Earthworms (Lumbricus terrestris L.) Mediate the Fertilizing Effect of Frass
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Earthworm Survival
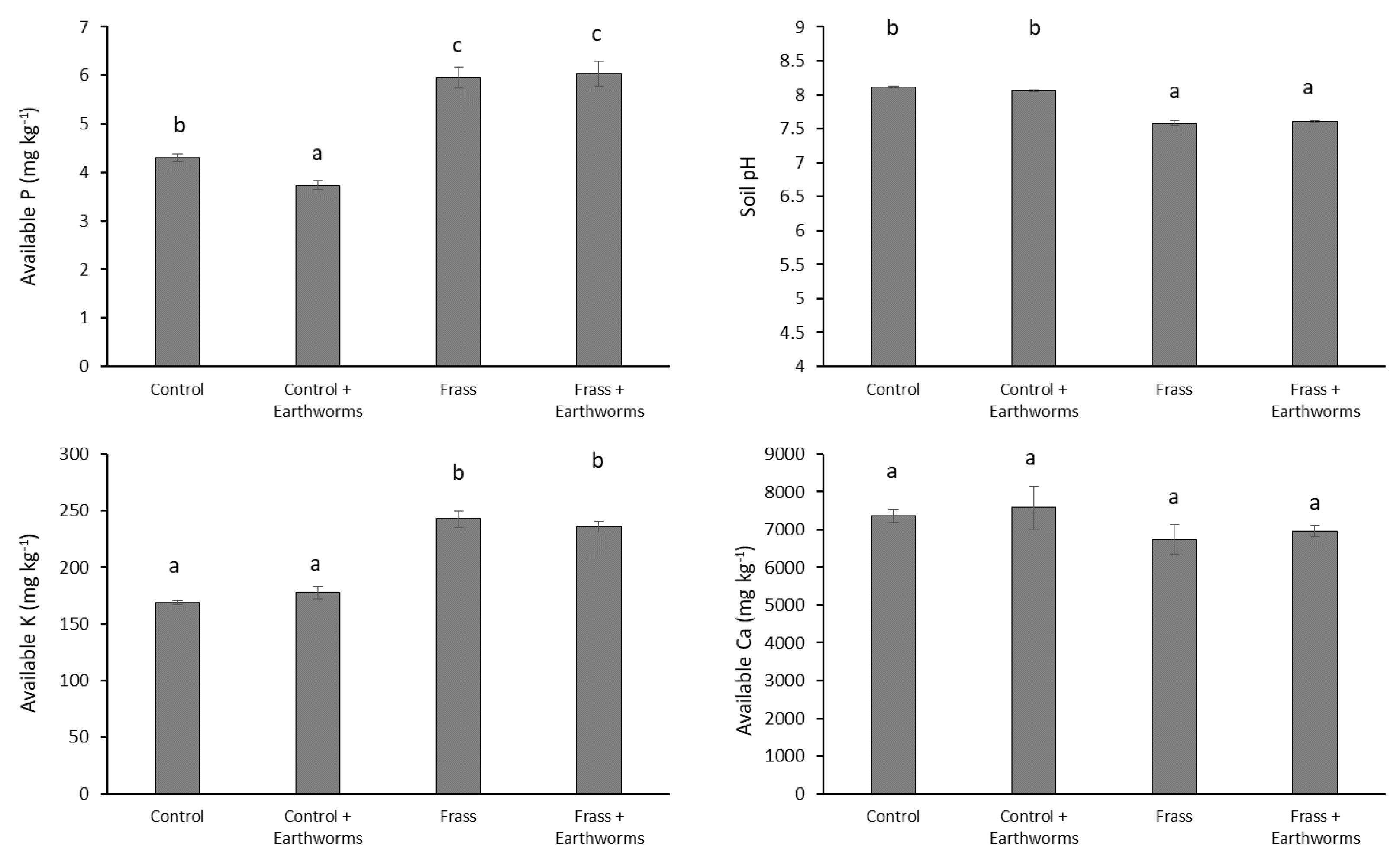
3.2. Impact of Earthworms on Nutrient Uptake and Crop Growth
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dicke, M. Insects as feed and the Sustainable Development Goals. J. Insects Food Feed 2018, 4, 147–156. [Google Scholar] [CrossRef]
- Poveda, J.; Jimenez-Gomez, A.; Saati-Santamaría, Z.; Usategui-Martín, R.; Rivas, R.; García-Fraile, P. Mealworm frass as a potential biofertilizer and abiotic stress tolerance-inductor in plants. Appl. Soil Ecol. 2019, 142, 110–122. [Google Scholar] [CrossRef]
- Houben, D.; Daoulas, G.; Faucon, M.-P.; Dulaurent, A.-M. Potential use of mealworm frass as a fertilizer: Impact on crop growth and soil properties. Sci. Rep. 2020, 10, 4659. [Google Scholar] [CrossRef]
- Schmitt, E.; de Vries, W. Potential benefits of using Hermetia illucens frass as a soil amendment on food production and for environmental impact reduction. Curr. Opin. Green Sustain. Chem. 2020. [Google Scholar] [CrossRef]
- Frost, C.J.; Hunter, M.D. Insect Canopy herbivory and frass deposition affect soil nutrient dynamics and export in oak mesocosms. Ecology 2004, 85, 3335–3347. [Google Scholar] [CrossRef]
- Kagata, H.; Ohgushi, T. Positive and negative impacts of insect frass quality on soil nitrogen availability and plant growth. Popul. Ecol. 2012, 54, 75–82. [Google Scholar] [CrossRef]
- Lovett, G.M.; Ruesink, A.E. Carbon and nitrogen mineralization from decomposing gypsy moth frass. Oecologia 1995, 104, 133–138. [Google Scholar] [CrossRef]
- van Groenigen, J.W.; Lubbers, I.M.; Vos, H.M.J.; Brown, G.G.; De Deyn, G.B.; van Groenigen, K.J. Earthworms increase plant production: A meta-analysis. Sci. Rep. 2014, 4, 6365. [Google Scholar] [CrossRef]
- Blouin, M.; Hodson, M.E.; Delgado, E.A.; Baker, G.; Brussaard, L.; Butt, K.R.; Dai, J.; Dendooven, L.; Peres, G.; Tondoh, J.E.; et al. A review of earthworm impact on soil function and ecosystem services. Eur. J. Soil Sci. 2013, 64, 161–182. [Google Scholar] [CrossRef]
- Blanchart, E.; Albrecht, A.; Alegre, J.; Duboisset, A.; Gilot, C.; Pashanasi, B.; Lavelle, P.; Brussaard, L. Effects of earthworms on soil structure and physical properties. In Earthworm Management in Tropical Agroecosystems; Lavelle, P., Brussaard, L., Hendrix, P., Eds.; CABI: New York, NY, USA, 1999; pp. 149–171. [Google Scholar]
- Chapuis-Lardy, L.; Le Bayon, R.-C.; Brossard, M.; Lopez-Hernandez, D.; Blanchart, E. Role of Soil Macrofauna in Phosphorus Cycling. In Phosphorus in Action: Biological Processes in Soil Phosphorus Cycling; Bünemann, E., Oberson, A., Frossard, E., Eds.; Soil Biology; Springer: Berlin/Heidelberg, Germany, 2011; pp. 199–213. ISBN 978-3-642-15271-9. [Google Scholar]
- Postma-Blaauw, M.B.; Bloem, J.; Faber, J.H.; van Groenigen, J.W.; de Goede, R.G.M.; Brussaard, L. Earthworm species composition affects the soil bacterial community and net nitrogen mineralization. Pedobiologia 2006, 50, 243–256. [Google Scholar] [CrossRef]
- Medina-Sauza, R.M.; Alvarez-Jimenez, M.; Delhal, A.; Reverchon, F.; Blouin, M.; Guerrero-Analco, J.A.; Cerdan, C.R.; Guevara, R.; Villain, L.; Barois, I. Earthworms Building Up Soil Microbiota, a Review. Front. Environ. Sci. 2019, 7, 81. [Google Scholar] [CrossRef]
- Waqar, A.; Shah, G.M.; Bakhat, H.F.; Shahid, M.; Aslam, M.; Ashraf, M.R.; Hafeez, R.; Murtaza, B.; Rashid, M.I. The earthworm species Pheretima hawayana influences organic wastes decomposition, nitrogen mineralization and maize N recovery. Eur. J. Soil Biol. 2019, 90, 1–8. [Google Scholar] [CrossRef]
- Sharpley, A.; McDowell, R.; Moyer, B.; Littlejohn, R. Land application of manure can influence earthworm activity and soil phosphorus distribution. Commun. Soil Sci. Plant Anal. 2011, 42, 194–207. [Google Scholar] [CrossRef]
- Jouquet, P.; Plumere, T.; Thu, T.D.; Rumpel, C.; Duc, T.T.; Orange, D. The rehabilitation of tropical soils using compost and vermicompost is affected by the presence of endogeic earthworms. Appl. Soil Ecol. 2010, 46, 125–133. [Google Scholar] [CrossRef]
- Clause, J.; Barot, S.; Richard, B.; Decaëns, T.; Forey, E. The interactions between soil type and earthworm species determine the properties of earthworm casts. Appl. Soil Ecol. 2014, 83, 149–158. [Google Scholar] [CrossRef]
- Houben, D.; Meunier, C.; Pereira, B.; Sonnet, P. Predicting the degree of phosphorus saturation using the ammonium acetate–EDTA soil test. Soil Use Manag. 2011, 27, 283–293. [Google Scholar] [CrossRef]
- Vos, H.M.J.; Ros, M.B.H.; Koopmans, G.F.; van Groenigen, J.W. Do earthworms affect phosphorus availability to grass? A pot experiment. Soil Biol. Biochem. 2014, 79, 34–42. [Google Scholar] [CrossRef]
- Coulis, M.; Bernard, L.; Gerard, F.; Hinsinger, P.; Plassard, C.; Villeneuve, M.; Blanchart, E. Endogeic earthworms modify soil phosphorus, plant growth and interactions in a legume–cereal intercrop. Plant Soil 2014, 379, 149–160. [Google Scholar] [CrossRef]
- Sissingh, H.A. Analytical technique of the Pw method, used for the assessment of the phosphate status of arable soils in the Netherlands. Plant Soil 1971, 34, 483–486. [Google Scholar] [CrossRef]
- Gomez-Suarez, A.D.; Nobile, C.; Faucon, M.-P.; Pourret, O.; Houben, D. Fertilizer potential of struvite as affected by nitrogen form in the rhizosphere. Sustainability 2020, 12, 2212. [Google Scholar] [CrossRef]
- R Core Team. R: A language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017; ISBN 3-900051-07-0. Available online: https://www.R-project.org (accessed on 22 December 2019).
- Fox, J. The R Commander: A Basic-Statistics Graphical User Interface to R. J. Stat. Softw. 2005, 14, 1–42. [Google Scholar] [CrossRef]
- Sizmur, T.; Martin, E.; Wagner, K.; Parmentier, E.; Watts, C.; Whitmore, A.P. Milled cereal straw accelerates earthworm (Lumbricus terrestris) growth more than selected organic amendments. Appl. Soil Ecol. 2017, 113, 166–177. [Google Scholar] [CrossRef] [PubMed]
- Griffith, B.; Türke, M.; Weisser, W.W.; Eisenhauer, N. Herbivore behavior in the anecic earthworm species Lumbricus terrestris L.? Eur. J. Soil Biol. 2013, 55, 62–65. [Google Scholar] [CrossRef]
- Cortez, J.; Bouche, M.B. Do earthworms eat living roots? Soil Biol. Biochem. 1992, 24, 913–915. [Google Scholar] [CrossRef]
- Scheu, S. Effects of earthworms on plant growth: Patterns and perspectives. Pedobiologia 2003, 47, 846–856. [Google Scholar] [CrossRef]
- Baker, G. Differences in nitrogen release from surface and incorporated plant residues by two endogeic species of earthworms (Lumbricidae) in a red–brown earth soil in southern Australia. Eur. J. Soil Biol. 2007, 43, S165–S170. [Google Scholar] [CrossRef]
- Lubbers, I.M.; Brussaard, L.; Otten, W.; Groenigen, J.W.V. Earthworm-induced N mineralization in fertilized grassland increases both N2O emission and crop-N uptake. Eur. J. Soil Sci. 2011, 62, 152–161. [Google Scholar] [CrossRef]
- Amador, J.A.; Gorres, J.H.; Savin, M.C. Effects of Lumbricus terrestris L. on nitrogen dynamics beyond the burrow. Appl. Soil Ecol. 2006, 33, 61–66. [Google Scholar] [CrossRef]
- Amador, J.A.; Gorres, J.H.; Savin, M.C. Carbon and nitrogen dynamics in Lumbricus terrestris (L.) burrow Soil. Soil Sci. Soc. Am. J. 2003, 67, 1755–1762. [Google Scholar] [CrossRef]
- Zhang, S.; Chao, Y.; Zhang, C.; Cheng, J.; Li, J.; Ma, N. Earthworms enhanced winter oilseed rape (Brassica napus L.) growth and nitrogen uptake. Agric. Ecosyst. Environ. 2010, 139, 463–468. [Google Scholar] [CrossRef]
- Amador, J.A.; Gorres, J.H. Role of the anecic earthworm Lumbricus terrestris L. in the distribution of plant residue nitrogen in a corn (Zea mays)–soil system. Appl. Soil Ecol. 2005, 30, 203–214. [Google Scholar] [CrossRef]
- Rashid, M.I.; de Goede, R.G.M.; Corral Nunez, G.A.; Brussaard, L.; Lantinga, E.A. Soil pH and earthworms affect herbage nitrogen recovery from solid cattle manure in production grassland. Soil Biol. Biochem. 2014, 68, 1–8. [Google Scholar] [CrossRef]
- Edwards, C.A. Earthworm Ecology, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2004; ISBN 978-0-429-12904-9. [Google Scholar]
- Lopez-Hernandez, D.; Lavelle, P.; Fardeau, J.C.; Nino, M. Phosphorus transformations in two P-sorption contrasting tropical soils during transit through Pontoscolex corethrurus (Glossoscolecidae: Oligochaeta). Soil Biol. Biochem. 1993, 25, 789–792. [Google Scholar] [CrossRef]
- Le Bayon, R.C.; Binet, F. Earthworms change the distribution and availability of phosphorous in organic substrates. Soil Biol. Biochem. 2006, 38, 235–246. [Google Scholar] [CrossRef]
- Vos, H.M.J.; Koopmans, G.F.; Beezemer, L.; de Goede, R.G.M.; Hiemstra, T.; van Groenigen, J.W. Large variations in readily-available phosphorus in casts of eight earthworm species are linked to cast properties. Soil Biol. Biochem. 2019, 138, 107583. [Google Scholar] [CrossRef]
- Li, H.; Xiang, D.; Wang, C.; Li, X.; Lou, Y. Effects of epigeic earthworm (Eisenia fetida) and arbuscular mycorrhizal fungus (Glomus intraradices) on enzyme activities of a sterilized soil–sand mixture and nutrient uptake by maize. Biol. Fertil. Soil 2012, 48, 879–887. [Google Scholar] [CrossRef]
- Nobile, C.; Houben, D.; Michel, E.; Firmin, S.; Lambers, H.; Kandeler, E.; Faucon, M.-P. Phosphorus-acquisition strategies of canola, wheat and barley in soil amended with sewage sludges. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Osone, Y.; Ishida, A.; Tateno, M. Correlation between relative growth rate and specific leaf area requires associations of specific leaf area with nitrogen absorption rate of roots. New Phytol. 2008, 179, 417–427. [Google Scholar] [CrossRef]
- Hodgson, J.G.; Montserrat-Martí, G.; Charles, M.; Jones, G.; Wilson, P.; Shipley, B.; Sharafi, M.; Cerabolini, B.E.L.; Cornelissen, J.H.C.; Band, S.R.; et al. Is leaf dry matter content a better predictor of soil fertility than specific leaf area? Ann. Bot. 2011, 108, 1337–1345. [Google Scholar] [CrossRef]
- Madani, N.; Kimball, J.S.; Running, S.W. Improving global gross primary productivity estimates by computing optimum light use efficiencies using flux tower data. J. Geophys. Res. Biogeosci. 2017, 122, 2939–2951. [Google Scholar] [CrossRef]
- Ordoñez, J.C.; Bodegom, P.M.V.; Witte, J.-P.M.; Wright, I.J.; Reich, P.B.; Aerts, R. A global study of relationships between leaf traits, climate and soil measures of nutrient fertility. Glob. Ecol. Biogeogr. 2009, 18, 137–149. [Google Scholar] [CrossRef]
- Lambers, H.; Poorter, H. Inherent variation in growth rate between pigher Plants: A search for physiological causes and ecological consequences. In Advances in Ecological Research; Begon, M., Fitter, A.H., Eds.; Academic Press: Amsterdam, The Netherlands, 1992; Volume 23, pp. 187–261. [Google Scholar]
- Gong, H.; Gao, J. Soil and climatic drivers of plant SLA (specific leaf area). Glob. Ecol. Conserv. 2019, 20, e00696. [Google Scholar] [CrossRef]
- Yao, H.; Zhang, Y.; Yi, X.; Zhang, X.; Zhang, W. Cotton responds to different plant population densities by adjusting specific leaf area to optimize canopy photosynthetic use efficiency of light and nitrogen. Field Crops Res. 2016, 188, 10–16. [Google Scholar] [CrossRef]
- Knops, J.M.; Reinhart, K. Specific leaf area along a nitrogen fertilization gradient. Am. Midl. Nat. 2000, 144, 265–272. [Google Scholar] [CrossRef]
- Ingham, R.E.; Detling, J.K. Effects of root-feeding nematodes on aboveground net primary production in a North American grassland. Plant Soil 1990, 121, 279–281. [Google Scholar] [CrossRef]


| Organic C g kg−1 | Total N g kg−1 | Total K g kg−1 | Total P g kg−1 | pH | Soluble Fraction %Corg | Hemicellulose-Like Fraction %Corg | Cellulose-Like Fraction %Corg | Lignin-Like Fraction %Corg |
|---|---|---|---|---|---|---|---|---|
| 393 | 50 | 17 | 20 | 5.8 | 49.3 | 31 | 15.2 | 4.4 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dulaurent, A.-M.; Daoulas, G.; Faucon, M.-P.; Houben, D. Earthworms (Lumbricus terrestris L.) Mediate the Fertilizing Effect of Frass. Agronomy 2020, 10, 783. https://doi.org/10.3390/agronomy10060783
Dulaurent A-M, Daoulas G, Faucon M-P, Houben D. Earthworms (Lumbricus terrestris L.) Mediate the Fertilizing Effect of Frass. Agronomy. 2020; 10(6):783. https://doi.org/10.3390/agronomy10060783
Chicago/Turabian StyleDulaurent, Anne-Maïmiti, Guillaume Daoulas, Michel-Pierre Faucon, and David Houben. 2020. "Earthworms (Lumbricus terrestris L.) Mediate the Fertilizing Effect of Frass" Agronomy 10, no. 6: 783. https://doi.org/10.3390/agronomy10060783
APA StyleDulaurent, A.-M., Daoulas, G., Faucon, M.-P., & Houben, D. (2020). Earthworms (Lumbricus terrestris L.) Mediate the Fertilizing Effect of Frass. Agronomy, 10(6), 783. https://doi.org/10.3390/agronomy10060783







