Effects of Short-Term Root Cooling before Harvest on Yield and Food Quality of Chinese Broccoli (Brassica oleracea var. Alboglabra Bailey)
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Materials and Experimental Setup
2.2. Evaluation of Photosynthesis, Transpiration, Stomatal Conductance and Leaf Temperature
2.3. Soluble Sugar, Total Chlorophyll and Starch Analysis
2.4. Mineral Elements Quantification
2.5. Glucosinolates Analysis
2.6. Statistical Analysis
3. Results
3.1. Greenhouse Climate Conditions
3.2. Biomass and Yield
3.3. Net Photosynthesis, Transpiration, Stomatal Conductance and Leaf Temperature
3.4. Soluble Sugars, Starch and Total Chlorophyll
3.5. Glucosinolates
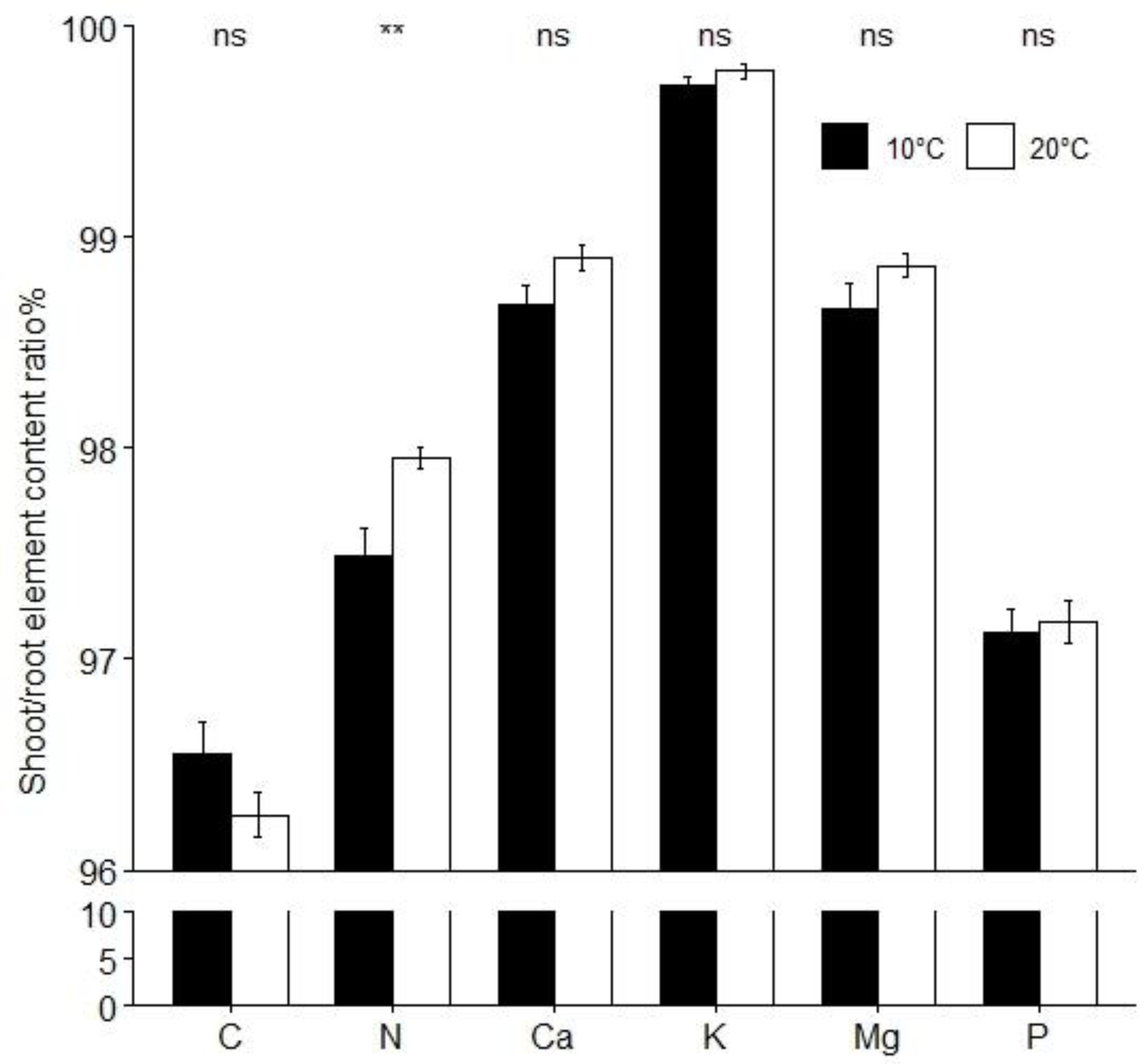
3.6. Mineral Elements
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Guo, J.-J.; Tan, X.; Fu, H.-L.; Chen, J.-X.; Lin, X.-X.; Ma, Y.; Yang, Z.-Y. Selection for Cd pollution-safe cultivars of Chinese kale (Brassica alboglabra LH Bailey) and biochemical mechanisms of the cultivar-dependent Cd accumulation involving in Cd subcellular distribution. J. Agric. Food Chem. 2018, 66, 1923–1934. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Yan, H.; Zhang, F.; Wang, Q. Effects of plant hormones on main health-promoting compounds and antioxidant capacity of Chinese kale. Food Res. Int. 2012, 48, 359–366. [Google Scholar] [CrossRef]
- Martínez-Ballesta, M.D.C.; Moreno, D.A.; Carvajal, M. The physiological importance of glucosinolates on plant response to abiotic stress in Brassica. Int. J. Mol. Sci. 2013, 14, 11607. [Google Scholar] [CrossRef] [PubMed]
- Fahey, J.W.; Wehage, S.L.; Holtzclaw, W.D.; Kensler, T.W.; Egner, P.A.; Shapiro, T.A.; Talalay, P. Protection of humans by plant glucosinolates: Efficiency of conversion of glucosinolates to isothiocyanates by the gastrointestinal microflora. Cancer Prev. Res. 2012, 5, 603–611. [Google Scholar] [CrossRef] [PubMed]
- Dinkova-Kostova, A.T.; Kostov, R.V. Glucosinolates and isothiocyanates in health and disease. Trends Mol. Med. 2012, 18, 337–347. [Google Scholar] [CrossRef] [PubMed]
- Rosa, E.; David, M.; Gomes, M.H. Glucose, fructose and sucrose content in broccoli, white cabbage and Portuguese cabbage grown in early and late seasons. J. Sci. Food Agric. 2001, 81, 1145–1149. [Google Scholar] [CrossRef]
- Kawashima, L.M.; Valente Soares, L.M. Mineral profile of raw and cooked leafy vegetables consumed in Southern Brazil. J. Food Compost. Anal. 2003, 16, 605–611. [Google Scholar] [CrossRef]
- Islam, M.Z.; Lee, Y.-T.; Mele, M.A.; Choi, I.-L.; Kang, H.-M. The effect of phosphorus and root zone temperature on anthocyanin of red romaine lettuce. Agronomy 2019, 9, 47. [Google Scholar] [CrossRef]
- Sakamoto, M.; Suzuki, T. Elevated root-zone temperature modulates growth and quality of hydroponically grown carrots. Agric. Sci. 2015, 6, 749–757. [Google Scholar] [CrossRef]
- Sakamoto, M.; Suzuki, T. Effect of root-zone temperature on growth and quality of hydroponically grown red leaf lettuce (Lactuca sativa L. cv. Red Wave). Am. J. Plant Sci. 2015, 6, 2350. [Google Scholar] [CrossRef]
- Ito, A.; Shimizu, H. Effect of temperature and duration of root chilling on the balance between antioxidant activity and oxidative stress in spinach. Environ. Control. Biol. 2020, 58, 115–121. [Google Scholar] [CrossRef]
- Variyar, P.S.; Banerjee, A.; Akkarakaran, J.J.; Suprasanna, P. Role of glucosinolates in plant stress tolerance. In Emerging Technologies and Management of Crop Stress Tolerance; Ahmad, P., Rasool, S., Eds.; Academic Press: San Diego, CA, USA, 2014; pp. 271–291. [Google Scholar]
- Lam, V.P.; Kim, S.J.; Bok, G.J.; Lee, J.W.; Park, J.S. The effects of root temperature on growth, physiology, and accumulation of bioactive compounds of Agastache rugosa. Agriculture 2020, 10, 162. [Google Scholar] [CrossRef]
- He, F.; Thiele, B.; Santhiraraja-Abresch, S.; Watt, M.; Kraska, T.; Ulbrich, A.; Kuhn, A.J. Effects of root temperature on the plant growth and food quality of Chinese broccoli (Brassica oleracea var. alboglabra Bailey). Agronomy 2020, 10, 702. [Google Scholar] [CrossRef]
- He, F.; Thiele, B.; Watt, M.; Kraska, T.; Ulbrich, A.; Kuhn, A.J. Effects of root cooling on plant growth and fruit quality of cocktail tomato during two consecutive seasons. J. Food Qual. 2019, 2019. [Google Scholar] [CrossRef]
- Hwang, I.; Manoharan, R.K.; Kang, J.-G.; Chung, M.-Y.; Kim, Y.-W.; Nou, I.-S. Genome-wide identification and characterization of bzip transcription factors in Brassica oleracea under Cold Stress. Biomed. Res. Int. 2016, 2016, 4376598. [Google Scholar] [CrossRef]
- Kayum, M.A.; Kim, H.-T.; Nath, U.K.; Park, J.-I.; Kho, K.H.; Cho, Y.-G.; Nou, I.-S. Research on biotic and abiotic stress related genes exploration and prediction in Brassica rapa and B. oleracea: A review. Plant Breed. Biotechnol. 2016, 4, 135–144. [Google Scholar] [CrossRef]
- Aroca, R.; Tognoni, F.; Irigoyen, J.J.; Sánchez-Díaz, M.; Pardossi, A. Different root low temperature response of two maize genotypes differing in chilling sensitivity. Plant Physiol. Biochem. 2001, 39, 1067–1073. [Google Scholar] [CrossRef]
- Schwarz, D.; Rouphael, Y.; Colla, G.; Venema, J.H. Grafting as a tool to improve tolerance of vegetables to abiotic stresses: Thermal stress, water stress and organic pollutants. Sci. Hortic. 2010, 127, 162–171. [Google Scholar] [CrossRef]
- Eremina, M.; Rozhon, W.; Poppenberger, B. Hormonal control of cold stress responses in plants. Cell. Mol. Life Sci. 2016, 73, 797–810. [Google Scholar] [CrossRef] [PubMed]
- Ramakrishna, A.; Ravishankar, G.A. Influence of abiotic stress signals on secondary metabolites in plants. Plant Signal. Behav. 2011, 6, 1720–1731. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, E.; Hikosaka, S.; Goto, E. Effects of nutrient solution temperature on the concentration of major bioactive compounds in red perilla. J. Agric. Meteorol. 2018, 74, 71–78. [Google Scholar] [CrossRef]
- Chadirin, Y.; Hidaka, K.; Takahashi, T.; Sago, Y.; Wajima, T.; Kitano, M. Application of temperature stress to roots of spinach I. Effect of the low temperature stress on quality. Environ. Control. Biol. 2011, 49, 133–139. [Google Scholar] [CrossRef][Green Version]
- Rosa, E.A.S.; Rodrigues, P.M.F. The effect of light and temperature on glucosinolate concentration in the leaves and roots of cabbage seedlings. J. Sci. Food Agric. 1998, 78, 208–212. [Google Scholar] [CrossRef]
- Füllner, K.; Temperton, V.M.; Rascher, U.; Jahnke, S.; Rist, R.; Schurr, U.; Kuhn, A.J. Vertical gradient in soil temperature stimulates development and increases biomass accumulation in barley. Plant Cell Environ. 2012, 35, 884–892. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Yan, H.; Liu, N.; Wei, J.; Wang, Q. Effect of 1-MCP treatment on postharvest quality characters, antioxidants and glucosinolates of Chinese kale. Food Chem. 2012, 131, 519–526. [Google Scholar] [CrossRef]
- Viola, R.; Davies, H.V. A microplate reader assay for rapid enzymatic quantification of sugars in potato tubers. Potato Res. 1992, 35, 55–58. [Google Scholar] [CrossRef]
- Xian, Y.; Yunying, Y. The effects of temperature on flower bud differentiation, yield and quality formation in Chinese kale (Brassica alboglabra Bailey). J. South China Agric. Univ. 2002, 23, 5–7. [Google Scholar] [CrossRef]
- Poiré, R.; Schneider, H.; Thorpe, M.R.; Kuhn, A.J.; Schurr, U.; Walter, A. Root cooling strongly affects diel leaf growth dynamics, water and carbohydrate relations in Ricinus communis. Plant Cell Environ. 2010, 33, 408–417. [Google Scholar] [CrossRef] [PubMed]
- Hao, H.-P.; Jiang, C.-D.; Zhang, S.-R.; Tang, Y.-D.; Shi, L. Enhanced thermal-tolerance of photosystem II by elevating root zone temperature in Prunus mira Koehne seedlings. Plant Soil 2012, 353, 367–378. [Google Scholar] [CrossRef]
- Aloni, B.; Cohen, R.; Karni, L.; Aktas, H.; Edelstein, M. Hormonal signaling in rootstock–scion interactions. Sci. Hortic. 2010, 127, 119–126. [Google Scholar] [CrossRef]
- Dodd, I.C.; He, J.; Turnbull, C.G.; Lee, S.K.; Critchley, C. The influence of supra-optimal root-zone temperatures on growth and stomatal conductance in Capsicum annuum L. J. Exp. Bot. 2000, 51, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Nagasuga, K.; Murai-Hatano, M.; Kuwagata, T. Effects of low root temperature on dry matter production and root water uptake in rice plants. Plant Prod. Sci. 2011, 14, 22–29. [Google Scholar] [CrossRef]
- Kuwagata, T.; Ishikawa-Sakurai, J.; Hayashi, H.; Nagasuga, K.; Fukushi, K.; Ahamed, A.; Takasugi, K.; Katsuhara, M.; Murai-Hatano, M. Influence of low air humidity and low root temperature on water uptake, growth and aquaporin expression in rice plants. Plant Cell Physiol. 2012, 53, 1418–1431. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhang, W.; Miao, Y.; Gao, L. Root-zone warming differently benefits mature and newly unfolded leaves of Cucumis sativus l. seedlings under sub-optimal temperature stress. PLoS ONE 2016, 11, e0155298. [Google Scholar] [CrossRef]
- Taranet, P.; Kirchhof, G.; Fujinuma, R.; Menzies, N. Root zone temperature alters storage root formation and growth of sweetpotato. J. Agron. Crop Sci. 2018, 204, 313–324. [Google Scholar] [CrossRef]
- Aidoo, M.K.; Sherman, T.; Lazarovitch, N.; Fait, A.; Rachmilevitch, S. Physiology and metabolism of grafted bell pepper in response to low root-zone temperature. Funct. Plant Biol. 2019, 46, 339–349. [Google Scholar] [CrossRef] [PubMed]
- Atkin, O.K.; Edwards, E.J.; Loveys, B.R. Response of root respiration to changes in temperature and its relevance to global warming: REVIEW Temperature and root respiration. New Phytol. 2000, 147, 141–154. [Google Scholar] [CrossRef]
- Shimono, H.; Hasegawa, T.; Fujimura, S.; Iwama, K. Responses of leaf photosynthesis and plant water status in rice to low water temperature at different growth stages. Field Crops Res. 2004, 89, 71–83. [Google Scholar] [CrossRef]
- Wang, Y.-Q.; Hu, L.-P.; Liu, G.-M.; Zhang, D.-S.; He, H.-J. Evaluation of the nutritional quality of Chinese kale (Brassica alboglabra Bailey) using UHPLC-Quadrupole-Orbitrap MS/MS-based metabolomics. Molecules 2017, 22. [Google Scholar] [CrossRef]
- Liu, W.; Su, J.; Li, S.; Lang, X.; Huang, X. Non-structural carbohydrates regulated by season and species in the subtropical monsoon broad-leaved evergreen forest of Yunnan Province, China. Sci. Rep. 2018, 8, 1083. [Google Scholar] [CrossRef] [PubMed]
- Arai-Sanoh, Y.; Ishimaru, T.; Ohsumi, A.; Kondo, M. Effects of soil temperature on growth and root function in rice. Plant Prod. Sci. 2010, 13, 235–242. [Google Scholar] [CrossRef]
- Sami, F.; Yusuf, M.; Faizan, M.; Faraz, A.; Hayat, S. Role of sugars under abiotic stress. Plant Physiol. Biochem. 2016, 109, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Peuke, A.D.; Windt, C.; Van As, H. Effects of cold-girdling on flows in the transport phloem in Ricinus communis: Is mass flow inhibited? Plant Cell Environ. 2006, 29, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Adebooye, O.C.; Schmitz-Eiberger, M.; Lankes, C.; Noga, G.J. Inhibitory effects of sub-optimal root zone temperature on leaf bioactive components, photosystem II (PS II) and minerals uptake in Trichosanthes cucumerina L. Cucurbitaceae. Acta Physiol. Plant. 2010, 32, 67–73. [Google Scholar] [CrossRef]
- Anwar, A.; Di, Q.; Yan, Y.; He, C.; Li, Y.; Yu, X. Exogenous 24-epibrassinolide alleviates the detrimental effects of suboptimal root zone temperature in cucumber seedlings. Arch. Agron. Soil Sci. 2019, 65, 1927–1940. [Google Scholar] [CrossRef]
- Bumgarner, N.R.; Scheerens, J.C.; Mullen, R.W.; Bennett, M.A.; Ling, P.P.; Kleinhenz, M.D. Root-zone temperature and nitrogen affect the yield and secondary metabolite concentration of fall- and spring-grown, high-density leaf lettuce. J. Sci. Food Agric. 2012, 92, 116–124. [Google Scholar] [CrossRef] [PubMed]
- La, G.-X.; Fang, P.; Li, Y.-J.; Wang, Y. Determination of desulpho-glucosinolates in bolting stems of Chinese kale by liquid chromatography-mass spectrometry. J. Zhejiang Univ. 2008, 34. [Google Scholar] [CrossRef]
- La, G.-X.; Fang, P.; Teng, Y.-B.; Li, Y.-J.; Lin, X.-Y. Effect of CO2 enrichment on the glucosinolate contents under different nitrogen levels in bolting stem of Chinese kale (Brassica alboglabra L.). J. Zhejiang Univ. Sci. B 2009, 10, 454–464. [Google Scholar] [CrossRef]
- Charron, C.S.; Sams, C.E. Glucosinolate content and myrosinase activity in rapid-cycling Brassica oleracea grown in a controlled environment. J. Am. Soc. Hortic. Sci. 2004, 129, 321–330. [Google Scholar] [CrossRef]
- Steindal, A.L.H.; Mølmann, J.; Bengtsson, G.B.; Johansen, T.J. Influence of day length and temperature on the content of health-related compounds in broccoli (Brassica oleracea L. var. italica). J. Agric. Food Chem. 2013, 61, 10779–10786. [Google Scholar] [CrossRef]
- Engelen-Eigles, G.; Holden, G.; Cohen, J.D.; Gardner, G. The effect of temperature, photoperiod, and light quality on gluconasturtiin concentration in watercress (Nasturtium officinale R. Br.). J. Agric. Food Chem. 2006, 54, 328–334. [Google Scholar] [CrossRef]
- Farnham, M.W.; Wilson, P.E.; Stephenson, K.K.; Fahey, J.W. Genetic and environmental effects on glucosinolate content and chemoprotective potency of broccoli. Plant Breed. 2004, 123, 60–65. [Google Scholar] [CrossRef]
- Cocetta, G.; Mishra, S.; Raffaelli, A.; Ferrante, A. Effect of heat root stress and high salinity on glucosinolates metabolism in wild rocket. J. Plant Physiol. 2018, 231, 261–270. [Google Scholar] [CrossRef]
- Kissen, R.; Eberl, F.; Winge, P.; Uleberg, E.; Martinussen, I.; Bones, A.M. Effect of growth temperature on glucosinolate profiles in Arabidopsis thaliana accessions. Phytochemistry 2016, 130, 106–118. [Google Scholar] [CrossRef] [PubMed]
- Steindal, A.L.H.; Rødven, R.; Hansen, E.; Mølmann, J. Effects of photoperiod, growth temperature and cold acclimatisation on glucosinolates, sugars and fatty acids in kale. Food Chem. 2015, 174, 44–51. [Google Scholar] [CrossRef]
- Baghour, M.; Ragala, L.; Moreno, D.A.; Villora, G.; Hernández, J.; Castilla, N.; Romero, L. Effect of root zone temperature on accumulation of molybdenum and nitrogen metabolism in potato plants. J. Plant Nutr. 2003, 26, 443–461. [Google Scholar] [CrossRef]
- Pettersson, S. Low root zone temperature effects on net mineral nutrient uptake and distribution in barley (Hordeum vulgare). J. Plant Physiol. 1995, 145, 459–464. [Google Scholar] [CrossRef]
- Starck, Z.; Niemyska, B.; Bogdan, J.; Tawalbeh, R.N.A. Response of tomato plants to chilling stress in association with nutrient or phosphorus starvation. Plant Soil 2000, 226, 99–106. [Google Scholar] [CrossRef]
- Aidoo, M.K.; Sherman, T.; Lazarovitch, N.; Fait, A.; Rachmilevitch, S. A bell pepper cultivar tolerant to chilling enhanced nitrogen allocation and stress-related metabolite accumulation in the roots in response to low root-zone temperature. Physiol. Plant. 2017, 161, 196–210. [Google Scholar] [CrossRef] [PubMed]
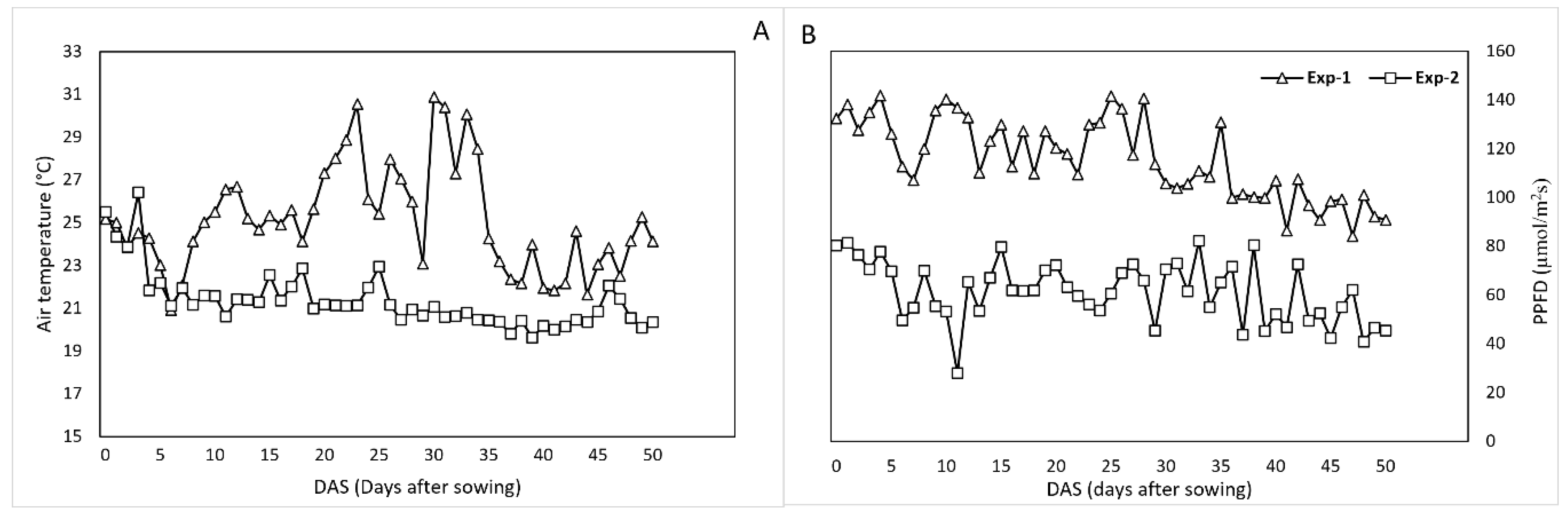
| Experiment | Exp-1 | Exp-2 | |||||
|---|---|---|---|---|---|---|---|
| Treatment | 10 °C | 20 °C | p-Value | 10 °C | 20 °C | p-Value | |
| Shoot | Yield (g) | 109.72 ± 13.74 | 120.97 ± 13.90 | 0.062 | 89.31 ± 17.91 | 110.25 ± 16.61 | 0.009 |
| FW (g) | 150.33 ± 18.67 | 166.91 ± 18.16 | 0.038 | 117.17 ± 20.93 | 140.13 ± 16.76 | 0.009 | |
| DW (g) | 11.5 ± 1.22 | 12.49 ± 1.48 | 0.052 | 10.39 ± 1.79 | 11.34 ± 1.40 | 0.177 | |
| Height (cm) | 30.9 ± 2.64 | 33.1 ± 3.40 | 0.091 | 30.55 ± 4.84 | 32.58 ± 3.45 | 0.264 | |
| Bolting stem diameter (mm) | 17.24 ± 0.89 | 17.86 ± 0.80 | 0.087 | 14.68 ± 1.31 | 15.47 ± 1.14 | 0.139 | |
| Shoot ratio % | 93.02 ± 0.61 | 93.19 ± 0.93 | 0.617 | 97.01 ± 0.41 | 96.90 ± 0.34 | 0.502 | |
| Root | FW (g) | 14.47 ± 2.70 | 16.56 ± 2.33 | 0.058 | 5.50 ± 1.15 | 6.52 ± 1.59 | 0.090 |
| DW (g) | 0.85 ± 0.13 | 0.91 ± 0.13 | 0.275 | 0.32 ± 0.06 | 0.36 ± 0.07 | 0.090 | |
| Length (cm) | 30.35 ± 4.45 | 31.31 ± 3.93 | 0.589 | 27.95 ± 5.81 | 33.17 ± 6.58 | 0.056 | |
| 47 DAS | 50 DAS | |||||
|---|---|---|---|---|---|---|
| 10 °C | 20 °C | p-Value | 10 °C | 20 °C | p-Value | |
| Leaf temperature (°C) | 22.29 ± 0.53 | 22.42 ± 0.43 | 0.588 | 22.17 ± 0.36 | 22.05 ± 0.46 | 0.568 |
| Transpiration rate (mmol H2O·m−2·s−1) | 7.01 ± 0.71 | 5.69 ± 1.12 | 0.016 | 5.60 ± 0.66 | 6.30 ± 0.65 | 0.051 |
| Stomatal conductance (mol H2O·m−2·s−1) | 0.63 ± 0.10 | 0.48 ± 0.16 | 0.043 | 0.46 ± 0.08 | 0.54 ± 0.11 | 0.142 |
| Net photosynthesis rate (µmol CO2·m−2·s−1) | 8.84 ± 0.45 | 8.66 ± 0.57 | 0.486 | 9.03 ± 1.49 | 9.35 ± 0.66 | 0.587 |
| Exp-1 | Exp-2 | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| (mg/g FW) | Treatment | 10 °C | 20 °C | Change [%] | p-Value | 10 °C | 20 °C | Change [%] | p-Value |
| Leaf | Glucose | 3.12 ± 0.68 | 2.59 ± 0.40 | +20.5 | 0.039 | 0.92 ± 0.27 | 0.43 ± 0.22 | +114.0 | <0.001 |
| Fructose | 3.37 ± 0.59 | 2.77 ± 0.31 | +21.7 | 0.009 | 1.15 ± 0.31 | 0.52 ± 0.23 | +121.2 | <0.001 | |
| Sucrose | 1.11 ± 0.38 | 0.86 ± 0.21 | 0.072 | 0.54 ± 0.31 | 0.52 ± 0.33 | 0.907 | |||
| Chlorophyll | 6.02 ± 0.38 | 5.69 ± 0.28 | +5.8 | 0.032 | 5.42 ± 0.54 | 5.31 ± 0.32 | 0.559 | ||
| Starch | 1.52 ± 0.65 | 1.84 ± 1.10 | 0.422 | 0.54 ± 0.23 | 0.32 ± 0.17 | +68.8 | 0.018 | ||
| Soluble sugar | 6.96 ± 2.54 | 5.69 ± 1.92 | 0.183 | 2.61 ± 0.50 | 1.47 ± 0.64 | +77.6 | <0.001 | ||
| Stem | Glucose | 7.35 ± 0.75 | 7.51 ± 0.71 | 0.612 | 9.50 ± 0.89 | 7.12 ± 1.38 | +33.4 | <0.001 | |
| Fructose | 5.62 ± 0.59 | 5.75 ± 0.64 | 0.608 | 7.96 ± 0.69 | 5.95 ± 1.03 | +33.8 | <0.001 | ||
| Sucrose | 2.40 ± 0.40 | 2.66 ± 0.50 | 0.180 | 3.15 ± 0.56 | 1.68 ± 0.46 | +87.5 | <0.001 | ||
| Chlorophyll | 0.37 ± 0.05 | 0.36 ± 0.04 | 0.668 | 0.34 ± 0.05 | 0.31 ± 0.11 | 0.498 | |||
| Starch | 0.21 ± 0.04 | 0.23 ± 0.07 | 0.422 | 0.19 ± 0.05 | 0.15 ± 0.06 | 0.076 | |||
| Soluble sugar | 14.09 ± 4.70 | 15.92 ± 1.36 | 0.217 | 20.61 ± 1.95 | 14.74 ± 2.43 | +39.8 | <0.001 | ||
| (µmol/100 g FW) | Exp-1 | Exp-2 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Treatment | 10 °C | 20 °C | Change [%] | p-Value | 10 °C | 20 °C | Change [%] | p-Value | ||
| Leaf | Aliphatic | Sinigrin | 28.87 ± 7.31 | 24.32 ± 11.25 | 0.276 | 16.72 ± 4.81 | 12.49 ± 2.55 | +33.9 | 0.020 | |
| Progoitrin | 2.47 ± 1.03 | 2.61 ± 1.72 | 0.821 | 1.56 ± 0.66 | 1.31 ± 0.34 | 0.280 | ||||
| Glucoraphanin | 7.27 ± 3.69 | 6.30 ± 4.37 | 0.582 | 3.40 ± 1.61 | 1.61 ± 0.91 | +111.2 | 0.005 | |||
| Gluconapin | 69.86 ± 21.84 | 58.45 ± 30.29 | 0.325 | 49.70 ± 17.22 | 34.39 ± 8.37 | +44.5 | 0.018 | |||
| Glucoiberin | 3.05 ± 1.38 | 2.60 ± 1.47 | 0.477 | 1.38 ± 0.73 | 0.80 ± 0.35 | +72.5 | 0.030 | |||
| Glucoalyssin | 0.15 ± 0.05 | 0.13 ± 0.06 | 0.383 | 0.08 ± 0.03 | 0.05 ± 0.02 | +60.0 | 0.026 | |||
| Total | 111.66 ± 34.66 | 94.42 ± 46.74 | 0.338 | 72.84 ± 23.70 | 50.65 ± 11.62 | +43.8 | 0.014 | |||
| Indolic | Glucobrassicin | 14.56 ± 6.12 | 12.59 ± 7.38 | 0.502 | 6.28 ± 2.52 | 4.31 ± 0.94 | +45.7 | 0.031 | ||
| 4-Methoxyglucobrassicin | 0.81 ± 0.12 | 0.76 ± 0.16 | 0.501 | 1.03 ± 0.26 | 0.74 ± 0.13 | +39.2 | 0.006 | |||
| Neoglucobrassicin | 0.80 ± 0.52 | 0.79 ± 0.61 | 0.988 | 1.37 ± 0.24 | 1.09 ± 0.12 | 0.552 | ||||
| 4-Hydroxyglucobrassicin | 2.37 ± 0.72 | 1.96 ± 0.65 | 0.179 | 1.87 ± 0.85 | 1.74 ± 1.06 | +7.5 | 0.004 | |||
| Total | 18.53 ± 7.03 | 16.10 ± 8.52 | 0.474 | 8.92 ± 2.96 | 6.35 ± 1.23 | +40.5 | 0.019 | |||
| Total | 130.19 ± 39.55 | 110.52 ± 54.71 | 0.347 | 81.76 ± 26.32 | 57.01 ± 12.37 | +43.4 | 0.013 | |||
| Stem | Aliphatic | Sinigrin | 23.75 ± 5.36 | 20.80 ± 4.00 | 0.142 | 18.75 ± 4.86 | 16.31 ± 8.73 | 0.415 | ||
| Progoitrin | 6.32 ± 1.41 | 4.23 ± 1.19 | +49.4 | <0.001 | 2.79 ± 1.03 | 3.19 ± 1.39 | 0.438 | |||
| Glucoraphanin | 11.23 ± 2.26 | 9.92 ± 2.39 | 0.182 | 7.80 ± 2.50 | 7.33 ± 5.08 | 0.777 | ||||
| Gluconapin | 77.15 ± 18.74 | 62.08 ± 14.61 | +24.3 | 0.039 | 51.51 ± 20.01 | 47.74 ± 28.87 | 0.718 | |||
| Glucoiberin | 2.63 ± 0.53 | 2.21 ± 0.64 | 0.098 | 1.87 ± 0.85 | 1.74 ± 1.06 | 0.759 | ||||
| Glucoalyssin | 0.17 ± 0.04 | 0.15 ± 0.02 | 0.099 | 0.12 ± 0.04 | 0.10 ± 0.06 | 0.420 | ||||
| Total | 121.24 ± 25.29 | 99.38 ± 21.67 | +22.0 | 0.033 | 82.84 ± 28.11 | 76.42 ± 44.80 | 0.683 | |||
| Indolic | Glucobrassicin | 4.18 ± 0.88 | 4.03 ± 0.90 | 0.687 | 3.54 ± 1.38 | 3.56 ± 1.81 | 0.971 | |||
| 4-Methoxyglucobrassicin | 2.41 ± 0.33 | 2.03 ± 0.32 | +18.7 | 0.009 | 2.37 ± 0.42 | 1.94 ± 0.52 | +22.2 | 0.039 | ||
| Neoglucobrassicin | 2.57 ± 0.71 | 2.19 ± 0.55 | 0.153 | 0.87 ± 0.46 | 0.96 ± 0.49 | 0.657 | ||||
| 4-Hydroxyglucobrassicin | 3.30 ± 0.77 | 2.49 ± 0.45 | +32.5 | 0.006 | 2.89 ± 0.85 | 2.36 ± 1.30 | 0.258 | |||
| Total | 12.46 ± 2.19 | 10.74 ± 1.67 | +16.0 | 0.042 | 9.67 ± 2.39 | 8.82 ± 3.73 | 0.522 | |||
| Total | 133.70 ± 27.12 | 110.12 ± 22.85 | +21.4 | 0.031 | 92.50 ± 30.34 | 85.24 ± 48.29 | 0.668 | |||
| (%) | Exp-1 | Exp-2 | |||||||
|---|---|---|---|---|---|---|---|---|---|
| 10 °C | 20 °C | Change [%] | p-Value | 10 | 20 °C | Change [%] | p-Value | ||
| Leaf | C | 39.43 ± 0.63 | 39.32 ± 0.95 | 0.727 | 34.92 ± 0.83 | 33.79 ± 0.76 | +3.3 | 0.003 | |
| N | 5.84 ± 0.20 | 6.21 ± 0.22 | −6.0 | <0.001 | 5.92 ± 0.10 | 6.27 ± 0.23 | −5.6 | <0.001 | |
| P | 0.67 ± 0.04 | 0.62 ± 0.05 | +8.1 | 0.012 | 0.50 ± 0.03 | 0.62 ± 0.02 | −19.4 | <0.001 | |
| K | 3.88 ± 0.36 | 4.88 ± 0.76 | −20.5 | <0.001 | 5.18 ± 0.25 | 5.27 ± 0.35 | 0.536 | ||
| Ca | 2.25 ± 0.23 | 2.07 ± 0.28 | 0.106 | 3.74 ± 0.24 | 4.14 ± 0.30 | −9.7 | 0.002 | ||
| Mg | 0.71 ± 0.04 | 0.78 ± 0.10 | −9.0 | 0.033 | 0.48 ± 0.02 | 0.51 ± 0.02 | −5.9 | <0.001 | |
| Stem | C | 38.32 ± 0.63 | 38.42 ± 0.41 | 0.621 | 39.43 ± 0.45 | 38.37 ± 0.43 | +2.8 | <0.001 | |
| N | 3.84 ± 0.30 | 3.60 ± 0.35 | 0.086 | 3.33 ± 0.19 | 3.57 ± 0.12 | −6.7 | 0.003 | ||
| P | 0.42 ± 0.03 | 0.49 ± 0.05 | −14.3 | 0.001 | 0.46 ± 0.02 | 0.53 ± 0.02 | −13.2 | <0.001 | |
| K | 6.15 ± 0.51 | 6.83 ± 0.73 | −10.0 | 0.017 | 5.31 ± 0.38 | 5.88 ± 0.25 | −9.7 | <0.001 | |
| Ca | 0.56 ± 0.08 | 0.60 ± 0.09 | 0.245 | 0.69 ± 0.05 | 0.78 ± 0.09 | −11.5 | 0.006 | ||
| Mg | 0.44 ± 0.05 | 0.43 ± 0.05 | 0.651 | 0.24 ± 0.02 | 0.26 ± 0.02 | −7.7 | 0.020 | ||
| Root | C | -- | -- | -- | -- | 42.63 ± 1.22 | 43.31 ± 1.55 | 0.256 | |
| N | -- | -- | -- | -- | 3.96 ± 0.37 | 3.41 ± 0.34 | +16.1 | 0.001 | |
| P | -- | -- | -- | -- | 0.41 ± 0.03 | 0.32 ± 0.04 | +28.1 | <0.001 | |
| K | -- | -- | -- | -- | 0.05 ± 0.02 | 0.04 ± 0.02 | 0.143 | ||
| Ca | -- | -- | -- | -- | 1.01 ± 0.13 | 0.97 ± 0.15 | 0.463 | ||
| Mg | -- | -- | -- | -- | 0.12 ± 0.01 | 0.12 ± 0.01 | 0.263 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, F.; Thiele, B.; Kraus, D.; Bouteyine, S.; Watt, M.; Kraska, T.; Schurr, U.; Kuhn, A.J. Effects of Short-Term Root Cooling before Harvest on Yield and Food Quality of Chinese Broccoli (Brassica oleracea var. Alboglabra Bailey). Agronomy 2021, 11, 577. https://doi.org/10.3390/agronomy11030577
He F, Thiele B, Kraus D, Bouteyine S, Watt M, Kraska T, Schurr U, Kuhn AJ. Effects of Short-Term Root Cooling before Harvest on Yield and Food Quality of Chinese Broccoli (Brassica oleracea var. Alboglabra Bailey). Agronomy. 2021; 11(3):577. https://doi.org/10.3390/agronomy11030577
Chicago/Turabian StyleHe, Fang, Björn Thiele, David Kraus, Souhaila Bouteyine, Michelle Watt, Thorsten Kraska, Ulrich Schurr, and Arnd Jürgen Kuhn. 2021. "Effects of Short-Term Root Cooling before Harvest on Yield and Food Quality of Chinese Broccoli (Brassica oleracea var. Alboglabra Bailey)" Agronomy 11, no. 3: 577. https://doi.org/10.3390/agronomy11030577
APA StyleHe, F., Thiele, B., Kraus, D., Bouteyine, S., Watt, M., Kraska, T., Schurr, U., & Kuhn, A. J. (2021). Effects of Short-Term Root Cooling before Harvest on Yield and Food Quality of Chinese Broccoli (Brassica oleracea var. Alboglabra Bailey). Agronomy, 11(3), 577. https://doi.org/10.3390/agronomy11030577








