An Organic Fertilizer ‘Doped’ with a Bacillus Strain Improves Melon and Pepper Yield, Modifying the Rhizosphere Microbiome with Negligible Changes in the Bulk Soil Microbiome
Abstract
1. Introduction
2. Materials and Methods
2.1. Doped Compost Production and Component Description
2.2. Field Trial Design
2.3. Sampling, Variables Measured and Data Analysis
2.4. DNA Extraction, Sequencing and Data Analysis
3. Results
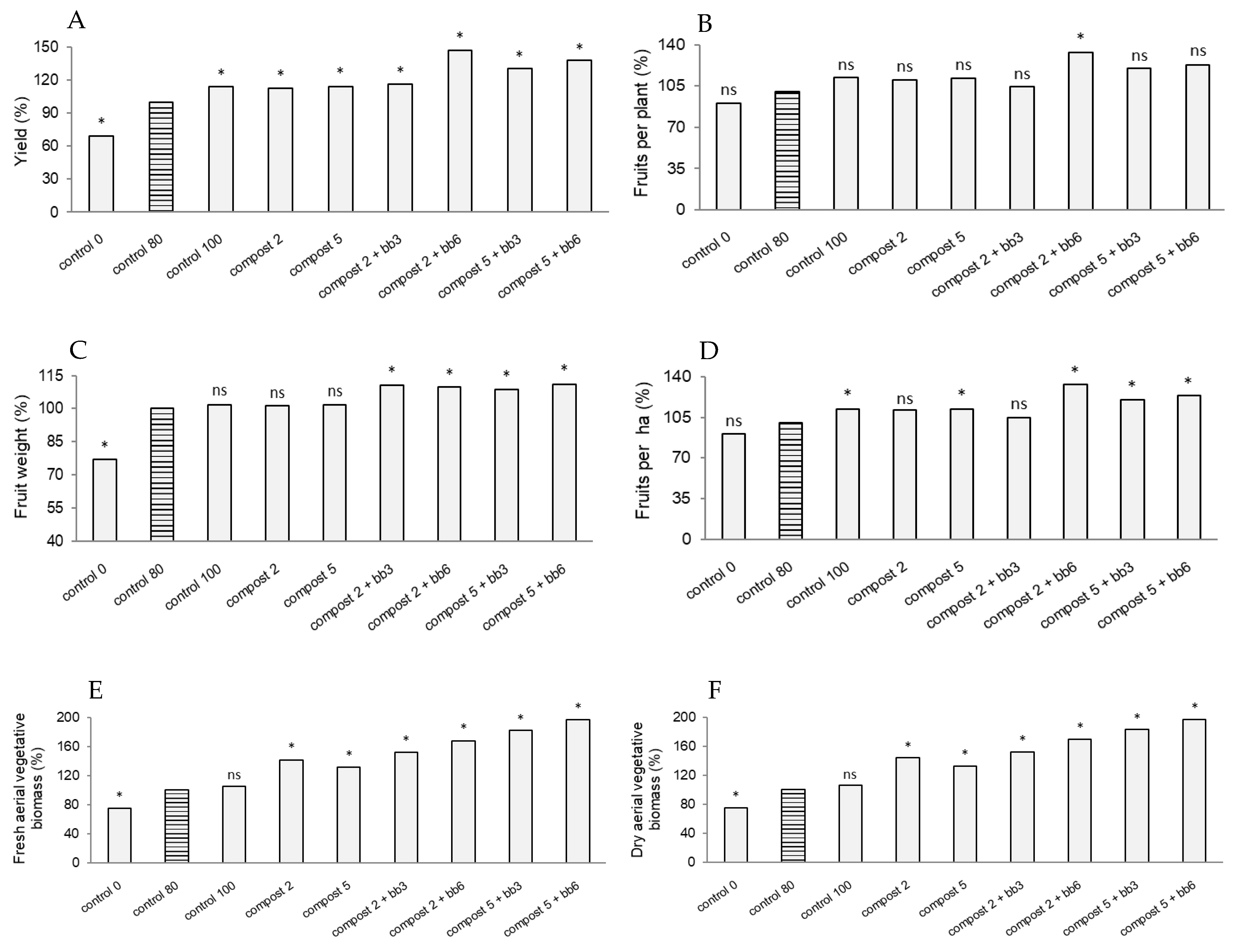
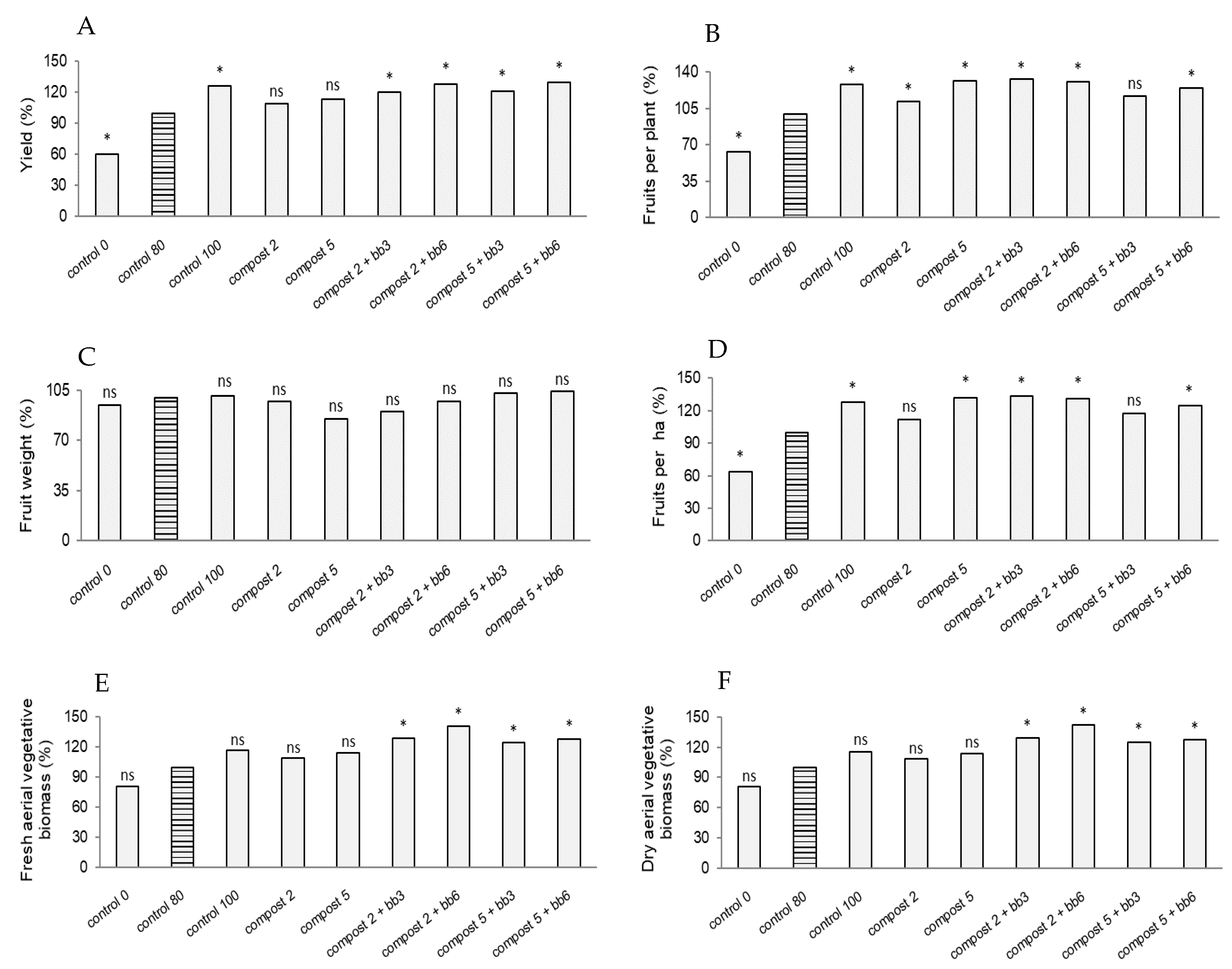
3.1. Yield, Yield Components and Biomass Production
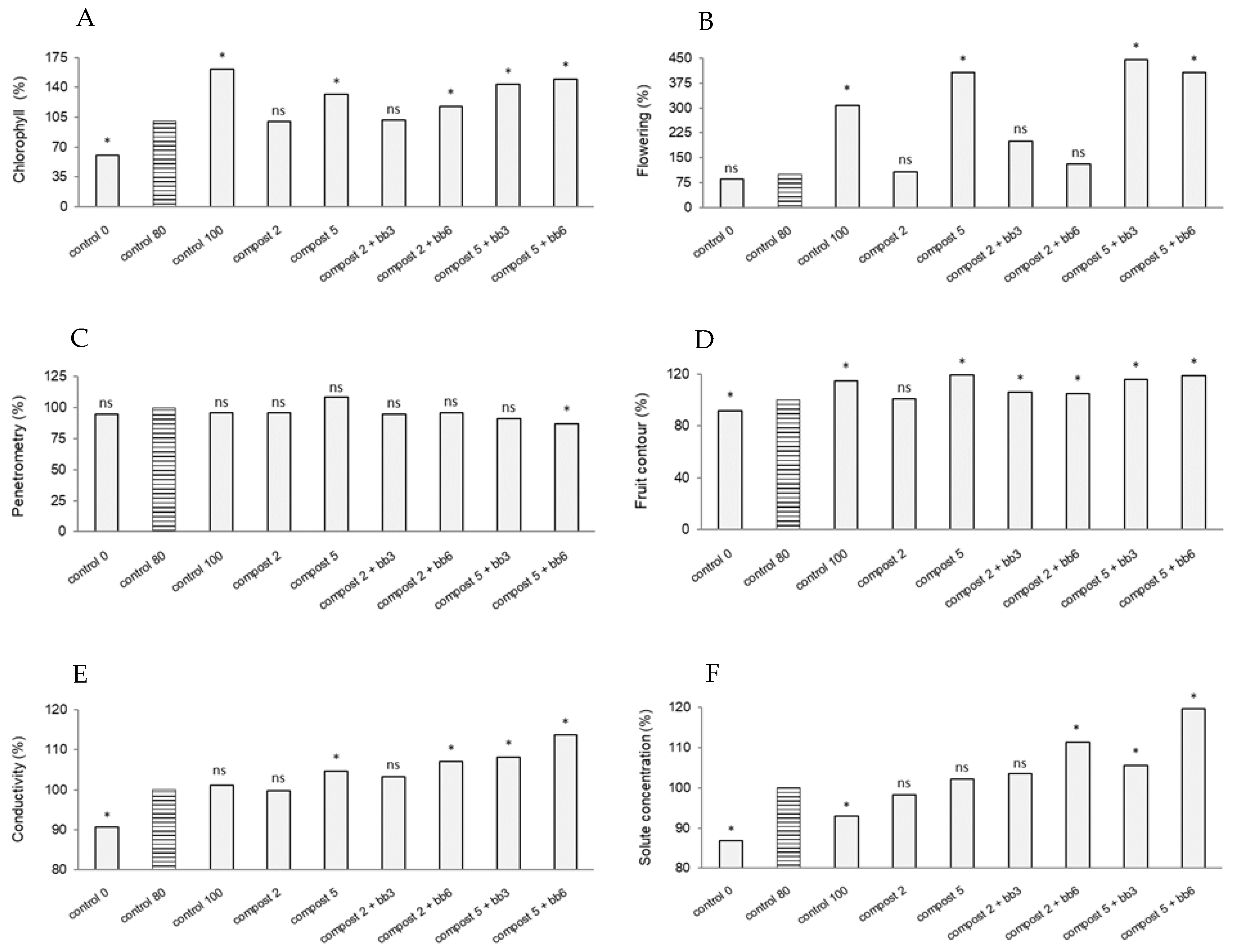
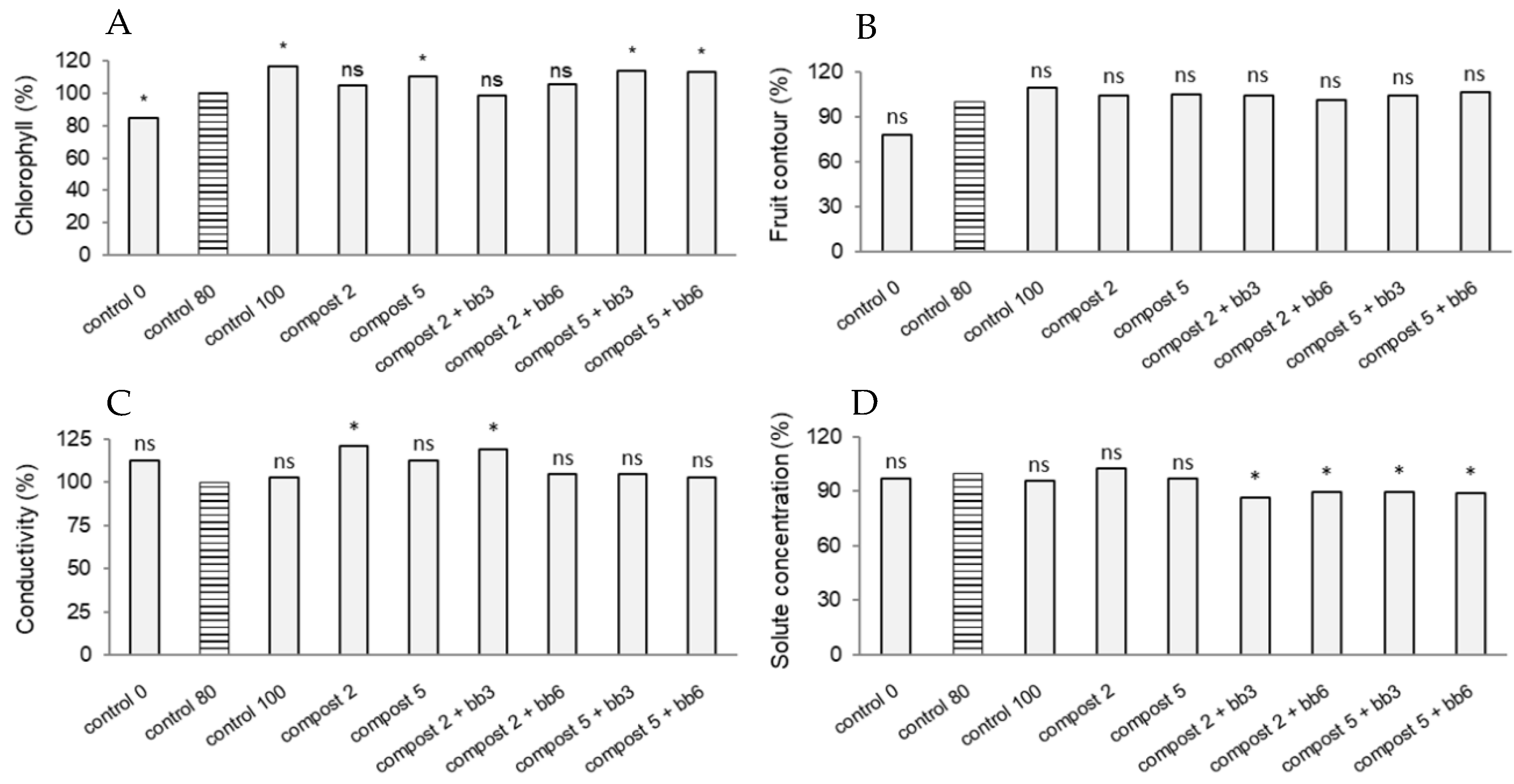
3.2. Chlorophyll Content, Flowering and Fruit Parameters
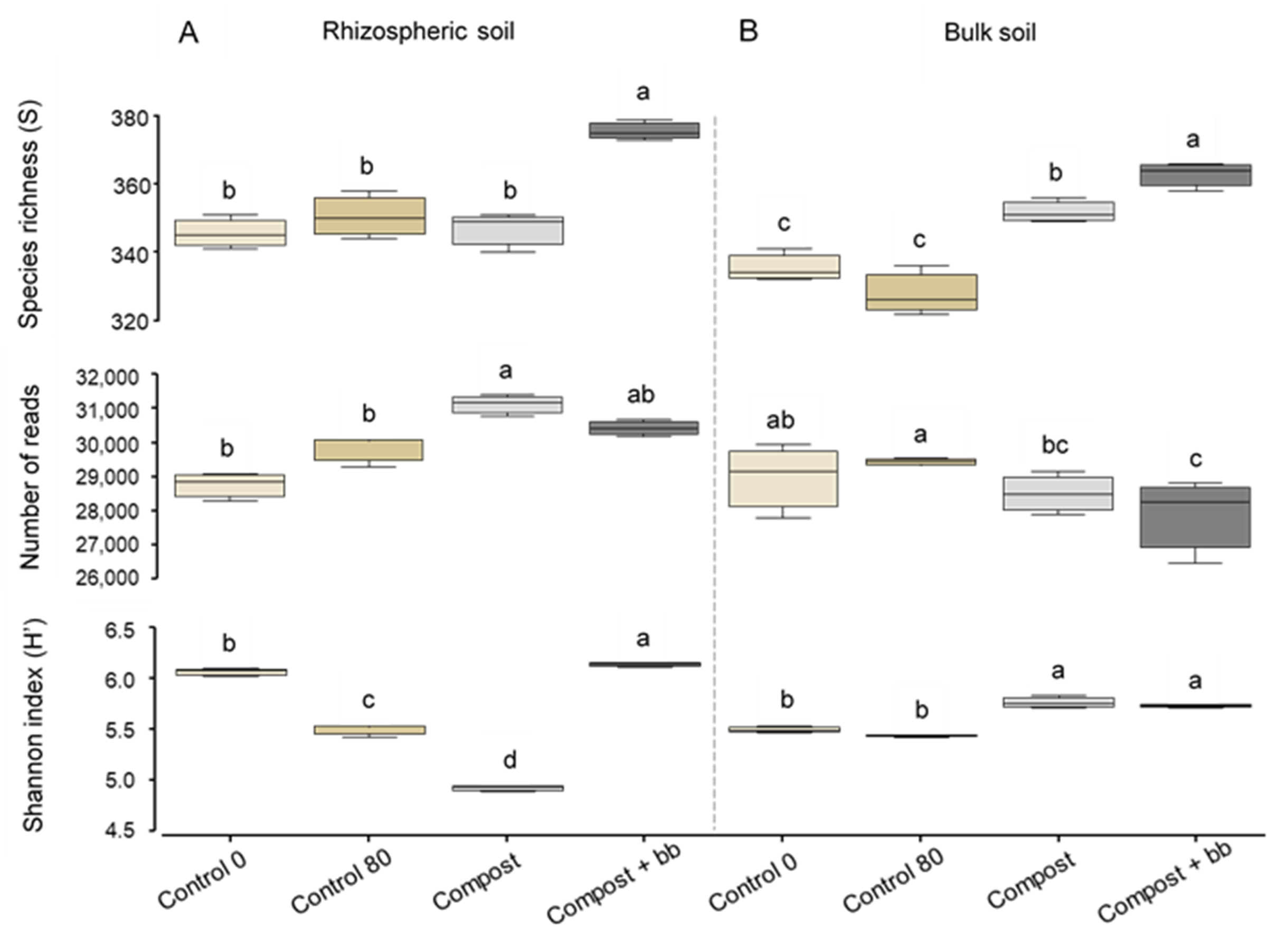
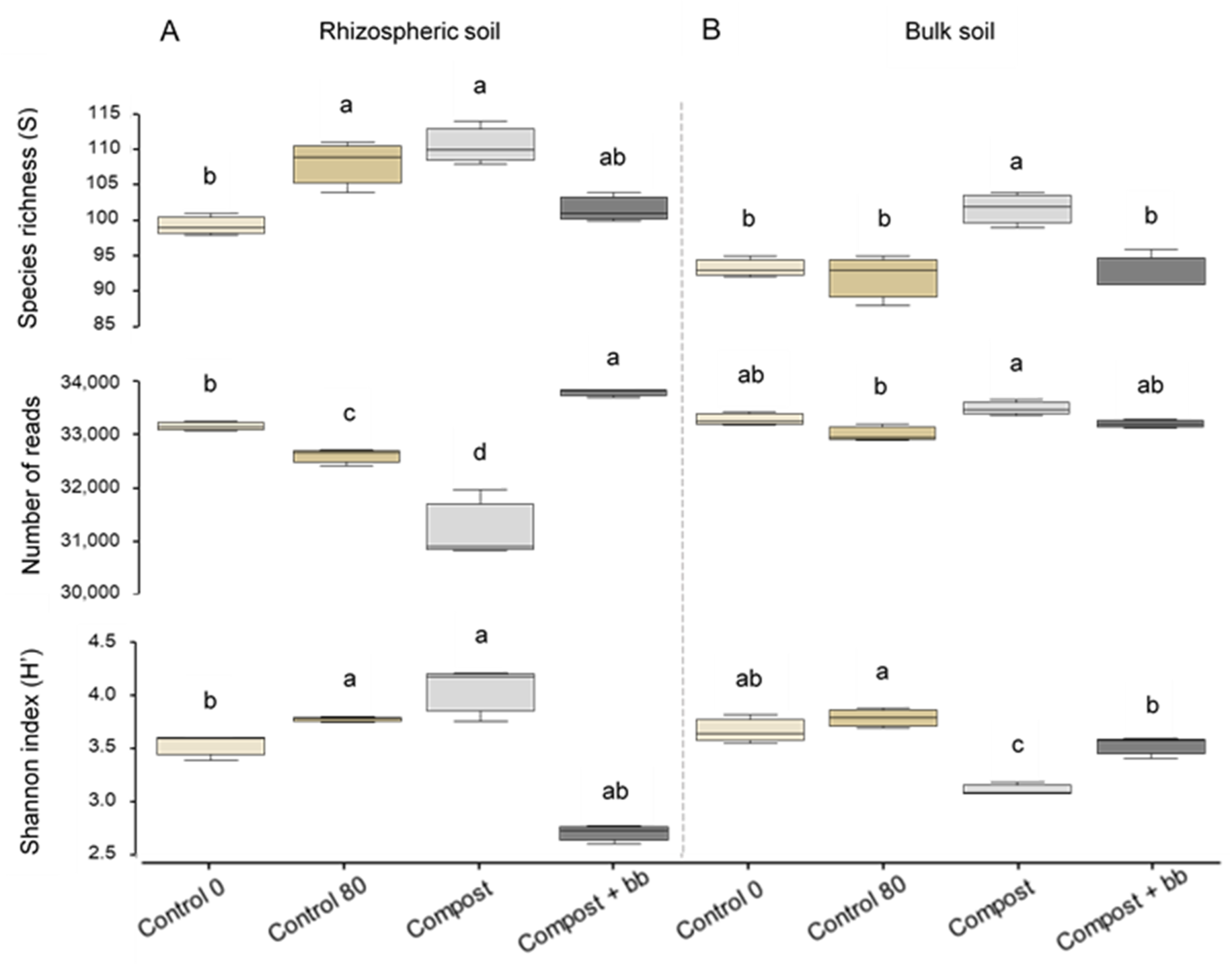
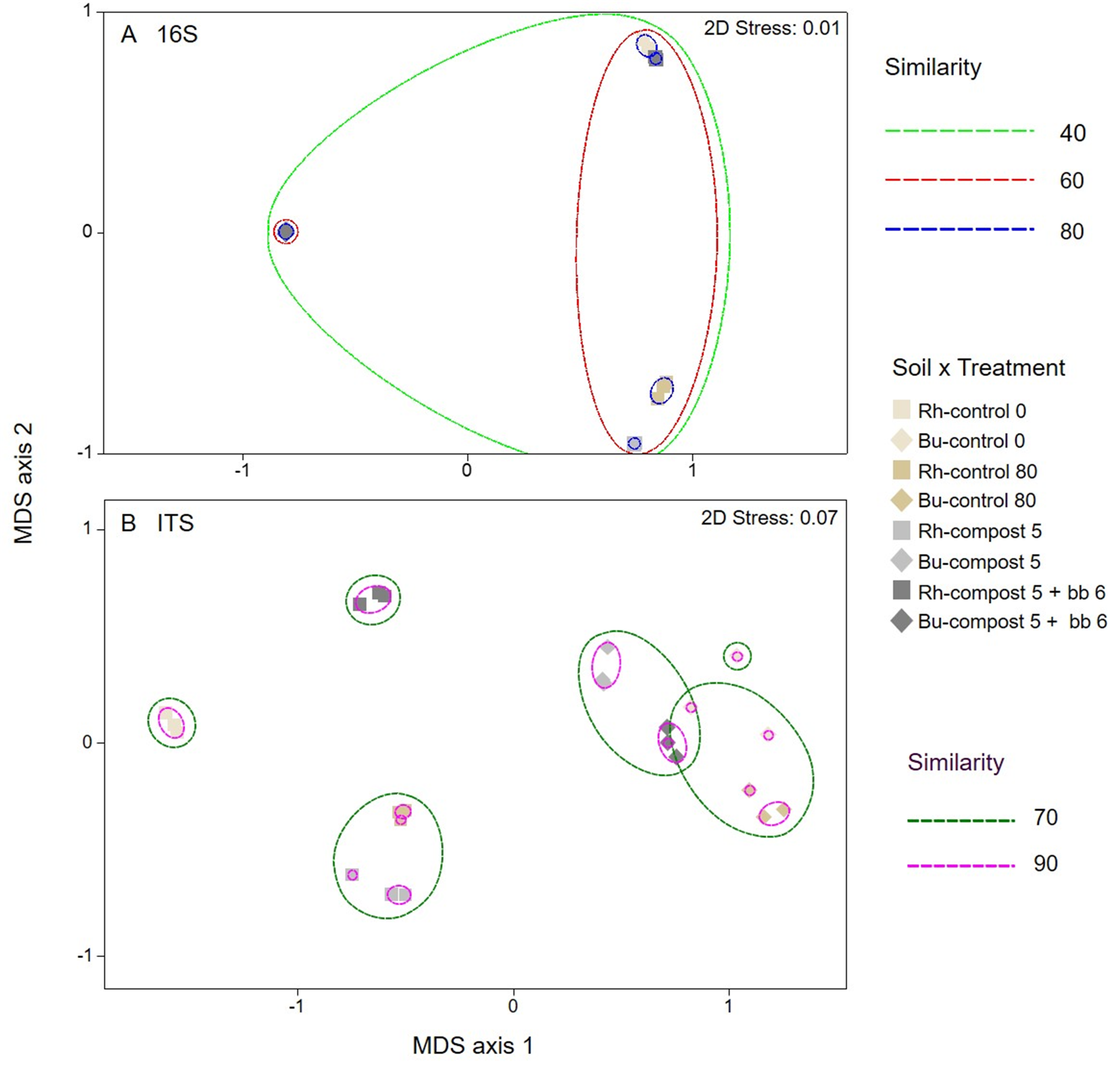
3.3. Bacterial and Fungal Diversity in Soil
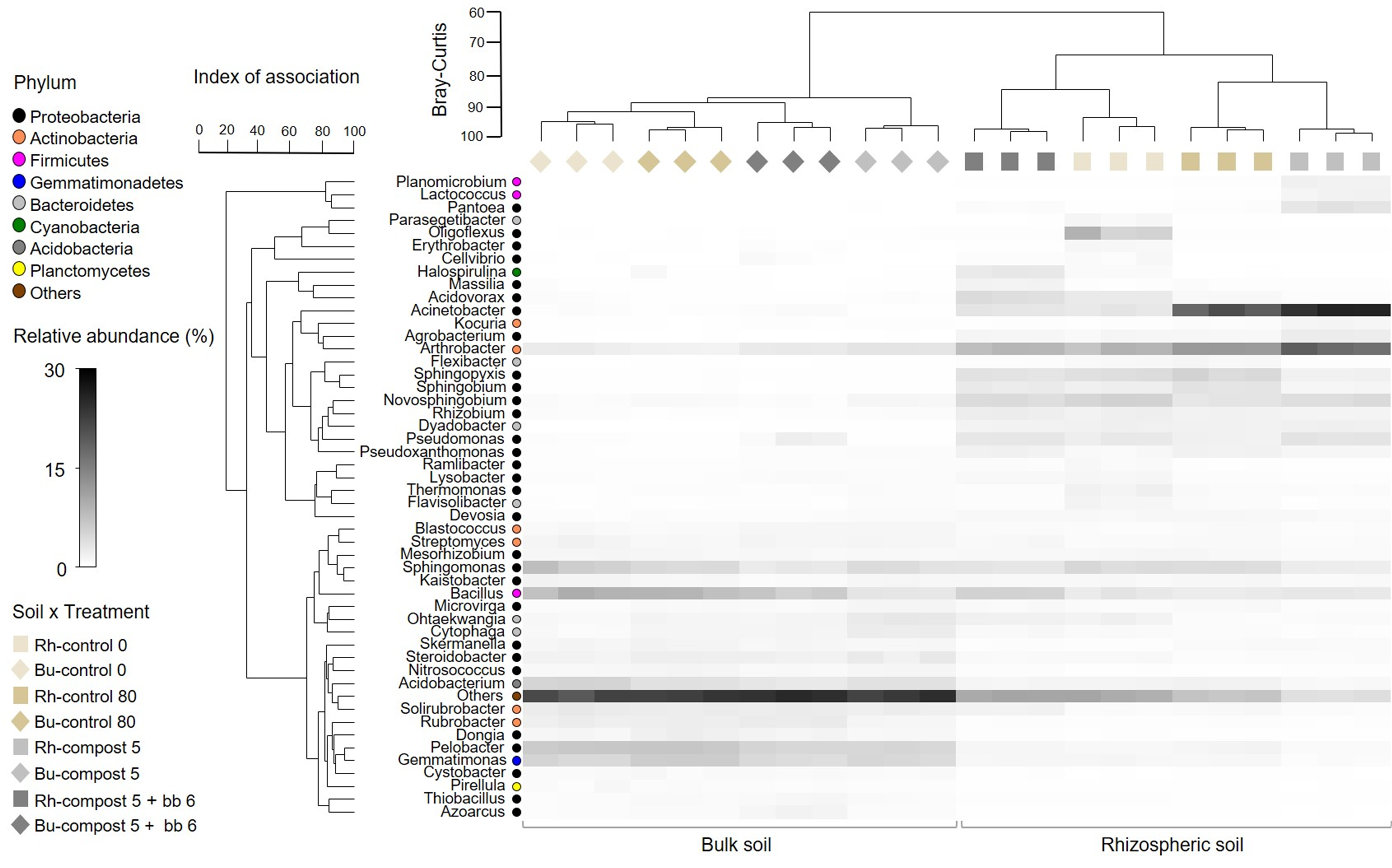
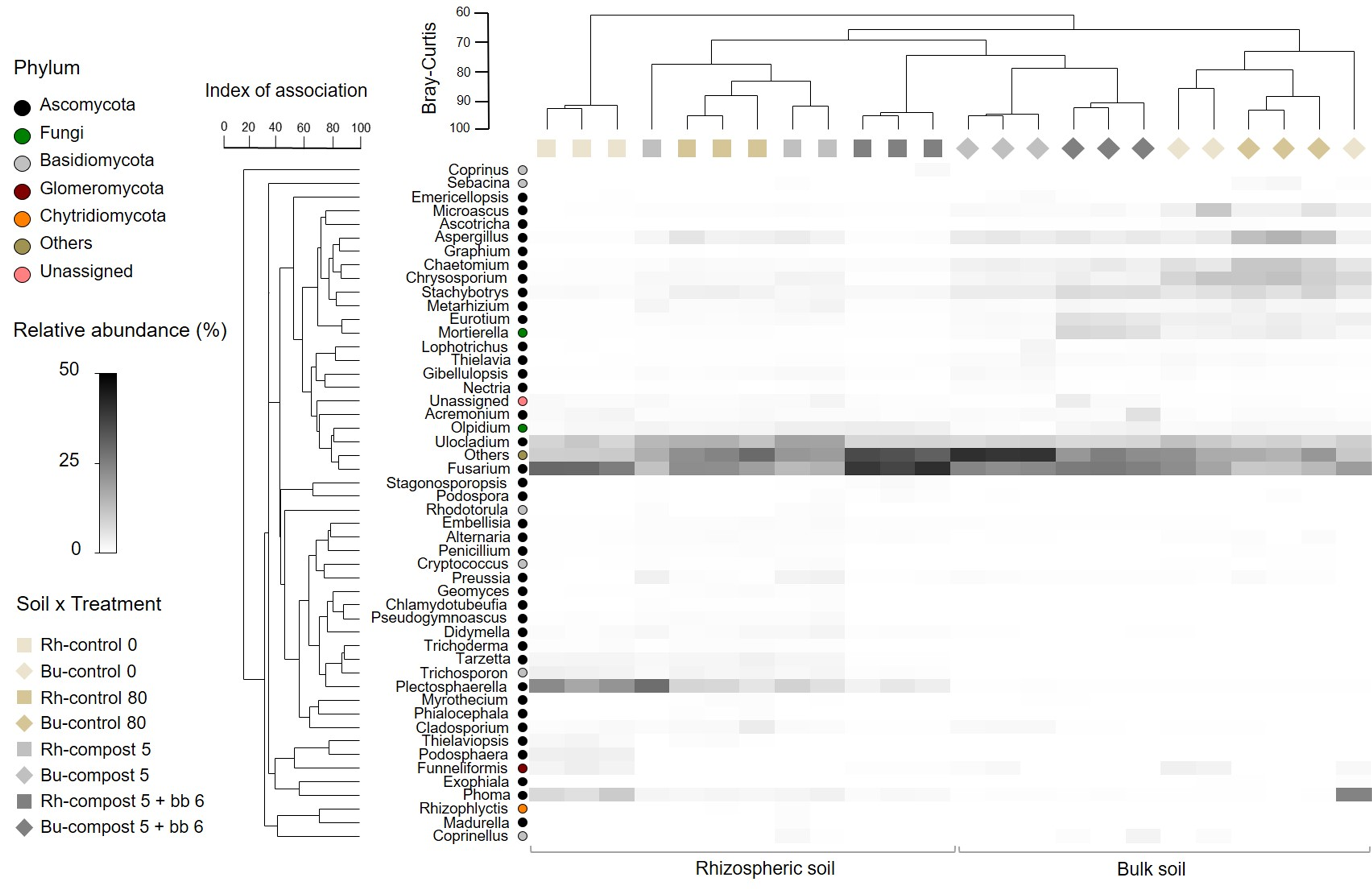
3.4. Composition of Bacterial and Fungal Soil Communities
4. Discussion
4.1. Yield, Yield Components and Biomass Production
4.2. Chlorophyll Content, Flowering and Fruit Parameters
4.3. Soil Microbiome: Bacteria
4.4. Soil Microbiome: Fungi
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Poveda, J. Insect Frass in the Development of Sustainable Agriculture. A Review. Agron. Sustain. Dev. 2021, 41, 5. [Google Scholar] [CrossRef]
- FAO. World Food and Agriculture-Statistical Pocketbook; FAO: Rome, Italy, 2018. [Google Scholar]
- Barea, J.M. Future Challenges and Perspectives for Applying Microbial Biotechnology in Sustainable Agriculture Based on a Better Understanding of Plant-Microbiome Interactions. J. Soil Sci. Plant Nutr. 2015, 15, 261–282. [Google Scholar] [CrossRef]
- Backer, R.; Rokem, J.S.; Ilangumaran, G.; Lamont, J.; Praslickova, D.; Ricci, E.; Subramanian, S.; Smith, D.L. Plant Growth-Promoting Rhizobacteria: Context, Mechanisms of Action, and Roadmap to Commercialization of Biostimulants for Sustainable Agriculture. Front. Plant Sci. 2018, 9, 1473. [Google Scholar] [CrossRef] [PubMed]
- Wezel, A.; Goris, M.; Bruil, J.; Félix, G.F.; Peeters, A.; Bàrberi, P.; Bellon, S.; Migliorini, P. Challenges and Action Points to Amplify Agroecology in Europe. Sustainability 2018, 10, 1598. [Google Scholar] [CrossRef]
- Besset-Manzoni, Y.; Rieusset, L.; Joly, P.; Comte, G.; Prigent-Combaret, C. Exploiting Rhizosphere Microbial Cooperation for Developing Sustainable Agriculture Strategies. Environ. Sci. Pollut. Res. 2018, 25, 29953–29970. [Google Scholar] [CrossRef]
- Cong, R.G.; Thomsen, M. Review of Ecosystem Services in a Bio-Based Circular Economy and Governance Mechanisms. Ecosyst. Ser. 2021, 50, 101298. [Google Scholar] [CrossRef]
- Barquero, M.; Pastor-Buies, R.; Urbano, B.; González-Andrés, F. Challenges, Regulations and Future Actions in Biofertilizers in the European Agriculture: From the Lab to the Field. In Microbial Probiotics for Agricultural Systems. Sustainability in Plant and Crop Protection; Zúñiga-Dávila, D., González-Andrés, F., Ormeño-Orrillo, E., Eds.; Springer: Cham, Switzerland, 2019; pp. 83–107. [Google Scholar] [CrossRef]
- Gutierrez-Albanchez, E.; García-Villaraco, A.; Lucas, J.A.; Horche, I.; Ramos-Solano, B.; Gutierrez-Mañero, F.J. Pseudomonas palmensis Sp. Nov., a Novel Bacterium Isolated From Nicotiana Glauca Microbiome: Draft Genome Analysis and Biological Potential for Agriculture. Front. Microbiol. 2021, 12, 1–15. [Google Scholar] [CrossRef]
- Poveda, J.; González-Andrés, F. Bacillus as a Source of Phytohormones for Use in Agriculture. Appl. Microbiol. Biotechnol. 2021, 105, 8629–8645. [Google Scholar] [CrossRef]
- Gamalero, E.; Glick, B.R. Plant Growth-Promoting Bacteria in Agriculture and Stressed Environments. In Modern Soil Microbiology; van Elsas, J.D., Trevors, J.T., Eds.; CRC Press: Boca Raton, FL, USA, 2019; pp. 361–380. [Google Scholar]
- Glick, B.R. Introduction to Plant Growth-Promoting Bacteria. In Beneficial Plant-Bacterial Interactions; Glick, B.R., Ed.; Springer: Cham, Switzerland, 2020; p. 383. [Google Scholar] [CrossRef]
- Ngalimat, M.; Yahaya, R.; Baharudin, M.; Yaminudin, S.; Karim, M.; Ahmad, S.; Sabri, S. A Review on the Biotechnological Applications of the Operational Group Bacillus Amyloliquefaciens. Microorganisms 2021, 9, 614. [Google Scholar] [CrossRef]
- European Council Regulation (EU) 2019/1009 of the European Parliament and of the Council of 5 June 2019 Laying Down Rules on the Making Available on the Market of EU Fertilising Products and Amending Regulations (EC) No 1069/2009 and (EC) No 1107/2009 and Repealing Regulation (EC) No 2003/2003. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex%3A32019R1009 (accessed on 17 July 2022).
- Yadav, A.N. Plant Microbiomes for Sustainable Agriculture: Current Research and Future Challenges. In Plant Microbiomes for Sustainable Agriculture; Yadav, A.N., Singh, J., Rastegari, A., Yadav, N., Eds.; Springer: Cham, Switzerland, 2020; pp. 475–482. [Google Scholar]
- Pastor-Bueis, R.; Mulas, R.; Gómez, X.; González-Andrés, F. Innovative Liquid Formulation of Digestates for Producing a Biofertilizer Based on Bacillus siamensis: Field Testing on Sweet Pepper. J. Plant Nutr. Soil Sci. 2017, 180, 748–758. [Google Scholar] [CrossRef]
- Thapa, R.; Tully, K.L.; Cabrera, M.L.; Dann, C.; Schomberg, H.H.; Timlin, D.; Reberg-Horton, C.; Gaskin, J.; Davis, B.W.; Mirsky, S.B. Effects of Moisture and Temperature on C and N Mineralization from Surface-Applied Cover Crop Residues. Biol. Fertil. Soils 2021, 57, 485–498. [Google Scholar] [CrossRef]
- Cabrera, M.L.; Kissel, D.E.; Vigil, M.F. Nitrogen Mineralization from Organic Residues. J. Environ. Qual. 2005, 34, 75–79. [Google Scholar] [CrossRef] [PubMed]
- Joshi, D.R.; Clay, D.E.; Clay, S.A.; Smart, A.J. Seasonal Losses of Surface Litter in Northern Great Plains Mixed-Grass Prairies. Rangel. Ecol. Manag. 2020, 73, 259–264. [Google Scholar] [CrossRef]
- Poffenbarger, H.J.; Mirsky, S.B.; Weil, R.R.; Kramer, M.; Spargo, J.T.; Cavigelli, M.A.; Poff, H.J.; Mirsky, S.B.; Cavigelli, M.A.; Spargo, J.T. Legume Proportion, Poultry Litter, and Tillage Effects on Cover Crop Decomposition. Agron. J. 2015, 107, 2083–2096. [Google Scholar] [CrossRef]
- Mazzoleni, S.; Bonanomi, G.; Incerti, G.; Chiusano, M.L.; Termolino, P.; Mingo, A.; Senatore, M.; Giannino, F.; Cartenì, F.; Rietkerk, M.; et al. Inhibitory and Toxic Effects of Extracellular Self-DNA in Litter: A Mechanism for Negative Plant-Soil Feedbacks? New Phytol. 2015, 205, 1195–1210. [Google Scholar] [CrossRef]
- Bonanomi, G.; Lorito, M.; Vinale, F.; Woo, S.L. Organic Amendments, Beneficial Microbes, and Soil Microbiota: Toward a Unified Framework for Disease Suppression. Annu. Rev. Phytopathol. 2018, 56, 1–20. [Google Scholar] [CrossRef]
- De Corato, U. Sustainability Editorial Towards New Soil Management Strategies for Improving Soil Quality and Ecosystem Services in Sustainable Agriculture: Editorial Overview. Sustainability 2020, 12, 9398. [Google Scholar] [CrossRef]
- Arif, M.S.; Shahzad, S.M.; Riaz, M.; Yasmeen, T.; Shahzad, T.; Akhtar, M.J.; Bragazza, L.; Buttler, A. Nitrogen-Enriched Compost Application Combined with Plant Growth-Promoting Rhizobacteria (PGPR) Improves Seed Quality and Nutrient Use Efficiency of Sunflower. J. Plant Nutr. Soil Sci. 2017, 180, 464–473. [Google Scholar] [CrossRef]
- Biederman, L.A.; Stanley Harpole, W. Biochar and Its Effects on Plant Productivity and Nutrient Cycling: A Meta-Analysis. GCB Bioenergy 2013, 5, 202–214. [Google Scholar] [CrossRef]
- Jeffery, S.; Abalos, D.; Prodana, M.; Bastos, A.C.; van Groenigen, J.W.; Hungate, B.A.; Verheijen, F. Biochar Boosts Tropical but Not Temperate Crop Yields. Environ. Res. Lett. 2017, 12, 053001. [Google Scholar] [CrossRef]
- Tan, G.; Wang, H.; Xu, N.; Liu, H.; Zhai, L. Biochar Amendment with Fertilizers Increases Peanut N Uptake, Alleviates Soil N 2 O Emissions without Affecting NH 3 Volatilization in Field Experiments. Environ. Sci. Pollut. Res. 2018, 25, 8817–8826. [Google Scholar] [CrossRef]
- Kalu, S.; Oyekoya, G.N.; Ambus, P.; Tammeorg, P.; Simojoki, A.; Pihlatie, M.; Karhu, K. Effects of Two Wood-Based Biochars on the Fate of Added Fertilizer Nitrogen—A 15N Tracing Study. Biol. Fertil. Soils 2021, 57, 457–470. [Google Scholar] [CrossRef]
- Glaser, B.; Parr, M.; Braun, C.; Biochar, K.G. Is Carbon Negative. Nat. Geosci. 2009, 2, 2. [Google Scholar] [CrossRef]
- Singh, B.P.; Cowie, A.L.; Smernik, R.J. Biochar Carbon Stability in a Clayey Soil as a Function of Feedstock and Pyrolysis Temperature. Environ. Sci. Technol. 2012, 46, 11770–11778. [Google Scholar] [CrossRef] [PubMed]
- Borchard, N.; Schirrmann, M.; Cayuela, M.L.; Kammann, C.; Wrage-Mönnig, N.; Estavillo, J.M.; Fuertes-Mendizábal, T.; Sigua, G.; Spokas, K.; Ippolito, J.A.; et al. Biochar, Soil and Land-Use Interactions That Reduce Nitrate Leaching and N2O Emissions: A Meta-Analysis. Sci. Total Environ. 2019, 651, 2354–2364. [Google Scholar] [CrossRef]
- Nguyen, T.T.N.; Xu, C.Y.; Tahmasbian, I.; Che, R.; Xu, Z.; Zhou, X.; Wallace, H.M.; Bai, S.H. Effects of Biochar on Soil Available Inorganic Nitrogen: A Review and Meta-Analysis. Geoderma 2017, 288, 79–96. [Google Scholar] [CrossRef]
- Hagemann, N.; Kammann, C.I.; Schmidt, H.P.; Kappler, A.; Behrens, S. Nitrate Capture and Slow Release in Biochar Amended Compost and Soil. PLoS ONE 2017, 12, 1–16. [Google Scholar] [CrossRef]
- Ye, J.; Zhang, R.; Nielsen, S.; Joseph, S.D.; Huang, D.; Thomas, T. A Combination of Biochar-Mineral Complexes and Compost Improves Soil Bacterial Processes, Soil Quality, and Plant Properties. Front. Microbiol. 2016, 7, 372. [Google Scholar] [CrossRef]
- Araujo, J.; Díaz-Alcántara, C.A.; Urbano, B.; González-Andrés, F. Inoculation with Native Bradyrhizobium Strains Formulated with Biochar as Carrier Improves the Performance of Pigeonpea (Cajanus cajan L.). Eur. J. Agron. 2020, 113, 125985. [Google Scholar] [CrossRef]
- Pastor-Bueis, R.; Sánchez-Cañizares, C.; James, E.K.; González-Andrés, F. Formulation of a Highly Effective Inoculant for Common Bean Based on an Autochthonous Elite Strain of Rhizobium leguminosarum bv. phaseoli, and Genomic-Based Insights Into Its Agronomic Performance. Front. Microbiol. 2019, 10, 1–16. [Google Scholar] [CrossRef]
- Faostat. Available online: http://www.fao.org/faostat/en/#home (accessed on 28 February 2022).
- Barquero, M. Caracterización y Selección de Bacterias y Hongos Micorrícicos Aislados En Raíces de Alubia y Pimiento En La Provincia de León, Para El Desarrollo de Biofertilizantes; Universidad de Salamanca: Salamanca, Spain, 2014. [Google Scholar]
- Rosas, J.G.; Gómez, N.; Cara, J.; Ubalde, J.; Sort, X.; Sánchez, M.E. Assessment of Sustainable Biochar Production for Carbon Abatement from Vineyard Residues. J. Anal. Appl. Pyrolysis 2015, 113, 239–247. [Google Scholar] [CrossRef]
- Urbano-Terrón, P. Tratado De Fitotecnia General, 2nd ed.; Mundiprensa: Madrid, Spain, 2008. [Google Scholar]
- Feller, C.; Bleiholder, H.; Buhr, L.; Hack, H.; He, M.; Klose, R.; Meier, U.; Stau, R.; van den Boom, T.; Weber, E. Phenological Growth Stages of Vegetable Crops II. Fruit Vegetables and Pulses. Nachrichtenbl. Dtsch. Pflanzenschutzdienst. 1995, 47, 217–232. [Google Scholar]
- Clarke, K.R.; Gorley, R.N. Getting Started with PRIMER V7, 1st ed.; Primer-E: Plymouth, UK, 2015. [Google Scholar]
- Joo, G.J.; Kim, Y.M.; Lee, I.J.; Song, K.S.; Rhee, I.K. Growth Promotion of Red Pepper Plug Seedlings and the Production of Gibberellins by Bacillus cereus, Bacillus macroides and Bacillus pumilus. Biotechnol. Lett. 2004, 26, 487–491. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Ruan, Y.; Wang, B.; Zhang, J.; Waseem, R.; Huang, Q.; Shen, Q. Plant Growth-Promoting Rhizobacteria Strain Bacillus amyloliquefaciens Njn-6-Enriched Bio-Organic Fertilizer Suppressed Fusarium Wilt and Promoted the Growth of Banana Plants. J. Agric. Food Chem. 2013, 61, 3774–3780. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.J.; Radhakrishnan, R.; Kang, S.M.; You, Y.H.; Jeong, E.J.; Kim, J.G.; Lee, I.J. Plant Growth Promontory Effect of Bacillus amyloliquefaciens H-2-5 on Crop Plants and Influence on Physiological Changes in Soybean under Soil Salinity. Physiol. Mol. Biol. Plants 2017, 23, 571–580. [Google Scholar] [CrossRef] [PubMed]
- Vicente-Hernández, A.; Salgado-Garciglia, R.; Valencia-Cantero, E.; Ramírez-Ordorica, A.; Hernández-García, A.; García-Juárez, P.; Macías-Rodríguez, L. Bacillus methylotrophicus M4-96 Stimulates the Growth of Strawberry (Fragaria × Ananassa ‘Aromas’) Plants In Vitro and Slows Botrytis cinerea Infection by Two Different Methods of Interaction. J. Plant Growth Regul. 2019, 38, 765–777. [Google Scholar] [CrossRef]
- Wang, X.; Xie, H.; Ku, Y.; Yang, X.; Chen, Y.; Yang, N.; Mei, X.; Cao, C. Chemotaxis of Bacillus cereus YL6 and Its Colonization of Chinese Cabbage Seedlings. Plant Soil 2020, 447, 413–430. [Google Scholar] [CrossRef]
- Ambawade, M.; Pathade, G. Production of Gibberellic Acid by Bacillus siamensis BE 76 Isolated from Banana Plant (Musa spp.). Int. J. Sci. Res. 2015, 4, 394–398. [Google Scholar]
- Idris, E.S.E.; Iglesias, D.J.; Talon, M.; Borriss, R. Tryptophan-Dependent Production of Indole-3-Acetic Acid (IAA) Affects Level of Plant Growth Promotion by Bacillus amyloliquefaciens FZB42. Mol. Plant Microbe Interact. 2007, 20, 619–626. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, L.; Zhang, N.; Li, Z.; Zhang, G.; Xu, Y.; Shen, Q.; Zhang, R. Plant-Microbe Communication Enhances Auxin Biosynthesis by a Root-Associated Bacterium, Bacillus amyloliquefaciens SQR9. Mol. Plant Microbe Interact. 2016, 29, 324–330. [Google Scholar] [CrossRef]
- Ji, C.; Tian, H.; Wang, X.; Hao, L.; Wang, C.; Zhou, Y.; Xu, R.; Song, X.; Liu, Y.; Du, J.; et al. Bacillus subtilis HG-15, a Halotolerant Rhizoplane Bacterium, Promotes Growth and Salinity Tolerance in Wheat (Triticum aestivum). BioMed Res. Int. 2020, 2022, 1–16. [Google Scholar] [CrossRef]
- Lim, J.H. Synergistic Plant Growth Promotion by the Indigenous Auxins-Producing PGPR Bacillus subtilis AH18 and Bacillus licheniforims K11. J. Korean Soc. Appl. Biol. Chem. 2009, 52, 531–538. [Google Scholar] [CrossRef]
- Kudoyarova, G.R.; Melentiev, A.I.; Martynenko, E.V.; Timergalina, L.N.; Arkhipova, T.N.; Shendel, G.V.; Kuz’mina, L.Y.; Dodd, I.C.; Veselov, S.Y. Cytokinin Producing Bacteria Stimulate Amino Acid Deposition by Wheat Roots. Plant Physiol. Biochem. 2014, 83, 285–291. [Google Scholar] [CrossRef]
- Qian, L.; Chen, L.; Joseph, S.; Pan, G.; Li, L.; Zheng, J.; Zhang, X.; Zheng, J.; Yu, X.; Wang, J. Biochar Compound Fertilizer as an Option to Reach High Productivity but Low Carbon Intensity in Rice Agriculture of China. Carbon Manage. 2014, 5, 145–154. [Google Scholar] [CrossRef]
- Joseph, S.; Graber, E.R.; Chia, C.; Munroe, P.; Donne, S.; Thomas, T.; Nielsen, S.; Marjo, C.; Rutlidge, H.; Pan, G.X.; et al. Shifting Paradigms: Development of High-Efficiency Biochar Fertilizers Based on Nano-Structures and Soluble Components. Carbon Manage. 2013, 4, 323–343. [Google Scholar] [CrossRef]
- Yao, C.; Joseph, S.; Li, L.; Pan, G.; Lin, Y.; Munroe, P.; Pace, B.; Taherymoosavi, S.; Van zwieten, L.; Thomas, T.; et al. Developing More Effective Enhanced Biochar Fertilisers for Improvement of Pepper Yield and Quality. Pedosphere 2015, 25, 703–712. [Google Scholar] [CrossRef]
- Frimpong, K.A.; Abban-Baidoo, E.; Marschner, B. Can Combined Compost and Biochar Application Improve the Quality of a Highly Weathered Coastal Savanna Soil? Heliyon 2021, 7, e07089. [Google Scholar] [CrossRef]
- Mukhopadhyay, S.; Masto, R.E.; Singh, A.K.; Singh, P.K. Impact of the Combined Application of Biochar and Compost on Mine Soil Quality and Growth of Lady’s Finger (Abelmoschus esculentus). Bull. Environ. Contam. Toxicol. 2020, 108, 396–402. [Google Scholar] [CrossRef]
- Ijaz, M.; Tahir, M.; Shahid, M.; Ul-Allah, S.; Sattar, A.; Sher, A.; Mahmood, K.; Hussain, M. Combined Application of Biochar and PGPR Consortia for Sustainable Production of Wheat under Semiarid Conditions with a Reduced Dose of Synthetic Fertilizer. Braz. J. Microbiol. 2019, 50, 449–458. [Google Scholar] [CrossRef]
- Nadeem, S.M.; Imran, M.; Naveed, M.; Khan, M.Y.; Ahmad, M.; Zahir, Z.A.; Crowley, D.E. Synergistic Use of Biochar, Compost and Plant Growth-Promoting Rhizobacteria for Enhancing Cucumber Growth under Water Deficit Conditions. J. Sci. Food Agric. 2017, 97, 5139–5145. [Google Scholar] [CrossRef]
- Rasool, M.; Akhter, A.; Soja, G.; Haider, M.S. Role of Biochar, Compost and Plant Growth Promoting Rhizobacteria in the Management of Tomato Early Blight Disease. Sci. Rep. 2021, 11, 6092. [Google Scholar] [CrossRef]
- Zafar-Ul-Hye, M.; Tahzeeb-ul-Hassan, M.; Abid, M.; Fahad, S.; Brtnicky, M.; Dokulilova, T.; Datta, R.; Danish, S. Potential Role of Compost Mixed Biochar with Rhizobacteria in Mitigating Lead Toxicity in Spinach. Sci. Rep. 2020, 10, 12159. [Google Scholar] [CrossRef] [PubMed]
- Zafar-Ul-Hye, M.; Tahzeeb-Ul-Hassan, M.; Wahid, A.; Danish, S.; Khan, M.J.; Fahad, S.; Brtnicky, M.; Hussain, G.S.; Leonardo Battaglia, M.; Datta, R. Compost Mixed Fruits and Vegetable Waste Biochar with ACC Deaminase Rhizobacteria Can Minimize Lead Stress in Mint Plants. Sci. Rep. 2021, 11, 6606. [Google Scholar] [CrossRef] [PubMed]
- Samreen, T.; Zahir, Z.A.; Naveed, M.; Asghar, M. Boron Tolerant Phosphorus Solubilizing Bacillus spp. MN-54 Improved Canola Growth in Alkaline Calcareous Soils. Int. J. Agric. Biol. 2019, 21, 538–546. [Google Scholar] [CrossRef]
- Tekaya, M.; El-Gharbi, S.; Mechri, B.; Chehab, H.; Bchir, A.; Chraief, I.; Ayachi, M.; Boujnah, D.; Attia, F.; Hammami, M. Improving Performance of Olive Trees by the Enhancement of Key Physiological Parameters of Olive Leaves in Response to Foliar Fertilization. Acta Physiol. Plant. 2016, 38, 101. [Google Scholar] [CrossRef]
- Akram, M.S.; Ashraf, M. Alleviation of Adverse Effects of Salt Stress on Sunflower (Helianthus annuus L.) by Exogenous Application of Potassium Nitrate. J. Appl. Bot. Food Qual. 2009, 83, 19–27. [Google Scholar]
- Hasnain, M.; Chen, J.; Ahmed, N.; Memon, S.; Wang, L.; Wang, Y.; Wang, P. The Effects of Fertilizer Type and Application Time on Soil Properties, Plant Traits, Yield and Quality of Tomato. Sustainability 2020, 12, 9065. [Google Scholar] [CrossRef]
- Radhakrishnan, R.; Lee, I.J. Gibberellins Producing Bacillus methylotrophicus KE2 Supports Plant Growth and Enhances Nutritional Metabolites and Food Values of Lettuce. Plant Physiol. Biochem. 2016, 109, 181–189. [Google Scholar] [CrossRef]
- Kaymak, H.C. Potential of PGPR in Improvement of Environmental-Friendly Vegetable Production. In Field Crops: Sustainable Management by PGPR; Maheshwari, D., Dheeman, S., Eds.; Springer: Cham, Switzerland, 2019; Volume 23, pp. 221–251. [Google Scholar]
- Glick, B.R.; Gamalero, E. Recent Developments in the Study of Plant Microbiomes. Microorganisms 2021, 9, 1533. [Google Scholar] [CrossRef]
- Schmidt, J.E.; Kent, A.D.; Brisson, V.L.; Gaudin, A.C.M. Agricultural Management and Plant Selection Interactively Affect Rhizosphere Microbial Community Structure and Nitrogen Cycling. Microbiome 2019, 7, 146. [Google Scholar] [CrossRef]
- Jacobs-Hoffman, I.; Hills, P.N. Effects of the Commercial Biostimulant BC204 on the Rhizosphere Microbial Community of Solanum Lycopersicum L. South Afr. J. Bot. 2021, 143, 52–60. [Google Scholar] [CrossRef]
- Thierry, S.; Macarie, H.; Iizuka, T.; Geißdörfer, W.; Assih, E.A.; Spanevello, M.; Verhe, F.; Thomas, P.; Fudou, R.; Monroy, O.; et al. Pseudoxanthomonas mexicana Sp. Nov. and Pseudoxanthomonas japonensis Sp. Nov., Isolated from Diverse Environments, and Emended Descriptions of the Genus Pseudoxanthomonas Finkmann et al. 2000 and of Its Type Species. Int. J. Syst. Evol. Microbiol. 2004, 54, 2245–2255. [Google Scholar] [CrossRef] [PubMed]
- Nayaka, S.; Chakraborty, B.; Swamy, P.S.; Bhat, M.P.; Airodagi, D.; Basavarajappa, D.S.; Rudrappa, M.; Hiremath, H.; Nagaraja, S.K.; Madhappa, C. Isolation, Characterization, and Functional Groups Analysis of Pseudoxanthomonas Indica RSA-23 from Rhizosphere Soil. J. Appl. Pharm. Sci. 2019, 9, 101–106. [Google Scholar] [CrossRef]
- Ofek, M.; Hadar, Y.; Minz, D. Ecology of Root Colonizing Massilia (Oxalobacteraceae). PLoS ONE 2012, 7, e40117. [Google Scholar] [CrossRef]
- Hayat, R.; Ali, S.; Amara, U.; Khalid, R.; Ahmed, I. Soil Beneficial Bacteria and Their Role in Plant Growth Promotion: A Review. Ann. Microbiol. 2010, 60, 579–598. [Google Scholar] [CrossRef]
- Vargas, L.K.; Volpiano, C.G.; Lisboa, B.B.; Giongo, A.; Beneduzi, A.; Passaglia, L.M.P. Potential of Rhizobia as Plant Growth-Promoting Rhizobacteria. In Microbes for Legume Improvement, 2nd ed.; Khan, M.S., Musarrat, J., Zaidi, A., Eds.; Springer: Vienna, Austria, 2017; pp. 153–174. [Google Scholar] [CrossRef]
- Zhao, Q.; Yue, S.; Bilal, M.; Hu, H.; Wang, W.; Zhang, X. Comparative Genomic Analysis of 26 Sphingomonas and Sphingobium Strains: Dissemination of Bioremediation Capabilities, Biodegradation Potential and Horizontal Gene Transfer. Sci. Total Environ. 2017, 609, 1238–1247. [Google Scholar] [CrossRef]
- Sharma, M.; Khurana, H.; Singh, D.N.; Negi, R.K. The Genus Sphingopyxis: Systematics, Ecology, and Bioremediation Potential—A Review. J. Environ. Manage. 2021, 280, 111744. [Google Scholar] [CrossRef]
- Wu, L.; Bi, E. Sorption of Ionic and Neutral Species of Pharmaceuticals to Loessial Soil Amended with Biochars. Environ. Sci. Pollut. Res. 2019, 26, 35871–35881. [Google Scholar] [CrossRef]
- Rodríguez-Caballero, G.; Caravaca, F.; Alguacil, M.M.; Fernández-López, M.; Fernández-González, A.J.; Roldán, A. Striking Alterations in the Soil Bacterial Community Structure and Functioning of the Biological N Cycle Induced by Pennisetum setaceum Invasion in a Semiarid Environment. Soil Biol. Biochem. 2017, 109, 176–187. [Google Scholar] [CrossRef]
- Dias, A.C.F.; Costa, F.E.C.; Andreote, F.D.; Lacava, P.T.; Teixeira, M.A.; Assumpção, L.C.; Araújo, W.L.; Azevedo, J.L.; Melo, I.S. Isolation of Micropropagated Strawberry Endophytic Bacteria and Assessment of Their Potential for Plant Growth Promotion. World J. Microbiol. Biotechnol. 2009, 25, 189–195. [Google Scholar] [CrossRef]
- Ma, L.J.; Geiser, D.M.; Proctor, R.H.; Rooney, A.P.; O’donnell, K.; Trail, F.; Gardiner, D.M.; Manners, J.M.; Kazan, K. Fusarium Pathogenomics. Annu. Rev. Microbiol. 2013, 67, 399–416. [Google Scholar] [CrossRef] [PubMed]
- Sajeena, A.; Nair, D.S.; Sreepavan, K. Non-Pathogenic Fusarium oxysporum as a Biocontrol Agent. Indian Phytopathol. 2020, 73, 177–183. [Google Scholar] [CrossRef]
- Carlucci, A.; Raimondo, M.L.; Santos, J.; Phillips, A.J.L. Plectospharaella Species Associated with Root and Collar Rots of Horticultural Crops in Southern Italy. Persoonia 2012, 28, 34–48. [Google Scholar] [CrossRef] [PubMed]
- Deb, D.; Khan, A.; Dey, N. Phoma Diseases: Epidemiology and Control. Plant Pathol. 2020, 69, 1203–1217. [Google Scholar] [CrossRef]
| Treatment | Mineral N Fertilizer | Organic Fertilizer | ||||
|---|---|---|---|---|---|---|
| Dose (kg N ha−1) | Type | Dose (t ha−1) | Type | Additive | ||
| Percentage of Additive in the Compost (w:w) (%) | Description | |||||
| Control 0 | 0 | 0 | ||||
| Control 80 | 176 (80% of full dose) | NH4NO3 (27% N) | 0 | |||
| Control 100 | 220 (full dose) | NH4NO3 (27% N) | 0 | |||
| Compost 2 | 176 | NH4NO3 (27% N) | 2 | Compost | 0 | - |
| Compost 5 | 176 | NH4NO3 (27% N) | 5 | Compost | 0 | - |
| Compost 2 + bb3 | 176 | NH4NO3 (27% N) | 2 | Compost + additive | 3 | biochar:bacterial suspension 1 × 109 cfu ml−1 (1:1 w:vol.) |
| Compost 2 + bb6 | 176 | NH4NO3 (27% N) | 2 | Compost + additive | 6 | biochar:bacterial suspension 1 × 109 cfu ml−1 (1:1 w:vol.) |
| Compost 5 + bb3 | 176 | NH4NO3 (27% N) | 5 | Compost + additive | 3 | biochar:bacterial suspension 1 × 109 cfu ml−1 (1:1 w:vol.) |
| Compost 5 + bb6 | 176 | NH4NO3 (27% N) | 5 | Compost + additive | 6 | biochar:bacterial suspension 1 × 109 cfu ml−1 (1:1 w:vol.) |
| Doses | Fresh Aerial Vegetative Biomass (g Per Plant) | Dry Aerial Vegetative Biomass (g Per Plant) | Yield (kg ha−1) | Number Fruits Per Plant | Fruit Weight (g) | Number Fruits Per ha | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean Square | F Statistic | Mean Square | F Statistic | Mean Square | F Statistic | Mean Square | F Statistic | Mean Square | F Statistic | Mean Square | F Statistic | |
| Compost dose (t/ha) | 1,445,378 | 33.32 *** | 802,250 | 33.43 *** | 48,158,351 | 9.19 *** | 0.23 | 1.13 ns | 2948 | 0.59 ns | 7,615,915 | 8.06 *** |
| Additive dose (%) | 1,170,231 | 26.98 *** | 629,323 | 26.22 *** | 323,276,628 | 61.67 *** | 1.01 | 4.94 ** | 151,818 | 30.27 *** | 29,944,528 | 31.69 *** |
| Compost dose × Additive dose | 283,338 | 6.53 *** | 163,831 | 6.83 *** | 53,606,898 | 10.23 *** | 0.47 | 2.27 ns | 4462 | 0.89 ns | 12,941,871 | 13.69 *** |
| Doses | Fresh Aerial Vegetative Biomass (g Per Plant) | Dry Aerial Vegetative Biomass (g Per Plant) | Yield (kg ha−1) | Number Fruits Per Plant | Fruit Weight (g) | Number Fruits Per ha | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean Square | F Statistic | Mean Square | F Statistic | Mean Square | F Statistic | Mean Square | F Statistic | Mean Square | F Statistic | Mean Square | F Statistic | |
| Compost dose (t/ha) | 4938 | 2.42 ns | - | - | 51,096,351 | 3.57 * | 40 | 5.71 ** | 152 | 2.02 ns | 24,859,770,931 | 5.71 ** |
| Additive dose (%) | 12,428 | 6.10 ** | 1284 | 5.43 ** | 171,270,302 | 11.95 *** | 4 | 0.59 ns | 242 | 3.23 * | 2,582,073,179 | 0.59 ns |
| Compost dose × Additive dose | 1813 | 0.89 ns | - | - | 1,602,240 | 0.11 ns | 45 | 6.47 *** | 436 | 5.81 ** | 28,180,202,318 | 6.47 *** |
| Doses | Chlorophyll (CCI) | Flowering (%) | Penetrometry (kg) | Fruit Contour (cm) | Conductivity (µS cm−1) | Solute Concentration (mg l−1) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean Square | F Statistic | Mean Square | F Statistic | Mean Square | F Statistic | Mean Square | F Statistic | Mean Square | F Statistic | Mean Square | F Statistic | |
| Compost dose (t/ha) | 460.030 | 9.640 *** | 3724.643 | 37.421*** | 0.059 | 2.566 ns | 222.610 | 31.083 *** | 0.591 | 15.926 *** | 81045.252 | 15.240 *** |
| Additive dose (%) | 71.741 | 1.503 ns | 147.024 | 1.477 ns | 0.247 | 10.80 *** | 1.427 | 0.199 ns | 0.564 | 15.186 *** | 222846.500 | 41.905 *** |
| Compost dose × Additive dose | 8.190 | 0.172 ns | 22.024 | 0.22 ns | 0.218 | 9.507 *** | 10.496 | 1.465 ns | 0.009 | 0.231 ns | 8879.786 | 1.670 ns |
| Doses | Chlorophyll (CCI) | Fruit Contour (mm) | Conductivity (µS cm−1) | Solute Concentration (mg l−1) | ||||
|---|---|---|---|---|---|---|---|---|
| Mean Square | F Statistic | Mean Square | F Statistic | Mean Square | F Statistic | Mean Square | F Statistic | |
| Compost dose (t/ha) | 383.867 | 6.790 ** | 66.404 | 0.051 ns | 0.532 | 7.733 ** | 7926.828 | 15.240 *** |
| Additive dose (%) | 18.672 | 0.330 ns | 0.141 | 0.000 ns | 0.357 | 5.194 * | 180,354.056 | 41.905 *** |
| Compost dose × Additive dose | 53.852 | 0.953 ns | 6.699 | 0.005 ns | 0.082 | 1.187 ns | 18,287.056 | 1.670 ns |
| Bacteria | Fungi | |||||||
|---|---|---|---|---|---|---|---|---|
| Rhizospheric Soil | Bulk Soil | Rhizospheric Soil | Bulk Soil | |||||
| Pseudo-F/t | p-Value | Pseudo-F/t | p-Value | Pseudo-F/t | p-Value | Pseudo-F/t | p-Value | |
| Control 0 vs. Control 80 | 6.7329 | 0.001 | 2.8431 | 0.005 | 5.7629 | 0.001 | 2.2273 | 0.027 |
| Control 0 vs. Compost 5 | 10.406 | 0.001 | 4.6153 | 0.002 | 4.4506 | 0.003 | 3.1415 | 0.009 |
| Control 0 vs. Compost 5+bb6 | 5.0538 | 0.004 | 3.7117 | 0.004 | 8.5004 | 0.001 | 2.3973 | 0.025 |
| Control 80 vs. Compost 5 | 8.1157 | 0.002 | 4.7808 | 0.002 | 2.1977 | 0.031 | 7.3687 | 0.001 |
| Control 80 vs. Compost 5+bb6 | 9.3350 | 0.001 | 3.7124 | 0.002 | 5.3430 | 0.003 | 4.7638 | 0.003 |
| Compost 5 vs. Compost 5+bb6 | 15.099 | 0.001 | 4.0405 | 0.005 | 5.7326 | 0.001 | 4.9925 | 0.002 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ortiz-Liébana, N.; Zotti, M.; Barquero, M.; González-Andrés, F. An Organic Fertilizer ‘Doped’ with a Bacillus Strain Improves Melon and Pepper Yield, Modifying the Rhizosphere Microbiome with Negligible Changes in the Bulk Soil Microbiome. Agronomy 2022, 12, 2620. https://doi.org/10.3390/agronomy12112620
Ortiz-Liébana N, Zotti M, Barquero M, González-Andrés F. An Organic Fertilizer ‘Doped’ with a Bacillus Strain Improves Melon and Pepper Yield, Modifying the Rhizosphere Microbiome with Negligible Changes in the Bulk Soil Microbiome. Agronomy. 2022; 12(11):2620. https://doi.org/10.3390/agronomy12112620
Chicago/Turabian StyleOrtiz-Liébana, Noemí, Maurizio Zotti, Marcia Barquero, and Fernando González-Andrés. 2022. "An Organic Fertilizer ‘Doped’ with a Bacillus Strain Improves Melon and Pepper Yield, Modifying the Rhizosphere Microbiome with Negligible Changes in the Bulk Soil Microbiome" Agronomy 12, no. 11: 2620. https://doi.org/10.3390/agronomy12112620
APA StyleOrtiz-Liébana, N., Zotti, M., Barquero, M., & González-Andrés, F. (2022). An Organic Fertilizer ‘Doped’ with a Bacillus Strain Improves Melon and Pepper Yield, Modifying the Rhizosphere Microbiome with Negligible Changes in the Bulk Soil Microbiome. Agronomy, 12(11), 2620. https://doi.org/10.3390/agronomy12112620










