Genotypic Variability of Photosynthetic Parameters in Maize Ear-Leaves at Different Cadmium Levels in Soil
Abstract
1. Introduction
2. Materials and Methods
2.1. Soil Preparation
2.2. Plant Material and Growth Conditions
2.3. Chlorophyll Fluorescence Measurements
2.4. Photosynthetic Pigments and Dry Weight Analysis
2.5. ICP-OES Analysis—Plant Material and Soil
2.6. Statistical Analysis
3. Results
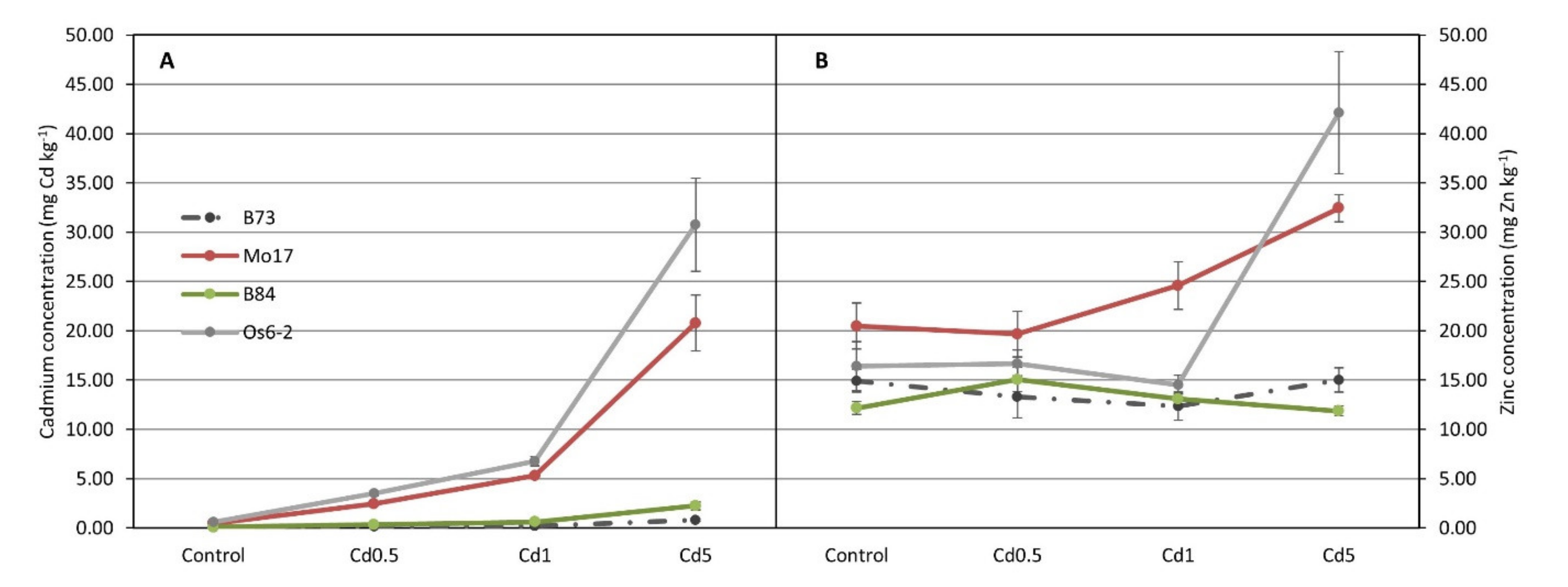
3.1. ICP Analysis for Cd and Zn
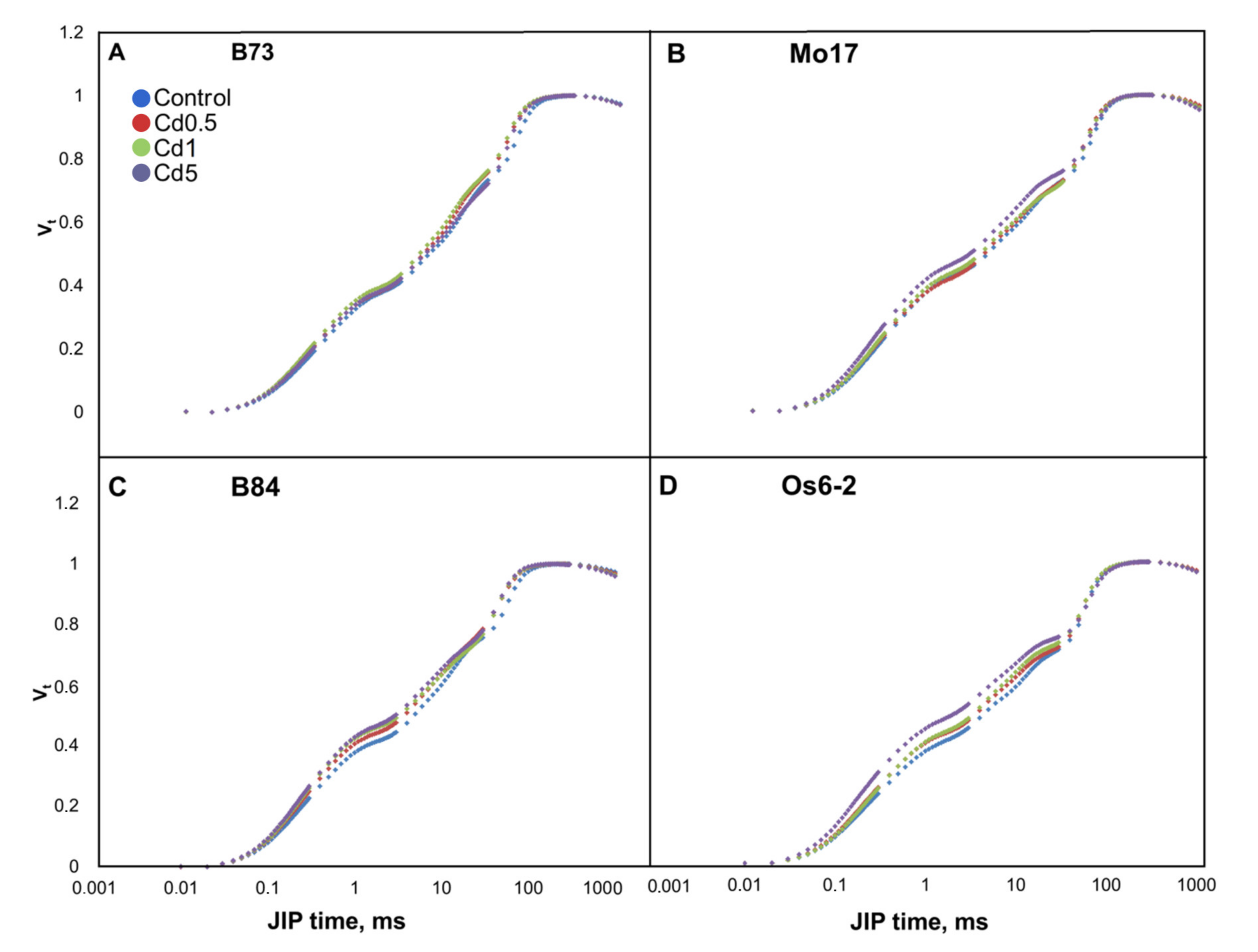
3.2. Chlorophyll a Fluorescence Transients
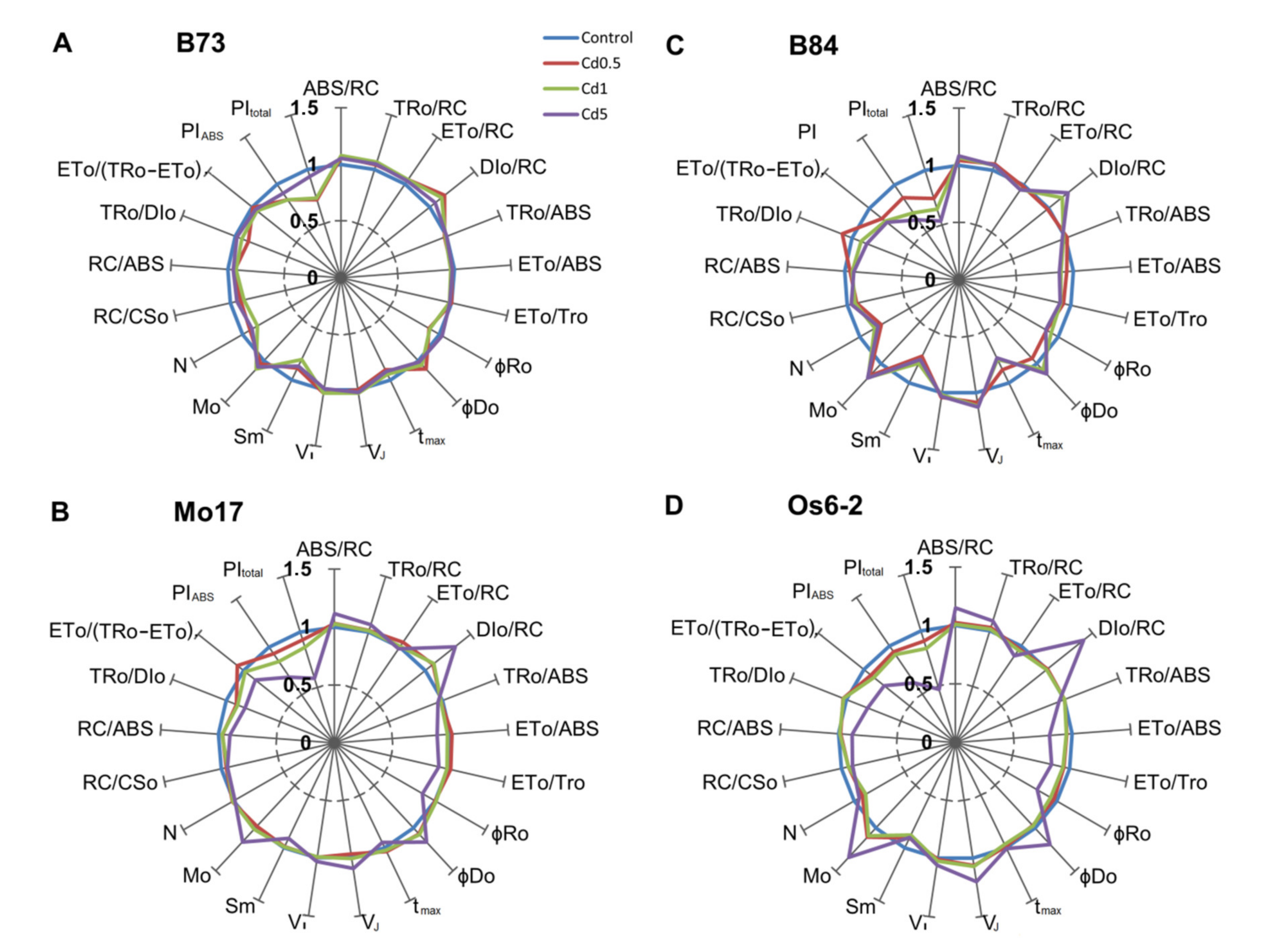
3.3. JIP-Test Parameters
3.4. Photosynthetic Pigments and Dry Weight
3.5. Correlation Analysis between JIP-Test Parameters and Trace-Metal Concentrations
4. Discussion
4.1. Effects of Cd on O-J-I-P Transients
4.2. Effects of Cd on JIP-Test Parameters
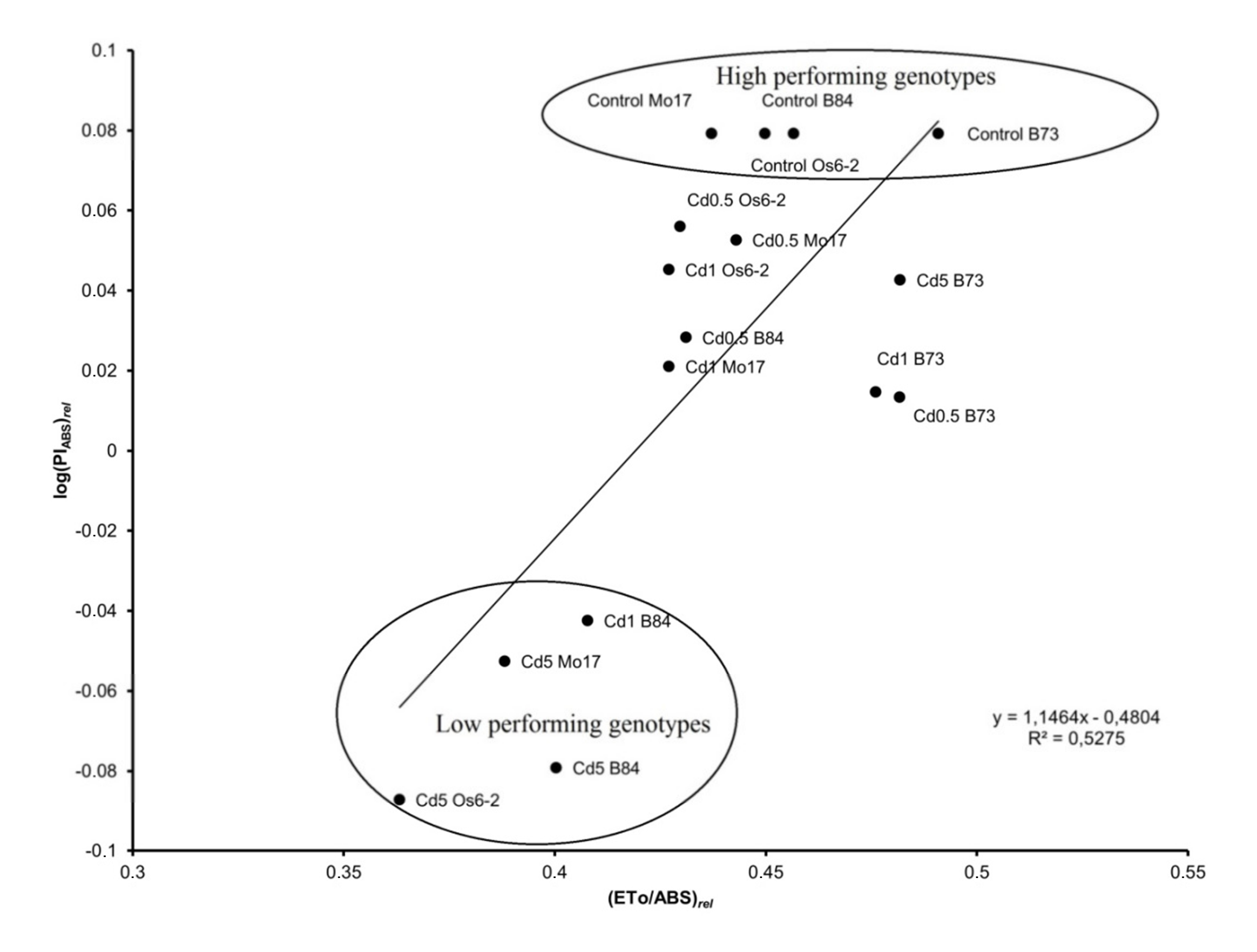
4.3. Performance of Light Utilization in Cd Stressed Plants
4.4. Photosynthetic Pigments
4.5. ICP-OES Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- McLaughlin, M.J.; Tiller, K.G.; Naidu, R.; Stevens, D.P. Review: The behaviour and environmental impact of contaminants in fertilizers. Aust. J. Soil Res. 1996, 34, 1–54. [Google Scholar] [CrossRef]
- Jacob, J.M.; Karthik, C.; Saratale, R.G.; Kumar, S.S.; Prabakar, D.; Kadirvelu, K.; Pugazhendhi, A. Biological approaches to tackle heavy metal pollution: A survey of literature. J. Environ. Manag. 2018, 217, 56–70. [Google Scholar] [CrossRef] [PubMed]
- Ekmekçi, Y.; Tanyolaç, D.; Ayhan, B. Effects of cadmium on antioxidant enzyme and photosynthetic activities in leaves of two maize cultivars. J. Plant. Physiol. 2008, 165, 600–611. [Google Scholar] [CrossRef]
- Huang, D.; Gong, X.; Liu, Y.; Zeng, G.; Lai, C.; Bashir, H.; Zhou, L.; Wang, D.; Xu, P.; Cheng, M.; et al. Effects of calcium at toxic concentrations of cadmium in plants. Planta 2017, 245, 863–873. [Google Scholar] [CrossRef]
- Ortega-Villasante, C.; Rellán-Álvarez, R.; Del Campo, F.F.; Carpena-Ruiz, R.O.; Hernández, L.E. Cellular damage induced by cadmium and mercury in Medicago sativa. J. Exp. Bot. 2005, 56, 2239–2251. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.S.; Dietz, K.J. The relationship between metal toxicity and cellular redox imbalance. Trends Plant. Sci. 2009, 14, 43–50. [Google Scholar] [CrossRef]
- Schützendübel, A.; Schwanz, P.; Teichmann, T.; Gross, K.; Langenfeld-Heyser, R.; Godbold, D.L.; Polle, A. Cadmium-induced changes in antioxidative systems, hydrogen peroxide content, and differentiation in Scots pine roots. Plant. Physiol. 2001, 127, 887–898. [Google Scholar] [CrossRef]
- Hendrix, S.; Keunen, E.; Mertens, A.I.G.; Beemster, G.T.S.; Vangronsveld, J.; Cuypers, A. Cell cycle regulation in different leaves of Arabidopsis thaliana plants grown under control and cadmium-exposed conditions. Environ. Exp. Bot. 2018, 155, 441–452. [Google Scholar] [CrossRef]
- Franić, M.; Galić, V. As, Cd, Cr, Cu, Hg: Physiological implications and toxicity in plants. In Plant Metallomics and Functional Omics: A System-Wide Perspective; Springer: Cham, Switzerland, 2019; pp. 209–251. ISBN 9783030191030. [Google Scholar]
- Tóth, G.; Hermann, T.; Da Silva, M.R.; Montanarella, L. Heavy metals in agricultural soils of the European Union with implications for food safety. Environ. Int. 2016, 88, 299–309. [Google Scholar] [CrossRef]
- Tóth, G.; Hermann, T.; Szatmári, G.; Pásztor, L. Maps of heavy metals in the soils of the European Union and proposed priority areas for detailed assessment. Sci. Total Environ. 2016, 565, 1054–1062. [Google Scholar] [CrossRef]
- Clijsters, H.; Van Assche, F. Inhibition of photosynthesis by heavy metals. Photosynth. Res. 1985, 7, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Sanità Di Toppi, L.; Gabbrielli, R. Response to cadmium in higher plants. Environ. Exp. Bot. 1999, 41, 105–130. [Google Scholar] [CrossRef]
- Perfus-Barbeoch, L.; Leonhardt, N.; Vavasseur, A.; Forestier, C. Heavy metal toxicity: Cadmium permeates through calcium channels and disturbs the plant water status. Plant J. 2002, 32, 539–548. [Google Scholar] [CrossRef] [PubMed]
- Tudoreanu, L.; Phillips, C.J.C. Empirical models of cadmium accumulation in maize, rye grass and soya bean plants. J. Sci. Food Agric. 2004, 84, 845–852. [Google Scholar] [CrossRef]
- Aravind, P.; Prasad, M.N.V. Zinc protects chloroplasts and associated photochemical functions in cadmium exposed Ceratophyllum demersum L., a freshwater macrophyte. Plant Sci. 2004, 166, 1321–1327. [Google Scholar] [CrossRef]
- Rout, G.R.; Das, P. Effect of metal toxicity on plant growth and metabolism: I. Zinc. In Sustainable Agriculture; Springer: Dordrecht, The Netherlands, 2009; pp. 873–884. ISBN 9789048126651. [Google Scholar]
- Cakmak, I.; Welch, P.M.; Hart, J.; Norvell, W.A.; Oztürk, L.; Kochian, L.V. Uptake and retranslocation of leaf-applied cadmium (109Cd) in diploid, tetraploid and hexaploid wheats. J. Exp. Bot. 2000, 51, 221–226. [Google Scholar] [CrossRef] [PubMed]
- Baryla, A.; Carrier, P.; Franck, F.; Coulomb, C.; Sahut, C.; Havaux, M. Leaf chlorosis in oilseed rape plants (Brassica napus) grown on cadmium-polluted soil: Causes and consequences for photosynthesis and growth. Planta 2001, 212, 696–709. [Google Scholar] [CrossRef]
- Strasser, R.J.; Govindjee. On the OJIP fluorescence transients in leaves and D1 mutants of Chlamydomonas reinhardtii. In Research in Photosynthesis; Murata, N., Ed.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1992; Volume 2, pp. 29–32. [Google Scholar]
- Strasser, B.J.; Strasser, R.J. Measuring Fast Fluorescence Transients to Address Environmental Questions: The JIP-Test. In Photosynthesis: From Light to Biosphere; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1995; pp. 977–980. [Google Scholar]
- Krüger, G.H.J.; Tsimilli-Michael, M.; Strasser, R.J. Light stress provokes plastic and elastic modifications in structure and function of photosystem II in camellia leaves. Physiol. Plant. 1997, 101, 265–277. [Google Scholar] [CrossRef]
- Tsimilli-Michael, M.; Pêcheux, M.; Strasser, R.J. Light and heat stress adaptation of the symbionts of temperate and coral reef foraminifers probed in Hospite by the chlorophyll a fluorescence kinetics. Zeitschrift für Naturforschung C 1999, 54, 671–680. [Google Scholar] [CrossRef]
- Zushi, K.; Kajiwara, S.; Matsuzoe, N. Chlorophyll A fluorescence OJIP transient as a tool to characterize and evaluate response to heat and chilling stress in tomato leaf and fruit. Sci. Hortic. 2012, 148, 39–46. [Google Scholar] [CrossRef]
- Kalaji, H.M.; Jajoo, A.; Oukarroum, A.; Brestic, M.; Zivcak, M.; Samborska, I.A.; Cetner, M.D.; Łukasik, I.; Goltsev, V.; Ladle, R.J. Chlorophyll A fluorescence as a tool to monitor physiological status of plants under abiotic stress conditions. Acta Physiol. Plant. 2016, 38, 102. [Google Scholar] [CrossRef]
- Hu, W.; Snider, J.L.; Chastain, D.R.; Slaton, W.; Tishchenko, V. Sub-optimal emergence temperature alters thermotolerance of thylakoid component processes in cotton seedlings. Environ. Exp. Bot. 2018, 155, 360–367. [Google Scholar] [CrossRef]
- Snider, J.L.; Thangthong, N.; Pilon, C.; Virk, G.; Tishchenko, V. OJIP-fluorescence parameters as rapid indicators of cotton (Gossypium hirsutum L.) seedling vigor under contrasting growth temperature regimes. Plant. Physiol. Biochem. 2018, 132, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Franić, M.; Jambrović, A.; Zdunić, Z.; Šimić, D.; Galić, V. Photosynthetic properties of maize hybrids under different environmental conditions probed by the chlorophyll a fluorescence. Maydica 2019, 64, 9. [Google Scholar]
- Reddy, A.V.R. Strasse Molecular Approaches for the Genetic Improvement of Cereals for Stable Production in Water-Limited Environments; Ribaut, J., Poland, D., Eds.; International Maize and Wheat Improvement Center: Veracruz, Mexico, 2000; pp. 29–35. [Google Scholar]
- Appenroth, K.J.; Stöckel, J.; Srivastava, A.; Strasser, R.J. Multiple effects of chromate on the photosynthetic apparatus of Spirodela polyrhiza as probed by OJIP chlorophyll a fluorescence measurements. Environ. Pollut. 2001, 115, 49–64. [Google Scholar] [CrossRef]
- Hermans, C.; Smeyers, M.; Maldonado Rodriguez, R.; Eyletters, M.; Strasser, R.J.; Delhaye, J.P. Quality assessment of urban trees: A comparative study of physiological characterisation, airborne imaging and on site fluorescence monitoring by the OJIP-test. J. Plant Phys. 2003, 160, 81–90. [Google Scholar] [CrossRef]
- Galic, V.; Franic, M.; Jambrovic, A.; Ledencan, T.; Brkic, A.; Zdunic, Z.; Simic, D. Genetic Correlations Between Photosynthetic and Yield Performance in Maize Are Different Under Two Heat Scenarios During Flowering. Front. Plant. Sci. 2019, 10, 566. [Google Scholar] [CrossRef] [PubMed]
- Galić, V.; Mazur, M.; Šimić, D.; Zdunić, Z.; Franić, M. Plant biomass in salt-stressed young maize plants can be modelled with photosynthetic performance. Photosynthetica 2020, 58, 194–204. [Google Scholar] [CrossRef]
- Franic, M.; Mazur, M.; Volenik, M.; Brkic, J.; Brkic, A.; Simic, D. Effect of Plant Density on Agronomic Traits and Photosynthetic Performance in the Maize IBM Population. Agriculture 2015, 21, 36–40. [Google Scholar] [CrossRef]
- Drązkiewicz, M.; Tukendorf, A.; Baszyński, T. Age-dependent response of maize leaf segments to cadmium treatment: Effect on chlorophyll fluorescence and phytochelatin accumulation. J. Plant. Physiol. 2003, 160, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Mallick, N.; Mohn, F.H. Use of chlorophyll fluorescence in metal-stress research: A case study with the green microalga Scenedesmus. Ecotoxicol. Environ. Saf. 2003, 55, 64–69. [Google Scholar] [CrossRef]
- Cherif, J.; Derbel, N.; Nakkach, M.; Von Bergmann, H.; Jemal, F.; Lakhdar, Z. Ben Spectroscopic studies of photosynthetic responses of tomato plants to the interaction of zinc and cadmium toxicity. J. Photochem. Photobiol. B Biol. 2012, 111, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Soric, R.; Loncaric, Z.; Kovacevic, V.; Brkic, I.; Simic, D. A major gene for leaf cadmium accumulation in maize (Zea mays L.). In The Proceedings of the International Plant Nutrition Colloquium XVI; University of California: Davis, CA, USA, 2009. [Google Scholar]
- Zhao, X.; Luo, L.; Cao, Y.; Liu, Y.; Li, Y.; Wu, W.; Lan, Y.; Jiang, Y.; Gao, S.; Zhang, Z.; et al. Genome-wide association analysis and QTL mapping reveal the genetic control of cadmium accumulation in maize leaf. BMC Genom. 2018, 19, 91. [Google Scholar] [CrossRef] [PubMed]
- Navarro-León, E.; Oviedo-Silva, J.; Ruiz, J.M.; Blasco, B. Possible role of HMA4a TILLING mutants of Brassica rapa in cadmium phytoremediation programs. Ecotoxicol. Environ. Saf. 2019, 180, 88–94. [Google Scholar] [CrossRef]
- Franić, M.; Galić, V.; Mazur, M.; Šimić, D. Effects of excess cadmium in soil on JIP-test parameters, hydrogen peroxide content and antioxidant activity in two maize inbreds and their hybrid. Photosynthetica 2018, 56, 660–669. [Google Scholar] [CrossRef]
- Anjum, S.A.; Tanveer, M.; Hussain, S.; Bao, M.; Wang, L.; Khan, I.; Ullah, E.; Tung, S.A.; Samad, R.A.; Shahzad, B. Cadmium toxicity in Maize (Zea mays L.): Consequences on antioxidative systems, reactive oxygen species and cadmium accumulation. Environ. Sci. Pollut. Res. 2015, 22, 17022–17030. [Google Scholar] [CrossRef]
- Tkalec, M.; Štefanić, P.P.; Cvjetko, P.; Šikić, S.; Pavlica, M.; Balen, B. The effects of cadmium-zinc interactions on biochemical responses in tobacco seedlings and adult plants. PLoS ONE 2014, 9, e87582. [Google Scholar] [CrossRef]
- Adamczyk-Szabela, D.; Lisowska, K.; Romanowska-Duda, Z.; Wolf, W.M. Combined cadmium-zinc interactions alter manganese, lead, copper uptake by Melissa officinalis. Sci. Rep. 2020, 10, 1–10. [Google Scholar] [CrossRef]
- Liedschulte, V.; Laparra, H.; Battey, J.N.D.; Schwaar, J.D.; Broye, H.; Mark, R.; Klein, M.; Goepfert, S.; Bovet, L. Impairing both HMA4 homeologs is required for cadmium reduction in tobacco. Plant. Cell Environ. 2017, 40, 364–377. [Google Scholar] [CrossRef] [PubMed]
- Lal, R. World Reference Base for Soil Resources. In Encyclopedia of Soil Science, 3rd ed.; CRC Press: Boca Raton, FL, USA, 2017. [Google Scholar]
- Egnér, H.; Riehm, H.; Domingo, W.R. Untersuchungen über die chemische Bodenanalyse als Grundlage für die Beurteilung des Nährstoffzustandes der Böden. II. Chemische Extraktionsmethoden zur Phosphor-und Kaliumbestimmung. Kungliga Lantbrukshögskolans Annaler 1960, 26, 199–215. [Google Scholar]
- Allen, R.G.; Pereira, L.S.; Raes, D.; Smith, M. Crop Evapotranspiration–Guidelines for Computing Crop Water Requirements–FAO Irrigation and Drainage Paper 56; FAO: Rome, Italy, 1998. [Google Scholar]
- Xu, D.Q.; Shen, Y.K. Photosynthetic efficiency and crop yield. In Handbook of Plant and Crop Physiology, 2nd ed.; Marcel Dekker: New York, NY, USA, 2001; ISBN 9780203908426. [Google Scholar]
- Ciganda, V.; Gitelson, A.; Schepers, J. Non-destructive determination of maize leaf and canopy chlorophyll content. J. Plant. Physiol. 2009, 166, 157–167. [Google Scholar] [CrossRef] [PubMed]
- Strasserf, R.J.; Srivastava, A.; Govindjee. Polyphasic Chlorophyll a Fluorescence Transient in Plants and Cyanobacteria. Photochem. Photobiol. 1995, 61, 32–42. [Google Scholar] [CrossRef]
- Strasser, R.J.; Srivastava, A.; Tsimilli-Michael, M. The fluorescence transient as a tool to characterize and screen photosynthetic samples. In Probing Photosynthesis Mechanisms Regulation and Adaptation; Taylor and Francis: Abingdon, UK, 2000. [Google Scholar]
- Strasser, R.J.; Tsimilli-Michael, M.; Srivastava, A. Analysis of the Chlorophyll a Fluorescence Transient. In Chlorophyll a Fluorescence; Springer: Dordrecht, The Netherlands, 2004; pp. 321–362. [Google Scholar]
- Strasser, R.J.; Tsimilli-Michael, M.; Qiang, S.; Goltsev, V. Simultaneous in vivo recording of prompt and delayed fluorescence and 820-nm reflection changes during drying and after rehydration of the resurrection plant Haberlea rhodopensis. Biochim. Biophys. Acta Bioenerg. 2010, 1797, 1313–1326. [Google Scholar] [CrossRef] [PubMed]
- Lichtenthaler, H.K. Chlorophylls and Carotenoids: Pigments of Photosynthetic Biomembranes. Methods Enzym. 1987, 148, 350–382. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- Fox, J.; Weisberg, S. Companion to Applied Regression, 2nd ed.; SAGE Publications: Los Angeles, CA, USA, 2011; ISBN 9781412975148. [Google Scholar]
- De, F.; Maintainer, M.; De Mendiburu, F. Package “Agricolae”. In Statistical Procedures for Agricultural Research; R Foundation for Statistical Computing: Vienna, Austria, 2017. [Google Scholar]
- Harrell, F.E. Hmisc: Harrell Miscellaneous, R Package Version 4.4-0; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Stirbet, A.; Govindjee. Chlorophyll a fluorescence induction: A personal perspective of the thermal phase, the J-I-P rise. Photosynth. Res. 2012, 113, 15–61. [Google Scholar] [CrossRef]
- Sheoran, I.S.; Singal, H.R.; Singh, R. Effect of cadmium and nickel on photosynthesis and the enzymes of the photosynthetic carbon reduction cycle in pigeonpea (Cajanus cajan L.). Photosynth. Res. 1990, 23, 345–351. [Google Scholar] [CrossRef]
- Ghori, N.H.; Ghori, T.; Hayat, M.Q.; Imadi, S.R.; Gul, A.; Altay, V.; Ozturk, M. Heavy metal stress and responses in plants. Int. J. Environ. Sci. Technol. 2019, 16, 1807–1828. [Google Scholar] [CrossRef]
- Schreiber, U.; Neubauer, C.; Klughammer, C. Devices and methods for room-temperature fluorescence analysis. Philos. Trans. R. Soc. B Biol. Sci. 1989, 323, 241–251. [Google Scholar]
- Ciscato, M.; Vangronsveld, J.; Valcke, R. Effects of heavy metals on the fast chlorophyll fluorescence induction kinetics of photosystem II: A comparative study. Zeitschrift fur Naturforschung-Section C 1999, 54, 9–10. [Google Scholar] [CrossRef]
- Kalaji, H.M.; Oukarroum, A.; Alexandrov, V.; Kouzmanova, M.; Brestic, M.; Zivcak, M.; Samborska, I.A.; Cetner, M.D.; Allakhverdiev, S.I.; Goltsev, V. Identification of nutrient deficiency in maize and tomato plants by invivo chlorophyll a fluorescence measurements. Plant. Physiol. Biochem. 2014, 81, 16–25. [Google Scholar] [CrossRef]
- Stirbet, A.; Govindjee. On the relation between the Kautsky effect (chlorophyll a fluorescence induction) and Photosystem II: Basics and applications of the OJIP fluorescence transient. J. Photochem. Photobiol. B Biol. 2011, 104, 236–257. [Google Scholar] [CrossRef]
- Pagliano, C.; Raviolo, M.; Dalla Vecchia, F.; Gabbrielli, R.; Gonnelli, C.; Rascio, N.; Barbato, R.; La Rocca, N. Evidence for PSII donor-side damage and photoinhibition induced by cadmium treatment on rice (Oryza sativa L.). J. Photochem. Photobiol. B Biol. 2006, 84, 70–78. [Google Scholar] [CrossRef]
- Lefèvre, I.; Marchal, G.; Corréal, E.; Zanuzzi, A.; Lutts, S. Variation in response to heavy metals during vegetative growth in Dorycnium pentaphyllum Scop. Plant. Growth Regul. 2009, 59, 1–11. [Google Scholar] [CrossRef]
- Cao, Y.; Zhang, Q.; Chen, Y.; Zhao, H.; Lang, Y.; Yu, C.; Yang, J. Identification of differential expression genes in leaves of rice (Oryza sativa L.) in response to heat stress by cDNA-AFLP analysis. Biomed. Res. Int. 2013, 2013, 11. [Google Scholar] [CrossRef] [PubMed]
- Christen, D.; Schönmann, S.; Jermini, M.; Strasser, R.J.; Défago, G. Characterization and early detection of grapevine (Vitis vinifera) stress responses to esca disease by in situ chlorophyll fluorescence and comparison with drought stress. Environ. Exp. Bot. 2007, 60, 504–514. [Google Scholar] [CrossRef]
- Shaw, B.P.; Sahu, S.K.; Mishra, R.K. Heavy Metal Induced Oxidative Damage in Terrestrial Plants. In Heavy Metal Stress in Plants; Springer: Heidelberg, Germany, 2004; pp. 84–126. [Google Scholar]
- Shaw, B.P. Effects of mercury and cadmium on the activities of antioxidative enzymes in the seedlings of Phaseolus aureus. Biol. Plant. 1995, 37, 587. [Google Scholar] [CrossRef]
- Sandalio, L.M.; Dalurzo, H.C.; Gómez, M.; Romero-Puertas, M.C.; Del Río, L.A. Cadmium-induced changes in the growth and oxidative metabolism of pea plants. J. Exp. Bot. 2001, 52, 2115–2126. [Google Scholar] [CrossRef] [PubMed]
- Lichtenthaler, H.K.; Buschmann, C.; Döll, M.; Fietz, H.J.; Bach, T.; Kozel, U.; Meier, D.; Rahmsdorf, U. Photosynthetic activity, chloroplast ultrastructure, and leaf characteristics of high-light and low-light plants and of sun and shade leaves. Photosynth. Res. 1981, 2, 115–141. [Google Scholar] [CrossRef] [PubMed]
- Lichtenthaler, H.K.; Kuhn, G.; Prenzel, U.; Buschmann, C.; Meier, D. Adaptation of chloroplast-ultrastructure and of chlorophyll-protein levels to high-light and low-light growth conditions. Zeitschrift für Naturforschung C 1982, 37, 464–475. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K.; Meier, D.; Buschmann, C. Development of chloroplasts at high and low light quanta fluence rates. Isr. J. Bot. 1984, 33, 185–194. [Google Scholar]
- Yamamoto, H.Y.; Bassi, R. Carotenoids: Localization and Function. In Oxygenic Photosynthesis: The Light Reactions; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2005. [Google Scholar]
- Maksymiec, W.; Baszyński, T. Different susceptibility of runner bean plants to excess copper as a function of the growth stages of primary leaves. J. Plant Physiol. 1996, 149, 217–221. [Google Scholar] [CrossRef]
- Shaw, B.P.; Rout, N.P. Age-dependent responses of Phaseolus aureus Roxb. to inorganic salts of mercury and cadmium. Acta Physiol. Plant. 1998, 20, 85–90. [Google Scholar] [CrossRef]
- Han, Y.L.; Yuan, H.Y.; Huang, S.Z.; Guo, Z.; Xia, B.; Gu, J. Cadmium tolerance and accumulation by two species of Iris. Ecotoxicology 2007, 16, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Ali, B.; Gill, R.A.; Yang, S.; Gill, M.B.; Farooq, M.A.; Liu, D.; Daud, M.K.; Ali, S.; Zhou, W. Regulation of cadmium-induced proteomic and metabolic changes by 5-aminolevulinic acid in leaves of Brassica napus L. PLoS ONE 2015, 10, e0123328. [Google Scholar] [CrossRef]
- Köleli, N.; Eker, S.; Cakmak, I. Effect of zinc fertilization on cadmium toxicity in durum and bread wheat grown in zinc-deficient soil. Environ. Pollut. 2004, 131, 453–459. [Google Scholar] [CrossRef]
- Troyer, A.F. Development of hybrid corn and the seed corn industry. In Handbook of Maize: Genetics and Genomics; Springer: New York, NY, USA, 2009; pp. 87–114. ISBN 9780387778631. [Google Scholar]
- Balen, B.; Tkalec, M.; Šikić, S.; Tolić, S.; Cvjetko, P.; Pavlica, M.; Vidaković-Cifrek, Ž. Biochemical responses of Lemna minor experimentally exposed to cadmium and zinc. Ecotoxicology 2011, 20, 815–826. [Google Scholar] [CrossRef]
- Wu, F.; Zhang, G. Genotypic variation in kernel heavy metal concentrations in barley and as affected by soil factors. J. Plant. Nutr. 2002, 25, 1163–1173. [Google Scholar] [CrossRef]
- Nan, Z.; Li, J.; Zhang, J.; Cheng, G. Cadmium and zinc interactions and their transfer in soil-crop system under actual field conditions. Sci. Total Environ. 2002, 285, 187–195. [Google Scholar] [CrossRef]
- Moraghan, J.T. Accumulation of cadmium and selected elements in flax seed grown on a calcareous soil. Plant Soil 1993, 150, 61–68. [Google Scholar] [CrossRef]
- Larbi, A.; Morales, F.; Abadia, A.; Gogorcena, Y.; Lucena, J.J.; Abadia, J. Effects of Cd and Pb in sugar beet plants grown in nutrient solution: Induced Fe deficiency and growth inhibition. Funct. Plant. Biol. 2002, 29, 1453–1464. [Google Scholar] [CrossRef]
- Smilde, K.W.; Van Luit, B.; Van Driel, W. The extraction by soil and absorption by plants of applied zinc and cadmium. Plant Soil 1992, 143, 233–238. [Google Scholar] [CrossRef]
- McKenna, I.M.; Chaney, R.L.; Williams, F.M. The effects of cadmium and zinc interactions on the accumulation and tissue distribution of zinc and cadmium in lettuce and spinach. Environ. Pollut. 1993, 79, 113–120. [Google Scholar] [CrossRef]
- Zha, H.G.; Jiang, R.F.; Zhao, F.J.; Vooijs, R.; Schat, H.; Barker, J.H.A.; McGrath, S.P. Co-segregation analysis of cadmium and zinc accumulation in Thlaspi caerulescens interecotypic crosses. New Phytol. 2004, 163, 299–312. [Google Scholar] [CrossRef]
- Florijn, P.J.; De Knecht, J.A.; Van Beusichem, M.L. Phytochelatin Concentrations and Binding State of Cd in Roots of Maize Genotypes Differing in Shoot/Root Cd Partitioning. J. Plant. Physiol. 1993, 142, 537–542. [Google Scholar] [CrossRef]
| Element | Value | Element | Value |
|---|---|---|---|
| pHKCl | 6.99 ± 0.03 | Cu (mg kg–1) | 24.00 ± 0.19 |
| pHH2O | 8.05 ± 0.02 | Fe (mg kg–1) | 29358.75 ± 208.80 |
| Organic matter (%) | 2.57 ± 0.08 | Mn (mg kg–1) | 676.05 ± 5.53 |
| P2O5-AL (%) | 29.58 ± 0.64 | Zn (mg kg–1) | 69.40 ± 0.64 |
| K2O-AL (%) | 25.60 ± 3.43 | Ni (mg kg–1) | 31.32 ± 0.22 |
| CaCO3 (%) | 1.26 ± 0 | Co (mg kg–1) | 13.83 ± 0.11 |
| Pb (mg/kg) | 20.50 ± 0.20 | Cr (mg kg–1) | 44.87 ± 0.56 |
| Cd Excluders | Cd Non-Excluders | |||
|---|---|---|---|---|
| B73 | B84 | Mo17 | Os6-2 | |
| Dry weight (%) | ||||
| Control | 25.25 ± 0.06 a | 23.38 ± 0.04 c | 20.35 ± 0.49 c | 21.95 ± 0.07 b |
| Cd0.5 | 22.80 ± 0.19 c | 22.98 ± 0.18 d | 22.89 ± 0.21 b | 21.20 ± 0.27 c |
| Cd1 | 20.95 ± 0.08 d | 25.84 ± 0.01 a | 23.26 ± 0.03 b | 22.40 ± 0.08 b |
| Cd5 | 23.90 ± 0.08 b | 25.32 ± 0.15 b | 24.14 ± 0.05 a | 24.90 ± 0.10 a |
| LSD0.05 | 0.3742 | 0.8805 | 0.3894 | 0.4971 |
| Chl a/DW | ||||
| Control | 5.01 ± 0.13 b | 4.66 ± 0.87 c | 5.71 ± 0.21 a | 4.85 ± 0.03 a |
| Cd0.5 | 5.27 ± 0.07 b | 6.04 ± 0.06 a | 4.40 ± 0.07 b | 4.90 ± 0.04 a |
| Cd1 | 5.88 ± 0.07 a | 5.46 ± 0.16 b | 4.35 ± 0.03 b | 4.13 ± 0.04 b |
| Cd5 | 4.33 ± 0.07 c | 4.53 ± 0.06 c | 3.52 ± 0.05 c | 3.74 ± 0.02 c |
| LSD0.05 | 0.2912 | 0.3769 | 0.3680 | 0.1172 |
| Chl b/DW | ||||
| Control | 1.40 ± 0.05 b | 1.36 ± 0.32 b | 1.56 ± 0.01 a | 1.30 ± 0.06 a |
| Cd0.5 | 1.34 ± 0.02 b | 1.75 ± 0.02 a | 1.18 ± 0.03 b | 1.32 ± 0.06 a |
| Cd1 | 1.63 ± 0.01 a | 1.66 ± 0.06 a | 1.15 ± 0.00 b | 1.10 ± 0.04 b |
| Cd5 | 1.09 ± 0.02 c | 1.22 ± 0.04 b | 0.95 ± 0.02 c | 0.96 ± 0.03 c |
| LSD0.05 | 0.0961 | 0.0568 | 0.1480 | 0.0616 |
| Chl a + b/DW | ||||
| Control | 6.41 ± 0.08 b | 6.01 ± 1.18 c | 7.27 ± 0.21 a | 6.15 ± 0.06 a |
| Cd0.5 | 6.61 ± 0.09 b | 7.79 ± 0.05 a | 5.57 ± 0.09 b | 6.22 ± 0.06 a |
| Cd1 | 7.51 ± 0.06 a | 7.13 ± 0.09 b | 5.50 ± 0.02 b | 5.24 ± 0.04 b |
| Cd5 | 5.42 ± 0.09 c | 5.76 ± 0.11 c | 4.47 ± 0.07 c | 4.70 ± 0.03 c |
| LSD0.05 | 0.2590 | 0.3927 | 0.2830 | 0.1603 |
| Car/DW | ||||
| Control | 1.32 ± 0.03 c | 1.27 ± 0.27 b | 1.35 ± 0.04 a | 1.25 ± 0.02 a |
| Cd0.5 | 1.40 ± 0.01 b | 1.59 ± 0.01 a | 1.09 ± 0.01 c | 1.23 ± 0.02 a |
| Cd1 | 1.70 ± 0.02 a | 1.54 ±0.04 a | 1.23 ± 0.01 b | 1.25 ± 0.01 a |
| Cd5 | 1.42 ± 0.02 b | 1.30 ± 0.02 b | 0.99 ± 0.01 d | 1.16 ± 0.00 b |
| LSD0.05 | 0.0641 | 0.0726 | 0.0820 | 0.0485 |
| Chl a/Chl b/DW | ||||
| Control | 14.25 ± 0.86 b | 14.75 ± 0.46 ab | 18.03 ± 1.00 a | 16.96 ± 0.24 ab |
| Cd0.5 | 17.26 ± 0.16 a | 15.03 ± 0.40 a | 16.33 ± 0.31 ab | 17.50 ± 0.12 a |
| Cd1 | 17.23 ± 0.32 a | 12.76 ± 0.85 b | 16.24 ± 0.17 b | 16.73 ± 0.31 b |
| Cd5 | 16.64 ± 0.18 a | 14.68 ± 0.37 b | 15.27 ± 0.19 b | 15.60 ± 0.22 c |
| LSD0.05 | 1.556 | 1.763 | 1.652 | 0.7586 |
| Chl a + b/Car/DW | ||||
| Control | 19.27 ± 0.13 b | 20.23 ± 0.78 b | 26.39 ± 0.65 a | 22.49 ± 0.17 b |
| Cd0.5 | 20.71 ± 0.29 a | 21.33 ± 0.20 a | 22.39 ± 0.27 b | 23.91 ± 0.17 a |
| Cd1 | 21.11 ± 0.09 a | 17.87 ± 0.20 c | 19.30 ± 0.05 c | 18.66 ± 0.04 c |
| Cd5 | 15.98 ± 0.08 c | 17.51 ± 0.06 c | 18.74 ± 0.05 c | 16.27 ± 0.14 d |
| LSD0.05 | 0.5611 | 1.149 | 0.5218 | 0.4589 |
| Variable | VJ | Mo | TRo/ABS | ETo/ABS | Eto/Tro |
|---|---|---|---|---|---|
| Zn (mg kg−1) | 0.67 ** | 0.74 *** | −0.65 ** | −0.67 ** | −0.67 ** |
| Cd (mg kg–1) | 0.73 *** | 0.82 *** | −0.76 *** | −0.74 *** | −0.73 *** |
| Soil Cd (mg kg–1) | 0.51 * | 0.55 * | −0.54 * | −0.52 * | −0.51 * |
| Variable | RC/CSo | RC/ABS | TRo/DIo | ETo/(TRo-ETo) | PIabs |
| Zn (mg kg–1) | −0.63 ** | −0.67 ** | −0.54 * | −0.54 * | −0.52 * |
| Cd (mg kg–1) | −0.58 * | −0.79 *** | −0.65 ** | −0.59 * | −0.60 * |
| Soil Cd (mg kg–1) | −0.20 n.s. | −0.58 * | −0.44 n.s. | −0.45 n.s. | −0.44 n.s. |
| Variable | ABS/RC | TRo/RC | ETo/RC | DIo/RC | DIo/CSo |
| Zn (mg kg–1) | 0.75 *** | 0.71 ** | −0.60 * | 0.73 ** | 0.48 n.s. |
| Cd (mg kg–1) | 0.86 *** | 0.81 *** | −0.62 ** | 0.84 *** | 0.64 ** |
| Soil Cd (mg kg–1) | 0.59 * | 0.56 * | −0.40 n.s. | 0.59 * | 0.56 * |
| Variable | PItotal | φDo | Zn (mg kg–1) | Cd (mg kg–1) | Soil Cd (mg kg–1) |
| Zn (mg kg–1) | −0.34 n.s. | 0.62 n.s. | 0.65 *** | 0.34 n.s. | |
| Cd (mg kg–1) | −0.46 * | 0.73 ** | 0.65 *** | 0.63 ** | |
| Soil Cd (mg kg–1) | −0.40 n.s. | 0.53 n.s. | 0.34 n.s. | 0.63 ** |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Franić, M.; Galić, V.; Lončarić, Z.; Šimić, D. Genotypic Variability of Photosynthetic Parameters in Maize Ear-Leaves at Different Cadmium Levels in Soil. Agronomy 2020, 10, 986. https://doi.org/10.3390/agronomy10070986
Franić M, Galić V, Lončarić Z, Šimić D. Genotypic Variability of Photosynthetic Parameters in Maize Ear-Leaves at Different Cadmium Levels in Soil. Agronomy. 2020; 10(7):986. https://doi.org/10.3390/agronomy10070986
Chicago/Turabian StyleFranić, Mario, Vlatko Galić, Zdenko Lončarić, and Domagoj Šimić. 2020. "Genotypic Variability of Photosynthetic Parameters in Maize Ear-Leaves at Different Cadmium Levels in Soil" Agronomy 10, no. 7: 986. https://doi.org/10.3390/agronomy10070986
APA StyleFranić, M., Galić, V., Lončarić, Z., & Šimić, D. (2020). Genotypic Variability of Photosynthetic Parameters in Maize Ear-Leaves at Different Cadmium Levels in Soil. Agronomy, 10(7), 986. https://doi.org/10.3390/agronomy10070986







