Effective Propagation of Selaginella tamariscina through Optimized Medium Composition
Abstract
1. Introduction
2. Materials and Methods
2.1. Plants
2.2. Basal Medium and Explant Length for Effective Propagation
2.3. Assessment of the Effect of Various Medium Components on Propagation and Growth
2.4. Acclimation in a Controlled Environment and Greenhouse
2.5. Statistics and Data Analysis
3. Results
3.1. Determination of Optimal MS Strength for Propagation and Growth
3.2. Effective Explant Length for Sporophyte Propagation
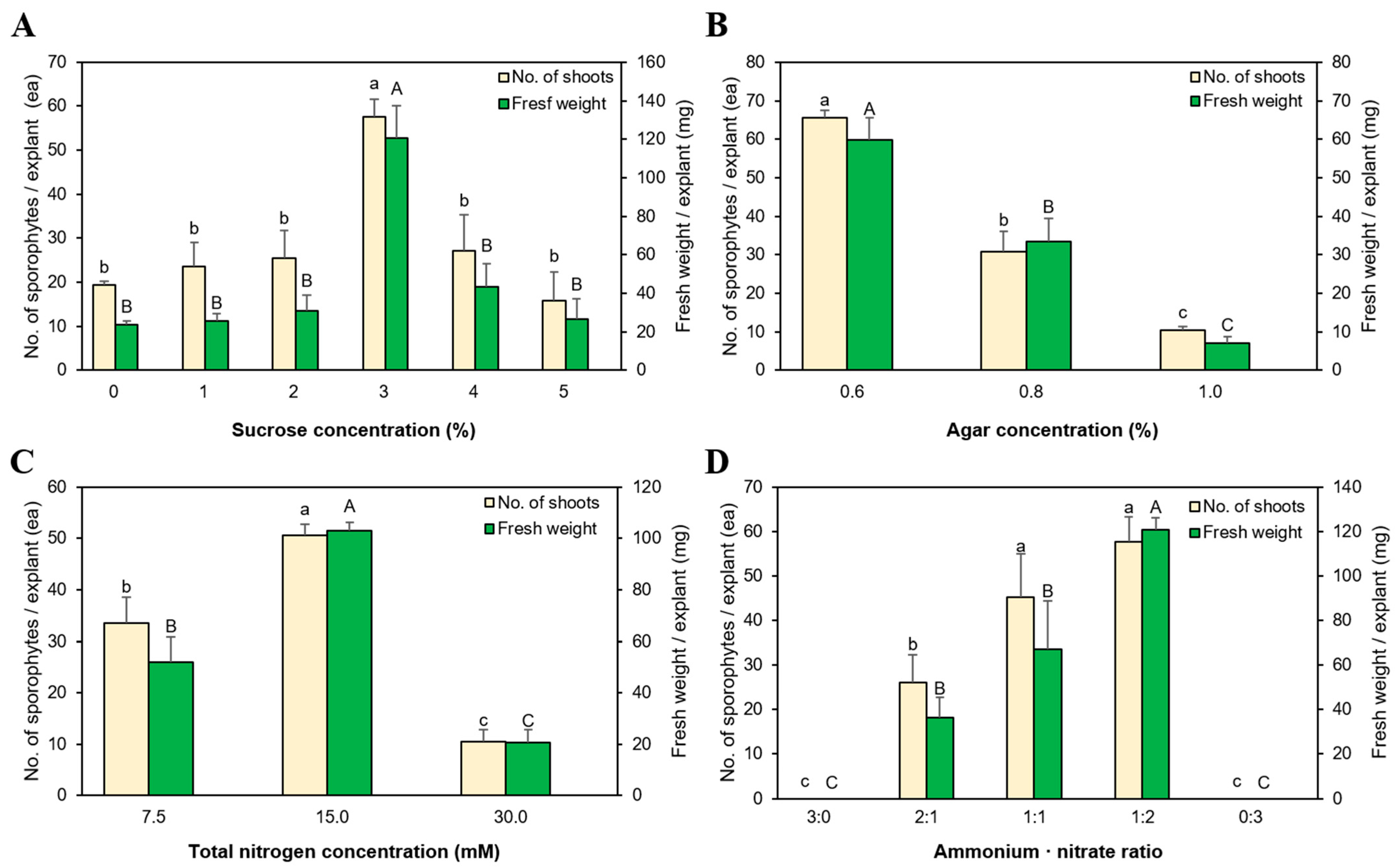
3.3. Sporophyte Regeneration According to Sucrose Concentration
3.4. Effect of Agar Concentration on Growth and Propagation
3.5. The Effect of Nitrogen Concentration and NH4+:NO3− Ratio on Growth and Propagation
3.6. Acclimation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ma, S.C.; But, P.P.H.; Ooi, V.E.C.; He, Y.H.; Lee, S.H.S.; Lee, S.F.; Lin, R.C. Antiviral amentoflavone from Selaginella sinensis. Biol. Pharm. Bull. 2001, 24, 311–312. [Google Scholar] [CrossRef]
- Su, Y.; Sun, C.M.; Chuang, H.H.; Chang, P.T. Studies on the cytotoxic mechanisms of ginkgetin in a human ovarian adenocarcinoma cell line. Naunyn Schmiedebergs Arch. Pharm. 2000, 362, 82–90. [Google Scholar] [CrossRef]
- Cao, Y.; Tan, N.H.; Chen, J.J.; Zeng, G.Z.; Ma, Y.B.; Wu, Y.P.; Yan, H.; Yang, J.; Lu, L.F.; Wang, Q. Bioactive flavones and biflavones from Selaginella moellendorffii Hieron. Fitoterapia 2010, 81, 253–258. [Google Scholar] [CrossRef]
- Silva, G.L.; Chai, H.; Gupta, M.P.; Farnsworth, N.R.; Cordell, G.A.; Pezzuto, J.M.; Beecher, C.W.; Kinghorn, A.D. Cytotoxic biflavonoids from Selaginella willdenowii. Phytochemistry 1995, 40, 129–134. [Google Scholar] [CrossRef]
- Lin, L.C.; Kuo, Y.C.; Chou, C.J. Cytotoxic biflavonoids from Selaginella delicatula. J. Nat. Prod. 2000, 63, 627–630. [Google Scholar] [CrossRef]
- Chao, L.R.; Seguin, E.; Tillequin, F.O.; Koch, M. New alkaloid glycosides from Selaginella doederleinii. J. Nat. Prod. 1987, 50, 422–426. [Google Scholar] [CrossRef]
- Lin, R.C.; Skaltsounis, A.L.; Seguin, E.; Tillequin, F.; Koch, M. Phenolic constituents of Selaginella doederleinii. Planta Med. 1994, 60, 168–170. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.S.; Oh, W.K.; Kim, B.Y.; Ahn, S.C.; Kang, D.O.; Shin, D.I.; Kim, J.; Mheen, T.I.; Ahn, J.S. Inhibition of phospholipase Cγl activity by amentoflavone isolated from Selaginella tamariscina. Planta Med. 1996, 62, 293–296. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.M.; Syu, W.J.; Huang, Y.T.; Chen, C.C.; Ou, J.C. Selective cytotoxicity of ginkgetin from Selaginella moellendorffii. J. Nat. Prod. 1997, 60, 382–384. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.J.; Sung, W.S.; Yeo, S.H.; Kim, H.S.; Lee, I.S.; Woo, E.R.; Lee, D.G. Antifungal effect of amentoflavone derived from Selaginella tamariscina. Arch. Pharm. Res. 2006, 9, 746–751. [Google Scholar] [CrossRef] [PubMed]
- Swamy, R.C.; Kunert, O.; Schuhly, W.; Bucar, F.; Ferreira, D.; Rani, V.S.; Kumar, B.R.; Rao, A.V.N.A. Structurally unique biflavonoids from Selaginella chrysocaulos and Selaginella bryopteris. Chem. Biodivers. 2006, 3, 405–413. [Google Scholar] [CrossRef]
- Lee, C.W.; Choi, H.J.; Kim, H.S.; Kim, D.H.; Chang, I.S.; Moon, H.T.; Lee, S.Y.; Oh, W.K.; Woo, E.R. Biflavonoids isolated from Selaginella tamariscina regulate the expression of matrix metalloproteinase in human skin fibroblasts. Bioorg. Med. Chem. 2008, 16, 732–738. [Google Scholar] [CrossRef]
- Wang, Y.H.; Long, C.L.; Yang, F.M.; Wang, X.; Sun, Q.Y.; Wang, H.S.; Shi, Y.N.; Tang, G.H. Pyrrolidinoindoline alkaloids from Selaginella moellendorfii. J. Nat. Prod. 2009, 72, 1151–1154. [Google Scholar] [CrossRef]
- Zheng, X.K.; Li, Y.J.; Zhang, L.; Feng, W.S.; Zhang, X. Antihyperglycemic activity of Selaginella tamariscina (Beauv.) Spring. J. Ethnopharmacol. 2011, 133, 531–537. [Google Scholar] [CrossRef]
- Ahn, D.K. Illustrated Book of Korean Medicinal Herbs; Kyohaksa: Seoul, Korea, 1998. [Google Scholar]
- Shin, D.I.; Kim, J.W. Flavonoid constituents of Selaginella tamariscina. Korean J. Pharmacogn. 1991, 22, 207–210. [Google Scholar]
- Lee, I.R.; Song, J.Y.; Lee, Y.S. Cytotoxicity of folkloric medicine in murine and human cancer cells. Korean J. Pharmacogn. 1992, 23, 132–134. [Google Scholar]
- Bae, K.H. The Medicinal Plants of Korea; Kyohaksa: Seoul, Korea, 2000. [Google Scholar]
- Yang, J.W.; Pokharel, Y.R.; Kim, M.R.; Woo, E.R.; Choi, H.K.; Kang, K.W. Inhibition of inducible nitric oxide synthase by sumaflavone isolated from Selaginella tamariscina. J. Ethnopharmacol. 2006, 105, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Fernández, H.; Bertrand, A.M.; Sánchez-Tamés, R. In vitro regeneration of Asplenium nidus L. from gametophytic and sporophytic tissue. Sci. Hort. 1993, 56, 71–77. [Google Scholar] [CrossRef]
- Park, K.; Jang, B.K.; Lee, H.M.; Cho, J.S.; Lee, C.H. An efficient method for in vitro shoot-tip culture and sporophyte production using Selaginella martensii Spring sporophyte. Plants 2020, 9, 235. [Google Scholar] [CrossRef] [PubMed]
- Barnicoat, H.; Cripps, R.; Kendon, J.; Sarasan, V. Conservation in vitro of rare and threatened ferns—case studies of biodiversity hotspot and island species. Cell. Dev. Biol. Plant. 2011, 47, 37–45. [Google Scholar] [CrossRef]
- Lee, J.; Kim, M.; Jeong, S.E.; Park, H.Y.; Jeon, C.O.; Park, W. Amentoflavone, a novel cyanobacterial killing agent from Selaginella tamariscina. J. Hazard. Mater. 2020, 384, 121312. [Google Scholar] [CrossRef] [PubMed]
- Woo, S.; Kang, K.B.; Kim, J.; Sung, S.H. Molecular networking reveals the chemical diversity of selaginellin derivatives, natural phosphodiesterase-4 inhibitors from Selaginella tamariscina. J. Nat. Prod. 2019, 82, 1820–1830. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Wang, S.; Yu, M.; Wu, D.; Lei, J.; Li, W.; He, Y.; Gang, W. Ultrasonic–Assisted Ionic Liquid Extraction of Two Biflavonoids from Selaginella tamariscina. ACS Omega 2020, 51, 33113–33124. [Google Scholar] [CrossRef] [PubMed]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Hicks, G.; Aderkas, P. A tissue culture of the ostrich fern Matteuccia struthiopteris L. Todaro. Plant Cell Tissue Org. Cult. 1986, 5, 199–204. [Google Scholar] [CrossRef]
- Higuchi, H.; Amaki, W.; Suzuki, S. In vitro propagation of Nephrolepis cordiflora. Sci. Hort. 1987, 32, 105–113. [Google Scholar] [CrossRef]
- Fernández, H.; Bertrand, A.M.; Sánchez-Tamés, R. Plantlet regeneration in Asplenium nidus L. and Pteris ensiformis L. by homogenisation of BA treated rhizomes. Sci. Hort. 1997, 68, 243–247. [Google Scholar] [CrossRef]
- Ambrožič-Dolinšek, J.; Camloh, M.; Bohanec, B.; Žel, J. Apospory in leaf culture of Staghorn fern (Platycerium bifurcatum). Plant Cell Rep. 2002, 20, 791–796. [Google Scholar] [CrossRef]
- Mikuła, A.; Pożoga, M.; Tomiczak, K.; Rybczyński, J.J. Somatic embryogenesis in ferns: A new experimental system. Plant Cell Rep. 2015, 34, 783–794. [Google Scholar] [CrossRef]
- Jha, T.B.; Mukherjee, S.; Basak, A.; Adhikari, J. In vitro morphogenesis in Selaginella microphylla (Kunth.) Spring. Plant Biotechnol. Rep. 2013, 7, 239–245. [Google Scholar] [CrossRef]
- Gamborg, O.L.; Murashige, T.; Thorpe, T.A.; Vasil, I.K. Plant tissue culture media. In Vitro 1976, 12, 473–478. [Google Scholar] [CrossRef]
- Morini, S. In vitro culture of Osmunda regalis fern. J. Hortic. Sci. Biotechnol. 2000, 75, 12–18. [Google Scholar] [CrossRef]
- Fernández, H.; Revilla, M.A. In vitro culture of ornamental ferns. Plant Cell Tissue Organ Cult. 2003, 73, 1–13. [Google Scholar] [CrossRef]
- Jung, J.A.; Lee, C.H. Factors affected on plant regeneration of Phyllitis scolopendrium (L.) Newman in vitro. Korean J. Plant. Res. 2006, 19, 365–373. [Google Scholar]
- Shin, S.L.; Lee, C.H. In vitro medium composition and culture method affecting mass propagation of Osmunda japonica Thunb. prothalli. Korean J. Hortic. Sci. 2009, 27, 299–304. [Google Scholar]
- Rybczyński, J.; Mikuła, A. Tree ferns biotechnology: From spores to sporophytes. In Working with Ferns: Issues and Applications, 1st ed.; Fernández, H., Kumar, A., Revilla, A., Eds.; Springer: Berlin/Heidelberg, Germany, 2010. [Google Scholar]
- Lee, C.S.; Lee, K.H. Pteridophytes of Korea: Lycophytes & Ferns, 2nd ed.; Geobook: Seoul, Korea, 2018. [Google Scholar]
- Shin, S.L.; Lee, C.H. Optimal culture conditions for masspropagation of gametophyte and sporophyte of Pyrrosia linearifolia by tissue culture. Flower Res. J. 2010, 18, 179–185. [Google Scholar]
- George, E.F. Plant. Propagation by Tissue Culture. Part. 1. The Technology, 2nd ed.; Exegetics Ltd.: Edington, UK, 1993. [Google Scholar]
- Dyer, A.F. The Experimental Biology of Ferns; Academic Press: London, UK, 1979. [Google Scholar]
- Fernández, H.; Bertrand, A.M.; Sánchez-Tamés, R. Biological and nutritional aspects involved in fern multiplication. Plant Cell Tissue Organ. Cult. 1999, 56, 211–214. [Google Scholar] [CrossRef]
- Paiva Neto, V.B.; Otoni, W.C. Carbon sources and their osmotic potential in plant tissue culture: Does it matter? Sci. Hortic. 2003, 97, 193–202. [Google Scholar] [CrossRef]
- Melan, M.A.; Whittier, D.P. Effects of inorganic nitrogen sources on spore germination and gametophyte growth in Botrychium dissectum. Plant Cell Environ. 1990, 13, 477–482. [Google Scholar] [CrossRef]
- Whittier, D.P.; Thomas, R.D. Gametophytes and young sporophytes of Botrychium jenmanii in axenic culture. Int. J. Plant. Sci. 1993, 154, 68–74. [Google Scholar] [CrossRef]
- Fernández, H.; Bertrand, A.M.; Sánchez-Tamés, R. Gemmation in Osmunda regalis L. gametophyte cultured in vitro. Plant Cell Rep. 1997, 16, 358–362. [Google Scholar] [CrossRef]
- Whittier, D.P. Rapid gametophyte maturation in Ophioglossum crotalophoroides. Am. Fern J. 2003, 93, 137–145. [Google Scholar] [CrossRef]
- Jung, J.A.; Lee, C.H. Mass propagation of Dryopteris varia (L.) O. Kuntze by sporophyte culture. Kor. Soc. Floricult. Sci. 2006, 14, 25–34. [Google Scholar]
- Jung, J.A.; Lee, C.H. Several factors affecting on in vitro culture of prothallus and ex vitro sporophyte formation from prothallus of Dyropteris varia (L.) O. Kuntze. Korean J. Plant. Res. 2006, 19, 252–258. [Google Scholar]
- Pevalek-Kozlina, B. Effects of sucrose and agar concentration, and medium pH on staghorn fern [Platycerium bifurcatum (Cav.) C.Chr.] shoot multiplication. Hortic. Sci. 1996, 28, 18–20. [Google Scholar]
- Fernández, H.; Bertrand, A.M.; Feito, I.; Sánchez-Tamés, R. Gametophyte culture in vitro and antheridiogen activity in Blechnum spicant. Plant Cell Tissue Org. Cult. 1997, 50, 71–74. [Google Scholar] [CrossRef]
- Yan, J.; Wang, B.; Zhou, Y.; Hao, S. Resistance from agar medium impacts the helical growth of Arabidopsis primary roots. J. Mech. Behav. Biomed. 2018, 85, 43–50. [Google Scholar] [CrossRef]
- Smith, M.A.L.; Spomer, L.A. Vessels, gels, liquid media, and support systems. In Automation and Environmental Control in Plant Tissue Culture; Aitken-Christie, J., Kozai, T., Smith, M.A.L., Eds.; Springer: Dordrecht, The Netherlands, 2013. [Google Scholar]
- Debergh, P. Effects of agar brand and concentration on the tissue culture medium. Physiol. Plant. 1983, 59, 270–276. [Google Scholar] [CrossRef]
- Debergh, P.; Aitken-Christie, J.; Cohen, D.; Grout, B.; Von Arnold, S.; Zimmerman, R.; Ziv, M. Reconsideration of the term ‘vitrification’ as used in micropropagation. Plant Cell Tissue Organ. Cult. 1992, 30, 135–140. [Google Scholar] [CrossRef]
- Hazarika, B.N. Acclimatization of tissue-cultured plants. Curr. Sci. 2003, 85, 1704–1712. [Google Scholar]
- Pospíšilová, J.; Tichá, I.; Kadleček, P.; Haisel, D.; Plzáková, S. Acclimatization of micropropagated plants to ex vitro conditions. Biol. Plantarum 1999, 42, 481–497. [Google Scholar] [CrossRef]
- Preece, J.E.; Sutter, E.G. Acclimatization of micropropagated plants to the greenhouse and field. In Micropropagation. Technology and Application; Debergh, P.C., Zimmerman, R.H., Eds.; Springer: Dordrecht, The Netherlands, 1991. [Google Scholar]
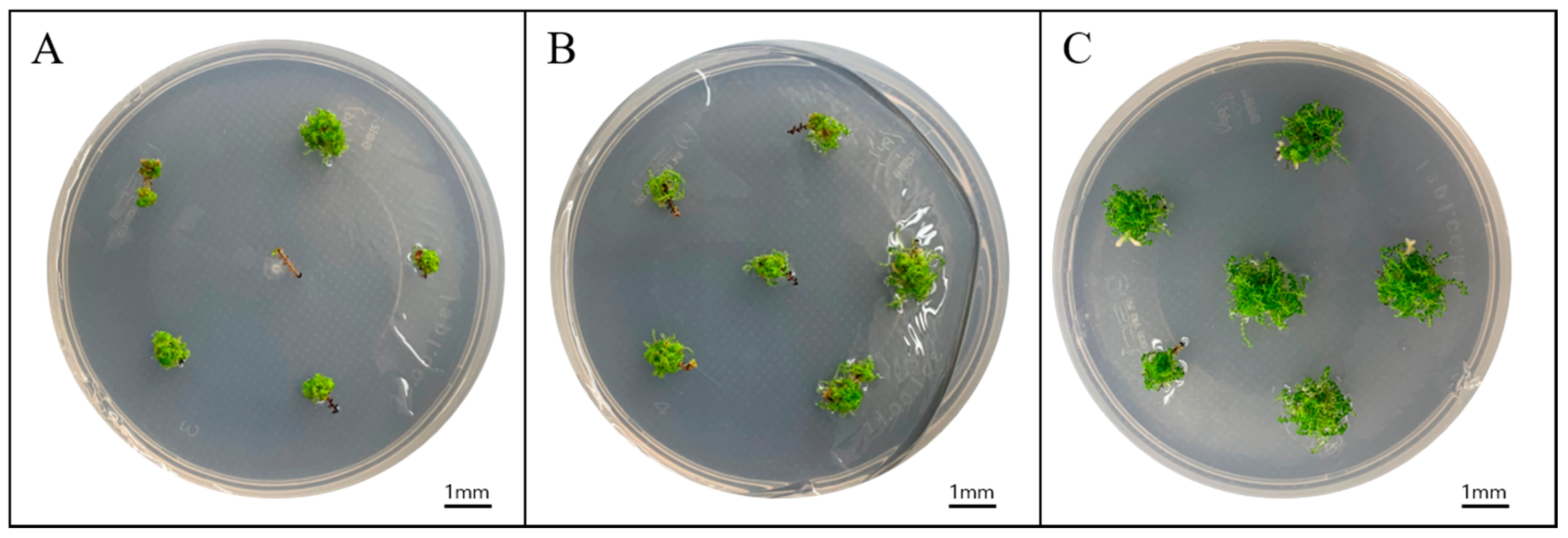

| Medium Strength | No. of Sporophytes/Explant | Fresh Weight/Explant (mg) |
|---|---|---|
| 1/16 MS | 11.2 ± 1.94 d * | 22.4 ± 4.73 cd |
| 1/8 MS | 23.5 ± 4.18 c | 43.5 ± 8.44 c |
| 1/4 MS | 65.7 ± 6.64 a | 221.0 ± 16.55 a |
| 1/2 MS | 32.7 ± 0.63 b | 139.7 ± 16.27 b |
| 1 MS | 8.6 ± 0.97 d | 27.1 ± 3.12 c |
| 2 MS | 0.00 e | 0.9 ± 0.01 d |
| Explant Length (mm) | Survival Rate (%) | No. of Sporophytes/Explant | Fresh Weight/Explant (mg) |
|---|---|---|---|
| 3 | 75.0 ± 8.33 a * | 29.2 ± 4.35 b | 32.4 ± 4.70 b |
| 6 | 87.5 ± 7.98 a | 28.1 ± 1.20 b | 30.2 ± 4.00 b |
| 12 | 88.9 ± 11.11 a | 48.7 ± 2.22 a | 81.8 ± 1.49 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, K.; Jang, B.K.; Lee, H.M.; Cho, J.S.; Lee, C.H. Effective Propagation of Selaginella tamariscina through Optimized Medium Composition. Agronomy 2021, 11, 578. https://doi.org/10.3390/agronomy11030578
Park K, Jang BK, Lee HM, Cho JS, Lee CH. Effective Propagation of Selaginella tamariscina through Optimized Medium Composition. Agronomy. 2021; 11(3):578. https://doi.org/10.3390/agronomy11030578
Chicago/Turabian StylePark, Kyungtae, Bo Kook Jang, Ha Min Lee, Ju Sung Cho, and Cheol Hee Lee. 2021. "Effective Propagation of Selaginella tamariscina through Optimized Medium Composition" Agronomy 11, no. 3: 578. https://doi.org/10.3390/agronomy11030578
APA StylePark, K., Jang, B. K., Lee, H. M., Cho, J. S., & Lee, C. H. (2021). Effective Propagation of Selaginella tamariscina through Optimized Medium Composition. Agronomy, 11(3), 578. https://doi.org/10.3390/agronomy11030578






