Spraying Agro-Industrial Compost Tea on Baby Spinach Crops: Evaluation of Yield, Plant Quality and Soil Health in Field Experiments
Abstract
1. Introduction
2. Materials and Methods
2.1. Compost Tea Production
2.2. Plant Material, Experimental Set-Up and Design
2.3. Harvesting
2.4. Soil Chemical and Biochemical Parameters
2.5. Soil-Borne Pathogens
2.6. Statistical Analysis
3. Results
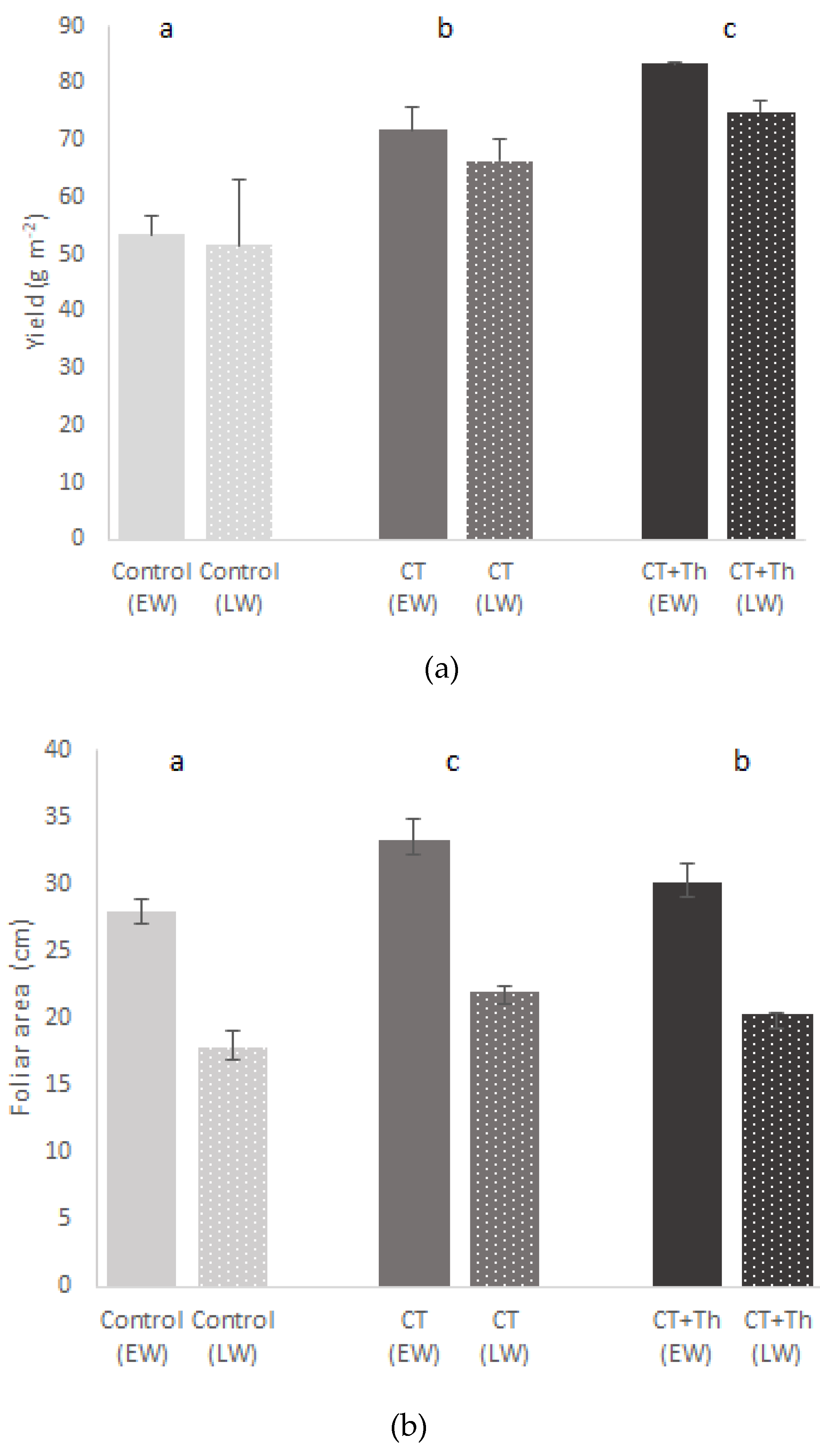
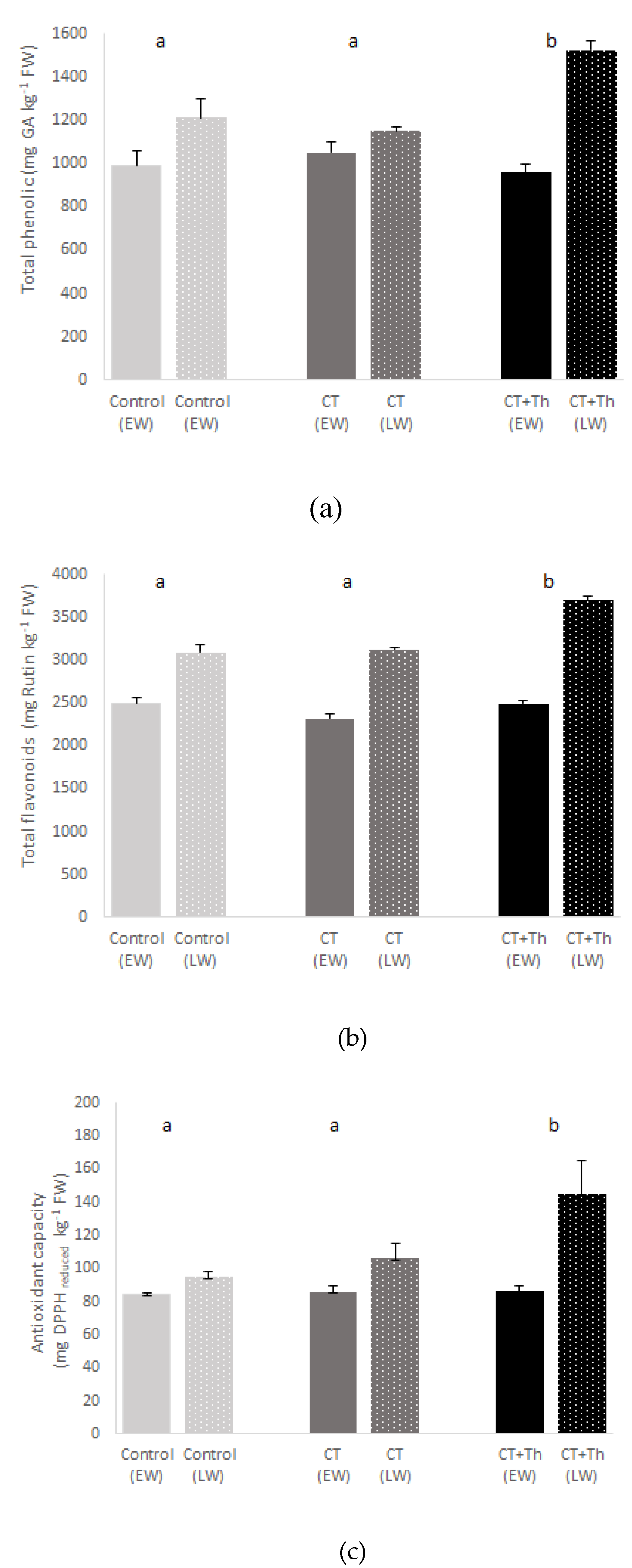
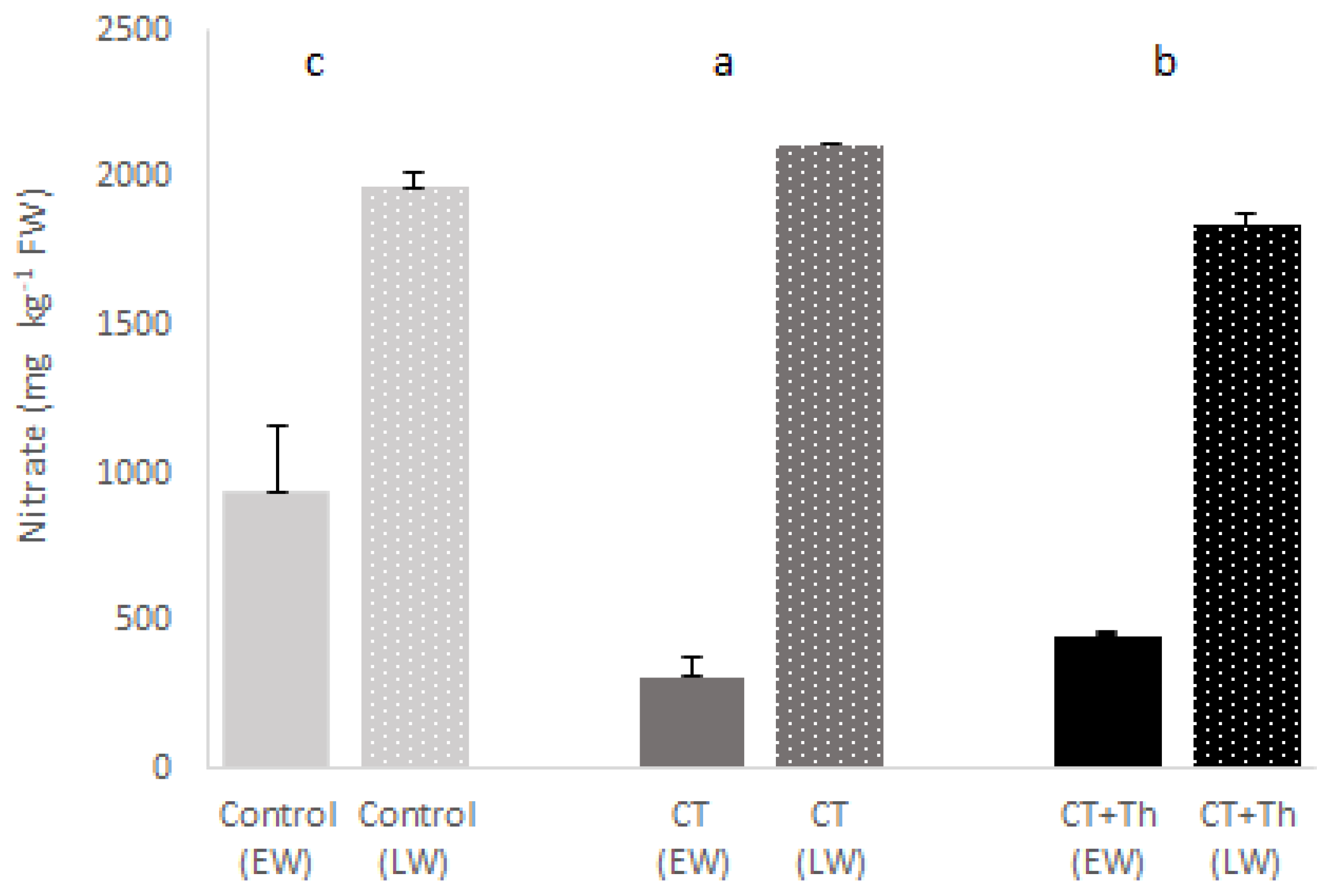
3.1. Effects of CT on Baby-Leaf Spinach Growth and Quality
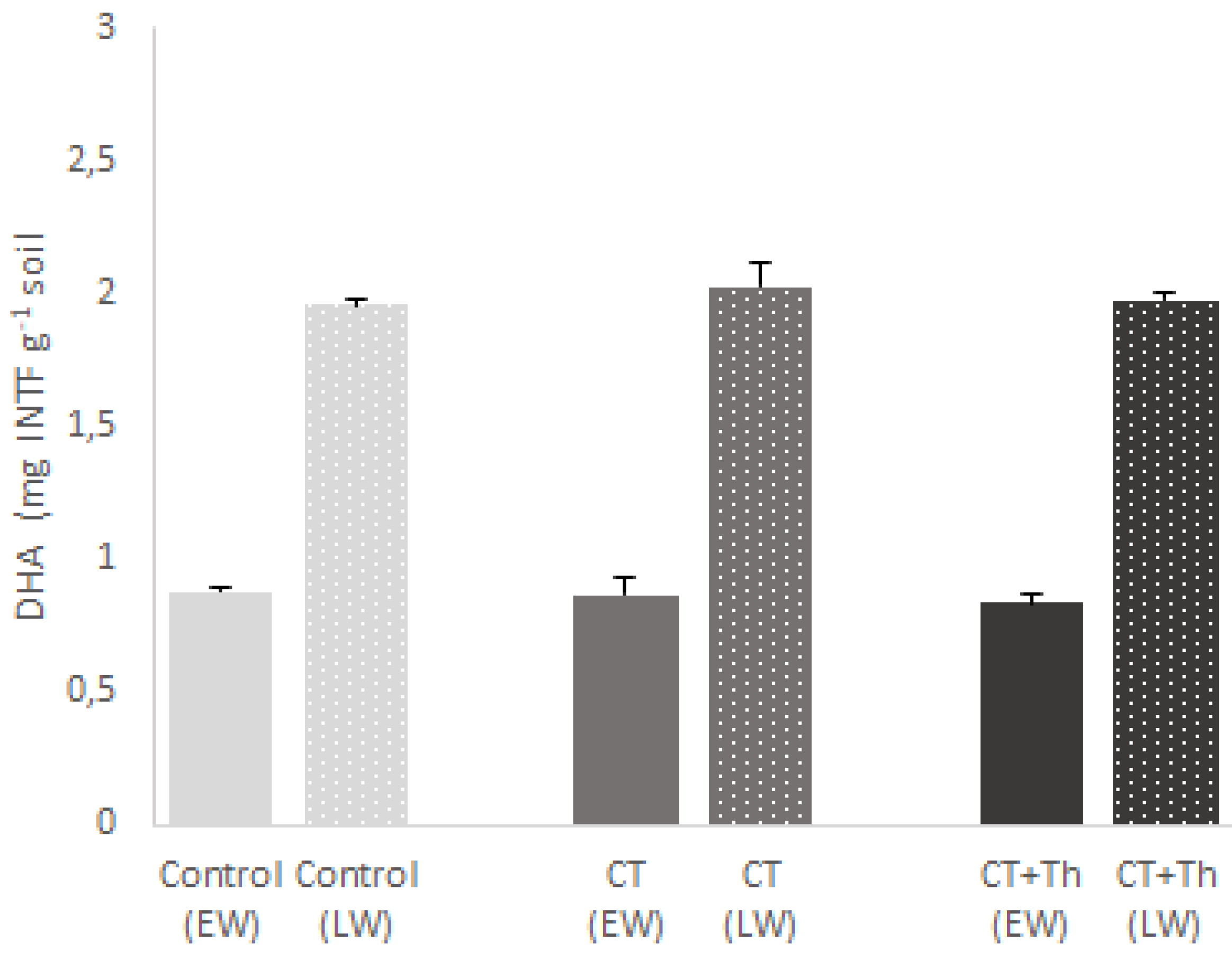
3.2. Effects of CT on Soil Chemical Properties and Soil Microbial Activity
3.3. Effects of CT on Soil-Borne Fungal Pathogen Abundance and Disease Incidence
4. Discussion
4.1. Effects of CT on Baby Spinach Growth and Quality
4.2. Effects of CT on Soil Chemical Properties and Soil Microbial Activity
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Morelock, T.E.; Correll, J.C. Spinach, in Vegetables I: Asteraceae, Brassicaceae, Chenopodiaceae, and Cucurbitaceae; Prohens, J., Nuez, F., Eds.; Springer: New York, NY, USA, 2008; pp. 189–218. [Google Scholar]
- Avsar, B. Genetic Diversity of Turkish Spinach Cultivars (Spinacia oleracea L.). Master’s Thesis, Izmir Institute of Technology, İzmir, Turkey, 2011. [Google Scholar]
- Siddiqui, Y.; Meon, S.; Ismail, R.; Rahmani, M.; Ali, A. Bio-efficiency of compost extracts on the wet rot incidence, morphological and physiological growth of okra (Abelmoschus esculentus [(L.) Moench]). Sci. Hortic. 2008, 117, 9–14. [Google Scholar] [CrossRef]
- Shaheen, M.; Rizkf, A.; Sawano, M.; Bakrym, O. Sustaining the quality and quantity of onion productivity throughout complementarity treatments between compost tea and amino acids. Middle East J. Agric. 2013, 2, 108–115. [Google Scholar]
- Piccolo, A.; Nardi, S.; Concheri, G. Structural characteristics of humic substances as related to nitrate uptake and growth regulation in plant systems. Soil Biol. Biochem. 1992, 24, 373–380. [Google Scholar] [CrossRef]
- Parr, J.F.; Hornick, S.B.; Papendick, R.I. Transition from Conventional Agriculture to Natural Farming Systems: The Role of Microbial Inoculants and Biofertilizer [Online]. International Nature Farming Research Center 2002. Available online: http://www.infrc.or.jp/knf/PDF%20KNF%20Conf%20Data/C4-4-120.pdf (accessed on 16 September 2019).
- Morales-Corts, M.R.; Pérez-Sanchez, R.; Gómez- Sánchez, M. Efficiency of garden waste compost teas on tomato growth and its suppressiveness against soilborne pathogens. Sci. Agric. 2018, 75, 400–409. [Google Scholar] [CrossRef]
- Moretti, S.M.L.; Bertoncini, I.B.; Abreu-Junior, C.H. Composting sewage sludge with green waste from tree pruning. Sci. Agric. 2015, 72, 432–439. [Google Scholar] [CrossRef]
- Ros, M.; Hernández, M.T.; García, C.; Bernal, A.; Pascual, J.A. Biopesticide effect on Green compost against fusarium wilt on melon plants. J. Appl. Microbiol. 2005, 98, 845–854. [Google Scholar] [CrossRef]
- Celar, F.; Valič, N. Effects of Trichoderma spp. and Gliocladium roseum culture filtrates on seed germination of vegetables and maize. J. Plant Dis. Prot. 2005, 112, 343–350. [Google Scholar]
- Rabeendran, N.; Moot, D.J.; Jones, E.E.; Stewart, A. Inconsistent growth promotion of cabbage and lettuce from Trichoderma isolates. N. Z. Plant Prot. 2000, 53, 143–146. [Google Scholar] [CrossRef]
- Hoyos-Carvajal, L.; Ordua, S.; Bissett, J. Growth stimulation in bean (Phaseolus vulgaris L.) by Trichoderma. Biol. Control 2009, 51, 409–416. [Google Scholar] [CrossRef]
- López-Mondéjar, R.; Bernal-Vicente, A.; Ros, M.; Tittarelli, F.; Canali, S.; Intrigliolo, F.; Pascual, J.A. Utilisation of citrus compost-based growing media implemented with Trichoderma harzianum T-78 in Cucumis melo L. seedling production. Bioresour. Technol. 2010, 101, 3718–3723. [Google Scholar] [CrossRef]
- Pane, C.; Celano, G.; Villecco, D.; Zaccardelli, M. Control of Botrytis cinerea, Alternaria alternata and Pyrenochaeta lycopersici on tomato with whey compost-tea applications. Crop Prot. 2012, 38, 80–86. [Google Scholar] [CrossRef]
- Lara, L.J.; Egea-Gilabert, C.; Niñirola, D.; Conesa, E.; Fernández, J.A. Effect of aeration of the nutrient solution on the growth and quality of purslane (Portulaca oleracea). J. Hortic. Sci. Biotechnol. 2011, 86, 603–610. [Google Scholar] [CrossRef]
- Everette, J.D.; Bryant, Q.M.; Green, A.M.; Abbey, Y.A.; Wangila, G.M.; Walker, R.B. Through study of reactivity of various compound classes toward the Folin-Ciocalteu reagent. J. Agric. Food Chem. 2010, 58, 8139–8144. [Google Scholar] [CrossRef] [PubMed]
- Meda, A.; Lamien, C.E.; Romito, M.; Millogo, J.; Nacoulma, O.G. Determination of the total phenolic, flavonoid and proline contents in Burkina Fasan honey, as well as their radical scavenging activity. Food Chem. 2005, 91, 571–577. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of free radical method to evaluate antioxidant activity. LWT—Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- García, C.; Hernández, T.; Costa, F.; Ceccanti, B.; Masciandaro, G. The Dehydrogenase Activity of Soil as an Ecological Marker in Processes of Perturbed System Regeneration. In Proceedings of the XI International Symposium of Environmental Biochemistry, CSIC, Salamanca, Spain, September 1993; pp. 89–100. [Google Scholar]
- Taskin, B.; Gozen, A.G.; Duran, M. Selective quantification of viable Escherichia coli bacteria in biosolids by quantitative PCR with propidium monoazide modification. Appl. Environ. Microbiol. 2011, 77, 4329–4335. [Google Scholar] [CrossRef]
- Santísima-Trinidad, A.B.; Montiel-Rozas, M.M.; Diéz-Rojo, M.A.; Pascual, J.A.; Ros, M. Impact of foliar fungicides on target and non-target soil microbial communities in cucumber crops. Ecotoxicol. Environ. Safe 2018, 166, 78–85. [Google Scholar] [CrossRef]
- Blaya, J.; Lloret, E.; Ros, M.; Pascual, J.A. Identification of predictor parameters to determine agro-industrial compost suppressiveness against Fusarium oxysporum and Phytophthora capsici disease in muskmelon and pepper seedlings. J. Sci. Food Agric. 2015, 95, 1482–1490. [Google Scholar] [CrossRef]
- Morales, A.B.; Ros, M.; Ayuso, L.M.; Bustamante, M.A.; Moral, R.; Pascual, J.A. Agroindustrial Composts to reduce the use of peat and fungicides in the cultivation of muskmelon seedlings. J. Sci. Food Agric. 2017, 97, 875–881. [Google Scholar] [CrossRef]
- Luo, Y.; Liang, J.; Zeng, G.; Chen, M.; Mo, D.; Li, G. Seed germination test for toxicity evaluation of compost: Its roles, problems and prospects. Waste Manag. 2018, 71, 109–114. [Google Scholar] [CrossRef]
- Bernal-Vicente, A.; Ros, M.; Tittarelli, F.; Intrigliolo, F.; Pascual, J.A. Citrus compost and its water extract for cultivation of melon plants in greenhouse nurseries: Evaluation of nutriactive and biocontrol effects. Bioresour. Technol. 2008, 99, 8722–8728. [Google Scholar] [CrossRef] [PubMed]
- Shaban, H.; Fazeli-nasab, B. An Overview of the Benefits of Compost tea on Plant and Soil Structure. Adv. Biomed. Res. 2015, 6, 154–158. [Google Scholar]
- Arancon, N.; Edwards, C.; Dick, R.; Dick, L. Vermicompost tea production and plant growth impacts. BioCycle 2007, 48, 51–52. [Google Scholar]
- Hargreaves, J.C.; Adl, M.; Warman, P.R. Are compost teas an effective nutrient amendment in the cultivation of strawberries? Soil and plant tissue effects. J. Sci. Food Agric. 2009, 89, 390–397. [Google Scholar] [CrossRef]
- Marin, F.; Diánez, F.; Santos, M.; Carretero, F.; Gea, F.J.; Castañeda, C.; Navarro, M.J.; Yau, J.A. Control of Phytophthora capsici and Phytophthora parasitica on pepper (capsicum annum L.) with compost teas form different sources, and their effects on plant growth promotion. Phytopathol. Mediterr. 2014, 53, 216–228. [Google Scholar]
- Edwards, C.C.; Arancon, N.Q.; Greytak, S. Effects of vermicompost teas on plant growth and disease. Biocycle 2006, 47, 28–31. [Google Scholar]
- Zamora-Nahum, S.; Danon, M.; Hadar, Y.; Chen, Y. Chemical properties of compost extracts inhibitory to germination of Sclerotium rolfsii. Soil Biol. Biochem. 2008, 40, 2523–2529. [Google Scholar] [CrossRef]
- Michael, G. The control of root hair formation: Suggested mechanisms. J. Plant Nutr. Soil Sci. 2001, 164, 111–119. [Google Scholar] [CrossRef]
- Contreras-Cornejo, H.; Macías-Rodríguez, L.; del-Val, E.; Larsen, J. Ecological functions of Trichoderma spp. and their secondary metabolites in the rhizosphere: Interactions with plants. FEMS Microbiol. Ecol. 2016, 92, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Harman, G.E.; Petzoldt, R.; Comis, A.; Chen, J. Interactions between Trichoderma harzianum strain T22 and maize inbred line Mo17 and effects of these interactions on disease caused by Pythium ultimum and Colletotrichum graminicola. Phytopathology 2004, 94, 147–153. [Google Scholar] [CrossRef]
- Haggag, W.M.; Abo-Sedera, S.A. Characteristics of three Trichoderma species in peanut haulms compost involved in biocontrol of cumin wilt disease. Int. J. Agric. Biol. 2005, 7, 222–229. [Google Scholar]
- Nahar, M.S.; Rahman, M.A.; Kibria, M.G.; Rezaul-Karim, A.N.M.; Miller, S.A. Use of Tricho-compost and Tricho-leachate for management of soil-borne pathogens and production on healthy cabbage seedlings. Bangladesh J. Agric. Res. 2012, 37, 653–664. [Google Scholar] [CrossRef]
- Topolovec-Pintarić, S. Trichoderma: Invisible Partner for Visible Impact on Agriculture. In Trichoderma—The Most Widely Used Fungicide; Manjur Shah, M., Sharif, U., Buhari, T.R., Eds.; IntechOpen: London, UK, 2019; pp. 1–21. [Google Scholar]
- Kris-Etherton, P.M.; Hecker, K.D.; Bonanome, A.; Coval, S.M.; Binkoski, A.E.; Hilpert, K.F.; Griel, A.E.; Etherton, T.D. Bioactive compounds in foods: Their role in the prevention of cardiovascular disease and cancer. Am. J. Med. 2002, 113 (Suppl. 9B), 71S–88S. [Google Scholar] [CrossRef]
- Giménez, A.; Fernández, J.A.; Pascual, J.A.; Ros, M.; López-Serrano, M.; Egea-Gilabert, C. An agroindustrial compost as alternative to peat for production of baby leaf red lettuce in a floating system. Sci. Hortic. 2019, 46, 907–915. [Google Scholar] [CrossRef]
- Hua-Bin, L.; Chi-Chun, W.; Ka-Wing, C.; Feng, C. Antioxidant properties in vitro and total phenolic contents in methanol extracts from medicinal plants. LWT—Food Sci. Technol. 2008, 41, 385–390. [Google Scholar]
- Pascale, A.; Vinale, F.; Manganiello, G.; Nigro, M.; Lanzuise, S.; Ruocco, M.; Marra, R.; Lombardi, N.; Woo, S.L.; Lorito, M. Trichoderma and its secondary metabolites improve yield and quality. Crop Prot. 2017, 92, 176–181. [Google Scholar] [CrossRef]
- Yedidia, I.; Shoresh, M.; Kerem, Z.; Benhamou, N.; Kapulnik, Y.; Chet, I. Concomitant induction of systemic resistance to Pseudomonas syringae pv. lachrymans in cucumber by Trichoderma asperellum (T-203) and accumulation of phytoalexins. Appl. Environ. Microbiol. 2003, 69, 7343–7353. [Google Scholar] [CrossRef]
- Hassan, S.K.; El–Abssawy, A.A.; Khoder, M.I. Characteristics of gas–phase nitric acid and ammonium–nitrate–sulfate aerosol, and their gas–phase precursors in a suburban area in Cairo, Egypt. Atmos. Pollut. Res. 2013, 4, 117–129. [Google Scholar] [CrossRef]
- Jensen, H.L. Effect of organic compounds on Nitrosomonas. Nature 1950, 165, 974. [Google Scholar] [CrossRef]
- Rittenberg, S.C. The roles of exogenous organic matter in the physiology of chemolithotrophic bacteria. Adv. Microb. Physiol. 1969, 3, 159–196. [Google Scholar]
- Smith, A.J.; Hoare, D.S. Specialist phototrophs, lithotrophs, and methylotrophs: A unity among a diversity of prokaryotes? Bacteriol. Rev. 1977, 41, 419–448. [Google Scholar] [CrossRef] [PubMed]
- Krummel, A.; Harms, H. Effect of organic matter on growth and cell yield of ammonia-oxidizing bacteria. Arch. Microbiol. 1982, 133, 50–54. [Google Scholar] [CrossRef]
- Takahashi, R.; Kondo, N.; Usui, K.; Kanehira, T.; Shinohara, M.; Tokuyama, T. Pure isolation of a new chemoautotrophic ammonia-oxidizing bacterium on gellan gum plate. J. Ferment. Bioeng. 1992, 74, 52–54. [Google Scholar] [CrossRef]
- Stutte, G.W. Nitrogen dynamics in the CELSS breadboard facility at Kennedy Space Center. Life Support Biosph. Sci. Int. J. Earth Space 1996, 3, 67–74. [Google Scholar]
- Xu, Z.; Zheng, S.; Yang, G.; Zhang, Q.; Wang, L. Nitrification inhibition by naphthalene derivatives and its relationship with copper. Bull. Environ. Contam. Toxicol. 2000, 64, 542–549. [Google Scholar] [CrossRef]
- Harman, G.E. Myths and Dogmas of Biocontrol. Changes in perceptions derived from research on Trichoderma harzianum T-22. Plant Dis. 2000, 84, 377–393. [Google Scholar] [CrossRef]
- Lynch, J.M.; Wilson, K.L.; Ousley, M.A.; Whipps, J.M. Response of lettuce to Trichoderma treatment. Lett. Appl. Microbiol. 1991, 12, 59–61. [Google Scholar] [CrossRef]
- Manojlovic, M.; Cabilovski, R.; Nikolic, L.J.; Dzigurski, D.; Seremesic, S.; Baveec, M. Ground cover management and farmyard manure effects on soil nitrogen dynamics, productivity and economics of organically grown lettuce (Lactuca sativa L. subsp. secalina). J. Integr. Agric. 2017, 16, 947–958. [Google Scholar]
- Kapoulas, N.; Koukounaras, A.; Ilic, Z.S. Nutritional quality of lettuce and onion as companion plants from organic and conventional production in north Greece. Sci. Hortic. 2017, 219, 310–318. [Google Scholar] [CrossRef]
- Burns, I.G.; Zhang, K.; Turner, M.K.; Edmondson, R. Iso-osmotic regulation of nitrate accumulation in lettuce. J. Plant Nutr. 2010, 34, 283–313. [Google Scholar] [CrossRef]
- Pascual, J.A.; García, C.; Hernández, T.; Moreno, J.; Ros, M. Soil microbial activity as a biomarker of degradation and remediation processes. Soil Biol. Biochem. 2000, 32, 1877–1883. [Google Scholar] [CrossRef]
- Ros, M.; García, C.; Hernández, T. Soil microbial Activity after restoration of a semiarid soil by organic amendments. Soil Biol. Biochem. 2003, 35, 463–469. [Google Scholar] [CrossRef]
| Parameters | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| pH | EC (dS/m) | Ct (%) | Nt (%) | C/N | P (%) | K (%) | Ca (%) | Na (%) | |
| Compost | 7.25 ± 0.20 | 7.52 ± 0.25 | 36.50 ± 1.00 | 2.00 ± 1.50 | 18 | 2.68 ± 0.30 | 1.58 ± 1.40 | 7.23 ± 2.00 | 1.07 ± 0.70 |
| CT | pH | EC (dS/m) | Ct (mg L−1) | Nt (mg L−1) | C/N | P (mg L−1) | K (mg L−1) | Ca (mg L−1) | Na (mg L−1) |
| EW | 8.33 ± 0.05 | 0.45 ± 0.20 | 69.06 ± 1.25 | 18.1 ± 1.00 | 3.8 | 5.8 ± 0.40 | 99.8 ± 1.50 | 23.6 ± 2.80 | 8.02 ± 0.60 |
| LW | 8.93 ± 0.30 | 0.45 ± 0.24 | 57.85 ± 2.85 | 15.61 ± 3.30 | 3.7 | 6.02 ± 0.40 | 91.6 ± 8.30 | 26.1 ± 1.20 | 9.26 ± 0.60 |
| F | P | ||
|---|---|---|---|
| Treatment | Yield (g m−2) | 35.137 | 0.000 |
| Foliar área (cm) | 31.972 | 0.000 | |
| Total phenolic (mg GA Kg−1 FW) | 6.761 | 0.009 | |
| Total flavonoids (mg Rutin kg−1 FW) | 9.272 | 0.003 | |
| Antioxidant capacity (mg DPPH reduced kg−1 FW) | 16.068 | 0.000 | |
| Nitrate (mg kg−1 FW) | 68.236 | 0.000 | |
| Experiment | Yield (g m−2) | 3.676 | 0.076 |
| Foliar area (cm) | 392.073 | 0.000 | |
| Total phenolic (mg GA kg−1 FW) | 69.912 | 0.000 | |
| Total flavonoids (mg Rutin kg−1 FW) | 145.327 | 0.000 | |
| Antioxidant capacity (mg DPPH reduced kg−1 FW) | 56.433 | 0.000 | |
| Nitrate (mg kg−1 FW) | 2929.441 | 0.000 | |
| Interaction | Yield (g m−2) | 0.505 | 0.614 |
| Foliar area (cm) | 2.441 | 0.123 | |
| Total phenolic (mg GA kg−1 FW) | 14.320 | 0.000 | |
| Total flavonoids (mg Rutin kg−1 FW) | 6.436 | 0.010 | |
| Antioxidant capacity (mg DPPH reduced kg−1 FW) | 13.463 | 0.001 | |
| Nitrate (mg kg−1 FW) | 7.675 | 0.007 |
| Treatment | P (g kg−1) | K (g kg−1) | Ca (g kg−1) | |||
| EW | LW | EW | LW | EW | LW | |
| Control | 1.49–10.19 b | 0.41–10.04 A | 6.22–10.19 a | 7.70–10.45 A | 0.78–10.06 b | 0.90–10.06 A |
| CT | 2.47–10.30 a | 0.41–10.03 A | 6.28–10.08 a | 8.14–10.36 A | 0.86–10.04 a | 0.62–10.04 B |
| CT + Th | 2.34–10.30 a | 0.40–10.03 A | 6.34–10.06 a | 7.99–10.49 A | 0.88–10.06 a | 0.57–10.04 B |
| Parameters | Multivariate Analysis of Variance | |||||
| Treatment | Experiment | Interaction | ||||
| F | P | F | P | F | P | |
| P (g kg−1) | 7.388 | 0.005 | 208.560 | 0.000 | 7.583 | 0.004 |
| K (g kg−1) | 2.822 | 0.087 | 142.904 | 0.000 | 1.551 | 0.241 |
| Ca (g kg−1) | 2.847 | 0.086 | 131.732 | 0.000 | 1.908 | 0.179 |
| Treatment | Total C (g kg−1) | Total N (g kg−1) | Total P (g kg−1) | Total K (g kg−1) | ||||
| EW | LW | EW | LW | EW | LW | EW | LW | |
| Control | 29.35–11.07 a | 29.35–11.07 a | 29.35–11.07 a | 29.35–11.07 a | 29.35–11.07 a | 29.35–11.07 a | 29.35–11.07 a | 29.35–11.07 a |
| CT | 30.23–10.75 a | 30.23–10.75 a | 30.23–10.75 a | 30.23–10.75 a | 30.23–10.75 a | 30.23–10.75 a | 30.23–10.75 a | 30.23–10.75 a |
| CT + Th | 30.60–11.90 a | 30.60–11.90 a | 30.60–11.90 a | 30.60–11.90 a | 30.60–11.90 a | 30.60–11.90 a | 30.60–11.90 a | 30.60–11.90 a |
| Treatment | Cycle | Interaction | ||||||
| F | P | F | P | F | SP | |||
| Total C (g kg−1) | 12.39 | 0.01 | 0.846 | 0.373 | 7.140 | 0.005 | ||
| Total N (g kg−1) | 1.122 | 0.353 | 19.873 | 0.001 | 1.366 | 0.287 | ||
| Total P (g kg−1) | 1,599 | 0.237 | 98.897 | 0.000 | 2.192 | 0.149 | ||
| Total K (g kg−1) | 1.272 | 0.311 | 6.229 | 0.026 | 3.012 | 0.082 | ||
| Fungal Pathogens | Treatment | EW | LW | |||
| Alternaria spp. | Control | 4.64–10.44 | 5.95–10.92 | |||
| CT | 4.88–10.35 | 5.79–10.20 | ||||
| CT + Th | 5.17–10.09 | 6.19–10.19 | ||||
| F. oxysporum | Control | 4.32–10.39 | 5.62–10.07 | |||
| CT | 4.23–10.13 | 5.95–10.21 | ||||
| CT + Th | 3.87–10.31 | 6.12–10.01 | ||||
| F. solani | Control | 4.99–10.05 | 6.47–10.07 | |||
| CT | 4.88–10.32 | 6.76–10.12 | ||||
| CT + Th | 4.81–10.47 | 6.82–10.03 | ||||
| Stemphylium botryosum | Control | 4.00–10.15 | 4.74–10.21 | |||
| CT | 3.95–10.12 | 4.97–10.22 | ||||
| CT + Th | 3.85–10.16 | 5.13–10.14 | ||||
| Treatment | Experiment | Interaction | ||||
| F | P | F | P | F | P | |
| Alternaria spp. | 1.165 | 0.340 | 22.772 | 0.000 | 0.291 | 0.752 |
| F. oxysporum | 0.517 | 0.607 | 269.759 | 0.000 | 7.011 | 0.008 |
| F. solani | 0.383 | 0.689 | 317.683 | 0.000 | 2.748 | 0.098 |
| Stemphylium botryosum | 286.72 | 0.000 | 967.035 | 0.000 | 308.609 | 0.000 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ros, M.; Hurtado-Navarro, M.; Giménez, A.; Fernández, J.A.; Egea-Gilabert, C.; Lozano-Pastor, P.; Pascual, J.A. Spraying Agro-Industrial Compost Tea on Baby Spinach Crops: Evaluation of Yield, Plant Quality and Soil Health in Field Experiments. Agronomy 2020, 10, 440. https://doi.org/10.3390/agronomy10030440
Ros M, Hurtado-Navarro M, Giménez A, Fernández JA, Egea-Gilabert C, Lozano-Pastor P, Pascual JA. Spraying Agro-Industrial Compost Tea on Baby Spinach Crops: Evaluation of Yield, Plant Quality and Soil Health in Field Experiments. Agronomy. 2020; 10(3):440. https://doi.org/10.3390/agronomy10030440
Chicago/Turabian StyleRos, Margarita, María Hurtado-Navarro, Almudena Giménez, Juan Antonio Fernández, Catalina Egea-Gilabert, Pedro Lozano-Pastor, and José Antonio Pascual. 2020. "Spraying Agro-Industrial Compost Tea on Baby Spinach Crops: Evaluation of Yield, Plant Quality and Soil Health in Field Experiments" Agronomy 10, no. 3: 440. https://doi.org/10.3390/agronomy10030440
APA StyleRos, M., Hurtado-Navarro, M., Giménez, A., Fernández, J. A., Egea-Gilabert, C., Lozano-Pastor, P., & Pascual, J. A. (2020). Spraying Agro-Industrial Compost Tea on Baby Spinach Crops: Evaluation of Yield, Plant Quality and Soil Health in Field Experiments. Agronomy, 10(3), 440. https://doi.org/10.3390/agronomy10030440








