Morphometric Characteristics, Polyphenols and Ascorbic Acid Variation in Brassica oleracea L. Novel Foods: Sprouts, Microgreens and Baby Leaves
Abstract
1. Introduction
2. Materials and Methods
2.1. Plants Materials, Seed Morphological Characteristics and Germination Test
2.2. Sprouts, Microgreens and Baby Leaves Production and Characterization
2.3. Polyphenol Analysis
2.4. Ascorbic Acid Analysis
2.5. Folin–Ciocalteu Index
2.6. Antioxidant Activity
2.7. Statistical Analysis
3. Results
3.1. Germination Process, Seed and Plant Characteristics
3.2. Multifactorial ANOVA
3.3. Total Polyphenol Content
3.4. Polyphenol Analysis
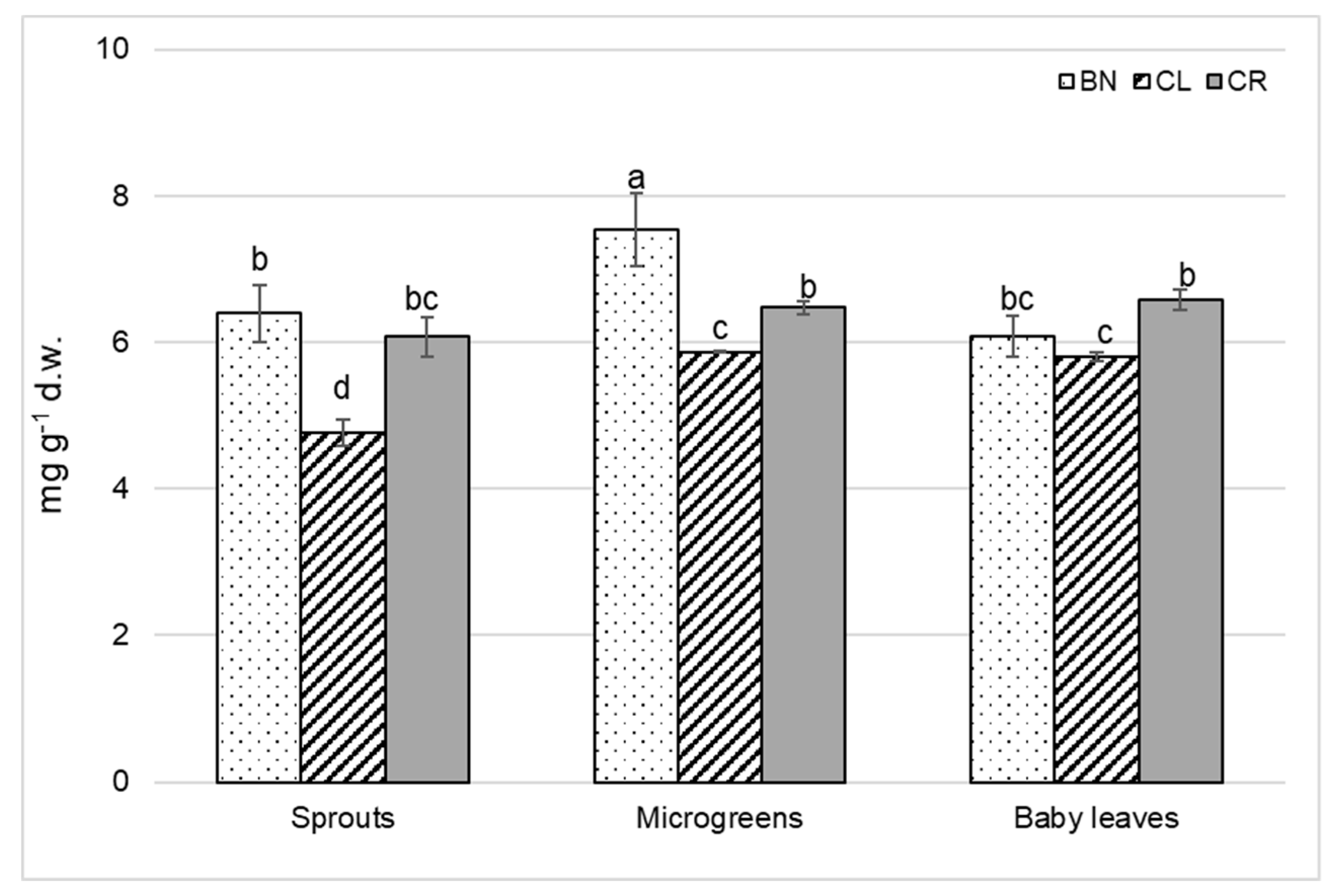
3.5. Ascorbic Acid Content
3.6. Antioxidant Activity
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Podsedek, A.; Sosnowska, D.; Redzynia, M.; Anders, B. Antioxidant capacity and content of Brassica oleracea dietary antioxidants. Int. J. Food Sci. Technol. 2006, 41, 49–58. [Google Scholar] [CrossRef]
- Garcia-Lafuente, A.; Guillamon, E.; Villares, A.; Rostagno, M.A.; Martinez, J.A. Flavonoids as anti-inflammatory agents: Implications in cancer and cardiovascular disease. Inflamm. Res. 2009, 58, 537–552. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Nagata, N.; Tsuguhito, O. Impact of glucoraphanin-mediated activation of Nrf2 on non-alcoholic fatty liver disease with a focus on mitochondrial dysfunction. Int. J. Mol. Sci. 2019, 20, 5920. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Song, X.; Feng, Y.; Liu, N.; Fu, Z.; Wu, J.; Li, T.; Chen, H.; Chen, J.; Chen, C.; et al. Natural ingredients-derivates antioxidants attenuate H2O2-induced oxidative stress and have chondroprotective effects on human osteoarthritic chondrocytes via Keap1/Nrf2 pathways. Free Radic. Biol. Med. 2020. [Google Scholar] [CrossRef]
- Branca, F.; Li, G.; Goyal, S.; Quiros, C.F. Survey of aliphatic glucosinolates in Sicilian wild and cultivated Brassicaceae. Phytochemistry 2002, 59, 717–724. [Google Scholar] [CrossRef]
- Branca, F.; Chiarenza, G.L.; Cavallaro, C.; Gu, Z.; Zhao, Z.; Tribulato, A. Diversity of Sicilian broccoli (Brassica oleracea var. italica) and cauliflower (Brassica oleracea var. botrytis) landraces and their distinctive bio-morphological, antioxidant, and genetic traits. Genet. Resour. Crop Evol. 2018, 65, 485. [Google Scholar] [CrossRef]
- Crozier, A.; Jaganath, I.B.; Clifford, M.N. Phenols, polyphenols and tannins: An overview. In Plant Secondary Metabolites: Occurrence, Structure and Role in the Human Diet; Crozier, A., Clifford, M., Ashihara, H., Eds.; Blackwell: Oxford, UK, 2006; pp. 1–24. [Google Scholar]
- Pereira, D.M.; Valentao, P.; Pereira, J.A.; Andrade, P.B. Phenolics: From chemistry to biology. Molecules 2009, 14, 2202–2211. [Google Scholar] [CrossRef]
- Toscano, S.; Trivellini, A.; Cocetta, G.; Bulgari, R.; Francini, A.; Romano, D.; Ferrante, A. Effect of preharvest abiotic stresses on the accumulation of bioactive compounds in horticultural produce. Front. Plant Sci. 2019, 10, 1212. [Google Scholar] [CrossRef]
- Lo Scalzo, R.; Genna, A.; Branca, F.; Chedin, M.; Chassaigne, H. Anthocyanin composition of cauliflower (Brassica oleracea L. var botrytis) and cabbage (B. oleracea L. var. capitata) and its stability in relation to thermal treatments. Food Chem. 2008, 107, 136–144. [Google Scholar] [CrossRef]
- Cartea, M.E.; Francisco, M.; Soengas, P.; Velasco, P. Phenolic compounds in Brassica vegetables. Molecules 2011, 16, 251–280. [Google Scholar] [CrossRef]
- Schlueter, A.K.; Johnston, C.S. Vitamin C: Overview and update. J. Evid. Based Complementary Altern. Med. 2011, 16, 49–57. [Google Scholar] [CrossRef]
- Miller, E.; Wachowicz, B.; Majsterek, I. Advances in antioxidative therapy of multiple sclerosis. Curr. Med. Chem. 2013, 20, 4720–4730. [Google Scholar] [CrossRef] [PubMed]
- Renna, M.; Di Gioia, F.; Leoni, B.; Santamaria, P. Due Espressioni Dell’agrobiodiversità in Orticoltura: Germogli e Micro Ortaggi. Italus Hortus 2016, 23, 31–44. Available online: https://www.soihs.it/ItalusHortus/Review/Review%2028/03%20Renna.pdf (accessed on 15 January 2020).
- Ebert, A.W. Sprouts, microgreens, and edible flowers: The potential for high value specialty produce in Asia. In SEAVEG 2012 High Value Vegetables in Southeast Asia: Production, Supply and Demand; AVRDC-The World Vegetable Center: Tainan, Taiwan, 2013; pp. 216–227. [Google Scholar]
- Paśko, P.; Bartoń, H.; Zagrodzki, P.; Gorinstein, S.; Fołta, M.; Zachwieja, Z. Anhocyanins, total polyphenols and antioxidant activity in amaranth and quinoa seeds and sprouts during their growth. Food Chem. 2009, 115, 994–998. [Google Scholar] [CrossRef]
- Moreira-Rodríguez, M.; Nair, V.; Benavides, J.; Cisneros-Zevallos, L.; Jacobo-Velázquez, D. UVA, UVB light doses and harvesting time differentially tailor glucosinolate and phenolic profiles in broccoli sprouts. Molecules 2017, 22, 1065. [Google Scholar] [CrossRef] [PubMed]
- Kyriacou, M.C.; Rouphael, Y.; Di Gioia, F.; Kyratzis, A.; Serio, F.; Renna, M.; De Pascale, S.; Santamaria, P. Micro-scale vegetable production and the rise of microgreens. Trends Food Sci. Technol. 2016, 57, 103–115. [Google Scholar] [CrossRef]
- Bulgari, R.; Baldi, A.; Ferrante, A.; Lenzi, A. Yield and quality of basil, Swiss chard, and rocket microgreens grown in a hydroponic system. N. Z. J. Crop Hortic. Sci. 2017, 45, 119–129. [Google Scholar] [CrossRef]
- Xiao, Z.; Lester, G.E.; Luo, Y.; Wang, Q. Assessment of vitamin and carotenoid concentrations of emerging food products: Edible microgreens. J. Agric. Food Chem. 2012, 60, 7644–7651. [Google Scholar] [CrossRef]
- Paradiso, V.M.; Castellino, M.; Renna, M.; Gattullo, C.E.; Calasso, M.; Terzano, R.; Allegretta, I.; Leoni, B.; Caponio, F.; Santamaria, P. Nutritional characterization and shelf-life of packaged microgreens. Food Funct. 2018, 9, 5629. [Google Scholar] [CrossRef]
- Pinto, E.; Almeida, A.A.; Aguiar, A.A.; Ferreira, I.M. Comparison between the mineral profile and nitrate content of microgreens and mature lettuces. J. Food Compos. Anal. 2015, 37, 38–43. [Google Scholar] [CrossRef]
- Liu, J.; Hirami, A.H.; Li, Z.; Wu, C.; McVetty, P.B.E.; Daayf, F.; Li, G. QTL controlling glucosinolate content in seeds of Brassica napus L. Aust. J. Crop Sci. 2016, 10, 152–160. [Google Scholar]
- Weber, C.F. Broccoli microgreens: A mineral-rich crop that can diversify food systems. Front. Nutr. 2017, 4, 7. [Google Scholar] [CrossRef] [PubMed]
- Ying, Q.; Kong, Y.; Jones-Baumgardt, C.; Zheng, Y. Responses of yield and appearance quality of four Brassicaceae microgreens to varied blue light proportion in red and blue light-emitting diodes lighting. Sci. Hortic. 2020, 259, 108857. [Google Scholar] [CrossRef]
- Kyriacou, M.C.; De Pascale, S.; Kyratzis, A.; Rouphael, Y. Microgreens as a component of space life support systems: A cornucopia of functional food. Front. Plant Sci. 2017, 8, 1587. [Google Scholar] [CrossRef]
- Martínez-Sánchez, A.; Luna, M.C.; Selma, M.V.; Tudela, J.A.; Abad, J.; Gil, M.I. Baby-leaf and multi-leaf of green and red lettuces are suitable raw materials for the fresh-cut industry. Postharvest Biol. Technol. 2012, 63, 1–10. [Google Scholar] [CrossRef]
- Koukounaras, A.; Bantis, F.; Karatolos, N.; Melissas, C.; Vezyroglou, A. Influence of pre-harvest factors on postharvest quality of fresh-cut and baby leafy vegetables. Agronomy 2020, 10, 172. [Google Scholar] [CrossRef]
- Francis, G.A.; Gallone, A.; Nychas, G.J.; Sofos, J.N.; Colelli, G.; Amodio, M.L.; Spano, G. Factors affecting quality and safety of fresh-cut produce. Crit. Rev. Food Sci. Nutr. 2012, 52, 595–610. [Google Scholar] [CrossRef]
- Scialabba, A.; Salvini, L.; Faqi, A.S.; Bellani, L.M. Tocopherol, fatty acid and phytosterol content in seeds of nine wild taxa of Sicilian Brassica (Cruciferae). Plant Biosyst. 2010, 144, 626–633. [Google Scholar] [CrossRef]
- Soengas, P.; Cartea, M.E.; Francisco, M.; Sotelo, T.; Velasco, P. New insights into antioxidant activity of Brassica crops. Food Chem. 2012, 134, 725–733. [Google Scholar] [CrossRef]
- Thillmans, J.; Hirsch, P.; Hitsch, W. The reducing property of plant foods and its relation to vitamin C. Z. Lebensm Unters Forsch 1932, 63, 1–21. [Google Scholar]
- Meda, A.; Lamien, C.E.; Romito, M.; Millogo, J.; Nacoulma, O.G. Determination of the total phenolic, flavonoid and proline contents in Burkina Fasan honey, as well as their radical scavenging activity. Food Chem. 2005, 91, 571–577. [Google Scholar] [CrossRef]
- Picchi, V.; Migliori, C.; Scalzo, R.L.; Campanelli, G.; Ferrari, V.; Di Cesare, L.F. Phytochemical content in organic and conventionally grown Italian cauliflower. Food Chem. 2012, 130, 501–509. [Google Scholar] [CrossRef]
- Lo Scalzo, R.L.; Todaro, A.; Rapisarda, P. Methods used to evaluate the peroxyl (ROO•) radical scavenging capacities of four common antioxidants. Eur. Food Res. Technol. 2012, 235, 1141–1148. [Google Scholar] [CrossRef]
- Martinez-Villaluenga, C.; Peñas, E.; Ciska, E.; Piskula, M.K.; Kozlowska, H.; Vidal-Valverde, C.; Frias, J. Time dependence of bioactive compounds and antioxidant capacity during germination of different cultivars of broccoli and radish seeds. Food Chem. 2010, 120, 710–716. [Google Scholar] [CrossRef]
- Ragusa, L.; Picchi, V.; Tribulato, A.; Cavallaro, C.; Lo Scalzo, R.; Branca, F. The effect of the germination temperature on the phytochemical content of broccoli and rocket sprouts. Int. J. Food Sci. Nutr. 2017, 68, 411–420. [Google Scholar] [CrossRef] [PubMed]
- Copeland, L.; Mcdonald, M.B. (Eds.) Principles of Seed Science and Technology, 3rd ed.; Chapman and Hall: New York, NY, USA, 2001. [Google Scholar]
- Cáceres, P.J.; Martínez-Villaluenga, C.; Amigo, L.; Frias, J. Maximising the phytochemical content and antioxidant activity of Ecuadorian brown rice sprouts through optimal germination conditions. Food Chem. 2014, 152, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Balibrea, S.; Moreno, D.A.; García-Viguera, C. Genotypic effects on the phytochemical quality of seeds and sprouts from commercial broccoli cultivars. Food Chem. 2011, 125, 348–354. [Google Scholar] [CrossRef]
- Martínez-Villaluenga, C.; Frías, J.; Gulewicz, P.; Gulewicz, K.; Vidal-Valverde, C. Food safety evaluation of broccoli and radish sprouts. Food Chem. Toxicol. 2008, 46, 1635–1644. [Google Scholar] [CrossRef]
- Yuan, G.; Wang, X.; Guo, R.; Wang, Q. Effect of salt stress on phenolic compounds, glucosinolates, myrosinase and antioxidant activity in radish sprouts. Food Chem. 2010, 121, 1014–1019. [Google Scholar] [CrossRef]
- Galati, G.; O‘Brien, P.J. Potential toxicity of flavonoids and other dietary phenolics: Significance for their chemopreventive and anticancer properties. Free Radic. Biol. Med. 2004, 37, 287–303. [Google Scholar] [CrossRef]
- Duenas, M.; Hernandez, T.; Estrella, I.; Farnandez, D. Germination as process to increase the polyphenol content and antioxidant activity of lupin seeds. Food Chem. 2009, 117, 599–607. [Google Scholar] [CrossRef]
- Vale, A.P.; Santos, J.; Brito, N.V.; Fernandes, D.; Rosa, E.; Oliveira, M.B.P. Evaluating the impact of sprouting conditions on the glucosinolate content of Brassica oleracea sprouts. Phytochemistry 2015, 115, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Basu, T.K.; Ooraikul, B. Studies on germination conditions and antioxidant contents of wheat grain. Int. J. Food Sci. Nutr. 2001, 52, 319–330. [Google Scholar] [CrossRef] [PubMed]
- Koh, E.; Wimalasiri, K.M.S.; Chassy, A.W.; Mitchell, A.E. Content of ascorbic acid, quercetin, kaempferol and total phenolics in commercial broccoli. J. Food Compos. Anal. 2009, 22, 637–643. [Google Scholar] [CrossRef]
- Velasco, P.; Francisco, M.; Moreno, D.A.; Ferreres, F.; García-Viguera, C.; Cartea, M.E. Phytochemical fingerprinting of vegetable Brassica oleracea and Brassica napus by simultaneous identification of glucosinolates and phenolics. Phytochem. Anal. 2011, 22, 144–152. [Google Scholar] [CrossRef]
- Pająk, P.; Socha, R.; Galkowska, D.; Rożnowki, J.; Fortuna, T. Phenolic profile and antioxidant activity in selected seeds and sprouts. Food Chem. 2014, 143, 300–306. [Google Scholar] [CrossRef]
- Gawlik-Dziki, U.; Jeżyna, M.; Świeca, M.; Dziki, D.; Baraniak, B.; Czyż, J. Effect of bioaccessibility of phenolic compounds on in vitro anticancer activity of broccoli sprouts. Food Res. Int. 2012, 49, 469–476. [Google Scholar] [CrossRef]
- Kurilich, A.C.; Tsau, G.J.; Brown, A.; Howard, L.; Klein, B.P.; Jeffery, E.H.; Kushad, M.; Wallig, M.A.; Juvik, J.A. Carotene, tocopherol, and ascorbate contents in subspecies of Brassica oleracea. J. Agric. Food Chem. 1999, 47, 1576–1581. [Google Scholar] [CrossRef]
- Vallejo, F.; Tomas-Barberan, F.A.; Garcia-Viguera, C. Potential bioactive compounds in health promotion from broccoli cultivars grown in Spain. J. Sci. Food Agric. 2002, 82, 1293–1297. [Google Scholar] [CrossRef]
- Halliwell, B.; Gutteridge, J.M.C. (Eds.) Free Radicals in Biology and Medicine; Oxford University Press: Oxford, UK, 1999. [Google Scholar]
- Ebert, A.W.; Wu, T.H.; Yang, R.Y. Amaranth sprouts and microgreens—A homestead vegetable production option to enhance food and nutrition security in the rural-urban continuum. In Proceedings of the Regional Symposium on Sustaining Small-Scale Vegetable Production and Marketing Systems for Food and Nutrition Security (SEAVEG 2014), Bangkok, Thailand, 25–27 February 2014; pp. 25–27. [Google Scholar]
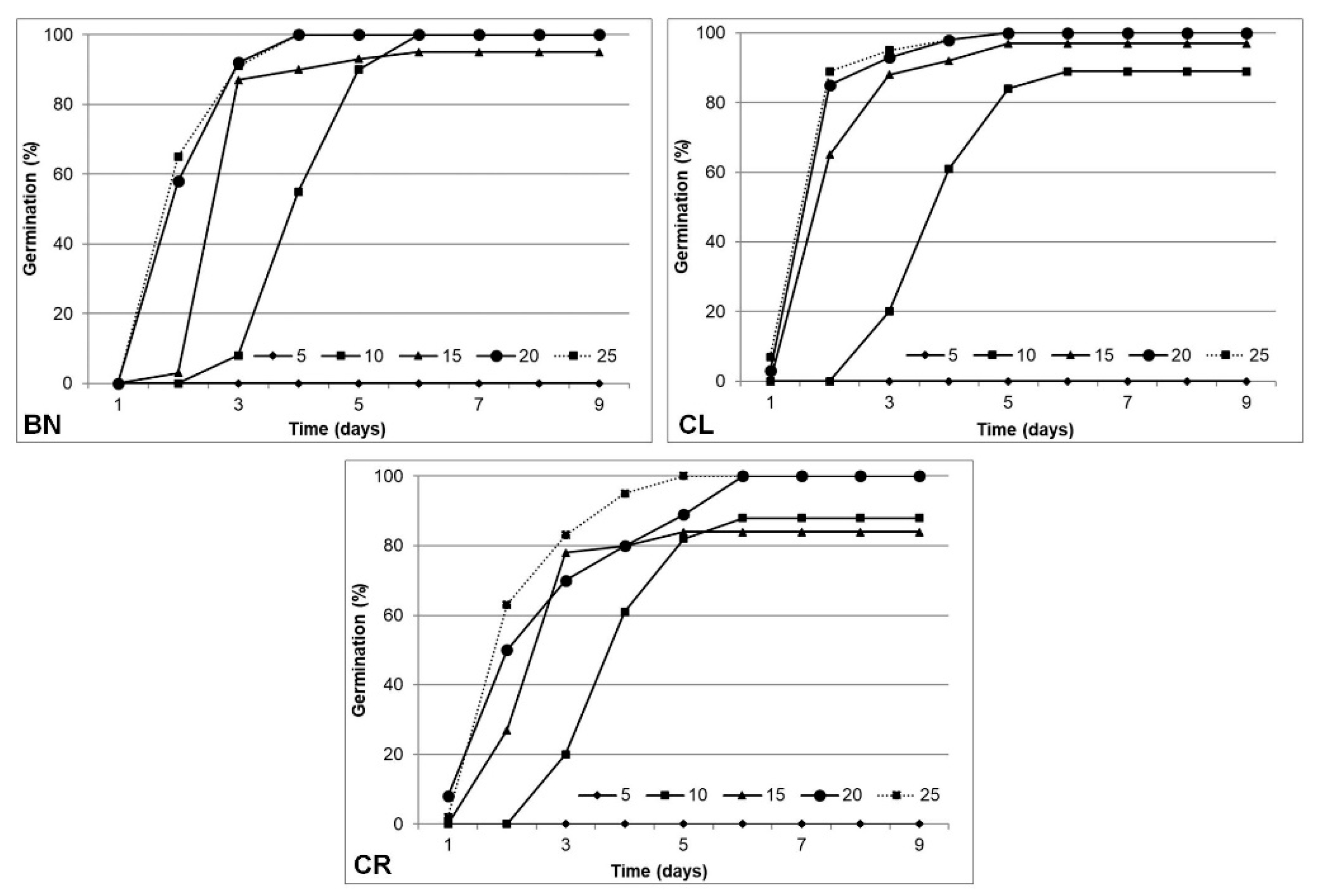
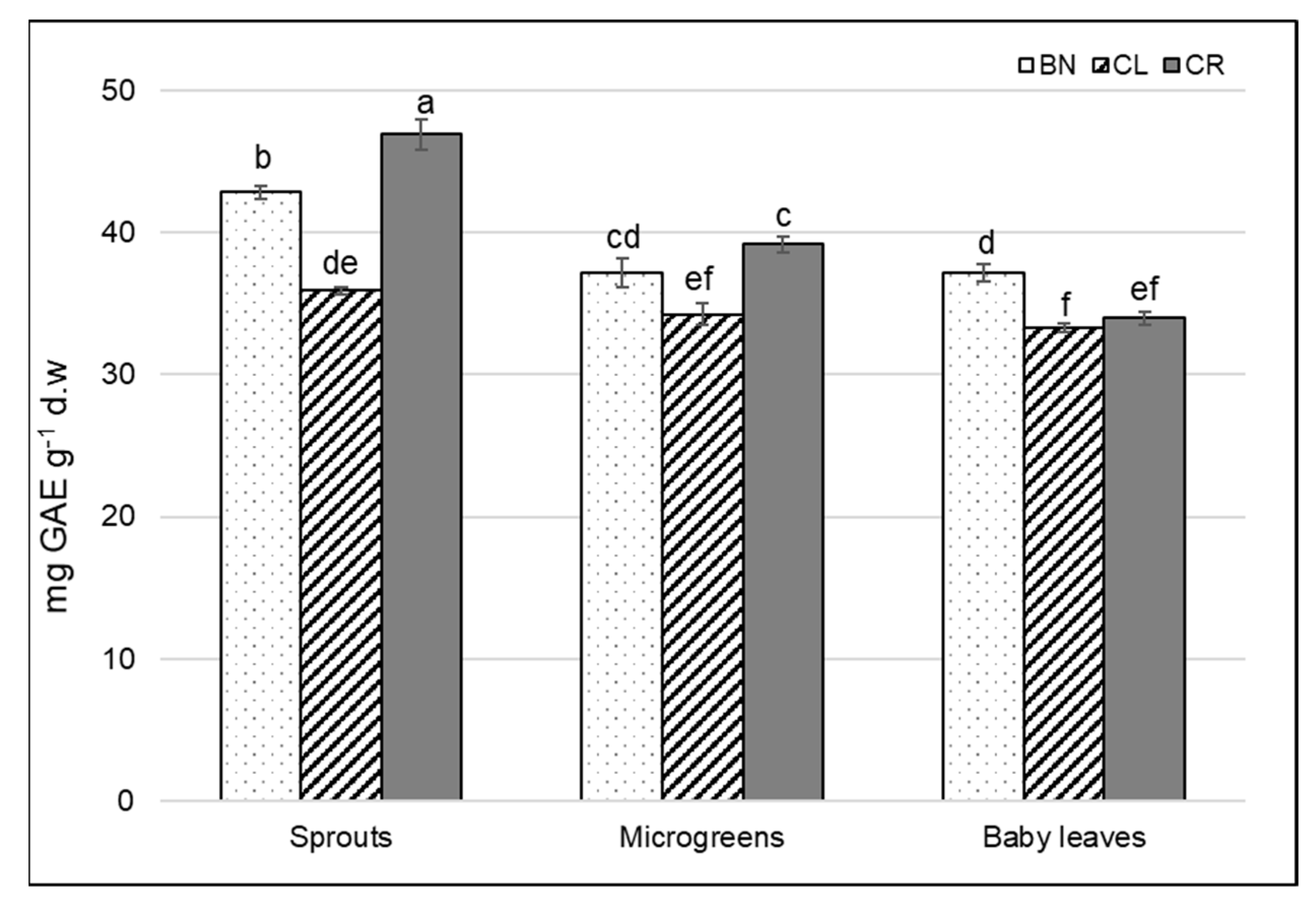

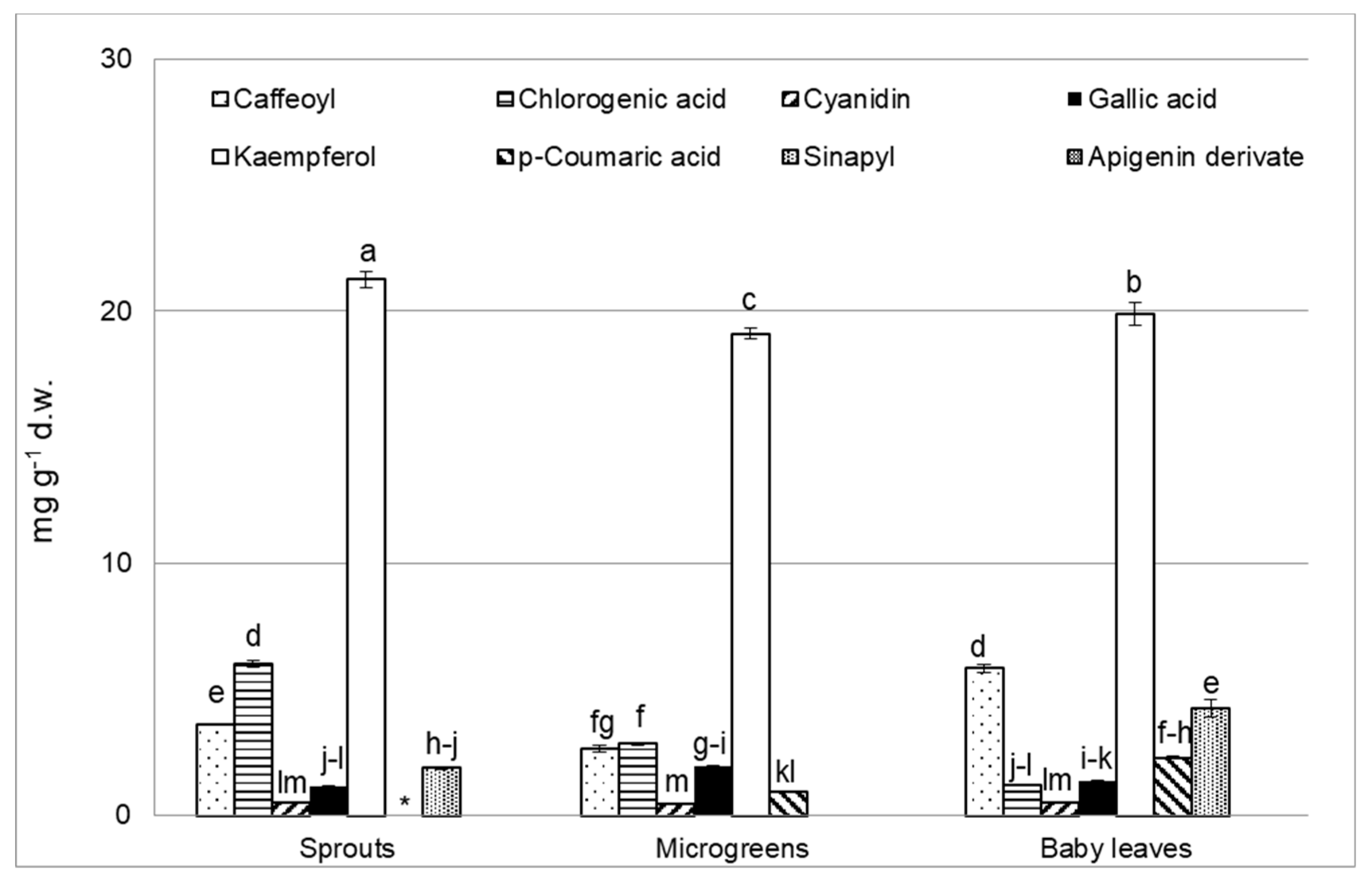
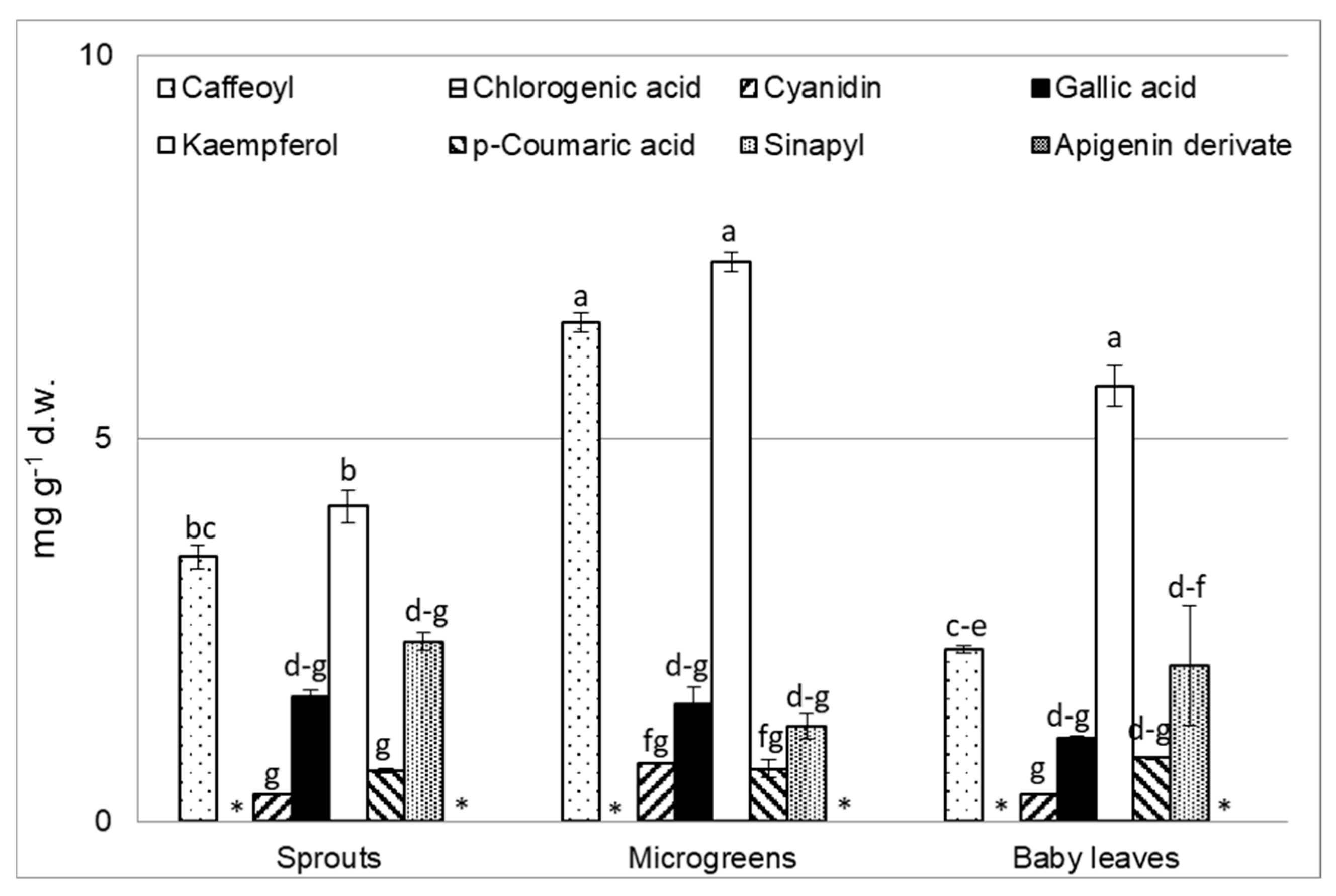

| Characteristics | BN | CL | CR | p Value |
|---|---|---|---|---|
| Weight of 1000 seeds (g) | 4.5 ± 0.1a | 4.1 ± 0.0b | 3.9 ± 0.0c | *** |
| Seeds per gram (n) | 223.6 ± 2.5c | 242.1 ± 2.7b | 254.4 ± 2.4a | *** |
| Perimeter (mm) | 6.0 ± 0.1 | 6.1 ± 0.1 | 5.8 ± 0.1 | ns |
| Longitudinal length (mm) | 1.8 ± 0.0 | 1.8 ± 0.0 | 1.8 ± 0.1 | ns |
| Transversal length (mm) | 2.0 ± 0.0 | 2.1 ± 0.1 | 2.0 ± 0.1 | ns |
| MGT (days) | 10 °C | 15 °C | 20 °C | 25 °C |
|---|---|---|---|---|
| BN | 4.5 ± 0.04a | 3.1 ± 0.04a | 2.5 ± 0.04b | 2.4 ± 0.05b |
| CL | 4.0 ± 0.06b | 2.5 ± 0.04c | 2.2 ± 0.11c | 2.1 ± 0.08c |
| CR | 4.1 ± 0.04b | 2.8 ± 0.05b | 3.0 ± 0.24a | 2.6 ± 0.03a |
| Characteristics | Sprouts | Microgreens | Baby leaves | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| BN | CL | CR | Means | BN | CL | CR | Means | BN | CL | CR | Means | |
| Weight of 10 individuals (g) | 1.0 ± 0.1a | 1.2 ± 0.2a | 1.4 ± 0.3a | 1.2 ± 0.0 | 3.4 ± 0.5a | 2.7 ± 0.4a | 2.8 ± 0.3a | 3.0 ± 0.1 | 8.4 ± 0.7a | 7.0 ± 0.8a | 7.4 ± 0.5a | 7.6 ± 0.5 |
| Hypocotyl length (mm) | 42.1 ± 0.8a | 29.2 ± 3.6b | 36.6 ± 2.3ab | 36.0 ± 1.7 | 43.3 ± 0.6a | 29.5 ± 2.6b | 28.9 ± 3.2b | 33.9 ± 1.5 | - | - | - | - |
| Cotyledon length (mm) | 10.6 ± 0.2ab | 10.3 ± 0.8b | 14.2 ± 1.2a | 11.7 ± 0.8 | 32.4 ± 0.4a | 23.5 ± 0.2b | 25.7 ±1.6b | 27.2 ± 0.6 | - | - | - | - |
| Cotyledon width (mm) | 14.5 ± 0.2b | 12.2 ± 0.8b | 16.4 ± 1.1a | 14.4 ± 0.8 | 23.1 ±0.4a | 17.6 ±1.0a | 17.8 ±2.2a | 19.5± 1.2 | - | - | - | - |
| Stem length (mm) | - | - | - | - | - | - | - | - | 107.8 ± 3.9a | 125.5 ± 6.9a | 122.4 ± 5.3a | 118.6 ± 3.9 |
| Number of true leaf (n) | - | - | - | - | 1.0 ± 0.0a | 1.0 ± 0.0a | 1.0 ± 0.0a | 1.0 ± 0 | 3.5 ± 0.2a | 3.6 ± 0.3a | 3.0 ± 0.0a | 3.4 ± 0.1 |
| Leaf length (mm) | - | - | - | - | 25.6 ± 2.4a | 23.7 ± 1.7a | 27.2 ± 2.7a | 25.2 ± 2.0 | 124.3 ± 2.1a | 113.5 ± 1.8b | 120.7 ± 1.5ab | 119.5 ± 0.6 |
| Leaf width (mm) | - | - | - | - | 16.4 ± 1.5a | 17.6 ± 1.0a | 19.0 ± 2.0a | 17.7 ± 0.9 | 42.5 ± 1.4a | 32.6 ± 0.9b | 35.3 ± 0.9b | 36.8 ± 0.7 |
| Weight of 10 individuals (g) | 42.1 ± 0.8a | 29.2 ± 3.6b | 36.6 ± 2.3ab | 36.0 ± 1.7 | 43.3 ± 0.6a | 29.5 ± 2.6b | 28.9 ± 3.2b | 33.9 ± 1.5 | - | - | - | - |
| Hypocotyl length (mm) | 10.6 ± 0.2ab | 10.3 ± 0.8b | 14.2 ± 1.2a | 11.7 ± 0.8 | 32.4 ±0.4a | 23.5 ± 0.2b | 25.7 ±1.6b | 27.2 ± 0.6 | - | - | - | - |
| FCI | AA | DPPH | ROO | |||
|---|---|---|---|---|---|---|
| Genotype (G) | ||||||
| BN | 39.06a * | 6.67a | 3.29b | 82.55a | ||
| CL | 34.48b | 5.48b | 2.91c | 80.19a | ||
| CR | 40.02a | 6.34a | 3.81a | 70.53b | ||
| P < 0.001 | P < 0.001 | P < 0.001 | P < 0.001 | |||
| Stage of growth (S) | ||||||
| Sprouts | 41.9a | 5.7c | 3.73a | 86.89a | ||
| Microgreens | 36.9b | 6.1b | 3.09b | 72.28b | ||
| Baby leaves | 34.8c | 6.3a | 3.20b | 74.11b | ||
| P < 0.001 | P < 0.001 | P < 0.001 | P < 0.001 | |||
| G x S | ||||||
| Sprouts | 42.9b | 6.4b | 3.22c | 93.28ab | ||
| BN | Microgreens | 37.2cd | 7.5a | 3.23c | 89.66b | |
| Baby leaves | 37.1d | 6.1bc | 3.43bc | 64.71d | ||
| Sprouts | 35.9de | 4.8d | 3.54b | 99.40a | ||
| CL | Microgreens | 34.2ef | 5.9c | 2.61d | 63.23d | |
| Baby leaves | 33.3f | 5.8c | 2.59d | 77.95c | ||
| Sprouts | 46.9a | 6.1bc | 4.41a | 67.99d | ||
| CR | Microgreens | 39.2c | 6.5b | 3.43bc | 63.94d | |
| Baby leaves | 34.0ef | 6.5b | 3.59b | 79.66c | ||
| P < 0.001 | P < 0.001 | P < 0.001 | P < 0.001 |
| Genotypes | Gallic Acid Derivate (mg g−1 d.w.) | Sinapyl Acid Derivate (mg g−1 d.w.) | Total Polyphenol Content (mg g−1 d.w.) |
|---|---|---|---|
| BN | 0.206 ± 0.021b | 2.826 ± 0.051b | 3.032 ± 0.252b |
| CL | 0.525 ± 0.062a | 3.413 ± 0.251a | 3.937 ± 0.313a |
| CR | 0.261 ± 0.047b | 3.572 ± 0.412a | 3.832 ± 0.456a |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Bella, M.C.; Niklas, A.; Toscano, S.; Picchi, V.; Romano, D.; Lo Scalzo, R.; Branca, F. Morphometric Characteristics, Polyphenols and Ascorbic Acid Variation in Brassica oleracea L. Novel Foods: Sprouts, Microgreens and Baby Leaves. Agronomy 2020, 10, 782. https://doi.org/10.3390/agronomy10060782
Di Bella MC, Niklas A, Toscano S, Picchi V, Romano D, Lo Scalzo R, Branca F. Morphometric Characteristics, Polyphenols and Ascorbic Acid Variation in Brassica oleracea L. Novel Foods: Sprouts, Microgreens and Baby Leaves. Agronomy. 2020; 10(6):782. https://doi.org/10.3390/agronomy10060782
Chicago/Turabian StyleDi Bella, Maria Concetta, Agnieszka Niklas, Stefania Toscano, Valentina Picchi, Daniela Romano, Roberto Lo Scalzo, and Ferdinando Branca. 2020. "Morphometric Characteristics, Polyphenols and Ascorbic Acid Variation in Brassica oleracea L. Novel Foods: Sprouts, Microgreens and Baby Leaves" Agronomy 10, no. 6: 782. https://doi.org/10.3390/agronomy10060782
APA StyleDi Bella, M. C., Niklas, A., Toscano, S., Picchi, V., Romano, D., Lo Scalzo, R., & Branca, F. (2020). Morphometric Characteristics, Polyphenols and Ascorbic Acid Variation in Brassica oleracea L. Novel Foods: Sprouts, Microgreens and Baby Leaves. Agronomy, 10(6), 782. https://doi.org/10.3390/agronomy10060782









