The Use of a Plant-Based Biostimulant Improves Plant Performances and Fruit Quality in Tomato Plants Grown at Elevated Temperatures
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Growth, Experimental Design, and Treatments
2.2. Pollen Viability
2.3. Ascorbic Acid Quantification
2.4. Total Carotenoids and Chlorophylls Content
2.5. Antioxidant Activity Determination
2.6. Fluorescence Emission Measurements
2.7. Leaf Functional Traits Determination
2.8. Statistical Analysis
3. Results
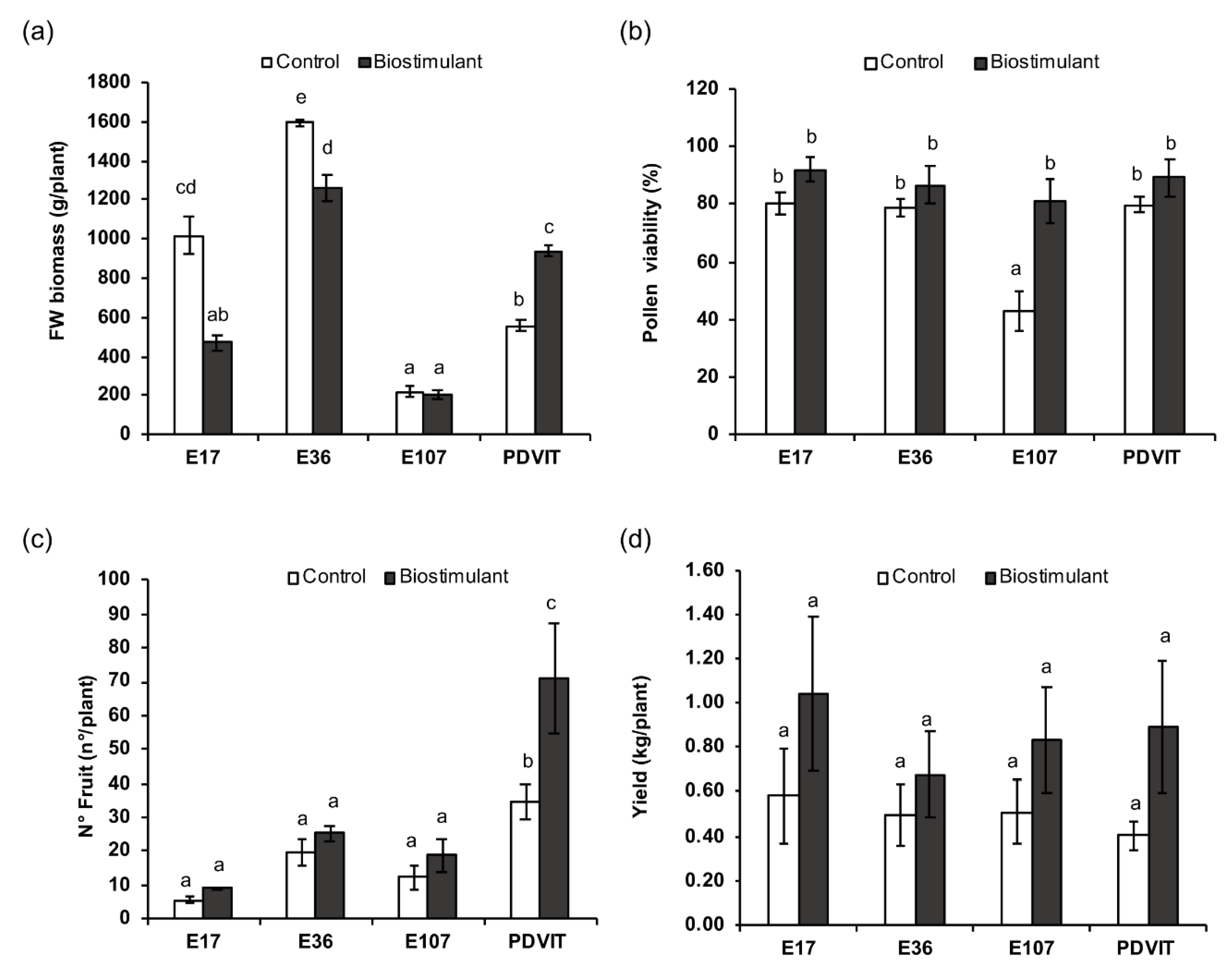
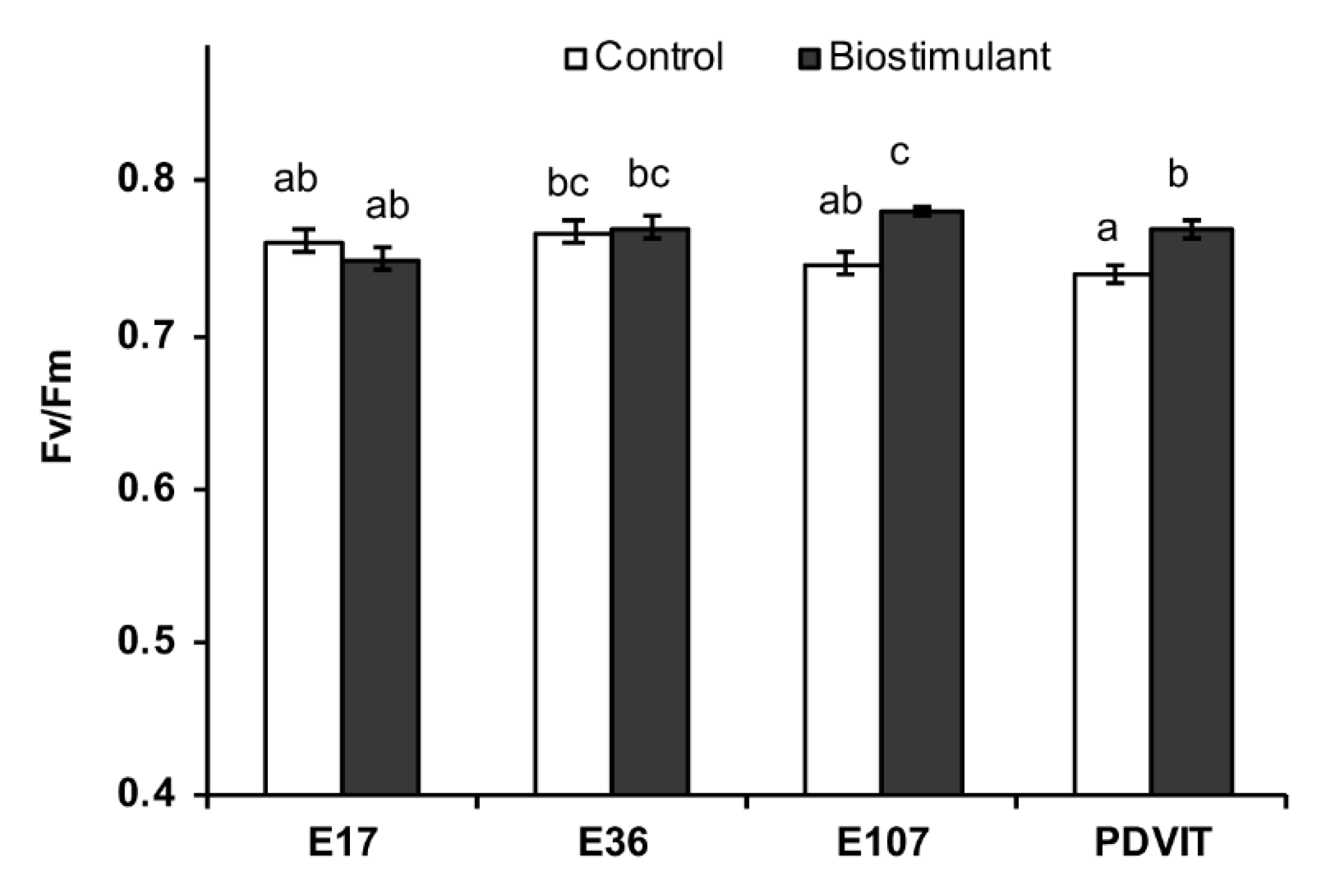
3.1. Phenotypic and Physiological Analyses
3.2. Leaf and Fruit Antioxidant Content
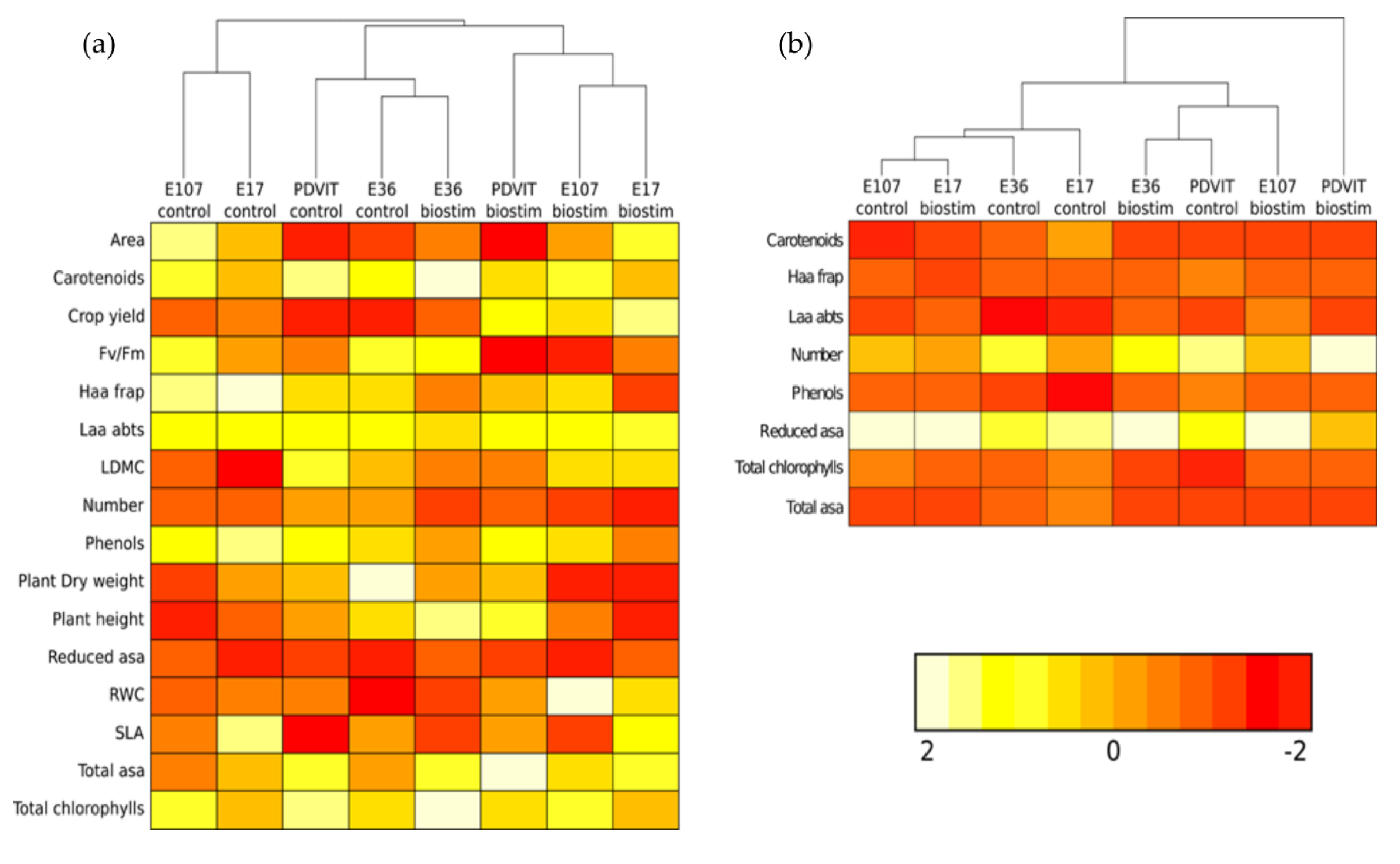
3.3. Heat Map Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Del Giudice, R.; Petruk, G.; Raiola, A.; Barone, A.; Monti, D.M.; Rigano, M.M. Carotenoids in Fresh and Processed Tomato (Solanum lycopersicum) Fruits Protect Cells from Oxidative Stress Injury. J. Sci. Food Agric. 2016, 97, 1616–1623. [Google Scholar] [CrossRef] [PubMed]
- Rigano, M.M.; Arena, C.; Di Matteo, A.; Sellitto, S.; Frusciante, L.; Barone, A. Eco-physiological response to water stress of drought-tolerant and drought-sensitive tomato genotypes. Plant Biosyst. 2016, 150, 682–691. [Google Scholar] [CrossRef]
- Ruggieri, V.; Calafiore, R.; Schettini, C.; Rigano, M.M.; Olivieri, F.; Frusciante, L.; Barone, A. Exploiting genetic and genomic resources to enhance heat-tolerance in tomatoes. Agronomy 2019, 9, 22. [Google Scholar] [CrossRef]
- Wen, J.; Jiang, F.; Weng, Y.; Sun, M.; Shi, X.; Zhou, Y.; Yu, L.; Wu, Z. Identification of heat-tolerance QTLs and high-temperature stress-responsive genes through conventional QTL mapping, QTL-seq and RNA-seq in tomato. BMC Plant Biol. 2019, 19, 398. [Google Scholar] [CrossRef] [PubMed]
- Szymanska, R.; Slesak, I.; Orzechowska, A.; Kruk, J. Physiological and biochemical responses to high light and temperature stress in plants. Environ. Exp. Bot. 2017, 139, 165–177. [Google Scholar] [CrossRef]
- Povero, G.; Mejia, J.F.; Di Tommaso, D.; Pioggesi, A.; Warrior, P. A systematic approach to discover and characterize natural plant biostimulant. Front. Plant Sci. 2016, 7, 435. [Google Scholar] [CrossRef]
- Di Stasio, E.; Van Oosten, M.J.; Silletti, S.; Raimondi, G.; Dell’Aversana, E.; Carillo, P.; Maggio, A. Ascophyllum nodosum- based algal extracts act as enhancers of growth, fruit quality, and adaptation to stress in salinized tomato plants. J. Appl. Physiol. 2018, 30, 2675–2686. [Google Scholar] [CrossRef]
- Du Jardin, P. Plant biostimulant: Definition, concept, main categories and regulation. Sci. Hortic. 2015, 196, 3–14. [Google Scholar] [CrossRef]
- Di Mola, I.; Ottaiano, L.; Cozzolino, E.; Senatore, M.; Giordano, M.; El-Nakhel, C.; Sacco, A.; Youssef, R.; Colla, G.; Mori, M. Plant-Based Biostimulants Influence the Agronomical, Physiological, and Qualitative Responses of Baby Rocket Leaves under Diverse Nitrogen Conditions. Plants 2019, 8, 522. [Google Scholar] [CrossRef]
- Colla, G.; Cardarelli, M.; Bonini, P.; Rouphael, Y. Foliar Application of Protein Hydrolysate, Plant and Seaweed Extracts Increase Yield but Differentially Modulate Fruit Quality of Greenhouse Tomato. Hortic. Sci. 2017, 52, 1214–1220. [Google Scholar] [CrossRef]
- Feitosa de Vasconcelos, A.C.; Zhang, X.; Ervin, E.H.; De Castro Kiehl, J. Enzymatic antioxidant responses to biostimulants in maize and soybean subjected to drought. Sci. Agric. 2009, 66, 395–402. [Google Scholar] [CrossRef]
- Petrozza, A.; Santaniello, A.; Summerer, S.; Di Tommaso, G.; Di Tommaso, D.; Paparelli, E.; Piaggesi, A.; Perata, P.; Cellini, F. Physiological responses to Megafol® treatments in tomato plants under drought stress: A phenomic and molecular approch. Sci. Hortic. 2014, 174, 185–192. [Google Scholar] [CrossRef]
- Ertani, A.; Schiavon, M.; Muscolo, A.; Nardi, S. Alfalfa plant-dericed biostimulant stimulate short-term growth of salt stressed Zea mays L. plants. Plant Soil 2013, 364, 145–158. [Google Scholar] [CrossRef]
- Lucini, L.; Rouphael, Y.; Cardarelli, M.; Canaguier, R.; Kumar, P.; Colla, G. The effect of a plant-derived biostimulant on metabolic profiling and crop performance of lettuce grown under saline conditions. Sci. Hortic. 2015, 182, 124–133. [Google Scholar] [CrossRef]
- Botta, A. Enhancing plant tolerance to temperature stress with amino acids: An approach to their mode of action. Acta Hortic. 2013, 1009, 29–35. [Google Scholar] [CrossRef]
- Verkleij, F.N. Seaweed extracts in agriculture and horticulture: A review. Biol. Agric. Hortic. 1992, 8, 309–324. [Google Scholar] [CrossRef]
- Battacharyya, D.; Babgohari, M.Z.; Rathor, P.; Prithiviraj, B. Seaweed extracts as biostimulants in horticulture. Sci. Hortic. 2015, 30, 39–48. [Google Scholar] [CrossRef]
- Van Oosten, M.J.; Pepe, O.; De Pascale, S.; Silletti, S.; Maggio, A. The role of biostimulants and bioeffectors as alleviators of abiotic stress in crop plants. Chem. Biol. Technol. Agric. 2017, 4, 5. [Google Scholar] [CrossRef]
- Hammad, S.A.R.; Ali, A.M.O. Physiological and biochemical studies on drought tolerance of wheat plants by application of amino acids and yeast extract. Ann. Agric. Sci. 2014, 59, 133–145. [Google Scholar] [CrossRef]
- De Pascale, S.; Rouphel, Y.; Colla, G. Plant biostimulants: Innovative tool for enhancing plant nutrition in organic farming. Eur. J. Hortic. Sci. 2017, 82, 277–285. [Google Scholar] [CrossRef]
- Paul, K.; Sorrentino, M.; Lucini, L.; Rouphael, Y.; Cardarelli, M.; Bonini, P.; Moreno, M.B.M.; Reynaud, H.; Canaguier, R.; Trtilek, M.; et al. A combined phenotypic and metabolic approach for elucidating the biostimulant action of a plant-derived protein hydrolysate on tomato grown under limited water availability. Front. Plant Sci. 2019, 10, 493. [Google Scholar] [CrossRef] [PubMed]
- Sorwong, A.; Sakhonwasee, S. Foliar Application of Glycine Betaine Mitigates the Effect of Heat Stress in Three Marigold (Tageteserecta) Cultivars. Hortic. J. 2015, 84, 161–171. [Google Scholar] [CrossRef]
- Dafni, A. Pollination Ecology: A Practical Approach; Oxford University Press: Oxford, UK, 1992; p. xiv +250. [Google Scholar]
- Stevens, R.; Buret, M.; Garchery, C.; Carretero, Y.; Causse, M. Technique for rapid small-scale analysis of vitamin C levels in fruit and application to a tomato mutant collection. J. Agric. Food Chem. 2006, 54, 6159–6165. [Google Scholar] [CrossRef] [PubMed]
- Rigano, M.M.; Raiola, A.; Tenore, G.C.; Monti, D.M.; Del Giudice, R.; Frusciante, L.; Barone, A. Quantitative trait loci pyramiding can improve the nutritional potential of tomato (Solanum lycopersicum) fruits. J. Agric. Food Chem. 2014, 62, 11519–11527. [Google Scholar] [CrossRef] [PubMed]
- Rigano, M.M.; Lionetti, V.; Raiola, A.; Bellincampi, D.; Barone, A. Pectic enzymes as potential enhancers of ascorbic acid production through the d-galacturonate pathway in Solanaceae. Plant Sci. 2018, 266, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Wellburn, A.R. The spectral determination of chlorophylls a and b, as well as total carotenoids, using various solvents with spectrophotometers of different resolution. J. Plant Physiol. 1994, 144, 307–313. [Google Scholar] [CrossRef]
- Zouari, I.; Salvioli, A.; Chialva, M.; Novero, M.; Miozzi, L.; Tenore, G.C.; Bagnaresi, P.; Bonfante, P. From root to fruit: RNA-Seq analysis shows that arbuscular mycorrhizal symbiosis may affect tomato fruit metabolism. BMC Genom. 2014, 15, 221. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef]
- Miller, J.N.; Rice-Evans, C.A. Factors influencing the antioxidant activity determined by the ABTS•+ radical cation assay. Free Radic. Res. 1997, 26, 195–199. [Google Scholar] [CrossRef]
- Figlioli, F.; Sorrentino, M.C.; Memoli, V.; Arena, C.; Maisto, G.; Giordano, S.; Capozzi, F.; Spagnuolo, V. Overall plant responses to Cd and Pb metal stress in maize: Growth pattern, ultrastructure, and photosynthetic activity. Environ. Sci. Pollut. Res. ESPR 2019, 26, 1781–1790. [Google Scholar] [CrossRef]
- Kitajima, M.; Butler, W.L. Quenching of chlorophyll fluorescence and primary photochemistry in chloroplasts by dibromothymoquinone. Biochim. Biophys. Acta-Bioenerg. 1975, 376, 105–115. [Google Scholar] [CrossRef]
- Arena, C.; De Micco, V.; De Maio, A.; Mistretta, C.; Aronne, G.; Vitale, L. Winter and summer leaves of Cistus incanus: Differences in leaf morpho-functional traits, photosynthetic energy partitioning and poly-ADP-ribose polymerase (PARP) activity. Botany 2013, 91, 805–813. [Google Scholar] [CrossRef]
- Hossain, M.I.; Khatun, A.; Talukder, M.S.A.; Dewan, M.M.R.; Uddin, M.S. Effect of drought o physiology and yield contributing characters of sunflower. Bangladesh J. Agric Res. 2010, 35, 113–124. [Google Scholar] [CrossRef]
- Paradikovic, N.; Vinkovic, T.; VinkovicVrcek, I.; Zuntar, I.; Bojic, M.; Medic-Saric, M. Effect of natural biostimulants on yield and nutritional quality: An example of sweet yellow pepper (Capsicum annuum L.) plants. J. Sci. Food Agric. 2011, 91, 2146–2152. [Google Scholar] [CrossRef]
- Chehade, L.A.; Al Chami, Z.; De Pascali, S.A.; Cavoski, I.; Fanizzi, F.P. Biostimulants from food processing by-poducts: Agronomic, quality and metabolic impacts on organic tomato (Solanum lycopersicum L.). J. Sci. Food Agric. 2017, 98, 1426–1436. [Google Scholar] [CrossRef]
- Kocira, S.; Szparaga, A.; Kocira, A.; Czerwinska, E.; Wojtowicz, A.; Bronowicka-Mielniczuk, U.; Koszel, M.; Findura, P. Modeling biometric traits, yield and nutritional and antioxidant properties of seeds of three soybean cultivars through the application of biostimulant containing seaweed and amino acids. Front. Plant Sci. 2018, 9, 388. [Google Scholar] [CrossRef]
- Cristiano, G.; Pallozzi, E.; Conversa, G.; Tufarelli, V.; De Lucia, B. Effect of an Animal-Derived Biostimulant on the Growth and Physiological Parameters of Potted Snapdragon (Antirrhinum majus L.). Front. Plant Sci. 2018, 9, 861. [Google Scholar] [CrossRef]
- Polo, J.; Mata, P. Evaluation of a biostimulant (pepton) based in enzymatic hydrolyzed animal protein in comparison to seaweed extracts on root development, vegetative growth, flowering, and yield of gold cherry tomatoes grown under low stress ambient field conditions. Front. Plant Sci. 2018, 8, 2261. [Google Scholar] [CrossRef]
- Hamasaki, T.; Kitagawa, T.; Yasuhara, T. Efficacy of yeast cell wall extract, a Byproduct of beer brewing, in Tomato (Solanum lycopersicum) culture. IPCBEE 2014, 76, 21–25. [Google Scholar] [CrossRef]
- Rouphael, Y.; Colla, G.; Giordano, M.; El-Nakhel, C.; Kyriacou, M.C.; De Pascale, S. Foliar applications of a legume-derived protein hydrolysate elicit dose-dependent increases of growth, leaf mineral composition, yield and fruit quality in two greenhouse tomato cultivars. Sci. Hortic. 2017, 226, 353–360. [Google Scholar] [CrossRef]
- Matsuo, S.; Kikuchi, K.; Fukuda, M.; Honda, I.; Imanishi, S. Roles and regulation of cytokinins in tomato fruit development. J. Exp. Bot. 2012, 63, 5569–5579. [Google Scholar] [CrossRef] [PubMed]
- Sato, S.; Kamiyama, M.; Iwara, T.; Makita, N.; Furukawa, H.; Ikeda, H. Moderate increase of mean daily temperature adversely affects fruit set of Lycopersicum esculentum by distrupting specific physiological processes in male reproductive development. Ann. Bot. 2006, 97, 731–738. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Li, F.; Wang, J.; Zhang, W.; Meng, Q.; Chen, T.H.H.; Murata, N.; Yang, X. Glycinebetaine enhances the tolerance of tomato plants to high temperature during germination of seeds and growth of seedlings. Plant Cell Environ. 2011, 34, 1931–1943. [Google Scholar] [CrossRef] [PubMed]
- Cristiano, G.; Pallozzi, E.; Conversa, G.; Tufarelli, V.; De Lucia, B. Effects of an animal-derived biostimulant on the growth and physiological parameters of potted snapdragon (Antirrhinum majus L.). Front. Plant Sci. 2017, 9, 861. [Google Scholar] [CrossRef]
- Xu, J.; Wolters-Arts, M.; Mariani, C.; Huber, H.; Rieu, I. Heat stress affects vegetative and reproductive performance and trait correlations in tomato (Solanum lycopersicum). Euphytica 2017, 213, 156. [Google Scholar] [CrossRef]
- Santaniello, A.; Scartazza, A.; Gresta, F.; Loreti, E.; Biasone, A.; Di Tommaso, D.; Piaggesi, A.; Perata, P. Ascophyllum nodosum Seeweed Extract Alleviates Drought Stress in Arabidopsis by Affecting Photosynthetic Performance and Related Gene Expression. Front. Plant Sci. 2017, 8, 1362. [Google Scholar] [CrossRef]
- Ertani, A.; Pizzeghello, D.; Francioso, O.; Sambo, P.; Sanchez-Cortes, S.; Nardi, S. Capsicum chinensis L. growth and nutraceutical properties are enhanced by biostimulants in a long-term period: Chemical and metabolomic approaches. Front. Plant Sci. 2014, 5, 375. [Google Scholar] [CrossRef]
- Parrado, J.; Bautista, J.; Romero, E.J.; Garcìa-Martìnez, A.M.; Friaza, V.; Tejada, M. Production of a carob enzymatic extract: Potential use as a biofertilizer. Bioresour. Technol. 2008, 99, 2312–2318. [Google Scholar] [CrossRef]
| Genotype | Origin | Common Accession | Fruit Size | Fruit Color |
|---|---|---|---|---|
| E17 | Italy | Pantano Romanesco | Big (200–250 g) | red |
| E36 | Italy | Riccia San Vito | Small (25–30 g) | red |
| E107 | Spain | E-L-19 | Medium (70–100 g) | yellow |
| PDVIT | Italy | Caramella | Small (10–15 g) | red |
| E17 | E36 | E107 | PDVIT | SIGNIFICANCE | |||
|---|---|---|---|---|---|---|---|
| Reduced AsA (mg/100 g FW) | control | 6 ± 0.43 a | 7.95 ± 1.33 a | 10.83 ± 1 ab | 18.20 ± 0.91 bc | G | ** |
| treated | 20.05 ± 3.30 c | 20.12 ± 1.42 c | 17.41 ± 1.91 bc | 19.34 ± 1.33 c | T | *** | |
| G X T | ** | ||||||
| Total AsA (mg/100 g FW) | control | 16.79 ± 0.73 ab | 14.45 ± 0.51 a | 24.52 ± 2.03 bc | 21.28 ± 0.86 bc | G | *** |
| treated | 21.15 ± 0.90 bc | 24.40 ± 2.55 cd | 20.27 ± 0.83 cd | 26.85 ± 0.69 d | T | *** | |
| G X T | *** | ||||||
| Phenols (mg/100 g FW) | control | 43.38 ± 0.98 e | 26.91 ± 1.19 a | 35.14 ± 0.48 c | 35.30 ± 0.56 c | G | *** |
| treated | 25.33 ± 1.20 a | 25.58 ± 0.27 a | 31.57 ± 0.52 b | 39.57 ± 0.54 d | T | *** | |
| G X T | *** | ||||||
| Carotenoids (mg/100 g FW) | control | 23.91 ± 1.06 ab | 26.06 ± 0.53 abc | 23.80 ± 0.75 a | 28.73 ± 0.23 de | G | *** |
| treated | 23.78 ± 0.48 a | 30.17 ± 0.24 e | 28.10 ± 0.47 cde | 26.42 ± 0.46 bcd | T | *** | |
| G X T | *** | ||||||
| Chl a (mg/100 g FW) | control | 108.78 ± 3.05 a | 113.30 ± 4.3 6 ab | 128.22 ± 5.34 bc | 140.30 ± 4.25 c | G | *** |
| treated | 110.13 ± 1.37 a | 137.08 ± 2.07 c | 138.61 ± 3.32 c | 130.20 ± 2.80 bc | T | ** | |
| G X T | *** | ||||||
| Chl b (mg/100 g FW) | control | 38.65 ± 3.96 a | 37.45 ± 2.12 a | 45.84 ± 3.67 ab | 55.75 ± 3.74 b | G | *** |
| treated | 37.29 ± 2.73 a | 55.41 ± 2.11 b | 59.47 ± 2.69 b | 45.85 ± 5.72 ab | T | ** | |
| G X T | *** | ||||||
| LAA (mg/100 g FW) | control | 18.88 ± 0.14 a | 18.75 ± 0.07 a | 18.86 ± 0.04 a | 18.62 ± 0.05 b | G | *** |
| treated | 18.98 ± 0.04 a | 19.07 ± 0.21 a | 19.90 ± 0.08 b | 19.75 ± 0.10 a | T | *** | |
| G X T | *** | ||||||
| HAA (mg/100 g FW) | control | 828.58 ± 140.08 a | 493.19 ± 220.27 bc | 599.85 ± 118.33 ab | 434.30 ± 88.34 cd | G | *** |
| treated | 255.57 ± 91.31 d | 390.49 ± 25.34 bc | 510.33 ± 53.59 ab | 438.26 ± 125.38 bc | T | *** | |
| G X T | *** | ||||||
| E17 | E36 | E107 | PDVIT | SIGNIFICANCE | |||
|---|---|---|---|---|---|---|---|
| Reduced AsA (mg/100 g FW) | control | 33.31 ± 2.99 a | 39.56 ± 2.30 ab | 47.14 ± 1.66 bc | 50.36 ± 1.84 bc | G | *** |
| treated | 47.36 ± 1.60 bc | 59.87 ± 4.34 cd | 74.79 ± 3.25 e | 64.83 ± 2.34 de | T | *** | |
| G X T | ns | ||||||
| Total AsA (mg/100 g FW) | control | 61.97 ± 0.57 ab | 78.03 ± 3.29 bc | 85.40 ± 3.75 c | 52.87 ± 4.24 a | G | ** |
| treated | 79.34 ± 4.44 bc | 87.15 ± 2.35 c | 93.36 ± 6.19 cd | 112.53 ± 4.08 d | T | *** | |
| G X T | *** | ||||||
| Phenols (mg/100 g FW) | control | 9.62 ± 0.46 a | 13.70 ± 0.68 b | 16.17 ± 0.58 c | 22.35 ± 0.37 e | G | *** |
| treated | 16.62 ± 0.46 c | 18.92 ± 0.76 d | 16.88 ± 0.44 c | 22.55 ± 0.19 e | T | *** | |
| G X T | *** | ||||||
| HAA (mg/100 g FW) | control | 129.28 ± 33.95 a | 189.22 ± 49.66 b | 179.38 ± 20.62 bc | 309.06 ± 39.51 d | G | *** |
| treated | 151.57 ± 8.71 c | 304.38 ± 30.92 c | 212.47 ± 7.08 c | 333.03 ± 46.91 d | T | *** | |
| G X T | *** | ||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Francesca, S.; Arena, C.; Hay Mele, B.; Schettini, C.; Ambrosino, P.; Barone, A.; Rigano, M.M. The Use of a Plant-Based Biostimulant Improves Plant Performances and Fruit Quality in Tomato Plants Grown at Elevated Temperatures. Agronomy 2020, 10, 363. https://doi.org/10.3390/agronomy10030363
Francesca S, Arena C, Hay Mele B, Schettini C, Ambrosino P, Barone A, Rigano MM. The Use of a Plant-Based Biostimulant Improves Plant Performances and Fruit Quality in Tomato Plants Grown at Elevated Temperatures. Agronomy. 2020; 10(3):363. https://doi.org/10.3390/agronomy10030363
Chicago/Turabian StyleFrancesca, Silvana, Carmen Arena, Bruno Hay Mele, Carlo Schettini, Patrizia Ambrosino, Amalia Barone, and Maria Manuela Rigano. 2020. "The Use of a Plant-Based Biostimulant Improves Plant Performances and Fruit Quality in Tomato Plants Grown at Elevated Temperatures" Agronomy 10, no. 3: 363. https://doi.org/10.3390/agronomy10030363
APA StyleFrancesca, S., Arena, C., Hay Mele, B., Schettini, C., Ambrosino, P., Barone, A., & Rigano, M. M. (2020). The Use of a Plant-Based Biostimulant Improves Plant Performances and Fruit Quality in Tomato Plants Grown at Elevated Temperatures. Agronomy, 10(3), 363. https://doi.org/10.3390/agronomy10030363






