Effect of Jasmonic Acid and Salicylic Acid on Growth and Biochemical Composition of In-Vitro-Propagated Lavandula angustifolia Mill
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Culture Conditions
2.2. Total Phenolic Content
2.3. Chlorophyll Content
Chl b (mg/g) = (18.61 × A645 − 3.96 × A663) × V/g
2.4. Carotenoid Content
2.5. Statistical Analysis
3. Results
3.1. Effect of Elicitors on the Morphological Characteristics of In Vitro Explants
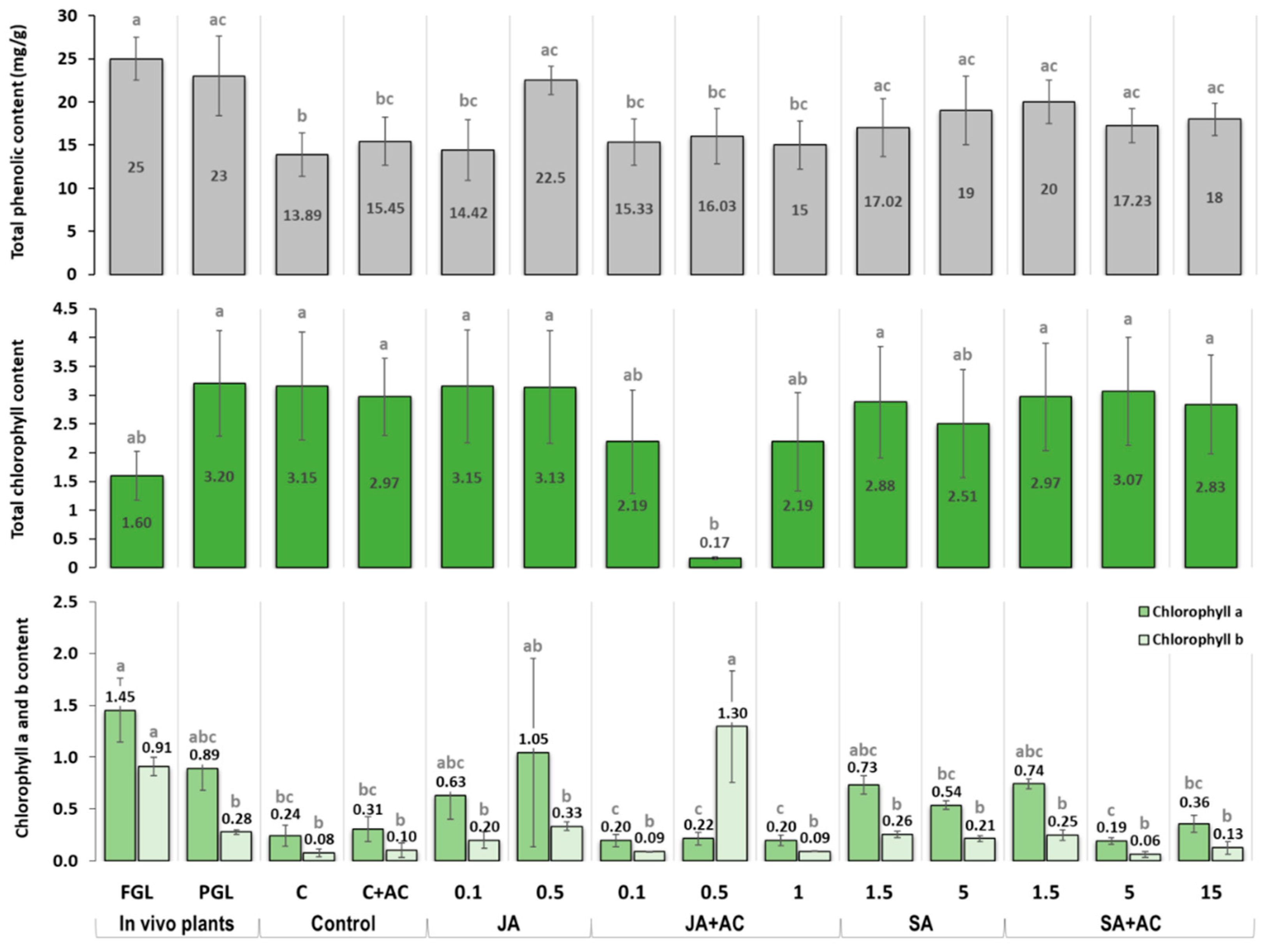
3.2. Effect of Elicitors on the Total Phenolic and Chlorophyll Contents of In Vitro Explants
3.3. Effect of Elicitors on the Carotenoid Profile of In Vitro Plantlets
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Demasi, S.; Caser, M.; Lonati, M.; Cioni, P.L.; Pistelli, L.; Najar, B.; Scariot, V. Latitude and altitude influence secondary metabolite production in peripheral alpine populations of the mediterranean species Lavandula angustifolia Mill. Front. Plant Sci. 2018, 9, 983. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, S.; Romano, A. In vitro culture of lavenders (Lavandula spp.) and the production of secondary metabolites. Biotechnol. Adv. 2013, 31, 166–174. [Google Scholar] [CrossRef] [PubMed]
- Andrys, D.; Kulpa, D. In vitro propagation affects the composition of narrow-leaved lavender essential oils. Acta Chromatogr. 2017, 30. [Google Scholar] [CrossRef]
- Sharopov, F.S.; Zhang, H.; Wink, M.; Setzer, W.N. Aromatic medicinal plants from Tajikistan (Central Asia). Medicines 2015, 2, 28–46. [Google Scholar] [CrossRef] [PubMed]
- De Rapper, S.; Viljoen, A.; Van Vuuren, S. The in vitro antimicrobial effects of Lavandula angustifolia essential oil in combination with conventional antimicrobial agents. Evid. Based Complement. Altern. Med. 2016, 2016, 2752739. [Google Scholar] [CrossRef]
- Shahdadi, H.; Bahador, R.; Eteghadi, A.; Boraiinejad, S. Levander a plant for medical uses: A literature review. Indian J. Public Health Res. Dev. 2017, 8, 328. [Google Scholar] [CrossRef]
- Białoń, M.; Krzyśko-Łupicka, T.; Nowakowska-Bogdan, E.; Wieczorek, P.P. Chemical composition of two different Lavender essential oils and their effect on facial skin microbiota. Molecules 2019, 24, 3270. [Google Scholar] [CrossRef]
- Mahmoudi, R.; Aghaei, S.; Salehpour, Z.; Mousavizadeh, A.; Khoramrooz, S.S.; Taheripour Sisakht, M.; Christiansen, G.; Baneshi, M.; Karimi, B.; Bardania, H. Antibacterial and antioxidant properties of phyto-synthesized silver nanoparticles using Lavandula stoechas extract. Appl. Organomet. Chem. 2020, 34, e5394. [Google Scholar] [CrossRef]
- Cory, H.; Passarelli, S.; Szeto, J.; Tamez, M.; Mattei, J. The role of polyphenols in human health and food systems: A mini-review. Front. Nutr. 2018, 5, 87. [Google Scholar] [CrossRef]
- Maiani, G.; Periago Castón, M.J.; Catasta, G.; Toti, E.; Cambrodón, I.G.; Bysted, A.; Granado-Lorencio, F.; Olmedilla-Alonso, B.; Knuthsen, P.; Valoti, M.; et al. Carotenoids: Actual knowledge on food sources, intakes, stability and bioavailability and their protective role in humans. Mol. Nutr. Food Res. 2009, 53, S194–S218. [Google Scholar] [CrossRef]
- Toti, E.; Chen, C.Y.O.; Palmery, M.; Villaño Valencia, D.; Peluso, I. Non-provitamin A and provitamin A carotenoids as immunomodulators: Recommended dietary allowance, therapeutic index, or personalized nutrition? Oxidative Med. Cell. Longev. 2018, 2018, 4637861. [Google Scholar] [CrossRef]
- Yue, W.; Ming, Q.L.; Lin, B.; Rahman, K.; Zheng, C.J.; Han, T.; Qin, L.P. Medicinal plant cell suspension cultures: Pharmaceutical applications and high-yielding strategies for the desired secondary metabolites. Crit. Rev. Biotechnol. 2016, 36, 215–232. [Google Scholar] [CrossRef] [PubMed]
- Debnath, M.; Malik, C.P.; Bisen, P.S. Micropropagation: A tool for the production of high quality plant-based medicines. Curr. Pharm. Biotechnol. 2006, 7, 33–49. [Google Scholar] [CrossRef]
- Chandran, H.; Meena, M.; Barupal, T.; Sharma, K. Plant tissue culture as a perpetual source for production of industrially important bioactive compounds. Biotechnol. Rep. 2020, 26, e00450. [Google Scholar] [CrossRef]
- Halder, M.; Sarkar, S.; Jha, S. Elicitation: A biotechnological tool for enhanced production of secondary metabolites in hairy root cultures. Eng. Life Sci. 2019, 19, 880–895. [Google Scholar] [CrossRef]
- Naik, P.; Al-Khayri, J. Abiotic and biotic elicitors–role in secondary metabolites production through In vitro culture of medicinal plants. In Abiotic and Biotic Stress in Plants-Recent Advances and Future Perspectives; Shanker, A.K., Shanker, C., Eds.; INTECH: London, UK, 2016; pp. 247–277. [Google Scholar]
- Giri, C.C.; Zaheer, M. Chemical elicitors versus secondary metabolite production in vitro using plant cell, tissue and organ cultures: Recent trends and a sky eye view appraisal. Plant Cell Tissue Organ Cult. 2016, 126, 1–18. [Google Scholar] [CrossRef]
- Cirak, C.; Radušienė, J.; Kurtarc, E.S.; Marksa, M.; Ivanauskas, L. In vitro plant regeneration and jasmonic acid induced bioactive chemical accumulations in two Hypericum species from Turkey. S. Afr. J. Bot. 2020, 128, 312–318. [Google Scholar] [CrossRef]
- Sirhindi, G.; Mushtaq, R.; Gill, S.S.; Sharma, P.; Abd_Allah, E.F.; Ahmad, P. Jasmonic acid and methyl jasmonate modulate growth, photosynthetic activity and expression of photosystem II subunit genes in Brassica oleracea L. Sci. Rep. 2020, 10, 9322. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Dwivedi, P. Methyl-jasmonate and salicylic acid as potent elicitors for secondary metabolite production in medicinal plants: A review. J. Pharmacogn. Phytochem. 2017, 7, 750–757. [Google Scholar]
- Zhai, X.; Jia, M.; Chen, L.; Zheng, C.J.; Rahman, K.; Han, T.; Qin, L.P. The regulatory mechanism of fungal elicitor-induced secondary metabolite biosynthesis in medical plants. Crit. Rev. Microbiol. 2017, 43, 238–261. [Google Scholar] [CrossRef]
- Ho, T.T.; Murthy, H.N.; Park, S.Y. Methyl jasmonate induced oxidative stress and accumulation of secondary metabolites in plant cell and organ cultures. Int. J. Mol. Sci. 2020, 21, 716. [Google Scholar] [CrossRef]
- Wang, J.; Song, L.; Gong, X.; Xu, J.; Li, M. Functions of jasmonic acid in plant regulation and response to abiotic stress. Int. J. Mol. Sci. 2020, 21, 1446. [Google Scholar] [CrossRef]
- Wasternack, C. Action of jasmonates in plant stress responses and development-applied aspects. Biotechnol. Adv. 2014, 32, 31–39. [Google Scholar] [CrossRef]
- Qi, X.; Torii, K.U. Hormonal and environmental signals guiding stomatal development. BMC Biol. 2018, 16, 21. [Google Scholar] [CrossRef]
- Yang, J.; Duan, G.; Li, C.; Liu, L.; Han, G.; Zhang, Y.; Wang, C. The crosstalks between jasmonic acid and other plant hormone signaling highlight the involvement of jasmonic acid as a core component in plant response to biotic and abiotic stresses. Front. Plant Sci. 2019, 10, 1349. [Google Scholar] [CrossRef]
- Zhao, M.L.; Wang, J.N.; Shan, W.; Fan, J.G.; Kuang, J.F.; Wu, K.Q.; Li, X.P.; Chen, W.X.; He, F.Y.; Chen, J.Y.; et al. Induction of jasmonate signalling regulators MaMYC2s and their physical interactions with MaICE1 in methyl jasmonate-induced chilling tolerance in banana fruit. Plant Cell Environ. 2013, 36, 30–51. [Google Scholar] [CrossRef]
- Hu, Y.; Jiang, Y.; Han, X.; Wang, H.; Pan, J.; Yu, D. Jasmonate regulates leaf senescence and tolerance to cold stress: Crosstalk with other phytohormones. J. Exp. Bot. 2017, 68, 1361–1369. [Google Scholar] [CrossRef] [PubMed]
- Ullah, A.; Akbar, A.; Yang, X. Chapter 7-Jasmonic acid (JA)-mediated signaling in leaf senescence. In Senescence Signalling and Control in Plants; Sarwat, M., Tuteja, N., Eds.; Academic Press: Cambridge, MA, USA, 2019; pp. 111–123. [Google Scholar]
- Huang, H.; Liu, B.; Liu, L.; Song, S. Jasmonate action in plant growth and development. J. Exp. Bot. 2017, 68, 1349–1359. [Google Scholar] [CrossRef] [PubMed]
- Janda, T.; Szalai, G.; Pál, M. Salicylic acid signalling in plants. Int. J. Mol. Sci. 2020, 21, 2655. [Google Scholar] [CrossRef] [PubMed]
- Lefevere, H.; Bauters, L.; Gheysen, G. Salicylic acid biosynthesis in plants. Front. Plant Sci. 2020, 11, 338. [Google Scholar] [CrossRef]
- Wani, A.B.; Chadar, H.; Wani, A.H.; Singh, S.; Upadhyay, N. Salicylic acid to decrease plant stress. Environ. Chem. Lett. 2017, 15, 101–123. [Google Scholar] [CrossRef]
- Mohammadi, H.; Nikjoyan, M.; Hazrati, S.; Hashempour, H. Improvement of yield and phytochemical compounds of Thymus vulgaris through foliar application of salicylic acid under water stress. Agric. For. 2020, 66, 129–142. [Google Scholar] [CrossRef]
- Pathirana, R.; McLachlan, A.; Hedderley, D.; Panis, B.; Carimi, F. Pre-treatment with salicylic acid improves plant regeneration after cryopreservation of grapevine (Vitis spp.) by droplet vitrification. Acta Physiol. Plant 2015, 38, 12. [Google Scholar] [CrossRef]
- Appu, M.; Muthukrishnan, S. Foliar application of salicylic acid stimulates flowering and induce defense related proteins in finger millet plants. Univers. J. Plant Sci. 2014, 2, 14–18. [Google Scholar] [CrossRef]
- Kazemi, M. Foliar application of salicylic acid and calcium on yield, yield component and chemical properties of strawberry. BEPLS 2013, 2, 19–23. [Google Scholar]
- Shaaban, M.M.; Abd El-Aal, A.M.K.; Ahmed, F.F. Insight into the effect of salicylic acid on apple trees growing under sandy saline soil. RJABS 2011, 7, 150–156. [Google Scholar]
- Ngullie, C.R.; Tank, R.; Bhanderi, D. Effect of salicylic acid and humic acid on flowering, fruiting, yield and quality of mango (Mangifera indica L.) cv. KESAR. Adv. Res. J. Crop Improv. 2014, 5, 136–139. [Google Scholar] [CrossRef]
- Xu, A.; Zhan, J.-C.; Huang, W.-D. Effects of ultraviolet C, methyl jasmonate and salicylic acid, alone or in combination, on stilbene biosynthesis in cell suspension cultures of Vitis vinifera L. cv. Cabernet Sauvignon. Plant Cell Tissue Organ Cult. 2015, 122, 197–211. [Google Scholar] [CrossRef]
- Ahmed, H.S.; Moawad, A.S.; AbouZid, S.F.; Owis, A.I. Salicylic acid increases flavonolignans accumulation in the fruits of hydroponically cultured Silybum marianum. Saudi Pharm. J. 2020, 28, 593–598. [Google Scholar] [CrossRef]
- Golkar, P.; Taghizadeh, M.; Yousefian, Z. The effects of chitosan and salicylic acid on elicitation of secondary metabolites and antioxidant activity of safflower under in vitro salinity stress. Plant Cell Tissue Organ Cult. 2019, 137, 575–585. [Google Scholar] [CrossRef]
- Gadzovska, S.; Maury, S.; Delaunay, A.; Spasenoski, M.; Hagège, D.; Courtois, D.; Joseph, C. The influence of salicylic acid elicitation of shoots, callus, and cell suspension cultures on production of naphtodianthrones and phenylpropanoids in Hypericum perforatum L. Plant Cell Tissue Organ Cult. 2013, 113, 25–39. [Google Scholar] [CrossRef]
- Andrys, D.; Adaszyńska-Skwirzyńska, M.; Kulpa, D. Jasmonic acid changes the composition of essential oil isolated from narrow-leaved lavender propagated in in vitro cultures. Nat. Prod. Res. 2018, 32, 834–839. [Google Scholar] [CrossRef]
- Andrys, D.; Kulpa, D.; Grzeszczuk, M.; Białecka, B. Influence of jasmonic acid on the growth and antimicrobial and antioxidant activities of Lavandula angustifolia Mill. propagated in vitro. Folia Hortic. 2018, 30, 3. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Clapa, D.; Bunea, C.I.; Borsai, O.; Pintea, A.; Harta, M.; Stefan, R.; Fira, A. The role of Sequestrene 138 in highbush blueberry (Vaccinium corymbosum L.) micropropagation. Hortscience 2018, 53, 1487–1493. [Google Scholar] [CrossRef]
- Bunea, C.-I.; Pop, N.; Babeş, A.C.; Matea, C.; Dulf, F.V.; Bunea, A. Carotenoids, total polyphenols and antioxidant activity of grapes (Vitis vinifera) cultivated in organic and conventional systems. Chem. Cent. J. 2012, 6, 66. [Google Scholar] [CrossRef]
- Koo, Y.M.; Heo, A.Y.; Choi, H.W. Salicylic acid as a safe plant protector and growth regulator. Plant Pathol. 2020, 36, 1–10. [Google Scholar] [CrossRef]
- Pérez-Tortosa, V.; López-Orenes, A.; Martínez-Pérez, A.; Ferrer, M.A.; Calderón, A.A. Antioxidant activity and rosmarinic acid changes in salicylic acid-treated Thymus membranaceus shoots. Food Chem. 2012, 130, 362–369. [Google Scholar] [CrossRef]
- Kovácik, J.; Klejdus, B.; Hedbavny, J.; Backor, M. Salicylic acid alleviates NaCl-induced changes in the metabolism of Matricaria chamomilla plants. Ecotoxicology 2009, 18, 544–554. [Google Scholar] [CrossRef] [PubMed]
- Jini, D.; Joseph, B. Physiological mechanism of salicylic acid for alleviation of salt stress in rice. Rice Sci. 2017, 24, 97–108. [Google Scholar] [CrossRef]
- El-Katony, T.M.; El-Bastawisy, Z.M.; El-Ghareeb, S.S. Timing of salicylic acid application affects the response of maize (Zea mays L.) hybrids to salinity stress. Heliyon 2019, 5, e01547. [Google Scholar] [CrossRef]
- Sakhanokho, H.; Kelley, R. Influence of salicylic acid on in vitro propagation and salt tolerance in Hibiscus acetosella and Hibiscus moscheutos (cv ‘Luna Red’). Afr. J. Biotechnol. 2009, 8, 1474–1481. [Google Scholar]
- Koul, A.; Mallubhotla, S. Elicitation and enhancement of bacoside production using suspension cultures of Bacopa monnieri (L.) Wettst. 3 Biotech 2020, 10, 256. [Google Scholar] [CrossRef]
- Krzyzanowska, J.; Czubacka, A.; Pecio, Ł.; Przybys, M.; Doroszewska, T.; Stochmal, A.; Oleszek, W. The effects of jasmonic acid and methyl jasmonate on rosmarinic acid production in Mentha x piperita cell suspension cultures. Plant Cell Tissue Organ Cult. 2011, 108, 73–81. [Google Scholar] [CrossRef]
- Ruzic, D.J.; Vujovic, T.; Cerovic, R.; Djordjevic, M. Potential application of jasmonic acid in in vitro rooting of low vigorous pear and cherry rootstocks. Acta Hortic. 2015, 1099, 895–900. [Google Scholar] [CrossRef]
- Kumlay, A. The effect of jasmonic acid on the micropropagation of potato (Solanum tuberosum L.) under long days conditions. Yüzüncü Yıl Üniversitesi Tarım Bilimleri Dergisi 2016, 26, 79–88. [Google Scholar] [CrossRef]
- Złotek, U.; Michalak-Majewska, M.; Szymanowska, U. Effect of jasmonic acid elicitation on the yield, chemical composition, and antioxidant and anti-inflammatory properties of essential oil of lettuce leaf basil (Ocimum basilicum L.). Food Chem. 2016, 213, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Li, N.-Y.; Tang, H.-R.; Ge, C.; Mo, F.; Xiao, Y.-H.; Luo, Y. Tissue culture of Lavandula angustifolia L. AIP Conf. Proc. 2019, 2079. [Google Scholar] [CrossRef]
- Pistelli, L.; Noccioli, C.; D’Angiolillo, F.; Pistelli, L. Composition of volatile in micropropagated and field grown aromatic plants from Tuscany Islands. Acta Biochim. Pol. 2013, 60, 43–50. [Google Scholar] [CrossRef]
- Hamza, A.M.; Abd El-Kafie, O.M.; Kasem, M.M. Direct micropropagation of english lavender (Lavandula angustifolia Munstead) plant. J. Plant Prod. 2011, 2, 81–96. [Google Scholar] [CrossRef]
- Mitrofanova, I.; Chirkov, S.; Lesnikova-Sedoshenko, N.; Chelombit, S.; Zakubanskiy, A.; Rabotyagov, V.; Mitrofanova, O. Micropropagation of Lavandula angustifolia Mill. ‘Record’ and ‘Belyanka’. Acta Hortic. 2017, 37–42. [Google Scholar] [CrossRef]
- De Bona, C.M.; Reinhart, V.; Biasi, L.A.; Zanette, F. Lavandula dentata and Lavandula angustifolia in vitro organogenesis. Plant Cell Cult. Micropropag. 2011, 7, 66–70. [Google Scholar]
- Falk, L.; Biswas, K.; Boeckelmann, A.; Lane, A.; Mahmoud, S.S. An effcient method for the micropropagation of Lavenders: Regeneration of a unique mutant. J. Essent Oil Res. 2009, 21, 225–228. [Google Scholar] [CrossRef]
- Ghiorghita, G.; Maftei, D.E.; Nicuta, D. Some aspects concerning the in vitro reaction of Lavandula angustifolia L. Propag. Ornam. Plants 2009, 9, 47–49. [Google Scholar]
- Wang, X.; Jin, L.; Li, M.; Zhao, M.; Zhao, H.; Xu, Y. Bioreactor culture and plant regeneration from cell clusters of the aromatic plant, Lavandula angustifolia ‘Munstead’. J. Hortic. Sci. Biotechnol. 2007, 82, 781–785. [Google Scholar] [CrossRef]
- Miclea, I.; Chifor, R. Germination, in vitro propagation and acclimatization in Lavandula angustifolia. Bull. USAMV Anim. Sci. Biotechnol. 2018, 75, 105. [Google Scholar] [CrossRef]
- Vinh, D.T.; Hoa, M.T.; Khai, P.C.; Minh, T.V. Micropropagation of lavender (Lavandula angustifolia). JIPBS 2017, 4, 7–11. [Google Scholar]
- Jordan, A.; Calvo, M.; Segura, J. Micropropagation of adult Lavandula dentata plants. J. Hortic. Sci. Biotech. 1998, 73, 93–96. [Google Scholar] [CrossRef]
- Sudria, C.; Palazon, J.; Cusido, R.; Bonfill, M.; Pinol, M.T.; Morales, C. Effect of benzyladenine and indolebutyric acid on ultrastructure, glands formation, and essential oil accumulation in Lavandula dentata plantlets. Biol. Plant 2001, 44, 1–6. [Google Scholar] [CrossRef]
- Sudriá, C.; Piñol, M.T.; Palazón, J.; Cusidó, R.M.; Vila, R.; Morales, C.; Bonfill, M.; Cañigueral, S. Influence of plant growth regulators on the growth and essential oil content of cultured Lavandula dentata plantlets. Plant Cell Tissue Organ Cult. 1999, 58, 177–184. [Google Scholar] [CrossRef]
- Tsuro, M.; Inoue, M.; Kameoka, H. Variation in essential oil components in regenerated lavender (Lavandula vera DC) plants. Sci. Hortic. 2001, 88, 309–317. [Google Scholar] [CrossRef]
- Andrade, L.B.; Echeverrigaray, S.; Fracaro, F.; Pauletti, G.F.; Rota, L. The effect of growth regulators on shoot propagation and rooting of common lavender (Lavandula vera DC). Plant Cell Tissue Organ Cult. 1999, 56, 79–83. [Google Scholar] [CrossRef]
- Sánchez-Gras, M.C.; Del Carmen Calvo, M. Micropropagation of Lavandula latifolia through nodal bud culture of mature plants. Plant Cell Tissue Organ Cult. 1996, 45, 259–261. [Google Scholar] [CrossRef]
- Zuzarte, M.R.; Dinis, A.M.; Cavaleiro, C.; Salgueiro, L.R.; Canhoto, J.M. Trichomes, essential oils and in vitro propagation of Lavandula pedunculata (Lamiaceae). Ind. Crop Prod. 2010, 32, 580–587. [Google Scholar] [CrossRef]
- Zuzarte, M.; Dinis, A.; Salgueiro, L.; Canhoto, J. A rapid and efficient protocol for clonal propagation of phenolic-rich Lavandula multifida. J. Agric. Sci. 2015, 7. [Google Scholar] [CrossRef]
- Costa, P.; Gonçalves, S.; Valentão, P.; Andrade, P.B.; Romano, A. Accumulation of phenolic compounds in in vitro cultures and wild plants of Lavandula viridis L’Hér and their antioxidant and anti-cholinesterase potential. Food Chem. Toxicol. 2013, 57, 69–74. [Google Scholar] [CrossRef]
- Dias, M.C.; Almeida, R.; Romano, A. Rapid clonal multiplication of Lavandula viridis L’Hér through in vitro axillary shoot proliferation. Plant Cell Tissue Organ Cult. 2002, 68, 99–102. [Google Scholar] [CrossRef]
- Nitzsche, A.; Tokalov, S.V.; Gutzeit, H.O.; Ludwig-Müller, J. Chemical and biological characterization of cinnamic acid derivatives from cell cultures of Lavender (Lavandula officinalis) induced by stress and jasmonic acid. J. Agric. Food Chem. 2004, 52, 2915–2923. [Google Scholar] [CrossRef]
- Kirimer, N.; Mokhtarzadeh, S.; Demirci, B.; Goger, F.; Khawar, K.M.; Demirci, F. Phytochemical profiling of volatile components of Lavandula angustifolia Miller propagated under in vitro conditions. Ind. Crop Prod. 2017, 96, 120–125. [Google Scholar] [CrossRef]
- Gonçalves, S.; Serra, H.; Nogueira, J.M.F.; Almeida, R.; Custódio, L.; Romano, A. Headspace-SPME of in vitro shoot-cultures and micropropagated plants of Lavandula viridis. Biol. Plant 2008, 52, 133–136. [Google Scholar] [CrossRef]
- Wesołowska, A.; Jadczak, P.; Kulpa, D.; Przewodowski, W. Gas chromatography-mass spectrometry (GC-MS) analysis of essential oils from AgNPs and AuNPs elicited Lavandula angustifolia in vitro cultures. Molecules 2019, 24, 606. [Google Scholar] [CrossRef]
- Duda, S.C.; Mărghitaş, L.A.; Dezmirean, D.; Duda, M.; Mărgăoan, R.; Bobiş, O. Changes in major bioactive compounds with antioxidant activity of Agastache foeniculum, Lavandula angustifolia, Melissa officinalis and Nepeta cataria: Effect of harvest time and plant species. Ind. Crop Prod. 2015, 77, 499–507. [Google Scholar] [CrossRef]
- Ebert, A.; Taylor, F.; Blake, J. Changes of 6-benzylaminopurine and 2,4-dichlorophenoxyacetic acid concentrations in plant tissue culture media in the presence of activated charcoal. Plant Cell Tissue Organ Cult. 1993, 33, 157–162. [Google Scholar] [CrossRef]
- Avato, P.; Fortunato, I.M.; Ruta, C.; D’Elia, R. Glandular hairs and essential oils in micropropagated plants of Salvia officinalis L. Plant Sci. 2005, 169, 29–36. [Google Scholar] [CrossRef]
- Garibaldi, A.; Bertetti, D.; Pensa, P.; Poli, A.; Gullino, M.L. First report of web blight on lavender (Lavandula officinalis) caused by Rhizoctonia solani AG-1-IB in Italy. J. Plant Pathol. 2013, 95, 661. [Google Scholar]
- Giannoulis, K.D.; Evangelopoulos, V.; Gougoulias, N.; Wogiatzi, E. Could bio-stimulators affect flower, essential oil yield, and its composition in organic lavender (Lavandula angustifolia) cultivation? Ind. Crop Prod. 2020, 154, 112611. [Google Scholar] [CrossRef]

| Treatment | Number of Shoots/Explant | Overall Shoot Height (cm) | Main Shoot Height (cm) | Tissue Weight/Explant (mg) | Explant Response Rate (%) |
|---|---|---|---|---|---|
| C | 4.70 ± 0.95 ac | 1.35 ± 0.08 bce | 2.96 ± 0.34 b | 127.38 ± 4.40 cd | 100.00 ± 0.00 a |
| C + AC | 3.97 ± 0.72 acf | 1.62 ± 0.11 bc | 2.90 ± 0.26 b | 102.45 ± 5.58 cd | 100.00 ± 0.00 a |
| 0.1 JA | 1.69 ± 0.49 dgh | 1.18 ± 0.40 cde | 2.31 ± 0.18 b | 127.60 ± 10.12 cd | 71.51 ± 2.03 a |
| 0.5 JA | 2.73 ± 0.33 bcdg | 1.57 ± 0.31 bc | 3.01 ± 0.77 b | 154.15 ± 6.19 bc | 100.00 ± 0.00 a |
| 1 JA | 0.00 h | 0.00 f | 0.00 c | 0.00 d | 0.00 b |
| 0.1 JA + AC | 4.18 ± 1.33 adf | 1.30 ± 0.09 bce | 2.94 ± 0.83 b | 112.43 ± 3.78 cd | 100.00 ± 0.00 a |
| 0.5 JA + AC | 2.38 ± 0.85 dfgi | 1.49 ± 0.40 bce | 2.75 ± 0.27 b | 64.63 ± 1.66 cd | 80.00 ± 2.83 a |
| 1 JA + AC | 1.72 ± 0.78 gh | 1.46 ± 0.30 bce | 2.39 ± 0.35 b | 63.08 ± 4.16 cd | 70.00 ± 294 a |
| 1.5 SA | 5.76 ± 1.68 a | 1.43 ± 0.33 bce | 2.98 ± 0.71 b | 164.90 ± 8.26 ac | 95.83 ± 4.82 a |
| 5 SA | 3.33 ± 1.05 cdgi | 1.18 ± 0.31 cde | 2.96 ± 0.57 b | 85.28 ± 3.41 cd | 79.02 ± 2.44 a |
| 15 SA | 1.50 ± 0.68 bhi | 0.86 ± 0.05 e | 2.48 ± 0.12 b | 27.35 ± 1.15 bd | 30.00 ± 0.00 b |
| 1.5 SA + AC | 4.41 ± 0.74 acf | 1.95 ± 0.56 ab | 4.41 ± 0.89 a | 223.55 ± 5.39 a | 100.00 ± 0.00 a |
| 5 SA + AC | 3.88 ± 0.93 adg | 1.88 ± 0.05 abd | 3.41 ± 0.25 ab | 222.30 ± 6.36 a | 93.94 ± 8.57 a |
| 15 SA + AC | 3.29 ± 0.50 bfgc | 1.04 ± 0.23 ce | 2.54 ± 0.13 b | 93.00 ± 3.01 acd | 73.47 ± 8.62 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miclea, I.; Suhani, A.; Zahan, M.; Bunea, A. Effect of Jasmonic Acid and Salicylic Acid on Growth and Biochemical Composition of In-Vitro-Propagated Lavandula angustifolia Mill. Agronomy 2020, 10, 1722. https://doi.org/10.3390/agronomy10111722
Miclea I, Suhani A, Zahan M, Bunea A. Effect of Jasmonic Acid and Salicylic Acid on Growth and Biochemical Composition of In-Vitro-Propagated Lavandula angustifolia Mill. Agronomy. 2020; 10(11):1722. https://doi.org/10.3390/agronomy10111722
Chicago/Turabian StyleMiclea, Ileana, Andreea Suhani, Marius Zahan, and Andrea Bunea. 2020. "Effect of Jasmonic Acid and Salicylic Acid on Growth and Biochemical Composition of In-Vitro-Propagated Lavandula angustifolia Mill" Agronomy 10, no. 11: 1722. https://doi.org/10.3390/agronomy10111722
APA StyleMiclea, I., Suhani, A., Zahan, M., & Bunea, A. (2020). Effect of Jasmonic Acid and Salicylic Acid on Growth and Biochemical Composition of In-Vitro-Propagated Lavandula angustifolia Mill. Agronomy, 10(11), 1722. https://doi.org/10.3390/agronomy10111722







