The Comparison of Constant and Dynamic Red and Blue Light Irradiation Effects on Red and Green Leaf Lettuce
Abstract
1. Introduction
2. Materials and Methods
2.1. Growing Conditions
2.2. Lighting Treatments
2.3. Biometric and Non-Destructive Measurements
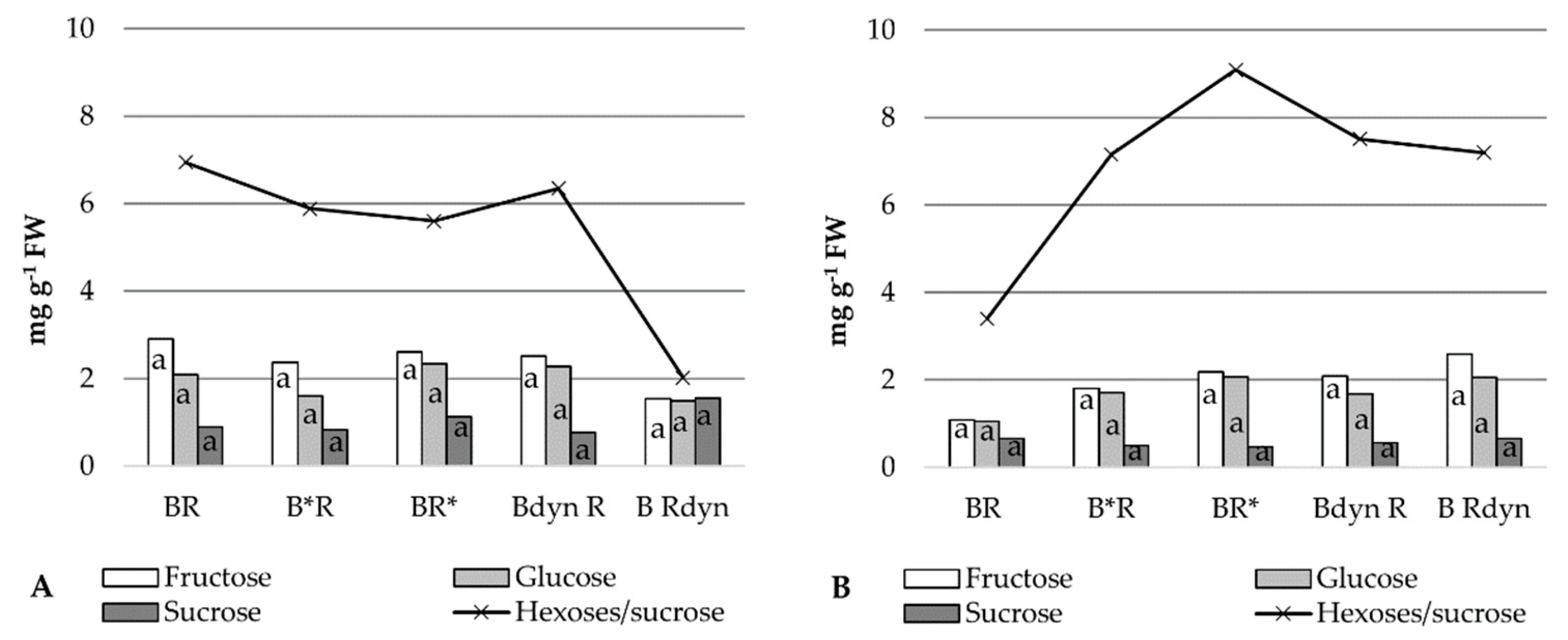
2.4. Soluble Sugars
2.5. Antioxidant Properties
2.6. Determination of Macro- and Microelements
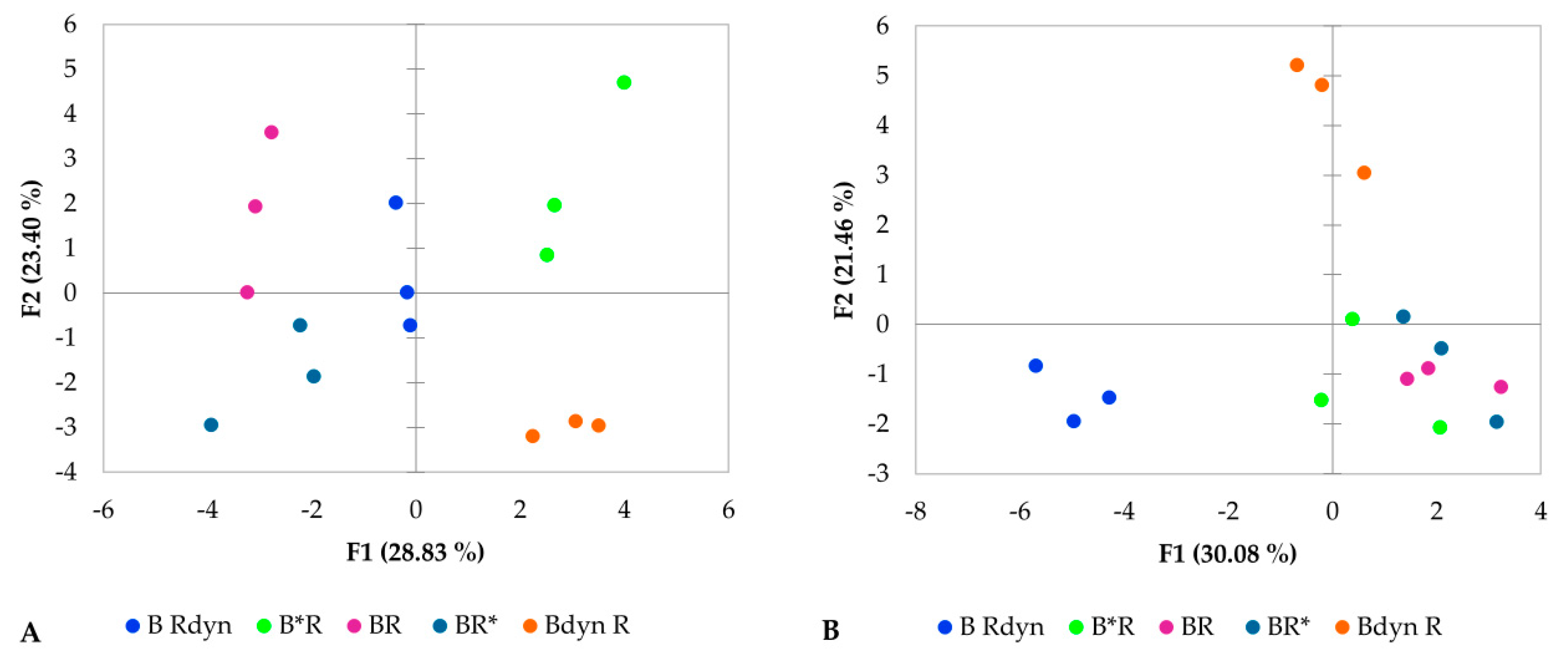
2.7. Statistical Analysis
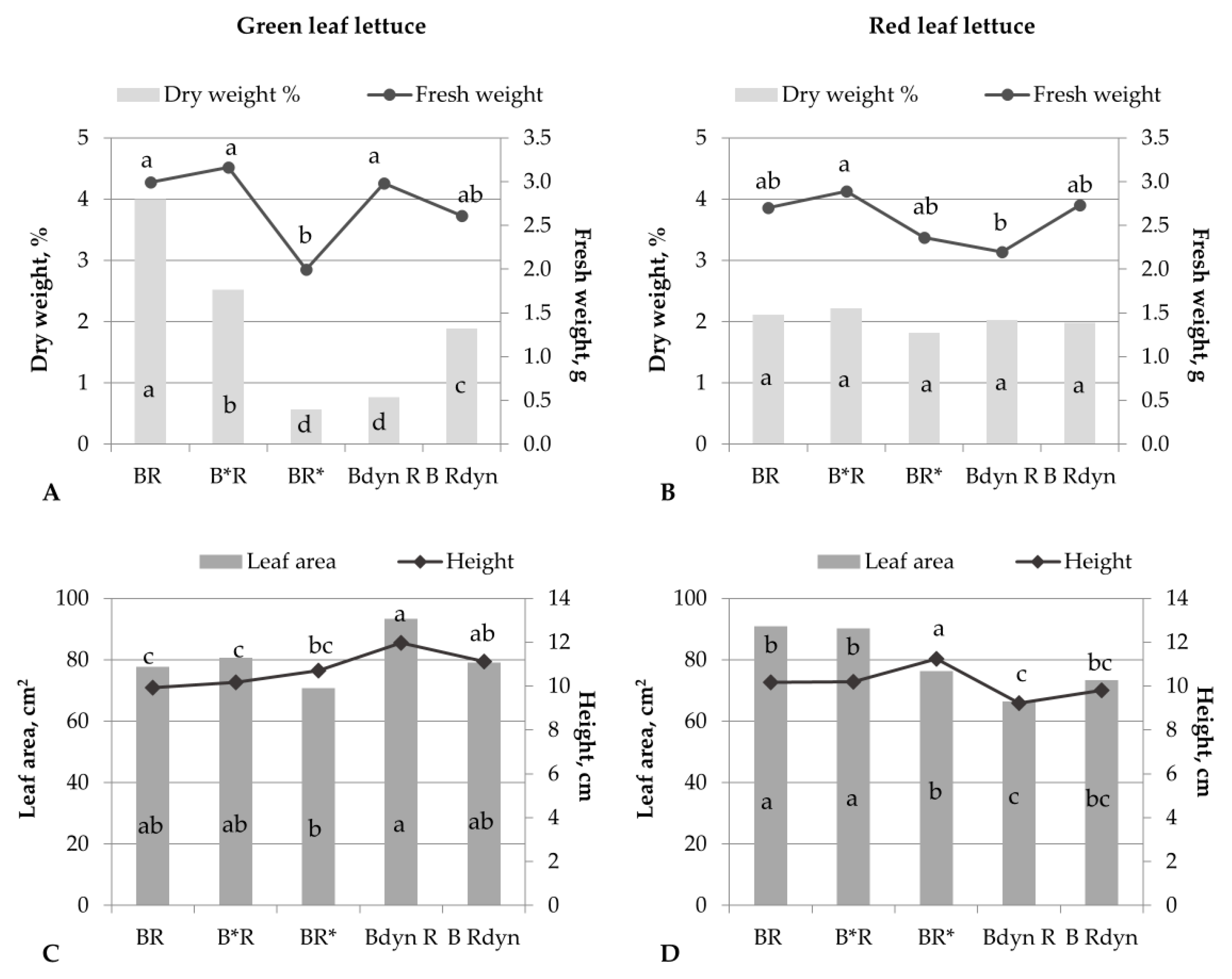
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Yamori, W. Photosynthetic response to fluctuating environments and photoprotective strategies under abiotic stress. J. Plant. Res. 2016, 129, 379–395. [Google Scholar] [CrossRef]
- Graham, P.J.; Nguyen, B.; Burdyny, T.; Sinton, D. A penalty on photosynthetic growth in fluctuating light. Sci. Rep. 2017, 7, 12513. [Google Scholar] [CrossRef]
- Yamori, W.; Makino, A.; Shikanai, T. A physiological role of cyclic electron transport around photosystem I in sustaining photosynthesis under fluctuating light in rice. Sci. Rep. 2016, 6, 20147. [Google Scholar] [CrossRef]
- Slattery, R.A.; Walker, B.J.; Weber, A.P.M.; Ort, D.R. The Impacts of Fluctuating Light on Crop Performance. Plant Physiol. 2018, 176, 990–1003. [Google Scholar] [CrossRef]
- Vialet-Chabrand, S.; Matthews, J.S.A.; Simkin, A.J.; Raines, C.A.; Lawson, T. Importance of Fluctuations in Light on Plant Photosynthetic Acclimation. Plant Physiol. 2017, 173, 2163–2179. [Google Scholar] [CrossRef] [PubMed]
- Kono, M.; Terashima, I. Long-term and short-term responses of the photosynthetic electron transport to fluctuating light. J. Photochem. Photobiol. B Biol. 2014, 137, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Annunziata, M.G.; Apelt, F.; Carillo, P.; Krause, U.; Feil, R.; Mengin, V.; Lauxmann, M.A.; Köhl, K.; Nikoloski, Z.; Stitt, M.; et al. Getting back to nature: A reality check for experiments in controlled environments. J. Exp. Bot. 2017, 68, 4463–4477. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Wang, L.; Li, T.; Yang, Q.; Guo, W. Sugar accumulation and growth of lettuce exposed to different lighting modes of red and blue LED light. Sci. Rep. 2019, 9, 6926. [Google Scholar] [CrossRef]
- Hosseini, A.; Zare Mehrjerdi, M.; Aliniaeifard, S.; Seif, M. Photosynthetic and growth responses of green and purple basil plants under different spectral compositions. Physiol. Mol. Biol. Plants 2019, 25, 741–752. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, C.A.; Dzakovich, M.P.; Gomez, C.; Lopez, R.; Burr, J.F.; Hernández, R.; Kubota, C.; Currey, C.J.; Meng, Q.; Runkle, E.S.; et al. Light-Emitting Diodes in Horticulture. In Horticultural Reviews: Volume 43; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2015; pp. 1–88. ISBN 9781119107781. [Google Scholar]
- Viršilė, A.; Olle, M.; Duchovskis, P. LED Lighting in Horticulture. In Light Emitting Diodes for Agriculture: Smart Lighting; Dutta Gupta, S., Ed.; Springer: Singapore, 2017; pp. 113–147. ISBN 9789811058073. [Google Scholar]
- Lin, K.-H.; Huang, M.-Y.; Huang, W.-D.; Hsu, M.-H.; Yang, Z.-W.; Yang, C.-M. The effects of red, blue, and white light-emitting diodes on the growth, development, and edible quality of hydroponically grown lettuce (Lactuca sativa L. var. capitata). Sci. Hortic. 2013, 150, 86–91. [Google Scholar] [CrossRef]
- Naznin, M.; Lefsrud, M.; Gravel, V.; Azad, M. Blue Light added with Red LEDs Enhance Growth Characteristics, Pigments Content, and Antioxidant Capacity in Lettuce, Spinach, Kale, Basil, and Sweet Pepper in a Controlled Environment. Plants 2019, 8, 93. [Google Scholar] [CrossRef] [PubMed]
- Pennisi, G.; Blasioli, S.; Cellini, A.; Maia, L.; Crepaldi, A.; Braschi, I.; Spinelli, F.; Nicola, S.; Fernandez, J.A.; Stanghellini, C.; et al. Unraveling the Role of Red:Blue LED Lights on Resource Use Efficiency and Nutritional Properties of Indoor Grown Sweet Basil. Front. Plant Sci. 2019, 10, 305. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; He, D.; Ji, F.; Zhang, S.; Zheng, J. Effects of Daily Light Integral and LED Spectrum on Growth and Nutritional Quality of Hydroponic Spinach. Agronomy 2020, 10, 1082. [Google Scholar] [CrossRef]
- Chiang, C.; Bånkestad, D.; Hoch, G. Reaching Natural Growth: Light Quality Effects on Plant Performance in Indoor Growth Facilities. Plants 2020, 9, 1273. [Google Scholar] [CrossRef] [PubMed]
- Pennisi, G.; Orsini, F.; Blasioli, S.; Cellini, A.; Crepaldi, A.; Braschi, I.; Spinelli, F.; Nicola, S.; Fernandez, J.A.; Stanghellini, C.; et al. Resource use efficiency of indoor lettuce (Lactuca sativa L.) cultivation as affected by red:blue ratio provided by LED lighting. Sci. Rep. 2019, 9, 14127. [Google Scholar] [CrossRef] [PubMed]
- Tamulaitis, G.; Duchovskis, P.; Bliznikas, Z.; Breive, K.; Ulinskaite, R.; Brazaityte, A.; Novičkovas, A.; Žukauskas, A. High-power light-emitting diode based facility for plant cultivation. J. Phys. D Appl. Phys. 2005, 38, 3182–3187. [Google Scholar] [CrossRef]
- Ma, C.; Sun, Z.; Chen, C.; Zhang, L.; Zhu, S. Simultaneous separation and determination of fructose, sorbitol, glucose and sucrose in fruits by HPLC–ELSD. Food Chem. 2014, 145, 784–788. [Google Scholar] [CrossRef]
- Brons, C.; Olieman, C. Study of the high-performance liquid chromatographic separation of reducing sugars, applied to the determination of lactose in milk. J. Chromatogr. A 1983, 259, 79–86. [Google Scholar] [CrossRef]
- Kraujalytė, V.; Venskutonis, P.R.; Pukalskas, A.; Česonienė, L.; Daubaras, R. Antioxidant properties and polyphenolic compositions of fruits from different European cranberrybush (Viburnum opulus L.) genotypes. Food Chem. 2013, 141, 3695–3702. [Google Scholar] [CrossRef]
- Kaiser, E.; Morales, A.; Harbinson, J. Fluctuating Light Takes Crop Photosynthesis on a Rollercoaster Ride. Plant. Physiol. 2018, 176, 977–989. [Google Scholar] [CrossRef]
- Chen, X.; Yang, Q.; Song, W.; Wang, L.; Guo, W.; Xue, X. Growth and nutritional properties of lettuce affected by different alternating intervals of red and blue LED irradiation. Sci. Hortic. 2017, 223, 44–52. [Google Scholar] [CrossRef]
- Chen, X.; Yang, Q. Effects of intermittent light exposure with red and blue light emitting diodes on growth and carbohydrate accumulation of lettuce. Sci. Hortic. 2018, 234, 220–226. [Google Scholar] [CrossRef]
- Ohtake, N.; Ishikura, M.; Suzuki, H.; Yamori, W.; Goto, E. Continuous Irradiation with Alternating Red and Blue Light Enhances Plant Growth While Keeping Nutritional Quality in Lettuce. Horts 2018, 53, 1804–1809. [Google Scholar] [CrossRef]
- Colonna, E.; Rouphael, Y.; Barbieri, G.; De Pascale, S. Nutritional quality of ten leafy vegetables harvested at two light intensities. Food Chem. 2016, 199, 702–710. [Google Scholar] [CrossRef]
- Samuolienė, G.; Sirtautas, R.; Brazaitytė, A.; Duchovskis, P. LED lighting and seasonality effects antioxidant properties of baby leaf lettuce. Food Chem. 2012, 134, 1494–1499. [Google Scholar] [CrossRef] [PubMed]
- Son, K.-H.; Oh, M.-M. Growth, photosynthetic and antioxidant parameters of two lettuce cultivars as affected by red, green, and blue light-emitting diodes. Hortic. Environ. Biotechnol. 2015, 56, 639–653. [Google Scholar] [CrossRef]
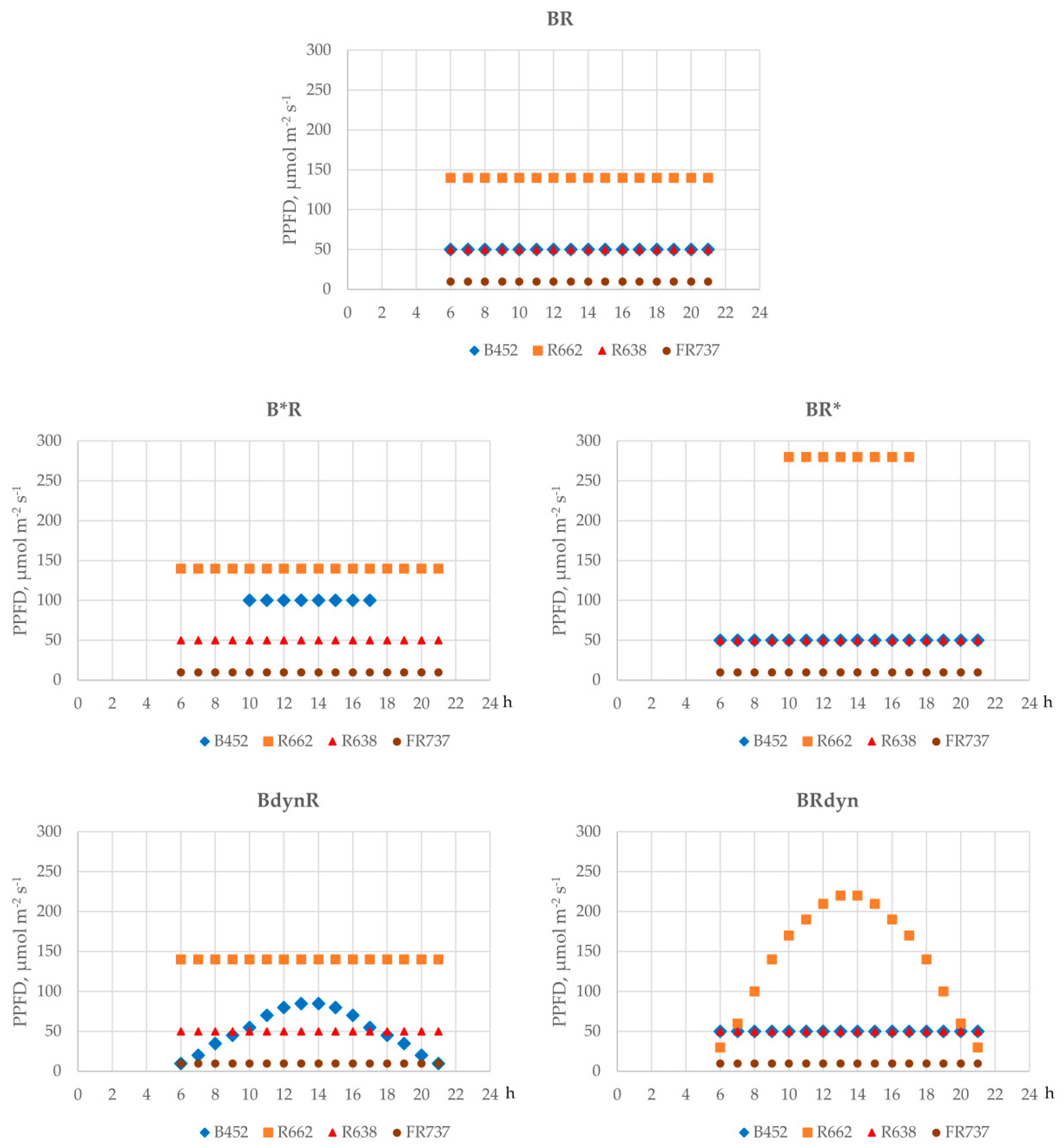
| % from Total DLI | DLI, mol/m2 day | |
|---|---|---|
| Total | 100% | 14.4 |
| B452 * | 20% | 2.88 |
| R638 | 20% | 2.88 |
| R662 * | 56% | 8.06 |
| FR737 | 4% | 0.58 |
| Treatment | Fv/m | Chlorophyll Index | Flavanol Index | NBI Index |
|---|---|---|---|---|
| Green leaf lettuce | ||||
| BR | 0.815a | 15.67a | 0.754b | 19.81a |
| B*R | 0.815a | 16.24a | 0.906a | 15.68b |
| BR* | 0.815a | 15.68a | 0.662b | 22.76a |
| Bdyn R | 0.815a | 15.68a | 0.734b | 20.97a |
| B Rdyn | 0.800a | 15.08a | 0.712b | 19.84a |
| Red leaf lettuce | ||||
| BR | 0.817ab | 23.99a | 0.404abc | 63.97ab |
| B*R | 0.820ab | 23.39a | 0.331bc | 78.09a |
| BR* | 0.829a | 23.27a | 0.296c | 77.65a |
| Bdyn R | 0.826a | 22.80a | 0.525a | 46.06b |
| B Rdyn | 0.812b | 25.36a | 0.469ab | 64.48ab |
| Treatment | DPPH µmol g−1 FW | ABTS µmol g−1 FW | FRAP | |
|---|---|---|---|---|
| Fe, µmol g−1 FW | TEAC, µmol Trolox g−1 FW | |||
| Green leaf lettuce | ||||
| BR | 13.30a | 50.35ab | 257.13ab | 0.062ab |
| B*R | 16.11a | 43.61ab | 329.02a | 0.080a |
| BR* | 11.06a | 42.92b | 195.19b | 0.041b |
| Bdyn R | 10.90a | 62.6a | 141.72b | 0.034b |
| B Rdyn | 13.98a | 53.57ab | 224.07ab | 0.053ab |
| Red leaf lettuce | ||||
| BR | 21.22a | 98.82a | 847.78a | 0.21a |
| B*R | 21.18a | 100.79a | 831.24a | 0.21a |
| BR* | 21.94a | 113.15a | 682.81a | 0.17a |
| Bdyn R | 21.51a | 109.33a | 674.96a | 0.17a |
| B Rdyn | 21.92a | 126.44a | 506.41a | 0.13a |
| Treatment | Macroelements, mg g−1 DW | Microelements mg g−1 DW | |||||||
|---|---|---|---|---|---|---|---|---|---|
| P | K | S | Ca | Mg | Fe | Zn | Mn | Cu | |
| Green leaf lettuce | |||||||||
| BR | 6.35c | 24.89d | 5.09d | 16.67d | 5.19d | 0.071e | 0.055b | 0.086c | 0.0066e |
| B*R | 6.81a | 25.36c | 5.39b | 18.48a | 5.52a | 0.084b | 0.062d | 0.089b | 0.0078b |
| BR* | 6.48b | 26.19a | 5.08d | 16.94c | 5.37c | 0.078d | 0.053e | 0.087c | 0.0067d |
| Bdyn R | 6.88a | 25.38c | 5.27c | 18.08b | 5.45ab | 0.089a | 0.066a | 0.092a | 0.0081a |
| B Rdyn | 6.85a | 25.91b | 6.19a | 16.71d | 5.42bc | 0.080c | 0.057c | 0.086c | 0.0073c |
| Red leaf lettuce | |||||||||
| BR | 7.38ab | 26.86b | 5.36b | 16.14a | 5.64a | 0.077d | 0.064b | 0.094a | 0.0089b |
| B*R | 7.22b | 27.02b | 5.17c | 15.23c | 5.59a | 0.084b | 0.060c | 0.089c | 0.0080c |
| BR* | 7.48a | 28.49a | 5.47a | 15.47b | 5.64a | 0.082c | 0.064b | 0.095a | 0.0080c |
| Bdyn R | 7.40a | 25.98c | 4.96d | 14.00d | 5.62a | 0.092a | 0.070a | 0.092b | 0.0094a |
| B Rdyn | 6.31c | 24.51d | 4.60e | 15.19c | 4.97b | 0.073e | 0.059d | 0.087d | 0.0076d |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Viršilė, A.; Miliauskienė, J.; Haimi, P.J.; Laužikė, K.; Samuolienė, G. The Comparison of Constant and Dynamic Red and Blue Light Irradiation Effects on Red and Green Leaf Lettuce. Agronomy 2020, 10, 1802. https://doi.org/10.3390/agronomy10111802
Viršilė A, Miliauskienė J, Haimi PJ, Laužikė K, Samuolienė G. The Comparison of Constant and Dynamic Red and Blue Light Irradiation Effects on Red and Green Leaf Lettuce. Agronomy. 2020; 10(11):1802. https://doi.org/10.3390/agronomy10111802
Chicago/Turabian StyleViršilė, Akvilė, Jurga Miliauskienė, Perttu Juhani Haimi, Kristina Laužikė, and Giedrė Samuolienė. 2020. "The Comparison of Constant and Dynamic Red and Blue Light Irradiation Effects on Red and Green Leaf Lettuce" Agronomy 10, no. 11: 1802. https://doi.org/10.3390/agronomy10111802
APA StyleViršilė, A., Miliauskienė, J., Haimi, P. J., Laužikė, K., & Samuolienė, G. (2020). The Comparison of Constant and Dynamic Red and Blue Light Irradiation Effects on Red and Green Leaf Lettuce. Agronomy, 10(11), 1802. https://doi.org/10.3390/agronomy10111802






