Sustainable Waterborne Polylactide Coatings Enabled by Hydrophobic Deep Eutectic Solvents Plasticization
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
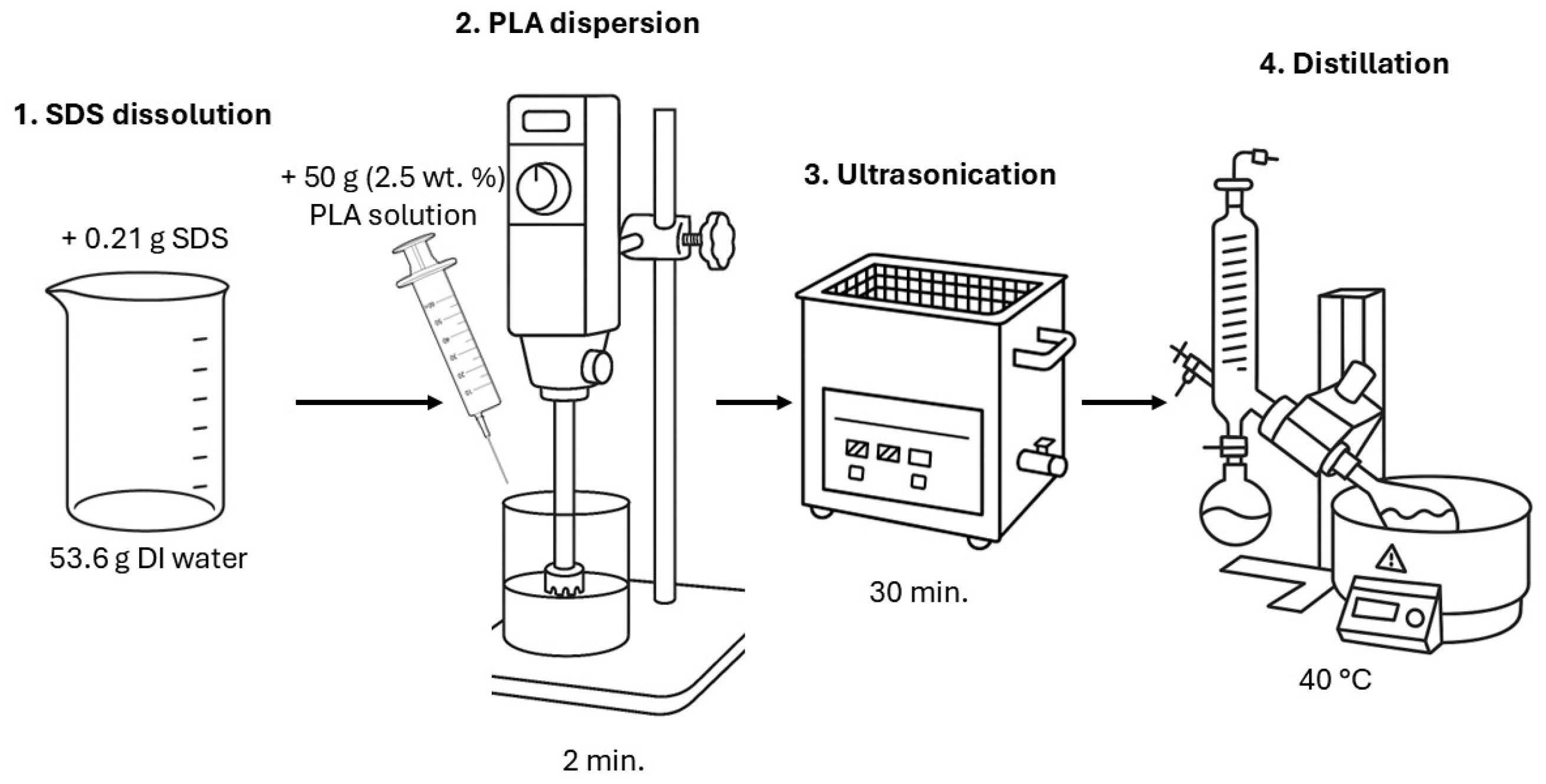
2.2. Obtaining Polylactide Dispersions
2.3. Obtaining Biofillers
2.4. Characterization Methods
3. Results and Discussion
NMR and FTIR Characterization of HDES
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PLA | Polylactide |
| MFFT | Minimum film formation temperature |
| HDES | Hydrophobic deep eutectic solvent |
| NMR | Nuclear magnetic resonance |
| FTIR | Fourier-transform infrared spectroscopy |
| RED | Relative energy difference |
| PEG | Polyethylene glycol |
| SDS | Sodium dodecyl sulfate |
| DSC | Differential scanning calorimetry |
References
- Righetti, G.I.C.; Faedi, F.; Famulari, A. Embracing Sustainability: The World of Bio-Based Polymers in a Mini Review. Polymers 2024, 16, 950. [Google Scholar] [CrossRef]
- Cywar, R.M.; Rorrer, N.A.; Hoyt, C.B.; Beckham, G.T.; Chen, E.Y.-X. Bio-Based Polymers with Performance-Advantaged Properties. Nat. Rev. Mater. 2021, 7, 83–103. [Google Scholar] [CrossRef]
- Naser, A.Z.; Deiab, I.; Darras, B.M. Poly(Lactic Acid) (PLA) and Polyhydroxyalkanoates (PHAs), Green Alternatives to Petroleum-Based Plastics: A Review. RSC Adv. 2021, 11, 17151–17196. [Google Scholar] [CrossRef] [PubMed]
- Barletta, M.; Aversa, C.; Ayyoob, M.; Gisario, A.; Hamad, K.; Mehrpouya, M.; Vahabi, H. Poly(Butylene Succinate) (PBS): Materials, Processing, and Industrial Applications. Prog. Polym. Sci. 2022, 132, 101579. [Google Scholar] [CrossRef]
- Sanders, J.H.; Cunniffe, J.; Carrejo, E.; Burke, C.; Reynolds, A.M.; Dey, S.C.; Islam, M.N.; Wagner, O.; Argyropoulos, D. Biobased Polyethylene Furanoate: Production Processes, Sustainability, and Techno-Economics. Adv. Sustain. Syst. 2024, 8, 2400074. [Google Scholar] [CrossRef]
- Hayes, G.; Laurel, M.; MacKinnon, D.; Zhao, T.; Houck, H.A.; Becer, C.R. Polymers without petrochemicals: Sustainable routes to conventional monomers. Chem. Rev. 2022, 123, 2609–2734. [Google Scholar] [CrossRef]
- Goliszek-Chabros, M.; Smyk, N.; Xu, T.; Matwijczuk, A.; Podkościelna, B.; Sevastyanova, O. Lignin Nanoparticle-Enhanced PVA Foils for UVB/UVC Protection. Sci. Rep. 2025, 15, 35735. [Google Scholar] [CrossRef]
- Goliszek, M.; Podkościelna, B.; Smyk, N.; Sevastyanova, O. Towards Lignin Valorization: Lignin as a UV-Protective Bio-Additive for Polymer Coatings. Pure Appl. Chem. 2023, 95, 475–486. [Google Scholar] [CrossRef]
- Anjum, A.; Zuber, M.; Zia, K.M.; Noreen, A.; Anjum, M.N.; Tabasum, S. Microbial production of polyhydroxyalkanoates (phas) and its copolymers: A review of recent advancements. Int. J. Biol. Macromol. 2016, 89, 161–174. [Google Scholar] [CrossRef]
- Khouri, N.G.; Bahú, J.O.; Blanco-Llamero, C.; Severino, P.; Concha, V.O.C.; Souto, E.B. Polylactic Acid (PLA): Properties, Synthesis, and Biomedical Applications—A Review of the Literature. J. Mol. Struct. 2024, 1309, 138243. [Google Scholar] [CrossRef]
- Belletti, G.; Buoso, S.; Ricci, L.; Guillem-Ortiz, A.; Aragón-Gutiérrez, A.; Bortolini, O.; Bertoldo, M. Preparations of Poly(lactic acid) Dispersions in Water for Coating Applications. Polymers 2021, 13, 2767. [Google Scholar] [CrossRef] [PubMed]
- Pieters, K.; Mekonnen, T.H. Stable aqueous dispersions of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) (PHBV) polymer for barrier paper coating. Prog. Org. Coat. 2023, 187, 108101. [Google Scholar] [CrossRef]
- Calosi, M.; D’Iorio, A.; Buratti, E.; Cortesi, R.; Franco, S.; Angelini, R.; Bertoldo, M. Preparation of high-solid PLA waterborne dispersions with PEG-PLA-PEG block copolymer as surfactant and their use as hydrophobic coating on paper. Prog. Org. Coat. 2024, 193, 108541. [Google Scholar] [CrossRef]
- Nguyen, H.T.H.; Qi, P.; Rostagno, M.; Feteha, A.; Miller, S.A. The Quest for High Glass Transition Temperature Bioplastics. J. Mater. Chem. A 2018, 6, 9298–9331. [Google Scholar] [CrossRef]
- Sun, S.; Weng, Y.; Zhang, C. Recent Advancements in Bio-Based Plasticizers for Polylactic Acid (PLA): A Review. Polym. Test. 2024, 140, 108603. [Google Scholar] [CrossRef]
- Mastalygina, E.E.; Aleksanyan, K.V. Recent Approaches to the Plasticization of Poly(Lactic Acid) (PLA) (A Review). Polymers 2023, 16, 87. [Google Scholar] [CrossRef]
- Li, D.; Jiang, Y.; Lv, S.; Liu, X.; Gu, J.; Chen, Q.; Zhang, Y. Preparation of Plasticized Poly (Lactic Acid) and Its Influence on the Properties of Composite Materials. PLoS ONE 2018, 13, e0193520. [Google Scholar] [CrossRef]
- Harte, I.; Birkinshaw, C.; Jones, E.; Kennedy, J.; DeBarra, E. The Effect of Citrate Ester Plasticizers on the Thermal and Mechanical Properties of Poly(DL-lactide). J. Appl. Polym. Sci. 2012, 127, 1997–2003. [Google Scholar] [CrossRef]
- Shamshina, J.L.; Berton, P. Ionic Liquids as Designed, Multi-Functional Plasticizers for Biodegradable Polymeric Materials: A Mini-Review. Int. J. Mol. Sci. 2024, 25, 1720. [Google Scholar] [CrossRef]
- Rebelo, L.P.N.; Lopes, J.N.C.; Esperança, J.M.S.S.; Filipe, E. On the Critical Temperature, Normal Boiling Point, and Vapor Pressure of Ionic Liquids. J. Phys. Chem. B 2005, 109, 6040–6043. [Google Scholar] [CrossRef]
- Rooney, D.; Jacquemin, J.; Gardas, R. Thermophysical Properties of Ionic Liquids. Top. Curr. Chem. 2009, 290, 185–212. [Google Scholar] [CrossRef]
- Dutkowski, K.; Kruzel, M.; Smuga-Kogut, M.; Walczak, M. A Review of the State of the Art on Ionic Liquids and Their Physical Properties during Heat Transfer. Energies 2025, 18, 4053. [Google Scholar] [CrossRef]
- Chaos, A.; Sangroniz, A.; Fernández, J.; del Río, J.; Iriarte, M.; Sarasua, J.R.; Etxeberria, A. Plasticization of poly(lactide) with poly(ethylene glycol): Low weight plasticizer vs triblock copolymers. Effect on free volume and barrier properties. J. Appl. Polym. Sci. 2020, 137, 48868. [Google Scholar] [CrossRef]
- Mohammed, A.A.B.A.; Hasan, Z.; Omran, A.A.B.; Elfaghi, A.M.; Khattak, M.A.; Ilyas, R.A.; Sapuan, S.M. Effect of Various Plasticizers in Different Concentrations on Physical, Thermal, Mechanical, and Structural Properties of Wheat Starch-Based Films. Polymers 2023, 15, 63. [Google Scholar] [CrossRef]
- Jarray, A.; Gerbaud, V.; Hemati, M. Polymer-plasticizer compatibility during coating formulation: A multi-scale investigation. Prog. Org. Coat. 2016, 101, 195–206. [Google Scholar] [CrossRef]
- Arjmandi, A.; Bi, H.; Nielsen, S.U.; Dam-Johansen, K. From wet to protective: Film formation in Waterborne Coatings. ACS Appl. Mater. Interfaces 2024, 16, 58006–58028. [Google Scholar] [CrossRef]
- Yomo, S. Curing Behavior of Waterborne Paint Containing Catalyst Encapsulated in Micelle. Coatings 2021, 11, 375. [Google Scholar] [CrossRef]
- Myronyuk, O.; Baklan, D.; Bilousova, A.; Smalii, I.; Vorobyova, V.; Halysh, V.; Trus, I. Plasticized Polylactide Film Coating Formation from Redispersible Particles. AppliedChem 2025, 5, 14. [Google Scholar] [CrossRef]
- De Los Ríos, M.D.; Belmonte, R.M. Extending Microsoft Excel and Hansen Solubility Parameters Relationship to Double Hansen’s Sphere Calculation. SN Appl. Sci. 2022, 4, 185. [Google Scholar] [CrossRef]
- De Los Ríos, M.D.; Ramos, E.H. Determination of the Hansen Solubility Parameters and the Hansen Sphere Radius with the Aid of the Solver Add-in of Microsoft Excel. SN Appl. Sci. 2020, 2, 676. [Google Scholar] [CrossRef]
- Fernandes, C.C.; Paiva, A.; Haghbakhsh, R.; Duarte, A.R.C. Application of Hansen Solubility Parameters in the Eutectic Mixtures: Difference between Empirical and Semi-Empirical Models. Sci. Rep. 2025, 15, 3862. [Google Scholar] [CrossRef] [PubMed]
- Alqarni, M.H.; Haq, N.; Alam, P.; Abdel-Kader, M.S.; Foudah, A.I.; Shakeel, F. Solubility Data, Hansen Solubility Parameters and Thermodynamic Behavior of Pterostilbene in Some Pure Solvents and Different (PEG-400 + Water) Cosolvent Compositions. J. Mol. Liq. 2021, 331, 115700. [Google Scholar] [CrossRef]
- Guo, J.; Liu, X.; Liu, M.; Han, M.; Liu, Y.; Ji, S. Effect of Molecular Weight of Poly(Ethylene Glycol) on Plasticization of Poly(l-Lactic Acid). Polymer 2021, 223, 123720. [Google Scholar] [CrossRef]
- Gonçalves, F.A.M.M.; Cruz, S.M.A.; Coelho, J.F.J.; Serra, A.C. The Impact of the Addition of Compatibilizers on Poly (lactic acid) (PLA) Properties after Extrusion Process. Polymers 2020, 12, 2688. [Google Scholar] [CrossRef]
- Xuan, W.; Hakkarainen, M.; Odelius, K. Levulinic Acid as a Versatile Building Block for Plasticizer Design. ACS Sustain. Chem. Eng. 2019, 7, 12552–12562. [Google Scholar] [CrossRef]
- Mascia, L.; Kouparitsas, Y.; Nocita, D.; Bao, X. Antiplasticization of Polymer Materials: Structural Aspects and Effects on Mechanical and Diffusion-Controlled Properties. Polymers 2020, 12, 769. [Google Scholar] [CrossRef]
- Ruiz, E.; Orozco, V.H.; Hoyos, L.M.; Giraldo, L.F. Study of Sonication Parameters on PLA Nanoparticles Preparation by Simple Emulsion-Evaporation Solvent Technique. Eur. Polym. J. 2022, 173, 111307. [Google Scholar] [CrossRef]
- Buoso, S.; Belletti, G.; Ragno, D.; Castelvetro, V.; Bertoldo, M. Rheological Response of Polylactic Acid Dispersions in Water with Xanthan Gum. ACS Omega 2022, 7, 12536–12548. [Google Scholar] [CrossRef]
- Keresztes, J.; Csóka, L. Characterisation of the Surface Free Energy of the Recycled Cellulose Layer that Comprises the Middle Component of Corrugated Paperboards. Coatings 2023, 13, 259. [Google Scholar] [CrossRef]
- Baklan, D.; Bilousova, A.; Wesolowski, M. UV Resistance and Wetting of PLA Webs Obtained by Solution Blow Spinning. Polymers 2024, 16, 2428. [Google Scholar] [CrossRef]
- Elsawy, M.A.; Kim, K.H.; Park, J.W.; Deep, A. Hydrolytic Degradation of Polylactic Acid (PLA) and Its Composites. Renew. Sustain. Energy Rev. 2017, 79, 1346–1352. [Google Scholar] [CrossRef]
- Lee, S.; Wee, J.-W. Effect of Temperature and Relative Humidity on Hydrolytic Degradation of Additively Manufactured PLA: Characterization and Artificial Neural Network Modeling. Polym. Degrad. Stab. 2024, 230, 111055. [Google Scholar] [CrossRef]
- Li, N.; Link, G.; Jelonnek, J. 3D Microwave Printing Temperature Control of Continuous Carbon Fiber Reinforced Composites. Compos. Sci. Technol. 2019, 187, 107939. [Google Scholar] [CrossRef]
- Asakura, T.; Ibe, Y.; Jono, T.; Naito, A. Structure and Dynamics of Biodegradable Polyurethane-Silk Fibroin Composite Materials in the Dry and Hydrated States Studied Using 13C Solid-State NMR Spectroscopy. Polym. Degrad. Stab. 2021, 190, 109645. [Google Scholar] [CrossRef]













| Title 1 | δd | δp | δh | R0 | Ra | RED | Ref. |
|---|---|---|---|---|---|---|---|
| Menthol–levulinic acid 1:1 (MenLev) | 17.15 | 5.17 | 10.96 | 6.7 | 0.79 | [31] | |
| Menthol–acetic acid 1:1 (MenAc) | 16.88 | 4.15 | 11.60 | 7.8 | 0.92 | [31] | |
| Menthol–lactic acid 1:1 (MenLac) | 16.7 | 2.5 | 8.8 | 7.8 | 0.92 | - | |
| Menthol–oleic acid 1:1 (MenOl) | 16.8 | 5.0 | 9.4 | 5.8 | 0.68 | - | |
| Epoxy oleic acid | 16.6 | 11.1 | 9.8 | 3.6 | 0.42 | [28] | |
| Epoxy linoleic acid | 16.6 | 11.4 | 10.5 | 4.4 | 0.51 | [28] | |
| PEG-400 | 14.6 | 7.5 | 9.4 | 5.4 | 0.64 | [32] | |
| PLA 4060D | 16.5 | 9.9 | 6.4 | 8.5 | [28] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Baklan, D.; Vorobyova, V.; Sevastyanova, O.; Karavayev, T.; Myronyuk, O. Sustainable Waterborne Polylactide Coatings Enabled by Hydrophobic Deep Eutectic Solvents Plasticization. Polymers 2026, 18, 154. https://doi.org/10.3390/polym18020154
Baklan D, Vorobyova V, Sevastyanova O, Karavayev T, Myronyuk O. Sustainable Waterborne Polylactide Coatings Enabled by Hydrophobic Deep Eutectic Solvents Plasticization. Polymers. 2026; 18(2):154. https://doi.org/10.3390/polym18020154
Chicago/Turabian StyleBaklan, Denys, Victoria Vorobyova, Olena Sevastyanova, Taras Karavayev, and Oleksiy Myronyuk. 2026. "Sustainable Waterborne Polylactide Coatings Enabled by Hydrophobic Deep Eutectic Solvents Plasticization" Polymers 18, no. 2: 154. https://doi.org/10.3390/polym18020154
APA StyleBaklan, D., Vorobyova, V., Sevastyanova, O., Karavayev, T., & Myronyuk, O. (2026). Sustainable Waterborne Polylactide Coatings Enabled by Hydrophobic Deep Eutectic Solvents Plasticization. Polymers, 18(2), 154. https://doi.org/10.3390/polym18020154







