Impact and Control of Powdery Mildew on Irrigated Soybean Varieties Grown in Southeast Australia
Abstract
1. Introduction
2. Materials and Methods
2.1. Field Site
2.2. Experimental Design
2.3. Data Collection and Statistical Analysis
3. Results
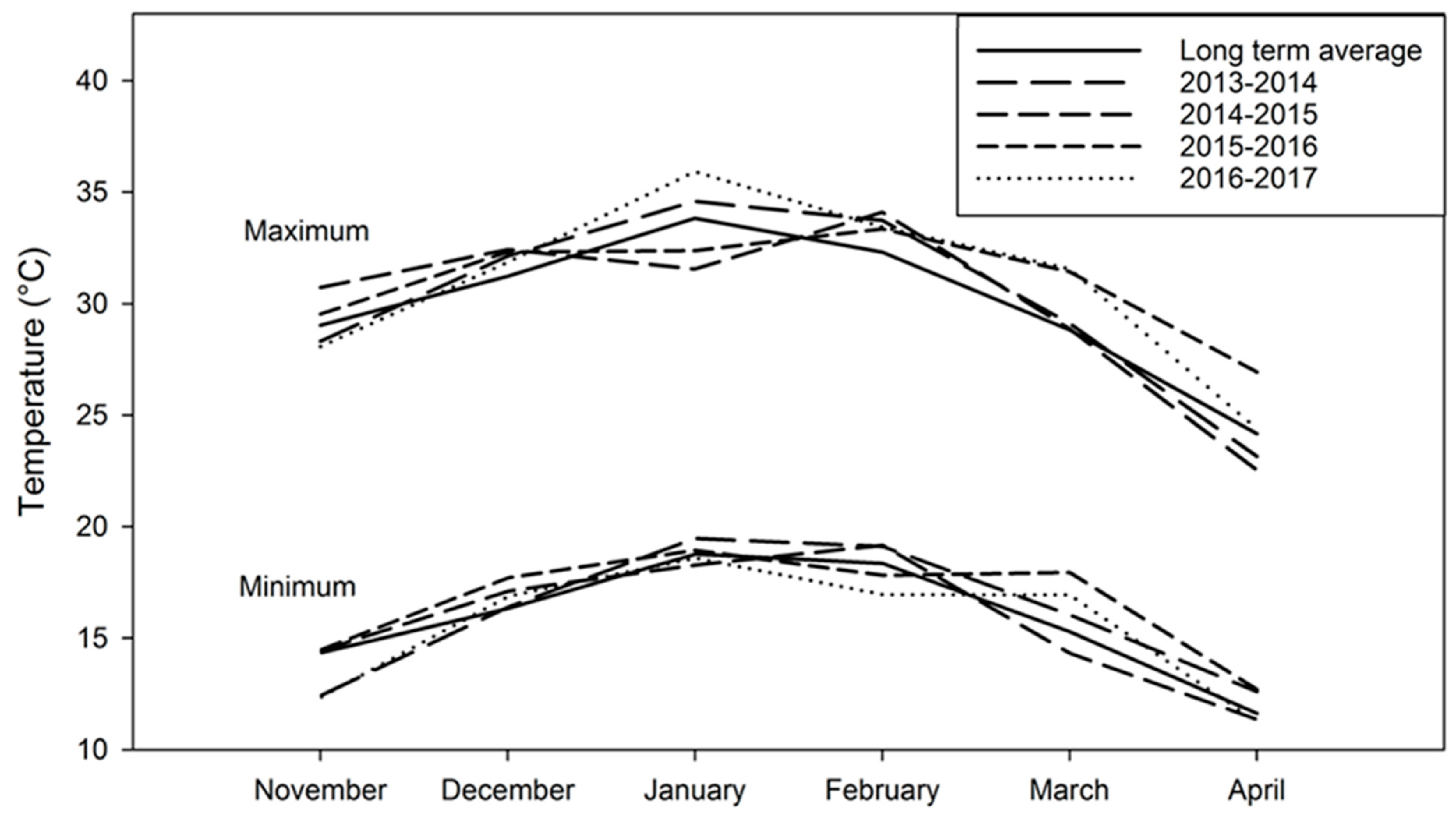
3.1. Temperature
3.2. Powdery Mildew Infection
3.3. Grain Yield and Other Agronomic Components
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dunleavy, J. Soybean seed yield losses caused by powdery mildew. Crop Sci. 1978, 18, 337–339. [Google Scholar] [CrossRef]
- McTaggart, A.R.; Ryley, M.J.; Shivas, R.G. First report of the powdery mildew Erysiphe diffusa on soybean in Australia. Australas. Plant Dis. Notes 2012, 7, 127–129. [Google Scholar] [CrossRef][Green Version]
- Phillips, D. Stability of Microsphaera diffusa and the effect of powdery mildew on yield of soybean. Plant Dis. 1984, 68, 953–956. [Google Scholar] [CrossRef]
- Ryley, M. Mildew Diseases of Soybean. 2015. Available online: http://www.australianoilseeds.com/__data/assets/pdf_file/0018/10368/Mildews_of_soybean_Feb_2015.pdf (accessed on 27 July 2016).
- Wrather, J.A.; Anderson, T.R.; Arsyad, D.M.; Tan, Y.; Ploper, L.D.; Porta-Puglia, A.; Ram, H.H.; Yorinori, J.T. Soybean disease loss estimates for the top ten soybean-producing counries in 1998. Can. J. Plant Pathol. 2001, 23, 115–121. [Google Scholar] [CrossRef]
- Yorinori, M.; Klingelfuss, L.; Paccola-Meirelles, L.; Yorinori, J. Effect of time of spraying of fungicide and foliar nutrient on soybean powdery mildew. J. Phytopathol. 2004, 152, 129–132. [Google Scholar] [CrossRef]
- Mignucci, J.S.; Lim, S. Powdery mildew development on soybeans with adult-plant resistance. Phytopathology 1980, 70, 919–921. [Google Scholar] [CrossRef]
- Gaynor, L.; O’Keeffe, K.; Lattimore, M. Southern Soybeans Shine in a Wet Year. 2012. Available online: http://irec.org.au/farmer_f/pdf_187/Soybean-success-in-southern-regions.pdf (accessed on 27 July 2016).
- Moore, N.; Serafin, L.; Jenkins, L. Summer Crop Production Guide. 2014. Available online: http://www.dpi.nsw.gov.au/__data/assets/pdf_file/0005/303485/Summer-crop-production-guide-2014.pdf (accessed on 27 July 2016).
- Rico, M.C. Interactions of Microsphaera diffusa with soybeans and other legumes. Phytopathology 1978, 68, 169–173. [Google Scholar]
- Mueller, J. Soybean Disease Control. In South Carolina Pest Management Handbook for Field Crops; Marshall, M., Ed.; Clemson Cooperative Extension: Clemson, SC, USA, 2015; pp. 256–260. [Google Scholar]
- Fletcher, R.A. Plant growth regulating properties of sterol-inhibiting fungicides. In Hormonal Regulation of Plant Growth and Development; Springer: Dordrecht, The Netherlands, 1987; pp. 103–113. [Google Scholar]
- Rademacher, W. Growth retardants: Effects on gibberellin biosynthesis and other metabolic pathways. Annu. Rev. Plant Biol. 2000, 51, 501–531. [Google Scholar] [CrossRef] [PubMed]
- Isbell, R. The Australian Soil Classification; CSIRO publishing: Collingwood, VIC, Australia, 1996. [Google Scholar]
- Fehr, W.R.; Caviness, C.E.; Burmood, D.T.; Pennington, J.S. Stage of Development Descriptions for Soybeans, Glycine max (L.) Merrill1. Crop Sci. 1971, 11, 929–931. [Google Scholar] [CrossRef]
- Bureau of Meteorology, AG, Climate Data Online. Australian Government, Bureau of Meteorology. Available online: www.bom.gov.au (accessed on 27 July 2016).
- Mignucci, J.S.; Boyer, J.S. Inhibition of photosynthesis and transpiration in soybean infected by Microsphaera diffusa. Phytopathology 1979, 69, 227–230. [Google Scholar] [CrossRef]
- Yulia, E.; Widiantini, F.; Karuniawan, A.; Comoc, R. Resistance Potential to Powdery Mildew (Microsphaera diffusa Cooke and Peck) of Several Yellow and Black Soybean (Glycine max (L.) Merr) Genotypes. KnE Life Sci. 2017, 9, 270–278. [Google Scholar] [CrossRef]
- Bowen, K.; Hagan, A.; Weeks, J. Number of tebuconazole applications for maximizing disease control and yield of peanut in growers’ fields in Alabama. Plant Dis. 1997, 81, 927–931. [Google Scholar] [CrossRef]
- Culbreath, A.; Kemerait, R., Jr.; Brenneman, T. Management of leaf spot diseases of peanut with prothioconazole applied alone or in combination with tebuconazole or trifloxystrobin. Peanut Sci. 2008, 35, 149–158. [Google Scholar] [CrossRef]
- Carmona, M.; Sautua, F.; Perelman, S.; Gally, M.; Reis, E.M. Development and validation of a fungicide scoring system for management of late season soybean diseases in Argentina. Crop Prot. 2015, 70, 83–91. [Google Scholar] [CrossRef]
| At R2 Growth Stage | Two Weeks after R2 | ||||
|---|---|---|---|---|---|
| Treatments | Application Regime | Rate (mL/ha) | Adjuvant 1 | Rate (mL/ha) | Adjuvant 1 |
| 2013–2014 | |||||
| Untreated control | – | – | – | – | – |
| Tebuconazole | Single 2 | 150 | 1% | – | – |
| Tebuconazole + prothioconazole | Single 2 | 400 | 1% | – | – |
| Tebuconazole + prothioconazole | Split 3 | 200 | 1% | 200 | 1% |
| 2014–2015, 2015–2016 and 2016–2017 | |||||
| Untreated control | – | – | – | – | – |
| Tebuconazole | Single 2 | 240 | 1% | – | – |
| Tebuconazole | Split 3 | 200 | 1% | 200 | 1% |
| Tebuconazole + prothioconazole | Single 2 | 400 | 1% | – | – |
| Tebuconazole + prothioconazole | Split 3 | 200 | 1% | 200 | 1% |
| Djakal | Snowy | N005A-80 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Treatments | 2013–2014 | 2014–2015 | 2015–2016 | 2016–2017 | 2013–2014 | 2014–2015 | 2015–2016 | 2016–2017 | 2013–2014 | 2014–2015 | 2015–2016 | 2016–2017 |
| Untreated control | 0 | 0 | 0 | 0 | 98 | 98 | 58 | 45 | 76 | 99 | 55 | 16 |
| Tebuconazole (single application) 1 | 0 | 0 | 0 | 0 | 62 | 74 | 16 | 4 | 54 | 54 | 10 | 0 |
| Tebuconazole (split application) | — 2 | 0 | 0 | 0 | — 2 | 6 | 1 | 1 | — 2 | 30 | 1 | 1 |
| Tebuconazole + prothioconazole (single application) | 0 | 0 | 0 | 0 | 15 | 0 | 0 | 0 | 6 | 5 | 3 | 1 |
| Tebuconazole + prothioconazole (split application) | 0 | 0 | 0 | 0 | 1 | 28 | 0 | 0 | 0 | 34 | 0 | 0 |
| l.s.d. (p = 0.05) | n.s. | n.s. | n.s. | n.s. | 16 | 14 | 9 | 7 | 16 | 14 | 9 | 7 |
| Djakal | Snowy | N005A-80 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Treatments | 2013–2014 | 2014–2015 | 2015–2016 | 2016–2017 | 2013–2014 | 2014–2015 | 2015–2016 | 2016–2017 | 2013–2014 | 2014–2015 | 2015–2016 | 2016–2017 |
| Untreated control | 3.46 | 3.56 | 3.20 | 3.60 | 3.37 | 2.23 | 2.82 | 3.08 | 3.25 | 3.83 | 3.63 | 3.52 |
| Tebuconazole (single application) 1 | 3.45 | 3.81 | 3.38 | 3.54 | 3.26 | 2.54 | 2.94 | 3.05 | 3.59 | 4.28 | 3.69 | 3.49 |
| Tebuconazole (split application) | — 2 | 3.92 | 3.22 | 3.42 | — 2 | 2.69 | 3.22 | 2.91 | — 2 | 4.20 | 3.77 | 3.73 |
| Tebuconazole + prothioconazole (single application) | 3.54 | 3.33 | 3.39 | 3.59 | 3.40 | 3.73 | 3.06 | 3.05 | 3.25 | 4.29 | 3.61 | 3.73 |
| Tebuconazole + prothioconazole (split application) | 3.13 | 3.55 | 3.30 | 3.77 | 3.70 | 2.77 | 2.96 | 3.34 | 3.55 | 4.05 | 3.56 | 3.75 |
| l.s.d. (p = 0.05) | n.s. | n.s. | n.s. | n.s. | n.s. | 0.46 | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dunn, M.W.; Gaynor, L.G. Impact and Control of Powdery Mildew on Irrigated Soybean Varieties Grown in Southeast Australia. Agronomy 2020, 10, 514. https://doi.org/10.3390/agronomy10040514
Dunn MW, Gaynor LG. Impact and Control of Powdery Mildew on Irrigated Soybean Varieties Grown in Southeast Australia. Agronomy. 2020; 10(4):514. https://doi.org/10.3390/agronomy10040514
Chicago/Turabian StyleDunn, Mathew W, and Luke G Gaynor. 2020. "Impact and Control of Powdery Mildew on Irrigated Soybean Varieties Grown in Southeast Australia" Agronomy 10, no. 4: 514. https://doi.org/10.3390/agronomy10040514
APA StyleDunn, M. W., & Gaynor, L. G. (2020). Impact and Control of Powdery Mildew on Irrigated Soybean Varieties Grown in Southeast Australia. Agronomy, 10(4), 514. https://doi.org/10.3390/agronomy10040514




