Evaluation of the Effects of the Application of Glauconitic Fertilizer on Oat Development: A Two-Year Field-Based Investigation
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples and Duration of Experiment
2.2. Plant Growth Tests
3. Results
3.1. Changes in Soil Physico–Chemical Properties
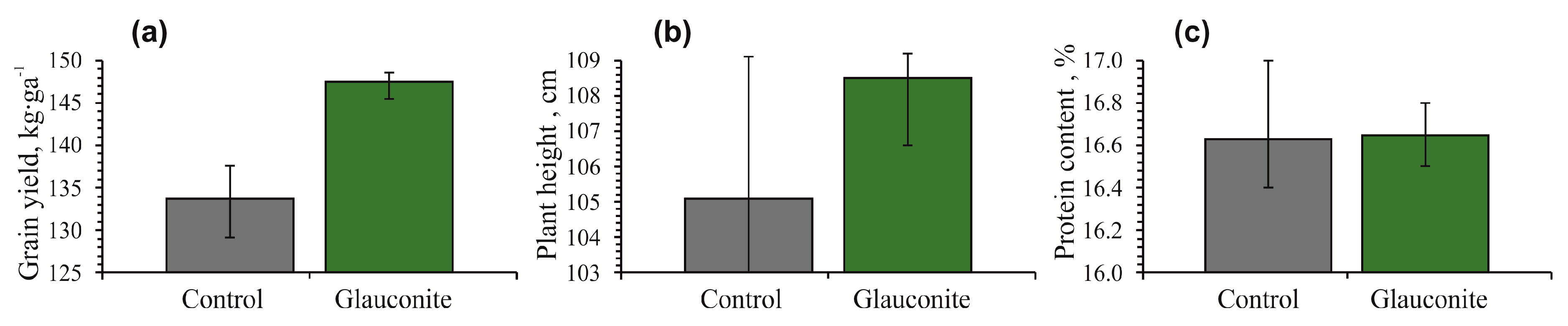
3.2. Changes in Plant Growth
4. Discussion
4.1. Improvement of Soil Characteristics with Glauconitic Fertilizers
4.2. Improvement of Soil Fertility
4.3. Advantages of Glauconitic Fertilizer
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Matson, P.A.; Parton, W.J.; Power, A.G.; Swift, M.J. Agricultural intensification and ecosystem properties. Science 1997, 277, 504–509. [Google Scholar] [CrossRef] [PubMed]
- Tilman, D.; Cassman, K.G.; Matson, P.A.; Naylor, R.; Polasky, S. Agricultural sustainability and intensive production practices. Nature 2002, 418, 671–677. [Google Scholar] [CrossRef] [PubMed]
- Zörb, C.; Senbayram, M.; Peiter, E. Potassium in agriculture – Status and perspectives. J. Plant Physiol. 2014, 171, 656–669. [Google Scholar] [CrossRef] [PubMed]
- Erisman, J.W.; Sutton, M.A.; Galloway, J.; Klimont, Z.; Winiwarter, W. How a century of ammonia synthesis changed the world. Nat. Geosci. 2008, 1, 636–639. [Google Scholar] [CrossRef]
- Rütting, T.; Aronsson, H.; Delin, S. Nutrient Cycling in Agroecosystems; Springer: Berlin/Heidelberg, Germany, 2018. [Google Scholar]
- Camargo, J.A.; Alonso, Á. Ecological and toxicological effects of inorganic nitrogen pollution in aquatic ecosystems: A global assessment. Environ. Int. 2006, 32, 831–849. [Google Scholar] [CrossRef]
- Conrad, R. Soil microorganisms as controllers of atmospheric trace gases (H2, CO, CH4, OCS, N2O, and NO). Microbiol. Rev. 1996, 60, 609–640. [Google Scholar] [CrossRef]
- Basak, B.B. Waste Mica as Alternative Source of Plant-Available Potassium: Evaluation of Agronomic Potential Through Chemical and Biological Methods. Nat. Resour. Res. 2018, 1–13. [Google Scholar] [CrossRef]
- Biswas, D.R.; Basak, B.B. Mobilization of potassium from waste mica by potassium-solubilizing bacteria (Bacillus mucilaginosus) as influenced by temperature and incubation period under in vitro laboratory conditions. Agrochimica 2014, 58, 309–320. [Google Scholar] [CrossRef]
- Feigenbaum, S.; Edelstein, R.; Shainberg, I. Release Rate of Potassium and Structural Cations from Micas to Ion Exchangers in Dilute Solutions. Soil Sci. Society Am. J. 1981, 45, 501. [Google Scholar] [CrossRef]
- Shirale, A.O.; Meena, B.P.; Gurav, P.P.; Srivastava, S.; Biswas, A.K.; Thakur, J.K.; Somasundaram, J.; Patra, A.K.; Rao, A.S. Prospects and challenges in utilization of indigenous rocks and minerals as source of potassium in farming. J. Plant Nutr. 2019, 42, 2682–2701. [Google Scholar] [CrossRef]
- Ciceri, D.; Allanore, A. Nutrient release from K-feldspar ore altered in hydrothermal conditions. Chem. Papers 2019, 1–10. [Google Scholar] [CrossRef]
- Castro, L.; Tourn, S. Direct Application of Phosphate Rocks and Glauconite as Alternative Sources of Fertilizer in Argentina. Explor. Min. Geol. 2003, 12, 71–78. [Google Scholar] [CrossRef]
- Shekhar, S.; Mishra, D.; Agrawal, A.; Sahu, K.K. Physical and chemical characterization and recovery of potash fertilizer from glauconitic clay for agricultural application. Appl. Clay Sci. 2017, 143, 50–56. [Google Scholar] [CrossRef]
- Rudmin, M.A.; Mazurov, A.K.; Makarov, B.I.; Galikhanov, A.V.; Stebletsov, M.D.; Chepala, K.K. On possibility of using Glauconite from rocks of Bakchar deposit (Western Siberia) in agriculture. Bull. Tomsk Polytech. Univ. Geo Assets Eng. 2016, 327, 6–16. [Google Scholar]
- Rudmin, M.; Banerjee, S.; Mazurov, A.; Makarov, B.; Martemyanov, D. Economic potential of glauconitic rocks in Bakchar deposit (S-E Western Siberia) for alternate potash fertilizer. Appl. Clay Sci. 2017, 150, 225–233. [Google Scholar] [CrossRef]
- Franzosi, C.; Castro, L.N.; Celeda, A.M. Technical Evaluation of Glauconies as Alternative Potassium Fertilizer from the Salamanca Formation, Patagonia, Southwest Argentina. Nat. Resour. Res. 2014, 23, 311–320. [Google Scholar] [CrossRef]
- Santos, W.O.; Mattiello, E.M.; da Costa, L.M.; Abrahão, W.A.P.; de Novais, R.F.; Cantarutti, R.B. Thermal and chemical solubilization of verdete for use as potassium fertilizer. Int. J. Miner. Process. 2015, 140, 72–78. [Google Scholar] [CrossRef]
- Shekhar, S.; Sinha, S.; Mishra, D.; Agrawal, A.; Sahu, K.K. A sustainable process for recovery of potash fertilizer from glauconite through simultaneous production of pigment grade red oxide. Sustain. Mater. Technol. 2019, e00129. [Google Scholar] [CrossRef]
- Santos, W.O.; Mattiello, E.M.; Vergutz, L.; Costa, R.F. Production and evaluation of potassium fertilizers from silicate rock. J. Plant Nutr. Soil Sci. 2016, 179, 547–556. [Google Scholar] [CrossRef]
- Shekhar, S.; Mishra, D.; Agrawal, A.; Sahu, K.K. Physico-chemical treatment of glauconitic sandstone to recover potash and magnetite. J. Clean. Prod. 2017, 147, 681–693. [Google Scholar] [CrossRef]
- Karimi, E.; Abdolzadeh, A.; Sadeghipour, H.R.; Aminei, A. The potential of glauconitic sandstone as a potassium fertilizer for olive plants. Arch. Agron. Soil Sci. 2012, 58, 983–993. [Google Scholar] [CrossRef]
- Merchant, R.J. Glauconite-the future potash for fertilisers in New Zealand. AusIMM Bull. 2012, 1, 78–81. [Google Scholar]
- Rudmin, M.; Banerjee, S.; Makarov, B.; Mazurov, A.; Ruban, A.; Oskina, Y.; Tolkachev, O.; Buyakov, A.; Shaldybin, M. An investigation of plant growth by the addition of glauconitic fertilizer. Appl. Clay Sci. 2019, 180, 1–8. [Google Scholar] [CrossRef]
- Guggenheim, S.; Adams, J.M.; Bain, D.C.; Bergaya, F.; Brigatti, M.F.; Drits, V.A.; Formoso, M.L.L.; Galan, E.; Kogure, T.; Stanjek, H. Summary of recommendations of nomenclature committees relevant to clay mineralogy: Report of the Association International pour l’Etude des Argiles (AIPEA) Nomenclature Committee for 2006 (Clays and Clay Minerals). Clays Clay Miner. 2007, 55, 761–772. [Google Scholar] [CrossRef]
- Odin, G.S.; Matter, A. De glauconiarum origine. Sedimentology 1981, 28, 611–641. [Google Scholar] [CrossRef]
- Meunier, A.; El Albani, A. The glauconite-Fe-illite-Fe-smectite problem: A critical review. Terra Nova 2007, 19, 95–104. [Google Scholar] [CrossRef]
- Drits, V.A. Isomorphous Cation Distribution in Celadonites, Glauconites and Fe-illites Determined by Infrared, Mössbauer and EXAFS Spectroscopies. Clay Miner. 1997, 32, 153–179. [Google Scholar] [CrossRef]
- Bailey, S.W. Summary of recommendations of AIPEA nomenclature committee on clay minerals. Am. Mineral. 1980, 65, 1–7. [Google Scholar] [CrossRef]
- Amorosi, A.; Sammartino, I.; Tateo, F. Evolution patterns of glaucony maturity: A mineralogical and geochemical approach. Deep Sea Res. Part II Top. Stud. Oceanogr. 2007, 54, 1364–1374. [Google Scholar] [CrossRef]
- Baldermann, A.; Warr, L.N.; Letofsky-Papst, I.; Mavromatis, V. Substantial iron sequestration during green-clay authigenesis in modern deep-sea sediments. Nat. Geosci. 2015, 8, 885–889. [Google Scholar] [CrossRef]
- Eder, V.G.; Marti’-Algarra, A.; Sanches-Navas, A.; Zanin, Y.N.; Zamiralova, A.G.; Lebedev, Y.N. Depositional controls on glaucony texture and composition, Upper Jurassic, West Siberian Basin. Sedimentology 2007, 54, 1365–1387. [Google Scholar] [CrossRef]
- Hegab, O.A.; Abd El-Wahed, A.G. Origin of the glauconite from the Middle Eocene, Qarara Formation, Egypt. J. Afr. Earth Sci. 2016, 123, 21–28. [Google Scholar] [CrossRef]
- Ivanovskaya, T.A. Glauconitites in Terrigenous Rocks of the Khaipakh Formation (Middle Riphean, Olenek Uplift). Lithol. Miner. Resour. 2009, 44, 348–366. [Google Scholar] [CrossRef]
- Baldermann, A.; Dietzel, M.; Mavromatis, V.; Mittermayr, F.; Warr, L.N.; Wemmer, K. The role of Fe on the formation and diagenesis of interstratified glauconite-smectite and illite-smectite: A case study of Upper Cretaceous shallow-water carbonates. Chem. Geol. 2017, 453, 21–34. [Google Scholar] [CrossRef]
- Banerjee, S.; Bansal, U.; Pande, K.; Meena, S.S. Compositional variability of glauconites within the Upper Cretaceous Karai Shale Formation, Cauvery Basin, India: Implications for evaluation of stratigraphic condensation. Sediment. Geol. 2016, 331, 12–29. [Google Scholar] [CrossRef]
- Banerjee, S.; Bansal, U.; Vilas Thorat, A. A review on palaeogeographic implications and temporal variation in glaucony composition. J. Palaeogeogr. 2016, 5, 43–71. [Google Scholar] [CrossRef]
- Banerjee, S.; Chattoraj, S.L.; Saraswati, P.K.; Dasgupta, S.; Sarkar, U.; Bumby, A. The origin and maturation of lagoonal glauconites: A case study from the Oligocene Maniyara Fort Formation, western Kutch, India. Geol. J. 2012, 47, 357–371. [Google Scholar] [CrossRef]
- Banerjee, S.; Chattoraj, S.L.; Saraswati, P.K.; Dasgupta, S.; Sarkar, U. Substrate control on formation and maturation of glauconites in the Middle Eocene Harudi Formation, western Kutch, India. Mar. Pet. Geol. 2012, 30, 144–160. [Google Scholar] [CrossRef]
- Banerjee, S.; Mondal, S.; Chakraborty, P.P.; Meena, S.S. Distinctive compositional characteristics and evolutionary trend of Precambrian glaucony: Example from Bhalukona Formation, Chhattisgarh basin, India. Precambrian Res. 2015, 271, 33–48. [Google Scholar] [CrossRef]
- Bansal, U.; Banerjee, S.; Pande, K.; Arora, A.; Meena, S.S. The distinctive compositional evolution of glauconite in the Cretaceous Ukra Hill Member (Kutch basin, India) and its implications. Mar. Pet. Geol. 2017, 82, 97–117. [Google Scholar] [CrossRef]
- Bansal, U.; Pande, K.; Banerjee, S.; Nagendra, R.; Jagadeesan, K.C. The timing of oceanic anoxic events in the Cretaceous succession of Cauvery Basin: Constraints from 40 Ar/39 Ar ages of glauconite in the Karai Shale Formation. Geol. J. 2019, 54, 308–315. [Google Scholar] [CrossRef]
- Yapparov, A.K.; Bikkinina, L.M.-K.; Yapparov, I.A.; Aliev, S.A.; Ezhkova, A.M.; Ezhkov, V.O.; Gazizov, R.R. Changes in the properties and productivity of leached chernozem and gray forest soil under the impact of ameliorants. Eurasian Soil Sci. 2015, 48, 1149–1158. [Google Scholar] [CrossRef]
- Mank, V.V.; Melnyk, L.N. Use of clay minerals for adsorptive clearing of aqueous-alcoholic solutions. Acta Geodyn. Geomater. 2005, 2, 113–117. [Google Scholar]
- Singh, S.; Bhadauria, R.; Tomar, R. Sorption of Cd2+, Hg2+ and Pb2+by synthetic analogue of the mica mineral glauconitic. Int. J. Appl. Chem. 2010, 6, 287–295. [Google Scholar]
- Voronina, A.V.; Blinova, M.O.; Semenishchev, V.S.; Gupta, D.K. Returning land contaminated as a result of radiation accidents to farming use. J. Environ. Radioact. 2015, 144, 103–112. [Google Scholar] [CrossRef]
- Ali, O.; Osman, H.H.; Sayed, S.A.; Shalabi, M.E.H. The removal of uranium and thorium from their aqueous solutions via glauconite. Desalin. Water Treat. 2015, 53, 760–767. [Google Scholar] [CrossRef]
- Yadav, V.; Sharma, T.; Saxena, V. Dissolution kinetics of potassium from glauconitic sandstone in acid lixiviant. Int. J. Miner. Process. 2000, 60, 15–36. [Google Scholar] [CrossRef]
- Yadav, V.P.; Sharma, T. Leaching of Glauconitic sandstone in acid lixiviants. Miner. Eng. 1992, 5, 715–720. [Google Scholar] [CrossRef]
- Rao, S.C.; Rao, S.A. Characterization of indigenous glauconitic sandstone for its potassium- supplying potential by chemical, biological, and electroultrafiltration methods. Commun. Soil Sci. Plant Anal. 1999, 30, 1105–1117. [Google Scholar] [CrossRef]
- Mazumder, A.; Sharma, T.; Rao, T. Extraction of potassium from glauconitic sandstone by the roast-leach method. Int. J. Miner. Process. 1993, 38, 111–123. [Google Scholar] [CrossRef]
- Rudmin, M.; Oskina, Y.; Banerjee, S.; Mazurov, A.; Soktoev, B.; Shaldybin, M. Roasting-leaching experiments on glauconitic rocks of Bakchar ironstone deposit (Western Siberia) for evaluation their fertilizer potential. Appl. Clay Sci. 2018, 162, 121–128. [Google Scholar] [CrossRef]
- Oze, C.; Smaill, J.B.; Reid, C.M.; Palin, M. Potassium and Metal Release Related to Glaucony Dissolution in Soils. Soil Syst. 2019, 3, 70. [Google Scholar] [CrossRef]
- Tanvar, H.; Dhawan, N. Kinetic and thermodynamic study of potassium recovery from silicate rocks. Miner. Process. Extr. Metall. 2019, 1–13. [Google Scholar] [CrossRef]
- Schimicoscki, R.S.; Oliveira, K.D.; Ávila-Neto, C.N. Potassium recovery from a Brazilian glauconitic siltstone via reaction with sulfuric acid in hydrothermal conditions. Hydrometallurgy 2020, 191, 105251. [Google Scholar] [CrossRef]
- Rudmin, M.; Abdullayev, E.; Ruban, A.; Buyakov, A.; Soktoev, B. Mechanochemical Preparation of Slow Release Fertilizer Based on Glauconite–Urea Complexes. Minerals 2019, 9, 507. [Google Scholar] [CrossRef]
- Torqueti, S.T.S.; Boldrin, K.V.F.; do Nascimento, Â.M.P.; Paiva, P.D.O.; Furtini Neto, A.E.; Luz, I.C.A. Alternative potassium source for the cultivation of ornamental sunflower|Fonte alternativa de potássio no cultivo do girassol ornamental. Cienc. Agrotecnol. 2016, 40, 257–264. [Google Scholar] [CrossRef]
- Dias, K.G.D.L.; Guimarães, P.T.G.; do Carmo, D.L.; Reis, T.H.P.; Lacerda, J.J.D.J. Alternative sources of potassium in coffee plants for better soil fertility, productivity, and beverage quality. Pesquisa Agropecuária Bras. 2018, 53, 1355–1362. [Google Scholar] [CrossRef]
- Rudmin, M.; Mazurov, A.; Banerjee, S. Origin of ooidal ironstones in relation to warming events: Cretaceous-Eocene Bakchar deposit, south-east Western Siberia. Mar. Pet. Geol. 2019, 100, 309–325. [Google Scholar] [CrossRef]
- Rudmin, M.; Banerjee, S.; Abdullayev, E.; Ruban, A.; Filimonenko, E.; Lyapina, E.; Kashapov, R.; Mazurov, A. Ooidal ironstones in the Meso-Cenozoic sequences in western Siberia: Assessment of formation processes and relationship with regional and global earth processes. J. Palaeogeogr. 2020, 9, 1–21. [Google Scholar] [CrossRef]
- Rudmin, M.; Banerjee, S.; Mazurov, A. Compositional variation of glauconites in Upper Cretaceous-Paleogene sedimentary iron-ore deposits in South-eastern Western Siberia. Sediment. Geol. 2017, 355, 20–30. [Google Scholar] [CrossRef]
- Rudmin, M.A.; Mazurov, A.K.; Reva, I.V.; Stebletsov, M.D. Prospects of integrated development of Bakchar iron deposit (Western Siberia, Russia). Bull. Tomsk Polytech. Univ. Geo Assets Eng. 2018, 329, 85–94. [Google Scholar]
- Sokolov, A.V. (Ed.) Agrochemical Methods of Soil Study; Nauka: Moscow, Russia, 1975. (In Russian) [Google Scholar]
- Aleksandrova, L.N.; Naidenova, O.A. Laboratory Practice in Soil Science; Kolos: Leningrad, Russia, 1976. (In Russian) [Google Scholar]
- Chang, S.S.; Shau, Y.H.; Wang, M.K.; Ku, C.T.; Chiang, P.N. Mineralogy and occurrence of glauconite in central Taiwan. Appl. Clay Sci. 2008, 42, 74–80. [Google Scholar] [CrossRef]
- Sorokin, I.B.; Gaag, A.V.; Chudinova, I.V.; Sirotina, E.A. Problems of sour soil fertility increasing in Tomsk region. Bull. NSAU 2018, 48, 31–37. [Google Scholar] [CrossRef]
- Basak, B.B. Phosphorus Supplying Capacity of Value Added Compost Prepared from Low-Grade Indian Rock Phosphates and Crop Residue. Waste Biomass Valorization 2017, 8, 2653–2662. [Google Scholar] [CrossRef]
- Shen, J.; Yuan, L.; Zhang, J.; Li, H.; Bai, Z.; Chen, X.; Zhang, W.; Zhang, F. Phosphorus dynamics: From soil to plant. Plant Physiol. 2011, 156, 997–1005. [Google Scholar] [CrossRef]
- Kumari, A.; Kapoor, K.K.; Kundu, B.S.; Mehta, R.K. Identification of organic acids produced during rice straw decomposition and their role in rock phosphate solubilization. Plant Soil Environ. 2008, 54, 72–77. [Google Scholar] [CrossRef]
- Singh, C.P.; Amberger, A. Organic acids and phosphorus solubilization in straw composted with rock phosphate. Bioresour. Technol. 1998, 63, 13–16. [Google Scholar] [CrossRef]
- Gillman, G.P.; Burkett, D.C.; Coventry, R.J. Amending highly weathered soils with finely ground basalt rock. Appl. Geochem. 2002, 17, 987–1001. [Google Scholar] [CrossRef]
- Manning, D.A.C. Mineral sources of potassium for plant nutrition. A review. Agron. Sustain. Dev. 2010, 30, 281–294. [Google Scholar] [CrossRef]
- Griffioen, J. Potassium adsorption ratios as an indicator for the fate of agricultural potassium in groundwater. J. Hydrol. 2001, 254, 244–254. [Google Scholar] [CrossRef]
- Ciceri, D.; de Oliveira, M.; Stokes, R.M.; Skorina, T.; Allanore, A. Characterization of potassium agrominerals: Correlations between petrographic features, comminution and leaching of ultrapotassic syenites. Miner. Eng. 2017, 102, 42–57. [Google Scholar] [CrossRef]
- Manning, D.A.C.; Baptista, J.; Sanchez Limon, M.; Brandt, K. Testing the ability of plants to access potassium from framework silicate minerals. Sci. Total Environ. 2017, 574, 476–481. [Google Scholar] [CrossRef] [PubMed]
- Pessoa, R.S.; Silva, C.A.; Moretti, B.S.; Furtini Neto, A.E.; Inda, A.V.; Curi, N. Solubilization of potassium from alternative rocks by humic and citric acids and coffee husk. Ciência Agrotecnol. 2015, 39, 553–564. [Google Scholar] [CrossRef]
- Mohammed, S.M.O.; Brandt, K.; Gray, N.D.; White, M.L.; Manning, D.A.C. Comparison of silicate minerals as sources of potassium for plant nutrition in sandy soil. Eur. J. Soil Sci. 2014, 65, 653–662. [Google Scholar] [CrossRef]
- Hinsinger, P.; Bolland, M.D.A.; Gilkes, R.J. Silicate rock powder: Effect on selected chemical properties of a range of soils from Western Australia and on plant growth as assessed in a glasshouse experiment. Fertil. Res. 1995, 45, 69–79. [Google Scholar] [CrossRef]
- Fu, J.; Wang, C.; Chen, X.; Huang, Z.; Chen, D. Classification research and types of slow controlled release fertilizers (SRFs) use-a review. Commun. Soil Sci. Plant. Anal. 2018, 49, 2219–2230. [Google Scholar] [CrossRef]
- Feigenbaum, S.; Shainberg, I. Dissolution of Illite-A Possible Mechanism of Potassium Release. Proc. Soil Sci Soc. Am. 1975, 39, 985–990. [Google Scholar] [CrossRef]
- Oertli, J.J. Controlled-release fertilizers. Fertil. Res. 1980, 1, 103–123. [Google Scholar] [CrossRef]
- Baldanza, V.A.R.; Souza, F.G.; Filho, S.T.; Franco, H.A.; Oliveira, G.E.; Caetano, R.M.J.; Hernandez, J.A.R.; Ferreira Leite, S.G.; Furtado Sousa, A.M.; Nazareth Silva, A.L. Controlled-release fertilizer based on poly(butylene succinate)/urea/clay and its effect on lettuce growth. J. Appl. Polym. Sci. 2018, e46858. [Google Scholar] [CrossRef]
- Weian, Z.; Wei, L.; Yue’e, F. Synthesis and properties of a novel hydrogel nanocomposites. Mater. Lett. 2005, 59, 2876–2880. [Google Scholar] [CrossRef]
- Chi, Y.; Zhang, G.; Xiang, Y.; Cai, D.; Wu, Z. Fabrication of reusable temperature-controlled-released fertilizer using a palygorskite-based magnetic nanocomposite. Appl. Clay Sci. 2018, 161, 194–202. [Google Scholar] [CrossRef]
- Trenkel, M.E. Controlled-Release and Stabilized Fertilizers in Agriculture; International Fertilizer Industry Association: Paris, France, 1997. [Google Scholar]

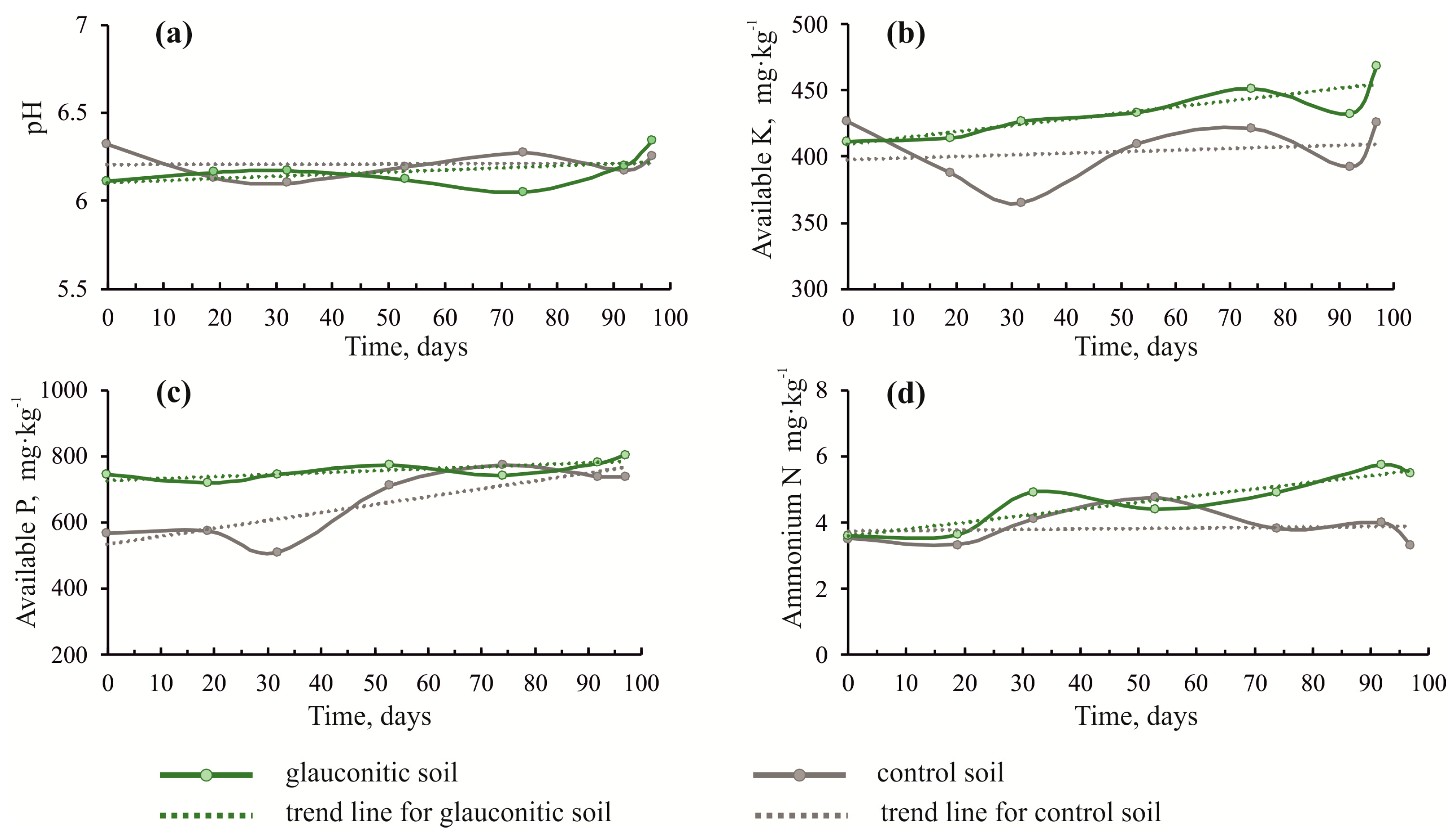
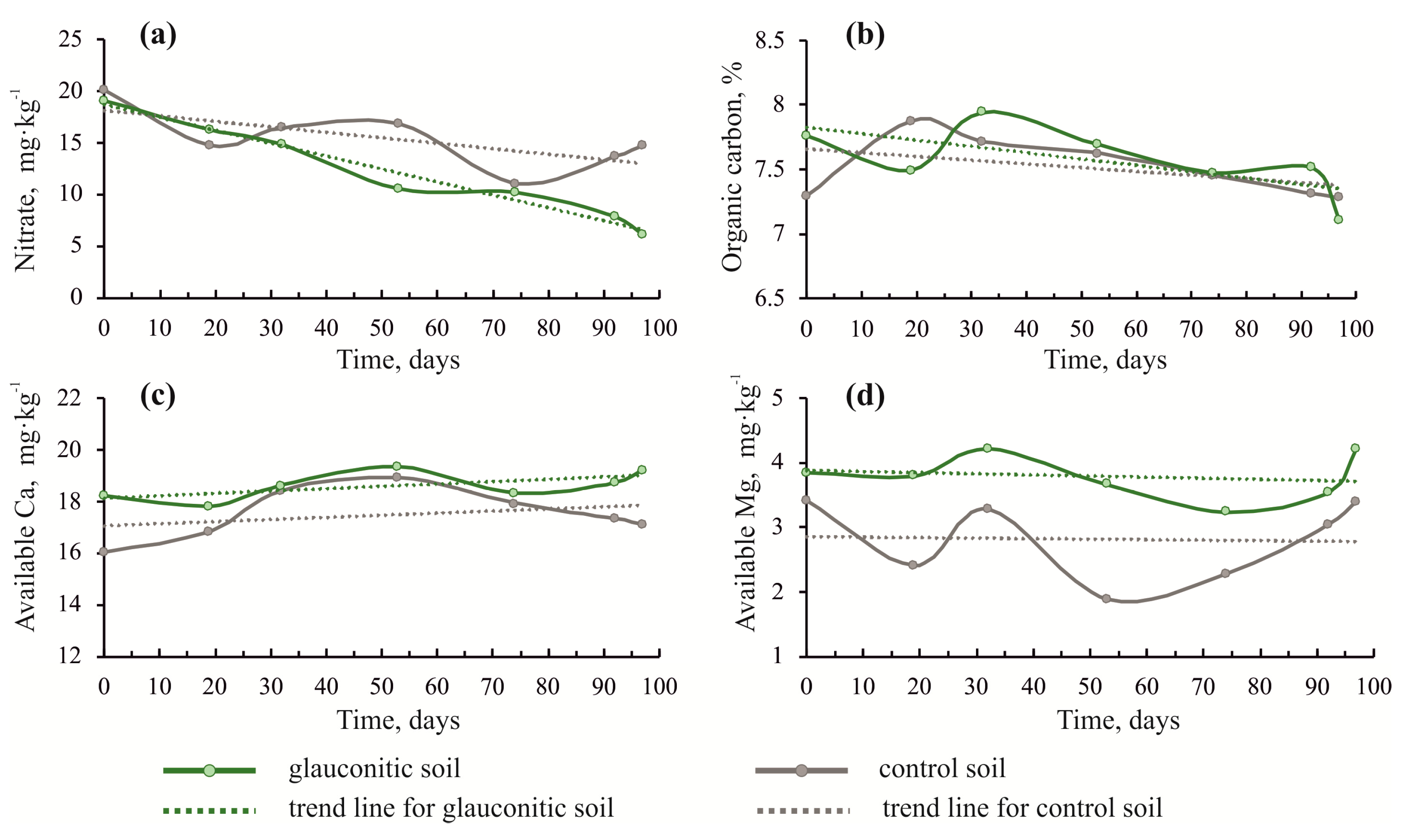
| Soil Sample | Time 2 | pH | Available K (mg∙kg−1) | Available P (mg∙kg−1) | Ammonium N (mg∙kg−1) | Nitrate (mg∙kg−1) | Organic Carbon (%) | Available Ca (mg∙kg−1) | Available Mg (mg∙kg−1) |
|---|---|---|---|---|---|---|---|---|---|
| Method | Potentiometric method | Kirsanov’s method | Kirsanov’s method | Trilonometric method | Kjeldahl’s method | Tyurin’s method | Kirsanov’s method | Kirsanov’s method | |
| Control | 1 | 6.32 ± 0.10 | 426 ± 64 | 565 ± 113 | 3.49 ± 0.52 | 20.1 ± 1.5 | 7.29 ± 0.73 | 16.0 ± 1.1 | 3.41 ± 0.26 |
| 19 | 6.13 ± 0.10 | 387 ± 58 | 573 ± 135 | 3.31 ± 0.5 | 14.7 ± 1.1 | 7.87 ± 0.79 | 16.8 ± 1.3 | 2.40 ± 0.18 | |
| 32 | 6.10 ± 0.10 | 365 ± 55 | 507 ± 101 | 4.09 ± 0.61 | 16.4 ± 1.2 | 7.71 ± 0.77 | 18.4 ± 1.4 | 3.28 ± 0.25 | |
| 53 | 6.19 ± 0.10 | 409 ± 72 | 710 ± 152 | 4.74 ± 0.83 | 16.8 ± 1.3 | 7.62 ± 0.76 | 18.9 ± 1.6 | 1.88 ± 0.09 | |
| 74 | 6.27 ± 0.10 | 421 ± 63 | 772 ± 192 | 3.79 ± 0.49 | 11.0 ± 0.8 | 7.45 ± 0.74 | 17.9 ± 1.3 | 2.27 ± 0.17 | |
| 92 | 6.17 ± 0.10 | 392 ± 59 | 737 ± 123 | 3.99 ± 0.60 | 13.6 ± 1.0 | 7.31 ± 0.73 | 17.3 ± 1.4 | 3.03 ± 0.23 | |
| 97 | 6.25 ± 0.10 | 425 ± 64 | 736 ± 123 | 3.29 ± 0.43 | 14.7 ± 1.1 | 7.28 ± 0.73 | 17.1 ± 1.2 | 3.39 ± 0.27 | |
| Glauconitolite | 1 | 6.11 ± 0.10 | 411 ± 55 | 742 ± 160 | 3.58 ± 0.54 | 19.0 ± 1.4 | 7.76 ± 0.78 | 18.2 ± 1.5 | 3.84 ± 0.30 |
| 19 | 6.16 ± 0.10 | 414 ± 56 | 718 ± 132 | 3.62 ± 0.54 | 16.2 ± 1.2 | 7.49 ± 0.75 | 17.8 ± 1.3 | 3.79 ± 0.28 | |
| 32 | 6.17 ± 0.10 | 426 ± 55 | 743 ± 149 | 4.91 ± 0.87 | 14.8 ± 1.1 | 7.94 ± 0.79 | 18.6 ± 1.4 | 4.21 ± 0.34 | |
| 53 | 6.12 ± 0.10 | 433 ± 55 | 772 ± 142 | 4.39 ± 0.66 | 10.5 ± 0.8 | 7.69 ± 0.72 | 19.3 ± 1.8 | 3.66 ± 0.27 | |
| 74 | 6.05 ± 0.10 | 451 ± 53 | 741 ± 148 | 4.88 ± 0.73 | 10.2 ± 0.8 | 7.47 ± 0.75 | 18.3 ± 1.4 | 3.23 ± 0.23 | |
| 92 | 6.20 ± 0.10 | 432 ± 43 | 778 ± 130 | 5.73 ± 1.01 | 7.8 ± 0.6 | 7.51 ± 0.75 | 18.7 ± 1.4 | 3.53 ± 0.25 | |
| 97 | 6.34 ± 0.10 | 468 ± 48 | 802 ± 126 | 5.47 ± 0.71 | 6.1 ± 0.5 | 7.10 ± 0.71 | 19.2 ± 1.3 | 4.20 ± 0.29 | |
| LSD 3 | 0.09 | 25 | 90 | 0.81 | 4.5 | 0.29 | 0.93 | 0.58 |
| Grain Yield (kg∙ga−1) | Plant Height (cm) | Protein Content (%) | ||||
|---|---|---|---|---|---|---|
| Control | With Glauconite | Control | With Glauconite | Control | With Glauconite | |
| Average | 133.7 | 147.5 | 105.1 | 108.5 | 16.6 | 16.6 |
| minimum | 129.1 | 145.6 | 101.4 | 106.8 | 16.4 | 16.5 |
| maximum | 137.6 | 148.6 | 109.4 | 109.6 | 17.0 | 16.8 |
| LSD 1 | 0.7 | 6.9 | 0.6 | |||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rudmin, M.; Banerjee, S.; Makarov, B. Evaluation of the Effects of the Application of Glauconitic Fertilizer on Oat Development: A Two-Year Field-Based Investigation. Agronomy 2020, 10, 872. https://doi.org/10.3390/agronomy10060872
Rudmin M, Banerjee S, Makarov B. Evaluation of the Effects of the Application of Glauconitic Fertilizer on Oat Development: A Two-Year Field-Based Investigation. Agronomy. 2020; 10(6):872. https://doi.org/10.3390/agronomy10060872
Chicago/Turabian StyleRudmin, Maxim, Santanu Banerjee, and Boris Makarov. 2020. "Evaluation of the Effects of the Application of Glauconitic Fertilizer on Oat Development: A Two-Year Field-Based Investigation" Agronomy 10, no. 6: 872. https://doi.org/10.3390/agronomy10060872
APA StyleRudmin, M., Banerjee, S., & Makarov, B. (2020). Evaluation of the Effects of the Application of Glauconitic Fertilizer on Oat Development: A Two-Year Field-Based Investigation. Agronomy, 10(6), 872. https://doi.org/10.3390/agronomy10060872






