Potassium Fertilisation Is Required to Sustain Cassava Yield and Soil Fertility
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design
2.2. Fresh Weights, Stem Lengths and Estimates of Leaf Loss
2.3. Starch Content
2.4. Analysis of K and Other Nutrients
2.5. Data Analysis
3. Results
3.1. Plant Growth
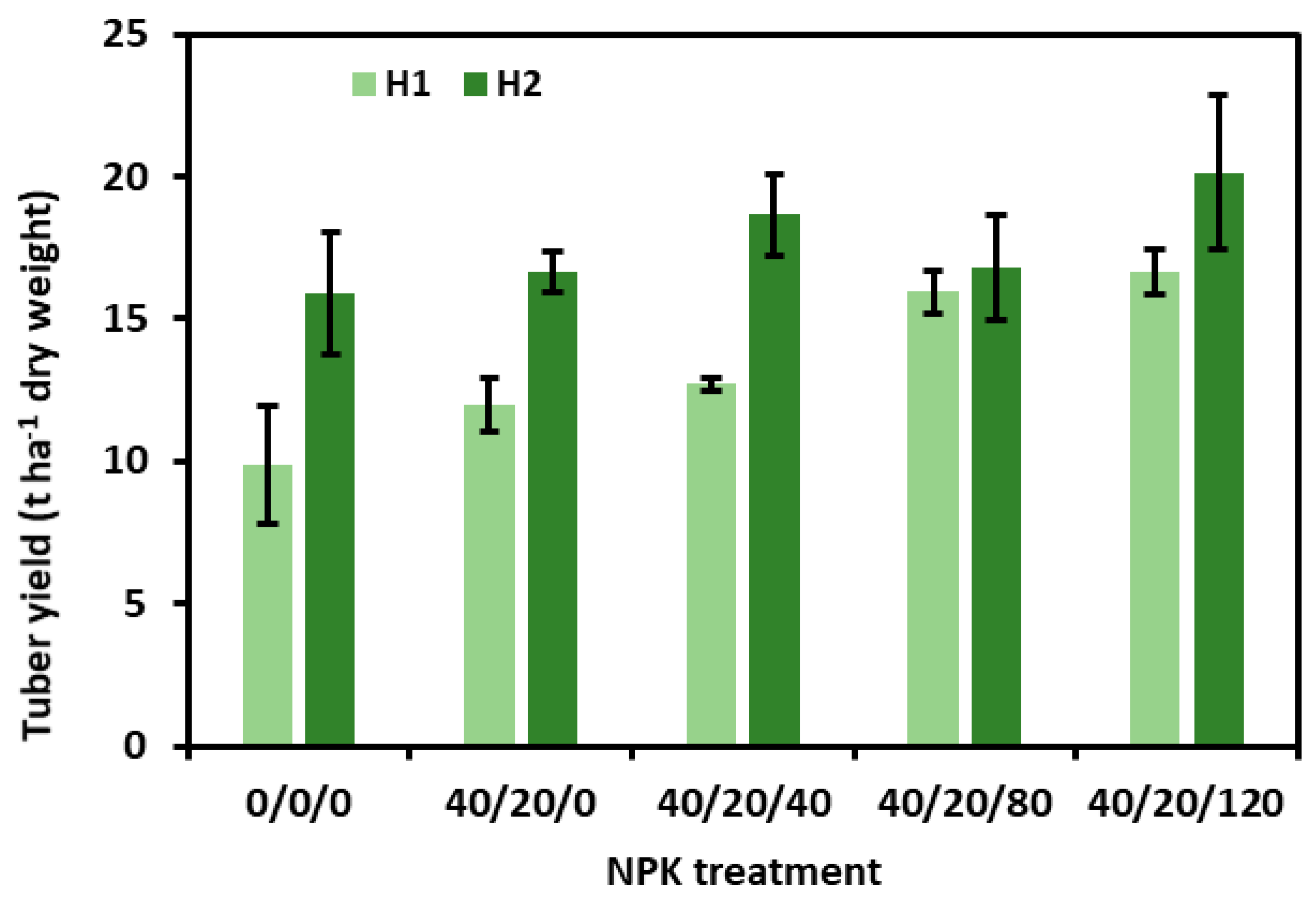
3.1.1. Tuber Production
3.1.2. Starch Yield
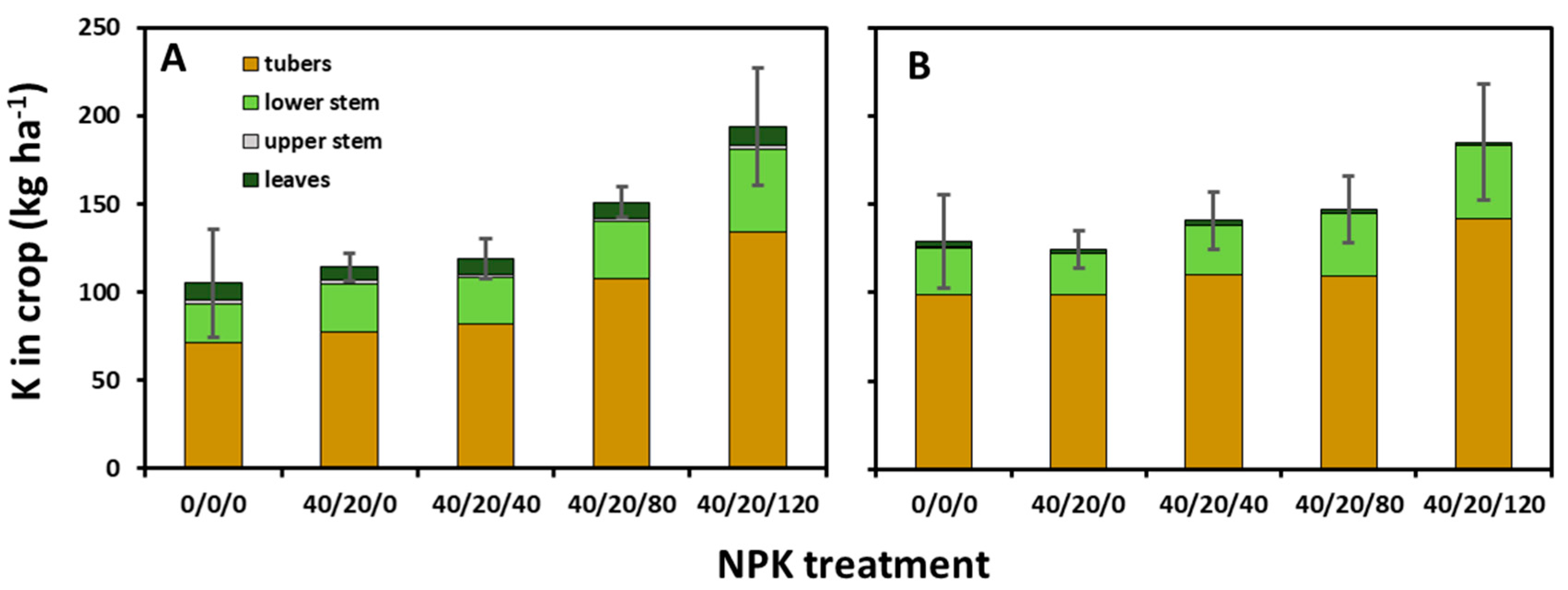
3.2. K Uptake, Distribution and Export in Yield
4. Discussion
4.1. K Fertilisation Increased Tuber Yields
4.2. K Fertilisation Affects Starch Content
4.3. More K Moved to Tubers Later in the Season
4.4. High K in Lower Stems may Benefit K Balance and Future Yield
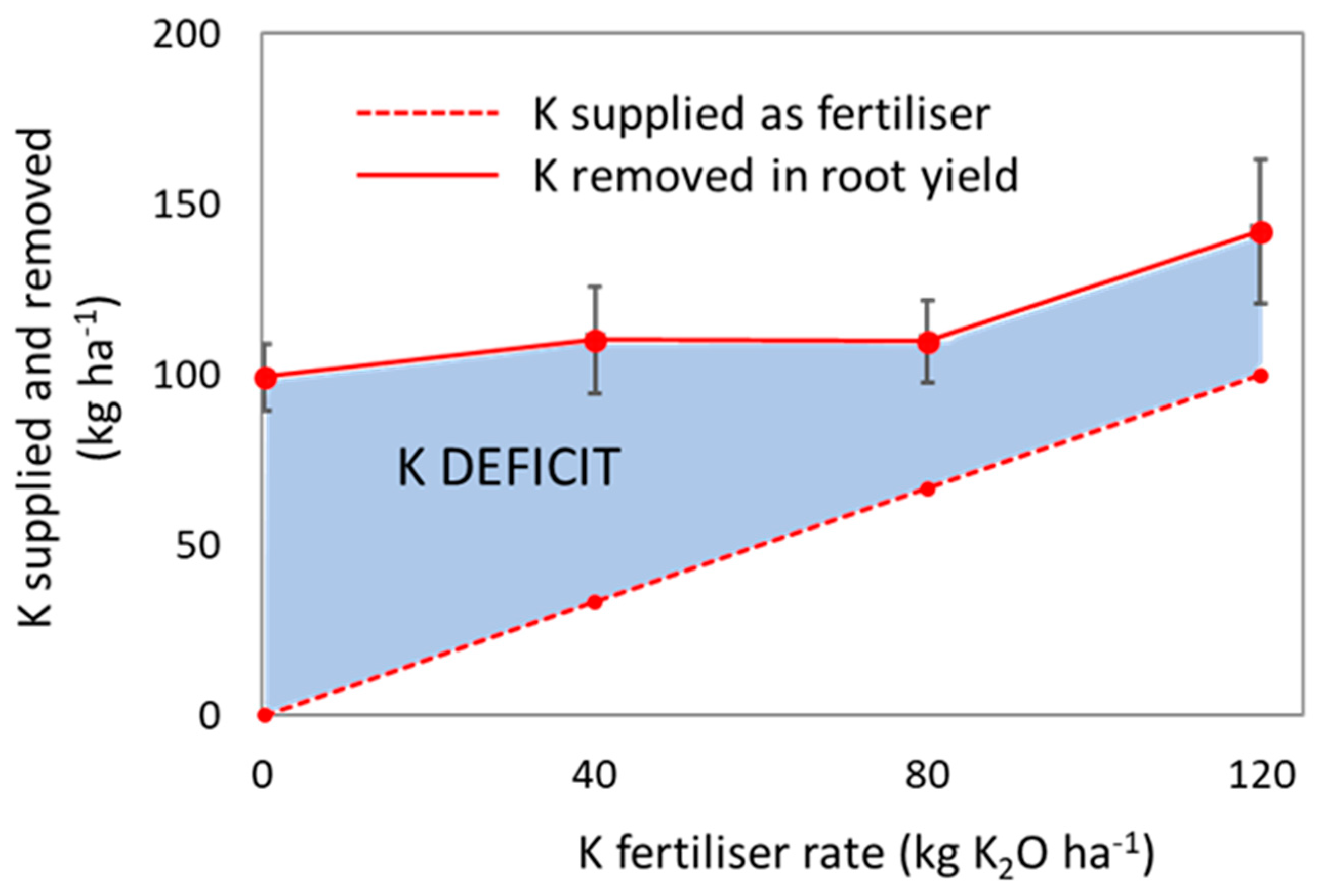
4.5. Reduction of K Deficit
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Malik, A.I.; Kongsil, P.; Nguyễn, V.A.; Ou, W.; Srean, P.; Sheela, M.N.; Luis Augusto Becerra, L.A.; Utsumi, Y.; Lu, C.; Kittipadakul, P.; et al. Cassava breeding and agronomy in Asia- 50 years of history and future directions. Breed Sci. 2020, 20, 145–166. [Google Scholar] [CrossRef] [PubMed]
- Howeler, R.H. Sustainable soil and crop management of cassava in Asia. In A Reference Manual; International Center for Tropical Agriculture (CIAT): Cali, Colombia, 2014; p. 280. [Google Scholar]
- Aye, T.M. Cassava Cultivation in Asia. In Achieving Sustainable Cultivation of Cassava; Hershey, C., Ed.; Burleigh Dodds Science Publishing: Cambridge, UK, 2017; pp. 101–122. [Google Scholar]
- Gleadow, R.; Pegg, A.; Blomstedt, C.K. Resilience of cassava (Manihot esculenta Crantz) to salinity: Implications for food security in low-lying regions. J. Exp. Bot. 2016, 67, 5403–5413. [Google Scholar] [CrossRef] [PubMed]
- Bechoff, A. Use and Nutritional Value of Cassava Roots and Leaves as a Traditional Food. In Achieving Sustainable Cultivation of Cassava Volume 1: Cultivation Techniques; Burleigh Dodds Series in Agricultural Science; Hershey, C., Ed.; Burleigh Dodds Science Publishing: Cambridge, UK, 2017; pp. 33–55. [Google Scholar]
- Alene, A.D.; Abdoulaye, T.; Rusike, J.; Labarta, R.; Creamer, B.; del Río, M. Identifying crop research priorities based on potential economic and poverty reduction impacts: The case of cassava in Africa, Asia, and Latin America. PLoS ONE 2018, 13, e0201803. [Google Scholar] [CrossRef] [PubMed]
- El-Sharkawy, M.A.; Mwanza, F. Drought-tolerant cassava for Africa, Asia, and Latin America. BioScience 1993, 43, 441–451. [Google Scholar] [CrossRef]
- Kawana, K.; Cock, J.H. Breeding cassava for underprivileged: Institutional, Socio-Economic and Biological Factors for success. J. Crop. Impro. 2005, 14, 197–214. [Google Scholar] [CrossRef]
- Henry, G.; Gottret, V. Global Cassava Trends: Reassessing the Crop’s Future. In Working Document No. 157; CIAT: Cali, Colombia, 1996. [Google Scholar]
- Viswanathan, P.K. Lao Census of Agriculture 2010/11: Analysis of Selected Themes, Ministry of Agriculture and Forestry, Government of the Lao People’s Democratic Republic, Vientiane. Food and Agriculture Organization of the United Nations: Vientiane, Lao PDR, 2014. Available online: http://www.fao.org/3/a-at767e.pdf (accessed on 7 May 2020).
- Smith, D.; Newby, J.; Malik, A.I.; Yadav, L.; Cramb, R. Fertiliser use patterns of smallholder farmers-implications for private sector involvement in technology dissemination, Cassava Program Discussion Paper Number 8, September 2018, School of Agriculture and Food Sciences, University of Queensland, 2018, ISSN 2209–2684. Available online: http://cassavavaluechains.net/wp-content/uploads/2018/09/Discussion-paper-number-8.pdf (accessed on 20 April 2020).
- Howeler, R.H. Long term effect of cassava cultivation on soil productivity. Field Crops Res. 1991, 26, 1–18. [Google Scholar] [CrossRef]
- Nguyen, H.; Schoenau, J.; Nguyen, D.; Van Rees, K.; Boehm, M. Effects of long-term nitrogen, phosphorus, and potassium fertilization on cassava yield and plant nutrient composition in North Vietnam. J. Plant Nutr. 2002, 25, 425–442. [Google Scholar] [CrossRef]
- Ezui, K.S.; Franke, A.C.; Mando, A.; Ahiabor, B.D.K.; Tetteh, F.M.; Sogbedji, J.; Janssen, B.H.; Giller, K.E. Fertiliser requirements for balanced nutrition of cassava across eight locations in West Africa. Field Crops Res. 2016, 185, 69–78. [Google Scholar] [CrossRef]
- Cadavid, L.F.; El-Sharkawy, M.A.; Ascota, A.; Sánchez, T. Long-term effects of mulch, fertilization and tillage on cassava grown in sandy soils in northern Colombia. Field Crops Res. 1998, 57, 45–56. [Google Scholar] [CrossRef]
- Howeler, R.H. Cassava Mineral Nutrition and Fertilization. In Cassava: Biology, Production and Utilization; Hillocks, R.J., Thresh, J.M., Bellotti, A., Eds.; CABI: Wallingford, UK, 2002; pp. 115–147. [Google Scholar]
- Fernandes, A.M.; Gazola, B.; Nunes, J.G.S.; Garcia, E.L.; Leonel, M. Yield and nutritional requirements of cassava in response to potassium fertilizer in the second cycle. J. Plant Nutr. 2017, 40, 2785–2796. [Google Scholar] [CrossRef]
- Howeler, R.H. Effect of cassava production on soil fertility and the long-term fertilizer requirements to maintain high yields. In The Cassava Handbook: A Reference Manual Based on the Asian Regional Cassava Training Course, Held in Thailand; Howeler, R.H., Ed.; Centro Internacional de Agricultura Tropical (CIAT): Bangkok, Thailand, 2012; pp. 411–428. [Google Scholar]
- Chan, S.K.; Lee, C. Relationships of Tuber Yield, Starch Content and Starch Yield of Cassava with Potassium Status of Fertilizer, Soil and Leaf. In Proceedings of the 5th International Symposium on Tropical Root and Tuber Crops, Manila, Philippines, 17–21 September 1979; pp. 461–465. [Google Scholar]
- Howeler, R.H. Diagnosis of Nutritional Disorders and Soil Fertility Maintenance of Cassava. In Tropical Tuber Crops: Problems, Prospects and Future Strategies; Kurup, G.T., Palaniswami, M.S., Potty, V.P., Padmaja, G., Kabeerathumma, S., Pilai, S.V., Eds.; Oxford and IBH Publishing Co.: New Delhi, India, 1983; pp. 181–192. [Google Scholar]
- Howeler, R.H.; Tan, S.L. Cassava’s Potential in Asia in the 21st Century: Present Situation and Future Research and Development Needs. In Proceedings of the Sixth Regional Workshop, Ho Chi Minh City, Vietnam, 21–25 February 2000; p. 666. [Google Scholar]
- Nguyen, H.H.; Nguyen, T.D.; Pham, V.B. Cassava agronomy research and adaptation of improved practices in Vietnam. In Cassava’s Potential in Asia in the 21st Century: Present Situation and Future Research and Development Needs, Proceedings of the Sixth Regional Workshop, Ho Chi Minh City, Vietnam, 21–25 February 2000; Howeler, R.H., Tan, S.L., Eds.; 2001; pp. 216–227. [Google Scholar]
- Hasanuzzaman, M.; Bhuyan, M.; Nahar, K.; Hossain, M.S.; Mahmud, J.A.; Hossen, M.S. Potassium: A Vital Regulator of Plant Responses and Tolerance to Abiotic Stresses. Agronomy 2018, 8, 31. [Google Scholar] [CrossRef]
- Jin, S.H.; Huang, J.Q.; Li, X.Q.; Zheng, B.S.; Wu, J.S.; Wang, Z.J. Effects of potassium supply on limitations of photosynthesis by mesophyll diffusion conductance in Carya cathayensis. Tree Physiol. 2011, 31, 1142–1151. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; Di Cera, E. Role of Na+ and K+ in enzyme function. Physiol. Rev. 2006, 86, 1049–1092. [Google Scholar] [CrossRef] [PubMed]
- Prajapati, K.; Modi, H. The importance of potassium in plant growth—A review. Indian J. Plant Sci. 2012, 1, 177–186. [Google Scholar]
- Siegel, G.J.; Goodwin, B. Sodium-Potassium-activated adenosine triphosphatase: Potassium regulation of enzyme phosphorylation sodium-stimulated, potassium-inhibited uridine triphosphate hydrolysis. J. Biol. Chem. 1972, 247, 3630–3637. [Google Scholar]
- Kang, B.T.; Okeke, J.E. Nitrogen and Potassium Responses of Two Cassava Varieties Grown on an Alfisol in Southern Nigeria. In Proceedings of the International Society of Tropical Root Crops Symposium, Lima, Peru, 21 February 1983; pp. 231–234. [Google Scholar]
- Spear, S.N.; Edwards, D.G.; Asher, C.J. Response of cassava, sunflower, and maize to potassium concentration in solution III. Interactions between potassium, calcium and magnesium. Field Crops Res. 1978, 1, 375–389. [Google Scholar] [CrossRef]
- Chatakanonda, P.; Chinachoti, P.; Sriroth, K.; Piyachomkwan, K.; Chotineeranat, S.; Tang, H.; Hills, B. The influence of time and condition of harvest on functional behaviour of cassava starch-a proton NMR relaxation study. Carbohydr. Polym. 2003, 53, 233–240. [Google Scholar] [CrossRef]
- Santisopasri, V.; Kurotjanawong, K.; Chotineeranat, S.; Piyachomkwan, K.; Sriroth, K.; Oates, C.G. Impact of water stress on yield and quality of cassava starch. Ind. Crops Prod. 2011, 13, 115–129. [Google Scholar] [CrossRef]
- Sriroth, K.; Santisopasri, V.; Petchalanuwat, C.; Kurotjanawong, K.; Piyachomkwan, K.; Oates, C.G. Cassava starch granule structure―function properties: Influence of time and conditions at harvest on four cultivars of cassava starch. Carbohydr. Polym. 1999, 28, 161–170. [Google Scholar] [CrossRef]
- Wholey, D.W.; Booth, R.H. Influence of variety and planting density on starch accumulation in cassava roots. J. Sci. Food Agric. 1979, 30, 65–170. [Google Scholar] [CrossRef]
- Baharuddin, N.H.; Mohamed, M.; Abdullah, M.M.A.B.; Muhammad, N.; Rahman, R.; Omar, M.N.; Amini, M.H.M.; Razab, M.K.A.A.; Rizman, Z.I. Potential of cassava root as raw material for bio composite development. ARPN J. Eng. Appl. Sci. 2016, 11, 6138–6147. [Google Scholar]
- Molina, J.L.; El-Sharkawy, M.A. Increasing crop productivity in cassava by fertilizing production of planting material. Field Crop. Res. 1995, 44, 151–157. [Google Scholar] [CrossRef]
- Keating, B.A.; Evenson, J.P.; Edwards, D.G. Effect of pre-harvest fertilization of cassava, prior to cutting for planting material on subsequent establishment and root yield. In Proceedings of the 5th International Symposium on Tropical Root and Tuber Crops, Manila, Philippines, 17–21 September 1979; Belen, E.H., Villanueva, M., Eds.; Philippine Council for Agriculture and Resources Research: Laguna, Philippines, 1982; pp. 301–306. [Google Scholar]
- Howeler, R.H.; Cadavid, L. Accumulation and distribution of dry matter and nutrients during a 12-month growth cycle of cassava. Field Crop. Res. 1983, 7, 123–139. [Google Scholar] [CrossRef]
- Carsky, R.; Toukourou, M. Identification of nutrients limiting cassava yield maintenance on a sedimentary soil in southern Benin, West Africa. Nutr. Cycl. Agroecosyst. 2005, 71, 151–162. [Google Scholar] [CrossRef]
- Turmel, M.S.; Speratti, A.; Baudron, F.; Verhust, N.; Govaerts, B. Crop residue management and soil health: A systems analysis. Agric. Syst. 2015, 134, 6–16. [Google Scholar] [CrossRef]
| Treatment | Total Stem Length (cm) | Total Plant Dry wt. (g) | Harvest Index HI | Starch Content (%) | Starch Yield (t ha−1) |
|---|---|---|---|---|---|
| Harvest 1 | |||||
| T1 | 411 a ± 62.3 | 1416 a ± 284.5 | 0.69 a ± 0.007 | 29.1 a ± 1.18 | 7.1 a ± 1.50 |
| T2 | 581 ab ± 51.8 | 1718 ab ± 125.7 | 0.69 a ± 0.006 | 28.8 a ± 0.66 | 8.7 ab ± 0.74 |
| T3 | 609 ab ± 60.7 | 1756 ab ± 6.0 | 0.72 a ± 0.014 | 30.7 a ± 0.79 | 8.9 ab ± 0.65 |
| T4 | 753 b ± 52.3 | 2243 b ± 92.4 | 0.71 a ± 0.005 | 29.3 a ± 1.08 | 11.9 b ± 0.53 |
| T5 | 699 b ± 62.0 | 2331 b ± 214.0 | 0.72 a ± 0.030 | 29.0 a ± 0.24 | 12.4 b ± 0.85 |
| Harvest 2 | |||||
| T1 | 729 a ± 118.9 | 2127 a ± 319.5 | 0.75 a ± 0.023 | 34.6 b ± 0.50 | 10.9 a ± 1.46 |
| T2 | 636 a ± 66.5 | 2176 a ± 85.8 | 0.77 a ± 0.007 | 33.5 ab ± 0.47 | 11.6 a ± 0.52 |
| T3 | 810 a ± 142.0 | 2473 a ± 153.1 | 0.75 a ± 0.012 | 34.5 b ± 0.10 | 12.7 a ± 0.64 |
| T4 | 746 a ± 129.8 | 2260 a ± 244.9 | 0.75 a ± 0.006 | 32.4 ab ± 0.98 | 11.7 a ± 1.38 |
| T5 | 742 a ± 75.6 | 2560 a ± 409.0 | 0.79 a ± 0.020 | 30.3 a ± 1.00 | 13.4 a ± 2.28 |
| Treatment (T) | P = 0.164 | P = 0.008 | P = 0.212 | P = 0.019 | P = 0.009 |
| Harvest (H) | P = 0.028 | P < 0.001 | P < 0.001 | P < 0.001 | P = 0.002 |
| T × H | P = 0.282 | P = 0.208 | P = 0.428 | P = 0.118 | P = 0.198 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chua, M.F.; Youbee, L.; Oudthachit, S.; Khanthavong, P.; Veneklaas, E.J.; Malik, A.I. Potassium Fertilisation Is Required to Sustain Cassava Yield and Soil Fertility. Agronomy 2020, 10, 1103. https://doi.org/10.3390/agronomy10081103
Chua MF, Youbee L, Oudthachit S, Khanthavong P, Veneklaas EJ, Malik AI. Potassium Fertilisation Is Required to Sustain Cassava Yield and Soil Fertility. Agronomy. 2020; 10(8):1103. https://doi.org/10.3390/agronomy10081103
Chicago/Turabian StyleChua, Ming Fung, Laothao Youbee, Saythong Oudthachit, Phanthasin Khanthavong, Erik J. Veneklaas, and Al Imran Malik. 2020. "Potassium Fertilisation Is Required to Sustain Cassava Yield and Soil Fertility" Agronomy 10, no. 8: 1103. https://doi.org/10.3390/agronomy10081103
APA StyleChua, M. F., Youbee, L., Oudthachit, S., Khanthavong, P., Veneklaas, E. J., & Malik, A. I. (2020). Potassium Fertilisation Is Required to Sustain Cassava Yield and Soil Fertility. Agronomy, 10(8), 1103. https://doi.org/10.3390/agronomy10081103





