· Radical Scavenging Rate
2.5.2. Measurement of ·OH Radical Scavenging Rate
2.5.3. Measurement of ABTS· Radical Scavenging Rate
2.5.4. Measurement of Ferric-Reducing Antioxidant Power
2.6. Statistical Analysis
3. Results
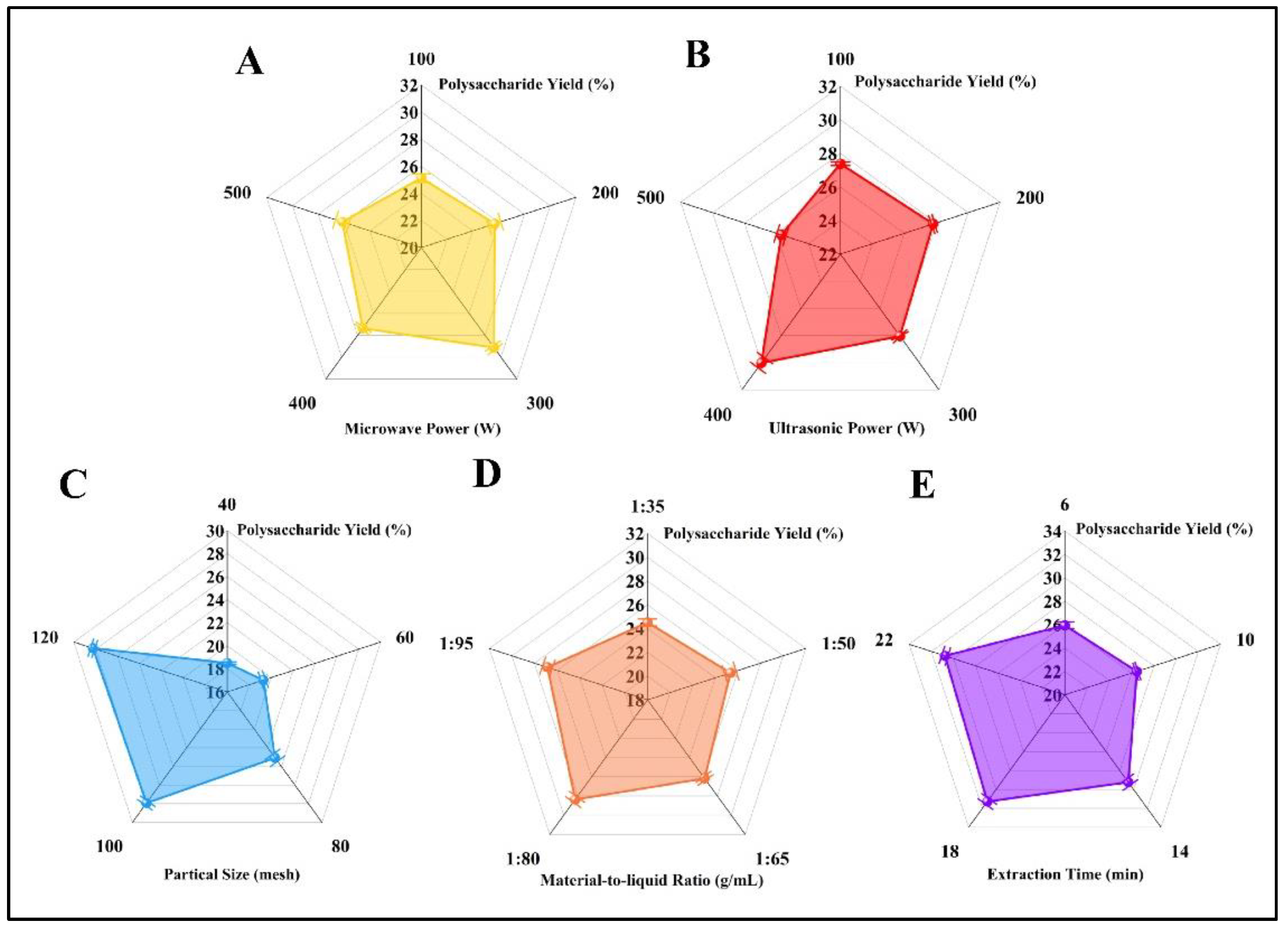
3.1. Analysis of One-Factor Experiment
3.1.1. Analysis of Microwave Power on Extraction
3.1.2. Analysis of Ultrasound Power on Extraction
3.1.3. Analysis of Particle Diameter on Extraction
3.1.4. Analysis of Liquid-to-Material Ratio on Extraction
3.1.5. Analysis of Time on Extraction
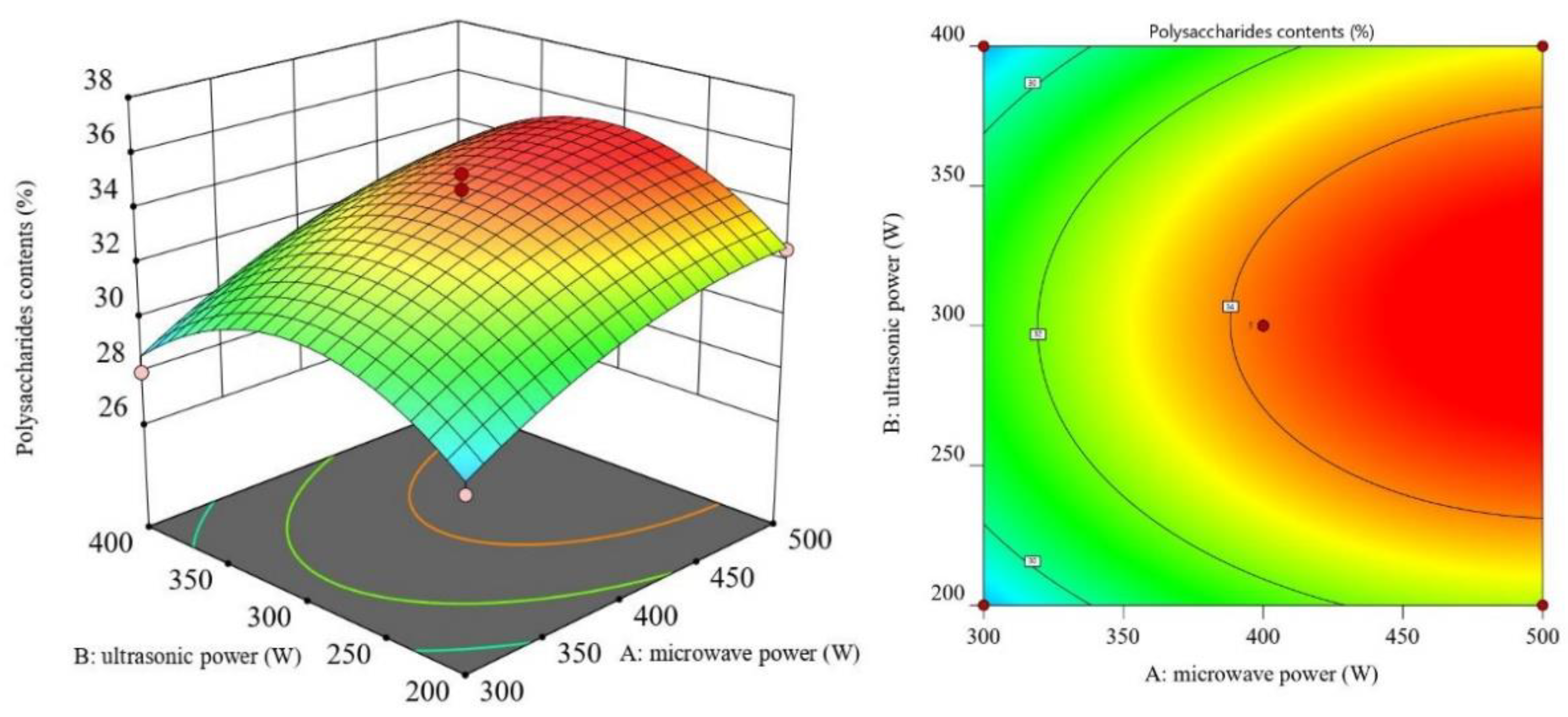
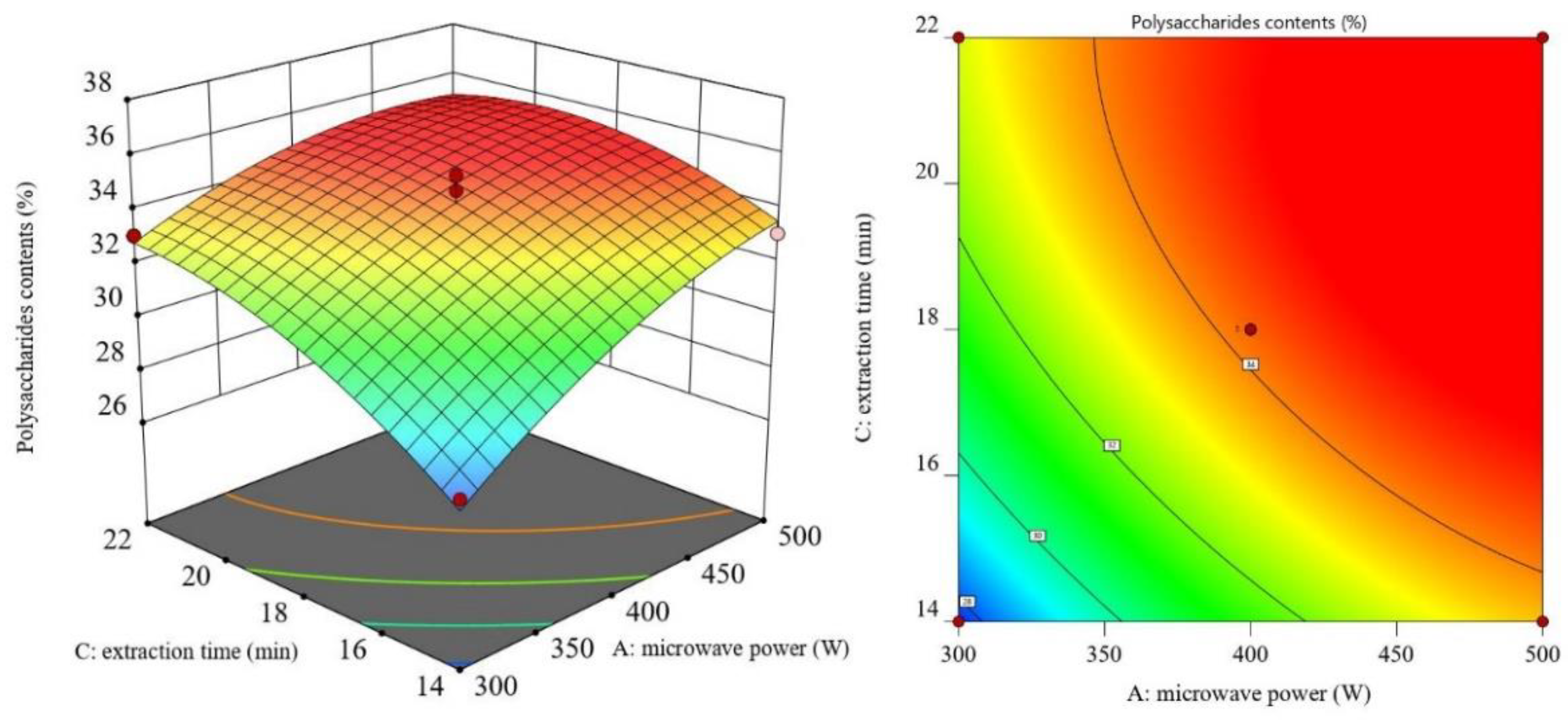
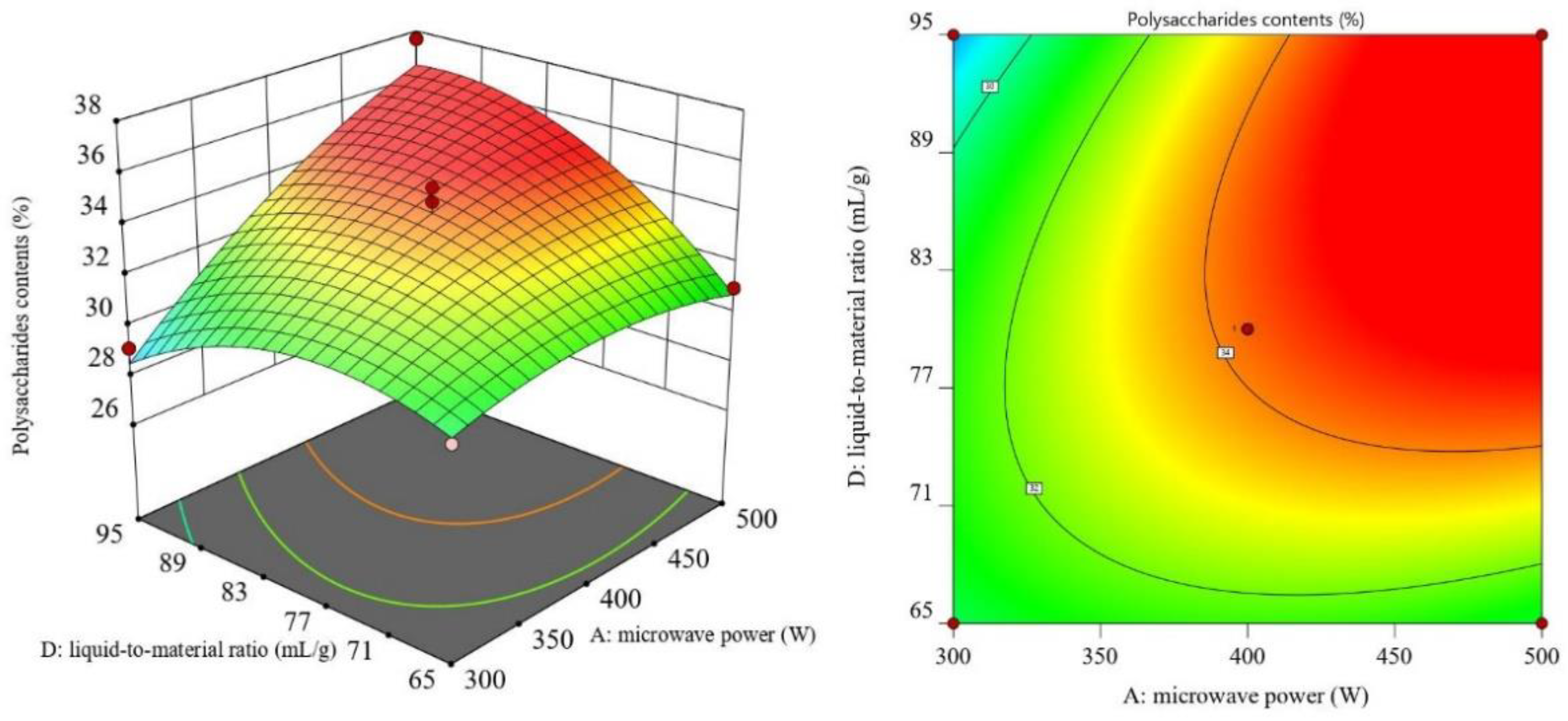
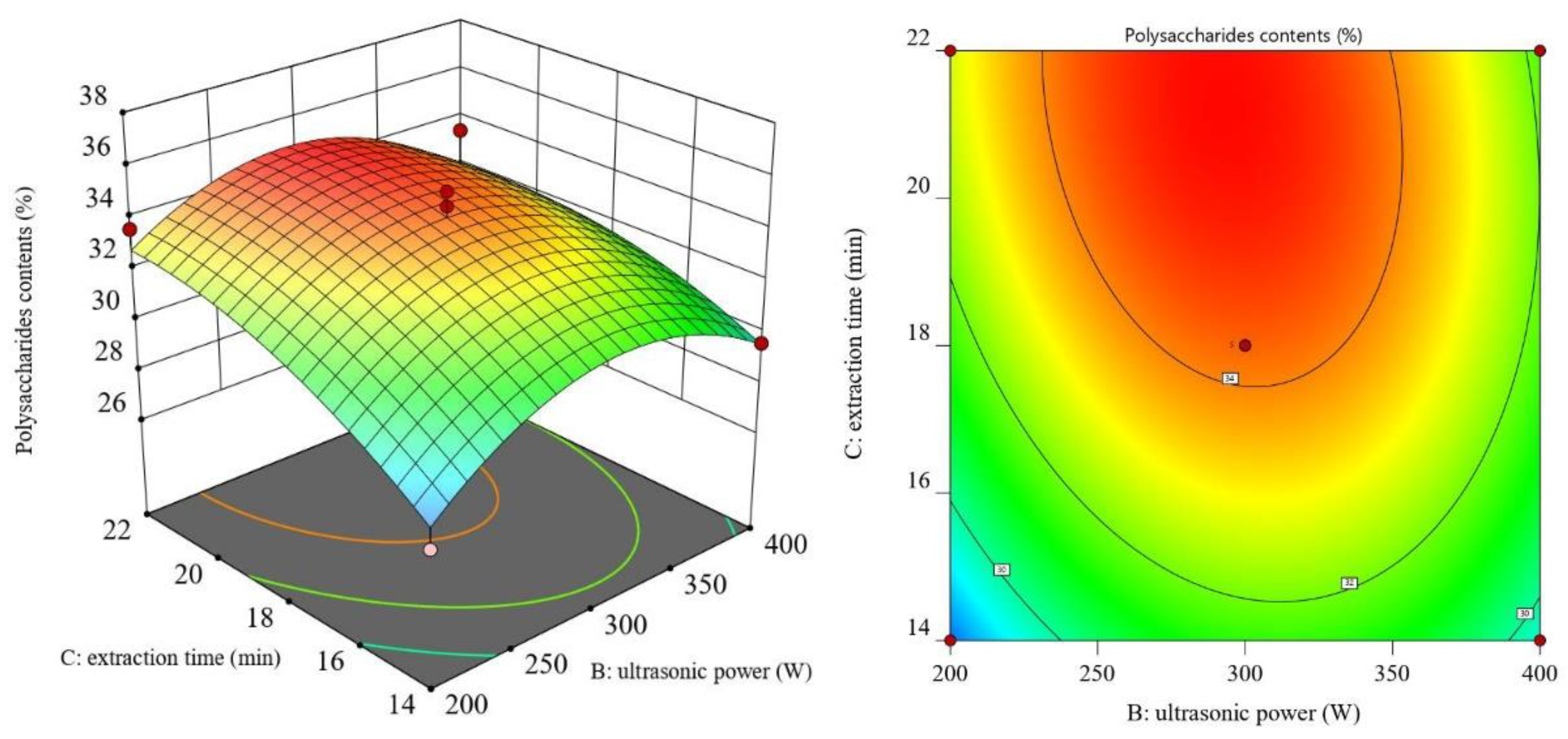
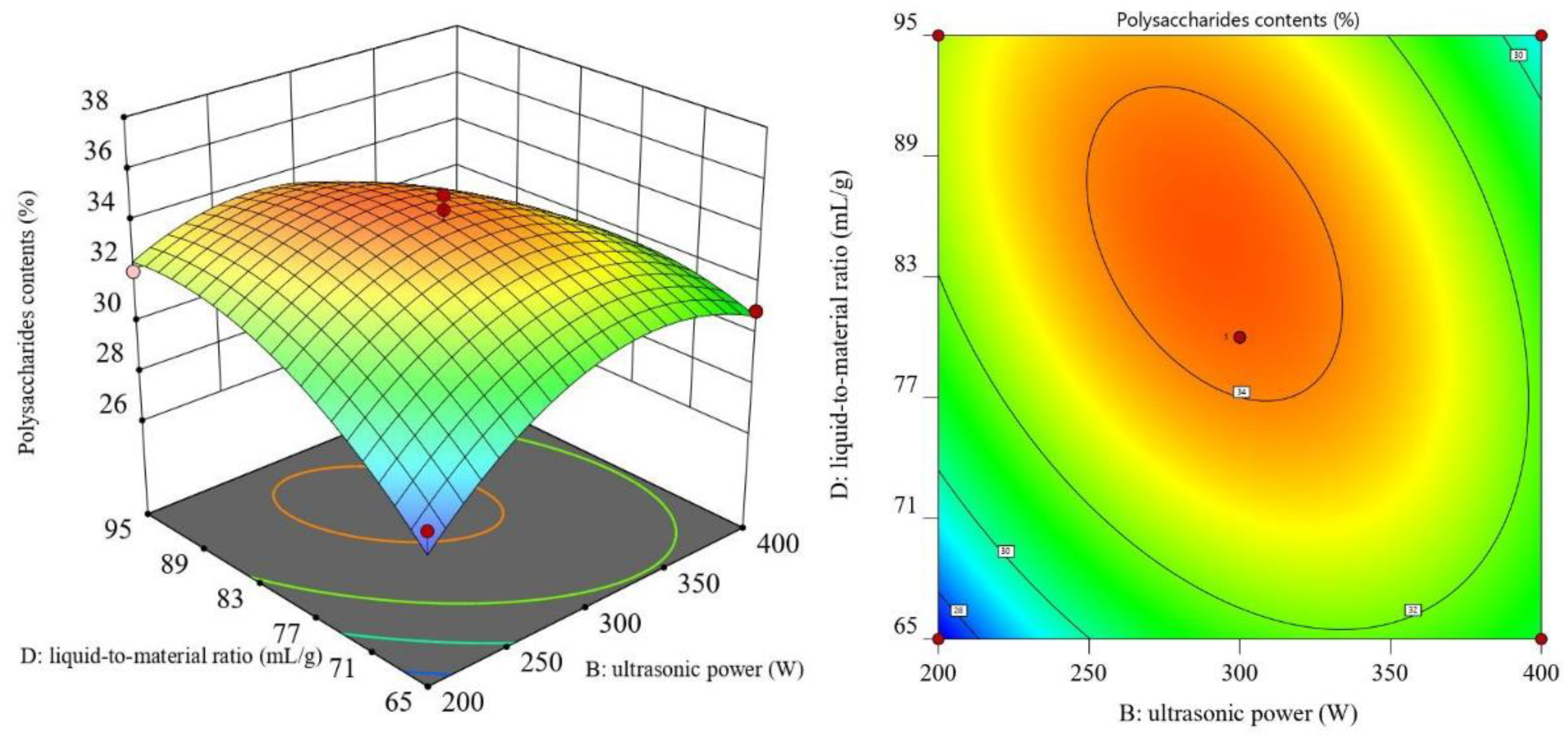
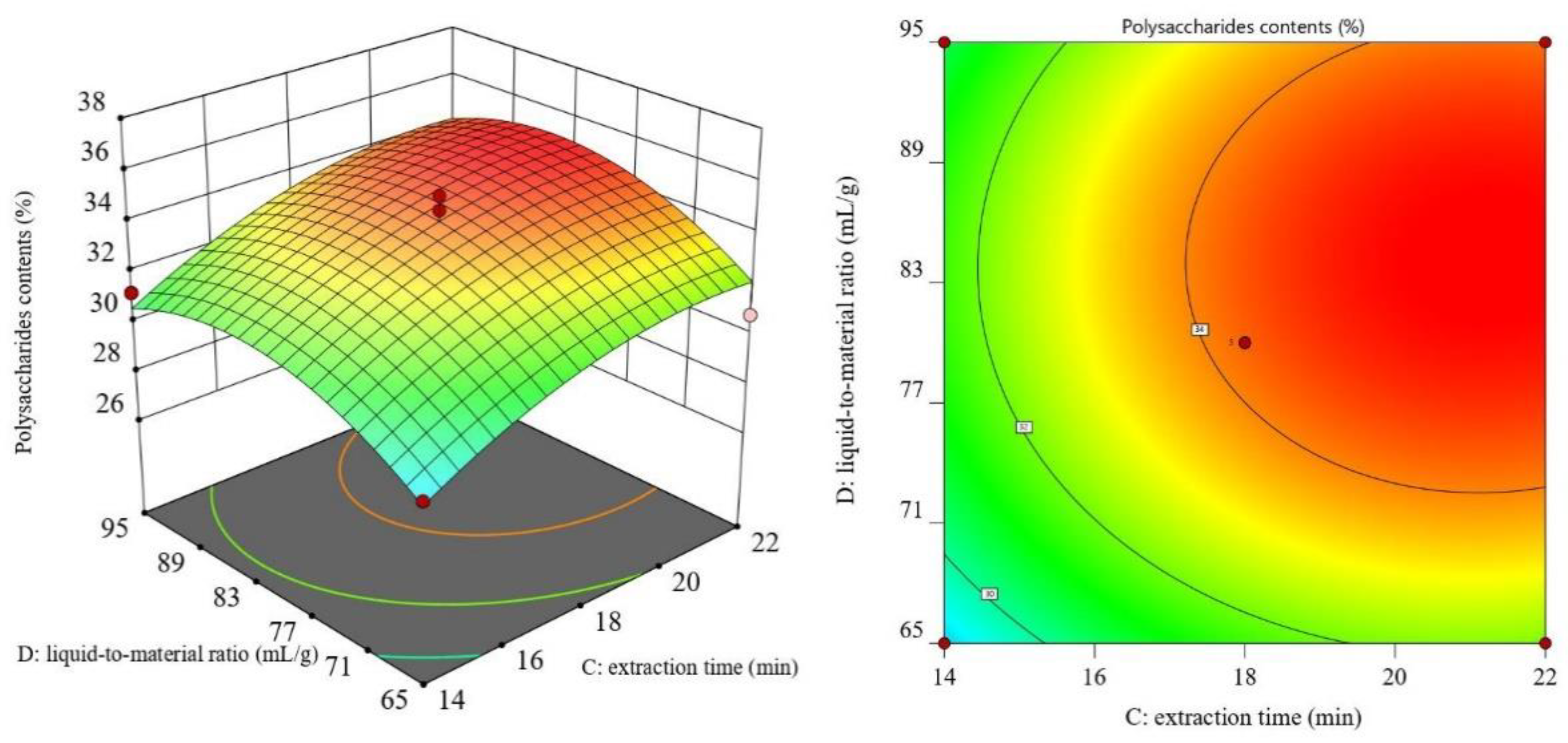
3.2. RSM Results and ANOVA
3.3. Optimal Process Conditions and Validation Experiments
3.4. Analysis of PFSI In Vitro Antioxidant
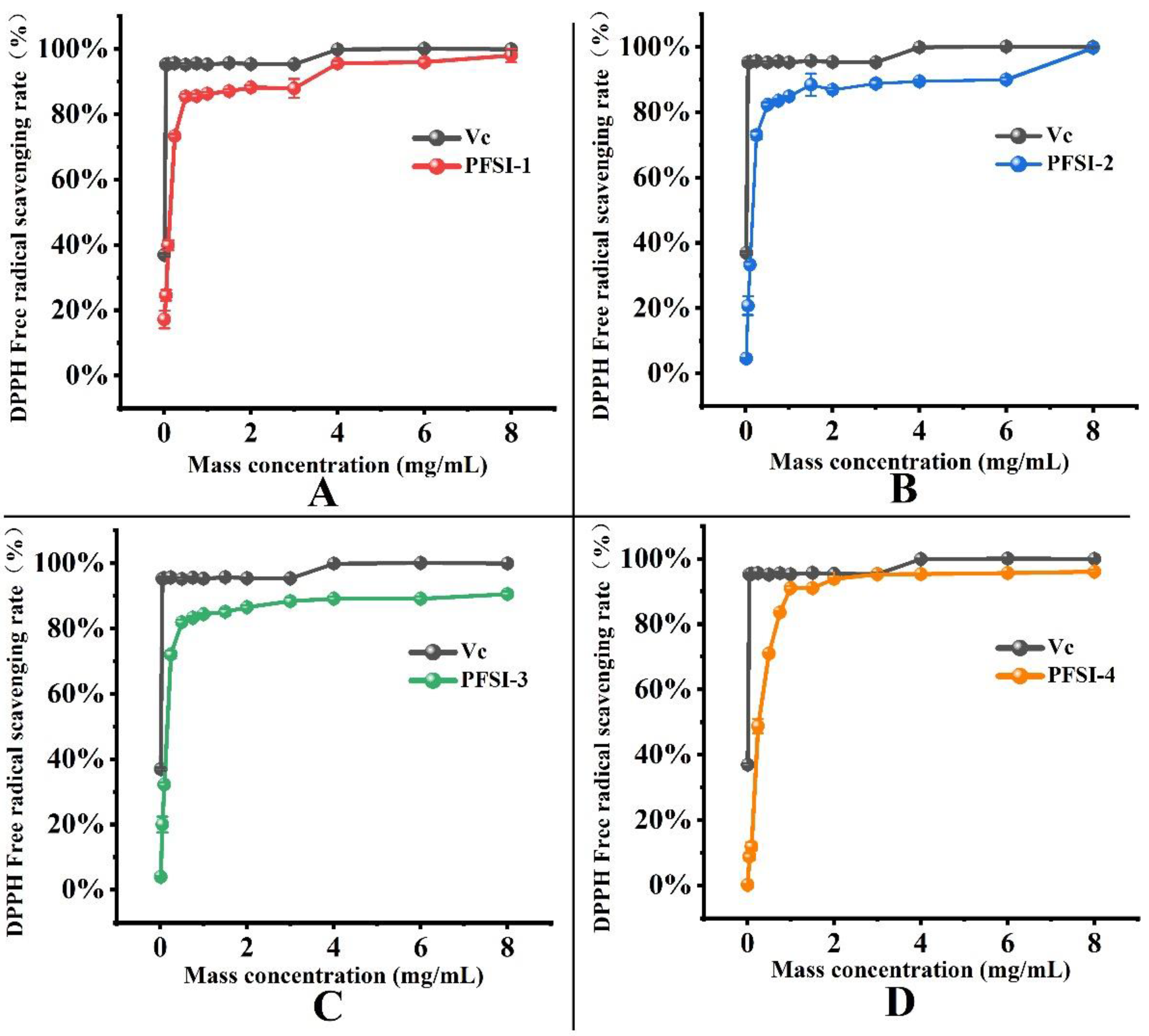
3.4.1. Analysis of DPPH· Radical Scavenging Rate
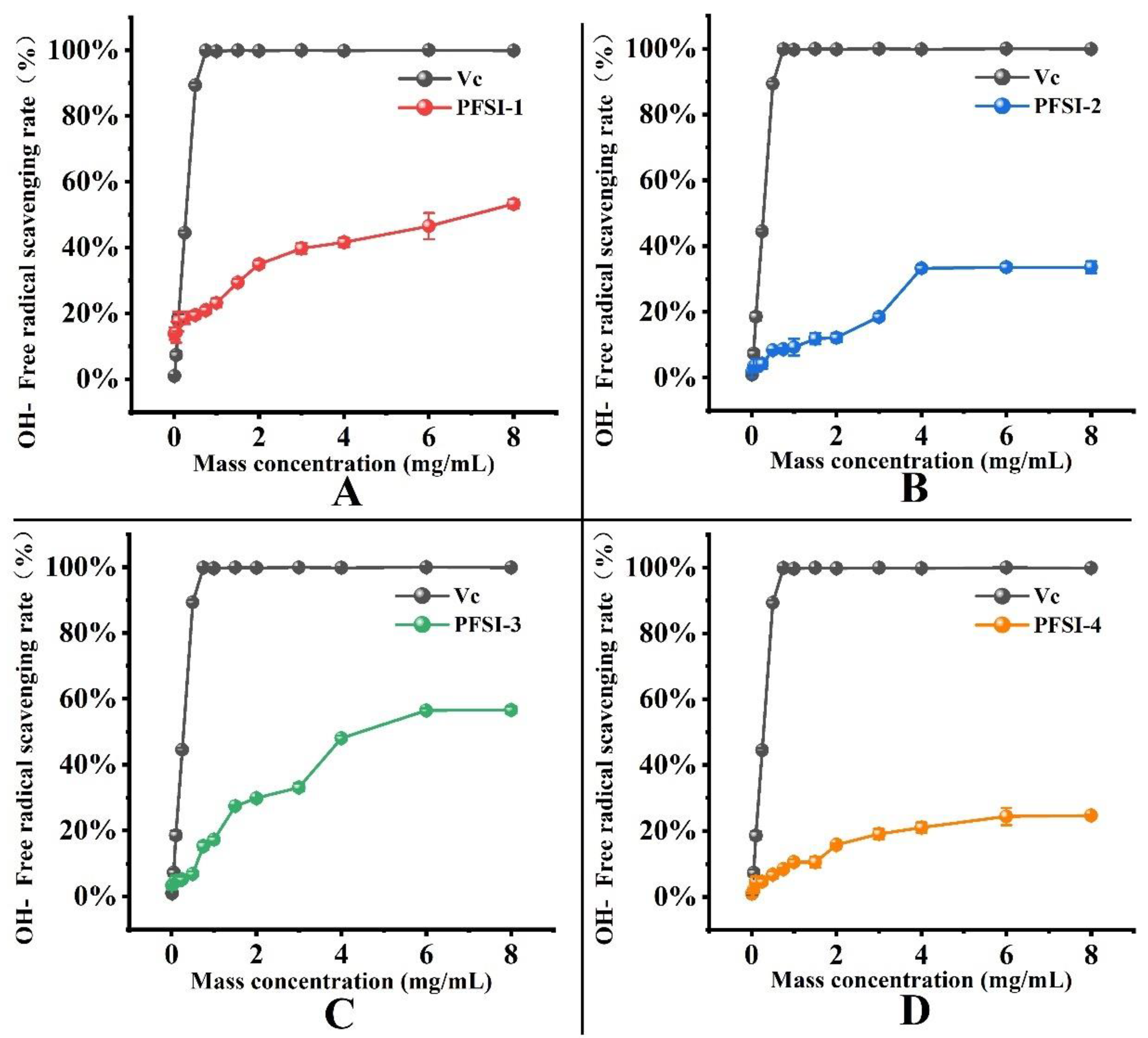
3.4.2. Analysis of ·OH Radical Scavenging Rate
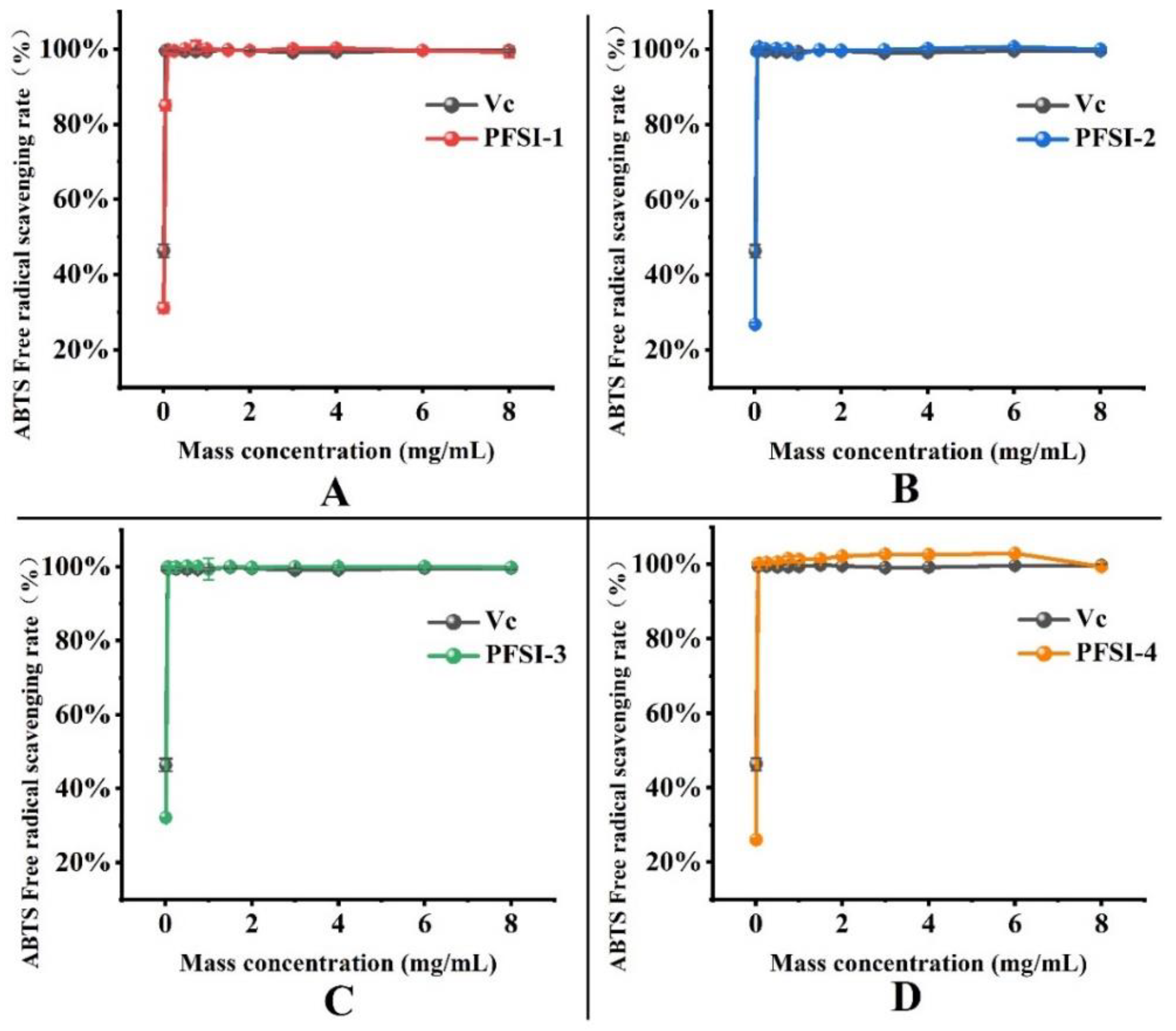
3.4.3. Analysis of ABTS· Radical Scavenging Rate
3.4.4. Analysis of Ferric-Reducing Antioxidant Power
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Liu, Y.; Sun, Y.Y.; Huang, G.L. Preparation and antioxidant activities of important traditional plant polysaccharides. Int. J. Biol. Macromol. 2018, 111, 780–786. [Google Scholar] [CrossRef] [PubMed]
- Yin, M.; Zhang, Y.; Li, H. Advances in Research on Immunoregulation of Macrophages by Plant Polysaccharides. Front. Immunol. 2019, 10, 145. [Google Scholar] [CrossRef] [PubMed]
- Hu, D.J.; Cheong, K.L.; Zhao, J.; Li, S.P. Chromatography in characterization of polysaccharides from medicinal plants and fungi. J. Sep. Sci. 2013, 36, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Zhou, X.; Chen, H.G. Structure-activity relationship of plant polysaccharides. China J. Chin. Mater. Med. 2017, 42, 4104–4109. [Google Scholar] [CrossRef]
- Huang, H.L.; Huang, G.L. Extraction, separation, modification, structural characterization, and antioxidant activity of plant polysaccharides. Chem. Biol. Drug Des. 2022, 96, 1209–1222. [Google Scholar] [CrossRef]
- Ahmad, M.M.; Shahid Chatha, S.A.; Iqbal, Y.; Hussain, A.I.; Khan, I.; Xie, F. Recent trends in extraction, purification, and antioxidant activity evaluation of plant leaf-extract polysaccharides. Biofuels Bioprod. Biorefining 2022, 16, 1820–1848. [Google Scholar] [CrossRef]
- Huang, G.L.; Chen, F.; Yang, W.J.; Huang, H.L. Preparation, deproteinization and comparison of bioactive polysaccharides. Trends Food Sci. Technol. 2021, 109, 264–568. [Google Scholar] [CrossRef]
- Spagnuolo, D.; Martino, A.D.; Zammuto, V.; Minicante, S.A.; Spanò, A.; Manghisi, A.; Gugliandolo, C.; Morabito, M.; Genovese, G. Conventional vs. Innovative Protocols for the Extraction of Polysaccharides from Macroalgae. Sustainability 2022, 14, 5750. [Google Scholar] [CrossRef]
- Essa, H.; Fleita, D.; Rifaat, D.; Samy, S.; EI-Sayed, M. Towards optimizing the conventional and ultrasonic-assisted extraction of sulfated polysaccharides from marine algae. IOP Conf. Ser. Mater. Sci. Eng. 2017, 464, 012007. [Google Scholar] [CrossRef]
- Guo, Z.B.; Zhao, B.B.; Li, H.; Miao, S.; Zheng, B.D. Optimization of ultrasound-microwave synergistic extraction of prebiotic oligosaccharides from sweet potatoes (Ipomoea batatas L.). Innov. Food Sci. Emerg. Technol. 2019, 54, 51–63. [Google Scholar] [CrossRef]
- Li, F.; Li, Q.H.; Li, Y.L. Research progress on the determination of active ingredients of Sophora japonica. Inf. Tradit. Chin. Med. 2008, 25, 19–21. [Google Scholar] [CrossRef]
- Hu, A.H. Herbal evidence on the use of Sophora as a medicine. Chin. Tradit. Pat. Med. 2018, 40, 2587–2589. [Google Scholar] [CrossRef]
- Wang, S.L.; Yu, H.; Li, C.Q.; Ge, B.E.; Xi, G.Q.; Liu, S.Y. Preparation and Purification of Rutin and Quercetin from Sophora Japonica by Semi-Preparative HPLC. Food Sci. Technol. 2022, 47, 245–250. [Google Scholar] [CrossRef]
- Ouyang, L.L.; Hua, P. Research Progress in Active Components and Functions of the Flower Plants Homology of Medicine and Food. Jiangxi Chem. Ind. 2020, 36, 58–60. [Google Scholar] [CrossRef]
- Kang, W.Y.; Wu, X.H. Analysis of fatty acids from flower, bud and leaves of Sophora japonica L. By GC-MS. J. Henan Univ. (Med. Sci.) 2009, 28, 17–20. [Google Scholar] [CrossRef]
- Wu, C.S.; Zhu, G.S. Comparation on Extraction Methods of Polysaccharide from Sophora Japonica. Farm Prod. Process. 2018, 14, 36–38+41. [Google Scholar] [CrossRef]
- Chen, D.W.; Li, J.H.; Wang, F. Detemination of Volatile compoents in Flos Sophorae immaturus by GC-MS. West China J. Pharm. Sci. 2006, 5, 450–451. [Google Scholar] [CrossRef]
- Zhong, W.T.; Yang, C.M.; Zhang, Y.Z.; Liu, Y.M.; Yang, D.S. The Chemical Profiling and Anticancer Potential of Functional Polysaccharides from Flos Sophorae Immaturus. Molecules 2022, 27, 5978. [Google Scholar] [CrossRef]
- Koltover, V.K. Free Radical Timer of Aging: From Chemistry of Free Radicals to Systems Theory of Reliability. Curr. Aging Sci. 2017, 10, 12–17. [Google Scholar] [CrossRef]
- Ivanova, D.G.; Yankova, T.M. The free radical theory of aging in search of a strategy for increasing life span. Folia Med. 2013, 55, 33–41. [Google Scholar] [CrossRef]
- Viña, J. The free radical theory of frailty: Mechanisms and opportunities for interventions to promote successful aging. Free Radic. Biol. Med. 2019, 134, 690–694. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; You, W.L.; Huang, J.; Yu, Y. Isolation and antioxidant property of the extracellular polysaccharide from Rhodella reticulata. World J. Microbiol. Biotechnol. 2010, 26, 833–840. [Google Scholar] [CrossRef]
- Darsih, C.; Apriyana, W.; Hayati, S.N.; Rosyida, V.T. The elevation effect on water-soluble polysaccharides and DPPH free radical scavenging activity of Ganoderma lucidum K. IOP Conf. Ser. Mater. Sci. Eng. 2017, 172, 012010. [Google Scholar] [CrossRef]
- Zhang, L.; Gu, J.; Chen, Y.; Zhang, L. A study on four antioxidation effects of Lycium barbarum polysaccharides In vitro. Afr. J. Online 2013, 10, 6. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.L.; Liu, H.Q.; Deng, Y.H.; Liu, M.L. Study on the purification and scavenging free radical activity of water-soluble polysaccharide of leave in Hippohae rhamnoides L. J. Chin. Med. Mater. 2006, 2, 151–154. [Google Scholar] [CrossRef]
- Fan, L.P.; Li, J.W.; Deng, K.Q.; Ai, L.Z. Effects of drying methods on the antioxidant activities of polysaccharides extracted from Ganoderma lucidum. Carbohydr. Polym. 2012, 87, 1849–1854. [Google Scholar] [CrossRef]
- Fuente, M.D. Effects of antioxidants on immune system ageing. Eur. J. Clin. Nutr. 2002, 56, S5–S8. [Google Scholar] [CrossRef]
- Ren, Y.; Bai, Y.; Zhang, Z.; Cai, W.; Del Rio Flores, A. The Preparation and Structure Analysis Methods of Natural Polysaccharides of Plants and Fungi: A Review of Recent Development. Molecules 2019, 24, 3122. [Google Scholar] [CrossRef]
- Shi, L. Bioactivities, isolation and purification methods of polysaccharides from natural products: A review. Int. J. Biol. Macromol. 2016, 92, 37–48. [Google Scholar] [CrossRef]
- Garcia-Vaquero, M.; Rajauria, G.; Doherty, J.V.; Sweeney, T. Polysaccharides from macroalgae: Recent advances, innovative technologies and challenges in extraction and purification. Food. Res. Int. 2017, 99, 1011–1020. [Google Scholar] [CrossRef]
- Ji, X.L.; Peng, Q.; Yuan, Y.P.; Shen, J.; Xie, X.Y.; Wang, M. Isolation, structures and bioactivities of the polysaccharides from jujube fruit (Ziziphus jujuba Mill.): A review. Food Chem. 2017, 227, 349–357. [Google Scholar] [CrossRef] [PubMed]
- Qin, K.; Liu, C.H.; Qi, Y.X.; Li, K. Evaluation of Antioxidant Activity of Polysaccharides from Tussilago farfara L. by Flow Injection Analysis. Asian J. Chem. 2014, 26, 3073–3076. [Google Scholar] [CrossRef]
- Xiang, J.F.; Xiang, Y.J.; Lin, S.M.; Xin, D.W.; Liu, X.Y.; Weng, L.L.; Chen, T.; Zhang, M.G. Anticancer effects of deproteinized asparagus polysaccharide on hepatocellular carcinoma In vitro and in vivo. Tumor Biol. 2013, 35, 3517–3524. [Google Scholar] [CrossRef]
- Li, S.F.; Wang, A.J.; Liu, L.N.; Tian, G.R.; Xu, F.F. Effect of deproteinization methods on the antioxidant activity of polysaccharides extracted from Lentinus edodes stipe. J. Food Meas. Charact. 2019, 13, 1382–1389. [Google Scholar] [CrossRef]
- Nie, L.R.; Hao, L.M.; Wang, T.T.; Liu, Y.; Zhang, L.M.; Lu, J.K.; Kang, C.C.; Cui, Y.; Han, P.P.; Jia, S.R. Antioxidant Activity of Crude Polysaccharides from Fomitopsis pinicola from Different Geographical Origins. J. Food Sci. 2019, 40, 60–68. [Google Scholar] [CrossRef]
- Liu, M.M.; Huang, Q.N.; Guo, L.Z. The Study on Extraction and Antioxidant Activity of Polysaccharides from Trametes robiniophila Murr. Edible Fungi China 2013, 32, 47–49. [Google Scholar] [CrossRef]
- Zong, A.Z.; Cao, H.Z.; Wang, F.S. Anticancer polysaccharides from natural resources: A review of recent research. Carbohydr. Polym. 2012, 90, 1395–1410. [Google Scholar] [CrossRef]
- Zhou, S.Y.; Huang, G.L.; Chen, G.Y. Extraction, structural analysis, derivatization and antioxidant activity of polysaccharide from Chinese yam. Food Chem. 2021, 361, 130089. [Google Scholar] [CrossRef]
- Cui, R.B.; Zhu, F. Ultrasound modified polysaccharides: A review of structure, physicochemical properties, biological activities and food applications. Trends Food Sci. Technol. 2021, 107, 491–508. [Google Scholar] [CrossRef]
- Liu, X.X.; Yan, Y.Y.; Liu, H.M.; Wang, X.D.; Qin, G.Y. Emulsifying and structural properties of polysaccharides extracted from Chinese yam by an enzyme-assisted method. LWT 2019, 111, 242–251. [Google Scholar] [CrossRef]
- Chen, L.; Huang, G.L.; Hu, J.C. Preparation, deproteinization, characterisation, and antioxidant activity of polysaccharide from cucumber (Cucumis saticus L.). Int. J. Biol. Macromol. 2017, 108, 408–411. [Google Scholar] [CrossRef]
- Zhou, Y.; Xia, X.K. Research on Extracting Technology of Polysaccharide from Flos Sophorae Immaturus. Hubei Agric. Sci. 2011, 50, 3161–3163. [Google Scholar] [CrossRef]
- Li, N.X.; Zhang, Y.P. Optimization of Ultrasound-assisted Polysaccharide Extraction from Flos Sophorae Immaturus by Central Composite Design-Response Surface Methodology. China Pharm. 2013, 16, 1809–1811. [Google Scholar] [CrossRef]

| Level | A | B | C | D |
|---|---|---|---|---|
| −1 | 200 | 300 | 14 | 65:1 |
| 0 | 300 | 400 | 18 | 80:1 |
| 1 | 400 | 500 | 22 | 95:1 |
| No. | A | B | C | D | Y/(%) |
|---|---|---|---|---|---|
| 1 | 200 | 300 | 18 | 80:1 | 28.26795 |
| 2 | 400 | 300 | 18 | 80:1 | 32.46507 |
| 3 | 200 | 500 | 18 | 80:1 | 27.97342 |
| 4 | 400 | 500 | 18 | 80:1 | 32.78722 |
| 5 | 300 | 400 | 14 | 65:1 | 29.04766 |
| 6 | 300 | 400 | 22 | 65:1 | 30.80509 |
| 7 | 300 | 400 | 14 | 95:1 | 31.21825 |
| 8 | 300 | 400 | 22 | 95:1 | 33.28401 |
| 9 | 200 | 400 | 18 | 65:1 | 30.32647 |
| 10 | 400 | 400 | 18 | 65:1 | 31.02944 |
| 11 | 200 | 400 | 18 | 95:1 | 29.13062 |
| 12 | 400 | 400 | 18 | 95:1 | 37.66694 |
| 13 | 300 | 300 | 14 | 80:1 | 27.30151 |
| 14 | 300 | 500 | 14 | 80:1 | 29.60256 |
| 15 | 300 | 300 | 22 | 80:1 | 33.58798 |
| 16 | 300 | 500 | 22 | 80:1 | 33.42231 |
| 17 | 200 | 400 | 14 | 80:1 | 28.01944 |
| 18 | 400 | 400 | 14 | 80:1 | 33.07562 |
| 19 | 200 | 400 | 22 | 80:1 | 33.07255 |
| 20 | 400 | 400 | 22 | 80:1 | 34.60045 |
| 21 | 300 | 300 | 18 | 65:1 | 27.97825 |
| 22 | 300 | 500 | 18 | 65:1 | 30.95466 |
| 23 | 300 | 300 | 18 | 95:1 | 32.05985 |
| 24 | 300 | 500 | 18 | 95:1 | 28.08134 |
| 25 | 300 | 400 | 18 | 80:1 | 33.93774 |
| 26 | 300 | 400 | 18 | 80:1 | 34.68328 |
| 27 | 300 | 400 | 18 | 80:1 | 35.24474 |
| 28 | 300 | 400 | 18 | 80:1 | 33.95615 |
| 29 | 300 | 400 | 18 | 80:1 | 33.45912 |
| Source | Sum of Squares | df | Mean Square | F-Value | p-Value | Significance |
|---|---|---|---|---|---|---|
| Model | 186.32 | 14 | 13.31 | 12.97 | <0.0001 | ** |
| A | 51.40 | 1 | 51.40 | 50.10 | <0.0001 | ** |
| B | 0.1123 | 1 | 0.1123 | 0.1095 | 0.7456 | -- |
| C | 35.05 | 1 | 35.05 | 34.16 | <0.0001 | ** |
| D | 10.64 | 1 | 10.64 | 10.37 | 0.0062 | ** |
| AB | 0.0951 | 1 | 0.0951 | 0.0927 | 0.7653 | -- |
| AC | 3.11 | 1 | 3.11 | 3.03 | 0.1035 | -- |
| AD | 15.34 | 1 | 15.34 | 14.95 | 0.0017 | ** |
| BC | 1.52 | 1 | 1.52 | 1.48 | 0.2434 | -- |
| BD | 12.09 | 1 | 12.09 | 11.79 | 0.0040 | ** |
| CD | 0.0238 | 1 | 0.0238 | 0.0232 | 0.8812 | -- |
| A2 | 5.25 | 1 | 5.25 | 5.12 | 0.0401 | * |
| B2 | 45.25 | 1 | 45.25 | 44.11 | <0.0001 | ** |
| C2 | 7.45 | 1 | 7.45 | 7.26 | 0.0174 | * |
| D2 | 19.95 | 1 | 19.95 | 19.44 | 0.0006 | ** |
| Residual | 14.36 | 14 | 1.03 | |||
| Lack of Fit | 12.37 | 10 | 1.24 | 2.49 | 0.1964 | -- |
| Pure Error | 1.99 | 4 | 0.4966 | |||
| Cor Total | 200.68 | 28 | ||||
| R2 = 0.9284 | Radj2 = 0.8569 | C.V.% = 3.20 |
| Mass Concentration (mg/mL) | DPPH· Free Radical Scavenging Rate | ||||
|---|---|---|---|---|---|
| VC | PFSI-1 | PFSI-2 | PFSI-3 | PFSI-4 | |
| 0.01 | 36.90% ± 0.49% d | 17.16% ± 2.66% f | 4.56% ± 0.22% h | 3.95% ± 0.25% i | 0.15% ± 0.15% i |
| 0.05 | 95.25% ± 0.18% c | 24.53% ± 1.65% e | 20.75% ± 2.84% g | 20.00% ± 2.41% h | 8.75% ± 1.09% h |
| 0.1 | 95.34% ± 0.37% bc | 39.85% ± 1.51% d | 33.25% ± 0.90% f | 32.24% ± 0.65% g | 11.89% ± 1.28% g |
| 0.25 | 95.61% ± 0.18% b | 73.30% ± 0.64% c | 72.89% ± 1.22% e | 71.99% ± 1.18% f | 48.76% ± 2.15% f |
| 0.5 | 95.13% ± 0.43% c | 85.39% ± 0.75% b | 82.23% ± 0.75% d | 81.90% ± 0.75% e | 70.90% ± 0.33% e |
| 0.75 | 95.55% ± 0.30% bc | 85.53% ± 0.61% b | 83.52% ± 0.32% cd | 83.23% ± 0.47% de | 83.52% ± 0.26% d |
| 1 | 95.25% ± 0.09% bc | 86.28% ± 0.42% b | 84.78% ± 0.07% cd | 84.34% ± 0.18% cd | 90.96% ± 0.58% c |
| 1.5 | 95.64% ± 0.11% b | 87.06% ± 0.54% b | 88.40% ± 3.38% b | 85.06% ± 0.90% cd | 90.99% ± 0.18% c |
| 2 | 95.32% ± 0.16% bc | 88.19% ± 0.64% b | 86.79% ± 0.57% bc | 86.50% ± 0.61% bc | 93.80% ± 0.26% b |
| 3 | 95.27% ± 0.39% bc | 87.89% ± 2.88% b | 88.73% ± 1.11% b | 88.37% ± 1.08% ab | 95.19% ± 0.22% ab |
| 4 | 99.82% ± 0.09% a | 95.55% ± 0.82% a | 89.41% ± 0.50% b | 89.08% ± 0.61% a | 95.30% ± 0.11% ab |
| 6 | 100.02% ± 0.21% a | 95.97% ± 1.06% a | 89.95% ± 0.61% b | 89.12% ± 0.65% a | 95.59% ± 0.29% a |
| 8 | 99.91% ± 0.09% a | 98.00% ± 2.05% a | 99.64% ± 0.83% a | 90.52% ± 0.75% a | 96.06% ± 0.44% a |
| Mass Concentration (mg/mL) | ·OH Free Radical Scavenging Rate | ||||
|---|---|---|---|---|---|
| VC | PFSI-1 | PFSI-2 | PFSI-3 | PFSI-4 | |
| 0.01 | 0.96% ± 0.30% f | 13.85% ± 1.84% h | 1.74% ± 0.09% d | 3.34% ± 0.32% j | 1.06% ± 0.09% g |
| 0.05 | 7.32% ± 1.01% e | 14.26% ± 3.19% h | 3.36% ± 0.31% d | 4.00% ± 0.78% ij | 2.06% ± 0.49% g |
| 0.1 | 18.56% ± 1.27% d | 17.49% ± 2.92% gh | 3.79% ± 1.92% d | 4.83% ± 1.55% ij | 4.75% ± 1.46% f |
| 0.25 | 44.50% ± 0.59% c | 18.66% ± 1.86% g | 4.18% ± 1.44% d | 5.26% ± 0.89% i | 4.63% ± 0.83% f |
| 0.5 | 89.33% ± 0.71% b | 19.48% ± 1.14% g | 8.37% ± 0.03% c | 6.93% ± 0.72% h | 6.75% ± 1.06% ef |
| 0.75 | 99.86% ± 0.18% a | 20.95% ± 1.04% fg | 8.61% ± 0.37% c | 15.33% ± 1.06% g | 8.47% ± 0.37% de |
| 1 | 99.66% ± 0.22% a | 23.19% ± 1.33% f | 9.25% ± 2.57% c | 17.20% ± 0.81% f | 10.61% ± 0.80% d |
| 1.5 | 99.92% ± 0.08% a | 29.39% ± 0.97% e | 11.85% ± 1.71% c | 27.44% ± 0.23% e | 10.61% ± 1.57% d |
| 2 | 99.80% ± 0.12% a | 34.92% ± 1.18% d | 12.19% ± 1.34% c | 29.80% ± 0.17% d | 15.76% ± 0.37% c |
| 3 | 99.92% ± 0.08% a | 39.74% ± 1.62% c | 18.45% ± 1.07% b | 33.07% ± 1.24% c | 19.08% ± 1.54% b |
| 4 | 99.80% ± 0.09% a | 41.60% ± 1.39% c | 33.26% ± 1.01% a | 48.03% ± 0.75% b | 21.00% ± 1.52% b |
| 6 | 100.00% ± 0.22% a | 46.49% ± 3.96% b | 33.57% ± 1.13% a | 56.43% ± 0.60% a | 24.37% ± 2.60% a |
| 8 | 99.84% ± 0.35% a | 53.24% ± 1.35% a | 33.60% ± 1.80% a | 56.57% ± 1.09% a | 24.66% ± 0.26% a |
| Mass Concentration (mg/mL) | ABTS· Free Radical Scavenging Rate | ||||
|---|---|---|---|---|---|
| VC | PFSI-1 | PFSI-2 | PFSI-3 | PFSI-4 | |
| 0.01 | 46.36% ± 1.71%b | 31.11% ± 1.43% d | 26.81% ± 0.47% d | 32.11% ± 0.08% b | 26.05% ± 0.32% e |
| 0.05 | 99.51% ± 0.49% a | 84.99% ± 1.43% c | 99.45% ± 0.16% bc | 99.92% ± 0.16% a | 100.08% ± 0.08% c |
| 0.1 | 99.76% ± 0.49% a | 99.54% ± 0.68% b | 100.62% ± 0.16% a | 99.76% ± 0.24% a | 100.16% ± 0.40% c |
| 0.25 | 99.45% ± 0.43% a | 99.49% ± 0.86% b | 100.08% ± 0.78% ab | 99.92% ± 0.40% a | 100.40% ± 0.40% c |
| 0.5 | 99.33% ± 0.31% a | 100.00% ± 0.23% ab | 100.23% ± 0.23% ab | 100.16% ± 0.16% a | 100.56% ± 0.32% c |
| 0.75 | 99.33% ± 0.55% a | 100.68% ± 1.60% a | 100.16% ± 0.16% ab | 100.08% ± 0.32% a | 101.45% ± 0.72% b |
| 1 | 99.39% ± 0.37% a | 99.94% ± 0.40% ab | 98.75% ± 1.09% c | 99.36% ± 2.94% a | 101.29% ± 0.80% b |
| 1.5 | 99.82% ± 0.31% a | 99.71% ± 0.17% b | 99.92% ± 0.16% ab | 100.08% ± 0.08% a | 101.37% ± 0.16% b |
| 2 | 99.57% ± 0.06% a | 99.54% ± 0.91% b | 99.77% ± 0.23% b | 99.84% ± 0.08% a | 102.17% ± 0.24% a |
| 3 | 99.08% ± 0.55% a | 100.06% ± 0.51% ab | 99.92% ± 0.16% ab | 100.00% ± 0.01% a | 102.65% ± 0.48% a |
| 4 | 99.20% ± 0.43% a | 100.17% ± 0.40% ab | 100.23% ± 0.47% ab | 100.00% ± 0.48% a | 102.57% ± 0.40% a |
| 6 | 99.63% ± 0.37% a | 99.60% ± 0.29% b | 100.70% ± 0.47% a | 100.08% ± 0.32% a | 102.89% ± 0.24% a |
| 8 | 99.63% ± 0.37% a | 99.24% ± 1.52% b | 100.00% ± 0.23% ab | 99.84% ± 0.08% a | 99.36% ± 0.08% d |
| Mass Concentration (mg/mL) | Absorbance (OD700nm) | ||||
|---|---|---|---|---|---|
| VC | PFSI-1 | PFSI-2 | PFSI-3 | PFSI-4 | |
| 0.01 | 0.049 ± 0.001 k | 0.014 ± 0.001 l | 0.060 ± 0.001 l | 0.062 ± 0.002 l | 0.058 ± 0.001 l |
| 0.05 | 0.196 ± 0.002 j | 0.027 ± 0.002 l | 0.068 ± 0.001 l | 0.072 ± 0.001 l | 0.071 ± 0.002 k |
| 0.1 | 0.382 ± 0.005 i | 0.041 ± 0.001 k | 0.083 ± 0.001 k | 0.088 ± 0.001 k | 0.081 ± 0.001 k |
| 0.25 | 0.862 ± 0.021 h | 0.075 ± 0.001 j | 0.121 ± 0.004 j | 0.136 ± 0.001 j | 0.118 ± 0.001 j |
| 0.5 | 1.654 ± 0.045 e | 0.135 ± 0.002 i | 0.166 ± 0.001 i | 0.202 ± 0.002 i | 0.175 ± 0.001 i |
| 0.75 | 1.713 ± 0.030 f | 0.185 ± 0.001 h | 0.218 ± 0.005 h | 0.277 ± 0.004 h | 0.222 ± 0.002 h |
| 1 | 1.731 ± 0.020 ef | 0.243 ± 0.005 g | 0.263 ± 0.001 g | 0.328 ± 0.004 g | 0.272 ± 0.004 g |
| 1.5 | 1.762 ± 0.030 cd | 0.329 ± 0.008 f | 0.460 ± 0.003 f | 0.398 ± 0.001 f | 0.298 ± 0.004 f |
| 2 | 1.772 ± 0.022 bc | 0.419 ± 0.024 e | 0.558 ± 0.010 e | 0.487 ± 0.009 e | 0.379 ± 0.003 e |
| 3 | 1.803 ± 0.043 ab | 0.605 ± 0.006 d | 0.710 ± 0.014 d | 0.597 ± 0.001 d | 0.480 ± 0.007 d |
| 4 | 1.811 ± 0.015 a | 0.751 ± 0.012 c | 1.084 ± 0.015 c | 0.731 ± 0.030 c | 0.569 ± 0.013 c |
| 6 | 1.792 ± 0.023 ab | 1.215 ± 0.026 b | 1.274 ± 0.013 b | 1.249 ± 0.020 b | 0.734 ± 0.017 b |
| 8 | 1.748 ± 0.019 a | 1.566 ± 0.025 a | 1.311 ± 0.012 a | 1.270 ± 0.008 a | 0.836 ± 0.024 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhong, W.; Yang, C.; Zhang, Y.; Yang, D. Effects of Different Deproteinization Methods on the Antioxidant Activity of Polysaccharides from Flos Sophorae Immaturus Obtained by Ultrasonic Microwave Synergistic Extraction. Agronomy 2022, 12, 2740. https://doi.org/10.3390/agronomy12112740
Zhong W, Yang C, Zhang Y, Yang D. Effects of Different Deproteinization Methods on the Antioxidant Activity of Polysaccharides from Flos Sophorae Immaturus Obtained by Ultrasonic Microwave Synergistic Extraction. Agronomy. 2022; 12(11):2740. https://doi.org/10.3390/agronomy12112740
Chicago/Turabian StyleZhong, Wenting, Chunmiao Yang, Yongze Zhang, and Dongsheng Yang. 2022. "Effects of Different Deproteinization Methods on the Antioxidant Activity of Polysaccharides from Flos Sophorae Immaturus Obtained by Ultrasonic Microwave Synergistic Extraction" Agronomy 12, no. 11: 2740. https://doi.org/10.3390/agronomy12112740
APA StyleZhong, W., Yang, C., Zhang, Y., & Yang, D. (2022). Effects of Different Deproteinization Methods on the Antioxidant Activity of Polysaccharides from Flos Sophorae Immaturus Obtained by Ultrasonic Microwave Synergistic Extraction. Agronomy, 12(11), 2740. https://doi.org/10.3390/agronomy12112740








