Supplementary Far-Red Light Did Not Affect Tomato Plant Growth or Yield under Mediterranean Greenhouse Conditions
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Setup
2.2. Plant Material and Growing Conditions
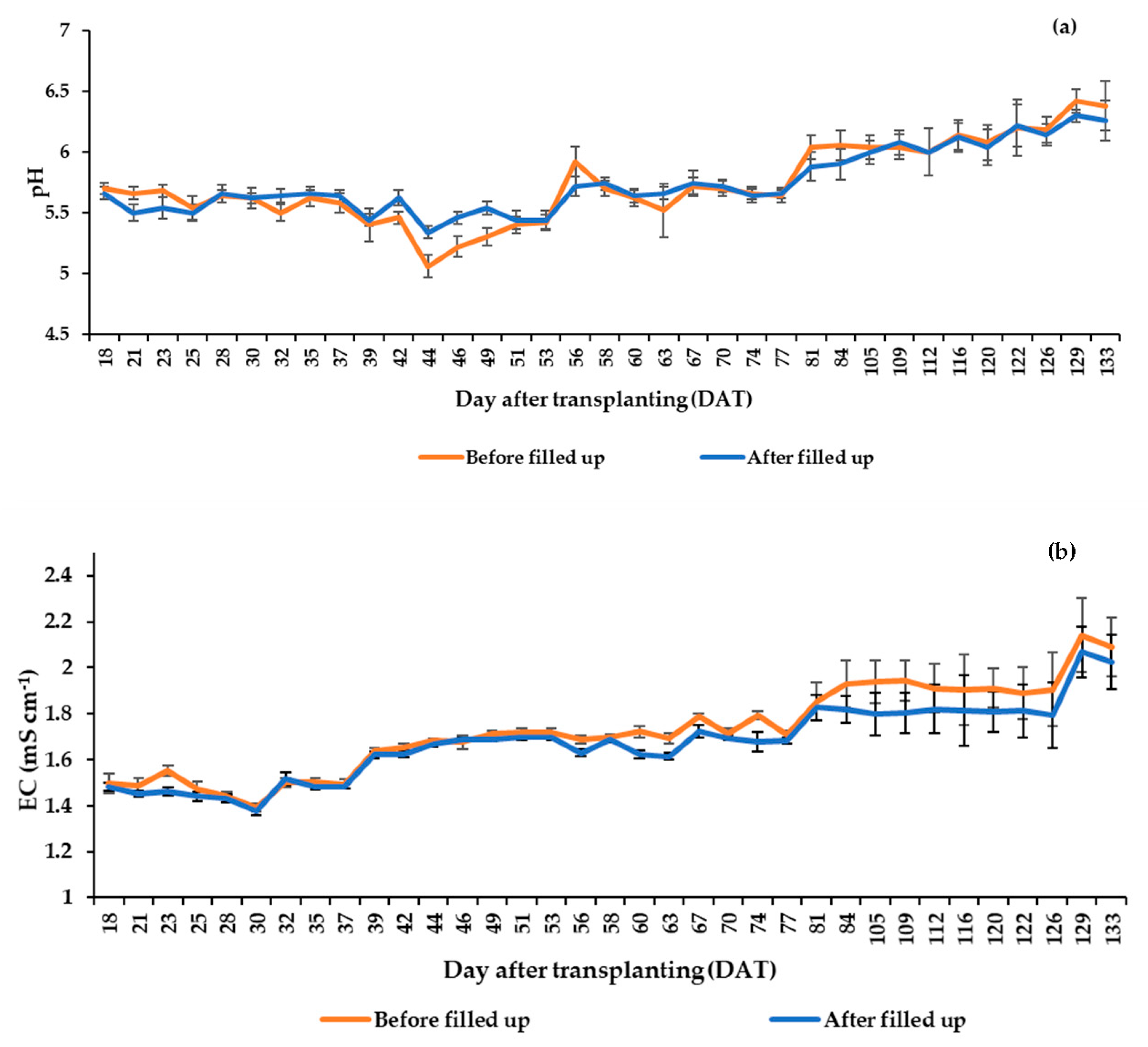
2.3. Nutrient Solution (NS) Management
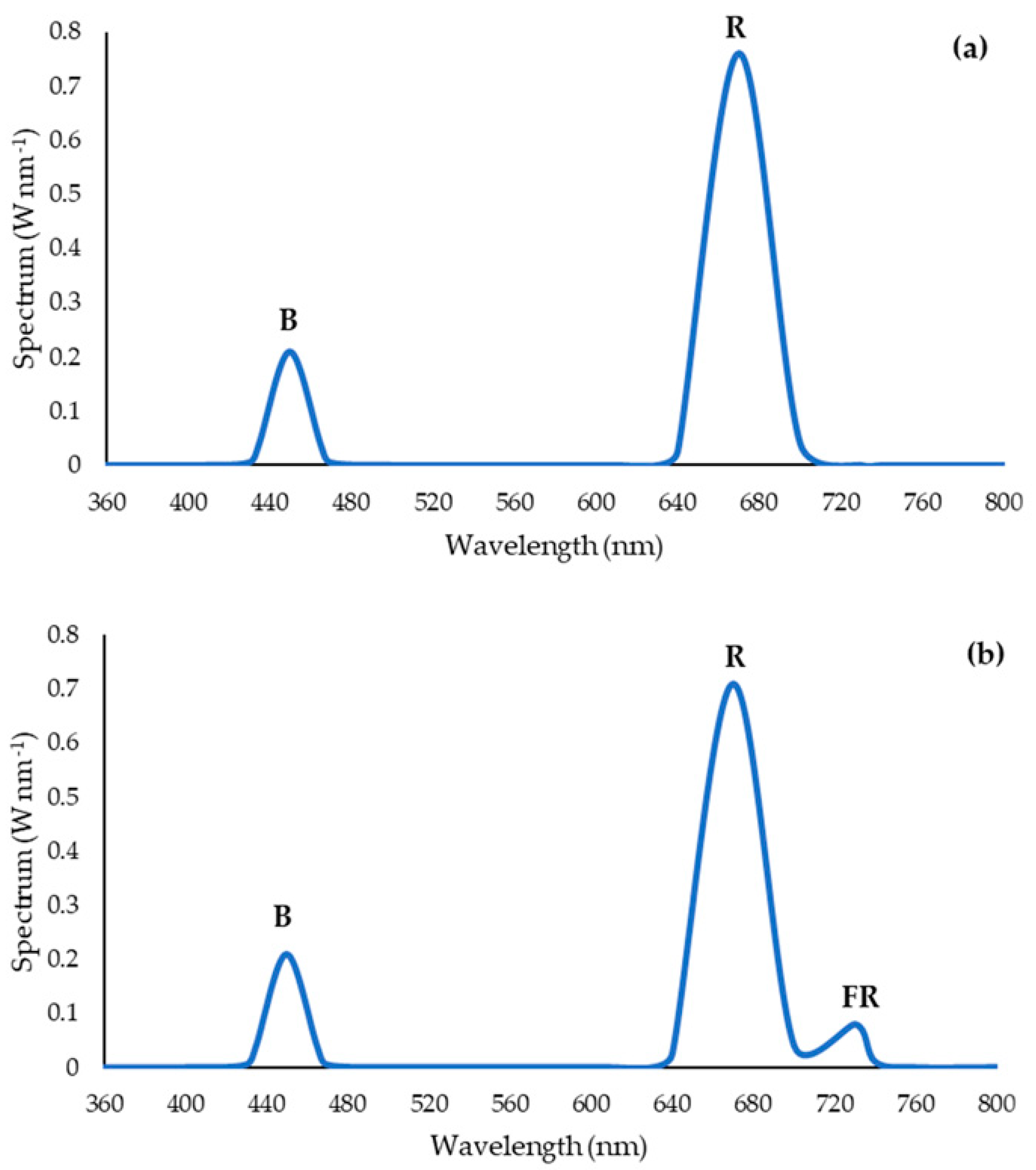
2.4. Supplementary Light Treatments
2.5. Daily Light Integral and Total Light Integral
2.6. Growth Analysis, Yield, Fruit Dry Weight and Total Soluble Solids
2.7. Gas Exchange Measurements
2.8. Experimental Design and Statistical Analysis
3. Results
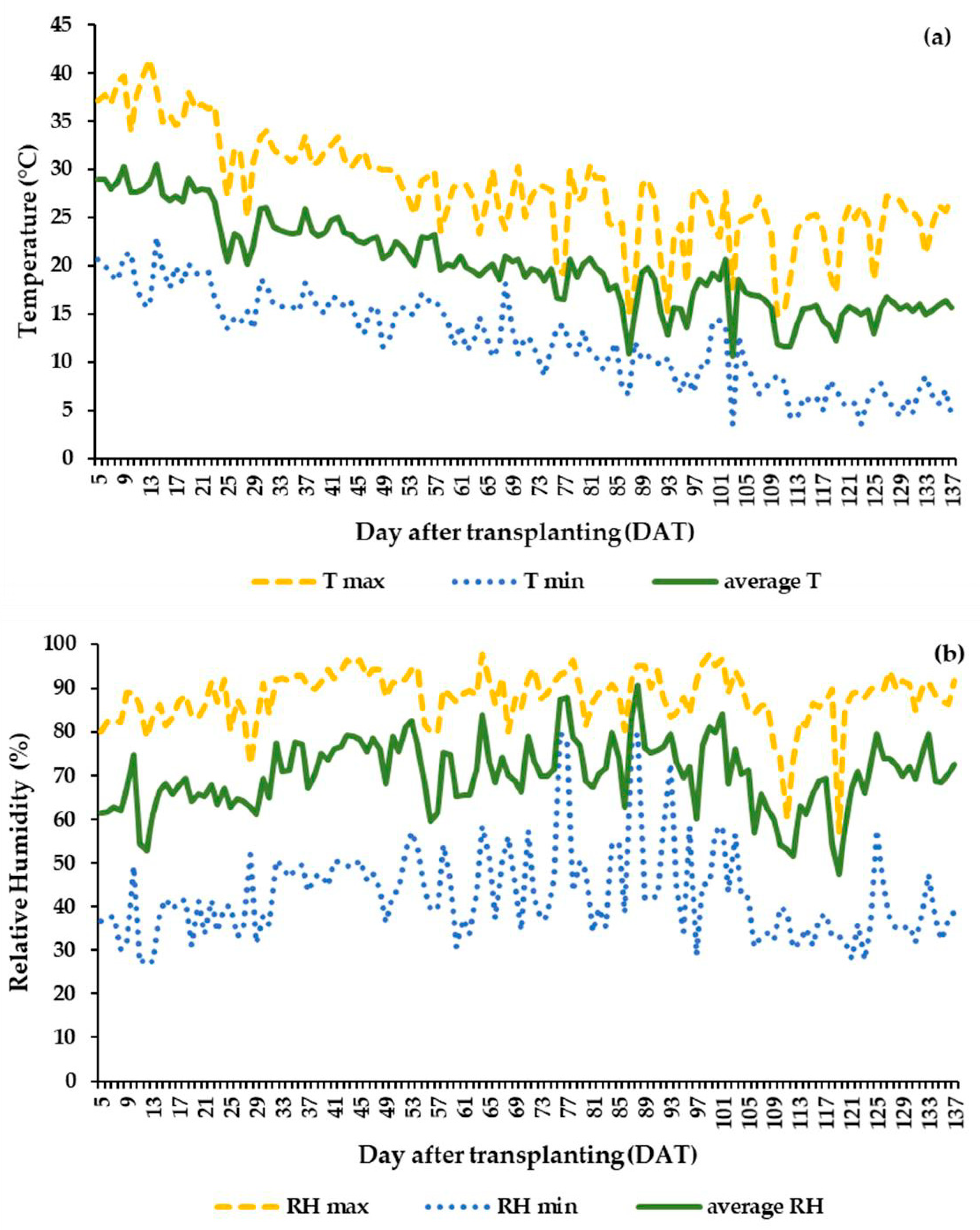
3.1. Greenhouse Climatic Conditions
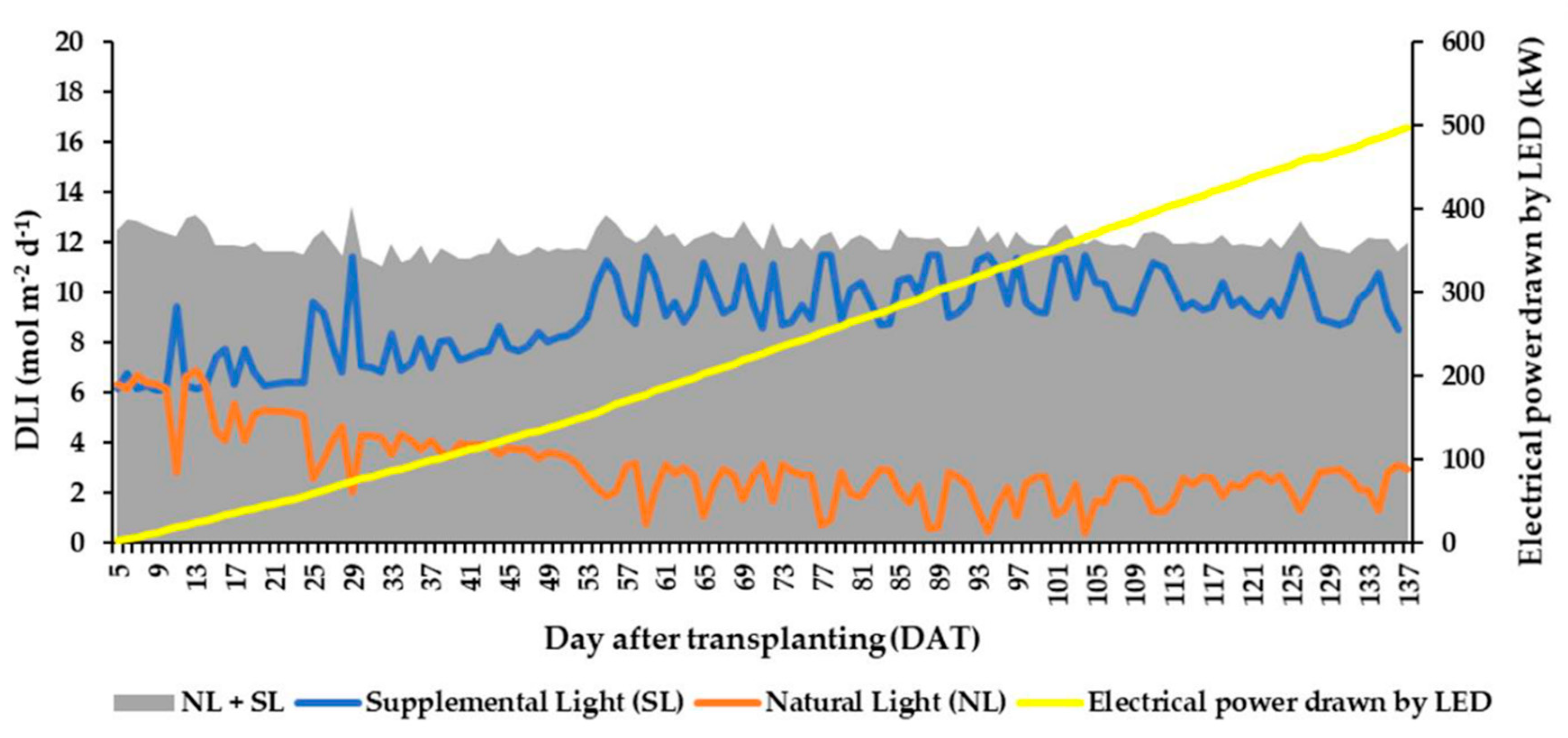
3.2. Daily Light Integral (DLI) and Total Light Integral (TLI)
3.3. Growth Analysis and Gas Exchange
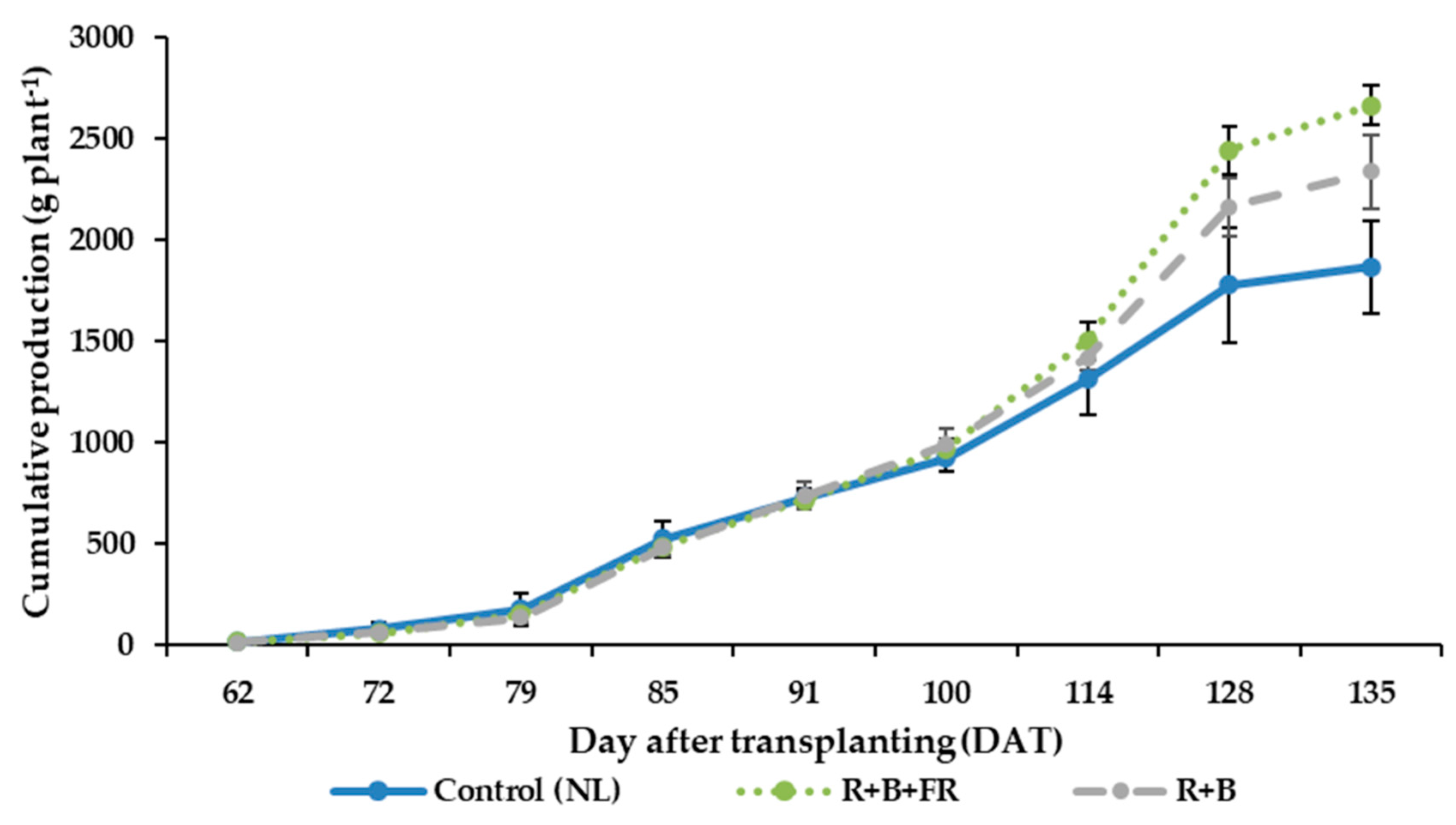
3.4. Fruit Quantitavive and Qualitative Analysis
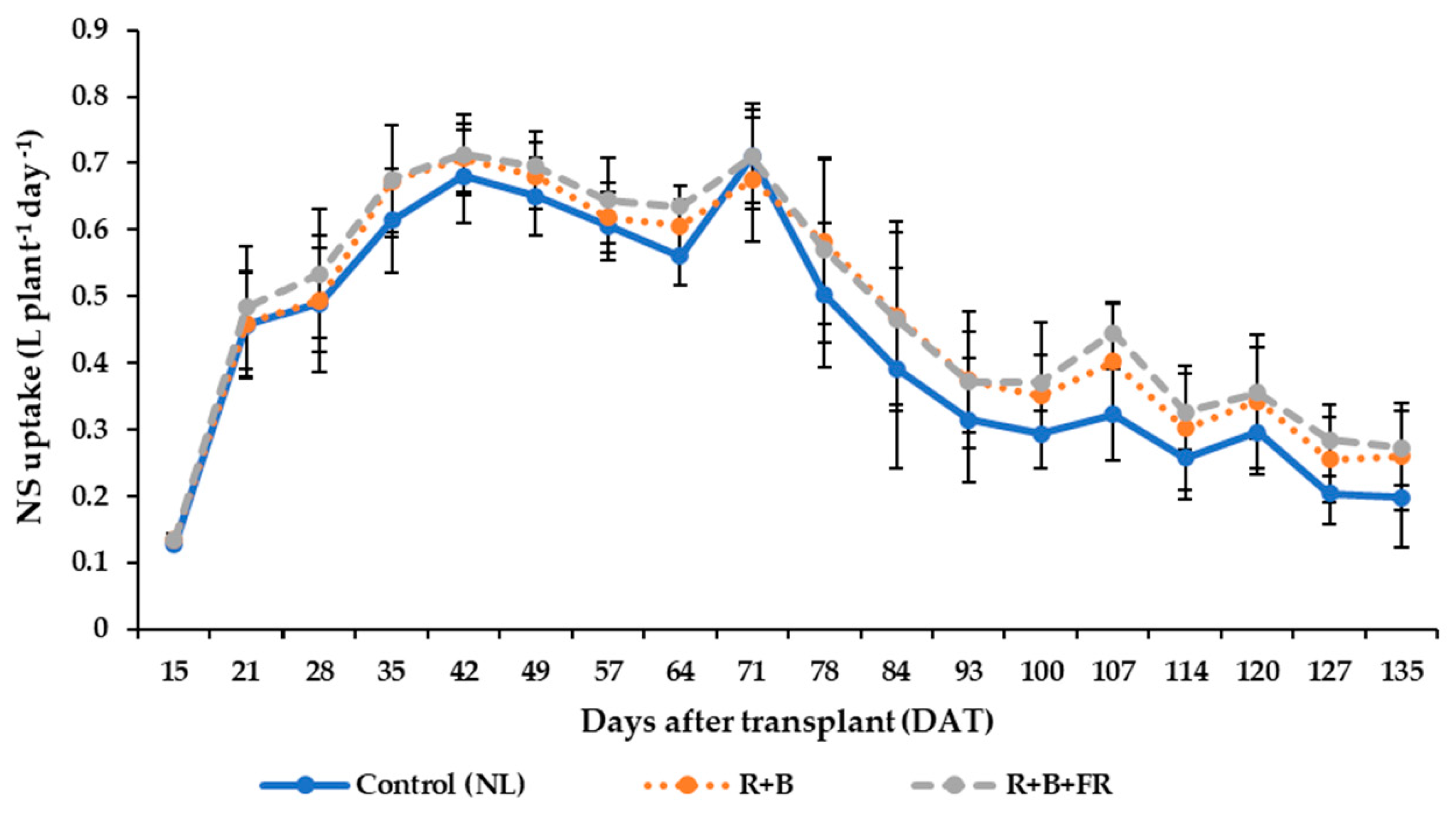
3.5. Nutrient Solution Consumption and Water Use Efficiency
4. Discussion
4.1. Supplemental Far-Red Did Not Impact Tomato Growth
4.2. Supplementary Interlight Increased Tomato Yield
4.3. Supplementary Interlight Did Not Influence Tomato Quality
4.4. Nutrient Solution Uptake and Water Use Efficiency (WUE)
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kubota, C.; de Gelder, A.; Peet, M.M. Grenhouse Tomato Production. In Tomatoes; CABI Publishing: Wallingford, UK, 2018; pp. 276–313. ISBN 9781780641942. [Google Scholar]
- Palmitessa, O.D.; Paciello, P.; Santamaria, P. Supplemental LED Increases Tomato Yield in mediterranean Semi-Closed Greenhouse. Agronomy 2020, 10, 1353. [Google Scholar] [CrossRef]
- Paponov, M.; Kechasov, D.; Lacek, J.; Verheul, M.J.; Paponov, I.A. Supplemental Light-Emitting Diode Inter-Lighting Increases Tomato Fruit Growth Through Enhanced Photosynthetic Light Use Efficiency and modulated Root Activity. Front. Plant Sci. 2019, 10, 1656. [Google Scholar] [CrossRef] [PubMed]
- Buttaro, D.; Santamaria, P.; Signore, A.; Cantore, V.; Boari, F.; Montesano, F.F.; Parente, A. Irrigation management of Greenhouse Tomato and Cucumber Using Tensiometer: Effects on Yield, Quality and Water Use. Agric. Agric. Sci. Procedia 2015, 4, 440–444. [Google Scholar] [CrossRef]
- Hovi-Pekkanen, T.; Tahvonen, R. Effects of interlighting on yield and external fruit quality in year-round cultivated cucumber. Sci. Hortic. 2008, 116, 152–161. [Google Scholar] [CrossRef]
- Hovi, T.; Näkkilä, J.; Tahvonen, R. Interlighting improves production of year-round cucumber. Sci. Hortic. 2004, 102, 283–294. [Google Scholar] [CrossRef]
- Särkkä, L.E.; Jokinen, K.; Ottosen, C.O.; Kaukoranta, T. Effects of HPS and LED lighting on cucumber leaf photosynthesis, light quality penetration and temperature in the canopy, plant morphology and yield. Agric. Food Sci. 2017, 26, 101–109. [Google Scholar] [CrossRef]
- Bantis, F.; Smirnakou, S.; Ouzounis, T.; Koukounaras, A.; Ntagkas, N.; Radoglou, K. Current status and recent achievements in the field of horticulture with the use of light-emitting diodes (LEDs). Sci. Hortic. 2018, 235, 437–451. [Google Scholar] [CrossRef]
- Kusuma, P.; Pattison, P.M. From physics to fixtures to food: Current and potential LED efficacy. Hortic. Res. 2020, 7, 1–9. [Google Scholar] [CrossRef]
- Tennessen, D.J.; Singsaas, E.L.; Sharkey, T.D. Light-emitting diodes as a light source for photosynthesis research. Photosynth. Res. 1994, 39, 85–92. [Google Scholar] [CrossRef]
- Lin, K.H.; Huang, M.Y.; Huang, W.D.; Hsu, M.H.; Yang, Z.W.; Yang, C.M. The effects of red, blue, and white light-emitting diodes on the growth, development, and edible quality of hydroponically grown lettuce (Lactuca sativa L. var. capitata). Sci. Hortic. 2013, 150, 86–91. [Google Scholar] [CrossRef]
- McCree, K.J. The action spectrum, absorptance and quantum yield of photosynthesis in crop plants. Agric. Meteorol. 1972, 9, 191–216. [Google Scholar] [CrossRef]
- Chen, M.; Blankenship, R.E. Expanding the solar spectrum used by photosynthesis. Trends Plant Sci. 2011, 16, 427–431. [Google Scholar] [CrossRef] [PubMed]
- Zhen, S.; Bugbee, B. Far-red photons have equivalent efficiency to traditional photosynthetic photons: Implications for redefining photosynthetically active radiation. Plant Cell Environ. 2020, 43, 1259–1272. [Google Scholar] [CrossRef] [PubMed]
- Zhen, S.; van Iersel, M.W. Far-red light is needed for efficient photochemistry and photosynthesis. J. Plant. Physiol. 2017, 209, 115–122. [Google Scholar] [CrossRef]
- Kim, H.; Yang, T.; Choi, S.; Wang, Y.; Lin, M.; Liceaga, A.M. Supplemental intracanopy far-red radiation to red LED light improves fruit quality attributes of greenhouse tomatoes. Sci. Hortic. 2020, 261, 108985. [Google Scholar] [CrossRef]
- Ji, Y.; Ouzounis, T.; Courbier, S.; Kaiser, E.; Nguyen, P.T.; Schouten, H.J.; Visser, R.G.F.; Pierik, R.; Marcelis, L.F.M.; Heuvelink, E. Far-red radiation increases dry mass partitioning to fruits but reduces Botrytis cinerea resistance in tomato. Environ. Exper. Bot. 2019, 103889. [Google Scholar] [CrossRef]
- Hoagland, D.R.; Arnon, D.I. The Water-Culture Method for Growing Plants without Soil. Californian Agricultural Experimental Station. Circular 347; University of California: Berkeley, CA, USA, 1950; p. 32. [Google Scholar]
- Faust, J.E.; Holcombe, V.; Rajapakse, N.C.; Layne, D.R. The effect of daily light integral on bedding plant growth and flowering. HortScience 2005, 40, 645–649. [Google Scholar] [CrossRef]
- Jiang, C.; Johkan, M.; Maruo, T.; Hohjo, M.; Tsukagoshi, S.; Ebihara, M.; Nakaminami, A. Effect of supplemental far-red light with blue and red LED lamps on leaf photosynthesis, stomatal regulation and plant development of protected cultivated tomato. Acta Hortic. 2018, 533–540. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, Y.; Yang, Q.; LI, T. Overhead supplemental Far-red light stimulates tomato growth under intra-canopy lighting with LEDs. J. Integr. Agric. 2018, 17, 62–69. [Google Scholar] [CrossRef]
- Hao, X.; Little, C.; Zheng, J.M.; Cao, R. Far-red LEDs improve fruit production in greenhouse tomato grown under high-pressure sodium lighting. Acta Hortic. 2016, 1134, 95–102. [Google Scholar] [CrossRef]
- Kalaitzoglou, P.; van Ieperen, W.; Harbinson, J.; van der Meer, M.; Martinakos, S.; Weerheim, K.; Nicole, C.C.S.; Marcelis, L.F.M. Effects of continuous or end-of-day far-red light on tomato plant growth, morphology, light absorption, and fruit production. Front. Plant Sci. 2019, 10, 322. [Google Scholar] [CrossRef] [PubMed]
- Cao, K.; Yu, J.; Xu, D.; Ai, K.; Bao, E.; Zou, Z. Exposure to lower red to far-red light ratios improve tomato tolerance to salt stress. BMC Plant Biol. 2018, 18–92. [Google Scholar] [CrossRef] [PubMed]
- Demotes-Mainard, S.; Péron, T.; Corot, A.; Bertheloot, J.; Le Gourrierec, J.; Pelleschi-Travier, S.; Crespel, L.; Morel, P.; Huché-Thélier, L.; Boumaza, R.; et al. Plant responses to red and far-red lights, applications in horticulture. Environ. Exp. Bot. 2016, 121, 4–21. [Google Scholar] [CrossRef]
- Arena, C.; Tsonev, T.; Doneva, D.; De Micco, V.; Michelozzi, M.; Brunetti, C.; Centritto, M.; Fineschi, S.; Velikova, V.; Loreto, F. The effect of light quality on growth, photosynthesis, leaf anatomy and volatile isoprenoids of a monoterpene-emitting herbaceous species (Solanum lycopersicum L.) and an isoprene-emitting tree (Platanus orientalis L.). Environ. Exp. Bot. 2016, 130, 122–132. [Google Scholar] [CrossRef]
- Palmitessa, O.D.; Montesano, F.F.; Serio, F.; Signore, A.; Santamaria, P. Supplemental lighting with LED for efficient year- round production of soilless tomato in a Mediterranean greenhouse. In Proceedings of the VI International Symposium on Applications of Modelling as an Innovative Technology in the Horticultural Supply Chain—Model.-IT 2019, Molfetta, Italy, 9–12 June 2019. (in press).
- Dorais, M.; Gosselin, A.; Trudel, M.J. Annual greenhouse tomato production under a sequential intercropping system using supplemental light. Sci. Hortic. 1991, 45, 225–234. [Google Scholar] [CrossRef]
- Paucek, I.; Pennisi, G.; Pistillo, A.; Appolloni, E.; Crepaldi, A.; Calegari, B.; Spinelli, F.; Cellini, A.; Gabarrell, X.; Orsini, F.; et al. Supplementary LED interlighting improves yield and precocity of greenhouse tomatoes in the mediterranean. Agronomy 2020, 10, 1002. [Google Scholar] [CrossRef]
- Grimstad, S.O. Supplementary lighting of early tomatoes after planting out in glass and acrylic greenhouses. Sci. Hortic. 1987, 33, 189–196. [Google Scholar] [CrossRef]
- Lu, N.; Maruo, T.; Johkan, M.; Hohjo, M. Effects of supplemental lighting with light-emitting diodes (Leds) on tomato yield and quality of single-truss tomato plants grown at high planting density. Environ. Control. Biol. 2012, 50, 63–74. [Google Scholar] [CrossRef]
- Hogewoning, S.W.; Trouwborst, G.; Maljaars, H.; Poorter, H.; van Ieperen, W.; Harbinson, J. Blue light dose-responses of leaf photosynthesis, morphology, and chemical composition of Cucumis sativus grown under different combinations of red and blue light. J. Exp. Bot. 2010, 61, 3107–3117. [Google Scholar] [CrossRef]
- Driever, S.; Labrie, C.; Verkerke, W. Increasing the level of vitamin C in tomato through light treatments during truss development. Presented at the Wageningen UR Greenhouse Horticolture; Available online: https://edepot.wur.nl/166504 (accessed on 8 April 2011).
- Sonneveld, C.; Voogt, W. Nutrient solutions for soilless cultures. In Plant Nutrition of Greenhouse Crops; Springer Publishing: Dordrecht, The Netherlands, 2009; pp. 257–275. ISBN 9789048125326. [Google Scholar]

| Leaves FW | Stem FW | Fruits FW | Leaves DW | Stem DW | Fruits DW | |
|---|---|---|---|---|---|---|
| Supplemental light (SL) | g plant−1 | g 100 g FW−1 | ||||
| Control (NL) | 692.4 ± 102.4 | 380.8 ± 47.7 | 380.8 ± 39.9 | 7.6 ± 0.3 | 8.3 ± 0.8 | 4.3 ± 0.8 |
| R + B | 623.9 ± 88.7 | 368.3 ± 35.6 | 374.8 ± 76.3 | 8.0 ± 0.1 | 7.7 ± 0.6 | 5.4 ± 0.6 |
| R + B + FR | 561.5 ± 63.8 | 375.8 ± 79.2 | 411.7 ± 67.0 | 8.5 ± 0.2 | 8.4 ± 0.7 | 4.9 ± 0.6 |
| Significance (1) | ||||||
| NL vs. SL | NS | NS | NS | * | NS | NS |
| R + B vs. R + B + FR | NS | NS | NS | ** | NS | NS |
| Net Photosynthetic Rate (A) | Stomatal Conductance (gs) | Transpiration Rate (E) | Intercellular CO2 Concentration (Ci) | |
|---|---|---|---|---|
| Supplementary light (SL) | µmol CO2 m−2 s−1 | mol H2O m−2 s−1 | mmol H2O m−2 s−1 | µmol CO2 mol−1 |
| Control (NL) | 9.0 ± 0.3 | 0.338 ± 0.010 | 3.72 ± 0.64 | 333 ± 6 |
| R + B | 8.4 ± 0.7 | 0.350 ± 0.011 | 3.75 ± 0.69 | 340 ± 5 |
| R + B + FR | 8.4 ± 0.7 | 0.353 ± 0.010 | 3.68 ± 0.56 | 340 ± 6 |
| Significance (1) | ||||
| NL vs. SL | NS | NS | NS | NS |
| R + B vs. R + B + FR | NS | NS | NS | NS |
| Yield | Marketable Yield | Harvested Fruit Number | Average Fruit Weight | Potential Production | ||
|---|---|---|---|---|---|---|
| Yield | Fruit Number | |||||
| Supplementary | kg plant−1 | kg plant−1 | n. plant−1 | g | kg plant−1 | n. plant−1 |
| light (SL) | ||||||
| Control (NL) | 1.89 ± 0.23 | 1.85 ± 0.23 | 22.7 ± 3.8 | 81.3 ± 3.2 | 2.07 ± 0.21 | 25.9 ± 3.8 |
| R + B | 2.26 ± 0.25 | 2.24 ± 0.24 | 26.2 ± 3.7 | 85.5 ± 4.3 | 2.54 ± 0.29 | 31.1 ± 3.2 |
| R + B + FR | 2.48 ± 0.28 | 2.46 ± 0.30 | 28.4 ± 3.9 | 86.5 ± 4.4 | 2.84 ± 0.32 | 34.5 ± 5.3 |
| Significance (1) | ||||||
| NL vs. SL | * | * | * | NS | * | * |
| R + B vs. R + B + FR | NS | NS | NS | NS | NS | NS |
| NS Uptake | WUE | |
|---|---|---|
| Supplementary light (SL) | L plant−1 | L kg−1 |
| Control (NL) | 56.1 ± 3.4 | 30.1 ± 3.1 |
| R + B | 64.6 ± 7.2 | 26.1 ± 3.8 |
| R + B + FR | 62.1 ± 1.3 | 27.4 ± 2.5 |
| Significance (1) | ||
| NL vs. SL | NS | NS |
| R + B vs. R + B + FR | NS | NS |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palmitessa, O.D.; Leoni, B.; Montesano, F.F.; Serio, F.; Signore, A.; Santamaria, P. Supplementary Far-Red Light Did Not Affect Tomato Plant Growth or Yield under Mediterranean Greenhouse Conditions. Agronomy 2020, 10, 1849. https://doi.org/10.3390/agronomy10121849
Palmitessa OD, Leoni B, Montesano FF, Serio F, Signore A, Santamaria P. Supplementary Far-Red Light Did Not Affect Tomato Plant Growth or Yield under Mediterranean Greenhouse Conditions. Agronomy. 2020; 10(12):1849. https://doi.org/10.3390/agronomy10121849
Chicago/Turabian StylePalmitessa, Onofrio Davide, Beniamino Leoni, Francesco Fabiano Montesano, Francesco Serio, Angelo Signore, and Pietro Santamaria. 2020. "Supplementary Far-Red Light Did Not Affect Tomato Plant Growth or Yield under Mediterranean Greenhouse Conditions" Agronomy 10, no. 12: 1849. https://doi.org/10.3390/agronomy10121849
APA StylePalmitessa, O. D., Leoni, B., Montesano, F. F., Serio, F., Signore, A., & Santamaria, P. (2020). Supplementary Far-Red Light Did Not Affect Tomato Plant Growth or Yield under Mediterranean Greenhouse Conditions. Agronomy, 10(12), 1849. https://doi.org/10.3390/agronomy10121849










