Can Nitrogen Fertilizer Management Improve Grain Iron Concentration of Agro-Biofortified Crops in Zimbabwe?
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Sites
2.2. Rationale for Field Selection and Experimental Design
2.3. Data Analysis
- 1.
- N application influences grain yield and grain Fe concentration when Fe fertilizer is also applied. This is contrast C1 tested with 1 df as: T1 vs. (T2,T3,T4,T5,T6,T7) (Table 5).
- 2.
- The effect of applied N on grain yield and grain Fe concentration depends on the total amount of N applied. This is contrast C2 tested with 1 df: (T2,T3,T4) vs. (T5,T6,T7) (equivalent to the main effect of N rate in the factorial subset of treatments) (Table 5).
- 3.
- The effect of applied N on grain yield and grain Fe concentration depends on the application strategy (organic, mineral, or mixed). This was tested with two more specific hypotheses as follows:
- 3.1.
- There are differences in grain yield and grain Fe concentration when N is applied as organic (T3 and T6) or as mixed organic and mineral N fertilizer (T4 and T7). This is contrast C3 with 1 df (Table 5).
- 3.2.
- There are differences in grain yield and grain Fe concentration when N is applied as mineral (T2 and T5) or as organic N (including mixed N treatments) (T3,T4,T6,T7). This is contrast C4 with 1 df (Table 5).
- 4.
- The effect of N application rate on grain yield and grain Fe concentration depends on the strategy. This was tested more specifically as:
- 4.1.
- The difference between the effect of applying sole organic N and applying mixed N depends on whether the overall rate of application of N is high (T6 and T7) or low (T3 and T4); this is contrast C5.
- 4.2.
- The difference between the effect of applying sole mineral N fertilizer and applying sole organic N (including mixed N) fertilizer depends on whether the overall rate of application of N is high (T5,T6,T7) or low (T2,T3,T4); this is contrast C6 with 1 df (Table 5).
3. Results
3.1. Effect of N Management Strategy and Fe Fertilization on Grain Yields
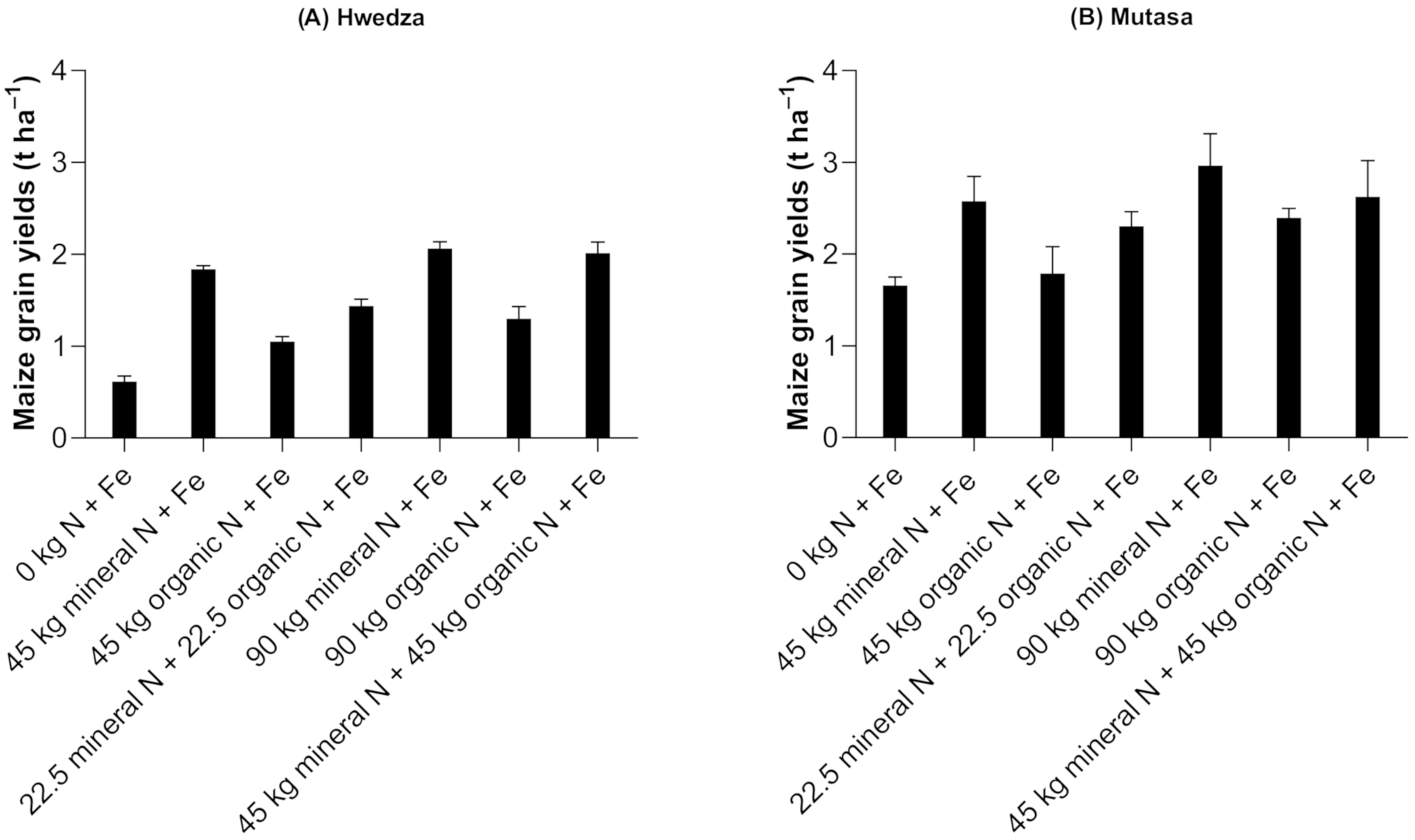
3.1.1. Maize Grain Yields
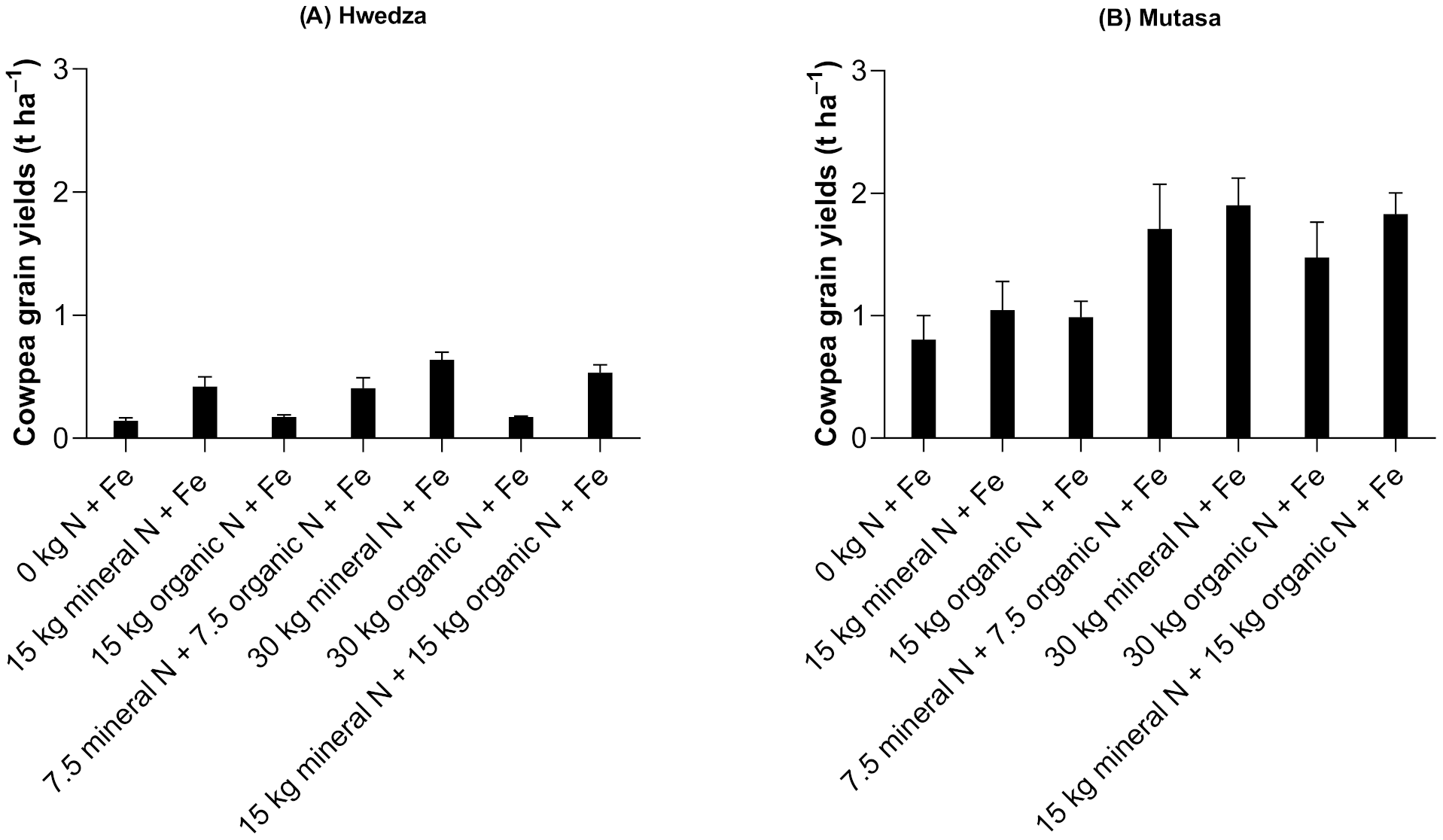
3.1.2. Cowpea Grain Yields
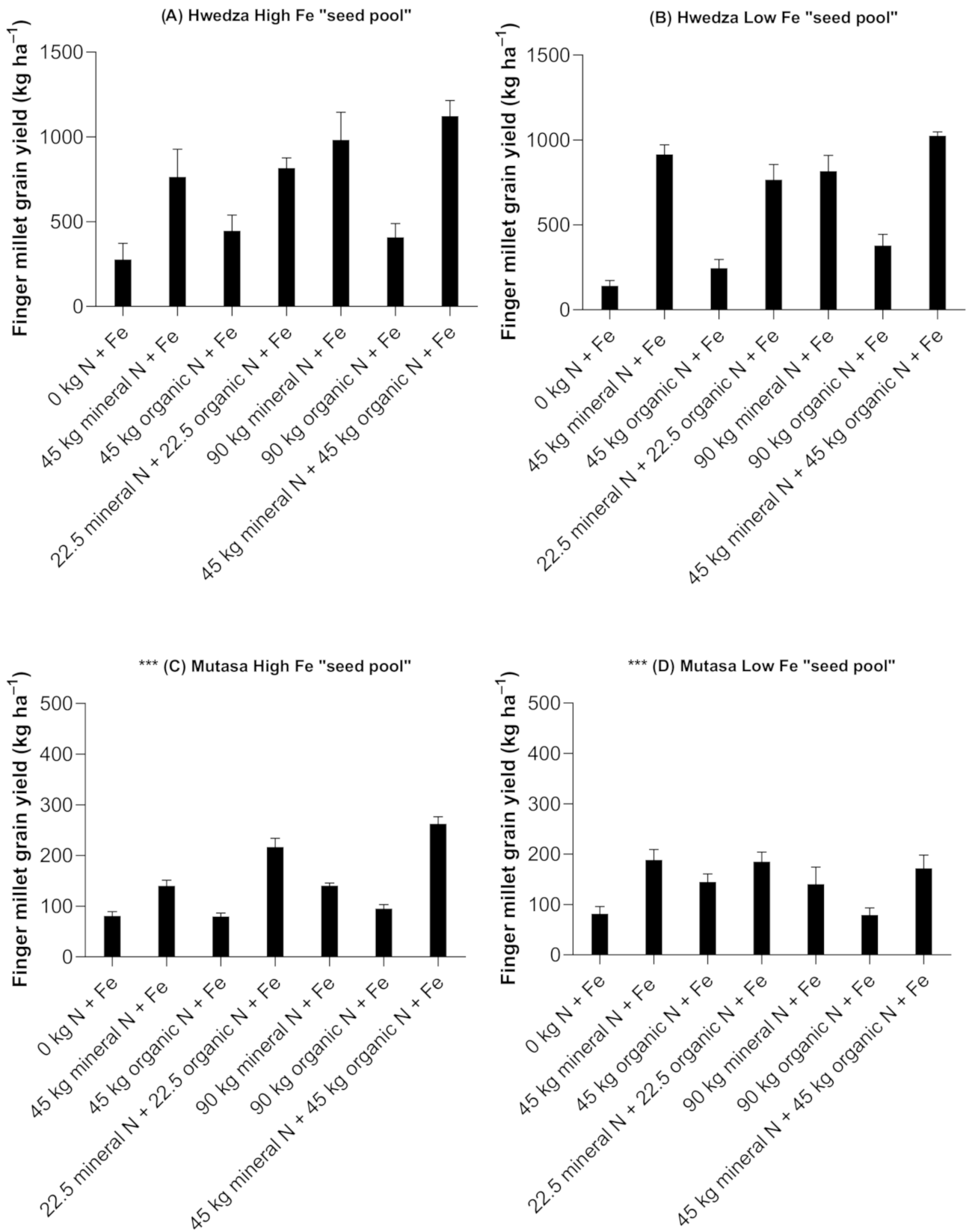
3.2. Finger Millet Grain Yields
3.2.1. High Fe “Seed Pool”
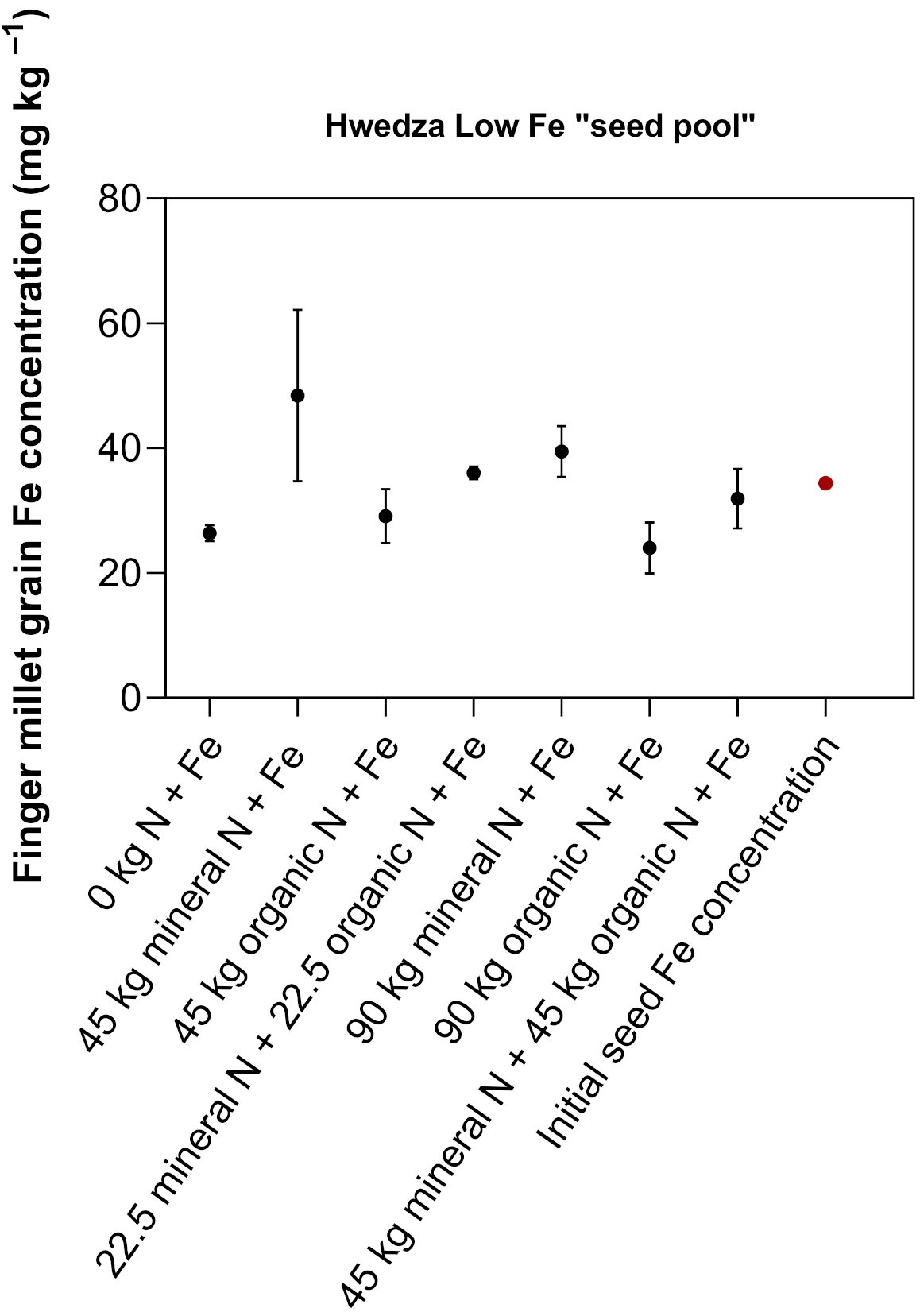
3.2.2. Low Fe “Seed Pool”
3.3. Influence of N Fertilization on Grain Fe Concentration of Maize, Cowpea, and Finger Millet
4. Discussion
4.1. Nitrogen Management Remains Crucial in Cereal and Legume Grain Yields Even When Fe is Supplied
4.2. Grain Fe Nutrition is Governed by Various Agronomic and Site-Specific Factors
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ems, T.; Lucia, K.; Huecker, M.R. Biochemistry, Iron Aabsorption; StatPearls: Treasure Island, FL, USA, 2020. [Google Scholar]
- Abbaspour, N.; Hurrell, R.; Kelishadi, R. Review on iron and its importance for human health. J. Res. Med. Sci. 2014, 19, 164–174. [Google Scholar] [PubMed]
- World Health Organization (WHO). Conclusions and recommendations of the WHO consultation on prevention and control of iron deficiency in infants and young children in malaria-endemic areas. Food Nutr. Bull. 2007, 28, S621–S627. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.L. Iron deficiency anemia: A common and curable disease. Cold Spring Harb. Perspect. Med. 2013, 3. [Google Scholar] [CrossRef] [PubMed]
- ZNSA and ICF. Zimbabwe Demographic and Health Survey 2015; Final Report for Zimbabwe National Statistics Agency (ZIMSTAT) and ICF International: Rockville, MD, USA, 2016; pp. 193–198. [Google Scholar]
- Food and Agriculture Organization of the United Nations. Current Worldwide Annual Meat Consumption per Capita; FAO: Rome, Italy, 2013. [Google Scholar]
- Kitalyi, A.J. Village Chicken Production Systems in Rural Africa: Household Food Security and Gender Issues. In FAO Animal Production and Health Paper 142; Food and Agriculture Organization: Rome, Italy, 1998; ISBN 92-5-104160-1. [Google Scholar]
- Katsidzira, L.; Laubscher, R.; Gangaidzo, I.T.; Swart, R.; Mutasa, R.; Manyanga, T.; Thomson, S.; Ramesar, R.; Matenga, J.A.; Rusakaniko, S. Dietary patterns and colorectal cancer risk in Zimbabwe: A population-based case-control study. Cancer Epidemiol. 2018, 57, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Bouis, H.E.; Saltzman, A. Improving nutrition through biofortification: A review of evidence from HarvestPlus, 2003 through 2016. Glob. Food Secur. 2017, 12, 49–58. [Google Scholar] [CrossRef]
- Scott, S.; Murray-Kolb, L.E.; Wenger, M.J.; A Udipi, S.; Ghugre, P.S.; Boy, E.; Haas, J.D. Cognitive Performance in Indian School-Going Adolescents Is Positively Affected by Consumption of Iron-Biofortified Pearl Millet: A 6-Month Randomized Controlled Efficacy Trial. J. Nutr. 2018, 148, 1462–1471. [Google Scholar] [CrossRef]
- Finkelstein, J.L.; Fothergill, A.; Hackl, L.S.; Haas, J.D.; Mehta, S. Iron biofortification interventions to improve iron status and functional outcomes. Proc. Nutr. Soc. 2019, 78, 197–207. [Google Scholar] [CrossRef]
- Fang, Y.; Wang, L.; Xin, Z.; Zhao, L.; An, X.; Hu, Q. Effect of Foliar Application of Zinc, Selenium, and Iron Fertilizers on Nutrients Concentration and Yield of Rice Grain in China. J. Agric. Food Chem. 2008, 56, 2079–2084. [Google Scholar] [CrossRef]
- Niyigaba, E.; Twizerimana, A.; Mugenzi, I.; Ngnadong, W.A.; Ye, Y.P.; Wu, B.M.; Hai, J.B. Winter Wheat Grain Quality, Zinc and Iron Concentration Affected by a Combined Foliar Spray of Zinc and Iron Fertilizers. Agronomy 2019, 9, 250. [Google Scholar] [CrossRef]
- Zou, C.; Du, Y.; Rashid, A.; Ram, H.; Savasli, E.; Pieterse, P.I.; Ortiz-Monasterio, I.; Yazici, A.; Kaur, C.; Mahmood, K.; et al. Simultaneous biofortification of wheat with zinc, iodine, selenium, and iron through foliar treatment of a micronutrient cocktail in six countries. J. Agric. Food Chem. 2019, 67, 8096–8106. [Google Scholar] [CrossRef]
- Godsey, C.B.; Schmidt, J.P.; Schlegel, A.J.; Taylor, R.K.; Thompson, C.R.; Gehl, R.J. Correcting Iron Deficiency in Corn with Seed Row–Applied Iron Sulfate. Agron. J. 2003, 95, 160–166. [Google Scholar] [CrossRef]
- Joy, E.J.M.; Stein, A.J.; Young, S.D.; Ander, E.L.; Watts, M.J.; Broadley, M.R. Zinc-enriched fertilisers as a potential public health intervention in Africa. Plant Soil 2015, 389, 1–24. [Google Scholar] [CrossRef]
- Manzeke, G.M.; Mtambanengwe, F.; Nezomba, H.; Mapfumo, P. Zinc fertilization influence on maize productivity and grain nutritional quality under integrated soil fertility management in Zimbabwe. Field Crop. Res. 2014, 166, 128–136. [Google Scholar] [CrossRef]
- Manzeke, M.G.; Mtambanengwe, F.; Nezomba, H.; Watts, M.J.; Broadley, M.R.; Mapfumo, P. Zinc fertilization increases productivity and grain nutritional quality of cowpea (Vigna unguiculata [L.] Walp.) under integrated soil fertility management. Field Crop. Res. 2017, 213, 231–244. [Google Scholar] [CrossRef]
- Manzeke, M.G.; Mtambanengwe, F.; Watts, M.J.; Broadley, M.R.; Lark, R.M.; Mapfumo, P. Nitrogen effect on efficient agronomic zinc biofortification of maize and cowpea in Zimbabwean smallholder farming systems. Agron. J. 2020, 112, 2256–2274. [Google Scholar] [CrossRef]
- Cakmak, I. Enrichment of cereal grains with zinc: Agronomic or genetic biofortification? Plant Soil 2008, 302, 1–17. [Google Scholar] [CrossRef]
- Kutman, U.B.; Yildiz, B.; Ozturk, L.; Cakmak, I. Biofortification of Durum Wheat with Zinc through Soil and Foliar Applications of Nitrogen. Cereal Chem. J. 2010, 87, 1–9. [Google Scholar] [CrossRef]
- Márquez-Quiroz, C.; de la Cruz-Lázaro, E.; Osorio-Osorio, R.; Sánchez-Chávez, E. Biofortification of cowpea beans with iron: Iron´s influence on mineral content and yield. J. soil Sci. Plant Nutr. 2015, 15, 839–847. [Google Scholar] [CrossRef]
- Mathers, A.C. Effect of ferrous sulfate and sulfuric acid on grain sorghum yields. Agron. J. 1970, 62, 555–556. [Google Scholar] [CrossRef]
- Manzeke, G.M.; Mapfumo, P.; Mtambanengwe, F.; Chikowo, R.; Tendayi, T.; Cakmak, I. Soil fertility management effects on maize productivity and grain zinc content in smallholder farming systems of Zimbabwe. Plant Soil 2012, 361, 57–69. [Google Scholar] [CrossRef]
- Manzeke, M.G.; Mtambanengwe, F.; Watts, M.J.; Hamilton, E.M.; Lark, R.M.; Broadley, M.R.; Mapfumo, P. Fertilizer management and soil type influence grain zinc and iron concentration under contrasting smallholder cropping systems in Zimbabwe. Sci. Rep. 2019, 9, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Kutman, U.B.; Yildiz, B.; Cakmak, I. Effect of nitrogen on uptake, remobilization and partitioning of zinc and iron throughout the development of durum wheat. Plant Soil 2011, 342, 149–164. [Google Scholar] [CrossRef]
- Kutman, U.B.; Yildiz, B.; Cakmak, I.; Yildiz-Kutman, B. Improved nitrogen status enhances zinc and iron concentrations both in the whole grain and the endosperm fraction of wheat. J. Cereal Sci. 2011, 53, 118–125. [Google Scholar] [CrossRef]
- Allen, L.H.; Carriquiry, A.L.; Murphy, S.P. Perspective: Proposed Harmonized Nutrient Reference Values for Populations. Adv. Nutr. 2019, 11, 469–483. [Google Scholar] [CrossRef]
- FAO. Fertiliser use by crop in Zimbabwe. Land and Plant Nutrition Management Service, Land and Water Development Division; Food and Agricultural Organization of the United Nations: Rome, Italy, 2006; Available online: http://www.fao.org/3/a0395e/a0395e00.htm (accessed on 6 July 2020).
- Lindsay, W.L.; Norvell, W.A. Development of a DTPA Soil Test for Zinc, Iron, Manganese, and Copper. Soil Sci. Soc. Am. J. 1978, 42, 421–428. [Google Scholar] [CrossRef]
- Kanonge, G.; Mtambanengwe, F.; Nezomba, H.; Manzeke, M.G.; Mapfumo, P. Assessing the potential benefits of organic and mineral fertilizer combinations on legume productivity under smallholder management in Zimbabwe. S. Afr. J. Plant Soil 2015, 32, 241–248. [Google Scholar] [CrossRef]
- Mtambanengwe, F.; Mapfumo, P. Organic Matter Management as an Underlying Cause for Soil Fertility Gradients on Smallholder Farms in Zimbabwe. Nutr. Cycl. Agroecosyst. 2005, 73, 227–243. [Google Scholar] [CrossRef]
- Yunta, F.; Martín, I.; Lucena, J.J.; Gárate, A. Iron Chelates Supplied Foliarly Improve the Iron Translocation Rate in Tempranillo Grapevine. Commun. Soil Sci. Plant Anal. 2012, 44, 794–804. [Google Scholar] [CrossRef]
- Lancashire, P.D.; Bleiholder, H.; Langelüddecke, P.; Stauss, R.; van den Boom, T.; Weber, E.; Witzen-Berger, A. A uniform decimal code for growth stages of crops and weeds. Ann. Appl. Biol. 1991, 119, 561–601. [Google Scholar] [CrossRef]
- Kurwakumire, N.; Chikowo, R.; Mtambanengwe, F.; Mapfumo, P.; Snapp, S.S.; Johnston, A.; Zingore, S. Maize productivity and nutrient and water use efficiencies across soil fertility domains on smallholder farms in Zimbabwe. Field Crop. Res. 2014, 164, 136–147. [Google Scholar] [CrossRef]
- Watts, M.J.; Middleton, D.R.S.; Marriott, A.L.; Humphrey, O.S.; Hamilton, E.M.; Gardner, A.; Smith, J.A.; McCormack, V.A.; Menya, D.; Munishi, M.O.; et al. Source apportionment of micronutrients in the diets of Kilimanjaro, Tanzania and Counties of Western Kenya. Sci. Rep. 2019, 9, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Joy, E.J.; Broadley, M.R.; Young, S.D.; Black, C.R.; Chilimba, A.D.; Ander, E.L.; Barlow, T.S.; Watts, M.J. Soil type influences crop mineral composition in Malawi. Sci. Total. Environ. 2015, 505, 587–595. [Google Scholar] [CrossRef] [PubMed]
- Okalebo, J.R.; Gathua, K.W.; Woomer, P.L. Laboratory Methods of Soil and Plant Analysis: A Working Manual, 2nd ed.; Sacred Africa: Nairobi, Kenya, 2002. [Google Scholar]
- Anderson, J.M.; Ingram, J.S.I. Tropical soil biology and fertility. In A Handbook of Methods, 2nd ed.; CAB International: Wallingford, UK, 1993. [Google Scholar]
- Murphy, J.; Riley, J.P. A modified single solution method for the determination of phosphate in natural waters. Anal. Chim. Acta 1962, 27, 31–36. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018; Available online: https://www.R-project.org/ (accessed on 10 November 2020).
- Nyamangara, J.; Mudhara, M.; Giller, K.E. Effectiveness of cattle manure and nitrogen fertilizer application on the agronomic and economic performance of maize. S. Afr. J. Plant Soil 2005, 22, 59–63. [Google Scholar] [CrossRef]
- Dunjana, N.; Nyamugafata, P.; Nyamangara, J.; Mango, N. Cattle manure and inorganic nitrogen fertilizer application effects on soil hydraulic properties and maize yield of two soils of Murewa district, Zimbabwe. Soil Use Manag. 2014, 30, 579–587. [Google Scholar] [CrossRef]
- Mtangadura, T.J.; Mtambanengwe, F.; Nezomba, H.; Rurinda, J.; Mapfumo, P. Why organic resources and current fertilizer formulations in Southern Africa cannot sustain maize productivity: Evidence from a long-term experiment in Zimbabwe. PLoS ONE 2017, 12, e0182840. [Google Scholar] [CrossRef] [PubMed]
- Mtambanengwe, F.; Mapfumo, P. Effects of Organic Resource Quality on Soil Profile N Dynamics and Maize Yields on Sandy Soils in Zimbabwe. Plant Soil 2006, 281, 173–191. [Google Scholar] [CrossRef]
- Jency, J.P.; Rajasekaran, R.; Singh, R.K.; Muthurajan, R.; Prabhakaran, J.; Mehanathan, M.; Prasad, M.; Ganesan, J. Induced mutagenesis enhances lodging resistance and photosynthetic efficiency of kodo millet (Paspalum Scrobiculatum). Agronomy 2020, 10, 227. [Google Scholar] [CrossRef]
- Li, P.; Brutnell, T.P. Setaria viridis and Setaria italica, model genetic systems for the Panicoid grasses. J. Exp. Bot. 2011, 62, 3031–3037. [Google Scholar] [CrossRef]
- Rurinda, J.; Mapfumo, P.; van Wijk, M.; Mtambanengwe, F.; Rufino, M.C.; Chikowo, R.; E Giller, K. Comparative assessment of maize, finger millet and sorghum for household food security in the face of increasing climatic risk. Eur. J. Agron. 2014, 55, 29–41. [Google Scholar] [CrossRef]
- Muthamilarasan, M.; Prasad, M. Small Millets for Enduring Food Security Amidst Pandemics. Trends Plant Sci. 2020. [Google Scholar] [CrossRef] [PubMed]
- Waters, B.M.; Chu, H.-H.; DiDonato, R.J.; Roberts, L.A.; Eisley, R.B.; Lahner, B.; Salt, D.E.; Walker, E. Mutations in Arabidopsis Yellow Stripe-Like1 and Yellow Stripe-Like3 Reveal Their Roles in Metal Ion Homeostasis and Loading of Metal Ions in Seeds. Plant Physiol. 2006, 141, 1446–1458. [Google Scholar] [CrossRef] [PubMed]
- Borg, S.; Brinch-Pedersen, H.; Tauris, B.; Holm, P.B. Iron transport, deposition and bioavailability in the wheat and barley grain. Plant Soil 2009, 325, 15–24. [Google Scholar] [CrossRef]
- Pahlavan-Rad, M.R.; Pessarakli, M. Response of Wheat Plants to Zinc, Iron, and Manganese Applications and Uptake and Concentration of Zinc, Iron, and Manganese in Wheat Grains. Commun. Soil Sci. Plant Anal. 2009, 40, 1322–1332. [Google Scholar] [CrossRef]
- Majeed, A.; Minhas, W.A.; Mehboob, N.; Farooq, S.; Hussain, M.; Cheema, S.A.; Rizwan, M.S. Iron application improves yield, economic returns and grain-Fe concentration of mungbean. PLoS ONE 2020, 15, e0230720. [Google Scholar] [CrossRef]
- Zhao, Q.-Y.; Xu, S.-J.; Zhang, W.-S.; Zhang, Z.; Yao, Z.; Chen, X.-P.; Zou, C. Identifying key drivers for geospatial variation of grain micronutrient concentrations in major maize production regions of China. Environ. Pollut. 2020, 266, 115114. [Google Scholar] [CrossRef]
- Mnyenyembe, P.H. Past and present research on finger millet in Malawi. In Advances in Small Millets; Riley, K.W., Gupta, S.C., Seetharam, A., Mushonga, J.N., Eds.; International Science Publisher: New York, NY, USA, 1994; pp. 29–37. [Google Scholar]
- Gabaza, M.; Muchuweti, M.; Vandamme, P.; Raes, K. Can fermentation be used as a sustainable strategy to reduce iron and zinc binders in traditional African fermented cereal porridges or gruels? Food Rev. Int. 2016, 33, 561–586. [Google Scholar] [CrossRef]
- Bouis, H.E.; Welch, R.M. Biofortification-A Sustainable Agricultural Strategy for Reducing Micronutrient Malnutrition in the Global South. Crop. Sci. 2010, 50, S20. [Google Scholar] [CrossRef]
- Morrissey, J.; Guerinot, M.L. Iron Uptake and Transport in Plants: The Good, the Bad, and the Ionome. Chem. Rev. 2009, 109, 4553–4567. [Google Scholar] [CrossRef]
- Nikolić, M.; Nikolic, N.; Kostić, L.; Pavlovic, J.; Bosnic, P.; Stevic, N.; Savic, J.; Hristov, N. The assessment of soil availability and wheat grain status of zinc and iron in Serbia: Implications for human nutrition. Sci. Total. Environ. 2016, 553, 141–148. [Google Scholar] [CrossRef]
- Shenker, M.; Chen, Y. Increasing Iron Availability to Crops: Fertilizers, Organo-Fertilizers, and Biological Approaches. Soil Sci. Plant Nutr. 2005, 51, 1–17. [Google Scholar] [CrossRef]
| Property | Hwedza | Mutasa | |
|---|---|---|---|
| Site 1 | Site 2 | ||
| Clay content (%) | 35 | 50 | 55 |
| Sand content (%) | 55 | 35 | 35 |
| Available P (mg kg−1) | 8.7 | 10.3 | 15.4 |
| Available N (mg kg−1) | 21.9 | 27.3 | 31.4 |
| † Available Fe (mg kg−1) | 9.3 | 11.9 | 15.4 |
| Total Fe (mg kg−1) | 9.3 × 104 | 3.9 × 104 | 1.6 × 105 |
| ¥ SOM (%) | 5.0 | 7.5 | 7.8 |
| Soil pH (0.01 M CaCl2) | 4.7 | 4.4 | 4.6 |
| Crops grown | Maize, cowpea, and finger millet | Maize and cowpea | Finger millet |
| Number of plots per crop | 28 | 28 | 28 |
| Number of plots per site | 112 | 56 | 56 |
| Treatment Code | Treatment |
|---|---|
| T1 | 0 N ha−1 + Fe |
| T2 | 45 kg mineral N fertilizer ha−1 * + Fe |
| T3 | 45 kg organic N fertilizer ha−1 + Fe |
| T4 | 22.5 kg mineral N fertilizer ha−1 + 22.5 kg organic N fertilizer ha−1 + Fe |
| T5 | 90 kg mineral N fertilizer ha−1 + Fe |
| T6 | 90 kg organic N fertilizer ha−1 + Fe |
| T7 | 45 kg mineral N fertilizer ha−1 + 45 kg organic N fertilizer ha−1 + Fe |
| Treatment Code | Treatment |
|---|---|
| T1 | 0 N ha−1 * + Fe |
| T2 | 15 kg mineral N fertilizer ha−1 + Fe |
| T3 | 15 kg organic N fertilizer ha−1 + Fe |
| T4 | 7.5 kg mineral N fertilizer ha−1 + 7.5 kg organic N fertilizer ha−1 + Fe |
| T5 | 30 kg mineral N fertilizer ha−1 + Fe |
| T6 | 30 kg organic N fertilizer ha−1 + Fe |
| T7 | 15 kg mineral N fertilizer ha−1 + 15 kg organic N fertilizer ha−1 + Fe |
| Property | Hwedza | Mutasa |
|---|---|---|
| a Total Zn (mg kg−1) | 10.0 | 6.0 |
| a Total Fe (g kg−1) | 10.4 | 5.0 |
| b Total N (g kg−1) | 10.1 | 16.1 |
| c Total P (g kg−1) | 2.3 | 0.8 |
| d Total K (g kg−1) | 4.3 | 2.6 |
| d Total Ca (mg kg−1) | 454 | 401 |
| d Total Mg (mg kg−1) | 1930 | 1196 |
| d Total Na (mg kg−1) | 257 | 101 |
| e Organic carbon (C, g kg−1) | 243 | 319 |
| C:N ratio | 24.1 | 19.8 |
| Contrast | Comparison | * df |
|---|---|---|
| C1 | 0 N versus some N fertilizer application | 1 |
| C2 | Low N versus High N fertilizer | 1 |
| C3 | Organic N fertilizer versus mixed N fertilizer | 1 |
| C4 | Mineral N fertilizer versus (organic N and mixed N) fertilizer | 1 |
| C5 | (Organic versus mixed N) fertilizer * (High versus Low N) fertilizer | 1 |
| C6 | (Mineral N fertilizer versus (Mixed and organic N)) fertilizer * (High versus Low N) fertilizer | 1 |
| A. Hwedza | B. Mutasa | ||||||
|---|---|---|---|---|---|---|---|
| Contrast | Comparison | d.f | Sum Sq | p-Value | d.f | Sum Sq | p-Value |
| C1 | 0 N vs. some N application | 1 | 3.84 | 0.030 | 1 | 1.55 | 0.039 |
| C2 | Low N vs. High N | 1 | 0.91 | 0.355 | 1 | 0.29 | 0.351 |
| C3 | Organic N vs. mixed N | 1 | 1.39 | 0.036 | 1 | 0.55 | 0.200 |
| C4 | Mineral N vs. (mixed N and organic N) | 1 | 1.85 | 0.012 | 1 | 1.25 | 0.062 |
| C5 | (Organic vs. mixed N) ● (High vs. Low N) | 1 | 0.90 | 0.956 | 1 | 0.86 | 0.116 |
| C6 | (Mineral N vs. (Mixed and organic N)) ● (High vs. Low N) | 1 | 0.04 | 0.694 | 1 | 0.07 | 0.647 |
| Block | Blocking effect | 2 | 1.25 | n.a. | 3 | 0.30 | n.a. |
| Residuals | Residuals | 18 | 3.98 | n.a. | 16 | 4.972 | n.a. |
| A. Hwedza | B. Mutasa | ||||||
|---|---|---|---|---|---|---|---|
| Contrast | Comparison | d.f | Sum Sq | p-Value | d.f | Sum Sq | p-Value |
| C1 | 0 N vs. some N application | 1 | 0.159 | 0.0059 | 1 | 1.615 | 0.020 |
| C2 | Low N vs. High N | 1 | 0.059 | 0.065 | 1 | 1.428 | 0.028 |
| C3 | Organic N vs. mixed N | 1 | 0.262 | 0.001 | 1 | 1.149 | 0.046 |
| C4 | Mineral N vs. (mixed N and organic N) | 1 | 0.173 | 0.004 | 1 | 0.0037 | 0.904 |
| C5 | (Organic vs. mixed N) ● (High vs. Low N) | 1 | 0.011 | 0.378 | 1 | 0.1335 | 0.475 |
| C6 | (Mineral N vs. (Mixed and organic N)) ● (High vs. Low N) | 1 | 0.023 | 0.227 | 1 | 0.4052 | 0.219 |
| Block | Blocking effect | 2 | 0.006 | n.a. | 3 | 4.509 | n.a. |
| Residuals | Residuals | 12 | 0.173 | n.a. | 18 | 0.387 | n.a. |
| High Fe “Seed Pool” | |||||||
|---|---|---|---|---|---|---|---|
| Contrast | Comparison | A. Hwedza | B. Mutasa | ||||
| d.f | Sum Sq | p-Value | d.f | Sum Sq | p-Value | ||
| C1 | 0 N vs. some N application | 1 | 788,814 | 0.0016 | 1 | 1.1459 | <0.0001 |
| C2 | Low N vs. High N | 1 | 157,324 | 0.1139 | 1 | 0.0980 | 0.06695 |
| C3 | Organic N vs. mixed N | 1 | 1,177,863 | 0.00025 | 1 | 4.0996 | <0.0001 |
| C4 | Mineral N vs. (mixed N and organic N) | 1 | 161,964 | 0.1091 | 1 | 0.0030 | 0.7373 |
| C5 | (Organic vs. mixed N) ● (High vs. Low N) | 1 | 119,093 | 0.1655 | 1 | 0.0003 | 0.9115 |
| C6 | (Mineral N vs. (Mixed and organic N)) ● (High vs. Low N) | 1 | 9478 | 0.688 | 1 | 0.0417 | 0.2196 |
| Block | Blocking effect | 3 | 52,897 | n.a. | 3 | 0.0729 | n.a. |
| Residuals | Residuals | 18 | 1,025,969 | n.a. | 18 | 0.4642 | n.a. |
| Low Fe “Seed Pool” | |||||||
|---|---|---|---|---|---|---|---|
| Contrast | Comparison | A. Hwedza | B. Mutasa | ||||
| d.f | Sum Sq | p-Value | d.f | Sum Sq | p-Value | ||
| C1 | 0 N vs. some N application | 1 | 1,036,219 | <0.0001 | 1 | 0.21065 | 0.002 |
| C2 | Low N vs. High N | 1 | 57,478 | 0.087 | 1 | 0.76236 | 0.011 |
| C3 | Organic N vs. mixed N | 1 | 1,364,545 | <0.0001 | 1 | 1.05983 | 0.003 |
| C4 | Mineral N vs. (mixed N and organic N) | 1 | 364,418 | <0.0001 | 1 | 0.10560 | 0.3079 |
| C5 | (Organic vs. mixed N) ● (High vs. Low N) | 1 | 15,381 | 0.3618 | 1 | 0.28978 | 0.099 |
| C6 | (Mineral N vs. (Mixed and organic N)) ● (High vs. Low N) | 1 | 116,303 | 0.019 | 1 | 0.0010 | 0.998 |
| Block | Blocking effect | 3 | 25,630 | n.a. | 3 | 0.28180 | n.a |
| Residuals | Residuals | 18 | 316,259 | n.a. | 18 | 1.726 | n.a. |
| Contrast | Comparison | d.f | Sum Sq | p-Value |
|---|---|---|---|---|
| C1 | 0 N vs. some N application | 1 | 0.161 | 0.160 |
| C2 | Low N vs. High N | 1 | 0.161 | 0.159 |
| C3 | Organic N vs. mixed N | 1 | 0.308 | 0.058 |
| C4 | Mineral N vs. (mixed N and organic N) | 1 | 0.651 | 0.008 |
| C5 | (Organic vs. mixed N) ● (High vs. Low N) | 1 | 0.004 | 0.822 |
| C6 | (Mineral N vs. (Mixed and organic N)) ● (High vs. Low N) | 1 | 0.005 | 0.805 |
| Block | Blocking effect | 3 | 0.563 | n.a. |
| Residuals | Residuals | 18 | 1.349 | n.a. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manzeke-Kangara, M.G.; Mtambanengwe, F.; Watts, M.J.; Broadley, M.R.; Lark, R.M.; Mapfumo, P. Can Nitrogen Fertilizer Management Improve Grain Iron Concentration of Agro-Biofortified Crops in Zimbabwe? Agronomy 2021, 11, 124. https://doi.org/10.3390/agronomy11010124
Manzeke-Kangara MG, Mtambanengwe F, Watts MJ, Broadley MR, Lark RM, Mapfumo P. Can Nitrogen Fertilizer Management Improve Grain Iron Concentration of Agro-Biofortified Crops in Zimbabwe? Agronomy. 2021; 11(1):124. https://doi.org/10.3390/agronomy11010124
Chicago/Turabian StyleManzeke-Kangara, Muneta G., Florence Mtambanengwe, Michael J. Watts, Martin R. Broadley, R. Murray Lark, and Paul Mapfumo. 2021. "Can Nitrogen Fertilizer Management Improve Grain Iron Concentration of Agro-Biofortified Crops in Zimbabwe?" Agronomy 11, no. 1: 124. https://doi.org/10.3390/agronomy11010124
APA StyleManzeke-Kangara, M. G., Mtambanengwe, F., Watts, M. J., Broadley, M. R., Lark, R. M., & Mapfumo, P. (2021). Can Nitrogen Fertilizer Management Improve Grain Iron Concentration of Agro-Biofortified Crops in Zimbabwe? Agronomy, 11(1), 124. https://doi.org/10.3390/agronomy11010124





