Changes in Growth and Physiological Parameters of ×Amarine Following an Exogenous Application of Gibberellic Acid and Methyl Jasmonate
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Location, Plant Materials, and Growth Conditions
2.2. Experimental Design
2.3. Determination of Plant Growth Parameters
2.4. Determination of Gas Exchange Rate, Chlorophyll Fluorescence, and Leaf Relative Chlorophyll Content
2.5. Determination of Total Sugars and Total Protein Content in Bulbs
2.6. Data Analysis
3. Results
3.1. Effects of Foliar Application of GA3 and MeJA Solutions on Growth, Flowering, and Bulb Yield
3.2. Effects of Foliar Application of GA3 and MeJA Solutions on Gas Exchange Rate
3.3. Effects of Foliar Application of GA3 and MeJA Solutions on Chlorophyll Fluorescence and SPAD
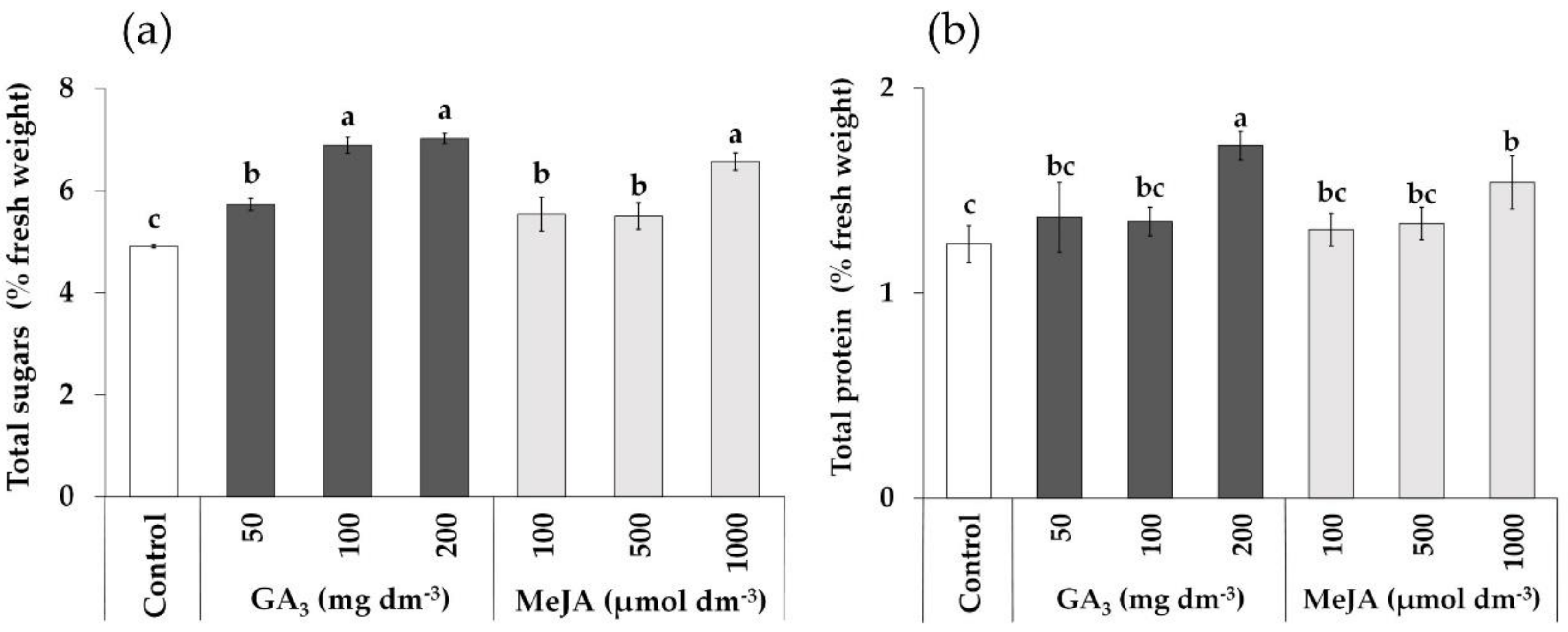
3.4. Effects of Foliar Application of GA3 and MeJA Solutions on Total Sugars and Total Protein Content
4. Discussion
4.1. Effect of GA3 and MeJA on Growth, Flowering, and Bulb Yield
4.2. Effect of GA3 and MeJA on Physiological Parameters
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Seaton, K.; Bettin, A.; Grüneberg, H. New ornamental plants for horticulture. In Horticulture: Plants for People and Places; Dixonand, G.R., Aldous, D.E., Eds.; Springer: Dordrecht, The Netherlands, 2014; pp. 435–463. [Google Scholar]
- Kamenetsky, R.; Miller, W.B. The global trade in ornamental geophytes. Chron. Hortic. 2010, 50, 27–30. [Google Scholar]
- Benschop, M.; Kamenetsky, R.; Nard, M.L.; Okubo, H.; De Hertogh, A. The Global flower bulb industry: Production, utilisation, research. Hortic. Rev. 2010, 36, 1–115. [Google Scholar]
- Hadas, R.; Kamenetsky, R.; Fragman-Sapir, O. Ex-Situ conservation of Israel’s native geophytes—Source for development of new ornamental crops. Isr. J. Plant Sci. 2009, 57, 277–285. [Google Scholar] [CrossRef]
- Coertze, A.F.; Louw, E. The breeding of interspecies and intergenera hybrids in the Amaryllidaceae. Acta Hortic. 1990, 266, 349–352. [Google Scholar] [CrossRef]
- Theron, K.I.; De Hertogh, A.A. Amaryllidaceae: Geophytic growth, development, and flowering. Hortic. Rev. 2001, 25, 1–70. [Google Scholar]
- Cahlíková, L.; Vaněčková, N.; Šafratová, M.; Breiterová, K.; Blunden, G.; Hulcová, D.; Opletal, L. The Genus Nerine Herb. (Amaryllidaceae): Ethnobotany, Phytochemistry, and Biological Activity. Molecules 2019, 24, 4238. [Google Scholar] [CrossRef]
- Duncan, G. Amaryllis magic: Feature. Veld Flora 2004, 90, 142–147. [Google Scholar]
- Amarine Belladiva. Available online: https://www.florapodium.com/index.php/en/amarine-belladiva-en (accessed on 5 June 2020).
- Theron, K.I.; Jacobs, G. Comparative growth and development of Nerine bowdenii W. Watson: Bulbs In Situ versus replanted. Hort. Sci. 1994, 29, 1493–1496. [Google Scholar] [CrossRef]
- Theron, K.I.; Jacobs, G. Inflorescence abortion in Nerine bowdenii W. Watts. Acta Hortic. 1992, 32, 97–104. [Google Scholar] [CrossRef]
- Theron, K.I.; Jacobs, G. The effect of irradiance, defoliation, and bulb size on flowering of Nerine bowdenii W. Watson (Amaryllidaceae). J. Amer. Soc. Hort. Sci. 1996, 121, 115–122. [Google Scholar] [CrossRef]
- Sajjad, Y.; Jaskani, M.J.; Asif, M.; Qasim, M. Application of plant growth regulators in ornamental plants: A review. Pak. J. Agric. Sci. 2017, 54. [Google Scholar] [CrossRef]
- Salachna, P.; Zawadzińska, A. Effect of daminozide and flurprimidol on growth, flowering and bulb yield of Eucomis autumnalis (Mill.) Chitt. Folia Hortic. 2017, 29, 33–38. [Google Scholar] [CrossRef][Green Version]
- Saniewski, M.; Kawa-Miszczak, L.; Wegrzynowicz-Lesiak, E. Role of ABA, gibberellins and auxin in dormancy and dormancy release of tulip bulbs. In Dormancy in Plants: From Whole Plant Behaviour to Cellular Control; Viémont, J., Crabbé, J., Eds.; CAB International Publishing: New York, NY, USA, 2000; pp. 227–245. [Google Scholar]
- Bonnet-Masimbert, M.; Zaerr, J.B. The role of plant growth regulators in promotion of flowering. J. Plant Growth Regul. 1987, 6, 13–35. [Google Scholar] [CrossRef]
- Ionescu, I.A.; Møller, B.L.; Sánchez-Pérez, R. Chemical control of flowering time. J. Exp. Bot. 2017, 68, 369–382. [Google Scholar] [CrossRef]
- Dos Santos, D.S.; Cardoso-Gustavson, P.; Nievola, C.C. Stem elongation of ornamental bromeliad in tissue culture depends on the temperature even in the presence of gibberellic acid. Acta Physiol. Plant. 2017, 39, 230. [Google Scholar] [CrossRef]
- Francis, D.; Sorrell, D.A. The interface between the cell cycle and plant growth regulators: A mini review. J. Plant Growth Regul. 2001, 33, 1–12. [Google Scholar] [CrossRef]
- Iftikhar, A.; Ali, S.; Yasmeen, T.; Arif, M.S.; Zubair, M.; Rizwan, M.; Alhaithloul, H.A.S.; Alayafi, A.A.M.; Soliman, M.H. Effect of gibberellic acid on growth, photosynthesis and antioxidant defense system of wheat under zinc oxide nanoparticle stress. Environ. Pollut. 2019, 254, 113109. [Google Scholar] [CrossRef]
- Koo, A.J.; Howe, G.A. The wound hormone jasmonate. Phytochemistry 2009, 70, 1571–1580. [Google Scholar] [CrossRef]
- Linkies, A.; Leubner-Metzger, G. Beyond gibberellins and abscisic acid: How ethylene and jasmonates control seed germination. Plant Cell Rep. 2012, 31, 253–270. [Google Scholar] [CrossRef]
- Golovatskaya, I.F.; Karnachuk, R.A. Effect of jasmonic acid on morphogenesis and photosynthetic pigment level in Arabidopsis seedlings grown under green light. Russ. J. Plant Physiol. 2008, 55, 220–224. [Google Scholar] [CrossRef]
- Reyes-Díaz, M.; Lobos, T.; Cardemil, L.; Nunes-Nesi, A.; Retamales, J.; Jaakola, L.; Alberdi, M.; Ribera-Fonseca, A. Methyl jasmonate: An alternative for improving the quality and health properties of fresh fruits. Molecules 2016, 21, 567. [Google Scholar] [CrossRef]
- Ahmad, P.; Rasool, S.; Gul, A.; Sheikh, S.A.; Akram, N.A.; Ashraf, M.; Kazi, A.M.; Gucel, S. Jasmonates: Multifunctional roles in stress tolerance. Front. Plant Sci. 2016, 7, 813. [Google Scholar] [CrossRef] [PubMed]
- Albrechtova, J.T.P.; Ullmann, J. Methyl jasmonate inhibits growth and flowering in Chenopodium rubrum. Biol. Plant. 1994, 36, 317–319. [Google Scholar] [CrossRef]
- Kazemi, M. Effect of foliar application with salicylic acid and methyl jasmonate on growth, flowering, yield and fruit quality of tomato. Bull. Environ. Pharmacol. Life Sci. 2014, 3, 154–158. [Google Scholar]
- Maciejewska, B.; Kopcewicz, J. Inhibitory effect of methyl jasmonate on flowering and elongation growth in Pharbitis nil. J. Plant Growth Regul. 2002, 21, 216–223. [Google Scholar] [CrossRef]
- Pak, H.; Guo, Y.; Chen, M.; Chen, K.; Li, Y.; Hua, S.; Shamsi, I.; Meng, H.; Shi, C.; Jiang, L.; et al. The effect of exogenous methyl jasmonate on the flowering time, floral organ morphology, and transcript levels of a group of genes implicated in the development of oilseed rape flowers (Brassica napus L.). Planta 2009, 231, 79–91. [Google Scholar] [CrossRef]
- Jásik, J.; De Klerk, G.J. Effect of methyl jasmonate on morphology and dormancy development in lily bulblets regenerated in vitro. J. Plant Growth Regul. 2006, 25, 45–51. [Google Scholar] [CrossRef]
- Podwyszyńska, M.; Kosson, R.; Treder, J. Polyamines and methyl jasmonate in bulb formation of in vitro propagated tulips. Plant Cell Tissue Organ Cult. 2015, 123, 591–605. [Google Scholar] [CrossRef]
- Mikiciuk, G.; Sas-Paszt, L.; Mikiciuk, M.; Derkowska, E.; Trzciński, P.; Głuszek, S.; Lisek, A.; Wera-Bryl, S.; Rudnicka, J. Mycorrhizal frequency, physiological parameters, and yield of strawberry plants inoculated with endomycorrhizal fungi and rhizosphere bacteria. Mycorrhiza 2019, 29, 489–501. [Google Scholar] [CrossRef]
- Kalaji, H.M.; Bosa, K.; Kościelniak, J.; Żuk-Gołaszewska, K. Effects of salt stress on photosystem II efficiency and CO2 assimilation of two Syrian barley landraces. Environ. Exp. Bot. 2011, 73, 64–72. [Google Scholar] [CrossRef]
- Manimaran, P.; Ghosh, S.; Priyanka, R. Bulb size and growth regulators on the growth and performance of bulbous ornamental crops—A review. Chem. Sci. Rev. Lett. 2017, 6, 1277–1284. [Google Scholar]
- Salachna, P.; Grzeszczuk, M.; Soból, M. Effects of chitooligosaccharide coating combined with selected ionic polymers on the stimulation of Ornithogalum saundersiae growth. Molecules 2017, 22, 1903. [Google Scholar] [CrossRef] [PubMed]
- Ramzan, F.; Younis, A.; Riaz, A.; Ali, S.; Siddique, M.I.; Lim, K.B. Pre-planting exogenous application of gibberellic acid influences sprouting, vegetative growth, flowering, and subsequent bulb characteristics of ‘Ad-Rem’ tulip. Hortic. Environ. Biotechnol. 2014, 55, 479–488. [Google Scholar] [CrossRef]
- Sauter, M.; Kende, H. Gibberellin-induced growth and regulation of the cell division cycle in deepwater rice. Planta 1992, 188, 362–368. [Google Scholar] [CrossRef] [PubMed]
- Diallo, A.O.; Agharbaoui, Z.; Badawi, M.A.; Ali-Benali, M.A.; Moheb, A.; Houde, M.; Sarhan, F. Transcriptome analysis of an mvp mutant reveals important changes in global gene expression and a role for methyl jasmonate in vernalization and flowering in wheat. J. Exp. Bot. 2014, 65, 2271–2286. [Google Scholar] [CrossRef]
- Mishra, S.K.; Mishra, S.; Bahadur, V. Effect of growth regulators on growth, yield and shelf life in amaryllis lily (Amaryllis belladona) cv. Zephyranthes. J. Pharmacogn. Phytochem. 2019, 8, 1217–1219. [Google Scholar]
- Fortanier, E.J.; Van Brenk, G.; Wellensiek, S.J. Growth and flowering of Nerine flexuosa alba. Sci. Hortic. 1979, 11, 281–290. [Google Scholar] [CrossRef]
- Brown, N.R. The Reproductive Biology of Nerine (Amaryllidaceae). Ph.D. Thesis, University of Tasmania, Tasmania, Australia, 1999. [Google Scholar]
- Zhai, Q.; Zhang, X.; Wu, F.; Feng, H.; Deng, L.; Xu, L.; Zhang, M.; Wang, Q.; Li, C. Transcriptional mechanism of jasmonate receptor COI1-mediated delay of flowering time in Arabidopsis. Plant Cell 2015, 27, 2814–2828. [Google Scholar]
- Cig, A.; Basdogan, G. In vitro propagation techniques for some geophyte ornamental plants with high economic value. Int. J. Second. Metab. 2015, 2, 27–49. [Google Scholar]
- Pogroszewska, E.; Laskowska, H.; Durlak, W. The effect of gibberellic acid and benzyladenine on the yield of (Allium karataviense Regel.) ‘Ivory Queen’. Acta Sci. Pol. Hortorum Cultus 2007, 6, 15–19. [Google Scholar]
- Liu, H.; Deng, R.; Huang, C.; Cheng, Z.; Meng, H. Exogenous gibberellins alter morphology and nutritional traits of garlic (Allium sativum L.) bulb. Sci. Hortic. 2019, 246, 298–306. [Google Scholar] [CrossRef]
- Rademacher, W. Chemical regulators of gibberellin status and their application in plant production. In Annual Plant Reviews; Wiley-Blackwell: Hoboken, NJ, USA, 2016; Volume 49, pp. 359–404. [Google Scholar]
- Žel, J.; Debeljak, N.; Ucman, R.; Ravnikar, M. The effect of jasmonic acid, sucrose and darkness on garlic (Allium sativum L. cv. Ptujski jesenski) bulb formation in vitro. In Vitro Cell. Dev. Biol. Plant 1997, 33, 231–235. [Google Scholar] [CrossRef]
- Santos, I.; Salema, R. Promotion by jasmonic acid of bulb formation in shoot cultures of Narcissus triandrus L. J. Plant Growth Regul. 2000, 30, 133–138. [Google Scholar] [CrossRef]
- Nojiri, H.; Yamane, H.; Seto, H.; Yamaguchi, I.; Murofushi, N.; Yoshihara, T.; Shibaoka, H. Qualitative and quantitative analysis of endogenous jasmonic acid in bulbing and non-bulbing onion plants. Plant Cell Physiol. 1992, 33, 1225–1231. [Google Scholar]
- Kumudini, B.S.; Patil, S.V. Role of plant hormones in improving photosynthesis. In Photosynthesis, Productivity and Environmental Stress; Ahmad, P., Ahanger, M.A., Alam, P., Alyemeni, M.N., Eds.; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 2019; pp. 215–240. [Google Scholar]
- Arteca, R.N. Manipulation of growth and photosynthetic processes by plant growth regulators. In Plant Growth Substances; Arteca, R.N., Ed.; Springer: Boston, MA, USA, 1996; pp. 240–272. [Google Scholar]
- Peng, S.; Krieg, D.R.; Girma, F.S. Leaf photosynthetic rate is correlated with biomass and grain production in grain sorghum lines. Photosynth. Res. 1991, 28, 1–7. [Google Scholar] [CrossRef]
- Ahmadi, F.I.; Karimi, K.; Struik, P.C. Effect of exogenous application of methyl jasmonate on physiological and biochemical characteristics of Brassica napus L. cv. Talaye under salinity stress. S. Afr. J. Bot. 2018, 115, 5–11. [Google Scholar] [CrossRef]
- Tavallali, V.; Karimi, S. Methyl jasmonate enhances salt tolerance of almond rootstocks by regulating endogenous phytohormones, antioxidant activity and gas-exchange. J. Plant Physiol. 2019, 234, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Miller, W.B. A review of carbohydrate metabolism in geophytes. Acta Hortic. 1992, 325, 239–246. [Google Scholar] [CrossRef]
- Wakchaure, G.C.; Minhas, P.S.; Meena, K.K.; Singh, N.P.; Hegade, P.M.; Sorty, A.M. Growth, bulb yield, water productivity and quality of onion (Allium cepa L.) as affected by deficit irrigation regimes and exogenous application of plant bio–regulators. Agric. Water Manag. 2018, 199, 1–10. [Google Scholar] [CrossRef]
- Sarkar, D.; Pandey, S.K.; Sharma, S. Cytokinins antagonize the jasmonates action on the regulation of potato (Solanum tuberosum) tuber formation in vitro. Plant Cell Tissue Organ Cult. 2006, 87, 285–295. [Google Scholar] [CrossRef]
- Theron, K.I.; Jacobs, G. Changes in carbohydrate composition of the different bulb components of Nerine bowdenii W. Watson (Amaryllidaceae). J. Amer. Soc. Hort. Sci. 1996, 121, 343–346. [Google Scholar] [CrossRef]


| Treatment | Leaves (no.) | Leaf Length (cm) | Stem Length (cm) | Florets (no.) | |
|---|---|---|---|---|---|
| Control | 9.00 ± 0.50 d,* | 49.9 ± 1.49 bc | 72.5 ± 4.39 a | 7.97 ± 0.64 a | |
| GA3 | 50 mg dm−3 | 11.2 ± 0.76 bc | 51.8 ± 1.07 bc | 72.5 ± 0.95 a | 8.33 ± 0.76 a |
| 100 mg dm−3 | 12.3 ± 0.25 ba | 53.8 ± 1.97 b | 74.0 ± 4.52 a | 8.33 ± 0.76 a | |
| 200 mg dm−3 | 12.8 ± 0.29 a | 58.1 ± 2.28 a | 75.6 ± 4.15 a | 7.97 ± 0.64 a | |
| MeJA | 100 µmol dm−3 | 9.00 ± 0.50 d | 49.1 ± 1.50 c | 72.3 ± 2.78 a | 7.87 ± 0.15 a |
| 500 µmol dm−3 | 10.5 ± 0.50 c | 51.1 ± 0.75 bc | 71.6 ± 3.45 a | 7.58 ± 0.38 a | |
| 1000 µmol dm−3 | 11.5 ± 0.50 abc | 50.4 ± 0.80 bc | 73.5 ± 4.20 a | 7.50 ± 0.43 a | |
| Treatment | Time to Anthesis (d) | Bulbs Flowering (%) | |
|---|---|---|---|
| Control | 188 ± 1.00 b* | 46.5 ± 8.85 bc | |
| GA3 | 50 mg dm−3 | 185 ± 2.65 bc | 82.2 ± 16.3 a |
| 100 mg dm−3 | 177 ± 2.65 c | 82.2 ± 16.3 a | |
| 200 mg dm−3 | 176 ± 1.00 c | 65.6 ± 4.21 ab | |
| MeJA | 100 µmol dm−3 | 206 ± 5.51 a | 30.4 ± 6.66 c |
| 500 µmol dm−3 | 206 ± 5.03 a | 22.8 ± 8.28 c | |
| 1000 µmol dm−3 | 203 ± 5.86 a | 28.0 ± 7.68 c | |
| Treatment | Bulb Weight (g) | Daughter Bulbs (no.) | |
|---|---|---|---|
| Control | 97.7 ± 1.46 c* | 1.28 ± 0.03 b | |
| GA3 | 50 mg dm−3 | 107 ± 3.61 b | 1.78 ± 0.20 ab |
| 100 mg dm−3 | 128 ± 5.51 a | 2.16 ± 0.15 a | |
| 200 mg dm−3 | 134 ± 4.26 a | 2.30 ± 0.30 a | |
| MeJA | 100 µmol dm−3 | 99.1 ± 2.80 c | 1.35 ± 0.13 b |
| 500 µmol dm−3 | 100 ± 3.61 c | 2.27 ± 0.25 a | |
| 1000 µmol dm−3 | 112 ± 4.63 b | 2.27 ± 0.25 a | |
| Treatment | A (μmol m−2 s−1) | E (mmol m−2 s−1) | ωW (mmol mol−1) | gS (mol m−2 s−1) | ci (μmol mol−1) | |
|---|---|---|---|---|---|---|
| Control | 6.67 ± 0.41 bc* | 0.82 ± 0.21 d | 4.67 ± 1.14 bc | 0.10 ± 0.02 d | 409 ± 40.3 c | |
| GA3 | 50 mg dm−3 | 7.40 ± 0.60 bc | 1.49 ± 0.12 bc | 6.99 ± 0.60 a | 0.28 ± 0.03 b | 449 ± 4.12 b |
| 100 mg dm−3 | 7.75 ± 0.65 ab | 1.41 ± 0.26 c | 5.75 ± 0.98 ab | 0.31 ± 0.07 b | 455 ± 9.66 ab | |
| 200 mg dm−3 | 8.99 ± 0.84 a | 1.44 ± 0.15 c | 5.40 ± 0.65 bc | 0.34 ± 0.07 b | 470 ± 9.35 ab | |
| MeJA | 100 µmol dm−3 | 6.32 ± 0.27 c | 1.75 ± 0.09 a | 4.32 ± 0.21 c | 0.43 ± 0.05 a | 444 ± 6.00 b |
| 500 µmol dm−3 | 6.16 ± 0.52 c | 1.72 ± 0.07 ab | 4.16 ± 0.40 c | 0.44 ± 0.04 a | 468 ± 6.57 ab | |
| 1000 µmol dm−3 | 7.10 ± 0.77 bc | 1.05 ± 0.14 d | 5.10 ± 1.48 bc | 0.21 ± 0.04 c | 482 ± 21.1 a | |
| Treatment | FV/FM | PI | Area | SPAD | |
|---|---|---|---|---|---|
| Control | 0.79 ± 0.03 ab* | 1.72 ± 0.39 ab | 49,428 ± 8097 ab | 52.2 ± 1.40 c | |
| GA3 | 50 mg dm−3 | 0.77 ± 0.03 ab | 1.71 ± 0.30 ab | 40,656 ± 6448 b | 56.9 ± 0.67 b |
| 100 mg dm−3 | 0.78 ± 0.02 ab | 1.66 ± 0.18 b | 47,401 ± 7553 ab | 58.5 ± 1.35 b | |
| 200 mg dm−3 | 0.82 ± 0.04 a | 2.02 ± 0.20 a | 59,420 ± 13,999 a | 66.0 ± 2.48 a | |
| MeJA | 100 µmol dm−3 | 0.77 ± 0.05 ab | 1.73 ± 0.08 ab | 41,702 ± 6330 b | 51.8 ± 0.40 c |
| 500 µmol dm−3 | 0.73 ± 0.04 b | 1.66 ± 0.06 b | 47,593 ± 7939 ab | 55.0 ± 1.50 bc | |
| 1000 µmol dm−3 | 0.76 ± 0.05 ab | 1.76 ± 0.12 ab | 37,339 ± 14,819 b | 59.5 ± 2.35 b | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salachna, P.; Mikiciuk, M.; Zawadzińska, A.; Piechocki, R.; Ptak, P.; Mikiciuk, G.; Pietrak, A.; Łopusiewicz, Ł. Changes in Growth and Physiological Parameters of ×Amarine Following an Exogenous Application of Gibberellic Acid and Methyl Jasmonate. Agronomy 2020, 10, 980. https://doi.org/10.3390/agronomy10070980
Salachna P, Mikiciuk M, Zawadzińska A, Piechocki R, Ptak P, Mikiciuk G, Pietrak A, Łopusiewicz Ł. Changes in Growth and Physiological Parameters of ×Amarine Following an Exogenous Application of Gibberellic Acid and Methyl Jasmonate. Agronomy. 2020; 10(7):980. https://doi.org/10.3390/agronomy10070980
Chicago/Turabian StyleSalachna, Piotr, Małgorzata Mikiciuk, Agnieszka Zawadzińska, Rafał Piechocki, Piotr Ptak, Grzegorz Mikiciuk, Anna Pietrak, and Łukasz Łopusiewicz. 2020. "Changes in Growth and Physiological Parameters of ×Amarine Following an Exogenous Application of Gibberellic Acid and Methyl Jasmonate" Agronomy 10, no. 7: 980. https://doi.org/10.3390/agronomy10070980
APA StyleSalachna, P., Mikiciuk, M., Zawadzińska, A., Piechocki, R., Ptak, P., Mikiciuk, G., Pietrak, A., & Łopusiewicz, Ł. (2020). Changes in Growth and Physiological Parameters of ×Amarine Following an Exogenous Application of Gibberellic Acid and Methyl Jasmonate. Agronomy, 10(7), 980. https://doi.org/10.3390/agronomy10070980








