Present Status and Perspective on the Future Use of Aflatoxin Biocontrol Products
Abstract
1. Introduction
1.1. The Aflatoxin Problem
1.2. Technologies to Limit Aflatoxin Crop Contamination
1.3. Biocontrol as a Tool to Decrease Crop Aflatoxin Contamination
1.4. From Single to Multiple Isolates
2. Basis for Selection of Atoxigenic Isolates as Biocontrol Agents
2.1. Use of Native Fungi
2.2. Membership in VCGs That Are Completely Atoxigenic
2.3. Lesions in the Aflatoxin Biosynthesis Gene Cluster
2.4. Area-Wide Adaptation
2.5. Superior Competitiveness
2.6. Efficacy Trials
3. Registration of Products
4. Delivery Methods
4.1. Grains as Carriers and Nutritional Sources.
4.2. Use of Bioplastics
4.3. Alternative Substrates
5. Effectiveness in Crops Treated with Biocontrol Products
5.1. Effectiveness of Aflatoxin Biocontrol Agents in Different Crops
5.2. Area-Wide Treatment for Increased Efficacy
6. Biocontrol Performance in Future Scenarios
7. Losses to Bird, Insect, or Rodent Predation
8. Concerns Posed for Using Biocontrol Products
9. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Klich, M.A. Environmental and developmental factors influencing aflatoxin production by Aspergillus flavus and Aspergillus parasiticus. Mycoscience 2007, 48, 71–80. [Google Scholar] [CrossRef]
- JECFA. Safety Evaluation of Certain Contaminants in Food: Prepared by the Eighty-Third Meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA); WHO Food A; World Health Organization and Food and Agriculture Organization of the United Nations: Geneva, Switzerland, 2018; ISBN 9241660554. [Google Scholar]
- Bandyopadhyay, R.; Kumar, M.; Leslie, J.F. Relative severity of aflatoxin contamination of cereal crops in West Africa. Food Addit. Contam. 2007, 24, 1109–1114. [Google Scholar] [CrossRef]
- Amaike, S.; Keller, N.P. Aspergillus flavus. Annu. Rev. Phytopathol. 2011, 49, 107–133. [Google Scholar] [CrossRef] [PubMed]
- Ortega-Beltran, A.; Moral, J.; Puckett, R.; Morgan, D.; Cotty, P.J.; Michailides, T.J. Fungal communities associated with almond throughout crop development: Implications for aflatoxin biocontrol management in California. PLoS ONE 2018, 13, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Frisvad, J.C.; Hubka, V.; Ezekiel, C.N.; Hong, S.B.; Nováková, A.; Chen, A.J.; Arzanlou, M.; Larsen, T.O.; Sklenář, F.; Mahakarnchanakul, W.; et al. Taxonomy of Aspergillus section Flavi and their production of aflatoxins, ochratoxins and other mycotoxins. Stud. Mycol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Klich, M.A. Aspergillus flavus: The major producer of aflatoxin. Mol. Plant Pathol. 2007, 8, 713–722. [Google Scholar] [CrossRef]
- Azziz-Baumgartner, E.; Lindblade, K.; Gieseker, K.; Rogers, H.S.; Kieszak, S.; Njapau, H.; Schleicher, R.; McCoy, L.F.; Misore, A.; DeCock, K.; et al. Case-control study of an acute aflatoxicosis outbreak, Kenya, 2004. Environ. Health Perspect. 2005, 113, 1779–1783. [Google Scholar] [CrossRef]
- Bryden, W.L. Mycotoxin contamination of the feed supply chain: Implications for animal productivity and feed security. Anim. Feed Sci. Technol. 2012, 173, 134–158. [Google Scholar] [CrossRef]
- Monson, M.; Coulombe, R.; Reed, K. Aflatoxicosis: Lessons from toxicity and responses to aflatoxin B1 in poultry. Agriculture 2015, 5, 742–777. [Google Scholar] [CrossRef]
- RASFF: Food and Feed Safety Alerts. European Commission, 2019. Available online: https://webgate.ec.europa.eu/rasff-window/portal/?event=notificationsList&StartRow=1 (accessed on 10 November 2019).
- Wu, F. Global impacts of aflatoxin in maize: Trade and human health. World Mycotoxin J. 2015, 8, 137–142. [Google Scholar] [CrossRef]
- Van de Perre, E.; Jacxsens, L.; Lachat, C.; El Tahan, F.; De Meulenaer, B. Impact of maximum levels in European legislation on exposure of mycotoxins in dried products: Case of aflatoxin B1 and ochratoxin A in nuts and dried fruits. Food Chem. Toxicol. 2015, 75, 112–117. [Google Scholar] [CrossRef] [PubMed]
- Logrieco, A.F.; Miller, J.D.; Eskola, M.; Krska, R.; Ayalew, A.; Bandyopadhyay, R.; Battilani, P.; Bhatnagar, D.; Chulze, S.; De Saeger, S.; et al. The mycotox charter: Increasing awareness of, and concerted action for, minimizing mycotoxin exposure worldwide. Toxins 2018, 10, 149. [Google Scholar] [CrossRef] [PubMed]
- Ortega-Beltran, A.; Moral, J.; Picot, A.; Puckett, R.D.; Cotty, P.J.; Michailides, T.J. Atoxigenic Aspergillus flavus isolates endemic to almond, fig, and pistachio orchards in California with potential to reduce aflatoxin contamination in these crops. Plant Dis. 2019, 103, 905–912. [Google Scholar] [CrossRef] [PubMed]
- Lubanga, D.; Bii, B. KEBS Bans 5 Maize Flour Brands over High Aflatoxin Levels. Daily Nation. 2019. Available online: https://www.nation.co.ke/news/Kebs-bans-5-maize-flour-brands/1056-5343060-eb9k78/index.html (accessed on 9 November 2019).
- Mwakio, P. State to Destroy 9.4 Tonnes of Contaminated Maize in Mombasa. Stand Digit. 2019. Available online: https://www.standardmedia.co.ke/article/2001309356/state-to-destroy-9-4-tonnes-of-contaminated-maize-in-mombasa (accessed on 14 January 2020).
- Omulo, C. Rwanda Bans Seven Peanut Butter Brands from Kenya. Businiss Daily. 2019. Available online: https://www.businessdailyafrica.com/news/Rwanda-bans-peanut-butter-brands-Kenya/539546-5345834-nyxhrl/index.html (accessed on 12 November 2019).
- CGTM Africa. Rwanda, Uganda Ban Kenyan-Produced Peanut Butter. CGTN Africa. 2019. Available online: https://africa.cgtn.com/2019/11/14/rwanda-uganda-ban-kenyan-produced-peanut-butter-over-contamination-fears/ (accessed on 14 November 2019).
- Guan, S.; Gong, M.; Yin, Y.; Huang, R.; Ruan, Z.; Zhou, T.; Xie, M. Occurrence of mycotoxins in feeds and feed ingredients in China. J. Food Agric. Environ. 2011, 9, 163–167. [Google Scholar] [CrossRef]
- Probst, C.; Bandyopadhyay, R.; Cotty, P.J. Diversity of aflatoxin-producing fungi and their impact on food safety in sub-Saharan Africa. Int. J. Food Microbiol. 2014, 174, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Waliyar, F.; Osiru, M.; Ntare, B.R.; Kumar, K.V.K.; Sudini, H.; Traore, A.; Diarra, B. Post-harvest management of aflatoxin contamination in groundnut. World Mycotoxin J. 2015, 8, 245–252. [Google Scholar] [CrossRef]
- Njoroge, S.M.C.; Matumba, L.; Kanenga, K.; Siambi, M.; Waliyar, F.; Maruwo, J.; Machinjiri, N.; Monyo, E.S. Aflatoxin B1 levels in groundnut products from local markets in Zambia. Mycotoxin Res. 2017, 33, 113–119. [Google Scholar] [CrossRef]
- Seetha, A.; Munthali, W.; Msere, H.W.; Swai, E.; Muzanila, Y.; Sichone, E.; Tsusaka, T.W.; Rathore, A.; Okori, P. Occurrence of aflatoxins and its management in diverse cropping systems of central Tanzania. Mycotoxin Res. 2017, 33, 323–331. [Google Scholar] [CrossRef]
- Lindahl, J.F.; Kagera, I.N.; Grace, D. Aflatoxin M1 levels in different marketed milk products in Nairobi, Kenya. Mycotoxin Res. 2018, 34, 289–295. [Google Scholar] [CrossRef]
- Bandyopadhyay, R.; Ortega-Beltran, A.; Akande, A.; Mutegi, C.; Atehnkeng, J.; Kaptoge, L.; Senghor, L.A.; Adhikari, B.N.; Cotty, P.J. Biological control of aflatoxins in Africa: Current status and potential challenges in the face of climate change. World Mycotoxin J. 2016, 9, 771–789. [Google Scholar] [CrossRef]
- Leroy, J.L.; Sununtnasuk, C.; García-Guerra, A.; Wang, J.S. Low level aflatoxin exposure associated with greater linear growth in southern Mexico: A longitudinal study. Matern. Child Nutr. 2018, 14, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Klich, M.A. Identification of Common Aspergillus Species; Centraalbureau Voor Schimmelcultures: Utrecht, The Netherlands, 2002; p. 116. [Google Scholar]
- Cotty, P.J.; Probst, C.; Jaime-Garcia, R. Etiology and management of aflatoxin contamination. In Mycotoxins: Detection Methods, Management, Public Health and Agricultural Trade; Leslie, J.F., Bandyopadhayay, R., Visconti, A., Eds.; CAB International: Wallingford, UK, 2008; pp. 287–299. ISBN 9781845930820. [Google Scholar]
- Mahuku, G.; Nzioki, H.S.; Mutegi, C.; Kanampiu, F.; Narrod, C.; Makumbi, D. Pre-harvest management is a critical practice for minimizing aflatoxin contamination of maize. Food Control 2019, 96, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Abbas, H.K.; Accinelli, C.; Zablotowicz, R.M.; Abel, C.A.; Bruns, H.A.; Dong, Y.; Shier, W.T. Dynamics of mycotoxin and Aspergillus flavus levels in aging Bt and non-Bt corn residues under Mississippi no-till conditions. J. Agric. Food Chem. 2008, 56, 7578–7585. [Google Scholar] [CrossRef] [PubMed]
- Abbas, H.K.; Wilkinson, J.R.; Zablotowicz, R.M.; Accinelli, C.; Abel, C.A.; Bruns, H.A.; Weaver, M.A. Ecology of Aspergillus flavus, regulation of aflatoxin production, and management strategies to reduce aflatoxin contamination of corn. Toxin Rev. 2009, 28, 142–153. [Google Scholar] [CrossRef]
- Doster, M.A.; Michailides, T.J. Aspergillus molds and aflatoxins in pistachio nuts in California. Phytopathology 1994, 84, 583–590. [Google Scholar] [CrossRef]
- Whitaker, T.B.; Dorner, J.W.; Lamb, M.; Slate, A.B. The effect of sorting farmers’ stock peanuts by size and color on partitioning aflatoxin into various shelled peanut grade sizes. Peanut Sci. 2005, 32, 103–118. [Google Scholar] [CrossRef]
- Matumba, L.; Van Poucke, C.; Njumbe Ediage, E.; Jacobs, B.; De Saeger, S. Effectiveness of hand sorting, flotation/washing, dehulling and combinations thereof on the decontamination of mycotoxin-contaminated white maize. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2015, 32, 960–969. [Google Scholar] [CrossRef]
- Peng, Z.; Chen, L.; Zhu, Y.; Huang, Y.; Hu, X.; Wu, Q.; Nüssler, A.K.; Liu, L.; Yang, W. Current major degradation methods for aflatoxins: A review. Trends Food Sci. Technol. 2018, 80, 155–166. [Google Scholar] [CrossRef]
- Campbell, B.C.; Molyneux, R.J.; Schatzki, T.F. Current research on reducing pre- and post-harvest aflatoxin contamination of U.S. almond, pistachio, and walnut. J. Toxicol. Toxin Rev. 2003, 22, 225–266. [Google Scholar] [CrossRef]
- Jaime-Garcia, R.; Cotty, P.J. Aspergillus flavus in soils and corncobs in south Texas: Implications for management of aflatoxins in corn-cotton rotations. Plant Dis. 2004, 88, 1366–1371. [Google Scholar] [CrossRef]
- Hell, K.; Fandohan, P.; Bandyopadhyay, R.; Kiewnick, S.; Sikora, R.; Cotty, P.J. Pre- and post-harvest management of aflatoxin in maize: An African perspective. In Mycotoxins: Detection Methods, Management, Public Health and Agricultural Trade; Leslie, J.F., Bandyopadhyay, R., Visconti, A., Eds.; CAB International: Wallingford, UK, 2008; p. 219. ISBN 9781845930820. [Google Scholar]
- Diao, E.; Dong, H.; Hou, H.; Zhang, Z.; Ji, N.; Ma, W. Factors influencing aflatoxin contamination in before and after harvest peanuts: A review. J. Food Res. 2014, 4, 148–154. [Google Scholar] [CrossRef]
- Ayalew, A.; Kimanya, M.; Matumba, L.; Bandyopadhyay, R.; Menkir, A.; Cotty, P.J. Controlling aflatoxins in maize in Africa: Strategies, challenges and opportunities for improvement. In Achieving Sustainable Cultivation of Maize. Volume 2: Cultivation Techniques, Pest and Disease Control; Watson, D., Ed.; Burleigh Dodds Science Publishing: Cambridge, MA, USA, 2017; pp. 1–24. [Google Scholar]
- Grubisha, L.C.; Cotty, P.J. Genetic isolation among sympatric vegetative compatibility groups of the aflatoxin-producing fungus Aspergillus flavus. Mol. Ecol. 2010, 19, 269–280. [Google Scholar] [CrossRef] [PubMed]
- Cotty, P.J.; Bayman, D.S.; Egel, D.S.; Elias, K.S. Agriculture, Aflatoxins and Aspergillus. In The Genus Aspergillus; Powell, K., Ed.; Plenum Press: New York, NY, USA, 1994; pp. 1–27. [Google Scholar]
- Cotty, P.J.; Jaime-Garcia, R. Influences of climate on aflatoxin producing fungi and aflatoxin contamination. Int. J. Food Microbiol. 2007, 119, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Adhikari, B.N.; Bandyopadhyay, R.; Cotty, P.J. Degeneration of aflatoxin gene clusters in Aspergillus flavus from Africa and North America. AMB Express 2016, 6, 62. [Google Scholar] [CrossRef]
- Atehnkeng, J.; Donner, M.; Ojiambo, P.S.; Ikotun, B.; Augusto, J.; Cotty, P.J.; Bandyopadhyay, R. Environmental distribution and genetic diversity of vegetative compatibility groups determine biocontrol strategies to mitigate aflatoxin contamination of maize by Aspergillus flavus. Microb. Biotechnol. 2016, 9, 75–88. [Google Scholar] [CrossRef] [PubMed]
- Agbetiameh, D.; Ortega-Beltran, A.; Awuah, R.T.; Atehnkeng, J.; Islam, M.-S.; Callicott, K.A.; Cotty, P.J.; Bandyopadhyay, R. Potential of atoxigenic Aspergillus flavus vegetative compatibility groups associated with maize and groundnut in Ghana as biocontrol agents for aflatoxin management. Front. Microbiol. 2019, 10, 2069. [Google Scholar] [CrossRef] [PubMed]
- Cotty, P.J.; Antilla, L.; Wakelyn, P.J. Competitive exclusion of aflatoxin producers: Farmer driven research and development. In Biological Control: A Global Perspective; Vincent, C., Goettel, N., Lazarovitis, G., Eds.; CAB International: Oxfordshire, UK, 2007; pp. 241–253. [Google Scholar]
- Mehl, H.L.; Jaime, R.; Callicott, K.A.; Probst, C.; Garber, N.P.; Ortega-Beltran, A.; Grubisha, L.C.; Cotty, P.J. Aspergillus flavus diversity on crops and in the environment can be exploited to reduce aflatoxin exposure and improve health. Ann. N. Y. Acad. Sci. 2012, 1273, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Atehnkeng, J.; Ojiambo, P.S.; Cotty, P.J.; Bandyopadhyay, R. Field efficacy of a mixture of atoxigenic Aspergillus flavus Link: FR vegetative compatibility groups in preventing aflatoxin contamination in maize (Zea mays L.). Biol. Control 2014, 72, 62–70. [Google Scholar] [CrossRef]
- Doster, M.A.; Cotty, P.J.; Michailides, T.J. Evaluation of the atoxigenic Aspergillus flavus strain AF36 in pistachio orchards. Plant Dis. 2014, 98, 948–956. [Google Scholar] [CrossRef]
- Mauro, A.; Garcia-Cela, E.; Pietri, A.; Cotty, P.J.; Battilani, P. Biological control products for aflatoxin prevention in Italy: Commercial field evaluation of atoxigenic Aspergillus flavus active ingredients. Toxins 2018, 10, 30. [Google Scholar] [CrossRef]
- Bandyopadhyay, R.; Cardwell, K.F.; Ortega-Beltran, A.; Schulthess, F.; Meikle, W.; Setamou, M.; Cotty, P.J. Identifying and managing plant health risks for key African crops: Maize. In Critical Issues in Plant Health: 50 Years of Research in African Agriculture; Neuenschwander, P., Tamò, M., Eds.; Burleigh Dodds Science Publishing: Cambridge, UK, 2019; pp. 173–212. [Google Scholar] [CrossRef]
- Brown, R.L.; Cotty, P.; Cleveland, T.E. Reduction in aflatoxin content of maize by atoxigenic strains of Aspergillus flavus. J. Food Prot. 1991, 54, 623–626. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cotty, P.J. Virulence and cultural characteristics of two Aspergillus flavus strains pathogenic on cotton. Phytopathology 1989, 79, 808–814. [Google Scholar] [CrossRef]
- Cotty, P.J. Biocompetitive exclusion of toxigenic fungi. In The Mycotoxin Factbook; Barug, D., Bhatnagar, D., van Egdmond, H.P., van der Kamp, J.W., van Osenbruggen, W.A., Visconti, A., Eds.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2006; pp. 179–197. [Google Scholar]
- Environmental Protection Agency. Aspergillus Flavus AF36; Amendment to Make Label Changes; 2011. Available online: https://www3.epa.gov/pesticides/chem_search/ppls/071693-00001-20110505.pdf (accessed on 15 February 2020).
- Dorner, J.W. Development of biocontrol technology to manage aflatoxin contamination in peanuts. Peanut Sci. 2009, 36, 60–67. [Google Scholar] [CrossRef][Green Version]
- Lavkor, I.; Arioglu, H.; Var, I.; Oztemiz, S. Biological control of aflatoxigenic fungi on peanut: For the pre-harvest approach. Turkish J. Field Crop. 2019, 24, 21–27. [Google Scholar] [CrossRef]
- Schreurs, F.; Bandyopadhyay, R.; Kooyman, C.; Ortega-Beltran, A.; Akande, A.; Konlambigue, M.; Van den Bosch, N. Commercial products promoting plant health in African agriculture. In Critical Issues in Plant Health: 50 Years of Research in African Agriculture; Neuenschwander, P., Tamò, M., Eds.; Burleigh Dodds Science Publishing: Cambridge, UK, 2019; pp. 345–364. [Google Scholar] [CrossRef]
- Probst, C.; Bandyopadhyay, R.; Price, L.E.; Cotty, P.J. Identification of atoxigenic Aspergillus flavus isolates to reduce aflatoxin contamination of maize in Kenya. Plant Dis. 2011, 95, 212–218. [Google Scholar] [CrossRef]
- Ortega-Beltran, A.; Cotty, P.J. Frequent shifts in Aspergillus flavus populations associated with maize production in Sonora, Mexico. Phytopathology 2018, 108, 412–420. [Google Scholar] [CrossRef]
- Shenge, K.C.; Mehl, H.L.; Cotty, P.J. Interactions among active ingredients of a multi-isolate aflatoxin biocontrol product. Phytopathology 2017, 107, S5.131. [Google Scholar]
- Environmental Protection Agency: Receipt of Information under the Toxic Substances Control Act. Federal Register 69059. 2016; Volume 81. Available online: https://www.govinfo.gov/app/details/FR-2016-10-05/2016-24112 (accessed on 10 November 2019).
- Donner, M.; Lichtemberg, P.; Doster, M.A.; Picot, A.; Cotty, P.J.; Puckett, R.D.; Michailides, T.J. Community structure of Aspergillus flavus and A. parasiticus in major almond-producing areas of California, United States. Plant Dis. 2015, 99, 1161–1169. [Google Scholar] [CrossRef]
- Picot, A.; Doster, M.A.; Islam, M.S.; Callicott, K.A.; Ortega-Beltran, A.; Cotty, P.J.; Michailides, T.J. Distribution and incidence of atoxigenic Aspergillus flavus VCG in tree crop orchards in California: A strategy for identifying potential antagonists, the example of almonds. Int. J. Food Microbiol. 2018, 265, 55–64. [Google Scholar] [CrossRef]
- Molo, M.S.; Heiniger, R.W.; Boerema, L.; Carbone, I. Trial summary on the comparison of various non-aflatoxigenic strains of Aspergillus flavus on mycotoxin levels and yield in maize. Agron. J. 2019, 1–5. [Google Scholar] [CrossRef]
- Camiletti, B.X.; Torrico, A.K.; Fernanda Maurino, M.; Cristos, D.; Magnoli, C.; Lucini, E.I.; de la Paz Giménez Pecci, M. Fungal screening and aflatoxin production by Aspergillus section Flavi isolated from pre-harvest maize ears grown in two Argentine regions. Crop Prot. 2017, 92, 41–48. [Google Scholar] [CrossRef]
- Camiletti, B.X.; Moral, J.; Asensio, C.M.; Torrico, A.K.; Lucini, E.I.; Giménez-Pecci, M.D.L.P.; Michailides, T.J. Characterization of Argentinian endemic Aspergillus flavus isolates and their potential use as biocontrol agents for mycotoxins in maize. Phytopathology 2018, 108, 818–828. [Google Scholar] [CrossRef] [PubMed]
- García-Lopez, M.T.; Jaime, R.; Camiletti, B.X.; Ortega-Beltran, A.; Moral, J.; Michailides, T.J. Contaminación de aflatoxinas en frutos secos: Un problema emergente. Phytoma 2018, 302, 38–42. [Google Scholar]
- Zhou, W.; Hu, L.; Zhao, Y.; Wang, M.; Zhang, H. Inhibition of fungal aflatoxin B1 biosynthesis by diverse botanically-derived polyphenols. Trop. J. Pharm. Res. 2015, 14, 605–609. [Google Scholar] [CrossRef]
- Hulikunte Mallikarjunaiah, N.; Jayapala, N.; Puttaswamy, H.; Siddapura Ramachandrappa, N. Characterization of non-aflatoxigenic strains of Aspergillus flavus as potential biocontrol agent for the management of aflatoxin contamination in groundnut. Microb. Pathog. 2017, 102, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Pitt, J.; Manthong, C.; Siriacha, P.; Chotechaunmanirat, S.; Markwell, P. Studies on the biocontrol of aflatoxin in maize in Thailand. Biocontrol Sci. Technol. 2015. [Google Scholar] [CrossRef]
- Tran-Dinh, N.; Pitt, J.I.; Markwell, P.J. Selection of non-toxigenic strains of Aspergillus flavus for biocontrol of aflatoxins in maize in Thailand. Biocontrol Sci. Technol. 2014, 24, 652–661. [Google Scholar] [CrossRef]
- Savić, Z.; Dudaš, T.; Loc, M.; Grahovac, M.; Budakov, D.; Jajić, I.; Krstović, S.; Barošević, T.; Krska, R.; Sulyok, M.; et al. Biological control of aflatoxin in maize grown in Serbia. Toxins 2020, 12, 162. [Google Scholar] [CrossRef]
- Grubisha, L.C.; Cotty, P.J. Genetic analysis of the Aspergillus flavus vegetative compatibility group to which a biological control agent that limits aflatoxin contamination in U.S. crops belongs. Appl. Environ. Microbiol. 2015, 81, 5889–5899. [Google Scholar] [CrossRef]
- Mauro, A.; Battilani, P.; Cotty, P.J. Atoxigenic Aspergillus flavus endemic to Italy for biocontrol of aflatoxins in maize. BioControl 2015, 60, 125–134. [Google Scholar] [CrossRef]
- Yu, J.; Chang, P.K.; Cary, J.W.; Wright, M.; Bhatnagar, D.; Cleveland, T.E.; Payne, G.A.; Linz, J.E. Comparative mapping of aflatoxin pathway gene clusters in Aspergillus parasiticus and Aspergillus flavus. Appl. Environ. Microbiol. 1995, 61, 2365–2371. [Google Scholar] [CrossRef] [PubMed]
- Yu, J. Current understanding on aflatoxin biosynthesis and future perspective in reducing aflatoxin contamination. Toxins 2012, 4, 1024–1057. [Google Scholar] [CrossRef]
- Prieto, R.; Yousibova, G.L.; Woloshuk, C.P. Identification of aflatoxin biosynthesis genes by genetic complementation in an Aspergillus flavus mutant lacking the aflatoxin gene cluster. Appl. Environ. Microbiol. 1996, 62, 3567–3571. [Google Scholar] [CrossRef] [PubMed]
- Chang, P.K.; Horn, B.W.; Dorner, J.W. Sequence breakpoints in the aflatoxin biosynthesis gene cluster and flanking regions in nonaflatoxigenic Aspergillus flavus isolates. Fungal Genet. Biol. 2005, 42, 914–923. [Google Scholar] [CrossRef] [PubMed]
- Ehrlich, K.C.; Cotty, P.J. An isolate of Aspergillus flavus used to reduce aflatoxin contamination in cottonseed has a defective polyketide synthase gene. Appl. Microbiol. Biotechnol. 2004, 65, 473–478. [Google Scholar] [CrossRef] [PubMed]
- Tominaga, M.; Lee, Y.-H.; Hayashi, R.; Suzuki, Y.; Yamada, O.; Sakamoto, K.; Gotoh, K.; Akita, O. Molecular analysis of an inactive aflatoxin biosynthesis gene cluster in Aspergillus oryzae RIB strains. Appl. Environ. Microbiol. 2006, 72, 484–490. [Google Scholar] [CrossRef]
- Mehl, H.L.; Cotty, P.J. Variation in competitive ability among isolates of Aspergillus flavus from different vegetative compatibility groups during maize infection. Phytopathology 2010, 100, 150–159. [Google Scholar] [CrossRef]
- Mehl, H.L.; Cotty, P.J. Influence of plant host species on intraspecific competition during infection by Aspergillus flavus. Plant Pathol. 2013, 62, 1310–1318. [Google Scholar] [CrossRef]
- Sweany, R.R.; Damann, K.E.; Kaller, M.D. Comparison of soil and corn kernel Aspergillus flavus populations: Evidence for niche specialization. Phytopathology 2011, 101, 952–959. [Google Scholar] [CrossRef] [PubMed]
- Bandyopadhyay, R.; Atehnkeng, J.; Ortega-Beltran, A.; Akande, A.; Falade, T.D.O.; Cotty, P.J. “Ground-truthing” efficacy of biological control for aflatoxin mitigation in farmers’ fields in Nigeria: From field trials to commercial usage, a 10-year study. Front. Microbiol. 2019, 10, 2528. [Google Scholar] [CrossRef]
- Senghor, L.A.; Ortega-Beltran, A.; Atehnkeng, J.; Callicott, K.A.; Cotty, P.J.; Bandyopadhyay, R. The atoxigenic biocontrol product Aflasafe SN01 is a valuable tool to mitigate aflatoxin contamination of both maize and groundnut cultivated in Senegal. Plant Dis. 2019, 104, 510–520. [Google Scholar] [CrossRef] [PubMed]
- Ortega-Beltran, A.; Bandyopadhyay, R. Comments on “Trial summary on the comparison of various non-aflatoxigenic strains of Aspergillus flavus on mycotoxin levels and yield in maize” by M.S. Molo, et al. Agron. J. 111: 942–946 (2019). Agron. J. 2019, 111, 2625–2631. [Google Scholar] [CrossRef]
- Weaver, M.; Abbas, H.K. Field displacement of aflatoxigenic Aspergillus flavus strains through repeated biological control applications. Front. Microbiol. 2019, 10, 1788. [Google Scholar] [CrossRef] [PubMed]
- Dorner, J.W. Biological control of aflatoxin contamination of crops. J. Toxicol. Toxin Rev. 2004, 23, 425–450. [Google Scholar] [CrossRef]
- Dorner, J.W.; Cole, R.J.; Connick, W.J.; Daigle, D.J.; McGuire, M.R.; Shasha, B.S. Evaluation of biological control formulations to reduce aflatoxin contamination in peanuts. Biol. Control 2003, 26, 318–324. [Google Scholar] [CrossRef]
- Dorner, J.W.; Cole, R.J.; Blankenship, P.D. Use of a biocompetitive agent to control preharvest aflatoxin in drought stressed peanuts. J. Food Prot. 1992, 55, 888–892. [Google Scholar] [CrossRef]
- Bock, C.H.; Cotty, P.J. Wheat seed colonized with atoxigenic Aspergillus flavus: Characterization and production of a biopesticide for aflatoxin control. Biocontrol Sci. Technol. 1999, 9, 529–543. [Google Scholar] [CrossRef]
- Jaime, R.; Foley, M.; Barker, G.; Liesner, L.; Antilla, L.; Bandyopadhyay, R.; Cotty, P.J. Evaluation of residence, sporulation and efficacy of two formulations of the biocontrol Aspergillus flavus AF36 in commercial cotton fields in Arizona. Phytopathology 2014, 104, S3.55. [Google Scholar] [CrossRef]
- Accinelli, C.; Abbas, H.K.; Little, N.S.; Kotowicz, J.K.; Mencarelli, M.; Shier, W.T. A liquid bioplastic formulation for film coating of agronomic seeds. Crop Prot. 2016, 89, 123–128. [Google Scholar] [CrossRef]
- Accinelli, C.; Abbas, H.K.; Little, N.S.; Kotowicz, J.K.; Shier, W.T. Biological control of aflatoxin production in corn using non-aflatoxigenic Aspergillus flavus administered as a bioplastic-based seed coating. Crop Prot. 2018, 107, 87–92. [Google Scholar] [CrossRef]
- Okike, I.; Samireddypalle, A.; Kaptoge, L.; Fauquet, C.; Atehnkeng, J.; Bandyopadhyay, R.; Kulakow, P.; Duncan, A.; Alabi, T.; Blummel, M. Technical innovations for small-scale producers and households to process wet cassava peels into high quality animal feed ingredients and aflasafeTM substrate. Food Chain 2015, 5, 71–90. [Google Scholar] [CrossRef]
- Michailides, T.J.; Puckett, R.D.; Moral, J.; García-Lopez, M.T.; Felts, D.; Doster, L.; Jaime, R. Factors Affecting the Efficacy of AF36 Improvement of the Biocontrol Agent and Monitoring Commercial Application. 2018. Available online: https://americanpistachios.org/sites/default/files/inlinefiles/5.%20Factors%20Affecting%20the%20Efficacy%20of%20AF36%20Improvement%20of%20the%20Biocontrol%20Agent%20and%20Monitoring%20Commercial%20Applications__Edited%2001-04-2018.pdf (accessed on 15 January 2020).
- Ezekiel, C.N.; Ortega-Beltran, A.; Oyedeji, E.; Atehnkeng, J.; Kössler, P.; Tairu, F.; Hoeschle-Zeledon, I.; Karlovsky, P.; Cotty, P.J.; Bandyopadhyay, R. Aflatoxin in chili peppers in Nigeria: Extent of contamination and control using atoxigenic Aspergillus flavus genotypes as biocontrol agents. Toxins 2019, 11, 429. [Google Scholar] [CrossRef] [PubMed]
- Dorner, J.W. Simultaneous quantitation of Aspergillus flavus/A. parasiticus and aflatoxins in peanuts. J. AOAC Int. 2002, 85, 911–916. [Google Scholar] [CrossRef] [PubMed]
- Atehnkeng, J.; Ojiambo, P.S.; Ikotun, T.; Sikora, R.A.; Cotty, P.J.; Bandyopadhyay, R. Evaluation of atoxigenic isolates of Aspergillus flavus as potential biocontrol agents for aflatoxin in maize. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2008, 25, 1264–1271. [Google Scholar] [CrossRef] [PubMed]
- Nelson, M.R.; Orum, T.V.; Jaime-Garcia, R.; Nadeem, A. Applications of geographic information systems and geostatistics in plant disease epidemiology and management. Plant Dis. 1999, 83, 308–319. [Google Scholar] [CrossRef]
- Jaime, R.; Cotty, P.J. Temporal dynamic of Aspergillus flavus community structure in soils of fields treated with the atoxigenic biocontrol A. flavus AF36 in Arizona. Phytopathology 2013, 103, S2.66. [Google Scholar] [CrossRef]
- Bidartondo, M.I.; Ellis, C.; Kennedy, P.G.; Lilleskov, E.A.; Suz, L.M.; Andrew, C. Climate change: Fungal responses and effects. In State of the World’s Fungi; Kew Garden: London, UK, 2018; pp. 62–72. [Google Scholar]
- Alcamo, J.; Henrichs, T.; Rösch, T. World Water in 2025–Global Modeling and Scenario Analysis for the World Commission on Water for the 21st Century; Report A0002; Center for Environmental Systems Research, University of Kassel: Kassel, Germany, 2000; p. 65. [Google Scholar]
- Jaime-Garcia, R.; Cotty, P.J. Aflatoxin contamination of commercial cottonseed in south Texas. Phytopathology 2003, 93, 1190–1200. [Google Scholar] [CrossRef]
- Jaime-Garcia, R.; Cotty, P.J. Crop rotation and soil temperature influence the community structure of Aspergillus flavus in soil. Soil Biol. Biochem. 2010, 42, 1842–1847. [Google Scholar] [CrossRef]
- Ortega-Beltran, A.; Grubisha, L.C.; Callicott, K.A.; Cotty, P.J. The vegetative compatibility group to which the US biocontrol agent Aspergillus flavus AF36 belongs is also endemic to Mexico. J. Appl. Microbiol. 2016, 120, 986–998. [Google Scholar] [CrossRef]
- Islam, M.-S.; Callicott, K.A.; Mutegi, C.; Bandyopadhyay, R.; Cotty, P.J. Aspergillus flavus resident in Kenya: High genetic diversity in an ancient population primarily shaped by clonal reproduction and mutation-driven evolution. Fungal Ecol. 2018, 35, 20–33. [Google Scholar] [CrossRef]
- Horn, B.W. Sexual reproduction in Aspergillus flavus. Mycologia 2009, 101, 423–429. [Google Scholar] [CrossRef] [PubMed]
- Horn, B.W.; Ramirez-Prado, J.H.; Carbone, I. Sexual reproduction and recombination in the aflatoxin-producing fungus Aspergillus parasiticus. Fungal Genet. Biol. 2009, 46, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Olarte, R.A.; Horn, B.W.; Dorner, J.W.; Monacell, J.T.; Singh, R.; Stone, E.A.; Carbone, I. Effect of sexual recombination on population diversity in aflatoxin production by Aspergillus flavus and evidence for cryptic heterokaryosis. Mol. Ecol. 2012, 21, 1453–1476. [Google Scholar] [CrossRef] [PubMed]
- Moore, G.G.; Elliott, J.L.; Singh, R.; Horn, B.W.; Dorner, J.W.; Stone, E.A.; Chulze, S.N.; Barros, G.G.; Naik, M.K.; Wright, G.C.; et al. Sexuality generates diversity in the aflatoxin gene cluster: Evidence on a global scale. PLoS Pathog. 2013, 8. [Google Scholar] [CrossRef]
- Horn, B.W.; Sorensen, R.B.; Lamb, M.C.; Sobolev, V.S.; Olarte, R.A.; Worthington, C.J.; Carbone, I. Sexual reproduction in Aspergillus flavus sclerotia naturally produced in corn. Phytopathology 2014, 104, 75–85. [Google Scholar] [CrossRef]
- Njoroge, S.M.C. A critical review of aflatoxin contamination of peanuts in Malawi and Zambia: The past, present, and future. Plant Dis. 2018, 102, 2394–2406. [Google Scholar] [CrossRef]
- Ehrlich, K.C.; Moore, G.G.; Mellon, J.E.; Bhatnagar, D. Challenges facing the biological control strategy for eliminating aflatoxin contamination. World Mycotoxin J. 2015, 8, 225–233. [Google Scholar] [CrossRef]
- Kagot, V.; Okoth, S.; De Boevre, M.; De Saeger, S. Biocontrol of Aspergillus and Fusarium mycotoxins in Africa: Benefits and limitations. Toxins 2019, 11, 109. [Google Scholar] [CrossRef]
- Ndemera, M.; De Boevre, M.; De Saeger, S. Mycotoxin management in a developing country context: A critical review of strategies aimed at decreasing dietary exposure to mycotoxins in Zimbabwe. Crit. Rev. Food Sci. Nutr. 2020, 60, 529–540. [Google Scholar] [CrossRef]
- Pitt, J. The pros and cons of using biocontrol by competitive exclusion as a means for reducing aflatoxin in maize in Africa. World Mycotoxin J. 2019, 12, 103–112. [Google Scholar] [CrossRef]
- Savary, S.; Willocquet, L.; Pethybridge, S.J.; Esker, P.; McRoberts, N.; Nelson, A. The global burden of pathogens and pests on major food crops. Nat. Ecol. Evol. 2019, 3, 430–439. [Google Scholar] [CrossRef] [PubMed]
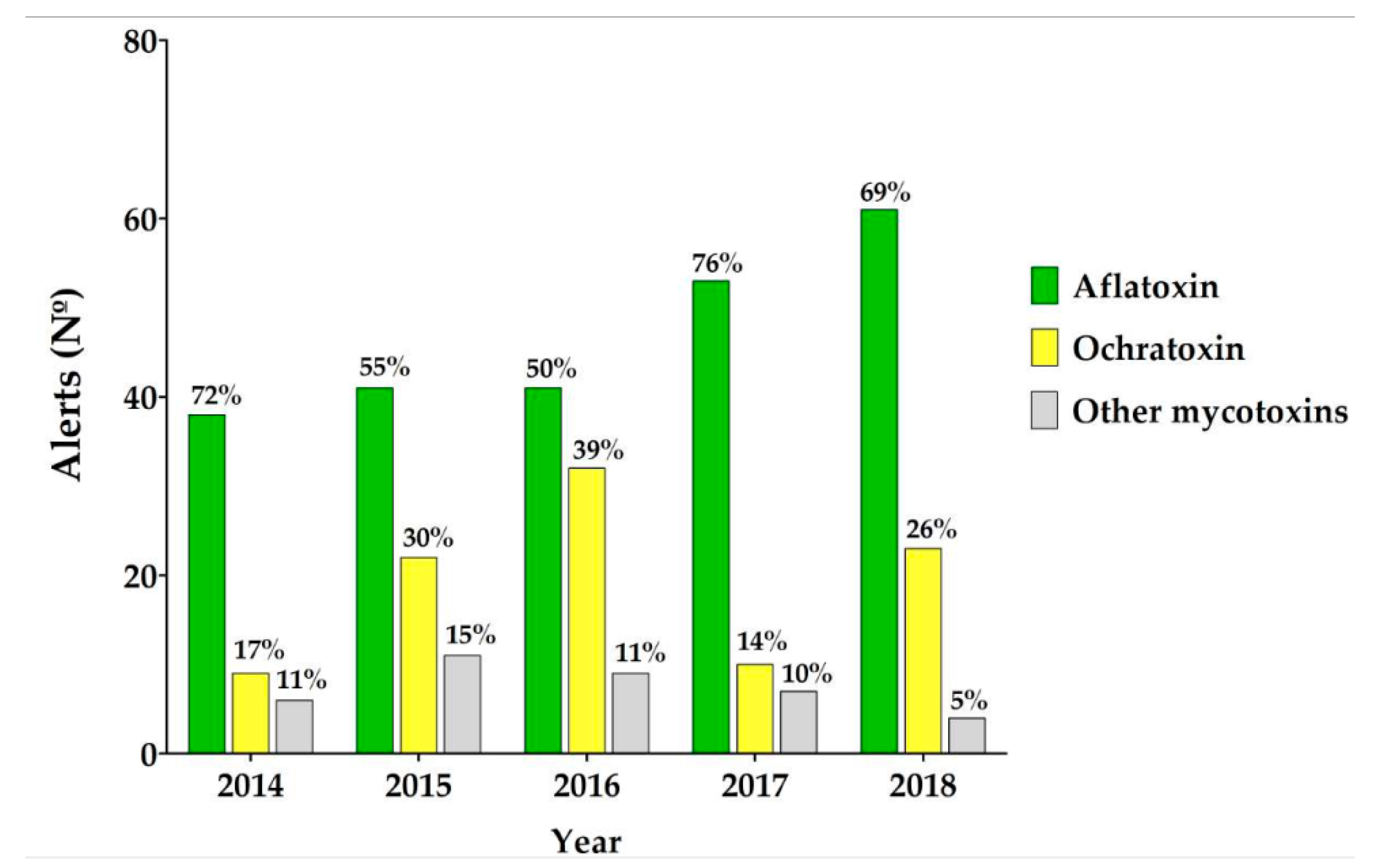
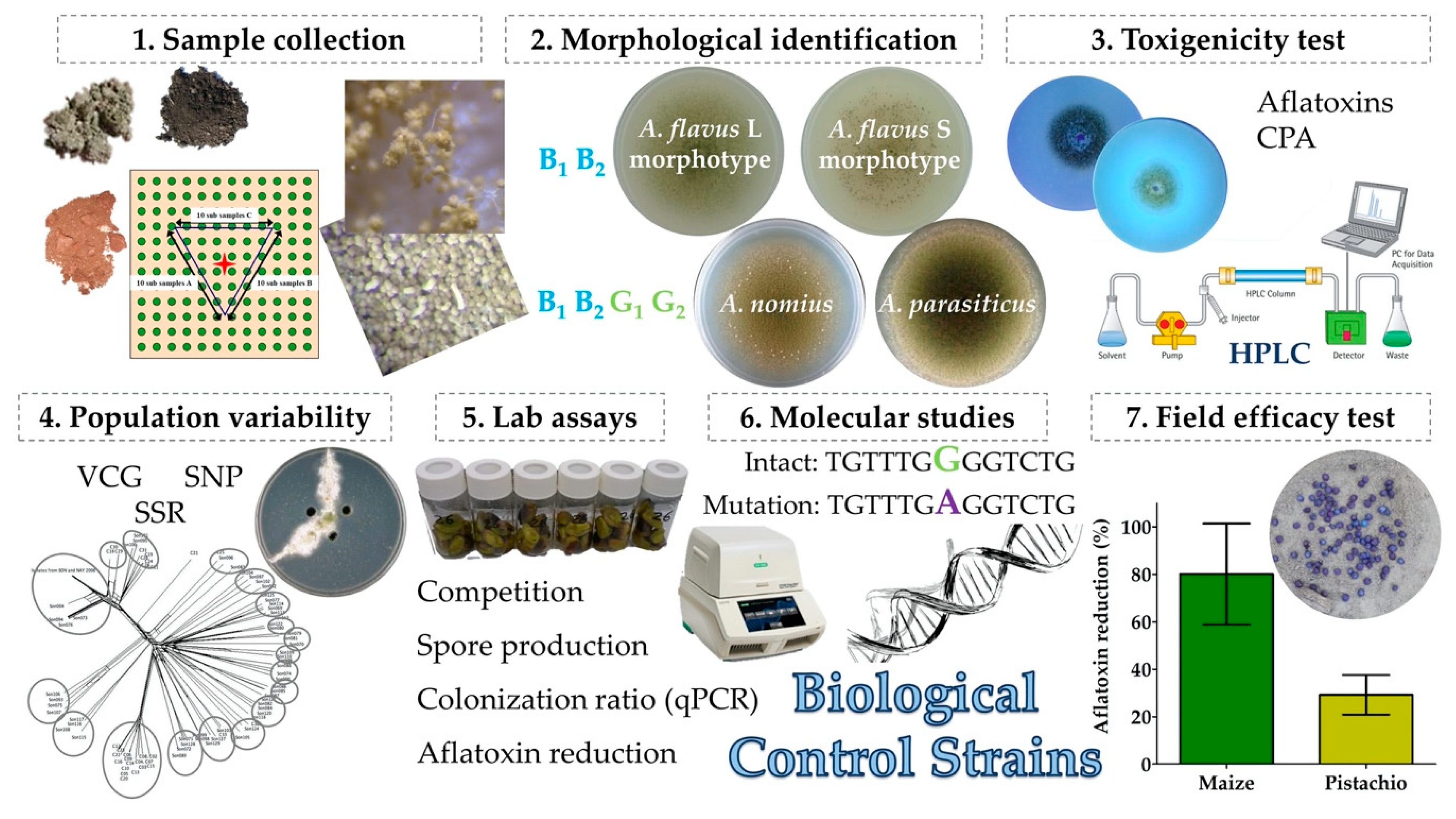
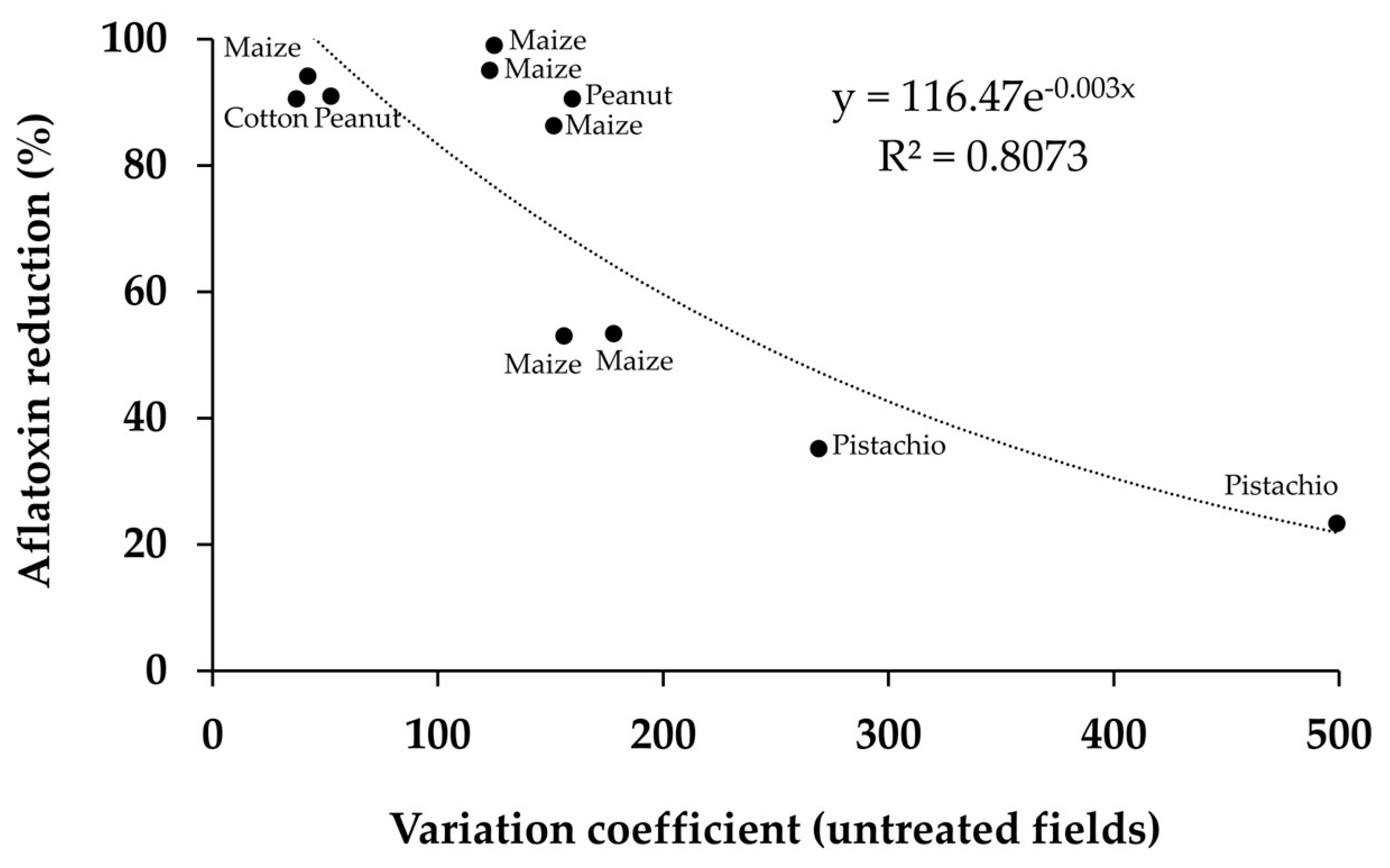
| Species | Aflatoxin B1 & B2 | Aflatoxin G1 & G2 | Aflatrem | CPA 2 | 3-Nit-Acid 3 | Tenuazonic Acid |
|---|---|---|---|---|---|---|
| A. aflatoxiformans | + | + | + | + | - | - |
| A. arachidicola | + | + | - | - | - | - |
| A. austwickii | + | + | + | + | - | - |
| A. cerealis | + | + | + | + | - | - |
| A. flavus | + | + 4 | + | + | + | - |
| A. luteovirescens | + | + | - | - | - | + |
| A. minisclerotigenes | + | + | + | + | - | - |
| A. mottae | + | + | - | + | - | - |
| A. nomius | + | + | - | - | - | + |
| A. novoparasiticus | + | + | - | - | - | - |
| A. parasiticus | + | + | - | - | - | - |
| A. pipericola | + | + | + | + | - | - |
| A. pseudocaelatus | + | + | - | + | - | + |
| A. pseudonomius | + | + | - | - | - | + |
| A. pseudotamarii | + | - | - | + | - | + |
| A. sergii | + | + | + | + | - | - |
| A. togoensis | + | - | - | - | - | - |
| A. transmontanensis | + | + | - | - | - | - |
| Product | Atoxigenic Aspergillus flavus Isolate(s) | Responsible Organization or Entity | Target Country | Crops for Use | Reference |
|---|---|---|---|---|---|
| Aspergillus flavus AF36 Prevail® | AF36 | Arizona Cotton Research and Protection Council | US | Cotton, maize, pistachio, almond, and figs | [5,48,49,51] |
| Afla-guard® | NRRL21882 | Syngenta® | US | Maize and groundnut | [91] |
| Aflasafe™ | Ka16127, La3279, La3304, Og0222 | IITA 3 | Nigeria | Maize and groundnut | [87] |
| Aflasafe KE01™ | C6-E, C8-F, E63-I, R7-H | IITA | Kenya | Maize | [45] |
| Aflasafe SN01 | M2-7, M21-11, Ms14-19, Ss19-14 | IITA | Senegal and The Gambia | Maize and groundnut | [45,88] |
| Aflasafe BF01 | M011-8, G018-2, M109-2, M110-7 | IITA | Burkina Faso | Maize and groundnut | [45] |
| Aflasafe GH01 | GHG079-4, GHG083-4, GHG321-2, GHM174-1 | IITA | Ghana | Maize, groundnut, and sorghum | [47] |
| Aflasafe GH02 | GHM511-3, GHM109-4, GHM001-5, GHM287-10 | IITA | Ghana | Maize, groundnut, and sorghum | [47] |
| Aflasafe TZ01 | TMS199-3, TMH104-9, TGS364-2, TMH 30-8 | IITA | Tanzania | Maize and groundnut | Unpublished registration document |
| Aflasafe TZ02 | TMS64-1, TGS55-6, TMS205-5, TMS137-3 | IITA | Tanzania | Maize and groundnut | Unpublished registration document |
| Aflasafe MWMZ01 2 | GP5G-8, GP1H-12, MZM594-1, MZM029-7 | IITA | Mozambique | Maize and groundnut | Unpublished registration document |
| Aflasafe MWMZ01 2 | MW199-1, MW097-8, MW246-2, MW238-2 | IITA | Malawi | Maize and groundnut | Unpublished registration document |
| Aflasafe MZ02 | GP5G-8, MZG071-6, MZM028-5, MZM250-8 | IITA | Mozambique | Maize and groundnut | Unpublished registration document |
| Aflasafe MW02 | MW258-6, MW332-10, MW248-11, MW204-7 | IITA | Malawi | Maize and groundnut | Unpublished registration document |
| Aflasafe ZM01 | 110MS-05, 38MS-03, 46MS-02, 03MS-10 | IITA | Zambia | Maize and groundnut | Unpublished registration document |
| Aflasafe ZM02 | 31MS-12, 12MS-10, 47MS-12, 64MS-03 | IITA | Zambia | Maize and groundnut | Unpublished registration document |
| AF-X1® | MUCL54911 | Pioneer® Int. | Italy | Maize | [52] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moral, J.; Garcia-Lopez, M.T.; Camiletti, B.X.; Jaime, R.; Michailides, T.J.; Bandyopadhyay, R.; Ortega-Beltran, A. Present Status and Perspective on the Future Use of Aflatoxin Biocontrol Products. Agronomy 2020, 10, 491. https://doi.org/10.3390/agronomy10040491
Moral J, Garcia-Lopez MT, Camiletti BX, Jaime R, Michailides TJ, Bandyopadhyay R, Ortega-Beltran A. Present Status and Perspective on the Future Use of Aflatoxin Biocontrol Products. Agronomy. 2020; 10(4):491. https://doi.org/10.3390/agronomy10040491
Chicago/Turabian StyleMoral, Juan, Maria Teresa Garcia-Lopez, Boris X. Camiletti, Ramon Jaime, Themis J. Michailides, Ranajit Bandyopadhyay, and Alejandro Ortega-Beltran. 2020. "Present Status and Perspective on the Future Use of Aflatoxin Biocontrol Products" Agronomy 10, no. 4: 491. https://doi.org/10.3390/agronomy10040491
APA StyleMoral, J., Garcia-Lopez, M. T., Camiletti, B. X., Jaime, R., Michailides, T. J., Bandyopadhyay, R., & Ortega-Beltran, A. (2020). Present Status and Perspective on the Future Use of Aflatoxin Biocontrol Products. Agronomy, 10(4), 491. https://doi.org/10.3390/agronomy10040491








