Application of Nostoc sphaericum and Opuntia ficus-indica Mucilage in the Coagulation–Flocculation Process of Sanitary Landfill Leachate: An Optimization Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Leachate Sampling and Characterization
2.2. Raw Materials and Reagents
2.3. Obtaining Biocoagulants
2.4. Methods for Characterization of the Biocoagulants
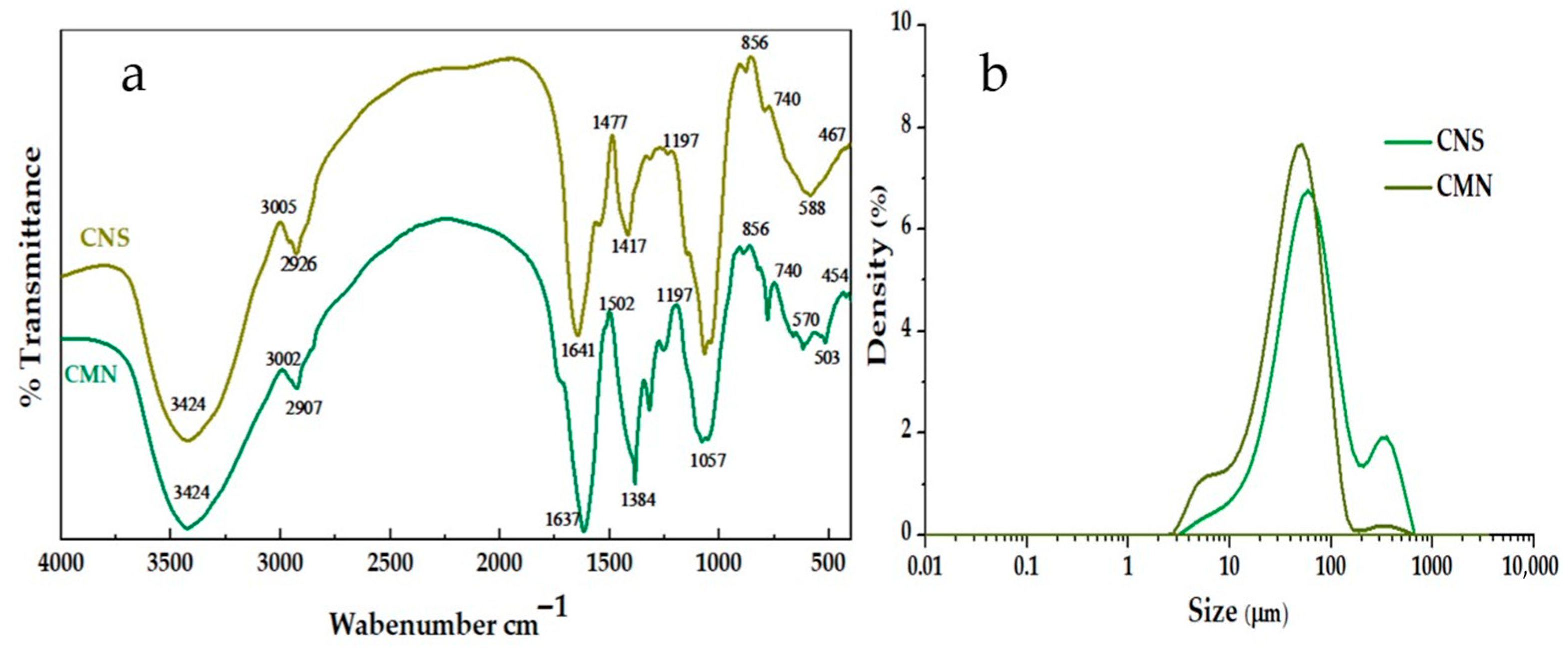
2.4.1. FTIR Analysis
2.4.2. Determination of Particle Size and Zeta Potential
2.4.3. Determination of the Point of Zero Charge
2.5. Experimental Procedure
Experimental Design and Process Configuration
2.6. Optimization and Experimental Validation
2.7. Statistical Techniques
3. Results and Discussion
3.1. Characterization of the Initial Leachate
3.2. Characterization of the Biocoagulants
3.2.1. FTIR Analysis of Biocoagulants
3.2.2. Particle Size Analysis of the Biocoagulants
3.2.3. Zeta Potential (ζ) and Point of Zero Charge (PZC) of the Biocoagulants
3.3. Analysis of Experimental Data
3.4. Model Adequacy Verification
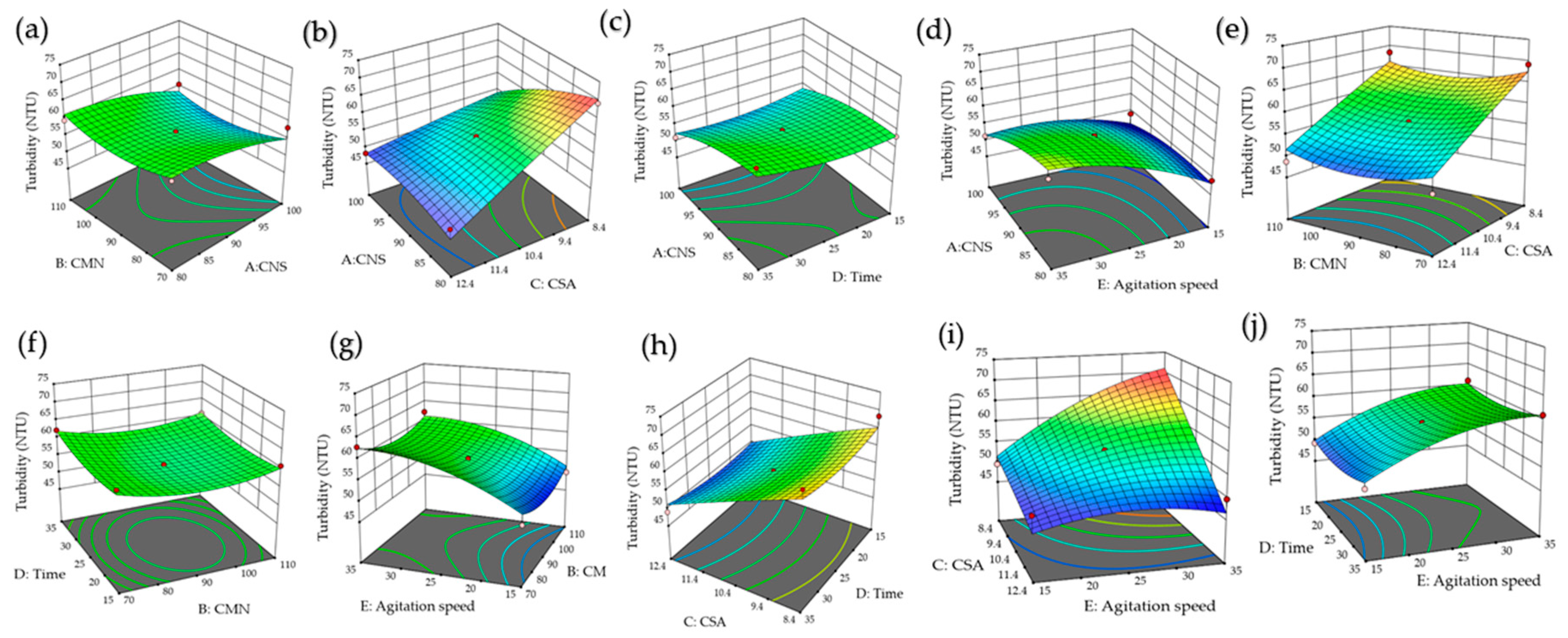
3.5. Response Surface Analysis for Turbidity
3.6. Optimization
3.7. Experimental Validation of the Optimal Treatment
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bouyakhsass, R.; Souabi, S.; Rifi, S.K.; Taleb, A.; Pala, A.; Madinzi, A. Optimization of coagulation-flocculation for landfill leachate treatment: An experimental design approach using response surface methodology. Environ. Nanotechnol. Monit. Manag. 2023, 20, 100841. [Google Scholar] [CrossRef]
- Bakraouy, H.; Souabi, S.; Digua, K.; Dkhissi, O.; Sabar, M.; Fadil, M. Optimization of the treatment of an anaerobic pretreated landfill leachate by a coagulation–flocculation process using experimental design methodology. Process Saf. Environ. Prot. 2017, 109, 621–630. [Google Scholar] [CrossRef]
- Kaur, H.; Kaur, P. Factors Determining Household Waste Segregation Behaviour: An Indian Case Study. Int. J. Exp. Res. Rev. 2024, 41, 83–95. [Google Scholar] [CrossRef]
- Alam, O.; Qiao, X. An in-depth review on municipal solid waste management, treatment and disposal in Bangladesh. Sustain. Cities Soc. 2020, 52, 101775. [Google Scholar] [CrossRef]
- Chen, H.; Xu, H.; Zhong, C.; Liu, M.; Yang, L.; He, J.; Sun, Y.; Zhao, C.; Wang, D. Treatment of landfill leachate by coagulation: A review. Sci. Total Environ. 2024, 912, 169294. [Google Scholar] [CrossRef]
- Abd El-Salam, M.M.; Abu-Zuid, G.I. Impact of landfill leachate on the groundwater quality: A case study in Egypt. J. Adv. Res. 2015, 6, 579–586. [Google Scholar] [CrossRef] [PubMed]
- Laines Canepa, J.R.; Goñi Arévalo, J.A.; Adams Schroeder, R.H.; Camacho Chiu, W. Mezclas con potencial coagulante para tratamiento de lixiviados de un relleno sanitario. Interciencia 2008, 33, 22–28. [Google Scholar]
- Miao, L.; Yang, G.; Tao, T.; Peng, Y. Recent advances in nitrogen removal from landfill leachate using biological treatments—A review. J. Environ. Manag. 2019, 235, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Gajewska, M.; Stosik, M.; Wojciechowska, E.; Obarska-Pempkowiak, H. Influence of wastewater treatment technology on particle size distribution in the effluent. Rocz. Ochr. Srodowiska 2013, 15, 1191–1206. [Google Scholar]
- Chaouki, Z.; El Mrabet, I.; Khalil, F.; Ijjaali, M.; Rafqah, S.; Anouar, S.; Nawdali, M.; Valdés, H.; Zaitan, H. Use of coagulation-flocculation process for the treatment of the landfill leachates of Casablanca city (Morocco). J. Mater. Environ. Sci. 2017, 8, 2781–2791. [Google Scholar]
- Cheng, Z.; Yang, J.; Li, L.; Chen, Y.; Wang, X. Flocculation inspired combination of layered double hydroxides and fulvic acid to form a novel composite adsorbent for the simultaneous adsorption of anionic dye and heavy metals. J. Colloid Interface Sci. 2022, 618, 386–398. [Google Scholar] [CrossRef]
- Chen, W.; Gu, Z.; Ran, G.; Li, Q. Application of membrane separation technology in the treatment of leachate in China: A review. Waste Manag. 2021, 121, 127–140. [Google Scholar] [CrossRef] [PubMed]
- Seifi, M.; Kamran-Pirzaman, A.; Dehghani Kiadehi, A.; Rahimnejad, M. A comprehensive comparison of various methods and hybrid systems in leachate treatment: A review. Int. J. Environ. Sci. Technol. 2025, 22, 8425–8520. [Google Scholar] [CrossRef]
- Tatsi, A.A.; Zouboulis, A.I.; Matis, K.A.; Samaras, P. Coagulation–flocculation pretreatment of sanitary landfill leachates. Chemosphere 2003, 53, 737–744. [Google Scholar] [CrossRef] [PubMed]
- Pashaki, S.G.A.; Khojastehpour, M.; Ebrahimi-Nik, M.; Tedesco, S. Potential of ash from agricultural waste as substitute of commercial FeCl3 in primary treatment of landfill leachate. J. Environ. Manag. 2024, 351, 119932. [Google Scholar] [CrossRef]
- Chaouki, Z.; Hadri, M.; Nawdali, M.; Benzina, M.; Zaitan, H. Treatment of a landfill leachate from Casablanca city by a coagulation-flocculation and adsorption process using a palm bark powder (PBP). Sci. Afr. 2021, 12, e00721. [Google Scholar] [CrossRef]
- Wern, T.Y.; Ardani, M.R.; Ramli, S.F.; Rezan, S.A.; Aziz, H.A.; Ibrahim, I. Coagulation performance of titanium tetrachloride for alor pongsu wastewater treatment. In AIP Conference Proceedings; AIP Publishing: New York, NY, USA, 2020. [Google Scholar]
- Wang, X.; Li, M.; Song, X.; Chen, Z.; Wu, B.; Zhang, S. Preparation and evaluation of titanium-based xerogel as a promising coagulant for water/wastewater treatment. Environ. Sci. Technol. 2016, 50, 9619–9626. [Google Scholar] [CrossRef]
- Aziz, H.A.; Rahim, N.A.; Ramli, S.F.; Alazaiza, M.Y.D.; Omar, F.M.; Hung, Y.-T. Potential use of Dimocarpus longan seeds as a flocculant in landfill leachate treatment. Water 2018, 10, 1672. [Google Scholar] [CrossRef]
- Saravanan, A.; Thamarai, P.; Kumar, P.S.; Rangasamy, G. Recent advances in polymer composite, extraction, and their application for wastewater treatment: A review. Chemosphere 2022, 308, 136368. [Google Scholar] [CrossRef]
- Choudhary, M.; Ray, M.B.; Neogi, S. Evaluation of the potential application of cactus (Opuntia ficus-indica) as a bio-coagulant for pre-treatment of oil sands process-affected water. Sep. Purif. Technol. 2019, 209, 714–724. [Google Scholar] [CrossRef]
- Nharingo, T.; Zivurawa, M.T.; Guyo, U. Exploring the use of cactus Opuntia ficus indica in the biocoagulation–flocculation of Pb(II) ions from wastewaters. Int. J. Environ. Sci. Technol. 2015, 12, 3791–3802. [Google Scholar] [CrossRef]
- Otálora, M.C.; Wilches-Torres, A.; Lara, C.R.; Gómez Castaño, J.A.; Cifuentes, G.R. Evaluation of Turbidity and Color Removal in Water Treatment: A Comparative Study between Opuntia ficus-indica Fruit Peel Mucilage and FeCl3. Polymers 2023, 15, 217. [Google Scholar] [CrossRef]
- Ibarra-Rodríguez, D.; Lizardi-Mendoza, J.; López-Maldonado, E.A.; Oropeza-Guzmán, M.T. Capacity of ‘nopal’ pectin as a dual coagulant-flocculant agent for heavy metals removal. Chem. Eng. J. 2017, 323, 19–28. [Google Scholar] [CrossRef]
- Choque-Quispe, D.; Ligarda-Samanez, C.A.; Choque-Quispe, Y.; Solano-Reynoso, A.M.; Ramos-Pacheco, B.S.; Zamalloa-Puma, M.M.; Álvarez-López, G.J.; Zamalloa-Puma, A.; Choque-Quispe, K.; Alzamora-Flores, H. Multimetal removal in aqueous medium using a potato starch/nopal mucilage copolymer: A study of adsorption kinetics and isotherms. Results Eng. 2023, 18, 101164. [Google Scholar] [CrossRef]
- Choque-Quispe, D.; Ligarda-Samanez, C.A.; Ramos-Pacheco, B.S.; Solano-Reynoso, A.M.; Quispe-Marcatoma, J.; Choque-Quispe, Y.; Peralta-Guevara, D.E.; Martínez-Huamán, E.L.; Correa-Cuba, O.; Masco-Arriola, M.L.; et al. Formulation of Novel Composite (Activated Nanoclay/Hydrocolloid of Nostoc sphaericum) and Its Application in the Removal of Heavy Metals from Wastewater. Polymers 2022, 14, 2803. [Google Scholar] [CrossRef]
- Herbas-De la Cruz, R.K.; Choque-Quispe, Y.; Choque-Quispe, D.; Ligarda-Samanez, C.A.; Froehner, S.; Buleje-Campos, D.; Ramos-Pacheco, B.S.; Peralta-Guevara, D.E.; Pérez-Salcedo, R.; Yauris-Silvera, C.R.; et al. Flocculant capacity of hydrocolloid extracted from high Andean algae (Nostoc sphaericum) in the treatment of artificial wastewater: An approach. Case Stud. Chem. Environ. Eng. 2023, 8, 100515. [Google Scholar] [CrossRef]
- Alnawajha, M.M.; Abdullah, S.R.S.; Hasan, H.A.; Othman, A.R.; Kurniawan, S.B. Effectiveness of using water-extracted Leucaena leucocephala seeds as a coagulant for turbid water treatment: Effects of dosage, pH, mixing speed, mixing time, and settling time. Biomass Convers. Biorefin. 2024, 14, 11203–11216. [Google Scholar] [CrossRef]
- Haddaji, C.; Ennaciri, K.; Driouich, A.; Digua, K.; Souabi, S. Optimization of the coagulation-flocculation process for vegetable oil refinery wastewater using a full factorial design. Process Saf. Environ. Prot. 2022, 160, 803–816. [Google Scholar] [CrossRef]
- Sakhi, D.; Elmchaouri, A.; Rakhila, Y.; Abouri, M.; Souabi, S.; Hamdani, M.; Jada, A. Optimization of the treatment of a real textile wastewater by coagulation– flocculation processes using central composite design. Desalination Water Treat. 2020, 196, 33–40. [Google Scholar] [CrossRef]
- Box, G.E.P.; Behnken, D.W. Simplex-Sum Designs: A Class of Second Order Rotatable Designs Derivable From Those of First Order. Ann. Math. Stat. 1960, 31, 838–864. [Google Scholar] [CrossRef]
- American Public Health, A. Standard Methods for the Examination of Water and Wastewater; American Public Health Association: Washington, DC, USA, 1926; Volume 6. [Google Scholar]
- Rodriguez-Cardenas, L.; Choque-Quispe, Y.; Solano-Reynoso, A.M.; Peralta-Guevara, D.E.; Carhuarupay-Molleda, Y.F.; Agreda-Cerna, H.W.; Correa-Cuba, O.; Palomino-Malpartida, Y.G.; Flores-Ccorisapra, Y.; Reynoso-Canicani, D.D.; et al. Leachate Pollution Index (LPI) in Sanitary Landfills in the High Andean Zones of Peru. Molecules 2025, 30, 3325. [Google Scholar] [CrossRef]
- Choque-Quispe, D.; Mojo-Quisani, A.; Ligarda-Samanez, C.A.; Calla-Florez, M.; Ramos-Pacheco, B.S.; Zamalloa-Puma, L.M.; Peralta-Guevara, D.E.; Solano-Reynoso, A.M.; Choque-Quispe, Y.; Zamalloa-Puma, A.; et al. Preliminary Characterization of a Spray-Dried Hydrocolloid from a High Andean Algae (Nostoc sphaericum). Foods 2022, 11, 1640. [Google Scholar] [CrossRef]
- Kurniawan, S.B.; Imron, M.F.; Chik, C.E.N.C.E.; Owodunni, A.A.; Ahmad, A.; Alnawajha, M.M.; Rahim, N.F.M.; Said, N.S.M.; Abdullah, S.R.S.; Kasan, N.A.; et al. What compound inside biocoagulants/bioflocculants is contributing the most to the coagulation and flocculation processes? Sci. Total Environ. 2022, 806, 150902. [Google Scholar] [CrossRef]
- Ahmed, H.M.; El-Khateeb, M.A.; Mohamed, N.Y.; Sobhy, N.A.; Fawzy, M.E. Evaluation of different natural waste materials as bio-coagulants for domestic wastewater treatment. Desalination Water Treat. 2024, 317, 100034. [Google Scholar] [CrossRef]
- Choque-Quispe, Y.; Solano-Reynoso, A.M.; Choque-Quispe, D.; Ligarda-Samanez, C.A.; Ramos-Pacheco, B.S.; Flores-Ccorisapra, Y.; Carhuarupay-Molleda, Y.F. Optimization of the flocculating capacity of a new micro-atomization extract from a Nostoc cyanobacterium species from the High Andes. Chem. Eng. J. Adv. 2025, 23, 100777. [Google Scholar] [CrossRef]
- Reji, M.; Kumar, R. Response surface methodology (RSM): An overview to analyze multivariate data. Indian J. Microbiol. Res 2022, 9, 241–248. [Google Scholar]
- Khayet, M.; Zahrim, A.Y.; Hilal, N. Modelling and optimization of coagulation of highly concentrated industrial grade leather dye by response surface methodology. Chem. Eng. J. 2011, 167, 77–83. [Google Scholar] [CrossRef]
- Nouj, N.; Majbar, Z.; Abelouah, M.R.; Ben Hamou, A.; Chaoui, A.; Hafid, N.; Benafqir, M.; El Alem, N.; Jada, A.; Ouachtak, H.; et al. Eco-friendly wastewater treatment using a crab shell-based liquid bio-coagulant: Multi-criteria decision analysis related to different pollutants separation. J. Environ. Chem. Eng. 2024, 12, 112318. [Google Scholar] [CrossRef]
- Mittal, M.; Phutela, K. Modeling and parameters optimization of biocomposite using box-Behnken response surface methodology. J. Appl. Res. Technol. 2023, 21, 991–1018. [Google Scholar] [CrossRef]
- Maghami, S.; Esmaeili-Faraj, S.H.; Zarei, M. Levofloxacin removal from wastewater by photocatalytic process with nano-porous SnO2 structure: Experimental analysis by response surface methodology. J. Water Process Eng. 2025, 72, 107557. [Google Scholar] [CrossRef]
- Sahu, U.K.; Mahapatra, S.S.; Patel, R.K. Application of Box–Behnken Design in response surface methodology for adsorptive removal of arsenic from aqueous solution using CeO2/Fe2O3/graphene nanocomposite. Mater. Chem. Phys. 2018, 207, 233–242. [Google Scholar] [CrossRef]
- Banch, T.J.H.; Hanafiah, M.M.; Alkarkhi, A.F.M.; Abu Amr, S.S. Factorial Design and Optimization of Landfill Leachate Treatment Using Tannin-Based Natural Coagulant. Polymers 2019, 11, 1349. [Google Scholar] [CrossRef]
- Otálora, M.C.; Wilches-Torres, A.; Lara, C.R.; Cifuentes, G.R.; Gómez Castaño, J.A. Use of Opuntia ficus-indica Fruit Peel as a Novel Source of Mucilage with Coagulant Physicochemical/Molecular Characteristics. Polymers 2022, 14, 3832. [Google Scholar] [CrossRef]
- Bouaouine, O.; Bourven, I.; Khalil, F.; Baudu, M. Efficiency of a coagulation–flocculation process using Opuntia ficus-indica for the treatment of a textile effluent. Desalination Water Treat. 2021, 240, 137–143. [Google Scholar] [CrossRef]
- Bouaouine, O.; Bourven, I.; Khalil, F.; Baudu, M. Identification of functional groups of Opuntia ficus-indica involved in coagulation process after its active part extraction. Environ. Sci. Pollut. Res. 2018, 25, 11111–11119. [Google Scholar] [CrossRef]
- Hernández Carrillo, C.G.; Gómez-Cuaspud, J.A.; Martínez Suarez, C.E. Compositional, thermal and microstructural characterization of the Nopal (opuntia ficus indica), for addition in commercial cement mixtures. J. Phys. Conf. Ser. 2017, 935, 012045. [Google Scholar] [CrossRef]
- Li, L.X.; Li, F. The effect of carbonyl, carboxyl and hydroxyl groups on the capacitance of carbon nanotubes. Xinxing Tan Cailiao/New Carbon Mater. 2011, 26, 224–228. [Google Scholar] [CrossRef]
- Sue, K.; Arai, K. Specific behavior of acid-base and neutralization reactions in supercritical water. J. Supercrit. Fluids 2004, 28, 57–68. [Google Scholar] [CrossRef]
- Han, Y.-L.; Gao, J.; Yin, Y.-Y.; Jin, Z.-Y.; Xu, X.-M.; Chen, H.-Q. Extraction optimization by response surface methodology of mucilage polysaccharide from the peel of Opuntia dillenii haw. fruits and their physicochemical properties. Carbohydr. Polym. 2016, 151, 381–391. [Google Scholar] [CrossRef]
- Fard, M.B.; Hamidi, D.; Yetilmezsoy, K.; Alavi, J.; Hosseinpour, F. Utilization of Alyssum mucilage as a natural coagulant in oily-saline wastewater treatment. J. Water Process Eng. 2021, 40, 101763. [Google Scholar] [CrossRef]
- Saranya, P.; Ramesh, S.T.; Gandhimathi, R. Effectiveness of natural coagulants from non-plant-based sources for water and wastewater treatment—A review. Desalination Water Treat. 2014, 52, 6030–6039. [Google Scholar] [CrossRef]
- Yin, C.Y. Emerging usage of plant-based coagulants for water and wastewater treatment. Process Biochem. 2010, 45, 1437–1444. [Google Scholar] [CrossRef]
- Mikol, V.; Vincendon, P.; Eriani, G.; Hirsch, E.; Giegé, R. Diagnostic of protein crystallization by dynamic light scattering; an application to an aminoacyl-tRNA synthetase. J. Cryst. Growth 1991, 110, 195–200. [Google Scholar] [CrossRef]
- Cuadros-Moreno, A.; Pimentel, R.G.C.; Martínez, E.S.M.; Fernandez, J.Y. Dispersión de luz dinámica en la determinación de tamaño de nanopartículas poliméricas. Lat.-Am. J. Phys. Educ. 2014, 8, 14. [Google Scholar]
- Lecca-Malo, S.; Martínez-Cerna, D.; Alvarado, C.; Alvarado-Quintana, H. Effects of the Concentration and Proportion of Nopal Cactus Coagulant on the Clarification of Water for Irrigation. In Proceedings of the LACCEI International Multi-Conference for Engineering, Education and Technology, San José, Costa Rica, 17–19 July 2024. [Google Scholar]
- Lugo-Arias, J.; Lugo-Arias, E.; Ovallos-Gazabon, D.; Arango, J.; de la Puente, M.; Silva, J. Effectiveness of the mixture of nopal and cassava starch as clarifying substances in water purification: A case study in Colombia. Heliyon 2020, 6, e04296. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Jiao, R.; Xu, H.; An, G.; Wang, D. The influence of particle size and concentration combined with pH on coagulation mechanisms. J. Environ. Sci. 2019, 82, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.; Manning, G.; Zakharova, J.; Arjunan, A.; Batool, M.; Hawkins, A.J. Particle size effect of Moringa oleifera Lam. seeds on the turbidity removal and antibacterial activity for drinking water treatment. Environ. Chem. Ecotoxicol. 2024, 6, 370–379. [Google Scholar] [CrossRef]
- Chasquibol, N.; Sotelo, A.; Tapia, M.; Paredes, Y.; Sajid, A.; Sahin, S.; Wu, M.; Goycoolea, F.M. Extraction and characterization of cushuro (Nostoc sphaericum) polysaccharides. LWT 2025, 230, 118239. [Google Scholar] [CrossRef]
- Choque-Quispe, D.; Ligarda-Samanez, C.A.; Choque-Quispe, Y.; Froehner, S.; Solano-Reynoso, A.M.; Moscoso-Moscoso, E.; Carhuarupay-Molleda, Y.F.; Peréz-Salcedo, R. Stability in Aqueous Solution of a New Spray-Dried Hydrocolloid of High Andean Algae Nostoc sphaericum. Polymers 2024, 16, 537. [Google Scholar] [CrossRef]
- Choque-Quispe, Y.; Choque-Quispe, D.; Ligarda-Samanez, C.A.; Solano-Reynoso, A.M.; Froehner, S.; Ramos-Pacheco, B.S.; Carhuarupay-Molleda, Y.F.; Sumarriva-Bustinza, L.A. A High Andean Hydrocolloid Extracted by Microatomization: Preliminary Optimization in Aqueous Stability. Polymers 2024, 16, 1777. [Google Scholar] [CrossRef]
- Chua, S.-C.; Malek, M.A.; Chong, F.-K.; Sujarwo, W.; Ho, Y.-C. Red Lentil (Lens culinaris) Extract as a Novel Natural Coagulant for Turbidity Reduction: An Evaluation, Characterization and Performance Optimization Study. Water 2019, 11, 1686. [Google Scholar] [CrossRef]
- Othmani, B.; Gamelas, J.A.F.; Mendes, C.V.T.; Rasteiro, M.G.; Khadhraoui, M. Green Flocculants from Cactus Cladodes: Physicochemical Characterization and Assessment of Their Flocculating Activity for Crystal Violet Dye Removal. Water Air Soil Pollut. 2024, 235, 458. [Google Scholar] [CrossRef]
- Otálora, M.C.; Wilches-Torres, A.; Lara, C.R.; Díaz-Gómez, J.; Gómez Castaño, J.A.; Cifuentes, G.R. Assessment of Prickly Pear Fruit Peel Mucilage in Form of Gel as a Green Coagulant for the Tertiary Treatment of Domestic Wastewater. Gels 2023, 9, 723. [Google Scholar] [CrossRef]
- Bakatula, E.N.; Richard, D.; Neculita, C.M.; Zagury, G.J. Determination of point of zero charge of natural organic materials. Environ. Sci. Pollut. Res. 2018, 25, 7823–7833. [Google Scholar] [CrossRef] [PubMed]
- Amin, N.K.; Abdelwahab, O.; El-Ashtoukhy, E.Z.; Abdel-Aziz, M.H. Comparative analysis of new natural coagulant extracts for turbidity removal in water systems. Water Sci. Technol. 2025, 91, 797–810. [Google Scholar] [CrossRef]
- Chen, D.; Dong, C.; Pan, J.; Engelhardt, G.R.; Macdonald, D.D. Development of the General Methodology for Determining the Point of Zero Charge of Oxide Films by Contact Angle Titration. J. Electrochem. Soc. 2023, 170, 081504. [Google Scholar] [CrossRef]
- Nasiruddin Khan, M.; Sarwar, A. Determination of points of zero charge of natural and treated adsorbents. Surf. Rev. Lett. 2007, 14, 461–469. [Google Scholar] [CrossRef]
- Ghebremichael, K.; Abaliwano, J.; Amy, G. Combined natural organic and synthetic inorganic coagulants for surface water treatment. J. Water Supply Res. Technol.—AQUA 2009, 58, 267–276. [Google Scholar] [CrossRef]
- Likando, N.M.; Dornack, C.; Hamutoko, J.T. Assessing the physicochemical parameters of leachate from biowaste fractions in a laboratory setting, using the elusion method. Environ. Earth Sci. 2023, 82, 610. [Google Scholar] [CrossRef]
- Elmoubarki, R.; Taoufik, M.; Moufti, A.; Tounsadi, H.; Mahjoubi, F.Z.; Bouabi, Y.; Qourzal, S.; Abdennouri, M.; Barka, N. Box-Behnken experimental design for the optimization of methylene blue adsorption onto Aleppo pine cones. J. Mater. Environ. Sci 2017, 8, 2184–2191. [Google Scholar]
- Semassel, S.E.; Elhadeuf, K.; Bougdah, N.; Messikh, N. Enhancing the Clarification of Azzaba Landfill Leachate Using Biocoagulants with Optimization by Box Behnken Design. Chem. Chem. Technol. 2025, 19, 307–326. [Google Scholar] [CrossRef]
- Wae AbdulKadir, W.A.F.; Che Omar, R.; Shamsudin, M.S. Ammoniacal nitrogen—A critical contaminant in leachate composition: Characteristics, potential risk, and adsorption treatment innovations. J. Water Process Eng. 2024, 62, 105424. [Google Scholar] [CrossRef]
- Baatache, O.; Benalia, A.; Derbal, K.; Khalfaoui, A.; Pizzi, A. Optimized Coagulation Flocculation of Drinking Water Using Pine cone-Based Bio-Coagulants: A Comparative Study of Different Extracts. Water 2025, 17, 1793. [Google Scholar] [CrossRef]
- Hogg, R. Bridging Flocculation by Polymers. KONA Powder Part. J. 2013, 30, 3–14. [Google Scholar] [CrossRef]
- Deong, J.L.; Mazatusziha, A.; Nur Sabrina, A. Comparative study on micronsized and nanosized carica papaya seed modified pullulan as biocoagulant in wastewater treatment. Solid State Phenom. 2021, 317, 276–282. [Google Scholar]
- Kurniawan, S.B.; Ahmad, A.; Said, N.S.M.; Gustinasari, K.; Abdullah, S.R.S.; Imron, M.F. The influence of preparation and pretreatment on the physicochemical properties and performance of plant-based biocoagulants in treating wastewater. Environ. Adv. 2023, 14, 100441. [Google Scholar] [CrossRef]
- Righetto, I.; Al-Juboori, R.A.; Kaljunen, J.U.; Mikola, A. Multipurpose treatment of landfill leachate using natural coagulants—Pretreatment for nutrient recovery and removal of heavy metals and micropollutants. J. Environ. Chem. Eng. 2021, 9, 105213. [Google Scholar] [CrossRef]
- Gunasheela, K.N.; Raju, V.K.; Ramakrishnan, M. Screening and Selection of Plant-Based Coagulants for Treating Household Greywater. Indian J. Environ. Prot. 2025, 45, 939–947. [Google Scholar]
- Shi, G. Application of plackett-burman design for determining key factors of ammoniacal nitrogen removal from wastewate. Chin. J. Environ. Eng. 2014, 8, 2936–2940. [Google Scholar]
- Thakare, N.P.; Bhanvase, B.A. A review on ammoniacal nitrogen removal and its recovery approaches from wastewater. Sep. Purif. Technol. 2025, 379, 134847. [Google Scholar] [CrossRef]
- Biswal, T.; Sethy, T.R.; Sahoo, P.K. Sustainable Removal of Contaminants by Biopolymers. In Bio-Based Polymers: Farm to Industry. Volume 2: Current Trends and Applications; American Chemical Society: Washington, DC, USA, 2024; Volume 1486, pp. 197–214. [Google Scholar]
- Moharana, S.; Sahu, B.B.; Maharana, T. Biological Methods of Heavy Metal Management—Biopolymer. In Heavy Metals in the Environment: Management Strategies for Global Pollution; American Chemical Society: Washington, DC, USA, 2023; Volume 1456, pp. 361–392. [Google Scholar]
- Heidari Farsani, M.; Jalilzadeh Yengejeh, R.; Hajiseyed Mirzahosseini, A.; Monavari, M.; Hassani, A.H.; Mengelizadeh, N. Study of the performance of bench-scale electro-membranes bioreactor in leachate treatment. Adv. Environ. Technol. 2021, 7, 209–220. [Google Scholar] [CrossRef]
- Prodanović, J.M.; Nastić, N.M.; Šćiban, M.B.; Lukić, D.V.; Vasić, V.M.; Blagojev, N.T. An Investigation into the Factors Influencing the Effectiveness of a Biodegradable Natural Coagulant Derived from Phaseolus vulgaris Seeds and Coagulation Mechanism. Sustainability 2025, 17, 1372. [Google Scholar] [CrossRef]




| Parameters | Method | Unit | Reference |
|---|---|---|---|
| pH | Selective electrode | - | Standard Methods 4500—H + B [32] |
| Electrical Conductivity (EC) | Selective electrode | mS/cm | Standard Methods 2510 B [32] |
| Turbidity | Nephelometry | NTU | Standard Methods 2130 B [32] |
| Biochemical Oxygen Demand (BOD5) | Respirometric, manometric oxytope method | mg/L | Standard Methods 5210D [32] |
| Chemical Oxygen Demand (COD) | Closed reflux, colorimetric method | mg/L | Standard Methods 5220B [32] |
| Metals (Fe, Cu, Cd, Zn, Pb, Cr, As, Ni, Al) | Optical emission spectrometry | mg/L | ICP-OES 9820, Standard Methods 3120 B [32] |
| Total Organic Carbon (TOC) | Catalytic combustion | mg/L | Standard Methods 5310 B [32] |
| Phosphates | Ascorbic acid method | mg/L | Standard Methods 4500-P E [32] |
| Ammonia Nitrogen (NH3-N) | Nessler method | mg/L | ASTM Standards, D 1426 [33] |
| Surfactants | Methylene blue method | mg/L | Standard Methods 5540 C [32] |
| Variable | Symbol | −1 | 0 | 1 |
|---|---|---|---|---|
| CNS (mg/L) | A | 80 | 90 | 100 |
| CMN (mg/L) | B | 70 | 90 | 110 |
| CSA (mg/L) | C | 8.4 | 10.4 | 12.4 |
| Mixing time (min) | D | 15 | 25 | 35 |
| Agitation speed (rpm) | E | 15 | 25 | 35 |
| Parameters | ±S | Limit mg/L | |
|---|---|---|---|
| PH | 8.01 | 0.00 | - |
| EC (mS/cm) | 12.6 | 0.00 | - |
| Turbidity (NTU) | 164.8 | 0.05 | - |
| NH3-N (mg/L) | 640.0 | 0.05 | 5 [44] |
| Phosphates (mg/L) | 0.23 | 0.00 | - |
| Surfactants (mg/L) | 0.31 | 0.00 | - |
| COD (mg/L) | 817 | 0.00 | 400 [44] |
| BOD5 (mg/L) | 563 | 0.58 | 20 [44] |
| BOD5/COD | 0.69 | - | - |
| TOC (mg/L) | 252.3 | 0.06 | - |
| As (mg/L) | 0.75 | 0.01 | 0.050 [44] |
| Cd (mg/L) | 0.48 | 0.01 | 0.010 [44] |
| Cu (mg/L) | 0.57 | 0.01 | 0.020 [44] |
| Cr (mg/L) | 0.38 | 0.00 | 0.010 [44] |
| Fe (mg/L) | 0.31 | 0.00 | 0.005 [44] |
| Pb (mg/L) | 0.38 | 0.00 | 0.010 [44] |
| Zn (mg/L) | 0.20 | 0.01 | 2 [44] |
| Al (mg/L) | 0.35 | 0.00 | - |
| Ni (mg/L) | 0.05 | 0.00 | - |
| Coefficient | Turbidity | p-Value | pH | p-Value | EC | p-Value |
|---|---|---|---|---|---|---|
| Intercept | 56.687 | - | 6.656 | - | 12.551 | - |
| A | −3.507 | <0.0001 | 0.001 | 0.9764 | 0.010 | 0.7861 |
| B | −0.467 | 0.4936 | −0.027 | 0.5251 | 0.025 | 0.4974 |
| C | −7.557 | <0.0001 | −0.567 | <0.0001 | 1.061 | <0.0001 |
| D | 0.460 | 0.4999 | −0.077 | 0.0817 | 0.001 | 0.9770 |
| E | 5.102 | <0.0001 | 0.157 | 0.0011 | −0.091 | 0.0173 |
| AB | −0.384 | 0.7778 | −0.034 | 0.6900 | 0.053 | 0.4690 |
| AC | 4.508 | 0.0026 | 0.033 | 0.7014 | 0.000 | 1.0000 |
| AD | −1.134 | 0.4072 | −0.005 | 0.9513 | 0.051 | 0.4831 |
| AE | −2.250 | 0.1067 | −0.001 | 0.9954 | −0.003 | 0.9723 |
| BC | 0.274 | 0.8405 | −0.014 | 0.8729 | 0.011 | 0.8806 |
| BD | 0.025 | 0.9853 | 0.048 | 0.5744 | −0.005 | 0.9447 |
| BE | −0.250 | 0.8540 | −0.106 | 0.2258 | −0.038 | 0.5961 |
| CD | 0.175 | 0.8973 | −0.222 | 0.0151 | −0.140 | 0.0611 |
| CE | −4.683 | 0.0018 | 0.068 | 0.4322 | 0.040 | 0.5803 |
| DE | −0.125 | 0.9267 | 0.027 | 0.7580 | −0.012 | 0.8715 |
| A2 | −1.787 | 0.0608 | −0.056 | 0.3364 | −0.074 | 0.1403 |
| B2 | 2.756 | 0.0056 | −0.094 | 0.1136 | −0.097 | 0.0565 |
| C2 | −0.237 | 0.7970 | −0.497 | <0.0001 | 0.054 | 0.2731 |
| D2 | 1.603 | 0.0905 | −0.149 | 0.0160 | −0.045 | 0.3579 |
| E2 | −2.786 | 0.0052 | −0.022 | 0.7105 | −0.093 | 0.0672 |
| Lack of fit | <0.0001 | 0.5674 | <0.0001 | |||
| R2 | - | 0.9165 | - | 0.9194 | - | 0.9732 |
| Adjusted R2 | - | 0.8496 | - | 0.8549 | - | 0.9517 |
| Adequate Precision | - | 13.9333 | - | 14.6613 | - | 25.2733 |
| predicted R2 | - | 0.6658 | 0.7195 | - | 0.8929 |
| Parameters | Initial | Treatment (CNS, CMN, CSA) | t-Test Significance | %R | ||||
|---|---|---|---|---|---|---|---|---|
| ±S | CV | ±S | CV | |||||
| PH | 8.02 | 0.00 | 0.00 | 6.47 | 0.03 | 0.45 | ** | -- |
| EC (mS/cm) | 12.61 | 0.00 | 0.00 | 11.45 | 0.00 | 0.01 | ** | 9.2 |
| Turbidity (NTU) | 163.52 | 0.01 | 0.00 | 49.02 | 0.02 | 0.04 | ** | 70.0 |
| NH3-N (mg/L) | 640.03 | 0.05 | 0.01 | 560.25 | 0.50 | 0.09 | ** | 12.5 |
| Phosphates (mg/L) | 0.23 | 0.00 | 0.25 | 0.12 | 0.00 | 0.83 | ** | 47.7 |
| Surfactants (mg/L) | 0.31 | 0.00 | 0.19 | 0.25 | 0.00 | 0.40 | ** | 19.5 |
| COD (mg/L) | 816.99 | 0.01 | 0.00 | 697.25 | 0.50 | 0.07 | ** | 14.7 |
| BOD5 (mg/L) | 563.33 | 0.58 | 0.10 | 424.00 | 0.00 | 0.00 | ** | 24.7 |
| BOD5/COD | 0.69 | - | - | 0.61 | - | - | - | - |
| TOC (mg/L) | 252.20 | 0.17 | 0.07 | 87.60 | 0.00 | 0.00 | ** | 65.3 |
| As (mg/L) | 0.75 | 0.01 | 0.77 | 0.33 | 0.00 | 0.02 | ** | 55.9 |
| Cd (mg/L) | 0.48 | 0.00 | 0.12 | 0.29 | 0.00 | 0.00 | ** | 40.0 |
| Cu (mg/L) | 0.57 | 0.00 | 0.10 | 0.19 | 0.00 | 0.83 | ** | 67.7 |
| Cr (mg/L) | 0.38 | 0.00 | 0.00 | 0.23 | 0.00 | 0.00 | ** | 39.2 |
| Fe (mg/L) | 0.31 | 0.00 | 0.19 | 0.20 | 0.00 | 0.00 | ** | 36.6 |
| Pb (mg/L) | 0.38 | 0.00 | 0.00 | 0.30 | 0.00 | 0.19 | ** | 20.8 |
| Zn (mg/L) | 0.20 | 0.00 | 0.36 | 0.16 | 0.00 | 0.37 | ** | 19.6 |
| Al (mg/L) | 0.35 | 0.00 | 0.17 | 0.28 | 0.00 | 0.00 | ** | 19.8 |
| Ni (mg/L) | 0.05 | 0.00 | 0.00 | 0.04 | 0.00 | 0.17 | ** | 35.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Choque-Quispe, Y.; Solano-Reynoso, A.M.; Dueñas-Valcarcel, C.E.; Arostegui-Leon, E.; Rodriguez-Cardenas, L.; Choque-Quispe, D. Application of Nostoc sphaericum and Opuntia ficus-indica Mucilage in the Coagulation–Flocculation Process of Sanitary Landfill Leachate: An Optimization Study. Polymers 2026, 18, 474. https://doi.org/10.3390/polym18040474
Choque-Quispe Y, Solano-Reynoso AM, Dueñas-Valcarcel CE, Arostegui-Leon E, Rodriguez-Cardenas L, Choque-Quispe D. Application of Nostoc sphaericum and Opuntia ficus-indica Mucilage in the Coagulation–Flocculation Process of Sanitary Landfill Leachate: An Optimization Study. Polymers. 2026; 18(4):474. https://doi.org/10.3390/polym18040474
Chicago/Turabian StyleChoque-Quispe, Yudith, Aydeé M. Solano-Reynoso, Carlos Eduardo Dueñas-Valcarcel, Edwar Arostegui-Leon, Liliana Rodriguez-Cardenas, and David Choque-Quispe. 2026. "Application of Nostoc sphaericum and Opuntia ficus-indica Mucilage in the Coagulation–Flocculation Process of Sanitary Landfill Leachate: An Optimization Study" Polymers 18, no. 4: 474. https://doi.org/10.3390/polym18040474
APA StyleChoque-Quispe, Y., Solano-Reynoso, A. M., Dueñas-Valcarcel, C. E., Arostegui-Leon, E., Rodriguez-Cardenas, L., & Choque-Quispe, D. (2026). Application of Nostoc sphaericum and Opuntia ficus-indica Mucilage in the Coagulation–Flocculation Process of Sanitary Landfill Leachate: An Optimization Study. Polymers, 18(4), 474. https://doi.org/10.3390/polym18040474








