Supplementary LED Interlighting Improves Yield and Precocity of Greenhouse Tomatoes in the Mediterranean
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Growing Conditions
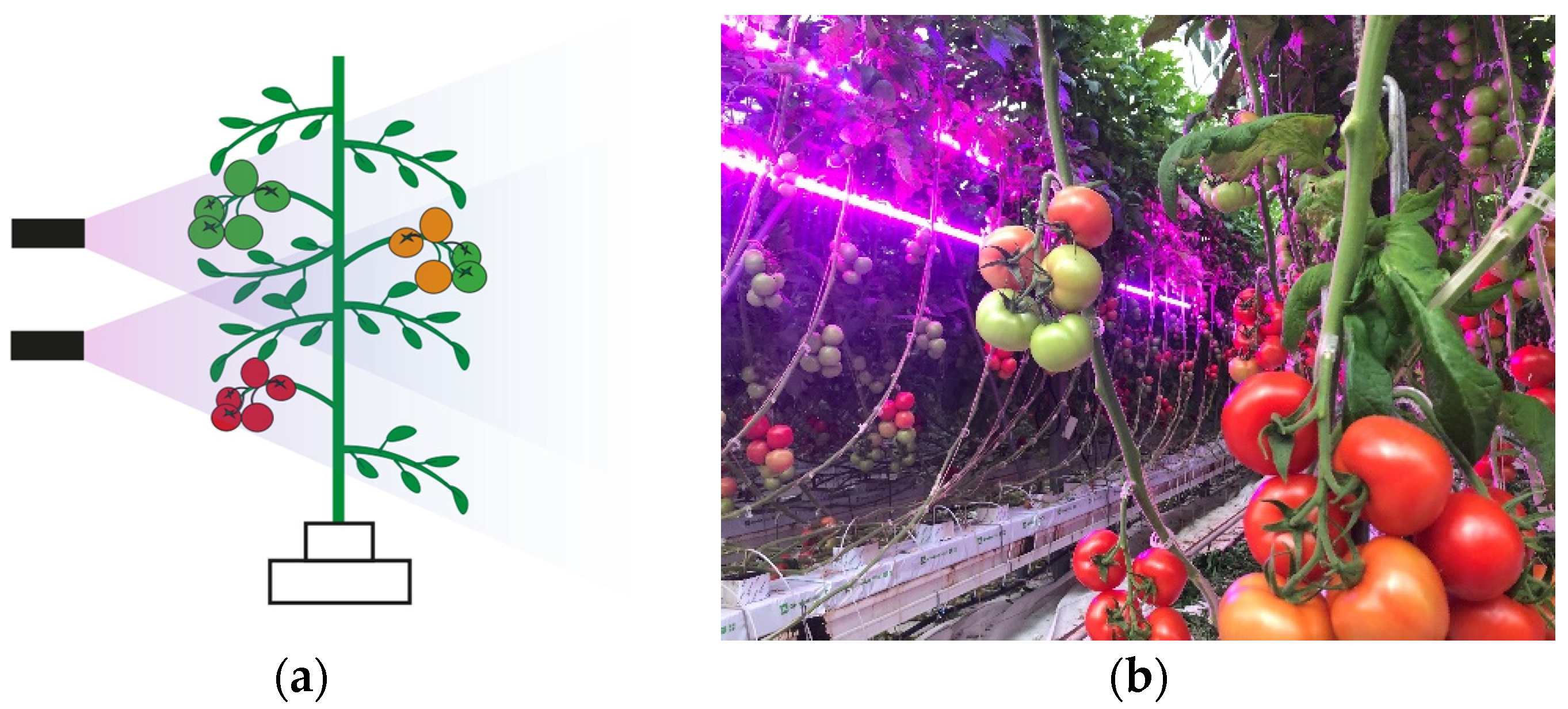
2.2. Light Treatment and Experimental Design
2.3. Vegetative and Fruit Growth Measurements
2.4. Physiological and Quality Measurements
2.5. Post-Harvest Measurements
2.6. Statistical Analysis
3. Results and Discussion
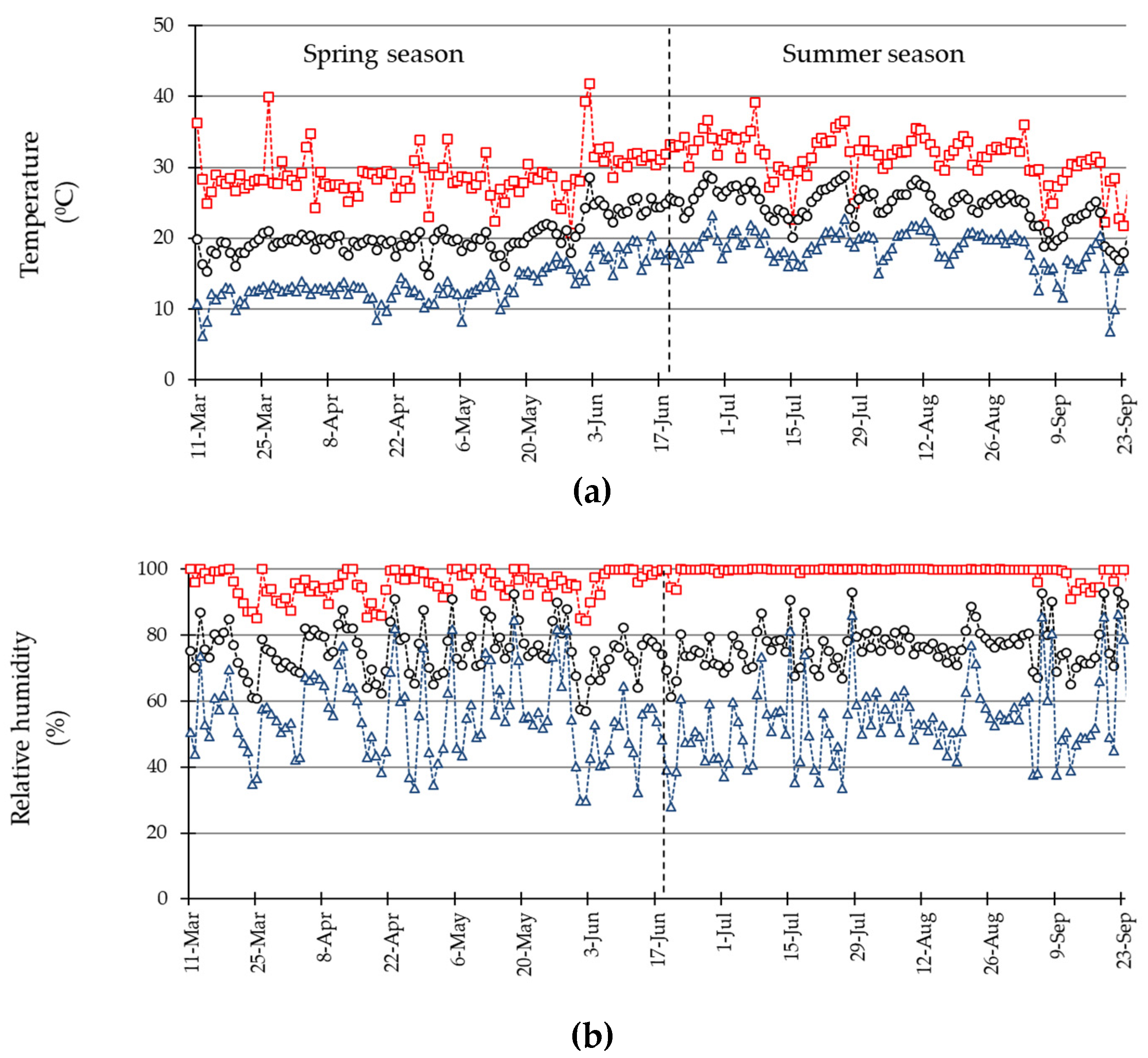
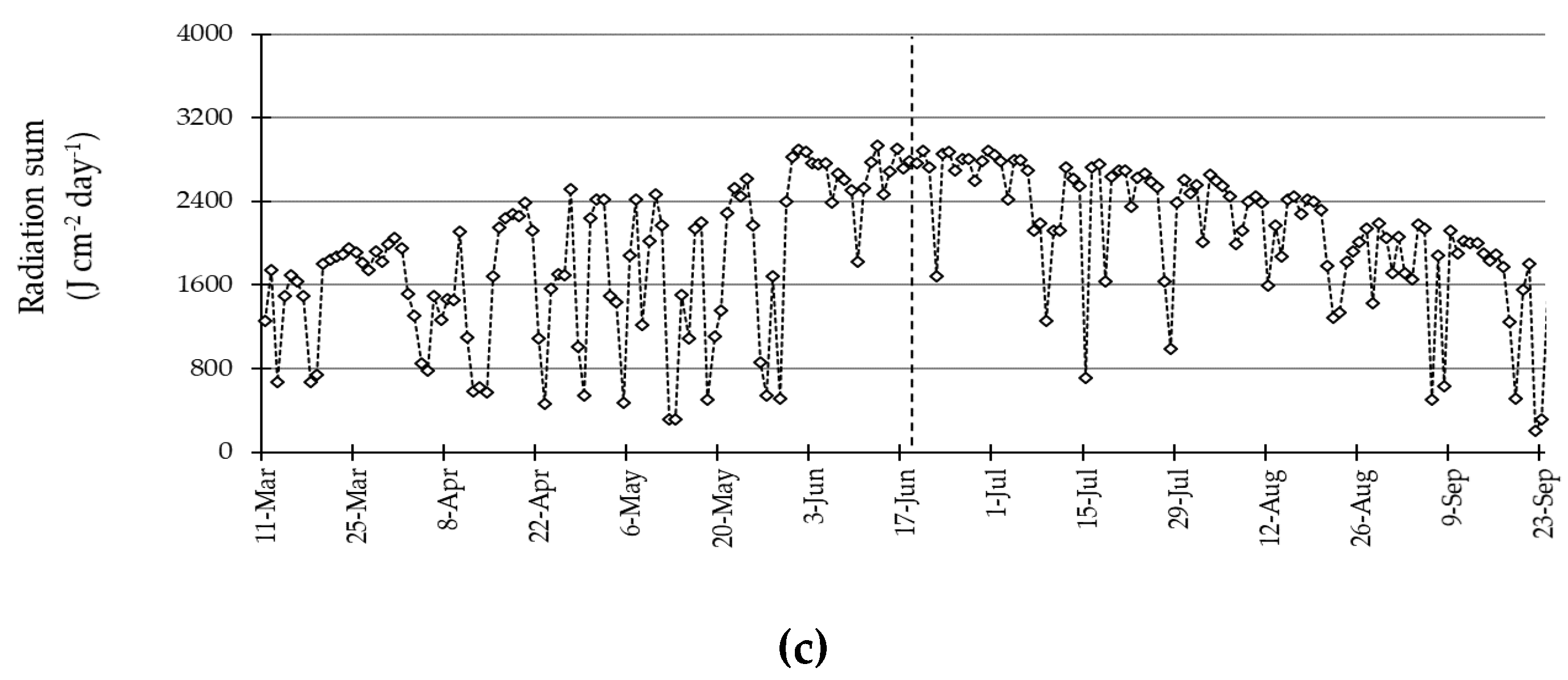
3.1. Environmental Conditions during the Experiment
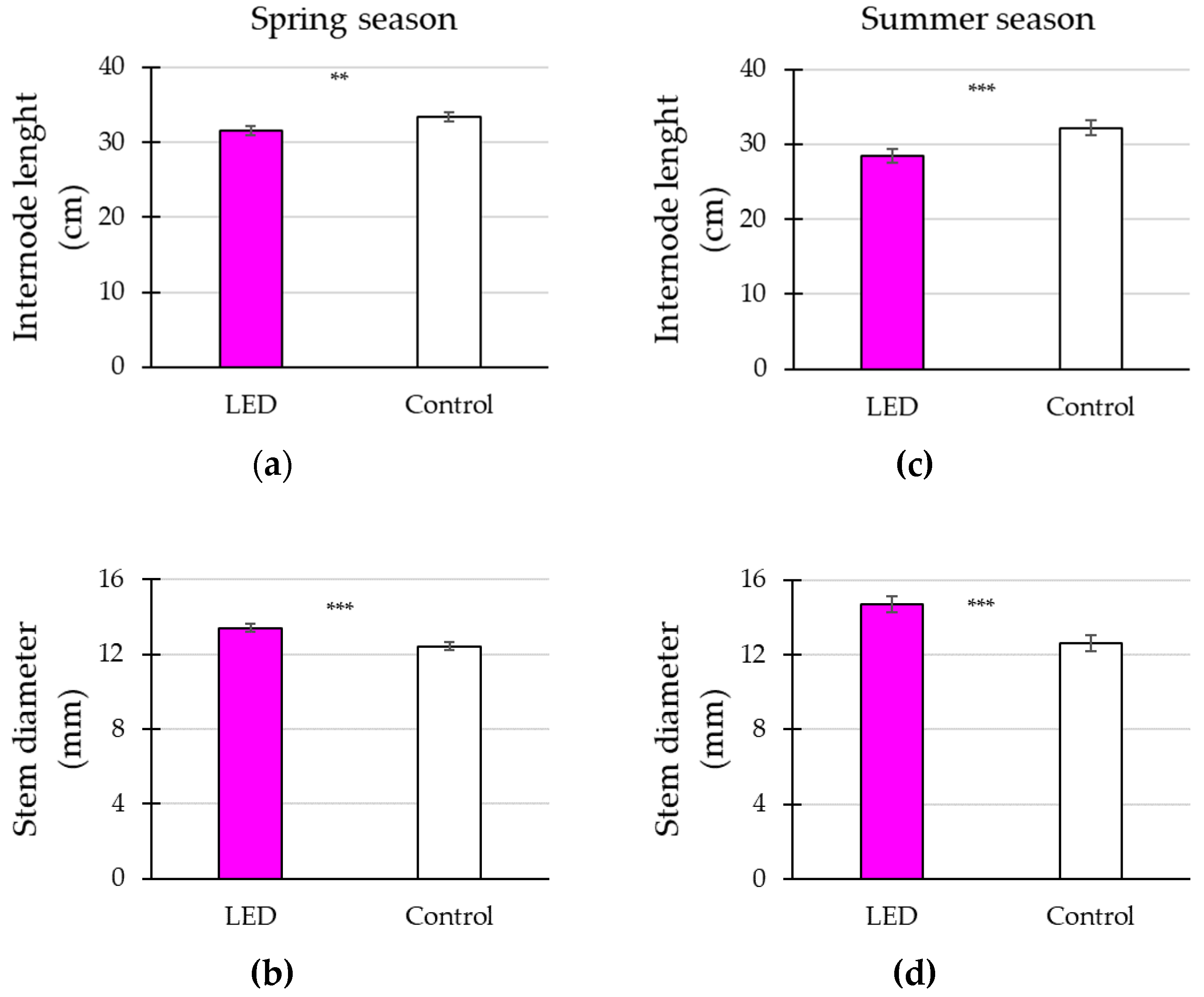
3.2. Plant Vegetative Growth in Response to Supplementary Lighting
3.3. LED Interlighting Improves Leaf Gas Exchanges and Alters Leaf Temperature
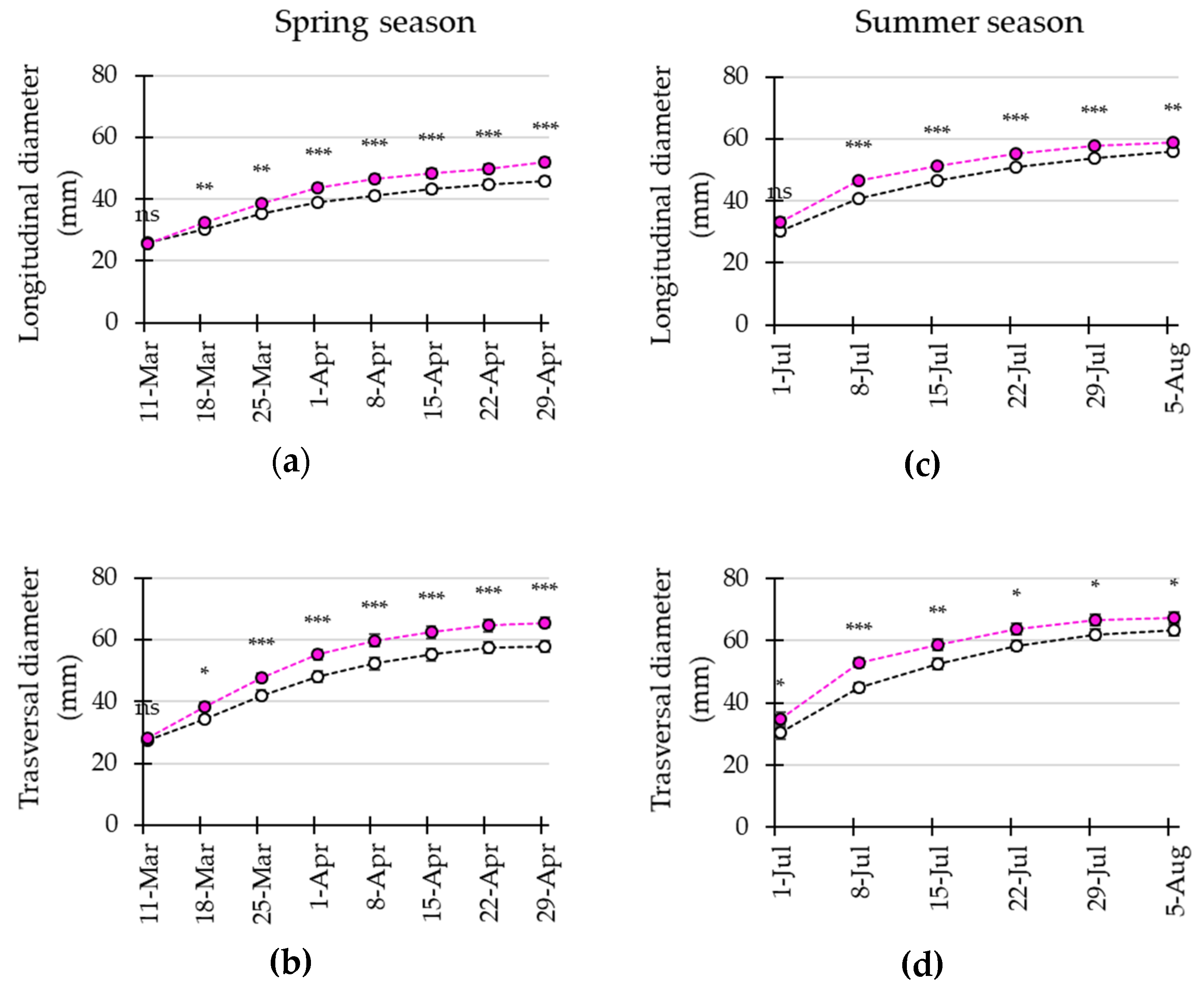
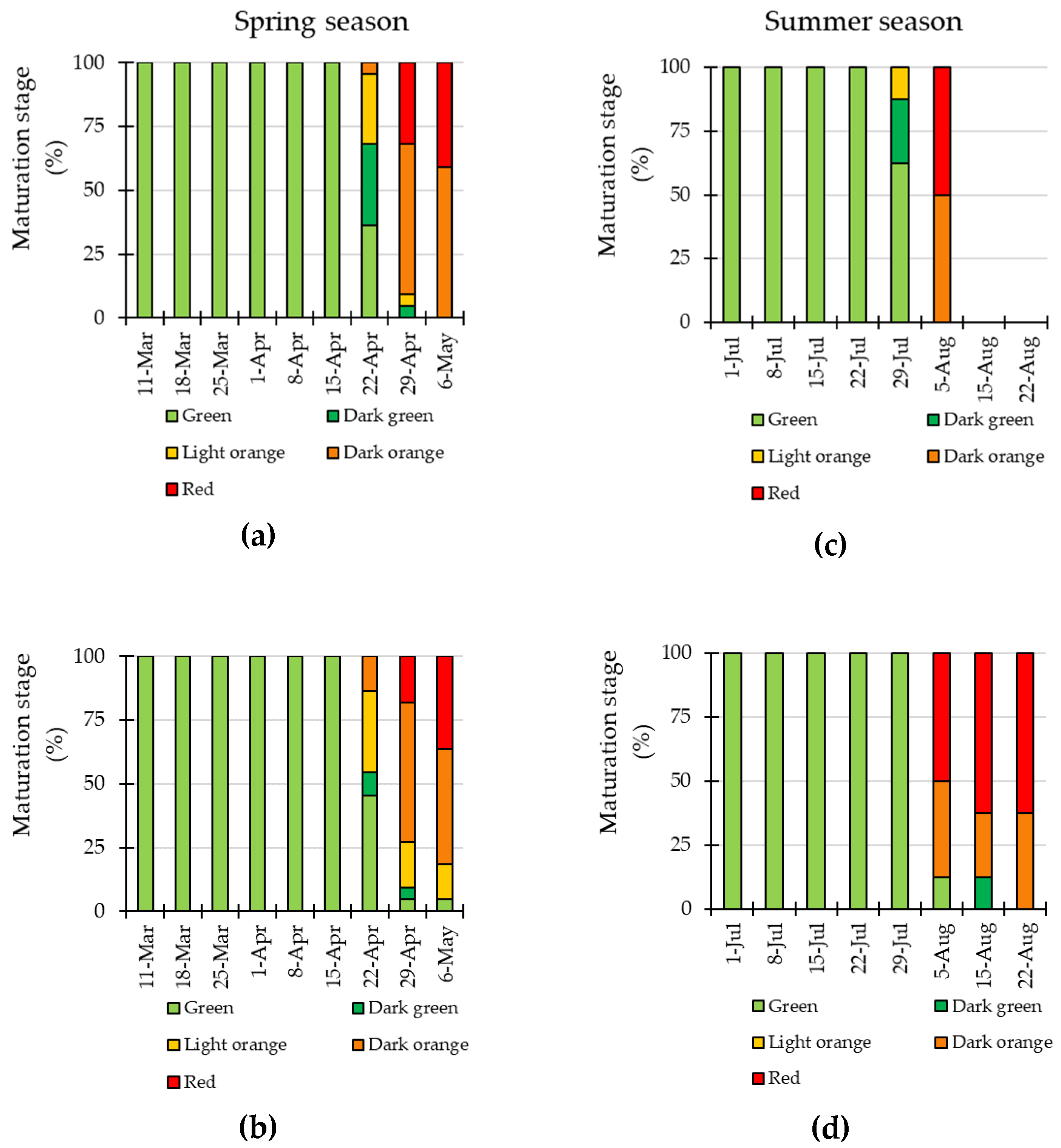
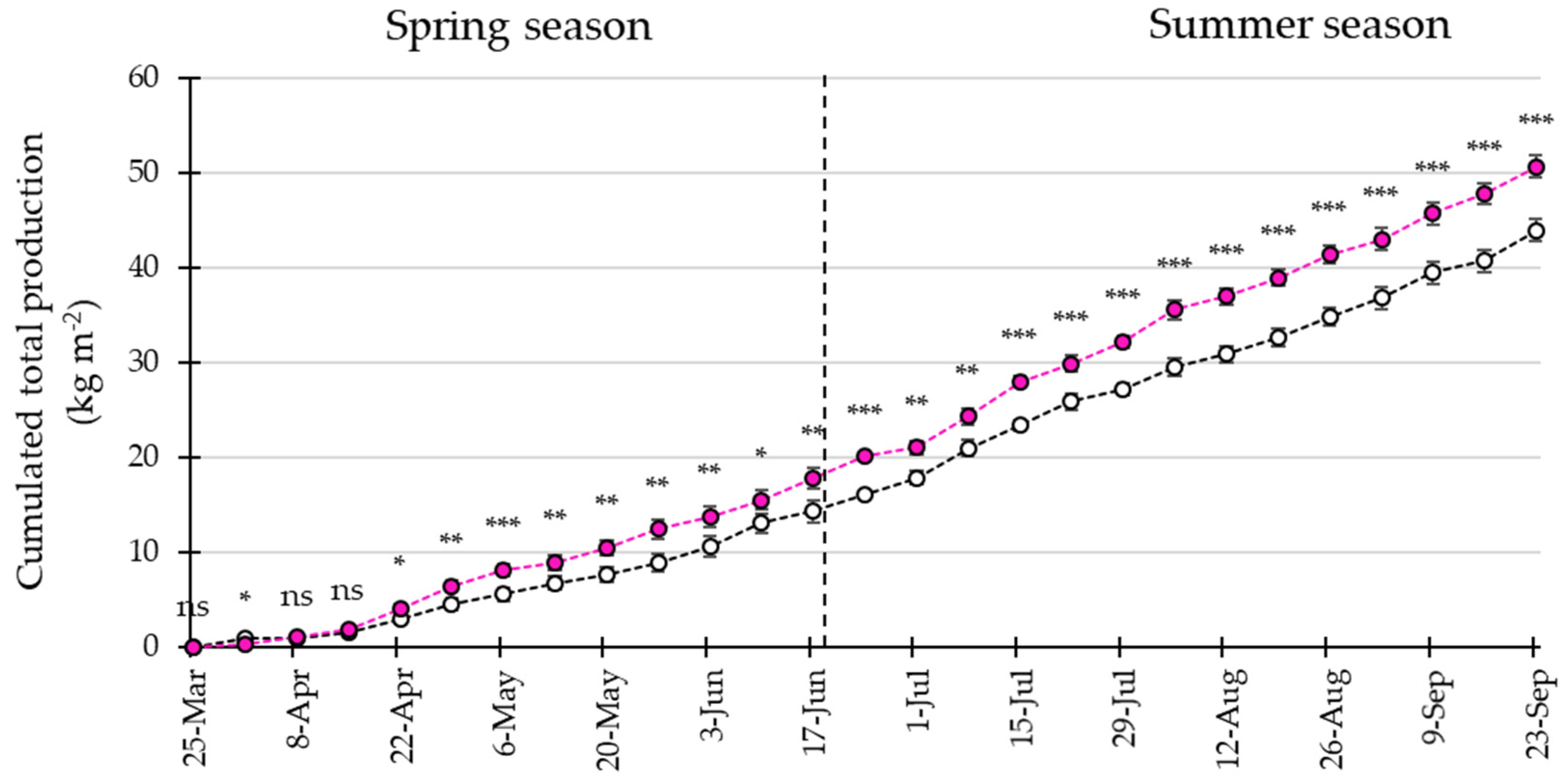
3.4. LED Interlighting Enhances Fruit Growth and Yield Parameters
3.5. Quality Parameters
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- FAOSTAT. Food and Agriculture Organization Corporate Statistical Database. Available online: http://www.fao.org/faostat/en/#data/QC (accessed on 13 March 2020).
- Jones, J.B., Jr. Tomato Plant Culture: In the Field, Greenhouse, and Home Garden, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2007; p. 16. [Google Scholar]
- Fernández, J.A.; Orsini, F.; Baeza, E.; Oztekin, G.B.; Muñoz, P.; Contreras, J.; Montero, J.I. Current trends in protected cultivation in Mediterranean climates. Eur. J. Hortic. Sci. 2018, 83, 294–305. [Google Scholar] [CrossRef]
- Gómez, C. Light-Emitting Diodes as an Alternative Supplemental Lighting Source for Greenhouse Tomato Propagation and Production. Ph.D. Dissertation, Purdue University, Lafayette, LA, USA, November 2014. [Google Scholar]
- De Pascale, S.; Orsini, F.; Caputo, R.; Palermo, M.A.; Barbieri, G.; Maggio, A. Seasonal and multiannual effects of salinisation on tomato yield and fruit quality. Funct. Plant Biol. 2012, 39, 689–698. [Google Scholar] [CrossRef] [PubMed]
- De Pascale, S.; Maggio, A.; Orsini, F.; Stanghellini, C.; Heuvelink, E. Growth response and radiation use efficiency in tomato exposed to short-term and long-term salinized soils. Sci. Hortic. 2015, 189, 139–149. [Google Scholar] [CrossRef]
- Lu, N.; Maruo, T.; Johkan, M.; Hohjo, M.; Tsukagoshi, S.; Ito, Y.; Ichimura, T.; Shinohara, Y. Effects of supplemental lighting within the canopy at different developing stages on tomato yield and quality of single-truss tomato plants grown at high density. Environ. Control Biol. 2012, 50, 1–11. [Google Scholar] [CrossRef]
- Lu, N.; Maruo, T.; Johkan, M.; Hohjo, M.; Tsukagoshi, S.; Ito, Y.; Ichimura, T.; Shinohara, Y. Effects of supplemental lighting with light-emitting diodes (LEDs) on tomato yield and quality of single-truss tomato plants grown at high planting density. Environ. Control Biol. 2012, 50, 63–74. [Google Scholar] [CrossRef]
- Pepin, S.; Fortier, E.; Béchard-Dubé, S.A.; Dorais, M.; Ménard, C.; Bacon, R. Beneficial Effects of Using a 3-D LED Interlighting System for Organic Greenhouse Tomato Grown in Canada under Low Natural Light Conditions. Acta Hortic. 2014, 1041, 239–246. [Google Scholar] [CrossRef]
- Tewolde, F.T.; Lu, N.; Shiina, K.; Maruo, T.; Takagaki, M.; Kozai, T.; Yamori, W. Nighttime Supplemental LED Inter-lighting Improves Growth and Yield of Single-Truss Tomatoes by Enhancing Photosynthesis in Both Winter and Summer. Front. Plant Sci. 2016, 7, 448. [Google Scholar] [CrossRef]
- Jiang, C.; Johkan, M.; Hohjob, M.; Tsukagoshi, S.; Ebihara, M.; Nakaminami, A.; Maruo, T. Photosynthesis, plant growth, and fruit production of single-truss tomato improves with supplemental lighting provided from underneath or within the inner canopy. Sci. Hortic. 2017, 222, 221–229. [Google Scholar] [CrossRef]
- Hao, X.; Little, C.; Zheng, J.M.; Cao, R. Far-red LEDs improve fruit production in greenhouse tomato grown under high-pressure sodium lighting. Acta Hortic. 2016, 1134, 95–102. [Google Scholar] [CrossRef]
- Park, Y.H.; Seo, B.S.; Seo, J.B.; Shin, G.H.; Yun, B.G. Effects of supplemental light on quality, yield, and growth rate of tomatoes. Acta Hortic. 2016, 1129, 105–110. [Google Scholar] [CrossRef]
- Pan, T.; Ding, J.; Qin, G.; Wang, Y.; Xi, L.; Yang, J.; Li, L.; Zhang, J.; Zou, Z. Interaction of supplementary light and CO2 enrichment improves growth, photosynthesis, yield, and quality of tomato in autumn through spring greenhouse production. HortScience 2019, 54, 246–252. [Google Scholar] [CrossRef]
- García-Caparros, P.; Chica, R.M.; Almansa, E.M.; Rull, A.; Rivas, L.A.; García-Buendía, A.; Barbero, F.J.; Lao, M.T. Comparisons of Different Lighting Systems for Horticultural Seedling Production Aimed at Energy Saving. Sustainability 2018, 10, 3351. [Google Scholar] [CrossRef]
- Bantis, F.; Smirnakou, S.; Ouzounis, T.; Koukounaras, A.; Ntagkas, N.; Radoglou, K. Current status and recent achievements in the field of horticulture with the use of light-emitting diodes (LEDs). Sci. Hortic. 2018, 235, 437–451. [Google Scholar] [CrossRef]
- Pennisi, G.; Blasioli, S.; Cellini, A.; Maia, L.; Crepaldi, A.; Braschi, I.; Spinelli, F.; Nicola, S.; Fernandez, J.A.; Stanghellini, C.; et al. Unravelling the role of red: Blue LED lights on resource use efficiency and nutritional properties of indoor grown sweet basil. Front. Plant Sci. 2019, 10, 305. [Google Scholar] [CrossRef] [PubMed]
- Pennisi, G.; Orsini, F.; Blasioli, S.; Cellini, A.; Crepaldi, A.; Braschi, I.; Spinelli, F.; Nicola, S.; Fernandez, J.A.; Stanghellini, C.; et al. Resource use efficiency of indoor lettuce (Lactuca sativa L.) cultivation as affected by red: Blue ratio provided by LED lighting. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Kittas, C.; Rigakis, N.; Katsoulas, N.; Bartzanas, T. Influence of shading screens on microclimate, growth and productivity of tomato. Acta Hortic. 2008, 807, 97–102. [Google Scholar] [CrossRef]
- Jun, H.; Imai, K.; Suzuki, Y. Effects of day temperature on gas exchange characteristics in tomato ecotypes. Sci. Hortic. 1990, 42, 321–327. [Google Scholar] [CrossRef]
- Kittas, C.; Katsoulas, N.; Bartzanas, T.; Bakker, S. Greenhouse climate control and energy use. In Good Agricultural Practices for Greenhouse Vegetable Crops: Principles for Mediterranean Climate Areas; Baudoin, W., Nono-Womdim, R., Lutaladio, N., Hodder, A., Castilla, N., Leonardi, C., De Pascale, S., Qaryouti, M., Eds.; FAO: Rome, Italy, 2013; Volume 217, pp. 63–95. [Google Scholar]
- Shamshiri, R.R.; Jones, J.W.; Thorp, K.R.; Ahmad, D.; Man, H.C.; Taheri, S. Review of optimum temperature, humidity, and vapour pressure deficit for microclimate evaluation and control in greenhouse cultivation of tomato: A review. Int. Agrophys. 2018, 32, 287–302. [Google Scholar] [CrossRef]
- Peet, M.M.; Welles, G. Greenhouse Tomato Production. In Tomatoes; Heuvelink, E., Ed.; CABI Publishing: Wallingford, UK, 2005; pp. 257–304. [Google Scholar]
- Tewolde, F.T.; Shiina, K.; Maruo, T.; Takagaki, M.; Kozai, T.; Yamori, W. Supplemental LED inter-lighting compensates for a shortage of light for plant growth and yield under the lack of sunshine. PLoS ONE 2018, 13. [Google Scholar] [CrossRef]
- Marcelis, L.F.M.; Broekhuijsen, A.G.M.; Meinen, E.; Nijs, E.M.F.M.; Raaphorst, M.G.M. Quantification of the growth response to light quantity of greenhouse grown crops. Acta Hortic. 2006, 711, 97–104. [Google Scholar] [CrossRef]
- Hirose, T. Development of the Monsi–Saeki theory on canopy structure and function. Ann. Bot. 2005, 95, 483–494. [Google Scholar] [CrossRef] [PubMed]
- Ménard, C.; Dorais, M.; Hovi, T.; Gosselin, A. Developmental and physiological responses of tomato and cucumber to additional blue light. Acta Hortic. 2006, 711, 291–296. [Google Scholar] [CrossRef]
- Särkkä, L.E.; Jokinen, K.; Ottosen, C.O.; Kaukoranta, T. Effects of HPS and LED lighting on cucumber leaf photosynthesis, light quality penetration and temperature in the canopy, plant morphology and yield. AFSci 2017, 26, 102–110. [Google Scholar] [CrossRef]
- Gajc-Wolska, J.; Kowalczyk, K.; Metera, A.; Mazur, K.; Bujalski, D.; Hemka, L. Effect of supplementary lighting on selected physiological parameters and yielding of tomato plants. Folia Hort. 2013, 25, 153–159. [Google Scholar] [CrossRef]
- Gómez, C.; Mitchell, C.A. In search of an optimized supplemental lighting spectrum for greenhouse tomato production with intracanopy lighting. Acta Hortic. 2016, 1134, 57–62. [Google Scholar] [CrossRef]
- XiaoYing, L.; ShiRong, G.; ZhiGang, X.; XueLei, J.; Tezuka, T. Regulation of chloroplast ultrastructure, cross-section anatomy of leaves, and morphology of stomata of cherry tomato by different light irradiations of light-emitting diodes. HortScience 2011, 46, 217–221. [Google Scholar] [CrossRef]
- Pettersen, R.I.; Torre, S.; Gislerød, H.R. Effects of intracanopy lighting on photosynthetic characteristics in cucumber. Sci. Hortic. 2010, 125, 77–81. [Google Scholar] [CrossRef]
- Kotiranta, S. The Effect of Light Quality on Tomato (Solanum lycopersicum L. cv ‘Efialto’) Growth and DroughtTolerance. Master’s Thesis, University of Helsinki, Helsinki, Finland, November 2014. [Google Scholar]
- Mawphlang, O.I.; Kharshiing, E.V. Photoreceptor mediated plant growth responses: Implications for photoreceptor engineering toward improved performance in crops. Front. Plant Sci. 2017, 8, 1181. [Google Scholar] [CrossRef]
- Gupta, S.K.; Sharma, S.; Santisree, P.; Kilambi, H.V.; Appenroth, K.; Sreelakshmi, Y.; Sharma, R. Complex and shifting interactions of phytochromes regulate fruit development in tomato. Plant. Cell Environ. 2014, 37, 1688–1702. [Google Scholar] [CrossRef]
- Gunnlaugsson, E.; Adalsteinsson, S. Interlight and plant density in year-round production of tomato at northern latitudes. Acta Hortic. 2006, 711, 71–76. [Google Scholar] [CrossRef]
- Heuvelink, E.; Bakker, M.J.; Hogendonk, L.; Janse, J.; Kaarsemaker, R.; Maaswinkel, R. Horticultural lighting in the Netherlands: New developments. Acta Hortic. 2006, 711, 25–34. [Google Scholar] [CrossRef]
- Trouwborst, G.; Oosterkamp, J.; Hogewoning, S.W.; Harbinson, J.; Ieperen, W.V. The responses of light interception, photosynthesis and fruit yield of cucumber to LED-lighting within the canopy. Physiol. Plant. 2010, 138, 289–300. [Google Scholar] [CrossRef]
- Gómez, C.; Mitchell, C.A. Physiological and Productivity Responses of High-wire Tomato as Affected by Supplemental Light Source and Distribution within the Canopy. J. Amer. Soc. Hort. Sci. 2016, 141, 196–208. [Google Scholar] [CrossRef]
- Hetherington, S.; Smillie, R.; Davies, W. Photosynthetic activities of vegetative and fruiting tissues of tomato. J. Exp. Bot. 1998, 49, 1173–1181. [Google Scholar] [CrossRef]
- Carrara, S.; Pardossi, A.; Soldatini, G.F.; Tognoni, F.; Guidi, L. Photosynthetic activity of ripening tomato fruit. Photosynthetica 2001, 39, 75–78. [Google Scholar] [CrossRef]
- Matas, A.J.; Yeats, T.H.; Buda, G.J.; Zheng, Y.; Chatterjee, S.; Tohge, T.; Ponnala, L.; Adato, A.; Aharoni, A.; Stark, R.; et al. Tissue- and cell-type specific transcriptome profiling of expanding tomato fruit provides insights into metabolic and regulatory specialization and cuticle formation. Plant Cell 2011, 23, 3893–3910. [Google Scholar] [CrossRef] [PubMed]
- Dzakovich, M.P.; Gómez, C.; Mitchell, C.A. Tomatoes grown with light-emitting diodes or high-pressure sodium supplemental lights have similar fruit-quality attributes. HortScience 2015, 50, 1498–1502. [Google Scholar] [CrossRef]
- Helyes, L.; Pék, Z.; Lugasi, A. Tomato fruit quality and content depend on stage of maturity. HortScience 2006, 41, 1400–1401. [Google Scholar] [CrossRef]

| Main Component | Unit | Spring Season | Summer Season |
|---|---|---|---|
| N-NO3− | mM | 21.1 | 21.1 |
| N-NH4+ | mM | 2.04 | 2.04 |
| P-H2PO4− | mM | 1.56 | 2.04 |
| SO4 | mM | 3.15 | 3.15 |
| H3O+ | mM | 8.34 | 8.34 |
| K | mM | 10.8 | 11.2 |
| Ca | mM | 8.05 | 8.05 |
| Mg | mM | 3.40 | 3.40 |
| Na | mM | 3.00 | 3.00 |
| Cl | mM | 1.00 | 1.00 |
| Fe | µM | 27.0 | 15.0 |
| Mn | µM | 12.0 | 12.0 |
| Zn | µM | 6.0 | 6.0 |
| B | µM | 20.0 | 20.0 |
| Mo | µM | 1.00 | 1.00 |
| LED | Control | |
|---|---|---|
| Leaf chlorophyll content (N-tester value) | 505.2 | 437.4 |
| * | ||
| Stomatal conductance (mmol m−2 s−1) | 367.1 | 126.8 |
| *** | ||
| Leaf temperature (°C) | 18.0 | 16.6 |
| *** | ||
| Spring | Summer | |||
|---|---|---|---|---|
| LED | Control | LED | Control | |
| Yield (kg plant−1) | 5.7 | 4.6 | 9.9 | 8.8 |
| * | * | |||
| Average fruit weight (g fruit−1) | 126.4 | 108.5 | 144.4 | 125.8 |
| ** | *** | |||
| Fruit number (n plant−1) | 45.0 | 42.2 | 56.3 | 57.1 |
| ns | ns | |||
| LED | Control | |
|---|---|---|
| Soluble solids content (°Brix) | 3.7 | 3.9 |
| ns | ||
| CIE Lab color space analysis | ||
| L | 41.3 | 42.4 |
| ns | ||
| a | 18.7 | 17.7 |
| ns | ||
| b | 21.6 | 21.7 |
| ns | ||
| a:b ratio | 0.87 | 0.81 |
| ns | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paucek, I.; Pennisi, G.; Pistillo, A.; Appolloni, E.; Crepaldi, A.; Calegari, B.; Spinelli, F.; Cellini, A.; Gabarrell, X.; Orsini, F.; et al. Supplementary LED Interlighting Improves Yield and Precocity of Greenhouse Tomatoes in the Mediterranean. Agronomy 2020, 10, 1002. https://doi.org/10.3390/agronomy10071002
Paucek I, Pennisi G, Pistillo A, Appolloni E, Crepaldi A, Calegari B, Spinelli F, Cellini A, Gabarrell X, Orsini F, et al. Supplementary LED Interlighting Improves Yield and Precocity of Greenhouse Tomatoes in the Mediterranean. Agronomy. 2020; 10(7):1002. https://doi.org/10.3390/agronomy10071002
Chicago/Turabian StylePaucek, Ivan, Giuseppina Pennisi, Alessandro Pistillo, Elisa Appolloni, Andrea Crepaldi, Barbara Calegari, Francesco Spinelli, Antonio Cellini, Xavier Gabarrell, Francesco Orsini, and et al. 2020. "Supplementary LED Interlighting Improves Yield and Precocity of Greenhouse Tomatoes in the Mediterranean" Agronomy 10, no. 7: 1002. https://doi.org/10.3390/agronomy10071002
APA StylePaucek, I., Pennisi, G., Pistillo, A., Appolloni, E., Crepaldi, A., Calegari, B., Spinelli, F., Cellini, A., Gabarrell, X., Orsini, F., & Gianquinto, G. (2020). Supplementary LED Interlighting Improves Yield and Precocity of Greenhouse Tomatoes in the Mediterranean. Agronomy, 10(7), 1002. https://doi.org/10.3390/agronomy10071002








