Oxytetracycline and Monensin Uptake by Tifton 85 Bermudagrass from Dairy Manure-Applied Soil
Abstract
1. Introduction
2. Materials and Methods
2.1. Site Description, Treatments and Greenhouse Conditions
2.2. Experimental Design
2.3. Monensin and Oxytetracycline Antibiotic Residue Analyses
2.4. Monensin Plant Tissue, Soil and Manure Extractions
2.5. Oxytetracycline Plant Tissue, Soil and Manure Extractions
2.6. ELISA Testing Protocol
2.7. Statistical Analysis
3. Results and Discussion
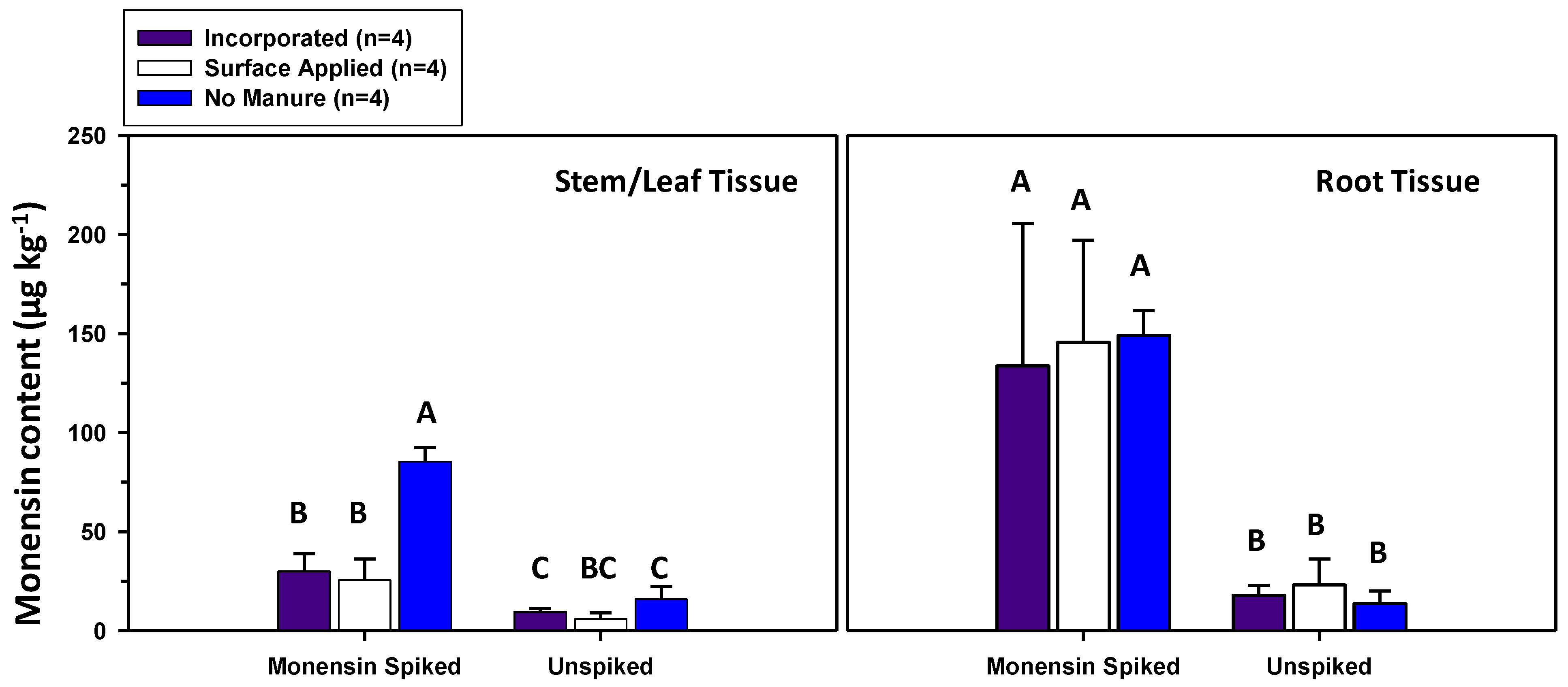
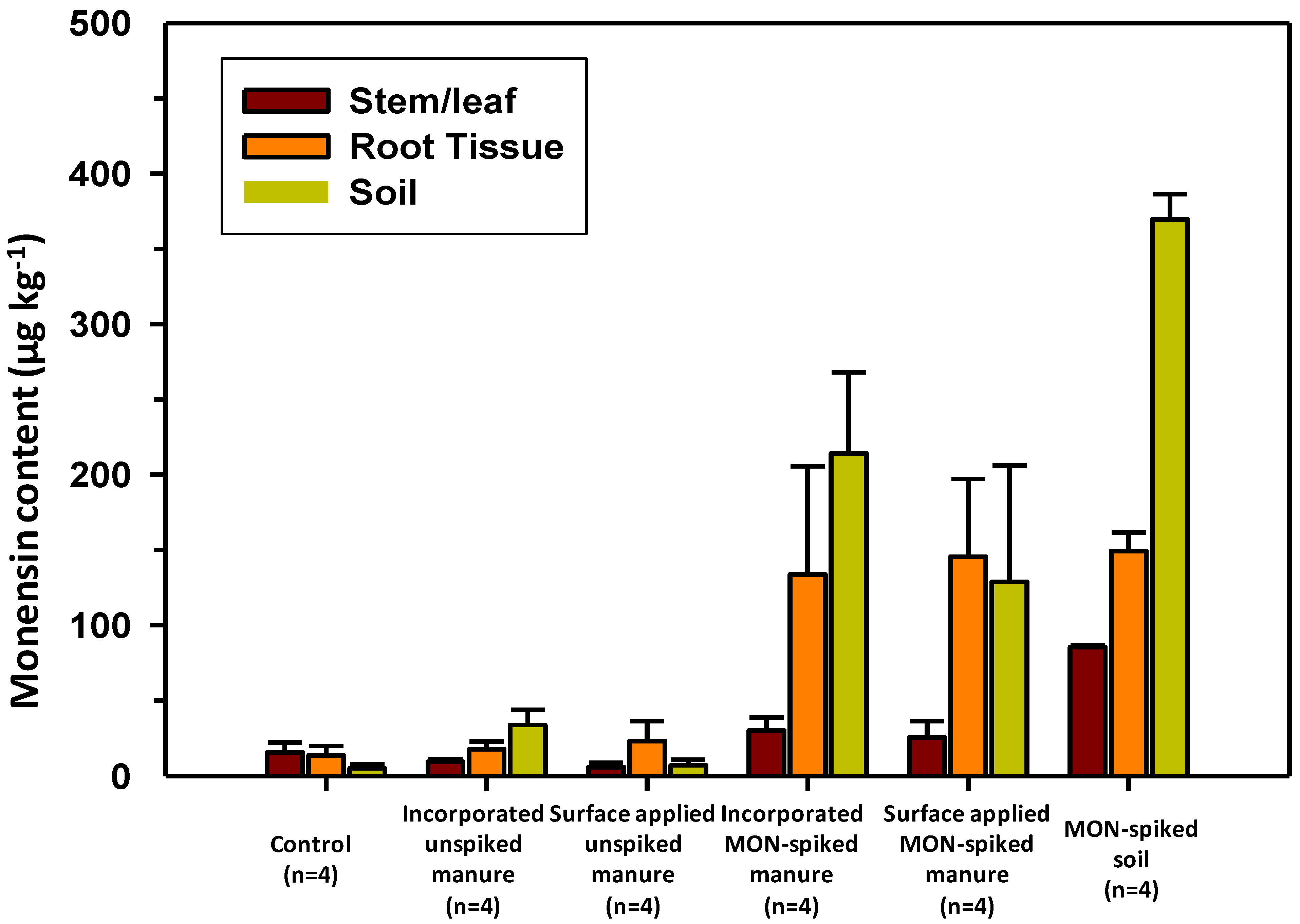
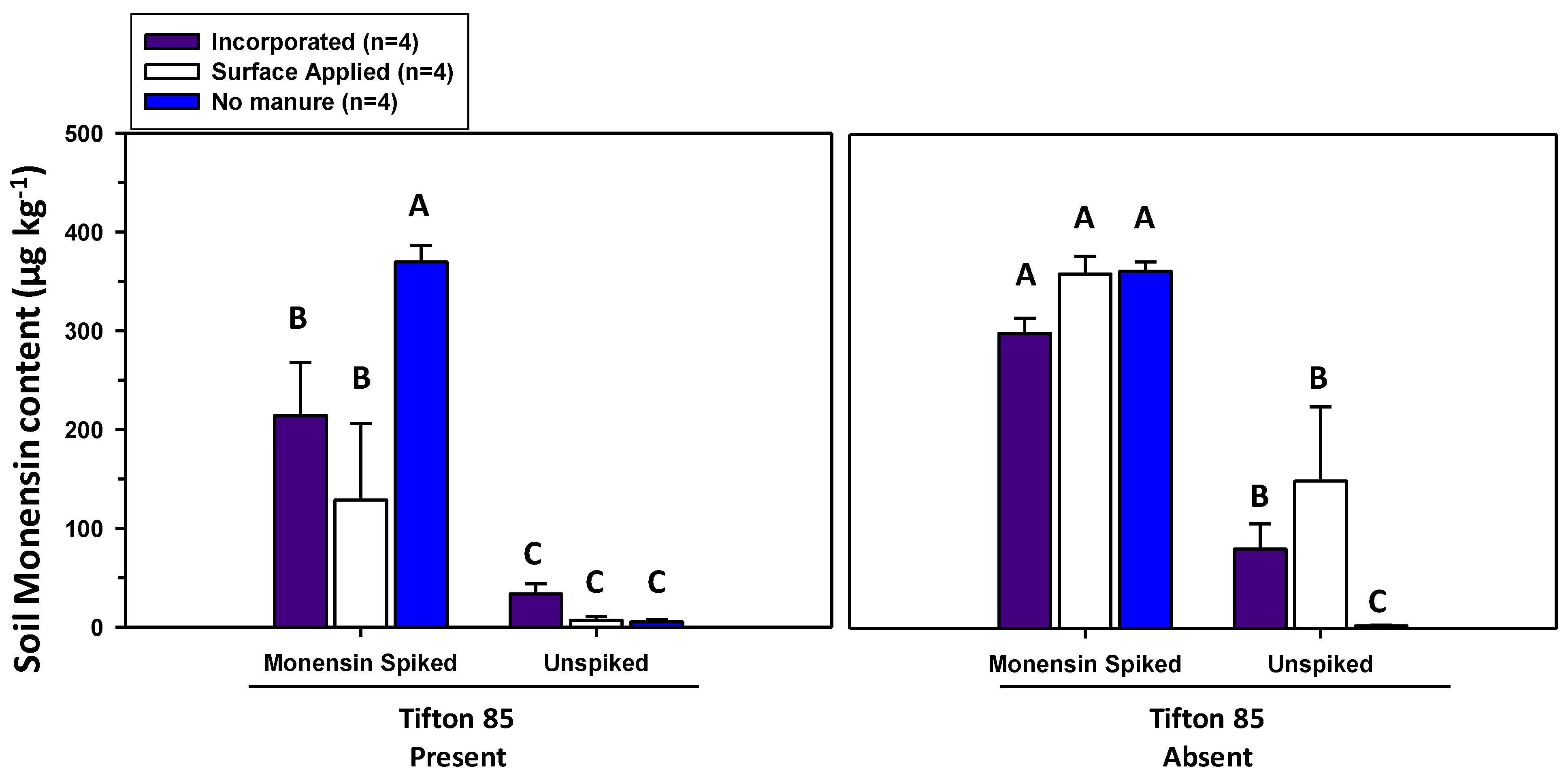
3.1. Monensin Content in Herbage and Soils
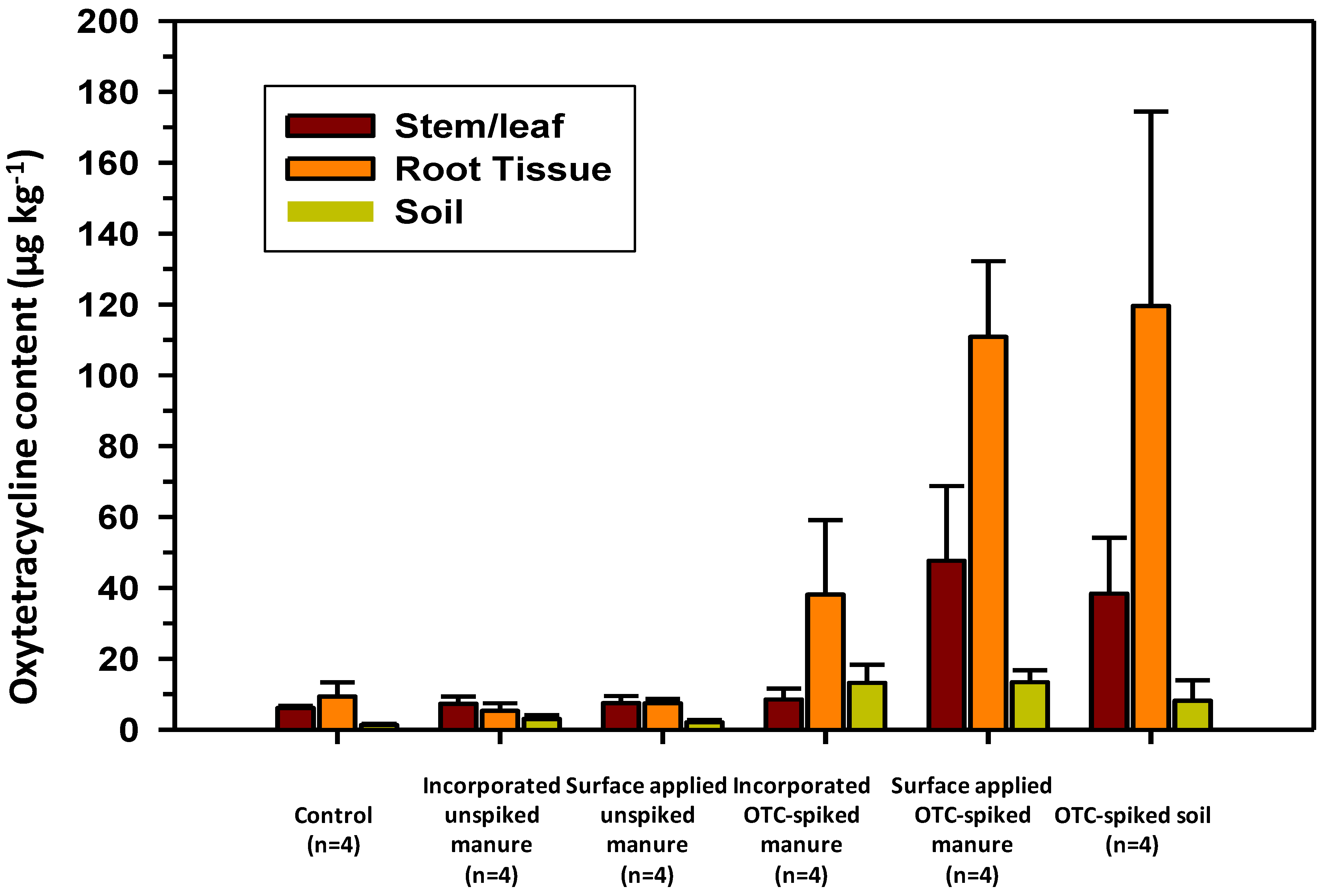
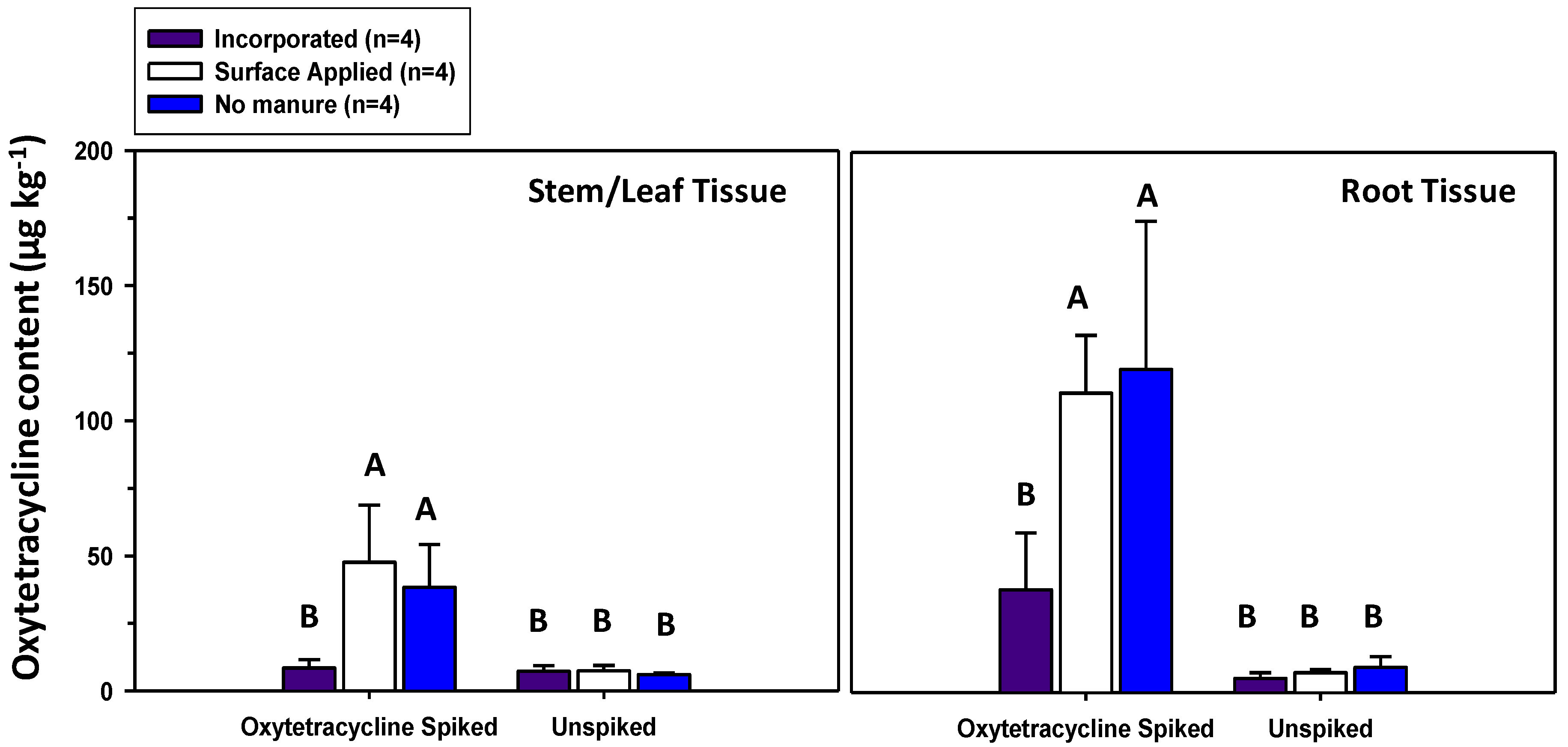
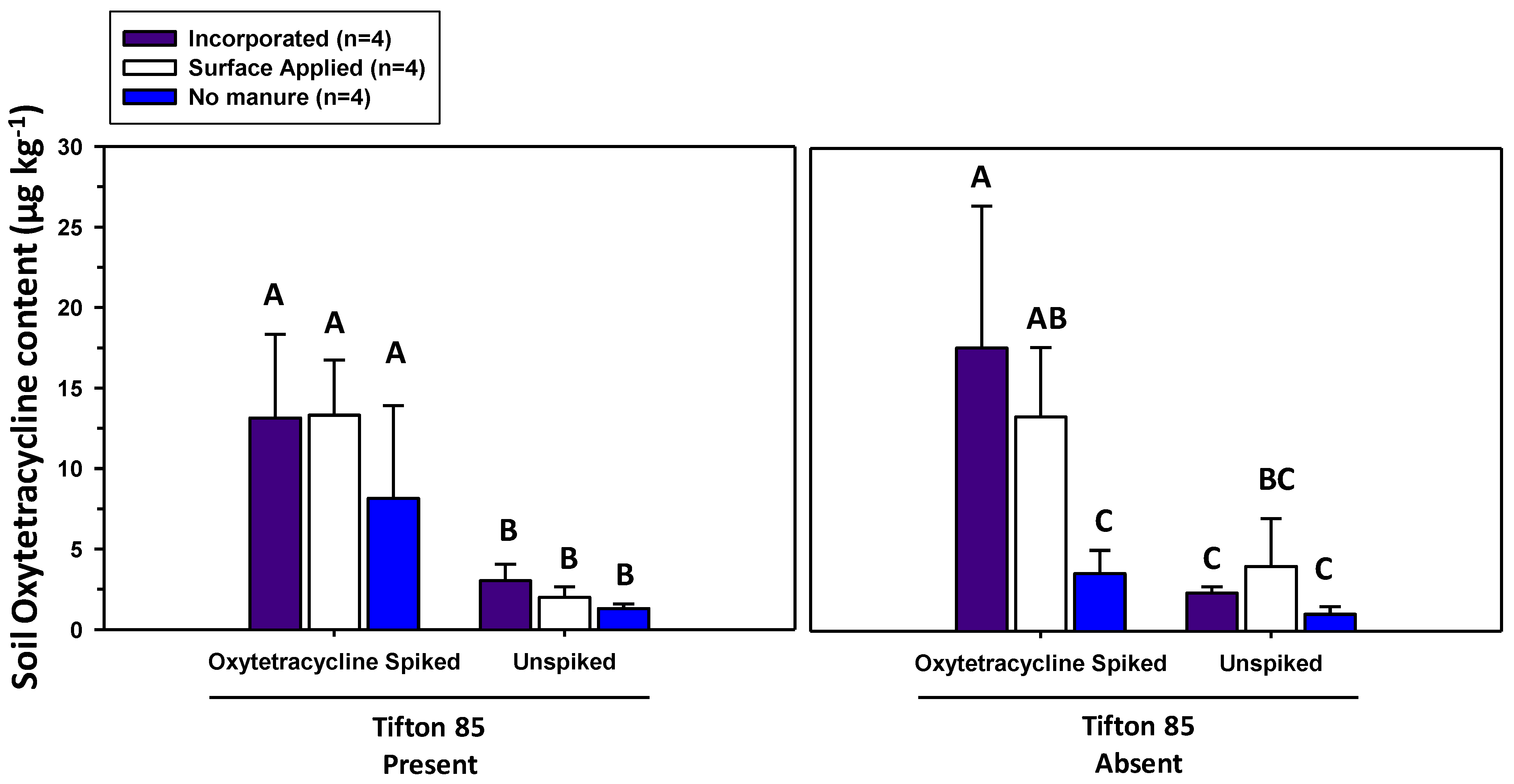
3.2. Oxytetracycline Contents in Soils and Herbage
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Huttner, A.; Harbarth, S.; Carlet, J.; Cosgrove, S.; Goossens, H.; Holmes, A.; Jarlier, V.; Voss, A.; Pittet, D. Antimicrobial resistance: A global view from the 2013 World Healthcare-Associated Infections Forum. Antimicrob Resist Infect Control 2013, 2, 31. [Google Scholar] [CrossRef] [PubMed]
- Silbergeld, E.K.; Graham, J.; Price, L.B. Industrial food animal production, antimicrobial resistance, and human health. Annu. Rev. Public Health 2008, 29, 151–169. [Google Scholar] [CrossRef] [PubMed]
- McGuffey, R.K.; Richardson, L.F.; Wilkinson, J.I.D. Ionophores for Dairy Cattle: Current Status and Future Outlook. J. Dairy Sci. 2001, 84, E194–E203. [Google Scholar] [CrossRef]
- Da Fonseca, M.P.; Da Costa Cruz Borges, A.L.; Reise Silva, R.; Lage, H.F.; Ferreira, A.L.; Lopes, F.C.F.; Pancoti, C.G.; Rodrigues, J.A.S. Intake, apparent digestibility, and methane emission in bulls receiving a feed supplement of monensin, virginiamycin, or a combination. Anim. Prod. Sci. 2016, 56, 1041–1045. [Google Scholar] [CrossRef]
- FDA. New Animal Drugs for Use in Animal Feed; Approval of New Animal Drug Applications; Withdrawal of Approval of New Animal Drug Applications; Food and Drug Administration: Rockville, MD, USA, 2016.
- Burkholder, J.; Libra, B.; Weyer, P.; Heathcote, S.; Kolpin, D.; Thorne, P.S.; Wichman, M. Impacts of waste from concentrated animal feeding operations on water quality. Environ. Health Perspect. 2007, 115, 308–312. [Google Scholar] [CrossRef]
- Massé, D.I.; Saady, N.M.C.; Gilbert, Y. Potential of biological processes to eliminate antibiotics in livestock manure: An overview. Animals 2014, 4, 146–163. [Google Scholar] [CrossRef]
- Kemper, N. Veterinary antibiotics in the aquatic and terrestrial environment. Ecol. Indic. 2008, 8, 1–13. [Google Scholar] [CrossRef]
- Lindsey, M.E.; Meyer, M.; Thurman, E.M. Analysis of trace levels of sulfonamide and tetracycline antimicrobials in groundwater and surface water using solid-phase extraction and liquid chromatography/mass spectrometry. Anal. Chem. 2001, 73, 4640–4646. [Google Scholar] [CrossRef]
- Sassman, S.A.; Lee, L.S. Sorption and Degradation in Soils of Veterinary Ionophore Antibiotics: Monensin and Lasalocid. Environ. Toxicol. Chem. 2007, 26, 1614–1621. [Google Scholar] [CrossRef]
- Wortmann, C.S.; Shapiro, C.A. The effects of manure application on soil aggregation. Nutr. Cycl. Agroecosystems 2008, 80, 173–180. [Google Scholar] [CrossRef]
- Xia, K.; Jeong, C.Y. Photodegradation of the Endocrine-Disrupting Chemical 4-Nonylphenol in Biosolids Applied to Soil. J. Environ. Qual. 2004, 33, 1568–1574. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Wang, H. Adsorption behavior of antibiotic in soil environment: A critical review. Front. Environ. Sci. Eng. 2015, 9, 565–574. [Google Scholar] [CrossRef]
- Thiele-Bruhn, S. Pharmaceutical antibiotic compounds in soils—A review. J. Plant Nutr. Soil Sci. 2003, 166, 145–167. [Google Scholar] [CrossRef]
- Pan, M.; Chu, L.M. Fate of antibiotics in soil and their uptake by edible crops. Sci. Total Environ. 2017, 599–600, 500–512. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Yates, S.R. Laboratory study of oxytetracycline degradation kinetics in animal manure and soil. J. Agric. Food Chem. 2008, 56, 1683–1688. [Google Scholar] [CrossRef]
- Pan, M.; Chu, L.M. Adsorption and degradation of five selected antibiotics in agricultural soil. Sci. Total Environ. 2016, 545–546, 48–56. [Google Scholar] [CrossRef]
- Klavarioti, M.; Mantzavinos, D.; Kassinos, D. Removal of residual pharmaceuticals from aqueous systems by advanced oxidation processes. Environ. Int. 2009, 35, 402–417. [Google Scholar] [CrossRef]
- Doydora, S.A.; Sun, P.; Cabrera, M.; Mantripragada, N.; Rema, J.; Pavlostathis, S.G.; Huang, C.H.; Thompson, A. Long-term broiler litter amendments can alter the soil’s capacity to sorb monensin. Environ. Sci. Pollut. Res. 2017, 24, 13466–13473. [Google Scholar] [CrossRef]
- Carlson, J.C.; Mabury, S.A. Dissipation kinetics and mobility of chlortetracycline, tylosin, and monensin in an agricultural soil in Northumberland County, Ontario, Canada. Environ. Toxicol. Chem. 2006, 1, 1–10. [Google Scholar] [CrossRef]
- Hu, X.; Zhou, Q.; Luo, Y. Occurrence and source analysis of typical veterinary antibiotics in manure, soil, vegetables and groundwater from organic vegetable bases, northern China. Environ. Pollut. 2010, 8, 2992–2998. [Google Scholar] [CrossRef]
- Boxall, A.B.A.; Johnson, P.; Smith, E.J.; Sinclair, C.J.; Stutt, E.; Levy, L.S. Uptake of veterinary medicines from soils into plants. J. Agric. Food Chem. 2006, 54, 2288–2297. [Google Scholar] [CrossRef] [PubMed]
- Chitescu, C.L.; Nicolau, A.I.; Stolker, A.A.M. Uptake of oxytetracycline, sulfamethoxazole and ketoconazole from fertilised soils by plants. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2013, 30, 1138–1146. [Google Scholar] [CrossRef] [PubMed]
- Kang, D.H.; Gupta, S.; Rosen, C.; Fritz, V.; Singh, A.; Chander, Y.; Murray, H.; Rohwer, C. Antibiotic uptake by vegetable crops from manure-applied soils. J. Agric. Food Chem. 2013, 61, 9992–10001. [Google Scholar] [CrossRef] [PubMed]
- Burton, G.W.; Gates, R.N.; Hill, G.M. Registration of “Tifton 85” Bermudagrasss. Crop Sci. 1993, 33, 644. [Google Scholar] [CrossRef]
- Kumar, K.; Gupta, S.C.; Baidoo, S.K.; Chander, Y.; Rosen, C.J. Antibiotic uptake by plants from soil fertilized with animal manure. J. Environ. Qual. 2005, 34, 2082–2085. [Google Scholar] [CrossRef]
- Brink, G.E.; Sistani, K.R.; Rowe, D.E. Nutrient uptake of hybrid and common bermudagrass fertilized with broiler litter. Agron. J. 2004, 96, 1509–1515. [Google Scholar] [CrossRef]
- Newton, G.L.; Bernard, J.K.; Hubbard, R.K.; Allison, J.R.; Lowrance, R.R.; Gascho, G.J.; Gates, R.N.; Vellidis, G. Managing Manure Nutrients Through Multi-crop Forage Production. J. Dairy Sci. 2003, 86, 2243–2252. [Google Scholar] [CrossRef]
- Sachi, S.; Ferdous, J.; Sikder, M.H.; Azizul Karim Hussani, S.M. Antibiotic residues in milk: Past, present, and future. J. Adv. Vet. Anim. Res. 2019, 6, 315–332. [Google Scholar] [CrossRef]
- Guerra, B.; Junker, E.; Schroeter, A.; Malorny, B.; Lehmann, S.; Helmuth, R. Phenotypic and genotypic characterization of antimicrobial resistance in German Escherichia coli isolates from cattle, swine and poultry. J. Antimicrob. Chemother. 2003, 57, 1210–1214. [Google Scholar] [CrossRef]
- Skočková, A.; Bogdanovičová, K.; Koláčková, I.; Karpíšková, R. Antimicrobial-resistant and extended-spectrum β-lactamase-producing Escherichia coli in raw cow’s milk. J. Food Prot. 2015, 78, 72–77. [Google Scholar] [CrossRef]
- Žižek, S.; Hrženjak, R.; Kalcher, G.T.; Šrimpf, K.; Šemrov, N.; Zidar, P. Does monensin in chicken manure from poultry farms pose a threat to soil invertebrates? Chemosphere 2011, 83, 517–523. [Google Scholar] [CrossRef] [PubMed]
- Granados-Chinchilla, F.; Arias-Andrés, M.D.J.; Fernández Montes de Oca, M.L.; Rodríguez, C. Effect of the veterinary ionophore monensin on the structure and activity of a tropical soil bacterial community. J. Environ. Sci. Health Part B Pestic. Food Contam. Agric. Wastes 2020, 55, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Cycoń, M.; Mrozik, A.; Piotrowska-Seget, Z. Antibiotics in the soil environment—Degradation and their impact on microbial activity and diversity. Front. Microbiol. 2019, 10, 338. [Google Scholar] [CrossRef] [PubMed]
- Hilaire, S.S.; Brady, J.A.; Muir, J.P.; Speshock, J.; Barbara, B. Changes in Soil and Plant Microbial Community Populations Following Administration of Manure Containing Oxytetracycline or Monensin. J. Soil Plant Biol. 2019, 2019, 94–105. [Google Scholar]
- Lee, L.S.; Carmosini, N.; Sassman, S.A.; Dion, H.M.; Sepulveda, M.S. Agricultural Contributions of Antimicrobials and Hormones on Soil and Water Quality. Adv. Agron. 2007, 93, 1–68. [Google Scholar]
- Stahnke, C.R.; Godfrey, C.L.; Moore, J.; Newman, J.S. Soils and Climate of the Texas A&M University Research and Extension Center at Stephenville in Relation to the Cross Timbers Land Resource Area; Texas Agricultural Experiment Station: College Station, TX, USA, 1980. [Google Scholar]
- Alekshun, M.N.; Levy, S.B. Molecular Mechanisms of Antibacterial Multidrug Resistance. Cell 2007, 128, 1037–1050. [Google Scholar] [CrossRef]
- Kumar, K.; Gupta, S.C.; Chander, Y.; Singh, A.K. Antibiotic Use in Agriculture and Its Impact on the Terrestrial Environment. Adv. Agron. 2005, 87, 1–54. [Google Scholar]
- Patten, D.K.; Wolf, D.C.; Kunkle, W.E.; Douglass, L.W. Effect of Antibiotics in Beef Cattle Feces on Nitrogen and Carbon Mineralization in Soil and on Plant Growth and Composition. J. Environ. Qual. 1980, 9, 167–172. [Google Scholar] [CrossRef]
- Donoho, A.L. Biochemical studies on the fate of monensin in animals and in the environment. J. Anim. Sci. 1984, 58, 1528–1539. [Google Scholar] [CrossRef]
- Reen, D. Enzyme-linked immunosorbent assay. Methods Mol. Biol. 1994, 32, 461–466. [Google Scholar]
- Aga, D.S.; Goldfish, R.; Kulshrestha, P. Application of ELISA in determining the fate of tetracyclines in land-applied livestock wastes. Analyst 2003, 128, 658–662. [Google Scholar] [CrossRef] [PubMed]
- Dolliver, H.; Kumar, K.; Gupta, S.; Singh, A. Application of enzyme-linked immunosorbent assay analysis for determination of monensin in environmental samples. J. Environ. Qual. 2008, 37, 1220–1226. [Google Scholar] [CrossRef] [PubMed]
- Dolliver, H.; Kumar, K.; Gupta, S. Sulfamethazine Uptake by Plants from Manure-Amended Soil. J. Environ. Qual. 2007, 36, 1224–1230. [Google Scholar] [CrossRef] [PubMed]
- SAS Institute Inc. JMP® 15 Basic Analysis; SAS Institute Inc.: Cary, NC, USA, 2019. [Google Scholar]
- Žižek, S.; Gombač, M.; Švara, T.; Pogačnik, M. Monensin—A review of factors influencing its presence in the environment and recommendations for safe storage and use of monensin-contaminated manure. Slov. Vet. Res. 2014, 51, 53–63. [Google Scholar]
- Yoshida, N.; Castro, M.J.L.; Cirelli, A.F. Degradation of monensin on soils: Influence of organic matter and water content. Chem. Ecol. 2010, 26, 27–33. [Google Scholar] [CrossRef]
- Dolliver, H.A.S.; Gupta, S.C. Antibiotic losses from unprotected manure stockpiles. J. Environ. Qual. 2008, 37, 1238–1244. [Google Scholar] [CrossRef]
- Miller, E.L.; Nason, S.L.; Karthikeyan, K.G.; Pedersen, J.A. Root Uptake of Pharmaceuticals and Personal Care Product Ingredients. Environ. Sci. Technol. 2016, 50, 525–541. [Google Scholar] [CrossRef]
- Dolliver, H.; Gupta, S.; Noll, S. Antibiotic degradation during manure composting. J. Environ. Qual. 2008, 37, 1245–1253. [Google Scholar] [CrossRef]
- Chopra, I.; Roberts, M. Tetracycline Antibiotics: Mode of Action, Applications, Molecular Biology, and Epidemiology of Bacterial Resistance. Microbiol. Mol. Biol. Rev. 2001, 65, 232–260. [Google Scholar] [CrossRef]
- Kulshrestha, P.; Giese, R.F.; Aga, D.S. Investigating the molecular interactions of oxytetracycline in clay and organic matter: Insights on factors affecting its mobility in soil. Environ. Sci. Technol. 2004, 38, 4097–4105. [Google Scholar] [CrossRef]
- Arikan, O.A.; Sikora, L.J.; Mulbry, W.; Khan, S.U.; Foster, G.D. Composting rapidly reduces levels of extractable oxytetracycline in manure from therapeutically treated beef calves. Bioresour. Technol. 2007, 98, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Aga, D.S.; Lenczewski, M.; Snow, D.; Muurinen, J.; Sallach, J.B.; Wallace, J.S. Challenges in the measurement of antibiotics and in evaluating their impacts in agroecosystems: A critical review. J. Environ. Qual. 2016, 45, 407–419. [Google Scholar] [CrossRef] [PubMed]
- Hamscher, G.; Pawelzick, H.T.; Hoper, H.; Hau, H.; Höper, H.; Nau, H. Different behaviour of tetracyclines and sulfonamides in sandy soils after repeated fertilization with liquid manure. Environ. Toxicol. Chem. 2004, 24, 861–868. [Google Scholar] [CrossRef] [PubMed]
- Goetsch, H.E.; Mylon, S.E.; Butler, S.; Zilles, J.L.; Nguyen, T.H. Oxytetracycline interactions at the soil-water interface: Effects of environmental surfaces on natural transformation and growth inhibition of Azotobacter vinelandii. Environ. Toxicol. Chem. 2012, 31, 2217–2224. [Google Scholar] [CrossRef]
- Cao, J.; Wang, C.; Dou, Z.; Liu, M.; Ji, D. Hyphospheric impacts of earthworms and arbuscular mycorrhizal fungus on soil bacterial community to promote oxytetracycline degradation. J. Hazard. Mater. 2018, 341, 346–354. [Google Scholar] [CrossRef]
- Jones, A.D.A.; Bruland, G.G.L.G.; Agrawal, S.G.; Vasudevan, D. Factors influencing the sorption of oxytetracycline to soils. Environ. Toxicol. Chem. 2005, 24, 761–770. [Google Scholar] [CrossRef]
- Kong, W.; Li, C.; Dolhi, J.M.; Li, S.; He, J.; Qiao, M. Characteristics of oxytetracycline sorption and potential bioavailability in soils with various physical-chemical properties. Chemosphere 2012, 87, 542–548. [Google Scholar] [CrossRef]
| pH | Eh | NO3− | P | K | Ca | Mg | S | Na | Fe | Zn | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ppm | ||||||||||||
| Soil | 5.3 | 120 | 7 | 16 | 92 | 403 | 91 | 6 | 4 | 12.24 | 0.15 | |
| DM | OTC | MON | ||||||||||
| 88.2% | 1.0 µg kg−1 | 2.5 µg kg−1 | ||||||||||
| DM | OTC | MON | % | ppm | ||||||||
| Manure | 84% | 0.009 µg kg−1 | 3.2 µg kg−1 | 1.94 | 0.38 | 0.43 | 1.26 | 0.64 | 0.15 | 4345 | 107.3 | |
| Treatment | Manure Application | Antibiotic Spike | Tifton 85 | Manure |
|---|---|---|---|---|
| 1 | Incorporated | None | Present | Present |
| 2 | Surface applied | None | Present | Present |
| 3 | Incorporated | Oxytetracycline | Present | Present |
| 4 | Surface applied | Oxytetracycline | Present | Present |
| 5 | Incorporated | Monensin | Present | Present |
| 6 | Surface applied | Monensin | Present | Present |
| 7 | Incorporated | None | Absent | Present |
| 8 | Surface applied | None | Absent | Present |
| 9 | Incorporated | Oxytetracycline | Absent | Present |
| 10 | Surface applied | Oxytetracycline | Absent | Present |
| 11 | Incorporated | Monensin | Absent | Present |
| 12 | Surface applied | Monensin | Absent | Present |
| 13 | N/A | None | Present | Absent |
| 14 | N/A | Oxytetracycline | Present | Absent |
| 15 | N/A | Monensin | Present | Absent |
| 16 | N/A | None | Absent | Absent |
| 17 | N/A | Oxytetracycline | Absent | Absent |
| 18 | N/A | Monensin | Absent | Absent |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hilaire, S.S.; Bellows, B.; Brady, J.A.; Muir, J.P. Oxytetracycline and Monensin Uptake by Tifton 85 Bermudagrass from Dairy Manure-Applied Soil. Agronomy 2020, 10, 468. https://doi.org/10.3390/agronomy10040468
Hilaire SS, Bellows B, Brady JA, Muir JP. Oxytetracycline and Monensin Uptake by Tifton 85 Bermudagrass from Dairy Manure-Applied Soil. Agronomy. 2020; 10(4):468. https://doi.org/10.3390/agronomy10040468
Chicago/Turabian StyleHilaire, Sheldon S., Barbara Bellows, Jeff A. Brady, and James P. Muir. 2020. "Oxytetracycline and Monensin Uptake by Tifton 85 Bermudagrass from Dairy Manure-Applied Soil" Agronomy 10, no. 4: 468. https://doi.org/10.3390/agronomy10040468
APA StyleHilaire, S. S., Bellows, B., Brady, J. A., & Muir, J. P. (2020). Oxytetracycline and Monensin Uptake by Tifton 85 Bermudagrass from Dairy Manure-Applied Soil. Agronomy, 10(4), 468. https://doi.org/10.3390/agronomy10040468






