Effectiveness of Humic Substances and Phenolic Compounds in Regulating Plant-Biological Functionality
Abstract
1. Introduction
2. Materials and Methods
2.1. Extraction and Characterization of Humic Substances and Soluble Soil Phenols
2.1.1. KOH Extract, (Humic Substances, HS), and Water Extract (Soluble Phenols, SWSP)
2.1.2. Fourier Transform Infrared Spectroscopy (FT-IR)
2.1.3. Surface-Enhanced Raman Scattering (SERS)
2.1.4. Fatty Acids, Organic Acids, and Carbohydrates
2.2. In Vitro Detection of Biological Activity of Humic Substances and Water Soluble Soil Phenols
2.2.1. Callus Growth, Antioxidant Compounds and Antioxidant Activity
2.2.2. Statistical Analysis
3. Results and discussion
3.1. Humic Substances and Soil Water Soluble Phenolic Fraction
3.2. Callus Growth and Metabolism
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- van Breemen, N.P.; Buurman, P. Soil Formation, 2nd ed.; Kluwer Academic Pub.: Boston, MA, USA, 1998. [Google Scholar]
- Stevenson, F.J. Humus Chemistry: Genesis, Composition, Reactions; John Wiley and Sons: New York, NY, USA, 1994. [Google Scholar]
- Garciá, A.C.; De Souza, L.G.A.; Pereira, M.G.; Castro, R.N.; Garciá-Mina, J.M.; Zonta, E.; Lisboa, F.J.G.; Berbara, R.L.L. Structure-Property-Function Relationship in Humic Substances to Explain the Biological Activity in Plants. Sci. Rep. 2016, 6, 20798. [Google Scholar] [CrossRef] [PubMed]
- Swift, R.S. Macromolecular properties of soil humic substances: Fact, fiction and opinion. Soil Sci. 1999, 164, 790–802. [Google Scholar] [CrossRef]
- Piccolo, A. The supramolecular structure of humic substances. Soil Sci. 2001, 166, 810–832. [Google Scholar] [CrossRef]
- Nardi, S.; Pizzeghello, D.; Muscolo, A.; Vianello, A. Physiological effects of humic substances in higher plants. Soil Biol. Biochem. 2002, 34, 1527–1537. [Google Scholar] [CrossRef]
- Muscolo, A.; Sidari, M.; Nardi, S. Humic substance: Relationship between structure and activity. Deeper information suggests univocal findings. J. Geochem. Explor. 2013, 129, 103–111. [Google Scholar] [CrossRef]
- Nardi, S.; Pizzeghello, D.; Schiavon, M.; Ertani, A. Plant biostimulants: Physiological responses induced by protein hydrolyzed-based products and humic substances in plant metabolism. Sci. Agric. 2016, 73, 18–23. [Google Scholar] [CrossRef]
- Nardi, S.; Ertani, A.; Francioso, O. Soil-root cross-talking: The role of humic substances. J. Plant Nutr. Soil Sci. 2017, 180, 5–13. [Google Scholar] [CrossRef]
- Mora, V.; Bacaicoa, E.; Baigorri, R.; Zamarreno, A.M.; Garcia-Mina, J.M. NO and IAA key regulators in the shoot growth promoting action of humic acid in Cucumis sativus L. J. Plant Growth Regul. 2014, 33, 430–439. [Google Scholar] [CrossRef]
- Canellas, L.P.; Olivares, F.L.; Natalia, O.A.; Canellas, L.P.; Mazzei, P.; Piccolo, A. Humic acids increase the maize seedlings exudation yield. Chem. Biol. Technol. Agric. 2019, 6, 3. [Google Scholar] [CrossRef]
- Pizzeghello, D.; Schiavon, M.; Francioso, O.; Dalla Vecchia, F.; Ertani, A.; Nardi, S. Bioactivity of size-fractionated and unfractionated humic substances from two forest soils and comparative effects on N and S metabolism, nutrition and root anatomy of Allium sativum L. Front. Plant Sci. 2020, 11, 1203. [Google Scholar] [CrossRef]
- Rose, M.T.; Patti, A.F.; Little, K.R.; Brown, A.L.; Jackson, W.R.; Cavagnaro, T.R. A meta analysis and review of plant-growth response in humic substances: Practical implications for agriculture. Adv. Agronom. 2014, 124, 37–98. [Google Scholar]
- Chen, Y.; Aviad, T. Effects of humic substances on plant growth. In Humic Substances in Soil and Crop Science: Selected Readings; Mac Carthy, P., Malcolm, R.L., Clapp, C.E., Bloom, P.R., Eds.; American Society of Agronomy and Soil Science Society of America: Madison, WI, USA, 1990; pp. 161–187. [Google Scholar]
- Nardi, S.; Concheri, G.; Dell’Agnola, G.; Scrimin, P. Nitrate uptake and ATPase activity in oat seedlings in the presence of two humic fractions. Soil Biol. Biochem. 1991, 23, 833–836. [Google Scholar] [CrossRef]
- Pinton, R.; Cesco, S.; Iacoletti, G.; Astolfi, S.; Varanini, Z. Modulation of NO3− uptake by water-extractable humic substances: Involvement of root plasma membrane H+ATPase. Plant Soil 1999, 215, 155–161. [Google Scholar] [CrossRef]
- Canellas, L.P.; Olivares, F.L.; Okorokova-Façanha, A.L.; Façanha, A.R. Humic acids isolated from earthworm compost enhance root elongation, lateral root emergence, and plasma membrane H+-ATPase activity in maize roots. Plant Physiol. 2002, 130, 1951–1957. [Google Scholar] [CrossRef] [PubMed]
- Canellas, L.P.; Olivares, F.L. Physiological responses to humic substances as plant growth promoter. Chem. Biol. Technol. Agric. 2014, 1, 3. [Google Scholar] [CrossRef]
- Min, K.; Freeman, C.; Kang, H.; Choi, S.-H. The regulation by phenolic compounds of soil organic matter dynamics under a changing environment. BioMed Res. Int. 2015, 2015, 825098. [Google Scholar] [CrossRef]
- Nardi, S.; Pizzeghello, D.; Bragazza, L.; Gerdol, R. Low-molecular-weight organic acids and hormone-like activity of dissolved organic matter in two forest soils in N Italy. J. Chem. Ecol. 2003, 29, 1549–1564. [Google Scholar] [CrossRef]
- Frierer, N.; Schimel, J.P.; Cates, R.G.; Zou, J.P. Influence of balsam poplar tannin fractions on carbon and nitrogen dynamics in Alaskan taiga floodplain soils. Soil Biol. Biochem. 2001, 33, 1827–1839. [Google Scholar] [CrossRef]
- Muscolo, A.; Sidari, M.; Texeira da Silva, J.A. Biological effects of water-soluble soil phenol and soil humic extracts on plant systems. Acta Physiol. Plant. 2013, 35, 309–320. [Google Scholar] [CrossRef]
- Griffiths, P.R.; DeHaseth, J.A. Fourier Transform Infrared Spectroscopy; Wiley-Interscience: Hoboken, NJ, USA, 2007. [Google Scholar]
- Francioso, O.; López-Tobar, E.; Torreggiani, A.; Iriarte, M.; Sanchez-Cortes, S. Stimulated adsorption of humic acids on capped plasmonic Ag nanoparticles investigated by surface-enhanced optical techniques. Langmuir 2019, 35, 4518–4526. [Google Scholar] [CrossRef]
- Aroca, R. Surface-Enhanced Vibrational Spectroscopy; John Wiley & Sons Ltd.: Chichester, UK, 2006; pp. 141–176. [Google Scholar]
- Francioso, O.; Sanchez-Cortes, S.; Tugnoli, V.; Ciavatta, C.; Sitti, L.; Gessa, C. Infrared, Raman, and Nuclear Magnetic Resonance (1 H, 13 C, and 31 P) Spectroscopy in the Study of Fractions of Peat Humic Acids. Appl. Spectrosc. 1996, 50, 1165–1174. [Google Scholar] [CrossRef]
- Mosier-Boss, P.A. Review of SERS Substrates for Chemical Sensing. Nanomaterials 2017, 7, 142. [Google Scholar] [CrossRef] [PubMed]
- Nelson, D.W.; Sommers, L.E. Total carbon, organic carbon and organic matter. In Methods of Soil Analysis. Part 2 Chemical and Microbiological Properties; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; Soil Science Society of America, Inc. Publisher: Madison, WI, USA, 1982; pp. 539–579. [Google Scholar]
- Bradford, M.A. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Leopold, N.; Lendl, B. A new method for fast preparation of highly surface-enhanced raman scattering (SERS) active silver colloids at room temperature by reduction of silver nitrate with hydroxylamine hydrochloride. J. Phys. Chem. B 2003, 107, 5723. [Google Scholar] [CrossRef]
- Sanchez-Cortes, S.; Corrado, G.; Trubetskaya, O.E.; Trubetskoj, O.A.; Hermosin, B.; Saiz-Jimenez, C. Surface-enhanced raman spectroscopy of chernozem humic acid and their fractions obtained by coupled size exclusion chromatography-polyacrylamide gel electrophoresis (SEC-PAGE). Appl. Spectrosc. 2006, 60, 48. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.; Findlay, R.H. The use of a classic lipid extraction method for simultaneous recovery of organic pollutants and microbial lipids from sediments. J. Microbiol. Methods 1996, 27, 63–71. [Google Scholar] [CrossRef]
- Salajova, T.; Salaj, J. Somatic embryogenesis in European black pine (Pinus nigra Arn.). Biol. Plant. 1992, 34, 213–218. [Google Scholar] [CrossRef]
- Muscolo, A.; Panuccio, M.R.; Sidari, M.; Nardi, S. The effects of humic substances on Pinus callus are reversed by 2,4-dichlorophenoxyacetic acid. J. Chem. Ecol. 2005, 31, 577–590. [Google Scholar] [CrossRef]
- Hunt, R. Basic Growth Analysis: Plant Growth Analysis for Beginners; Unwin Hyman Ltd.: London, UK, 1990. [Google Scholar]
- Muscolo, A.; Sidari, M.; Settineri, G.; Papalia, T.; Mallamaci, C.; Attinà, E. Influence of soil properties on bioactive compounds and antioxidant capacity of Brassica rupestris Raf. J. Soil Sci. Plant Nutr. 2019, 19, 808–815. [Google Scholar] [CrossRef]
- Muscolo, A.; Papalia, T.; Settineri, G.; Mallamaci, C.; Panuccio, M.R. Sulfur bentonite-organic-based fertilizers as tool for improving bio-compounds with antioxidant activities in red onion. J. Sci. Food Agric. 2020, 100, 785–793. [Google Scholar] [CrossRef]
- Dávalos, A.; Gómez-Cordovés, C.; Bartolomé, B. Extending applicability of the oxygen radical absorbance capacity (ORAC–fluorescein) assay. J. Agric. Food Chem. 2004, 52, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Sokal, R.R.; Rohlf, R.F. Biometry: The Principles and Practice of Statistics in Biological Research; Freeman: San Francisco, CA, USA, 1981; p. 859. [Google Scholar]
- Mao, J.; Olk, D.C.; Fang, X.; He, Z.; Schmidt-Rohr, K. Influence of animal manure application on the chemical structures of soil organic matter as investigated by advanced solid-state NMR and FT-IR spectroscopy. Geoderma 2008, 146, 353–362. [Google Scholar] [CrossRef]
- Spaccini, R.; Piccolo, A.; Haberhauer, G.; Stemmer, M.; Gerzabek, M.H. Decomposition of maize straw in three European soils as revealed by DRIFT spectra of soil particle fractions. Geoderma 2001, 99, 245–260. [Google Scholar] [CrossRef]
- Lehmann, J.; Kinyangi, J.; Solomon, D. Organic matter stabilization in soil microaggregates: Implications from spatial heterogeneity of organic carbon contents and carbon forms. Biogeochemistry 2007, 85, 45–57. [Google Scholar] [CrossRef]
- Haberhauer, G.; Gerzabek, M.H. Drift and transmission FT-IR spectroscopy of forest soils: An approach to determine decomposition processes of forest litter. Vib. Spectrosc. 1999, 19, 413–417. [Google Scholar] [CrossRef]
- Madari, B.E.; Reeves, J.B.; Machado, P.L.O.A.; Guimarães, C.M.; Torres, E.; McCarty, G.W. Mid- and near-infrared spectroscopic assessment of soil compositional parameters and structural indices in two Ferralsols. Geoderma 2006, 136, 245–259. [Google Scholar] [CrossRef]
- Madejová, J. FTIR techniques in clay mineral studies. Vib. Spectrosc. 2003, 31, 1–10. [Google Scholar] [CrossRef]
- Nuopponen, M.H.; Birch, G.M.; Sykes, R.J.; Lee, S.J.; Stewart, D. Estimation of wood density and chemical composition by means of diffuse reflectance mid-infrared Fourier transform (DRIFT-MIR) spectroscopy. J. Agric. Food Chem. 2006, 54, 34–40. [Google Scholar] [CrossRef]
- Alvarez-Ros, M.C.; Sanchez-Corte, S.; Francioso, O.; Garcıa-Ramos, J.V. Catalytic modification of gallic acid on a silver surface studied by surface-enhanced Raman spectroscopy. J. Raman Spectrosc. 2001, 32, 143–145. [Google Scholar] [CrossRef]
- Alvarez-Ros, M.C.; Sanchez-Corte, S.; Francioso, O.; Garcıa-Ramos, J.V. Adsorption and chemical modification of gallic acid on silver nanoparticles studied by Raman Spectroscopy: Effect of anions and cationic pesticide Paraquat. Can. J. Anal. Sci. Spectr. 2003, 48, 132–138. [Google Scholar]
- Sánchez-Cortés, S.; Francioso, O.; Ciavatta, C.; García-Ramos, J.V.; Gessa, C. pH-Dependent adsorption of fractionated peat humic substances on different silver colloids studied by surface-enhanced Raman spectroscopy. J. Colloid Interface Sci. 1998, 198, 308–318. [Google Scholar] [CrossRef]
- Leyton, P.; Córdova, I.; Lizama-Vergara, P.A.; Gómez-Jeria, J.S.; Aliaga, A.E.; Campos-Vallette, M.M.; Clavijo, E.; García-Ramos, J.V.; Sanchez-Cortes, S. Humic acids as molecular assemblers in the surface-enhanced Raman scattering detection of polycyclic aromatic hydrocarbons. Vib. Spectrosc. 2008, 46, 77–81. [Google Scholar] [CrossRef]
- Dukema, C.; De Vries, S.C.; Booij, H.; Schaafsma, T.J.; Van Kammen, A. Substrate utilization by suspension cultures and somatic embryos of Daucus carota L. measured by 13C NMR. Plant Physiol. 1988, 88, 1332–1337. [Google Scholar] [CrossRef] [PubMed]
- Borisjuk, L.; Rolletschek, H.; Radchuk, R.; Weschke, W.; Wobus, U.; Weber, H. Seed development and differentiation: A role for metabolic regulation. Plant Biol. 2004, 6, 375–386. [Google Scholar] [CrossRef] [PubMed]
- Adeleke, R.A.; Cloete, T.E.; Bertrand, A.; Khasa, D.P. Relationship between plant growth and organic acid exudates from ectomycorrhizal and non-ectomycorrhizal Pinus patula. S. Afr. J. Plant Soil 2015, 32, 183–188. [Google Scholar] [CrossRef]
- Meï, C.; Michaud, M.; Cussac, M.; Albrieux, C.; Gros, V.; Maréchal, E.; Block, M.A.; Jouhet, J.; Rébeillé, F. Levels of polyunsaturated fatty acids correlate with growth rate in plant cell cultures. Sci. Rep. 2015, 5, 15207. [Google Scholar] [CrossRef]
- Cheng, F.; Cheng, Z. Research progress on the use of plant allelopathy in agriculture and the physiological and ecological mechanisms of allelopathy. Review. Front. Plant. Sci. 2015, 6, 1020. [Google Scholar] [CrossRef] [PubMed]

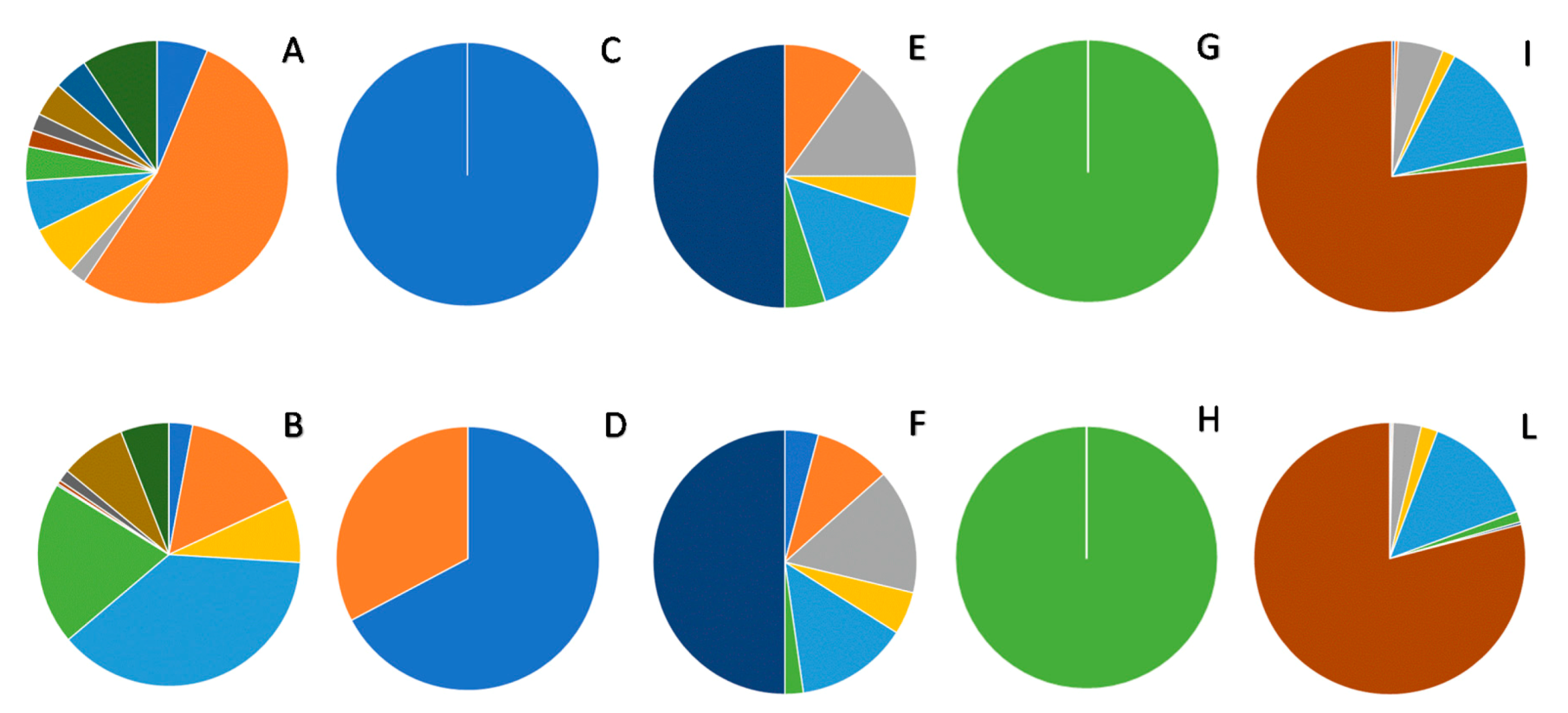

| Class of Compounds | HSs | SWSP | |
|---|---|---|---|
| Phenolic acids | Vanillic acid | 0.6 b* | 4.12 a |
| Benzoic acid | 5.1 b | 21.5 a | |
| Cinnamic acid | 0.2 a | 0.06 b | |
| Salicylic acid | 0.6 b | 11.10 a | |
| Syringic acid | 0.6 b | 53.64 a | |
| Ferulic acid | 0.4 b | 28.3 a | |
| o-Coumaric acid | Nd † | 0.30 | |
| m-Coumaric acid | 0.2 b | 0.65 a | |
| p-Coumaric acid | 0.2 b | 2.07 a | |
| Protocatechuic acid | 0.4 b | 11.5 a | |
| Caffeic acid | 0.4 | Nd | |
| p-Hydroxibenzoic acid | 0.9 b | 8.4 a | |
| Gentisisc acid | Nd | Nd | |
| Aldehydes | Vanillin | 0.4 b | 8.4 a |
| Syringaldehyde | Nd | 4.1 a | |
| Carbohydrates | Arabinose | Nd | 0.11 |
| Glucose | 0.02 b | 0.25 a | |
| Galactose | 0.03 b | 0.41 a | |
| Glucuronic acid | 0.01 b | 0.14 a | |
| Mannose | 0.03 b | 0.37 a | |
| Galacturonic acid | 0.01 b | 0.06 a | |
| Total sugars | 0.1 b | 1.34 a | |
| Organic Acids | Citric | Nd | Nd |
| Fumaric | Nd | Nd | |
| Malic | Nd | Nd | |
| Succinic | Nd | Nd | |
| Oxalic | Nd | Nd | |
| Tartaric | 77 a | 33 b | |
| Fatty Acids | Myristic | 0.07 a | 0.02 b |
| Palmitoleic | 0.08 a | 0.02 b | |
| Palmitic | 1.07 a | 0.34 b | |
| Linoleic | 0.30 a | 0.2 a | |
| Oleic | 2.73 a | 1.38 b | |
| Stearic | 0.36 a | 0.13 b | |
| Arachic | 0.01 a | 0.03 a | |
| Total lipids | 15.2 a | 0.80 b | |
| Treatments | Biomass (g f.w−1) |
|---|---|
| Control | 2.55 ± 0.6 c* |
| 2,4-D+BAP | 5.10 ± 0.2 a |
| HSs | 4.30 ± 0.3 b |
| SWSP | 2.64 ± 0.7 c |
| Treatments | Total Carbohydrates | Sucrose | Glucose | Fructose |
|---|---|---|---|---|
| Control | 20 ± 1.0 a* | 0.33 ± 0.03 c | 4.14 ± 0.3 b | 10.90 ± 2 a |
| 2,4-D+BAP | 14 ± 3 b | 0.33 ± 0.02 c | 1.46 ± 0.4 d | 2.95 ± 0.5 d |
| HSs | 16 ± 2 b | 0.48 ± 0.06 b | 2.50 ± 0.5 c | 3.55 ± 0.7 d |
| SWSP | 23 ± 2 a | 5.25 ± 2.00 a | 4.70 ± 0.2 a | 8.80 ± 3 a |
| Treatments | Prot | TP | Vit. C | Vit. E |
|---|---|---|---|---|
| Control | 1.24 ± 0.3 c* | 1.54 ± 0.1 b | 6.2 ± 2 c | 2.9 ± 1 d |
| 2,4-D+BAP | 1.87 ± 0.2 a | 1.13 ± 0.2 c | 11.4 ± 2 b | 12 ± 3 b |
| HSs | 2.11 ± 0.2 a | 1.19 ± 0.3 c | 16.5 ± 2 a | 20 ± 2 a |
| SWSP | 0.99 ± 0.3 c | 2.99 ± 0.2 a | 6.1 ± 1 c | 6.8 ± 2 c |
| T Carb | Sucr | Glu | Fru | Prot | TP | Vit. C | Vit. E | DPPH | ORAC | ABTS | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Biomass | −0.674 * | −0.371 | −0.838 *** | −0.685 * | 0.887 *** | −0.574 * | 0.772 ** | 0.732 ** | −0.744 ** | −0.921 *** | −0.815 *** |
| T Carb | 0.733 ** | 0.953 *** | 0.823 *** | −0.585 * | 0.875 *** | −0.514 * | −0.451 | 0.875 *** | 0.885 *** | 0.933 *** | |
| Sucr | 0.632 * | 0.437 | −0.501 | 0.927 *** | −0.413 | −0.226 | 0.873 *** | 0.603 * | 0.563 * | ||
| Glu | 0.865 *** | −0.703 ** | 0.814 *** | −0.610 * | −0.570 * | 0.883 *** | 0.971 *** | 0.990 *** | |||
| Fru | −0.577 * | 0.568 * | −0.646 * | −0.691 ** | 0.636 * | 0.864 *** | 0.889 *** | ||||
| Prot | −0.656 * | 0.945 *** | 0.867 *** | −0.771 ** | −0.836 *** | −0.626 * | |||||
| TP | −0.541 * | −0.396 | 0.965 *** | 0.777 ** | 0.740 ** | ||||||
| Vit. C | 0.967 *** | −0.635 * | −0.762 ** | −0.542 * | |||||||
| Vit. E | −0.497 | −0.718 ** | −0.524 | ||||||||
| DPPH | 0.882 *** | 0.818 *** | |||||||||
| ORAC | 0.949 *** |
| Treatments | DPPH | ORAC | ABTS |
|---|---|---|---|
| Control | 100 b* | 1890 ± 7 b | 21 ± 2 a |
| 2,4-D+BAP | 67 d | 1130 ± 9 d | 5.4 ± 2 c |
| HS | 80 c | 1319 ± 12 c | 12.5 ± 3 b |
| WP | 150 a | 1999 ± 6 a | 22 ± 2 a |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muscolo, A.; Pizzeghello, D.; Francioso, O.; Sanchez Cortes, S.; Nardi, S. Effectiveness of Humic Substances and Phenolic Compounds in Regulating Plant-Biological Functionality. Agronomy 2020, 10, 1553. https://doi.org/10.3390/agronomy10101553
Muscolo A, Pizzeghello D, Francioso O, Sanchez Cortes S, Nardi S. Effectiveness of Humic Substances and Phenolic Compounds in Regulating Plant-Biological Functionality. Agronomy. 2020; 10(10):1553. https://doi.org/10.3390/agronomy10101553
Chicago/Turabian StyleMuscolo, Adele, Diego Pizzeghello, Ornella Francioso, Santiago Sanchez Cortes, and Serenella Nardi. 2020. "Effectiveness of Humic Substances and Phenolic Compounds in Regulating Plant-Biological Functionality" Agronomy 10, no. 10: 1553. https://doi.org/10.3390/agronomy10101553
APA StyleMuscolo, A., Pizzeghello, D., Francioso, O., Sanchez Cortes, S., & Nardi, S. (2020). Effectiveness of Humic Substances and Phenolic Compounds in Regulating Plant-Biological Functionality. Agronomy, 10(10), 1553. https://doi.org/10.3390/agronomy10101553









