Selenium Biofortification of Three Wild Species, Rumex acetosa L., Plantago coronopus L., and Portulaca oleracea L., Grown as Microgreens
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Growth Conditions
2.2. Determinations
2.3. Contribution to Selenium Dietary Intake and Health Risk Assessment
2.4. Statistical Analysis
3. Results and Discussion
3.1. First Experiment
3.1.1. Biomass Production of Microgreens
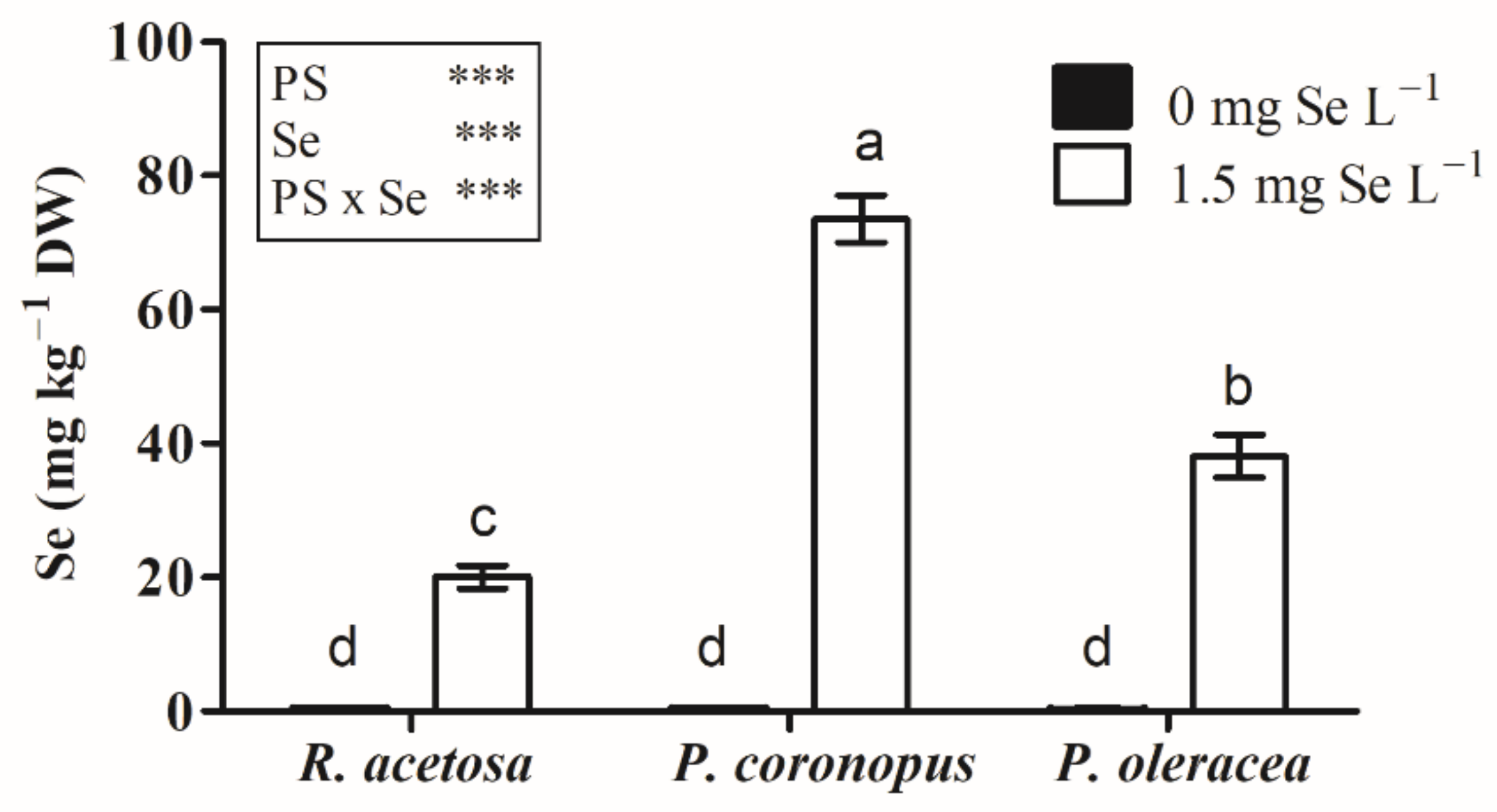
3.1.2. Se Concentration in Microgreens
3.1.3. Contribution to Se requirement in Humans
3.1.4. Photosynthetic Pigments
3.1.5. Total Phenols, Flavonoids, and Antioxidant Capacity
3.2. Second Experiment
3.2.1. Biomass Production of Microgreens
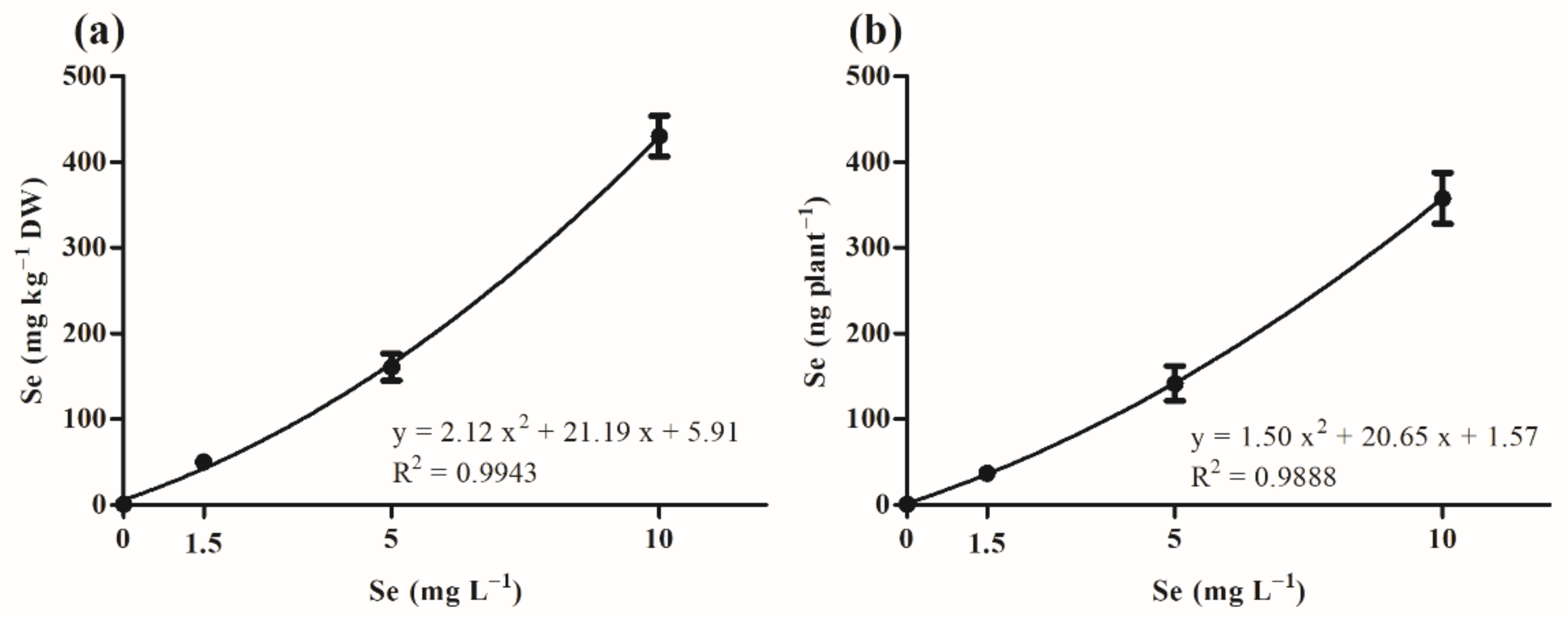
3.2.2. Relationship between Se Concentration in Microgreens and Concentration Applied
3.2.3. Contribution to Se Requirement in Humans
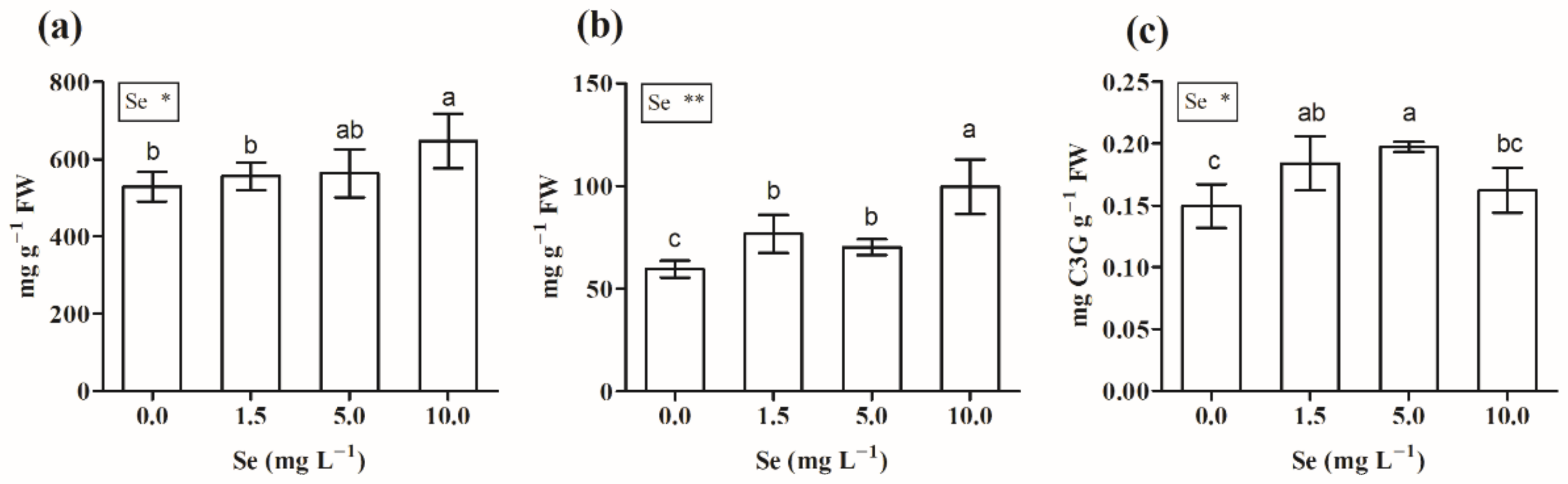
3.2.4. Pigments
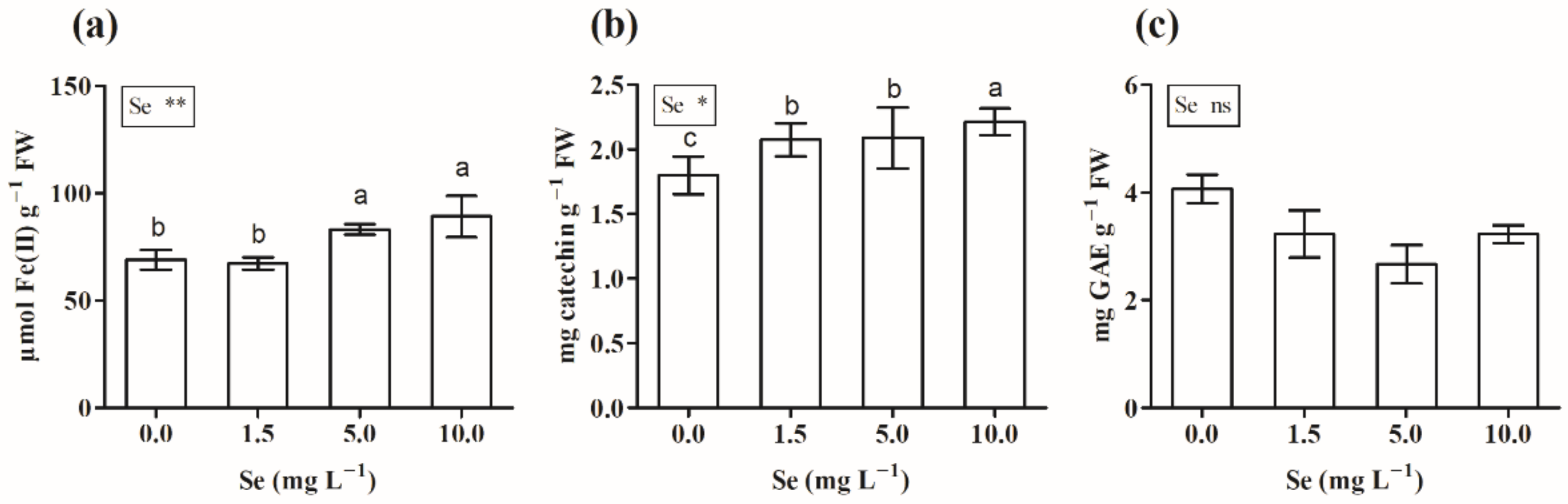
3.2.5. Total Phenols, Flavonoids, and Antioxidant Capacity
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Carvalho, A.M.; Barata, A.M. The Consumption of Wild Edible Plants. In Wild Plants, Mushrooms and Nuts: Functional Food Properties and Applications; Wiley: Hoboken, NJ, USA, 2016; pp. 159–198. ISBN 9781118944653. [Google Scholar]
- De Cortes Sánchez-Mata, M.; Matallana-González, M.C.; Morales, P. The contribution of wild plants to dietary intakes of micronutrients (I): Vitamins. In Mediterranean Wild Edible Plants: Ethnobotany and Food Composition Tables; Springer: New York, NY, USA, 2016; pp. 111–139. ISBN 9781493933297. [Google Scholar]
- García-Herrera, P.; de Cortes Sánchez-Mata, M. The contribution of wild plants to dietary intakes of micronutrients (II): Mineral elements. In Mediterranean Wild Edible Plants: Ethnobotany and Food Composition Tables; Springer: New York, NY, USA, 2016; pp. 141–171. ISBN 9781493933297. [Google Scholar]
- Barros, L.; Morales, P.; Carvalho, A.M.; Ferreira, I.C.F.R. Antioxidant potential of wild plant foods. In Mediterranean Wild Edible Plants: Ethnobotany and Food Composition Tables; Springer: New York, NY, USA, 2016; pp. 209–232. ISBN 9781493933297. [Google Scholar]
- Di Venere, D.; Gatto, M.A.; Ippolito, A.; Bianco, V.V. Antimicrobial potential of wild edible herbaceous species. In Mediterranean Wild Edible Plants: Ethnobotany and Food Composition Tables; Springer: New York, NY, USA, 2016; pp. 233–252. ISBN 9781493933297. [Google Scholar]
- Kyriacou, M.C.; El-Nakhel, C.; Graziani, G.; Pannico, A.; Soteriou, G.A.; Giordano, M.; Ritieni, A.; de Pascale, S.; Rouphael, Y. Functional Quality in Novel Food Sources: Genotypic Variation in the Nutritive and Phytochemical Composition of Thirteen Microgreens Species. Food Chem. 2019, 277, 107–118. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Z.; Lester, G.E.; Park, E.; Saftner, R.A.; Luo, Y.; Wang, Q. Evaluation and Correlation of Sensory Attributes and Chemical Compositions of Emerging Fresh Produce: Microgreens. Postharvest Biol. Technol. 2015, 110, 140–148. [Google Scholar] [CrossRef]
- Di Gioia, F.; Renna, M.; Santamaria, P. Sprouts, Microgreens and “Baby Leaf” Vegetables; Springer: Boston, MA, USA, 2017; pp. 403–432. [Google Scholar]
- Kyriacou, M.C.; Rouphael, Y.; di Gioia, F.; Kyratzis, A.; Serio, F.; Renna, M.; de Pascale, S.; Santamaria, P. Micro-Scale Vegetable Production and the Rise of Microgreens. Trends Food Sci. Technol. 2016, 57, 103–115. [Google Scholar] [CrossRef]
- Lenzi, A.; Orlandini, A.; Bulgari, R.; Ferrante, A.; Bruschi, P. Antioxidant and Mineral Composition of Three Wild Leafy Species: A Comparison between Microgreens and Baby Greens. Foods 2019, 8, 487. [Google Scholar] [CrossRef]
- Birringer, M.; Pilawa, S.; Flohé, L. Trends in Selenium Biochemistry. Nat. Prod. Rep. 2002, 19, 693–718. [Google Scholar] [CrossRef] [PubMed]
- Fordyce, F.M. Selenium deficiency and toxicity in the environment. In Essentials of Medical Geology: Revised Edition; Springer: Dordrecht, The Netherlands, 2013; pp. 375–416. ISBN 9789400743755. [Google Scholar]
- Rayman, M.P. Selenium and Human Health. Lancet 2012, 379, 1256–1268. [Google Scholar] [CrossRef]
- Hamilton, S.J. Review of Selenium Toxicity in the Aquatic Food Chain. Sci. Total Environ. 2004, 326, 1–31. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Wang, S.; Zhou, R.; Sun, S. Endemic Selenium Intoxication of Humans in China. Am. J. Clin. Nutr. 1983, 37, 872–881. [Google Scholar] [CrossRef] [PubMed]
- Raisbeck, M.F. Selenosis. Vet. Clin. N. Am. Food Anim. Pract. 2000, 16, 465–480. [Google Scholar] [CrossRef]
- Diao, M.; Ma, L.; Wang, J.; Cui, J.; Fu, A.; Liu, H. Selenium Promotes the Growth and Photosynthesis of Tomato Seedlings Under Salt Stress by Enhancing Chloroplast Antioxidant Defense System. J. Plant Growth Regul. 2014, 33, 1–12. [Google Scholar] [CrossRef]
- Malorgio, F.; Diaz, K.E.; Ferrante, A.; Mensuali-Sodi, A.; Pezzarossa, B. Effects of Selenium Addition on Minimally Processed Leafy Vegetables Grown in a Floating System. J. Sci. Food Agric. 2009, 89, 2243–2251. [Google Scholar] [CrossRef]
- Islam, M.Z.; Park, B.J.; Kang, H.M.; Lee, Y.T. Influence of Selenium Biofortification on the Bioactive Compounds and Antioxidant Activity of Wheat Microgreen Extract. Food Chem. 2020, 309, 125763. [Google Scholar] [CrossRef]
- Skrypnik, L.; Novikova, A.; Tokupova, E. Improvement of Phenolic Compounds, Essential Oil Content and Antioxidant Properties of Sweet Basil (Ocimum basilicum L.) Depending on Type and Concentration of Selenium Application. Plants 2019, 8, 458. [Google Scholar] [CrossRef] [PubMed]
- Germ, M.; Stibilj, V.; Šircelj, H.; Jerše, A.; Kroflič, A.; Golob, A.; Maršić, N.K. Biofortification of Common Buckwheat Microgreens and Seeds with Different Forms of Selenium and Iodine. J. Sci. Food Agric. 2019, 99, 4353–4362. [Google Scholar] [CrossRef]
- Puccinelli, M.; Malorgio, F.; Rosellini, I.; Pezzarossa, B. Production of Selenium-Biofortified Microgreens from Selenium-Enriched Seeds of Basil. J. Sci. Food Agric. 2019, 99, 4353–4362. [Google Scholar] [CrossRef]
- Pannico, A.; El-Nakhel, C.; Graziani, G.; Kyriacou, M.C.; Giordano, M.; Soteriou, G.A.; Zarrelli, A.; Ritieni, A.; de Pascale, S.; Rouphael, Y. Selenium Biofortification Impacts the Nutritive Value, Polyphenolic Content, and Bioactive Constitution of Variable Microgreens Genotypes. Antioxidants 2020, 9, 272. [Google Scholar] [CrossRef] [PubMed]
- Bandiera, M.; Dal Cortivo, C.; Barion, G.; Mosca, G.; Vamerali, T. Phytoremediation Opportunities with Alimurgic Species in Metal-Contaminated Environments. Sustainability 2016, 8, 357. [Google Scholar] [CrossRef]
- Tardío, J.; de Cortes Sánchez-Mata, M.; Morales, R.; Molina, M.; García-Herrera, P.; Morales, P.; Díez-Marqués, C.; Fernández-Ruiz, V.; Cámara, M.; Pardo-De-Santayana, M.; et al. Ethnobotanical and food composition monographs of selected mediterranean wild edible plants. In Mediterranean Wild Edible Plants: Ethnobotany and Food Composition Tables; Springer: New York, NY, USA, 2016; pp. 273–470. ISBN 9781493933297. [Google Scholar]
- Bello, O.M.; Fasinu, P.S.; Bello, O.E.; Ogbesejana, A.B.; Adetunji, C.O.; Dada, A.O.; Ibitoye, O.S.; Aloko, S.; Oguntoye, O.S. Wild Vegetable Rumex Acetosa Linn.: Its Ethnobotany, Pharmacology and Phytochemistry—A Review. S. Afr. J. Bot. 2019, 125, 149–160. [Google Scholar] [CrossRef]
- Koyro, H.W. Effect of Salinity on Growth, Photosynthesis, Water Relations and Solute Composition of the Potential Cash Crop Halophyte Plantago coronopus (L.). Environ. Exp. Bot. 2006, 56, 136–146. [Google Scholar] [CrossRef]
- Jdey, A.; Falleh, H.; ben Jannet, S.; Mkadmini Hammi, K.; Dauvergne, X.; Ksouri, R.; Magné, C. Phytochemical Investigation and Antioxidant, Antibacterial and Anti-Tyrosinase Performances of Six Medicinal Halophytes. S. Afr. J. Bot. 2017, 112, 508–514. [Google Scholar] [CrossRef]
- Liu, L.; Howe, P.; Zhou, Y.F.; Xu, Z.Q.; Hocart, C.; Zhang, R. Fatty Acids and β-Carotene in Australian Purslane (Portulaca oleracea) Varieties. J. Chromatogr. A 2000, 893, 207–213. [Google Scholar] [CrossRef]
- Petropoulos, S.; Karkanis, A.; Fernandes, Â.; Barros, L.; Ferreira, I.C.F.R.; Ntatsi, G.; Petrotos, K.; Lykas, C.; Khah, E. Chemical Composition and Yield of Six Genotypes of Common Purslane (Portulaca oleracea L.): An Alternative Source of Omega-3 Fatty Acids. Plant Foods Hum. Nutr. 2015, 70, 420–426. [Google Scholar] [CrossRef]
- Nursery | Manifattura_Maiano. Available online: http://www.maiano.it/eng/nursery.html (accessed on 4 April 2021).
- Hoagland, D.R.; Arnon, D.I. The Water-Culture Method for Growing Plants without Soil: University of California. Calif. Agric. Exp. Stn. 1938, 347, 36–39. [Google Scholar]
- Kang, H.M.; Saltveit, M.E. Antioxidant Capacity of Lettuce Leaf Tissue Increases after Wounding. J. Agric. Food Chem. 2002, 50, 7536–7541. [Google Scholar] [CrossRef]
- Kim, D.O.; Chun, O.K.; Kim, Y.J.; Moon, H.Y.; Lee, C.Y. Quantification of Polyphenolics and Their Antioxidant Capacity in Fresh Plums. J. Agric. Food Chem. 2003, 51, 6509–6515. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J. The Ferric Reducing Ability of Plasma (FRAP) as a Measure of “Antioxidant Power”: The FRAP Assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K. [34] Chlorophylls and Carotenoids: Pigments of Photosynthetic Biomembranes. Methods Enzymol. 1987, 148, 350–382. [Google Scholar] [CrossRef]
- Hrazdina, G.; Marx, G.A.; Hoch, H.C. Distribution of Secondary Plant Metabolites and Their Biosynthetic Enzymes in Pea (Pisum sativum L.) Leaves. Plant Physiol. 1982, 70, 745–748. [Google Scholar] [CrossRef]
- UNI EN 13657. Characterization of Waste—Digestion for Subsequent Determination of Aqua Regia Soluble Portion of Elements; International Standard: Geneva, Switzerland, 2004. [Google Scholar]
- UNI EN ISO 17294-2. Water Quality—Application of Inductively Coupled Plasma Mass Spectrometry (ICP-MS)—Part 2: Determination of Selected Elements Including Uranium Isotopes; International Standard: Geneva, Switzerland, 2016; p. 31. [Google Scholar]
- EFSA. Scientific Opinion on Dietary Reference Values for Selenium. EFSA J. 2014, 12, 3846. [Google Scholar] [CrossRef]
- Kumar, M.; Rahman, M.M.; Ramanathan, A.L.; Naidu, R. Arsenic and Other Elements in Drinking Water and Dietary Components from the Middle Gangetic Plain of Bihar, India: Health Risk Index. Sci. Total Environ. 2016, 539, 125–134. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Tolerable Upper Intake Levels for Vitamins and Minerals; European Food Safety Authority: Parma, Italy, 2006. [Google Scholar]
- Paradiso, V.M.; Castellino, M.; Renna, M.; Gattullo, C.E.; Calasso, M.; Terzano, R.; Allegretta, I.; Leoni, B.; Caponio, F.; Santamaria, P. Nutritional Characterization and Shelf-Life of Packaged Microgreens. Food Funct. 2018, 9, 5629–5640. [Google Scholar] [CrossRef]
- Bulgari, R.; Baldi, A.; Ferrante, A.; Lenzi, A. Yield and Quality of Basil, Swiss Chard, and Rocket Microgreens Grown in a Hydroponic System. N. Z. J. Crop Hortic. Sci. 2017, 45, 119–129. [Google Scholar] [CrossRef]
- Renna, M.; Castellino, M.; Leoni, B.; Paradiso, V.; Santamaria, P.; Renna, M.; Castellino, M.; Leoni, B.; Paradiso, V.M.; Santamaria, P. Microgreens Production with Low Potassium Content for Patients with Impaired Kidney Function. Nutrients 2018, 10, 675. [Google Scholar] [CrossRef] [PubMed]
- Dall’Acqua, S.; Ertani, A.; Pilon-Smits, E.A.H.; Fabrega-Prats, M.; Schiavon, M. Selenium Biofortification Differentially Affects Sulfur Metabolism and Accumulation of Phytochemicals in Two Rocket Species (Eruca sativa Mill. and Diplotaxis tenuifolia) Grown in Hydroponics. Plants 2019, 8, 68. [Google Scholar] [CrossRef] [PubMed]
- Paradiso, V.M.; Castellino, M.; Renna, M.; Leoni, B.; Caponio, F.; Santamaria, P. Simple Tools for Monitoring Chlorophyll in Broccoli Raab and Radish Microgreens on Their Growing Medium during Cold Storage. Prog. Nutr. 2018, 20, 415–422. [Google Scholar] [CrossRef]
- Xiao, Z.; Luo, Y.; Lester, G.E.; Kou, L.; Yang, T.; Wang, Q. Postharvest Quality and Shelf Life of Radish Microgreens as Impacted by Storage Temperature, Packaging Film, and Chlorine Wash Treatment. LWT Food Sci. Technol. 2014, 55, 551–558. [Google Scholar] [CrossRef]
- Tan, L.; Nuffer, H.; Feng, J.; Kwan, S.H.; Chen, H.; Tong, X.; Kong, L. Antioxidant Properties and Sensory Evaluation of Microgreens from Commercial and Local Farms. Food Sci. Hum. Wellness 2020, 9, 45–51. [Google Scholar] [CrossRef]
- Lobiuc, A.; Vasilache, V.; Oroian, M.; Stoleru, T.; Burducea, M.; Pintilie, O.; Zamfirache, M.-M.; Lobiuc, A.; Vasilache, V.; Oroian, M.; et al. Blue and Red LED Illumination Improves Growth and Bioactive Compounds Contents in Acyanic and Cyanic Ocimum basilicum L. Microgreens. Molecules 2017, 22, 2111. [Google Scholar] [CrossRef]
- Xiao, Z.; Lester, G.E.; Luo, Y.; Xie, Z.; Yu, L.; Wang, Q. Effect of Light Exposure on Sensorial Quality, Concentrations of Bioactive Compounds and Antioxidant Capacity of Radish Microgreens during Low Temperature Storage. Food Chem. 2014, 151, 472–479. [Google Scholar] [CrossRef]
- Samuolienė, G.; Viršilė, A.; Brazaitytė, A.; Jankauskienė, J.; Sakalauskienė, S.; Vaštakaitė, V.; Novičkovas, A.; Viškelienė, A.; Sasnauskas, A.; Duchovskis, P. Blue Light Dosage Affects Carotenoids and Tocopherols in Microgreens. Food Chem. 2017, 228, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Brazaityte, A.; Sakalauskiene, S.; Samuoliene, G.; Jankauskiene, J.; Viršile, A.; Novičkovas, A.; Sirtautas, R.R.; Miliauskiene, J.; Vaštakaite, V.; Dabašinskas, L.; et al. The Effects of LED Illumination Spectra and Intensity on Carotenoid Content in Brassicaceae Microgreens. Food Chem. 2015, 173, 600–606. [Google Scholar] [CrossRef]
- Hartikainen, H.; Xue, T.; Piironen, V. Selenium as an Anti-Oxidant and pro-Oxidant in Ryegrass. Plant Soil 2000, 225, 193–200. [Google Scholar] [CrossRef]
- Terry, N.; Zayed, A.M.; de Souza, M.P.; Tarun, A.S. Selenium in Higher Plants. Annu. Rev. Plant Physiol. Plant Mol. Biol. 2000, 51, 401–432. [Google Scholar] [CrossRef]
- D’Amato, R.; Fontanella, M.C.; Falcinelli, B.; Beone, G.M.; Bravi, E.; Marconi, O.; Benincasa, P.; Businelli, D. Selenium Biofortification in Rice (Oryza sativa L.) Sprouting: Effects on Se Yield and Nutritional Traits with Focus on Phenolic Acid Profile. J. Agric. Food Chem. 2018, 66, 4082–4090. [Google Scholar] [CrossRef]
- Di Gioia, F.; Petropoulos, S.A.; Ozores-Hampton, M.; Morgan, K.; Rosskopf, E.N. Zinc and Iron Agronomic Biofortification of Brassicaceae Microgreens. Agronomy 2019, 9, 677. [Google Scholar] [CrossRef]
- Akladious, S.A. Influence of Different Soaking Times with Selenium on Growth, Metabolic Activities of Wheat Seedlings under Low Temperature Stress. Afr. J. Biotechnol. 2012, 11, 14792–14804. [Google Scholar] [CrossRef]
- Feng, R.; Wei, C.; Tu, S. The Roles of Selenium in Protecting Plants against Abiotic Stresses. Environ. Exp. Bot. 2013, 87, 58–68. [Google Scholar] [CrossRef]
- Jiang, C.; Zu, C.; Shen, J.; Shao, F.; Li, T. Effects of Selenium on the Growth and Photosynthetic Characteristics of Flue-Cured Tobacco (Nicotiana tabacum L.). Acta Soc. Bot. Pol. 2015, 84, 71–77. [Google Scholar] [CrossRef]
- Yao, X.; Chu, J.; Ba, C. Antioxidant Responses of Wheat Seedlings to Exogenous Selenium Supply under Enhanced Ultraviolet-B. Biol. Trace Elem. Res. 2010, 136, 96–105. [Google Scholar] [CrossRef]
- Xia, Q.; Yang, Z.; Shui, Y.; Liu, X.; Chen, J.; Khan, S.; Wang, J.; Gao, Z. Methods of Selenium Application Differentially Modulate Plant Growth, Selenium Accumulation and Speciation, Protein, Anthocyanins and Concentrations of Mineral Elements in Purple-Grained Wheat. Front. Plant Sci. 2020, 11, 1114. [Google Scholar] [CrossRef]
- Liu, D.; Li, H.; Wang, Y.; Ying, Z.; Bian, Z.; Zhu, W.; Liu, W.; Yang, L.; Jiang, D. How Exogenous Selenium Affects Anthocyanin Accumulation and Biosynthesis-Related Gene Expression in Purple Lettuce. Pol. J. Environ. Stud. 2017, 26, 717–722. [Google Scholar] [CrossRef]
- Lin, L.; Zhou, W.; Dai, H.; Cao, F.; Zhang, G.; Wu, F. Selenium Reduces Cadmium Uptake and Mitigates Cadmium Toxicity in Rice. J. Hazard. Mater. 2012, 235–236, 343–351. [Google Scholar] [CrossRef]
- Combs, G.F. Selenium in Global Food Systems. Br. J. Nutr. 2001, 85, 517–547. [Google Scholar] [CrossRef]
- Castillo-Godina, R.G.; Foroughbakhch-Pournavab, R.; Benavides-Mendoza, A. Effect of Selenium on Elemental Concentration and Antioxidant Enzymatic Activity of Tomato Plants. J. Agric. Sci. Technol. 2016, 18, 233–244. [Google Scholar]
- Puccinelli, M.; Pezzarossa, B.; Rosellini, I.; Malorgio, F. Selenium Enrichment Enhances the Quality and Shelf Life of Basil Leaves. Plants 2020, 9, 801. [Google Scholar] [CrossRef]
- Emam, M.M.; Khattab, H.E.; Helal, N.M.; Deraz, A.E. Effect of Selenium and Silicon on Yield Quality of Rice Plant Grown under Drought Stress. Aust. J. Crop Sci. 2014, 8, 596–605. [Google Scholar]
- Shahidi, F.; Janitha, P.K.; Wanasundara, P.D. Phenolic Antioxidants. Crit. Rev. Food Sci. Nutr. 1992, 32, 67–103. [Google Scholar] [CrossRef]
- Tian, X.-Y.; Wang, Z.Y. Effects of Selenium Application on Content, Distribution and Accumulation of Selenium, Flavonoids and Rutin in Tartary Buckwheat. Plant Nutr. Fertil. Sci. 2008, 14, 721–727. [Google Scholar]
| Plant Species | Se added mg L−1 | FW kg m−2 | DW kg m−2 | DW/FW % |
|---|---|---|---|---|
| R. acetosa | 0 | 0.874 | 0.071 | 7.95 |
| 1.5 | 0.852 | 0.077 | 9.00 | |
| P. coronopus | 0 | 1.972 | 0.098 | 5.02 |
| 1.5 | 1.751 | 0.086 | 4.92 | |
| P. oleracea | 0 | 0.638 | 0.034 | 5.29 |
| 1.5 | 0.505 | 0.037 | 7.37 | |
| MEAN EFFECT | ||||
| R. acetosa | 0.863b | 0.074b | 8.47a | |
| P. coronopus | 1.861a | 0.092a | 4.97c | |
| P. oleracea | 0.564c | 0.036c | 6.45b | |
| 0 | 1.199a | 0.070 | 6.14b | |
| 1.5 | 1.046b | 0.067 | 7.09a | |
| ANOVA | ||||
| Plant Species (PS) | *** | *** | *** | |
| Selenium concentration (Se) | * | ns | * | |
| PS x Se | ns | ns | ns | |
| Plant Species | Se Added mg L−1 | EDI µg day−1 | EDI % | HRI |
|---|---|---|---|---|
| R. acetosa | 0 | 0.15d | 0.21d | 0.001d |
| 1.5 | 18.81c | 26.87c | 0.063c | |
| P. coronopus | 0 | 0.09d | 0.13d | 0.0003d |
| 1.5 | 37.40a | 53.43a | 0.1247a | |
| P. oleracea | 0 | 0.08d | 0.11d | 0.0003d |
| 1.5 | 30.14b | 43.05b | 0.1005b | |
| MEAN EFFECT | ||||
| R. acetosa | 9.48c | 13.54c | 0.0316c | |
| P. coronopus | 18.75a | 26.78a | 0.0625a | |
| P. oleracea | 15.11b | 21.54b | 0.0504b | |
| 0 | 0.105b | 0.15b | 0.0004b | |
| 1.5 | 28.78a | 41.12a | 0.0959a | |
| ANOVA | ||||
| Plant Species (PS) | *** | *** | *** | |
| Selenium concentration (Se) | *** | *** | *** | |
| PS x Se | *** | *** | *** | |
| Plant Species | Se added mg L−1 | Chls tot mg g−1 | Car mg g−1 | FRAP µmol Fe(II)g−1 | Phen tot. mg GAE g−1 | Flav mg catechin g−1 |
|---|---|---|---|---|---|---|
| R. acetosa | 0 | 0.564 | 0.098 | 34.3 | 3.34 | 1.90 |
| 1.5 | 0.542 | 0.095 | 35.7 | 3.43 | 2.11 | |
| P. coronopus | 0 | 0.762 | 0.080 | 26.3 | 2.30 | 2.00 |
| 1.5 | 0.842 | 0.093 | 39.0 | 2.90 | 2.80 | |
| P. oleracea | 0 | 0.629 | 0.101 | 28.0 | 2.30 | 1.90 |
| 1.5 | 0.663 | 0.108 | 32.7 | 2.60 | 1.90 | |
| MEAN EFFECT | ||||||
| R. acetosa | 0.554c | 0.096ab | 35.0 | 3.38a | 2.01ab | |
| P. coronopus | 0.802a | 0.087b | 32.7 | 2.59b | 2.42a | |
| P. oleracea | 0.645b | 0.104a | 30.4 | 2.42b | 1.89b | |
| 0 | 0.652 | 0.093 | 29.5b | 2.60 | 1.90b | |
| 1.5 | 0.683 | 0.099 | 35.8a | 3.00 | 2.30a | |
| ANOVA | ||||||
| Plant Species (PS) | *** | * | ns | ** | * | |
| Selenium concentration (Se) | ns | ns | * | ns | * | |
| PS x Se | ns | ns | ns | ns | ns | |
| Se added mg L−1 | FW kg m−2 | DW kg m−2 | DW/FW % | Se mg kg−1 DW | EDI µg day −1 | EDI % | HRI |
|---|---|---|---|---|---|---|---|
| 0 | 0.476 | 0.035 | 7.34 | 1.2d | 0.88d | 1.250d | 0.003d |
| 1.5 | 0.508 | 0.035 | 6.98 | 50.4c | 32.69c | 46.70c | 0.109c |
| 5.0 | 0.518 | 0.039 | 7.58 | 160.7b | 118.51b | 169.29b | 0.395b |
| 10.0 | 0.480 | 0.036 | 8.01 | 430.3a | 328.73a | 469.61a | 1.096a |
| ANOVA | |||||||
| Se concentration | ns | ns | ns | *** | *** | *** | *** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Puccinelli, M.; Pezzarossa, B.; Pintimalli, L.; Malorgio, F. Selenium Biofortification of Three Wild Species, Rumex acetosa L., Plantago coronopus L., and Portulaca oleracea L., Grown as Microgreens. Agronomy 2021, 11, 1155. https://doi.org/10.3390/agronomy11061155
Puccinelli M, Pezzarossa B, Pintimalli L, Malorgio F. Selenium Biofortification of Three Wild Species, Rumex acetosa L., Plantago coronopus L., and Portulaca oleracea L., Grown as Microgreens. Agronomy. 2021; 11(6):1155. https://doi.org/10.3390/agronomy11061155
Chicago/Turabian StylePuccinelli, Martina, Beatrice Pezzarossa, Lucia Pintimalli, and Fernando Malorgio. 2021. "Selenium Biofortification of Three Wild Species, Rumex acetosa L., Plantago coronopus L., and Portulaca oleracea L., Grown as Microgreens" Agronomy 11, no. 6: 1155. https://doi.org/10.3390/agronomy11061155
APA StylePuccinelli, M., Pezzarossa, B., Pintimalli, L., & Malorgio, F. (2021). Selenium Biofortification of Three Wild Species, Rumex acetosa L., Plantago coronopus L., and Portulaca oleracea L., Grown as Microgreens. Agronomy, 11(6), 1155. https://doi.org/10.3390/agronomy11061155






