QTL Mapping for Drought-Responsive Agronomic Traits Associated with Physiology, Phenology, and Yield in an Andean Intra-Gene Pool Common Bean Population
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Greenhouse Experiments
2.3. Trait Evaluation
2.4. DNA Extraction and Population Genotyping
2.5. Genetic Linkage Map Construction and QTL Analysis
2.6. Consensus Map Integration and QTL Validation
3. Results
3.1. A Novel Andean Intra-Gene Pool Genetic Linkage Map of Common Bean
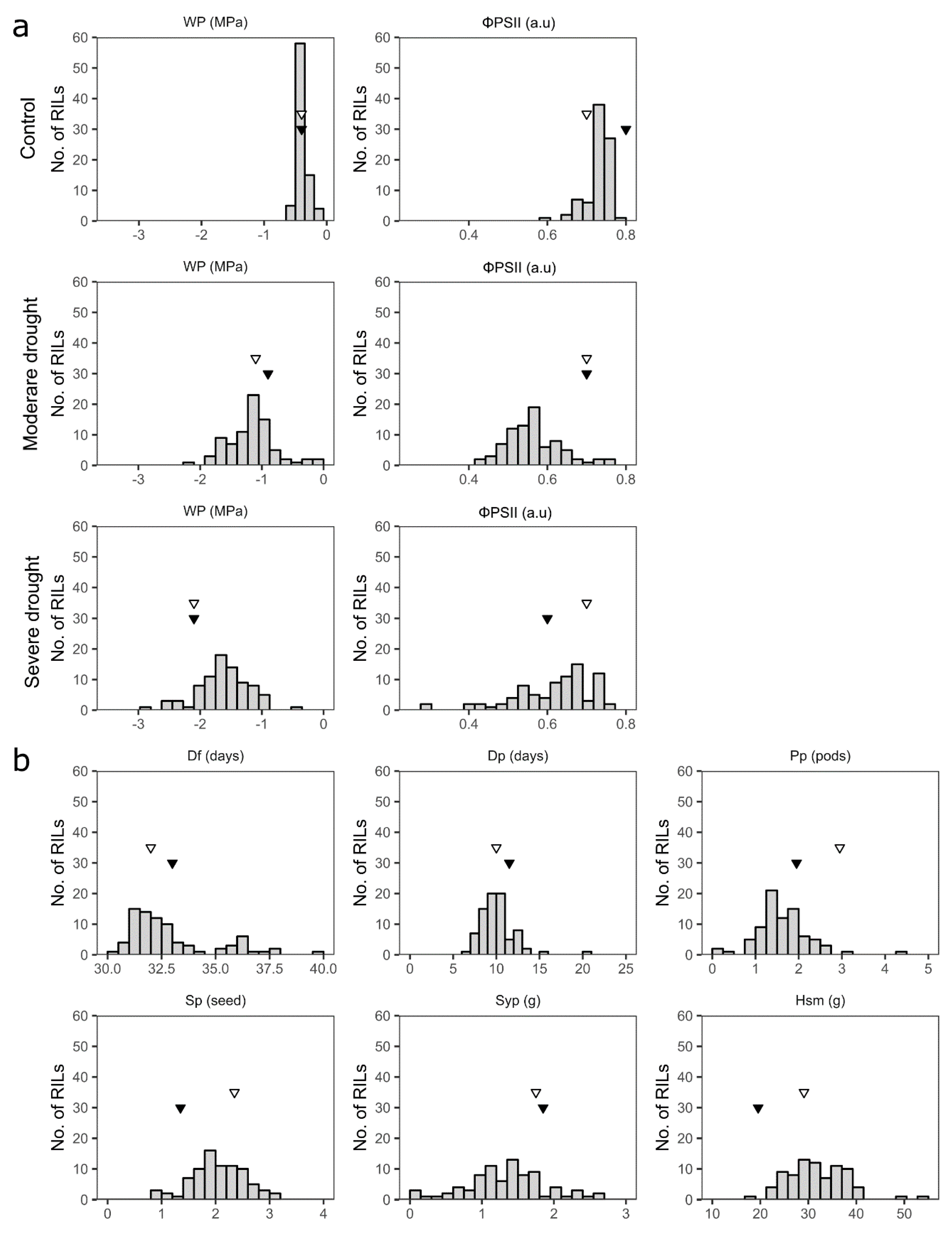
3.2. Phenotypic Variation of Drought Tolerance-Related Traits in the Parents and the Mapping Population
3.3. Drought Tolerance-Related QTL Identification Using the Novel Andean Intra-Gene Pool Genetic Linkage Map
3.3.1. Water Potential (Wp)
3.3.2. The Effective Quantum Yield of PSII (ΦPSII)
3.3.3. Days to Flowering (Df)
3.3.4. Days to Pod-Setting (Dp)
3.3.5. Pods per Plant (Pp)
3.3.6. Seeds per Pod (Sp)
3.3.7. Seed Yield per Plant (Syp)
3.3.8. One Hundred Seed Mass (Hsm)
3.3.9. Pod Harvest Index (Phi)
3.4. QTL × Environment Interaction
3.5. Transgressive Segregation
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Broughton, W.J.; Hernández, G.; Blair, M.W.; Beebe, S.E.; Gepts, P.L.; Vanderleyden, J. Beans (Phaseolus spp.)—model food legumes. Plant Soil 2003, 252, 55–128. [Google Scholar] [CrossRef]
- Beebe, S.E.; Rao, I.M.; Blair, M.W.; Acosta-Gallegos, J.A. Phenotyping common beans for adaptation to drought. Front. Physiol. 2013, 4, 35. [Google Scholar] [CrossRef] [PubMed]
- Beaver, J.S.; Osorno, J.M. Achievements and limitations of contemporary common bean breeding using conventional and molecular approaches. Euphytica 2009, 168, 145–175. [Google Scholar] [CrossRef]
- Levitt, J. Responses of Plants to Environmental Stresses; Academic Press: New York, NY, USA, 1972; p. 697. [Google Scholar]
- Polania, J.A.; Poschenrieder, C.; Beebe, S.; Rao, I.M. Effective use of water and increased dry matter partitioned to grain contribute to yield of common bean improved for drought resistance. Front. Plant Sci. 2016, 7, 660. [Google Scholar] [CrossRef] [PubMed]
- Yadav, S.; Sharma, K.D. Molecular and morphophysiological analysis of drought stress in plants. In Plant growth; Everlon, R., Ed.; IntechOpen: London, UK, 2016; pp. 149–173. [Google Scholar]
- Singh, S.P.; Terán, H.; Gutiérrez, J.P. Registration of SEA 5 and SEA 13 drought tolerant dry bean germplasm. Crop Sci. 2001, 41, 276–277. [Google Scholar] [CrossRef]
- Pérez-Vega, J.C.; Blair, M.W.; Monserrate, F.; Ligaretto, M. Evaluation of an Andean common bean reference collection under drought stress. Agron. Colomb. 2011, 29, 17–26. [Google Scholar]
- Dramadri, I.O.; Nkalubo, S.T.; Kelly, J.D. Identification of QTL associated with drought tolerance in Andean common bean. Crop Sci. 2019, 59, 1007–1020. [Google Scholar] [CrossRef]
- Mukeshimana, G.; Butare, L.; Cregan, P.B.; Blair, M.W.; Kelly, J.D. Quantitative trait loci associated with drought tolerance in common bean. Crop Sci. 2014, 54, 923–938. [Google Scholar] [CrossRef]
- Blair, M.W.; Astudillo, C.; Rengifo, J.; Beebe, S.E.; Graham, R. QTL analyses for seed iron and zinc concentrations in an intra-genepool population of Andean common beans (Phaseolus vulgaris L.). Theor. Appl. Genet. 2010, 122, 511–521. [Google Scholar] [CrossRef]
- Cichy, K.A.; Blair, M.W.; Galeano Mendoza, C.H.; Snapp, S.S.; Kelly, J.D. QTL analysis of root architecture traits and low phosphorus tolerance in an Andean bean population. Crop Sci. 2009, 49, 59–68. [Google Scholar] [CrossRef]
- Cichy, K.A.; Caldas, G.V.; Snapp, S.S.; Blair, M.W. QTL Analysis of seed iron, zinc, and phosphorus levels in an Andean bean population. Crop Sci. 2009, 49, 1742–1750. [Google Scholar] [CrossRef]
- Yuste-Lisbona, F.J.; Santalla, M.; Capel, C.; García-Alcázar, M.; De La Fuente, M.; Capel, J.; De Ron, A.M.; Lozano, R. Marker-based linkage map of Andean common bean (Phaseolus vulgaris L.) and mapping of QTLs underlying popping ability traits. BMC Plant Biol. 2012, 12, 136. [Google Scholar] [CrossRef] [PubMed]
- Blair, M.W.; Galeano, C.H.; Tovar, E.; Muñoz Torres, M.C.; Castrillón, A.V.; Beebe, S.E.; Rao, I.M. Development of a Mesoamerican intra-genepool genetic map for quantitative trait loci detection in a drought tolerant × susceptible common bean (Phaseolus vulgaris L.) cross. Mol. Breed 2012, 29, 71–88. [Google Scholar] [CrossRef] [PubMed]
- Asfaw, A.; Blair, M.W.; Struik, P.C. Multienvironment quantitative trait loci analysis for photosynthate acquisition, accumulation, and remobilization traits in common bean under drought stress. G3 (Bethesda) 2012, 2, 579–595. [Google Scholar] [CrossRef]
- Angioi, S.A.; Rau, D.; Nanni, L.; Belluci, E.; Papa, R.; Attene, G. The genetic make-up of the European landraces of the common bean. Plant Genet. Resour. 2011, 9, 197–201. [Google Scholar] [CrossRef]
- Maras, M.; Šuštar-Vozlič, J.; Kainz, W.; Meglič, V. Genetic diversity and dissemination pathways of common bean in Central Europe. J. Am. Soc. Hortic. Sci. 2013, 138, 297–305. [Google Scholar] [CrossRef]
- Maras, M.; Pipan, B.; Šuštar-Vozlič, J.; Todorović, V.; Đurić, G.; Vasić, M.; Kratovalieva, S.; Ibusoska, A.; Agić, R.; Matotan, Z.; et al. Examination of genetic diversity of common bean from the Western Balkans. J. Am. Soc. Hortic. Sci. 2015, 140, 308–316. [Google Scholar] [CrossRef]
- Sedlar, A.; Kidrič, M.; Šuštar-Vozlič, J.; Pipan, B.; Zadražnik, T.; Meglič, V. Drought stress response in agricultural plants: A case study of common bean (Phaseolus vulgaris L.). In Drought—Detection and Solutions; Ondrasek, G., Ed.; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef]
- Asfaw, A.; Ambachew, D.; Shah, T.; Blair, M.W. Trait associations in diversity panels of the two common bean (Phaseolus vulgaris L.) gene pools grown under well-watered and water-stress conditions. Front. Plant Sci. 2017, 8, 733. [Google Scholar] [CrossRef]
- Polania, J.; Rao, I.M.; Cajiao, C.; Rivera, M.; Raatz, B.; Beebe, S. Physiological traits associated with drought resistance in Andean and Mesoamerican genotypes of common bean (Phaseolus vulgaris L.). Euphytica 2016, 210, 17–29. [Google Scholar] [CrossRef]
- Sánchez-Reinoso, A.D.; Ligarreto-Moreno, G.A.; Restrepo-Díaz, H. Evaluation of drought indices to identify tolerant genotypes in common bean bush (Phaseoulus vulgaris L.). J. Integr. Agric. 2019, 18, 2–10. [Google Scholar]
- Zadražnik, T.; Hollung, K.; Egge-Jacobsen, W.; Meglič, V.; Šuštar-Vozlič, J. Differential proteomic analysis of drought stress response in leaves of common bean (Phaseolus vulgaris L.). J. Proteomics 2013, 78, 254–272. [Google Scholar] [CrossRef] [PubMed]
- Zupin, M.; Sedlar, A.; Kidrič, M.; Meglič, V. Drought-induced expression of aquaporin genes in leaves of two common bean cultivars differing in tolerance to drought stress. J. Plant Res. 2017, 130, 735–745. [Google Scholar] [CrossRef] [PubMed]
- Scholander, P.F.; Bradstreet, E.D.; Hemmingsen, E.A.; Hammel, H.T. Sap pressure in vascular plants. Science 1965, 148, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Brooks, M.D.; Niyogi, K.K. Use of a pulse-amplitude modulated chlorophyll fluorometer to study the efficiency of photosynthesis in Arabidopsis plants. In Chloroplast Research in Arabidopsis; Jarvis, R., Ed.; Humana Press: Totowa, NJ, USA, 2011; p. 432. [Google Scholar]
- Lichtenthaler, H.K.; Buschmann, C.; Knapp, M. How to correctly determine the different chlorophyll fluorescence parameters and the chlorophyll fluorescence decrease ratio RFd of leaves with the PAM fluorometer. Photosynthetica 2005, 43, 379–393. [Google Scholar] [CrossRef]
- Blair, M.W.; Torres, M.M.; Pedraza, F.; Giraldo, M.C.; Buendía, H.F.; Hurtado, N. Development of microsatellite markers for common bean (Phaseolus vulgaris L.) based on screening of non-enriched, small-insert genomic libraries. Genome. 2009, 52, 772–782. [Google Scholar] [CrossRef]
- Gaitán-Solís, E.; Duque, M.C.; Edwards, K.J.; Tohme, J. Microsatellite repeats in common bean (Phaseolus vulgaris): Isolation, characterization, and cross-species amplification in Phaseolus ssp. Crop Sci. 2002, 42, 2128–2136. [Google Scholar] [CrossRef]
- Blair, M.W.; Torres, M.M.; Giraldo, M.C.; Pedraza, F. Development and diversity of Andean-derived, gene-based microsatellites for common bean (Phaseolus vulgaris L.). BMC Plant Biol. 2009, 9, 100. [Google Scholar] [CrossRef]
- Blair, M.W.; Pedraza, F.; Buendia, H.F.; Gaitán-Solís, E.; Beebe, S.E.; Gepts, P.; Tohme, J. Development of a genome-wide anchored microsatellite map for common bean (Phaseolus vulgaris L.). Theor. Appl. Genet. 2003, 107, 1362–1374. [Google Scholar] [CrossRef]
- Blair, M.W.; Hurtado, N.; Sharma, P. New gene-derived simple sequence repeat markers for common bean (Phaseolus vulgaris L.). Mol. Ecol. Resour. 2012, 12, 661–668. [Google Scholar] [CrossRef]
- Hanai, L.R.; Santini, L.; Camargo, L.E.; Fungaro, M.H.; Gepts, P.; Tsai, S.M.; Vieira, M.L. Extension of the core map of common bean with EST-SSR, RGA, AFLP, and putative functional markers. Mol. Breed 2010, 25, 25–45. [Google Scholar] [CrossRef]
- Benchimol, L.L.; de Campos, T.; Carbonell, S.A.M.; Colombo, C.A.; Chioratto, A.F.; Formighieri, E.F.; Gouvêa, L.R.L.; de Souza, A.P. Structure of genetic diversity among common bean (Phaseolus vulgaris L.) varieties of Mesoamerican and Andean origins using new developed microsatellite markers. Genet. Resour. Crop Evol. 2007, 54, 1747–1762. [Google Scholar] [CrossRef]
- Vos, P.; Hogers, R.; Bleeker, M.; Reijans, M.; van de Lee, T.; Hornes, M.; Frijters, A.; Pot, J.; Peleman, J.; Kuiper, M. AFLP: A new technique for DNA fingerprinting. Nucleic Acids Res. 1995, 23, 4407–4414. [Google Scholar] [CrossRef] [PubMed]
- Šuštar-Vozlič, J.; Maras, M.; Javornik, B.; Meglič, V. Genetic diversity and origin of Slovene common bean (Phaseolus vulgaris L.) germplasm as revealed by AFLP markers and phaseolin analysis. J. Am. Soc. Hortic. Sci. 2006, 131, 242–249. [Google Scholar]
- Meng, L.; Li, H.; Zhang, L.; Wang, J. QTL IciMapping: Integrated software for genetic linkage map construction and quantitative trait locus mapping in biparental populations. Crop J. 2015, 3, 269–283. [Google Scholar] [CrossRef]
- Galeano, C.H.; Fernandez, A.C.; Franco-Herrera, N.; Cichy, K.A.; McClean, P.E.; Vanderleyden, J.; Blair, M.W. Saturation of an intra-gene pool linkage map: Towards a unified consensus linkage map for fine mapping and synteny analysis in common bean. PLoS ONE 2011, 6, e28135. [Google Scholar] [CrossRef]
- Kosambi, D.D. The estimation of map distances from recombination values. Ann Eugen 1943, 12, 172–175. [Google Scholar] [CrossRef]
- Wang, S.; Basten, C.J.; Zeng, Z.B. Windows QTL Cartographer 2.5. Department of Statistics, North Carolina State University, Raleigh, North Carolina, 2012. Available online: http://statgen.ncsu.edu/qtlcart/WQTLCart.htm (accessed on 3 February 2020).
- Li, S.; Wang, J.; Zhang, L. Inclusive composite interval mapping of QTL by environment interactions in biparental populations. PLoS ONE 2015, 10, e0132414. [Google Scholar] [CrossRef]
- Voorips, R.E. MapChart: Software for the Graphical Presentation of Linkage Maps and QTLs. J. Hered. 2002, 93, 77–78. [Google Scholar] [CrossRef]
- Sosnowski, O.; Charcosset, A.; Joets, J. BioMercator V3: An upgrade of genetic map compilation and quantitative trait loci meta-analysis algorithms. Bioinformatics 2012, 28, 2082–2083. [Google Scholar] [CrossRef]
- Yan, X.L.; Liao, H.; Beebe, S.E.; Blair, M.W.; Lynch, J.P. QTL mapping of root hair and acid exudation traits and their relationship to phosphorus uptake in common bean. Plant Soil 2004, 265, 17–29. [Google Scholar] [CrossRef]
- Beebe, S.E.; Rojas-Pierce, M.; Yan, X.L.; Blair, M.W.; Pedraza, F.; Muñoz, F.; Tohme, J.; Lynch, J.P. Quantitative trait loci for root architecture traits correlated with phosphorus acquisition in common bean. Crop Sci. 2006, 46, 413–423. [Google Scholar] [CrossRef]
- Pérez-Vega, E.; Pañeda, A.; Rodríguez-Suárez, C.; Campa, A.; Giraldez, R.; Ferreira, J.J. Mapping of QTLs for morpho-agronomic and seed quality traits in a RIL population of common bean (Phaseolus vulgaris L.). Theor. Appl. Genet. 2010, 120, 1367–1380. [Google Scholar] [CrossRef]
- Blair, M.W.; Iriarte, G.; Beebe, S. QTL analysis of yield traits in an advanced backcross population derived from a cultivated Andean x wild common bean (Phaseolus vulgaris L.) cross. Theor. Appl. Genet. 2006, 112, 1149–1163. [Google Scholar] [CrossRef] [PubMed]
- Pan, L.; Wang, N.; Wu, Z.; Gao, R.; Yu, X.; Zheng, Y.; Xia, Q.; Gui, S.; Chen, C. A high density genetic map derived from RAD sequencing and its application in QTL analysis of yield-related traits in Vigna unguiculata. Front Plant Sci. 2017, 8, 1544. [Google Scholar] [CrossRef] [PubMed]
- Valdisser, P.A.M.R.; Pappas, G.J., Jr.; de Menezes, I.P.P.; Müller, B.S.F.; Pereira, W.J.; Narciso, M.G.; Brondani, C.; Souza, T.L.P.O.; Borba, T.C.O.; Vianello, R.P. SNP discovery in common bean by restriction-associated DNA (RAD) sequencing for genetic diversity and population structure analysis. Mol. Genet. Genomics 2016, 291, 1277–1291. [Google Scholar] [PubMed]
- Li, J.; Cang, Z.; Jiao, F.; Bai, X.; Zhang, D.; Zhai, R. Influence of drought stress on photosynthetic characteristics and protective enzymes of potato seedling stage. J. Saudi Soc. Agric. Sci. 2017, 16, 82–88. [Google Scholar] [CrossRef]
- Santos, M.G.; Ribeiro, R.V.; Machado, E.C.; Pimentel, C. Photosynthetic parameters and leaf water potential of five common bean genotypes under mild water deficit. Biol. Plant 2009, 53, 229–236. [Google Scholar] [CrossRef]
- Anyia, A.O.; Herzog, H. Genotypic variability in drought performance and recovery in cowpea under controlled environment. J. Agron. Crop Sci. 2004, 190, 151–159. [Google Scholar] [CrossRef]
- Xu, M.; Yamagishi, N.; Zhao, C.; Takeshima, R.; Kasai, M.; Watanabe, S.; Kanazawa, A.; Yoshikawa, N.; Liu, B.; Yamada, T.; et al. Soybean-specific E1 family of floral repressors controls night-break responses through down-regulation of FLOWERING LOCUS T orthologs. Plant Physiol. 2015, 168, 1735–1746. [Google Scholar] [CrossRef]
- McKay, J.K.; Richards, J.; Nemali, K.S.; Sen, S.; Mitchell-Olds, T.; Boles, S.; Stahl, E.A.; Wayne, T.; Juenger, T.E. Genetics of drought adaptation in Arabidopsis thaliana II. QTL analysis of a new mapping population, KAS-1 x TSU-1. Evol. Int. J. Org. Evol. 2008, 62, 3014–3026. [Google Scholar] [CrossRef]
- Ramirez-Vallejo, P.; Kelly, J.D. Traits related to drought resistance in common bean. Euphytica 1998, 99, 127–136. [Google Scholar] [CrossRef]
- Trapp, J.J.; Urrea, C.A.; Cregan, P.B.; Miklas, P.N. Quantitative trait loci for yield under multiple stress and drought conditions in a dry bean population. Crop Sci. 2015, 55, 1596–1607. [Google Scholar] [CrossRef]
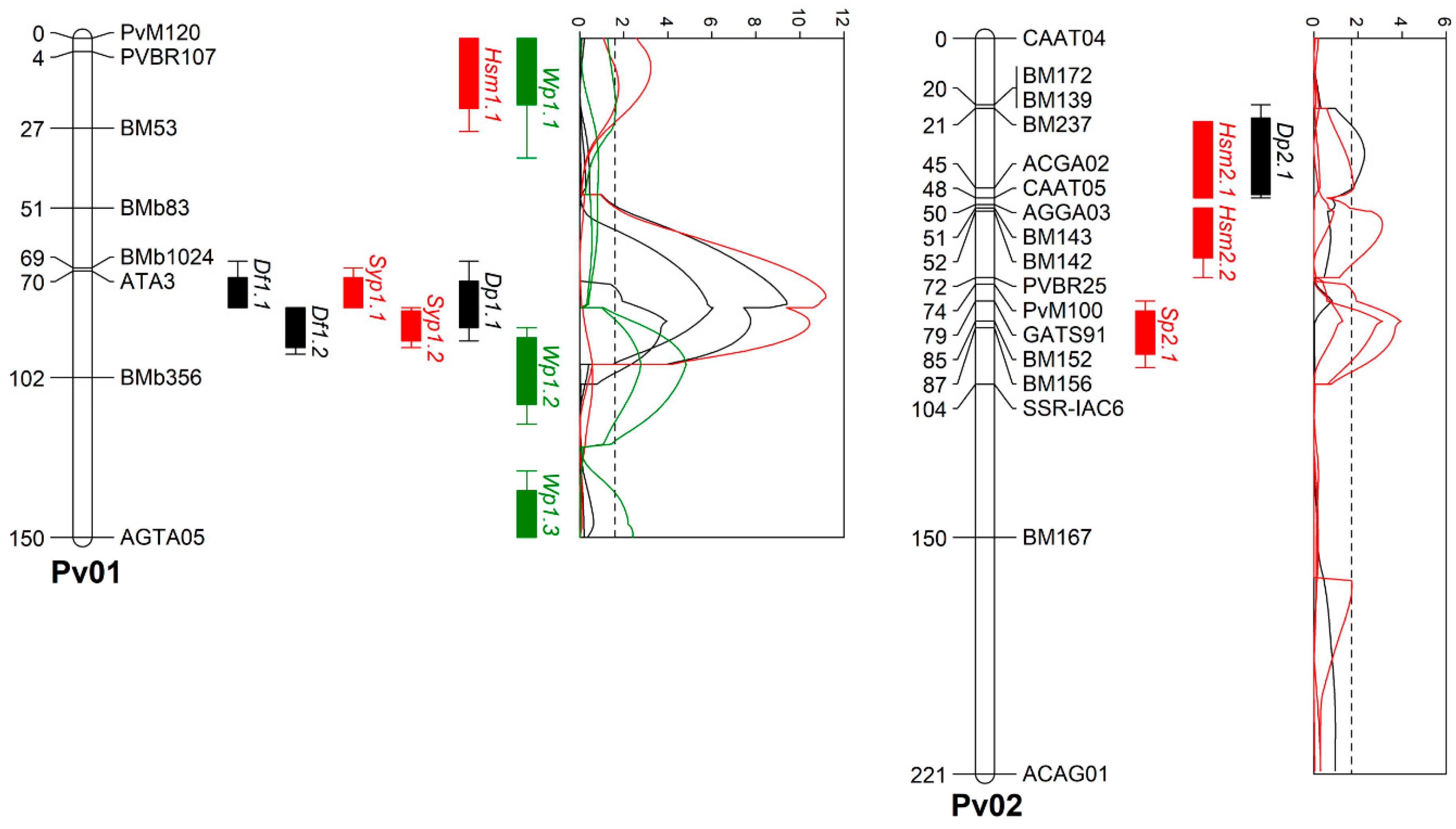

| LG 1 | SSR | AFLP | Total No. of Markers | Total Linkage Group Length (cM) | Average Distance all Markers (cM) | Anchor Markers | Total Linkage Group Length in Consensus Map (cM) |
|---|---|---|---|---|---|---|---|
| Pv01 | 7 | 1 | 8 | 149.9 | 21.4 | 8 | 191.9 |
| Pv02 | 14 | 5 | 19 | 221.1 | 12.3 | 12 | 192.4 |
| Pv03 | 3 | 0 | 3 | 65.9 | 33.0 | 3 | 235.8 |
| Pv04 | 6 | 6 | 12 | 194.4 | 17.7 | 4 | 228.7 |
| Pv05 | 10 | 3 | 13 | 56.4 | 4.7 | 8 | 132.9 |
| Pv06 | 7 | 4 | 11 | 40.3 | 4.0 | 6 | 129 |
| Pv07 | 7 | 0 | 7 | 172.2 | 28.7 | 6 | 180 |
| Pv08 | 11 | 3 | 14 | 78.4 | 6.0 | 7 | 166.1 |
| Pv09 | 14 | 1 | 15 | 135.5 | 9.7 | 7 | 128.2 |
| Pv10 | 13 | 2 | 15 | 100.1 | 7.1 | 9 | 131.9 |
| Pv11 | 5 | 1 | 6 | 74.2 | 14.8 | 5 | 163.8 |
| Total | 97 | 26 | 123 | 1288.4 | 11.5 | 73 | 1880.7 |
| Trait | QTL | Treatment 1 | LG | Position | Left Marker | Right Marker | LOD 2 | LOD Threshold | LOD (A) 2 | LOD (AbyE) 2 | PVE 3 | PVE (A) 3 | PVE (AbyE) 3 | Add 4 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Wp | Wp5.2 | Control | 5 | 44 | AGTC02 | BMd53 | 4.3 | 3.4 | 2.0 | 2.2 | 62.8 | 19.2 | 43.6 | 0.0 |
| Wp6.1 | Control | 6 | 13 | SSR-IAC47 | BMb519 | 3.7 | 3.4 | 0.0 | 3.7 | 16.7 | 0.3 | 16.4 | 0.0 | |
| ΦPSII | ΦPSII7.1 | Severe | 7 | 95 | BMb502 | BM150 | 3.3 | 3.1 | 1.9 | 1.5 | 9.3 | 6.3 | 3.0 | 0.0 |
| ΦPSII10.1 | Severe | 10 | 23 | BM212 | BMd42 | 3.3 | 3.1 | 2.0 | 1.2 | 9.0 | 6.6 | 2.4 | 0.0 | |
| ΦPSII11.1 | Severe | 11 | 0 | BMd22 | BM239 | 4.6 | 3.1 | 3.2 | 1.5 | 13.7 | 11.1 | 2.6 | 0.1 | |
| Df | Df1.1 | 1 | 79 | BMb356 | ATA3 | 13.0 | 3.3 | 11.0 | 2.1 | 30.1 | 29.1 | 1.0 | −1.1 | |
| Dp | Dp1.1 | 1 | 81 | ATA3 | BMb1024 | 5.9 | 3.2 | 4.5 | 1.4 | 13.6 | 13.1 | 0.5 | −0.6 | |
| Sp | Sp2.1 | 2 | 86 | BM236 | BM156 | 4.9 | 3.3 | 4.4 | 0.5 | 11.3 | 11.3 | 0.0 | 0.2 | |
| Syp | Syp1.1 | 1 | 78 | BMb356 | ATA3 | 10.4 | 3.4 | 0.3 | 10.1 | 3.3 | 1.7 | 1.6 | 0.1 | |
| Syp2.1 | 2 | 4 | CAAT04 | BM139 | 3.7 | 3.4 | 0.9 | 2.8 | 4.8 | 4.3 | 0.5 | 0.1 | ||
| Syp4.1 | 4 | 187 | CCTA05 | AGGT07 | 3.7 | 3.4 | 1.9 | 1.8 | 10.7 | 7.8 | 2.9 | −0.2 | ||
| Hsm | Hsm2.2 | 2 | 53 | BM142 | PVBR25 | 4.1 | 3.3 | 3.2 | 0.9 | 14.2 | 10.7 | 3.5 | −2.0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sedlar, A.; Zupin, M.; Maras, M.; Razinger, J.; Šuštar-Vozlič, J.; Pipan, B.; Meglič, V. QTL Mapping for Drought-Responsive Agronomic Traits Associated with Physiology, Phenology, and Yield in an Andean Intra-Gene Pool Common Bean Population. Agronomy 2020, 10, 225. https://doi.org/10.3390/agronomy10020225
Sedlar A, Zupin M, Maras M, Razinger J, Šuštar-Vozlič J, Pipan B, Meglič V. QTL Mapping for Drought-Responsive Agronomic Traits Associated with Physiology, Phenology, and Yield in an Andean Intra-Gene Pool Common Bean Population. Agronomy. 2020; 10(2):225. https://doi.org/10.3390/agronomy10020225
Chicago/Turabian StyleSedlar, Aleš, Mateja Zupin, Marko Maras, Jaka Razinger, Jelka Šuštar-Vozlič, Barbara Pipan, and Vladimir Meglič. 2020. "QTL Mapping for Drought-Responsive Agronomic Traits Associated with Physiology, Phenology, and Yield in an Andean Intra-Gene Pool Common Bean Population" Agronomy 10, no. 2: 225. https://doi.org/10.3390/agronomy10020225
APA StyleSedlar, A., Zupin, M., Maras, M., Razinger, J., Šuštar-Vozlič, J., Pipan, B., & Meglič, V. (2020). QTL Mapping for Drought-Responsive Agronomic Traits Associated with Physiology, Phenology, and Yield in an Andean Intra-Gene Pool Common Bean Population. Agronomy, 10(2), 225. https://doi.org/10.3390/agronomy10020225







