Grain Endogenous Selenium and Moderate Salt Stress Work as Synergic Elicitors in the Enrichment of Bioactive Compounds in Maize Sprouts
Abstract
1. Introduction
2. Materials and Methods
2.1. Sprout Production
2.2. Se Speciation
2.3. Ion Concentrations
2.4. Metabolomics Analyses
2.5. Statistical Analysis
3. Results
3.1. Growth Indexes
3.2. Selenium Concentration
3.3. Mineral Concentrations
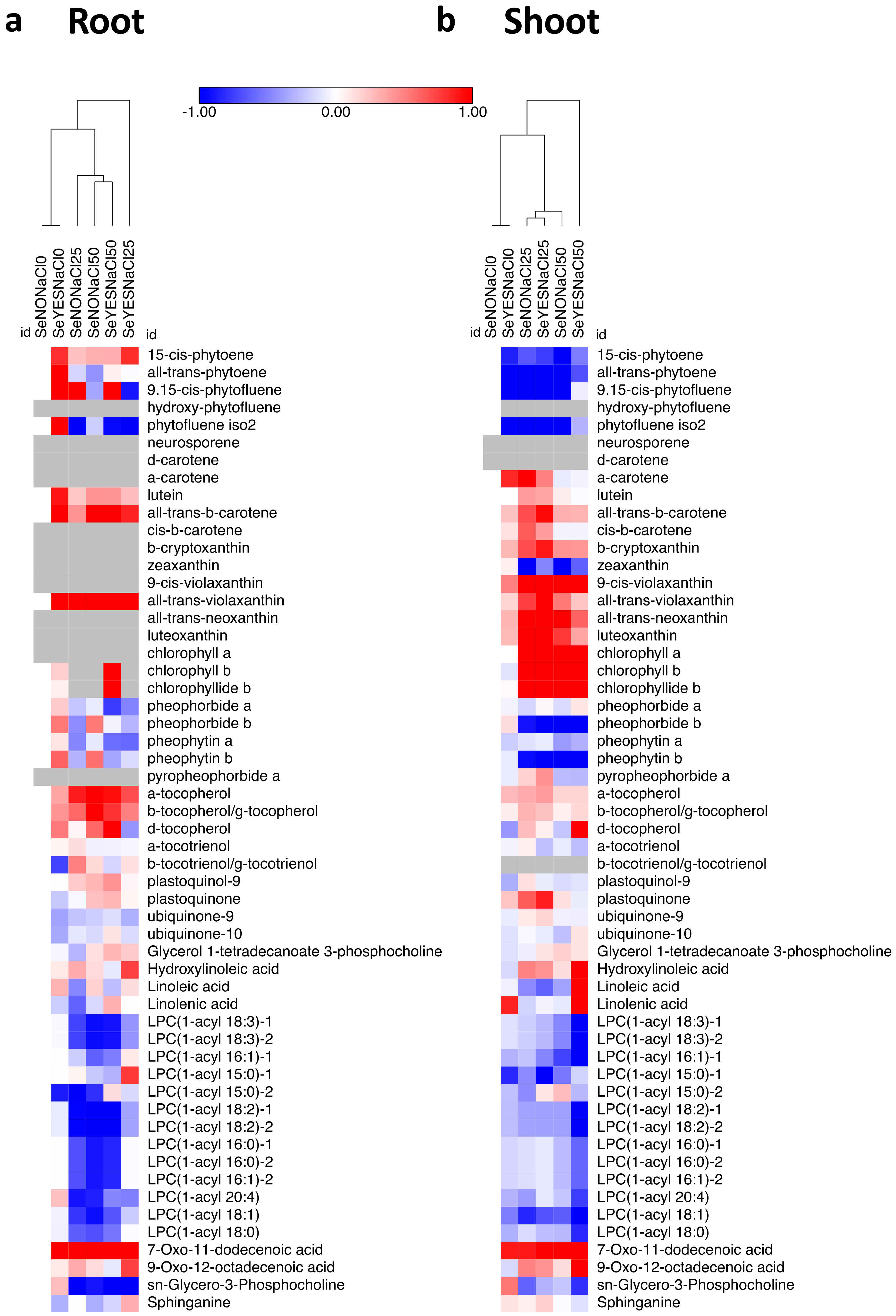
3.4. Polar and Non-Polar Metabolomics
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Benincasa, P.; Falcinelli, B.; Lutts, S.; Stagnari, F.; Galieni, A. Sprouted Grains: A Comprehensive Review. Nutrients 2019, 11, 421. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.K.; Kang, Y.F.; Zhao, X.Y.; Liu, Y.P.; Zhang, X.W.; Zhang, S.J. Effects of elicitation on bioactive compounds and biological activities of sprouts. J. Funct. Foods 2019, 53, 136–145. [Google Scholar] [CrossRef]
- Świeca, M. Elicitation with abiotic stresses improves pro-health constituents, antioxidant potential and nutritional quality of lentil sprouts. Saudi J. Biol. Sci. 2015, 22, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Yao, Y.; Zhu, Y.; Ren, G. Isoflavone content and composition in chickpea (Cicer arietinum L.) sprouts germinated under different conditions. J. Agric. Food Chem. 2015, 63, 2701–2707. [Google Scholar] [CrossRef]
- Natella, F.; Maldini, M.; Nardini, M.; Azzini, E.; Foddai, M.S.; Giusti, A.M.; Baima, S.; Morelli, G.; Scaccini, C. Improvement of the nutraceutical quality of broccoli sprouts by elicitation. Food Chem. 2016, 201, 101–109. [Google Scholar] [CrossRef]
- Falcinelli, B.; Sileoni, V.; Marconi, O.; Perretti, G.; Quinet, M.; Lutts, S.; Benincasa, P. Germination under moderate salinity increases phenolic content and antioxidant activity in rapeseed (Brassica napus var. oleifera Del.) sprouts. Molecules 2017, 22, 1377. [Google Scholar] [CrossRef]
- Falcinelli, B.; Benincasa, P.; Calzuola, I.; Gigliarelli, L.; Lutts, S.; Marsili, V. Phenolic content and antioxidant activity in raw and denatured aqueous extracts from sprouts and wheatgrass of einkorn and emmer obtained under salinity. Molecules 2017, 22, 2132. [Google Scholar] [CrossRef]
- Stagnari, F.; Galieni, A.; D’Egidio, S.; Falcinelli, B.; Pagnani, G.; Pace, R.; Pisante, M.; Benincasa, P. Effects of sprouting and salt stress on polyphenol composition and antiradical activity of einkorn, emmer and durum wheat. Ital. J. Agron. 2017, 12. [Google Scholar] [CrossRef]
- Guardado-Félix, D.; Serna-Saldivar, S.O.; Cuevas-Rodríguez, E.O.; Jacobo-Velázquez, D.A.; Gutiérrez-Uribe, J.A. Effect of sodium selenite on isoflavonoid contents and antioxidant capacity of chickpea (Cicer arietinum L.) sprouts. Food Chem. 2017, 226, 69–74. [Google Scholar] [CrossRef]
- Ávila, F.W.; Yang, Y.; Faquin, V.; Ramos, S.J.; Guilherme, L.R.G.; Thannhauser, T.W.; Li, L. Impact of selenium supply on Se-methylselenocysteine and glucosinolate accumulation in selenium-biofortified Brassica sprouts. Food Chem. 2014, 165, 578–586. [Google Scholar] [CrossRef]
- Tian, M.; Xu, X.; Liu, Y.; Xie, L.; Pan, S. Effect of Se treatment on glucosinolate metabolism and health-promoting compounds in the broccoli sprouts of three cultivars. Food Chem. 2016, 190, 374–380. [Google Scholar] [CrossRef] [PubMed]
- D’Amato, R.; Fontanella, M.C.; Falcinelli, B.; Beone, G.M.; Bravi, E.; Marconi, O.; Benincasa, P.; Businelli, D. Selenium Biofortification in Rice (Oryza sativa L.) Sprouting: Effects on Se Yield and Nutritional Traits with Focus on Phenolic Acid Profile. J. Agric. Food Chem. 2018, 66, 4082–4090. [Google Scholar] [CrossRef] [PubMed]
- Hasanuzzam, M.; Hossain, M.A.; Fujita, M. Selenium in Higher Plants: Physiological Role, Antioxidant Metabolism and Abiotic Stress Tolerance. J. Plant. Sci. 2010, 5, 354–375. [Google Scholar] [CrossRef]
- Bocchini, M.; D’Amato, R.; Ciancaleoni, S.; Fontanella, M.C.; Palmerini, C.A.; Beone, G.M.; Onofri, A.; Negri, V.; Marconi, G.; Albertini, E.; et al. Soil Selenium (Se) Biofortification Changes the Physiological, Biochemical and Epigenetic Responses to Water Stress in Zea mays L. by Inducing a Higher Drought Tolerance. Front. Plant. Sci. 2018, 9, 389. [Google Scholar] [CrossRef] [PubMed]
- D’Amato, R.; De Feudis, M.; Hasuoka, P.E.; Regni, L.; Pacheco, P.H.; Onofri, A.; Businelli, D.; Proietti, P. The Selenium Supplementation Influences Olive Tree Production and Oil Stability Against Oxidation and Can Alleviate the Water Deficiency Effects. Front. Plant. Sci. 2018, 9, 1–8. [Google Scholar] [CrossRef]
- Ahmad, R.; Waraich, E.A.; Nawaz, F.; Ashraf, M.Y.; Khalid, M. Selenium (Se) improves drought tolerance in crop plants—A myth or fact? J. Sci. Food Agric. 2016, 96, 372–380. [Google Scholar] [CrossRef]
- Fontanella, M.C.; D’Amato, R.; Regni, L.; Proietti, P.; Beone, G.M.; Businelli, D. Selenium speciation profiles in biofortified sangiovese wine. J. Trace Elem. Med. Biol. 2017, 43, 87–92. [Google Scholar] [CrossRef]
- Mattioli, S.; Dal Bosco, A.; Duarte, J.M.M.; D’Amato, R.; Castellini, C.; Beone, G.M.; Fontanella, M.C.; Beghelli, D.; Regni, L.; Businelli, D.; et al. Use of Selenium-enriched olive leaves in the feed of growing rabbits: Effect on oxidative status, mineral profile and Selenium speciation of Longissimus dorsi meat. J. Trace Elem. Med. Biol. 2019, 51, 98–105. [Google Scholar] [CrossRef]
- Thiry, C.; Ruttens, A.; De Temmerman, L.; Schneider, Y.J.; Pussemier, L. Current knowledge in species-related bioavailability of selenium in food. Food Chem. 2012, 130, 767–784. [Google Scholar] [CrossRef]
- Xiang, N.; Guo, X.; Liu, F.; Li, Q.; Hu, J.; Brennan, C.S. Effect of light- and dark-germination on the phenolic biosynthesis, phytochemical profiles, and antioxidant activities in sweet corn (Zea mays L.) sprouts. Int. J. Mol. Sci. 2017, 18, 1246. [Google Scholar] [CrossRef]
- Chalorcharoenying, W.; Lomthaisong, K.; Suriharn, B.; Lertrat, K. Germination process increases phytochemicals in corn. Int. Food Res. J. 2017, 24, 552–558. [Google Scholar]
- Randhir, R.; Shetty, K. Developmental stimulation of total phenolics and related antioxidant activity in light- and dark-germinated corn by natural elicitors. Process. Biochem. 2005, 40, 1721–1732. [Google Scholar] [CrossRef]
- Diowksz, A.; Kordialik-Bogacka, E.; Ambroziak, W. Se-enriched sprouted seeds as functional additives in sourdough fermentation. LWT Food Sci. Technol. 2014, 56, 524–528. [Google Scholar] [CrossRef]
- Puccinelli, M.; Malorgio, F.; Rosellini, I.; Pezzarossa, B. Production of selenium-biofortified microgreens from selenium-enriched seeds of basil. J. Sci. Food Agric. 2019, 99, 5601–5605. [Google Scholar] [CrossRef] [PubMed]
- D’Amato, R.; De Feudis, M.; Guiducci, M.; Businelli, D. Zea mays L. Grain: Increase in Nutraceutical and Antioxidant Properties Due to Se Fortification in Low and High Water Regimes. J. Agric. Food Chem. 2019, 67, 7050–7059. [Google Scholar] [CrossRef] [PubMed]
- Mimmo, T.; Tiziani, R.; Valentinuzzi, F.; Lucini, L.; Nicoletto, C.; Sambo, P.; Scampicchio, M.; Pii, Y.; Cesco, S. Selenium Biofortification in Fragaria × ananassa: Implications on Strawberry Fruits Quality, Content of Bioactive Health Beneficial Compounds and Metabolomic Profile. Front. Plant. Sci. 2017, 8, 1–12. [Google Scholar] [CrossRef]
- Cappelli, G.; Giovannini, D.; Basso, A.L.; Demurtas, O.C.; Diretto, G.; Santi, C.; Girelli, G.; Bacchetta, L.; Mariani, F. A Corylus avellana L. extract enhances human macrophage bactericidal response against Staphylococcus aureus by increasing the expression of anti-inflammatory and iron metabolism genes. J. Funct. Foods 2018, 45, 499–511. [Google Scholar] [CrossRef]
- Marsili, A.; Larsen, P.R.; Zavacki, A.M. Tissue-Specific Regulation of Thyroid Status by Selenodeiodinases. In Selenium; Springer: Cham, Switzerland, 2016; pp. 487–498. [Google Scholar]
- Diretto, G.; Frusciante, S.; Fabbri, C.; Schauer, N.; Busta, L.; Wang, Z.; Matas, A.J.; Fiore, A.; Rose, K.C.J.; Fernie, A.R.; et al. Manipulation of β-carotene levels in tomato fruits results in increased ABA content and extended shelf life. Plant. Biotechnol. J. 2019, 18, 1185–1199. [Google Scholar] [CrossRef]
- Sulli, M.; Mandolino, G.; Sturaro, M.; Onofri, C.; Diretto, G.; Parisi, B.; Giuliano, G. Molecular and biochemical characterization of a potato collection with contrasting tuber carotenoid content. PLoS ONE 2017, 12, e0184143. [Google Scholar] [CrossRef]
- Wen, W.; Li, D.; Li, X.; Gao, Y.; Li, W.; Li, H.; Liu, J.; Liu, H.; Chen, W.; Luo, J.; et al. Metabolome-based genome-wide association study of maize kernel leads to novel biochemical insights. Nat. Commun. 2014, 5, 1–10. [Google Scholar]
- Diretto, G.; Jin, X.; Capell, T.; Zhu, C.; Gomez-Gomez, L. Differential accumulation of pelargonidin glycosides in petals at three different developmental stages of the orange-flowered gentian (Gentiana lutea L. var. aurantiaca). PLoS ONE 2019, 14, e0212062. [Google Scholar] [CrossRef] [PubMed]
- Menzel, C.; González-Martínez, C.; Vilaplana, F.; Diretto, G.; Chiralt, A. Incorporation of natural antioxidants from rice straw into renewable starch films. Int. J. Biol. Macromol. 2020, 146, 976–986. [Google Scholar] [CrossRef] [PubMed]
- Perera, C.O.; Yen, G.M. Functional properties of carotenoids in human health. Int. J. Food Prop. 2007, 10, 201–230. [Google Scholar] [CrossRef]
- Adhikari, K.B.; Tanwir, F.; Gregersen, P.L.; Steffensen, S.K.; Jensen, B.M.; Poulsen, L.K.; Nielsen, C.H.; Høyer, S.; Borre, M.; Fomsgaard, I.S. Benzoxazinoids: Cereal phytochemicals with putative therapeutic and health-protecting properties. Mol. Nutr. Food Res. 2015, 59, 1324–1338. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; Wen, W.; Alseekh, S.; Yang, X.; Guo, H.; Li, W.; Wang, L.; Pan, Q.; Zhan, W.; Liu, J.; et al. Large-scale metabolite quantitative trait locus analysis provides new insights for high-quality maize improvement. Plant. J. 2019, 99, 216–230. [Google Scholar] [CrossRef] [PubMed]
- Cantero, I.; Abete, I.; Del Bas, J.M.; Caimari, A.; Arola, L.; Zulet, M.A.; Martinez, J.A. Changes in lysophospholipids and liver status after weight loss: The RESMENA study. Nutr. Metab. 2018, 15, 1–11. [Google Scholar] [CrossRef]
- Yu, A.M. Indolealkylamines: Biotransformations and potential drug-drug interactions. AAPS J. 2008, 10, 242–253. [Google Scholar] [CrossRef]
- Carpici, E.B.; Celik, N.; Bayram, G. Effects of salt stress on germination of some maize (Zea mays L.) cultivars. Afr. J. Biotechnol. 2009, 8, 4918–4922. [Google Scholar]
- Khodarahmpour, Z. Effects of NaCl salinity on maize (Zea mays L.) at germination and early seedling stage. Afr. J. Biotechnol. 2011, 11, 298–303. [Google Scholar] [CrossRef]
- Akram, M.; Ashraf, M.Y.; Ahmad, R.; Waraich, E.A.; Iqbal, J.; Mohsan, M. Screening for salt tolerance in maize (Zea mays L.) hybrids at an early seedling stage. Pak. J. Bot. 2010, 42, 141–154. [Google Scholar]
- Aliu, S.; Rusinovci, I.; Fetahu, S.; Gashi, B.; Simeonovska, E.; Rozman, L. The effect of salt stress on the germination of maize (Zea mays L.) seeds and photosynthetic pigments. Acta Agric. Slov. 2015, 105, 85–94. [Google Scholar] [CrossRef]
- Yuan, G.; Wang, X.; Guo, R.; Wang, Q. Effect of salt stress on phenolic compounds, glucosinolates, myrosinase and antioxidant activity in radish sprouts. Food Chem. 2010, 121, 1014–1019. [Google Scholar] [CrossRef]
- Guo, L.; Yang, R.; Wang, Z.; Guo, Q.; Gu, Z. Effect of NaCl stress on health-promoting compounds and antioxidant activity in the sprouts of three broccoli cultivars. Int. J. Food Sci. Nutr. 2014, 65, 476–481. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific Opinion on Dietary Reference Values for selenium. EFSA J. 2014, 12. [Google Scholar] [CrossRef]
- Nothstein, A.K.; Eiche, E.; Riemann, M.; Nick, P.; Winkel, L.H.E.; Göttlicher, J.; Steininger, R.; Brendel, R.; von Brasch, M.; Konrad, G.; et al. Tracking Se Assimilation and Speciation through the Rice Plant—Nutrient Competition, Toxicity and Distribution. PLoS ONE 2016, 11, e0152081. [Google Scholar] [CrossRef]
- Saha, U. Selenium in the Soil-Plant Environment: A Review. Int. J. Appl. Agric. Sci. 2017, 3, 1. [Google Scholar] [CrossRef]
- Hassini, I.; Baenas, N.; Moreno, D.A.; Carvajal, M.; Boughanmi, N.; Martinez Ballesta, M.D.C. Effects of seed priming, salinity and methyl jasmonate treatment on bioactive composition of Brassica oleracea var. capitata (white and red varieties) sprouts. J. Sci. Food Agric. 2017, 97, 2291–2299. [Google Scholar] [CrossRef]
- Roberts, S.K. Plasma membrane anion channels in higher plants and their putative functions in roots. New Phytol. 2006, 169, 647–666. [Google Scholar] [CrossRef]
- Demigné, C.; Sabboh, H.; Rémésy, C.; Meneton, P. Protective Effects of High Dietary Potassium: Nutritional and Metabolic Aspects. J. Nutr. 2004, 134, 2903–2906. [Google Scholar] [CrossRef]
- Soetan, K.O.; Olaiya, C.O.; Oyewole, O.E. The importance of mineral elements for humans, domestic animals and plants: A review. Afr. J. Food Sci. 2010, 4, 200–222. [Google Scholar]
- Tei, F.; Benincasa, P.; Guiducci, M. Critical nitrogen concentration in lettuce. In Proceedings of the Acta Horticulturae, Toronto, Canada, 11 August 2002; International Society for Horticultural Science: Leuven, Belgium, 2003; Volume 627, pp. 187–194. [Google Scholar]
- Federer, L.M.; Belter, C.W.; Joubert, D.J.; Livinski, A.; Lu, Y.L.; Snyders, L.N.; Thompson, H. Data sharing in PLoS ONE: An analysis of Data Availability Statements. PLoS ONE 2018, 13, e0194768. [Google Scholar] [CrossRef] [PubMed]
- Roldan, M.V.G.; Engel, B.; de Vos, R.C.H.; Vereijken, P.; Astola, L.; Groenenboom, M.; van de Geest, H.; Bovy, A.; Molenaar, J.; van Eeuwijk, F.; et al. Metabolomics reveals organ-specific metabolic rearrangements during early tomato seedling development. Metabolomics 2014, 10, 958–974. [Google Scholar] [CrossRef]
- Wang, S.; Alseekh, S.; Fernie, A.R.; Luo, J. The Structure and Function of Major Plant Metabolite Modifications. Mol. Plant. 2019, 12, 899–919. [Google Scholar] [CrossRef] [PubMed]
- Havaux, M. Carotenoid oxidation products as stress signals in plants. Plant. J. 2014, 79, 597–606. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Qiu, D.; Pang, Y.; Gao, H.; Wang, X.; Qin, Y. Diverse roles of tocopherols in response to abiotic and biotic stresses and strategies for genetic biofortification in plants. Mol. Breed. 2020, 40, 1–15. [Google Scholar] [CrossRef]
- Sharma, A.; Shahzad, B.; Rehman, A.; Bhardwaj, R.; Landi, M.; Zheng, B. Response of phenylpropanoid pathway and the role of polyphenols in plants under abiotic stress. Molecules 2019, 24, 2452. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Richter, A.; Jander, G. Beyond defense: Multiple functions of benzoxazinoids in maize metabolism. Plant. Cell Physiol. 2018, 59, 1528–1533. [Google Scholar] [CrossRef]
- Wouters, F.C.; Blanchette, B.; Gershenzon, J.; Vassão, D.G. Plant defense and herbivore counter-defense: Benzoxazinoids and insect herbivores. Phytochem. Rev. 2016, 15, 1127–1151. [Google Scholar] [CrossRef]
- Puccinelli, M.; Malorgio, F.; Terry, L.A.; Tosetti, R.; Rosellini, I.; Pezzarossa, B. Effect of selenium enrichment on metabolism of tomato (Solanum lycopersicum) fruit during postharvest ripening. J. Sci. Food Agric. 2019, 99, 2463–2472. [Google Scholar] [CrossRef]
- Borghesi, E.; González-Miret, M.L.; Escudero-Gilete, M.L.; Malorgio, F.; Heredia, F.J.; Meléndez-Martínez, A.J. Effects of salinity stress on carotenoids, anthocyanins, and color of diverse tomato genotypes. J. Agric. Food Chem. 2011, 59, 11676–11682. [Google Scholar] [CrossRef]
- Robbins, R.J.; Keck, A.-S.; Banuelos, G.; Finley, J.W. Cultivation Conditions and Selenium Fertilization Alter the Phenolic Profile, Glucosinolate, and Sulforaphane Content of Broccoli. J. Med. Food 2005, 8, 204–214. [Google Scholar] [CrossRef] [PubMed]
- Van Hoewyk, D.; Takahashi, H.; Inoue, E.; Hess, A.; Tamaoki, M.; Pilon-Smits, E.A.H. Transcriptome analyses give insights into selenium-stress responses and selenium tolerance mechanisms in Arabidopsis. Physiol. Plant. 2008, 132, 236–253. [Google Scholar] [CrossRef] [PubMed]
- Tamaoki, M.; Freeman, J.L.; Marquès, L.; Pilon-Smits, E.A.H. New insights into the roles of ethylene and jasmonic acid in the acquisition of selenium resistance in plants. Plant. Signal. Behav. 2008, 3, 865–867. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Cappa, J.J.; Harris, J.P.; Edger, P.P.; Zhou, W.; Pires, J.C.; Adair, M.; Unruh, S.A.; Simmons, M.P.; Schiavon, M.; et al. Transcriptome-wide comparison of selenium hyperaccumulator and nonaccumulator Stanleya species provides new insight into key processes mediating the hyperaccumulation syndrome. Plant. Biotechnol. J. 2018, 16, 1582–1594. [Google Scholar] [CrossRef]


| Treatment | Germination (%) | Lengths (mm) | Fresh Mass (mg) | Dry Matter (%) | |||
|---|---|---|---|---|---|---|---|
| Roots | Shoot | Roots | Shoot | Roots | Shoot | ||
| SeNONaCl0 | 99 (1.2) | 59 (14.9) | 26 (9.3) | 133 (33.6) | 83 (32.2) | 8.9 (2.25) | 11.9 (4.61) |
| SeNONaCl25 | 96 (3.5) | 65 (10.8) | 37 (7.2) | 147 (24.4) | 98 (19.1) | 9.1 (1.51) | 12.3 (2.39) |
| SeNONaCl50 | 94 (2.0) | 52 (17.3) | 32 (3.5) | 126 (41.9) | 103 (11.3) | 10.3 (3.43) | 11.8 (1.29) |
| SeYESNaCl0 | 97 (2.3) | 78 (22.8) | 39 (13.2) | 180 (52.6) | 99 (33.5) | 6.9 (2.02) | 11.6 (3.93) |
| SeYESNaCl25 | 99 (2.3) | 68 (19.5) | 35 (5.8) | 161 (46.2) | 109 (18.4) | 8.9 (2.55) | 11.9 (1.97) |
| SeYESNaCl50 | 93 (3.1) | 53 (16.5) | 26 (3.2) | 91 (28.3) | 84 (10.3) | 10.2 (3.18) | 15.4 (1.90) |
| F Test | |||||||
| Se fertilization (Se) | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. |
| NaCl treatment (NaCl) | n.s. | ** | * | n.s. | n.s. | n.s. | n.s. |
| Se X NaCl | n.s. | n.s. | * | n.s. | n.s. | n.s. | n.s. |
| LSD of the interaction | 3.44 | 15.22 | 6.75 | 69.6 | 40.39 | 4.58 | 5.21 |
| Treatments | Se Species (μg kg−1 DM) | TSeC | ||||||
|---|---|---|---|---|---|---|---|---|
| Inorganic Species | Organic Species | |||||||
| SeO32− | SeO42− | Total | SeCys2 | SeMeSeCys | SeMet | Total | ||
| Roots | ||||||||
| SeNONaCl0 | 172 (20) | 16 (1.2) | 188 (19.3) | 12 (2.2) | 6 (2.8) | 32 (3.8) | 52 (7.8) | 240 (14.2) |
| SeNONaCl25 | 160 (6.9) | 20 (1.6) | 180 (5.3) | 14 (2.0) | 17 (7.9) | 19 (4.1) | 47 (11.4) | 228 (16.7) |
| SeNONaCl50 | 167 (27.5) | 44 (9.8) | 211 (35.8) | 12 (0.7) | 16 (4.2) | 55 (8.4) | 141 (69.5) | 352 (73.1) |
| SeYESNaCl0 | 867 (87.2) | 37 (7.6) | 904 (92.1) | 81 (22.0) | 10 (5.3) | 139 (43.6) | 236 (92.5) | 1140 (179) |
| SeYESNaCl25 | 943 (160.5) | 82 (21.3) | 1025 (139.2) | 77 (19.1) | 13 (3) | 237 (60.0) | 382 (63.6) | 1407 (196.9) |
| SeYESNaCl50 | 876 (136.2) | 29 (20.5) | 904 (156.1) | 131 (13.6) | 14 (2.9) | 226 (24.8) | 317 (39.5) | 1221 (190.3) |
| F Test | ||||||||
| Se fertilization | ** | ** | ** | ** | n.s. | ** | ** | ** |
| NaCl treatment | n.s. | ** | n.s. | ** | * | * | n.s. | n.s. |
| Se x NaCl | n.s. | ** | n.s. | ** | n.s. | n.s. | n.s. | n.s. |
| LSD of interaction | 167 | 9 | 169 | 23 | 8 | 76 | 100 | 197 |
| Shoots | ||||||||
| SeNONaCl0 | 221 (34.8) | 16 (0.65) | 237 (34.3) | 13 (0.7) | 9 (0.58) | 31 (1.8) | 52 (2.6) | 289 (35.9) |
| SeNONaCl25 | 279 (25.5) | 53 (10.9) | 332 (14.6) | 86 (11.2) | 14 (1.9) | 95 (25.8) | 195 (34.8) | 527 (48.3) |
| SeNONaCl50 | 168 (58.8) | 64 (3.4) | 232 (56.6) | 34 (12.2) | 8 (0.6) | 33 (3.5) | 75 (16.2) | 306 (53.6) |
| SeYESNaCl0 | 1512 (153.4) | 30 (2.7) | 1541 (153.4) | 94 (7.0) | 15 (0.8) | 104 (5.8) | 212 (2.2) | 1754 (153.2) |
| SeYESNaCl25 | 1512 (70.6) | 31 (0.7) | 1543 (69.9) | 112 (1.1) | 21 (2.2) | 63 (3.7) | 196 (5.6) | 1738 (64.3) |
| SeYESNaCl50 | 1489 (173.5) | 24 (1.5) | 1512 (174.7) | 96 (10.2) | 15 (1.2) | 93 (5.0) | 204 (11.8) | 1716 (162.9) |
| F Test | ||||||||
| Se fertilization | ** | ** | ** | ** | ** | ** | ** | ** |
| NaCl treatment | n.s. | * | n.s. | ** | ** | n.s. | n.s. | n.s. |
| Se x NaCl | n.s. | ** | n.s. | ** | n.s. | ** | n.s. | n.s. |
| LSD of interaction | 184 | 23 | 149 | 15 | 2 | 20 | 196 | 222 |
| Treatments | Mineral Concentrations (mg kg−1 DM) | |||||
|---|---|---|---|---|---|---|
| K | Na | Chlorides | Nitrates | Phosphates | Sulphates | |
| SeNONaCl0 | 853.0 (32.9) | 2011 (27) | 37.9 (5.4) | 1.86 (0.50) | 91.2 (15.5) | 14.5 (0.9) |
| SeNONaCl25 | 991.7 (82.2) | 9689 (1959) | 64.0 (5.6) | 63.9 (2.5) | 210.7 (40.9) | 44.2 (1.7) |
| SeNONaCl50 | 1576.7 (153.6) | 20988 (980) | 114.6 (11.0) | 2.9 (1.4) | 117.7 (70.2) | 21.9 (7.1) |
| SeYESNaCl0 | 1641.7 (520.9) | 2104 (164) | 30.4 (6.9) | 1.5 (0.8) | 122.9 (31.1) | 19.0 (8.8) |
| SeYESNaCl25 | 1490.7 (271.6) | 12176 (2972) | 50.6 (28.3) | 200.4 (2.4) | 233.3 (4.0) | 40.4 (1.4) |
| SeYESNaCl50 | 1667.0 (85.2) | 18443 (1727) | 95.5 (14.8) | 1.4 (0.2) | 36.0 (0.9) | 13.6 (0.6) |
| F Test | ||||||
| Se fertilization | ** | n.s. | n.s. | ** | n.s. | n.s. |
| NaCl treatment | * | ** | ** | ** | ** | ** |
| Se x NaCl | n.s. | n.s. | n.s. | ** | * | n.s. |
| LSD of interaction | 449.4 | 2963 | 25.7 | 2.8 | 64.2 | 8.4 |
| Class of Metabolites | Roots | Shoots | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SeYESNaCl0 | SeNONaCl25 | SeYESNaCl25 | SeNONaCl50 | SeYESNaCl50 | SeYESNaCl0 | SeNONaCl25 | SeYESNaCl25 | SeNONaCl50 | SeYESNaCl50 | |||||||||||||
| UP- | DW- | UP- | DW- | UP- | DW- | UP- | DW- | UP- | DW- | TOT | UP- | DW- | UP- | DW- | UP- | DW- | UP- | DW- | UP- | DW- | TOT | |
| Non polar | ||||||||||||||||||||||
| carotenoids | 4 | - | 3 | - | 3 | - | 2 | - | 4 | - | 16 | 1 | 3 | 9 | 6 | 9 | 5 | 4 | 4 | 3 | 3 | 47 |
| chlorophylls | 2 | - | - | 2 | - | 2 | 2 | 2 | 1 | 1 | 12 | - | - | 3 | 2 | 4 | 2 | 3 | 3 | 3 | 3 | 23 |
| tococromanols | 2 | 1 | 2 | - | 2 | - | 3 | - | 2 | - | 12 | 1 | 2 | 2 | 1 | 1 | 2 | - | 1 | 1 | 2 | 13 |
| quinones | - | 2 | - | - | - | - | - | - | - | 1 | 3 | - | - | 1 | - | 1 | - | - | - | - | - | 2 |
| lipids | 1 | 1 | 3 | 12 | 3 | 1 | 3 | 11 | 1 | 7 | 43 | 3 | 2 | 3 | 3 | 4 | 3 | 1 | 4 | 2 | 8 | 33 |
| Tot | 9 | 4 | 8 | 14 | 8 | 3 | 10 | 13 | 8 | 9 | 5 | 7 | 18 | 12 | 19 | 12 | 8 | 12 | 9 | 16 | ||
| Semipolar | ||||||||||||||||||||||
| acids | 1 | 1 | 2 | 2 | 1 | 1 | 2 | 2 | 1 | 2 | 15 | - | 1 | - | 1 | 2 | - | - | - | - | - | 4 |
| alkaloids | - | 3 | - | 4 | - | 1 | - | 1 | - | 1 | 10 | - | - | - | - | 1 | - | 1 | - | 1 | - | 3 |
| amides | - | 1 | - | 2 | - | - | - | 2 | - | 2 | 7 | - | 1 | - | 3 | 1 | 2 | - | 3 | - | 2 | 12 |
| amino acids and derivatives | 1 | 8 | 2 | 20 | 2 | 15 | 5 | 13 | 4 | 16 | 86 | - | 2 | 3 | 5 | 6 | 4 | 4 | 7 | 1 | 7 | 39 |
| anthocyanins | - | - | - | - | - | - | - | 1 | - | - | 1 | 1 | - | - | - | 1 | - | 1 | - | 1 | - | 4 |
| benzoxazinoids | 2 | 3 | - | 7 | 2 | 2 | 4 | 7 | 3 | 3 | 33 | 11 | 3 | 6 | 1 | 13 | 2 | 10 | 1 | 10 | - | 57 |
| flavonoids | 11 | 2 | 14 | 8 | 5 | 1 | 7 | 16 | 3 | - | 67 | 16 | - | 14 | - | 27 | - | 19 | - | 10 | - | 86 |
| nucleosides and nucleotides | - | 1 | 3 | 3 | 3 | 1 | 1 | 3 | 2 | 2 | 19 | 2 | 2 | - | 1 | 4 | - | 3 | 1 | 2 | 2 | 17 |
| others | - | - | - | - | - | - | 1 | - | - | - | 1 | - | - | 1 | - | 1 | - | - | - | - | 1 | 3 |
| other phenylpropanoids | 1 | - | - | - | - | - | - | 2 | 1 | - | 4 | - | - | - | - | 1 | - | - | - | - | - | 1 |
| peptides | - | - | 1 | 1 | 1 | 1 | 1 | 1 | 2 | 1 | 9 | - | - | - | - | 1 | - | 2 | - | 1 | - | 4 |
| phenolics precursors | - | 2 | 2 | 2 | 4 | 1 | 2 | 1 | 3 | 1 | 18 | 4 | - | 5 | - | 4 | - | 5 | 1 | 5 | 1 | 25 |
| sugars and phosphates | 1 | - | - | 2 | - | - | - | 2 | - | 2 | 7 | - | - | - | - | 1 | - | - | - | - | - | 1 |
| vitamins | 1 | - | 1 | 2 | - | 2 | 1 | 3 | - | 3 | 13 | 1 | 1 | - | 1 | 3 | - | 4 | 1 | 1 | 12 | |
| Tot | 18 | 21 | 25 | 53 | 18 | 25 | 24 | 54 | 19 | 33 | 35 | 10 | 29 | 12 | 66 | 8 | 49 | 13 | 32 | 14 | ||
| Class of Metabolites | Roots | Shoots | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SeYESNaCl0 | SeNONaCl25 | SeYESNaCl25 | SeNONaCl50 | SeYESNaCl50 | SeYESNaCl0 | SeNONaCl25 | SeYESNaCl25 | SeNONaCl50 | SeYESNaCl50 | |||||||||||
| UP- | DW- | UP- | DW- | UP- | DW- | UP- | DW- | UP- | DW- | UP- | DW- | UP- | DW- | UP- | DW- | UP- | DW- | UP- | DW- | |
| non polar pro-nutritional | 8 | 3 | 5 | 2 | 5 | 2 | 7 | 2 | 7 | 2 | 2 | 5 | 15 | 9 | 15 | 9 | 7 | 8 | 7 | 8 |
| up-/dw- ratio | 2.67 | 2.50 | 2.50 | 3.50 | 3.50 | 0.40 | 1.67 | 1.67 | 0.88 | 0.88 | ||||||||||
| non polar anti-nutritional | 0 | 1 | 0 | 8 | 0 | 0 | 0 | 9 | 0 | 6 | 0 | 2 | 0 | 1 | 0 | 3 | 0 | 3 | 0 | 7 |
| up-/dw- ratio | - | - | - | - | - | - | - | - | - | - | ||||||||||
| semipolar pro-nutritional | 18 | 17 | 25 | 47 | 18 | 24 | 23 | 51 | 19 | 30 | 35 | 9 | 28 | 9 | 63 | 6 | 48 | 10 | 31 | 11 |
| up-/dw- ratio | 1.06 | 0.53 | 0.75 | 0.45 | 0.63 | 3.89 | 3.11 | 10.50 | 4.80 | 2.82 | ||||||||||
| semipolar anti-nutritional | 0 | 4 | - | 6 | 0 | 1 | 0 | 3 | 0 | 3 | 0 | 1 | 0 | 3 | 2 | 2 | 1 | 3 | 1 | 2 |
| up-/dw- ratio | - | - | - | - | - | - | - | 1.00 | 0.33 | 0.50 | ||||||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Benincasa, P.; D’Amato, R.; Falcinelli, B.; Troni, E.; Fontanella, M.C.; Frusciante, S.; Guiducci, M.; Beone, G.M.; Businelli, D.; Diretto, G. Grain Endogenous Selenium and Moderate Salt Stress Work as Synergic Elicitors in the Enrichment of Bioactive Compounds in Maize Sprouts. Agronomy 2020, 10, 735. https://doi.org/10.3390/agronomy10050735
Benincasa P, D’Amato R, Falcinelli B, Troni E, Fontanella MC, Frusciante S, Guiducci M, Beone GM, Businelli D, Diretto G. Grain Endogenous Selenium and Moderate Salt Stress Work as Synergic Elicitors in the Enrichment of Bioactive Compounds in Maize Sprouts. Agronomy. 2020; 10(5):735. https://doi.org/10.3390/agronomy10050735
Chicago/Turabian StyleBenincasa, Paolo, Roberto D’Amato, Beatrice Falcinelli, Elisabetta Troni, Maria Chiara Fontanella, Sarah Frusciante, Marcello Guiducci, Gian Maria Beone, Daniela Businelli, and Gianfranco Diretto. 2020. "Grain Endogenous Selenium and Moderate Salt Stress Work as Synergic Elicitors in the Enrichment of Bioactive Compounds in Maize Sprouts" Agronomy 10, no. 5: 735. https://doi.org/10.3390/agronomy10050735
APA StyleBenincasa, P., D’Amato, R., Falcinelli, B., Troni, E., Fontanella, M. C., Frusciante, S., Guiducci, M., Beone, G. M., Businelli, D., & Diretto, G. (2020). Grain Endogenous Selenium and Moderate Salt Stress Work as Synergic Elicitors in the Enrichment of Bioactive Compounds in Maize Sprouts. Agronomy, 10(5), 735. https://doi.org/10.3390/agronomy10050735









