Investigations on Thermal Transitions in PDPP4T/PCPDTBT/AuNPs Composite Films Using Variable Temperature Ellipsometry
Abstract
1. Introduction
2. Experimental
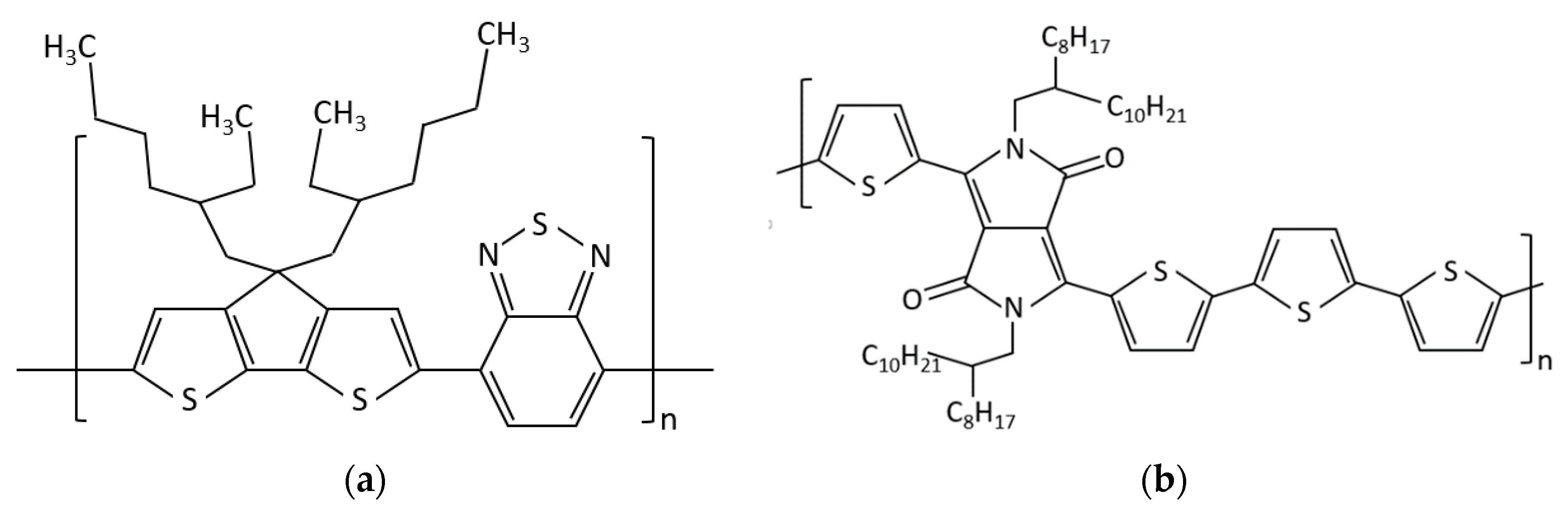
2.1. Materials
2.2. Samples Preparation
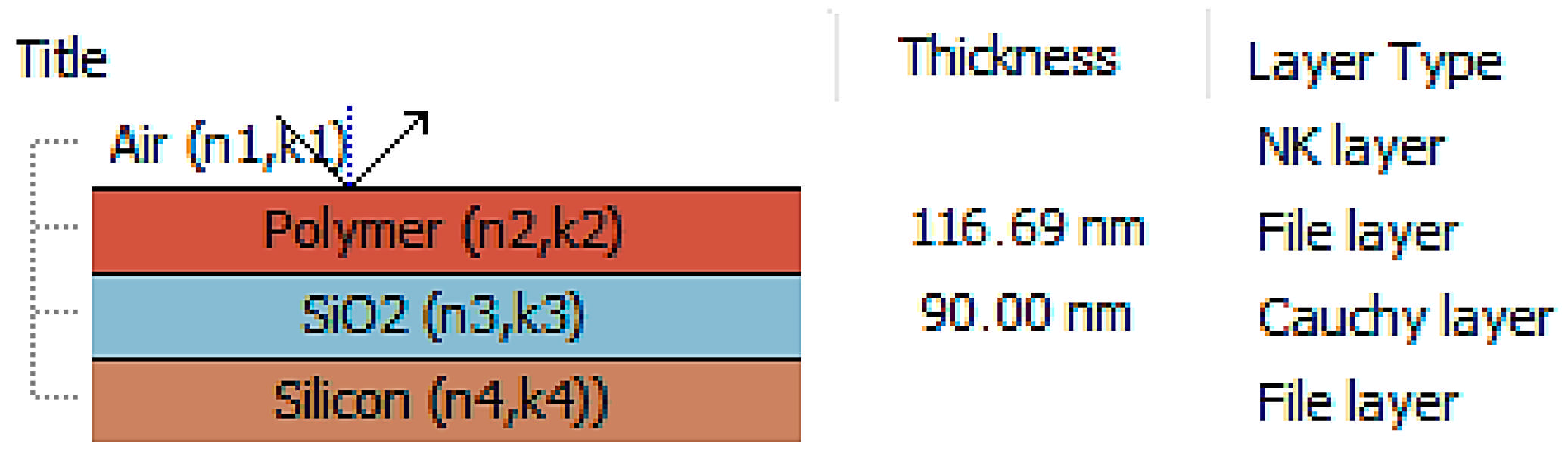
2.3. Methods
3. Results and Discussion
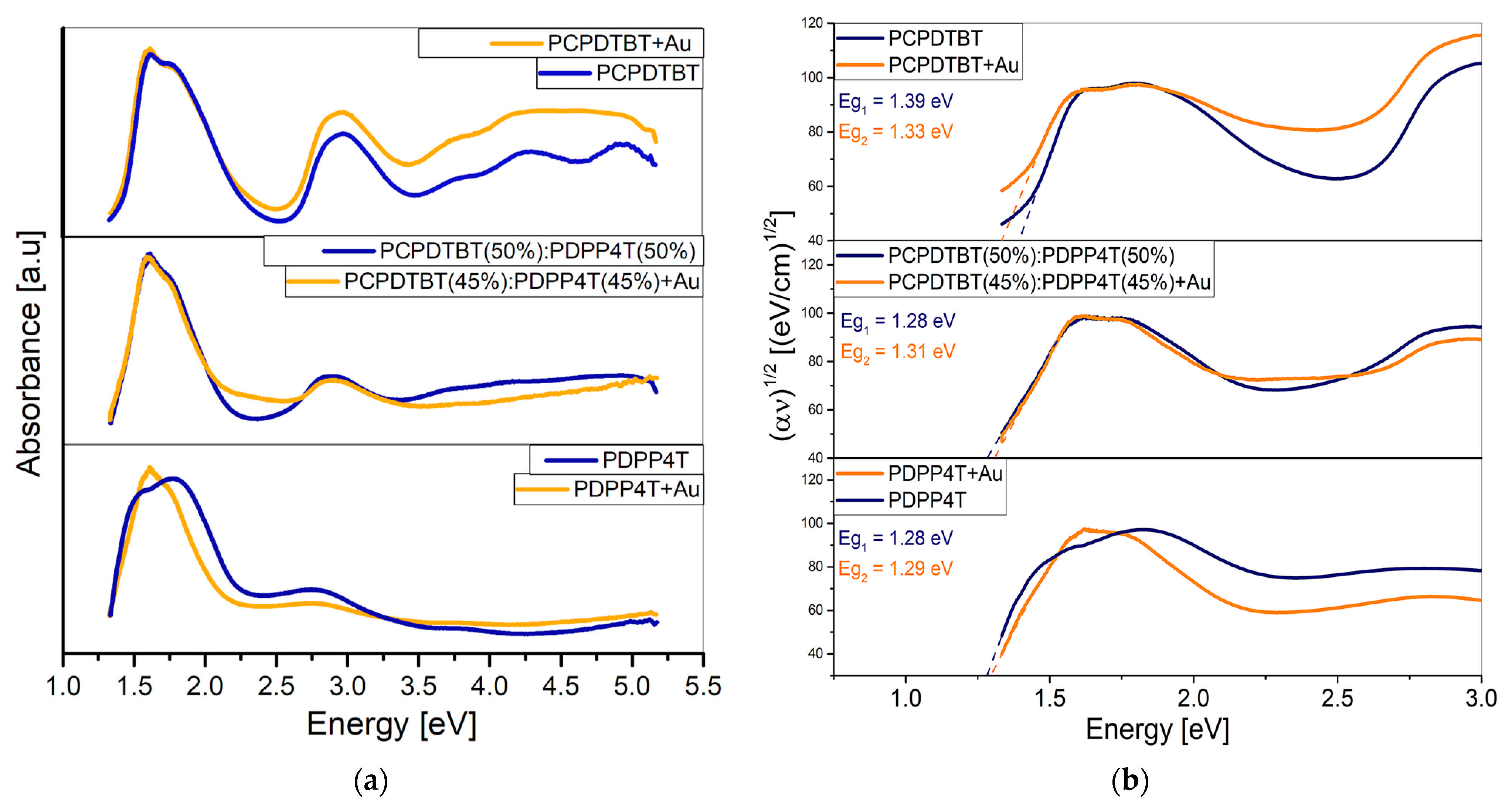
3.1. Optical Analysis
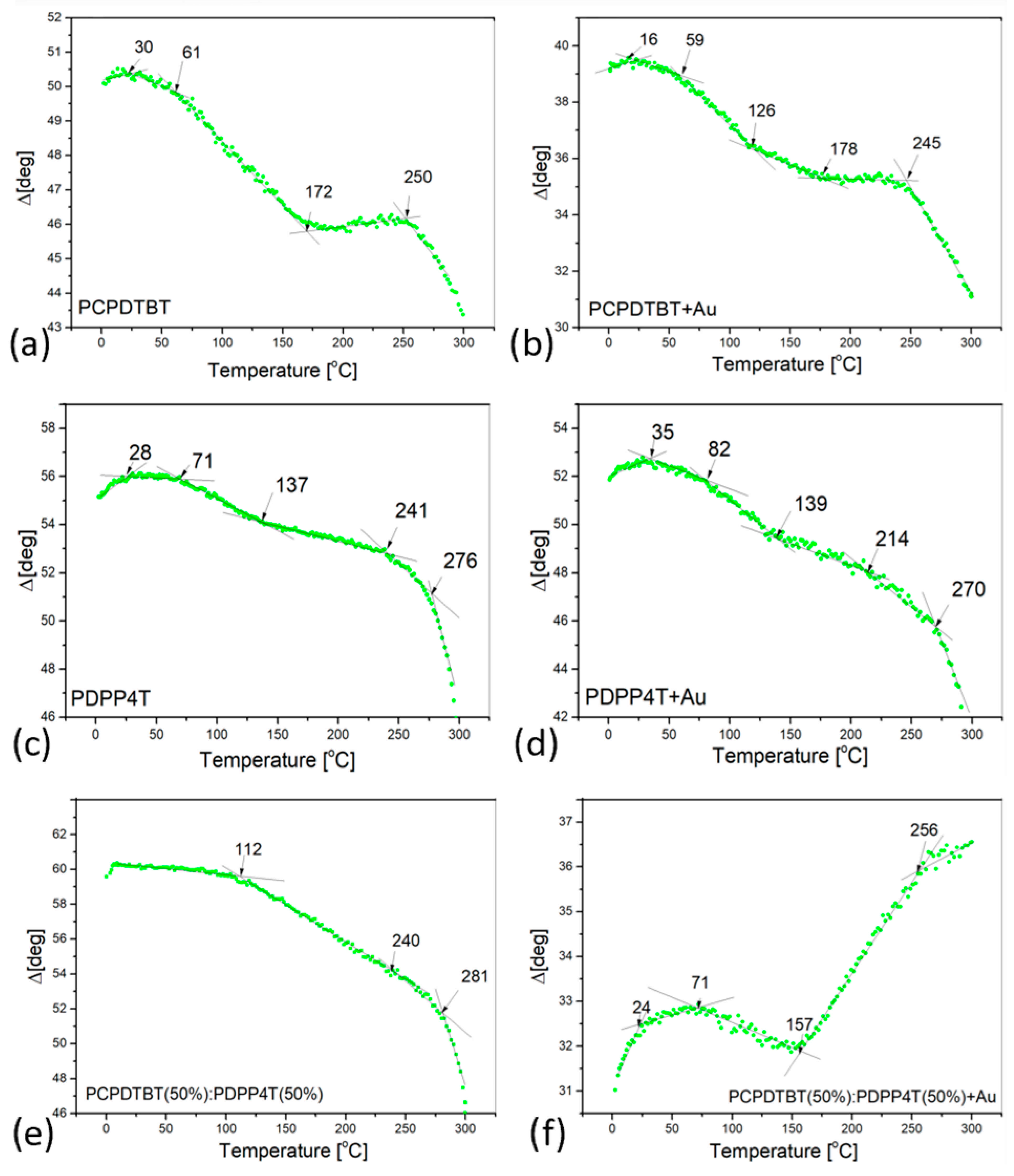
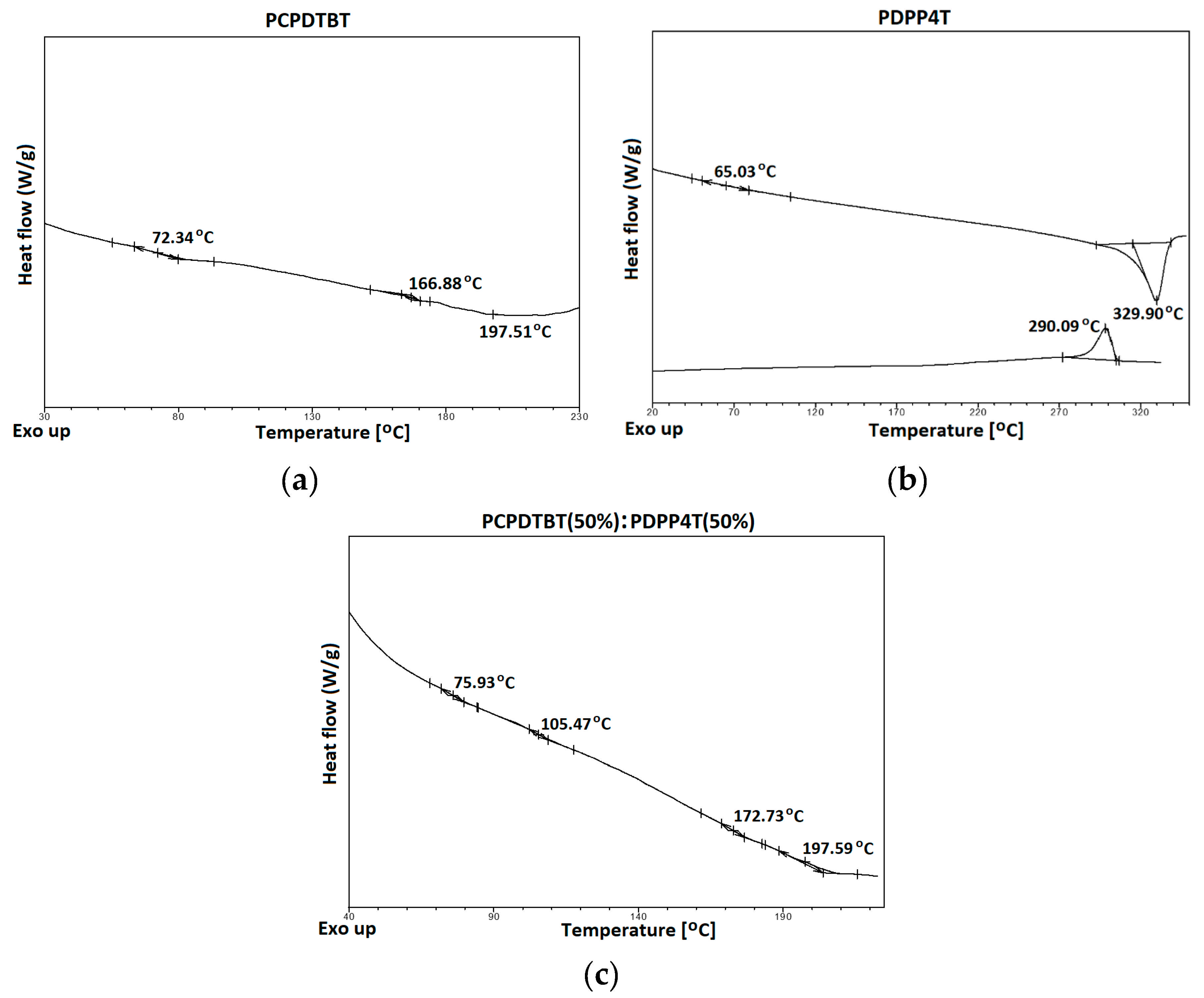
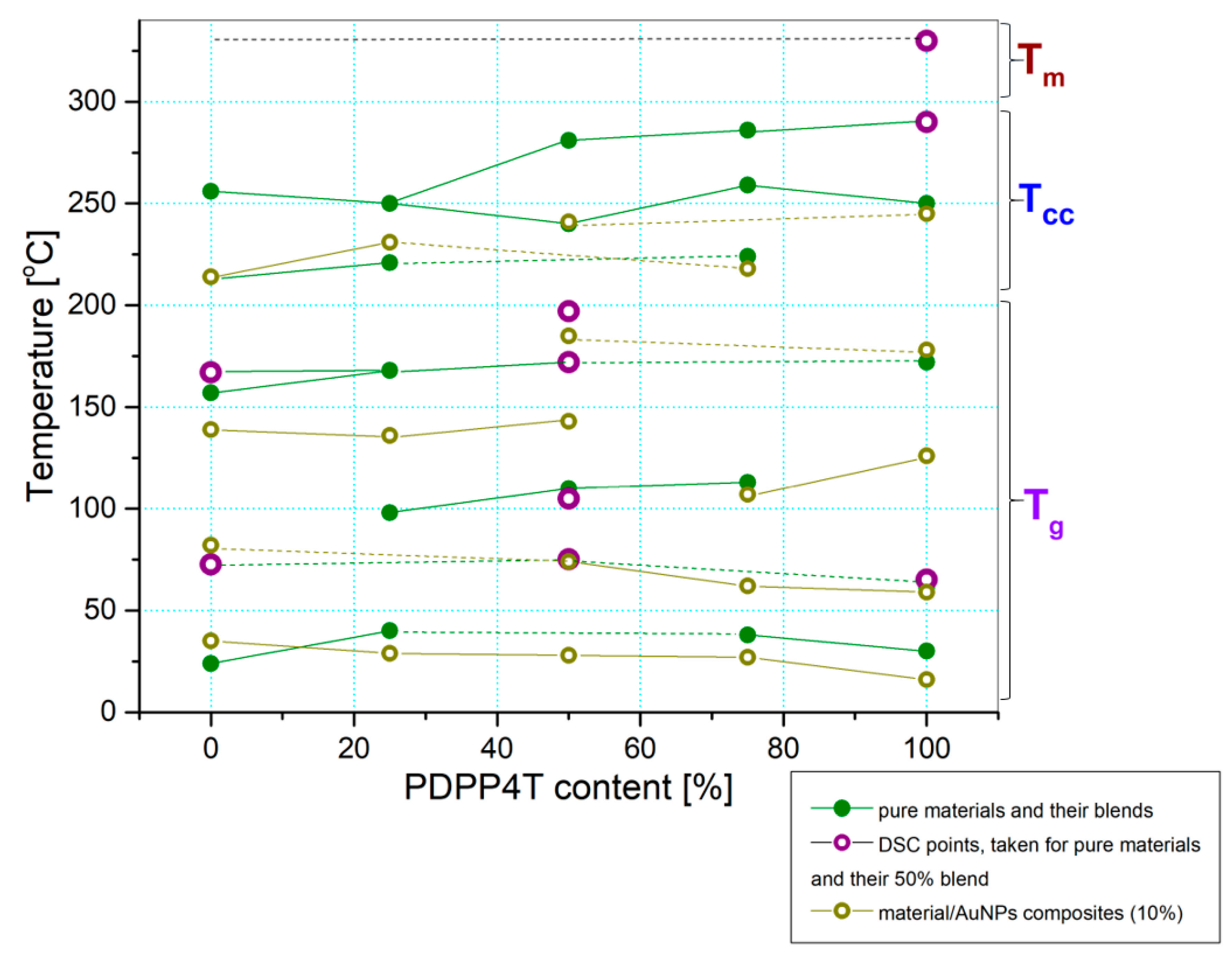
3.2. Thermal Analysis
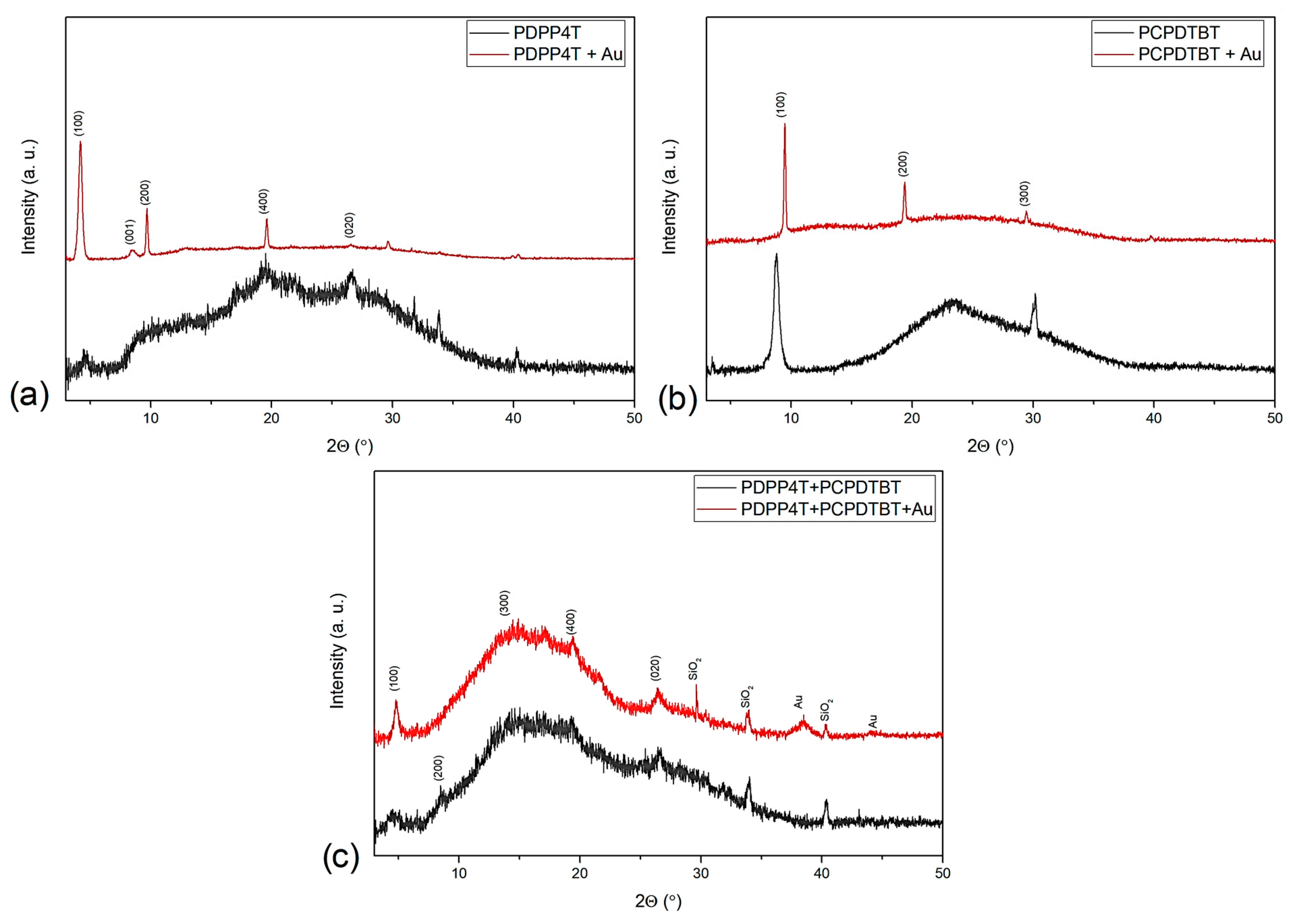
3.3. XRD Analysis
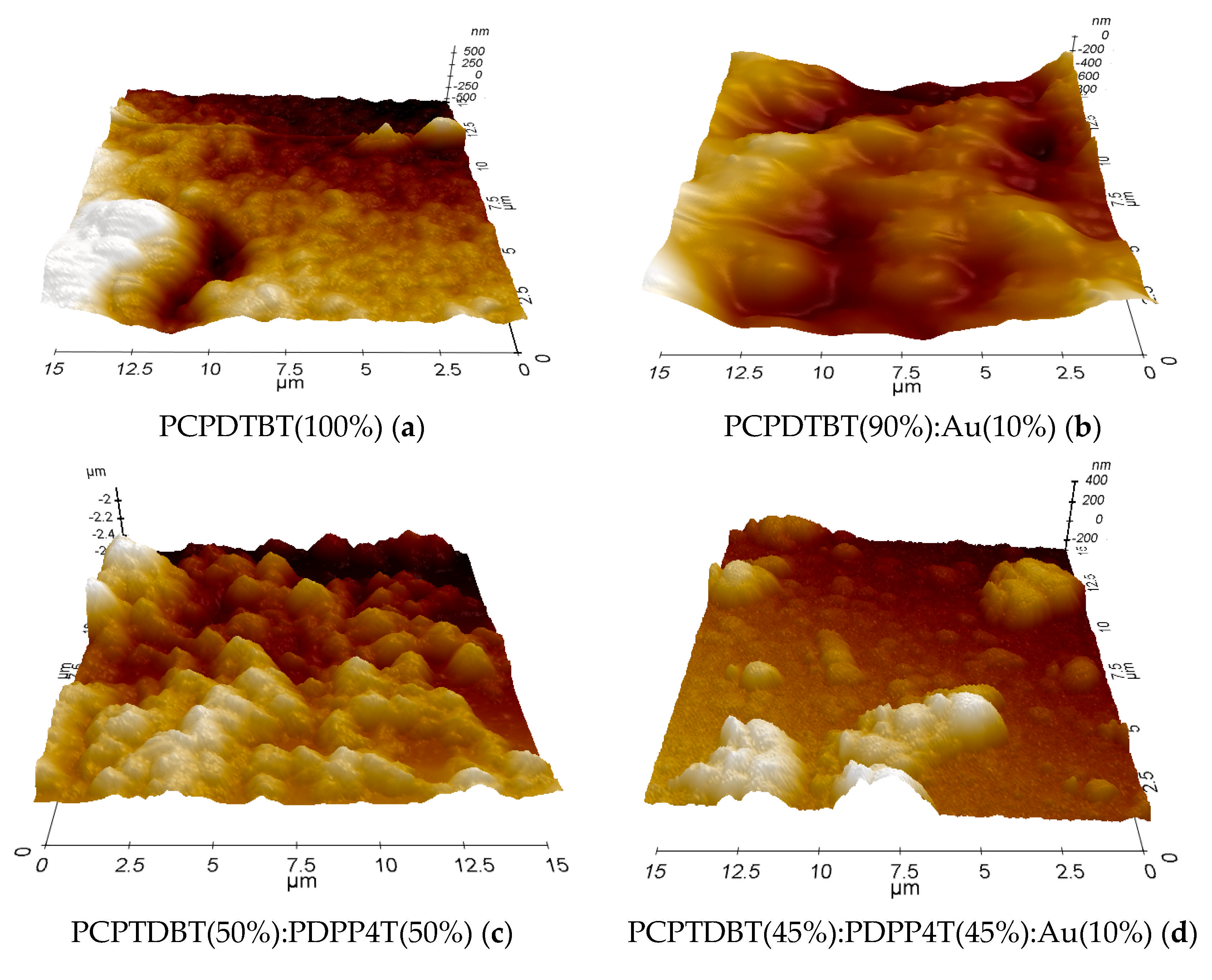
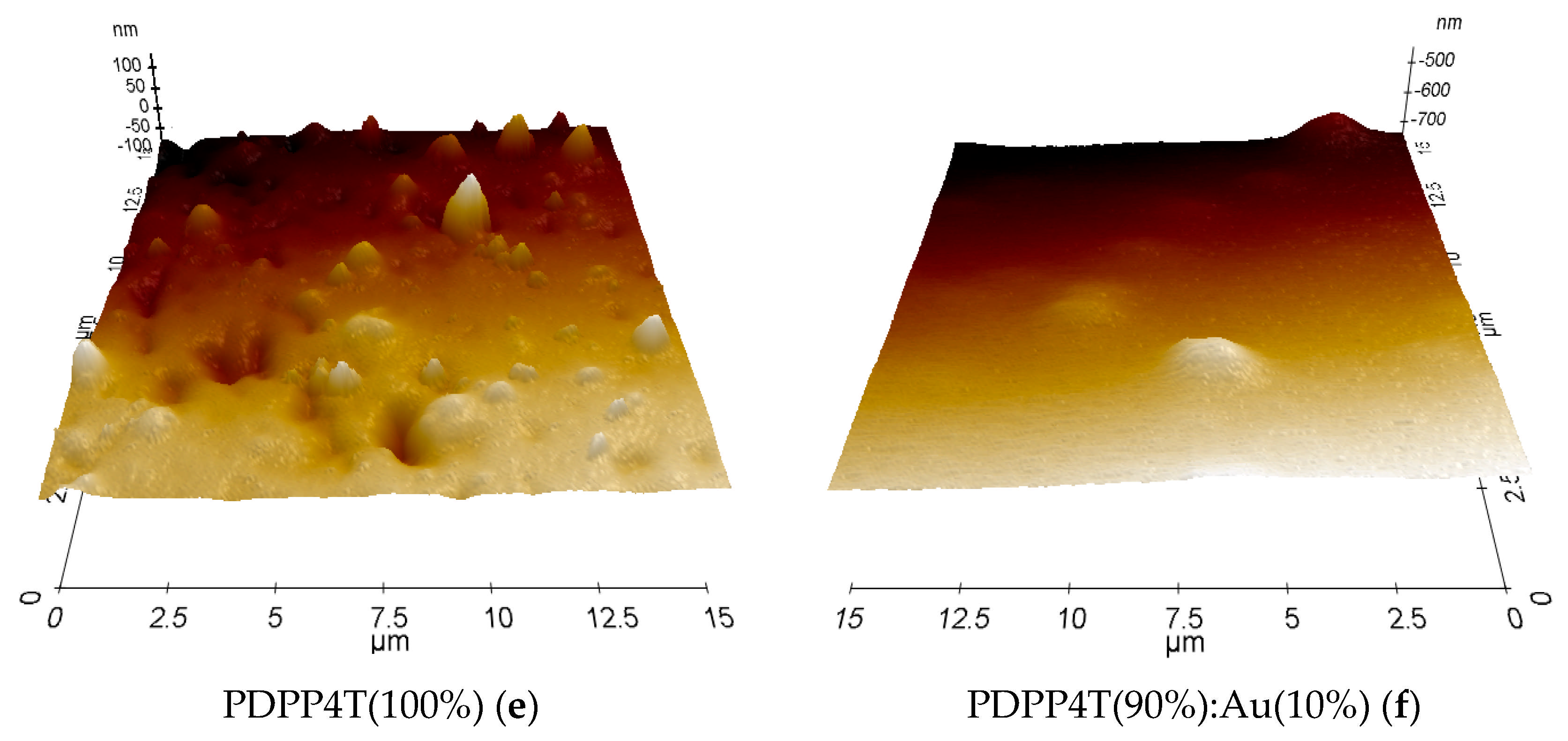
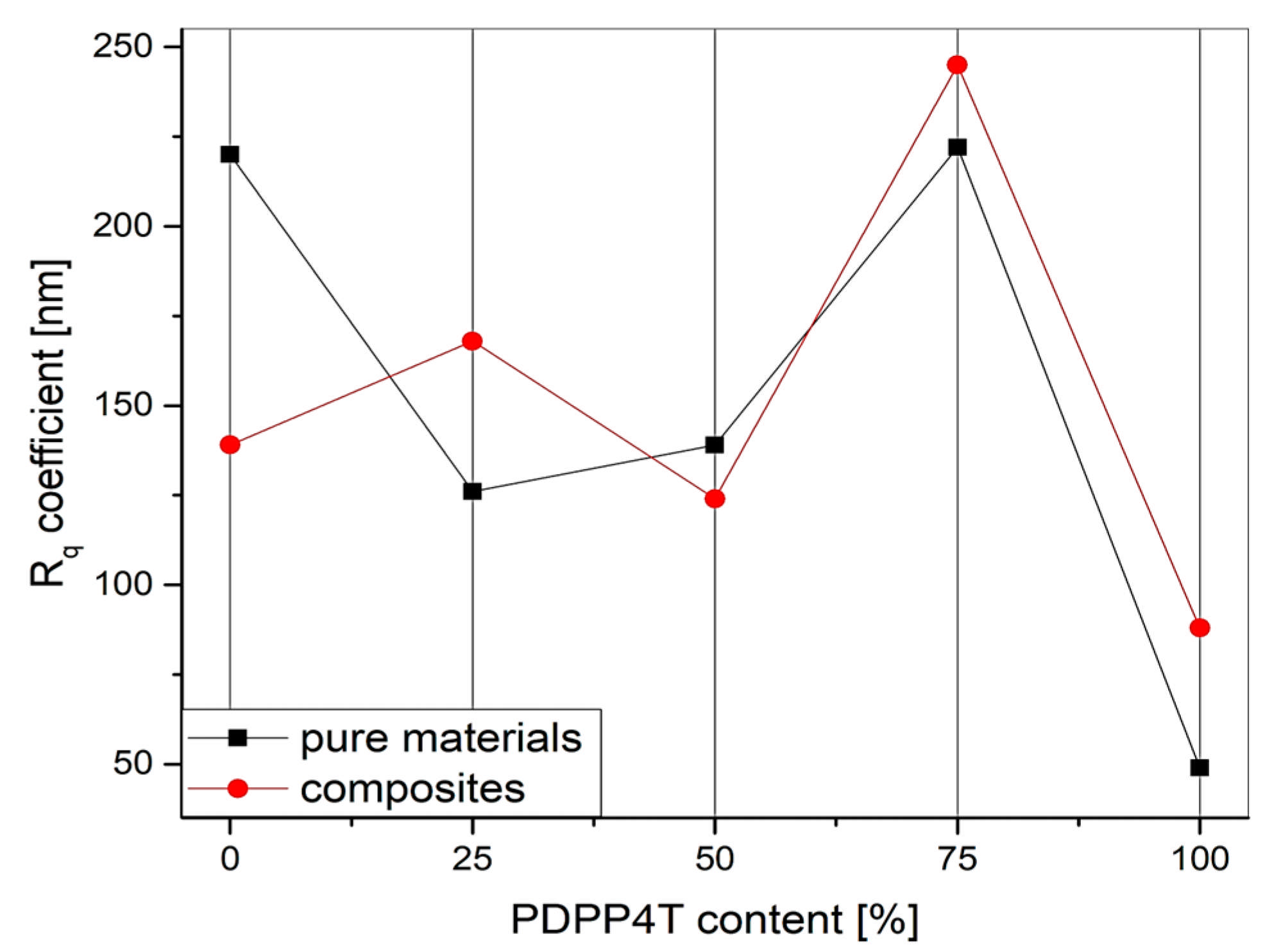
3.4. Microscopic Analysis
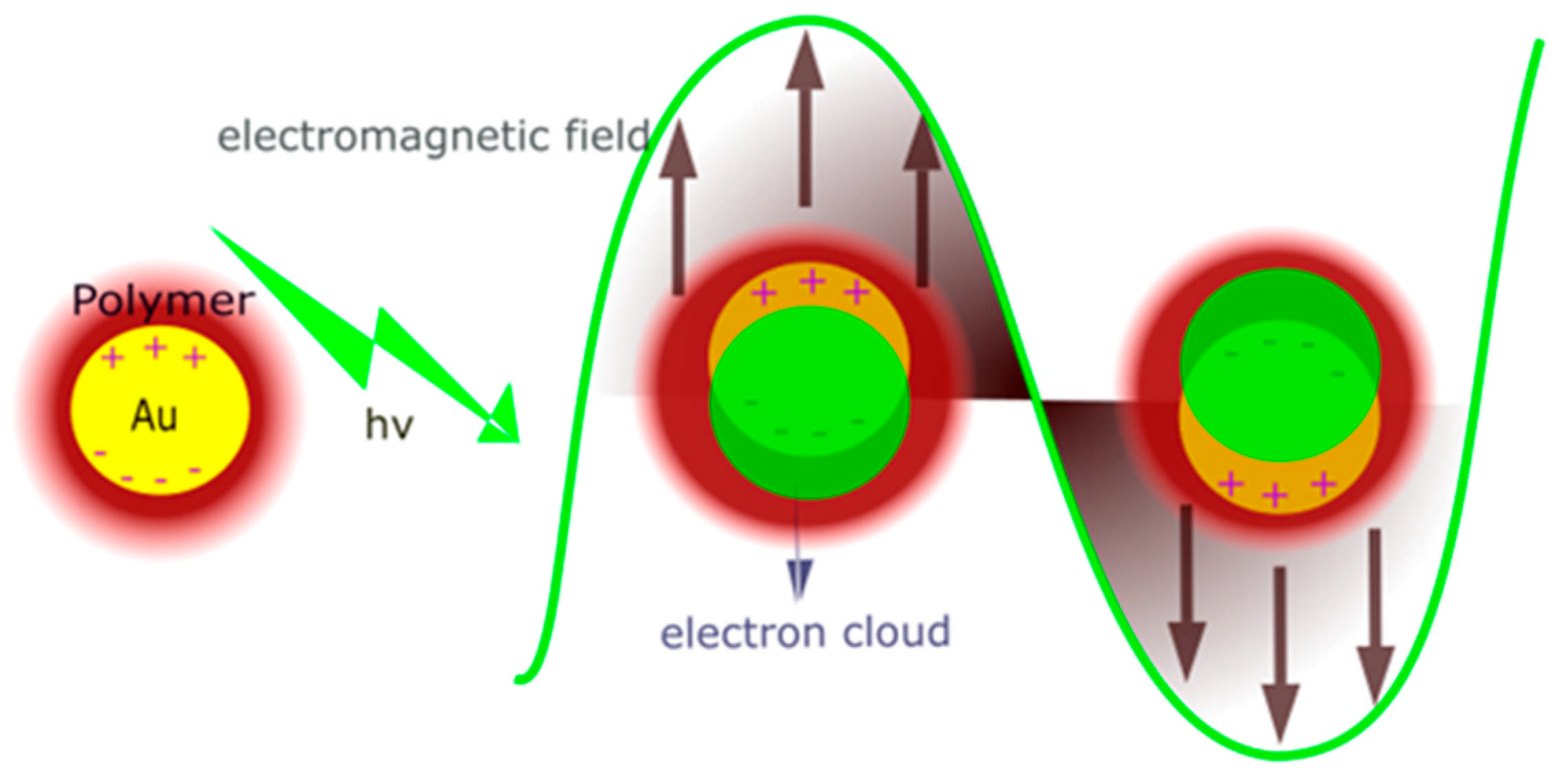
3.5. General Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Sulaiman, A.K.; Yap, C.C.; Touati, F. Binary blend based dye sensitized photo sensor using PCPDTBT and MEH-PPV composite as a light sensitizer. Synth. Met. 2015, 210, 392–397. [Google Scholar] [CrossRef]
- Li, Y.; Wei, Y.; Feng, K.; Hao, Y.; Pei, J.; Zhang, Y.; Sun, B. Introduction of PCPDTBT in P3HT:Spiro-OMeTAD blending system for solid-state hybrid solar cells with dendritic TiO2/Sb2S3 nanorods composite film. J. Sol. St. Chem. 2019, 276, 278–284. [Google Scholar] [CrossRef]
- Banerjee, S.; Singh, P.; Purkayastha, P.; Ghosh, S.K. Evolution of Organic Light Emitting Diode (OLED) Materials and their Impact on Display Technology. Chem. Asian J. 2024, 20, e202401291. [Google Scholar] [CrossRef] [PubMed]
- Nayak, D.; Choudhary, R.B. A survey of the structure, fabrication, and characterization of advanced organic light emitting diodes. Microelectron. Reliab. 2023, 144, 114959. [Google Scholar] [CrossRef]
- Luo, L.; Liu, Z. Recent progress in organic field-effect transistor-based chem/bio-sensors. View 2022, 3, 20200115. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, W.; He, Z.; Peng, B.; Di, C.-A.; Li, H. High-performance and multifunctional organic field-effect transistors. Chin. Chem. Lett. 2023, 34, 108094. [Google Scholar] [CrossRef]
- Kim, O.; Kwon, J.; Kim, S.; Xu, B.; Seo, K.; Park, C.; Do, W.; Bae, J.; Kang, S. Efect of PVP-capped ZnO nanoparticles with enhanced charge transport on the performance of P3HT/PCBM polymer solar cells. Polymers 2019, 11, 1818. [Google Scholar] [CrossRef]
- Liu, C.; Zhang, D.; Li, Z.; Zhang, X.; Guo, W.; Zhang, L.; Ruan, S.; Long, Y. Decreased charge transport barrier and recombination of organic solar cells by constructing interfacial nanojunction with annealing-free ZnO and Al flms. ACS Appl. Mater. Interfaces 2017, 9, 22068–22075. [Google Scholar] [CrossRef]
- Mohtaram, F.; Borhani, S.; Ahmadpour, M.; Fojan, P.; Behjat, A.; Rubahn, H.G.; Madsen, M. Electrospun ZnO nanofber interlayers for enhanced performance of organic photovoltaic devices. Sol. Energy 2020, 197, 311–316. [Google Scholar] [CrossRef]
- Swart, H.C.; Ntwaeaborwa, O.M.; Mbule, P.S.; Dhlamini, M.S.; Mothudi, B.B. P3HT: PCBM based solar cells: A short review focusing on ZnO nanoparticles bufer layer, post-fabrication annealing and an inverted geometry. J. Mater. Sci. Eng. B 2015, 5, 12–35. [Google Scholar] [CrossRef][Green Version]
- Wanninayake, A.P.; Church, B.C.; Abu-Zahra, N. Efect of ZnO nanoparticles on the power conversion efciency of organic photovoltaic devices synthesized with CuO nanoparticles. AIMS Mater. Sci. 2016, 3, 927–937. [Google Scholar] [CrossRef]
- Dong, J.; Yan, C.; Chen, Y.; Zhou, W.; Peng, Y.; Zhang, Y.; Wang, L.-N.; Huang, Z.-H. Organic semiconductor nanostructures: Optoelectronic properties, modification strategies, and photocatalytic applications. J. Mater. Sci. Technol. 2022, 113, 175–198. [Google Scholar] [CrossRef]
- Cheng, Z.; Javed, N.; O’Carroll, D.M. Optical and Electrical Properties of Organic Semiconductor Thin Films on Aperiodic Plasmonic Metasurfaces. ACS Appl. Mater. Interfaces 2020, 12, 35579–35587. [Google Scholar] [CrossRef] [PubMed]
- Balakrishnan, T.S.; Sultan, M.T.H.; Shahar, F.S.; Basri, A.A.; Shah, A.U.M.; Sebaey, T.A.; Łukaszewicz, A.; Józwik, J.; Grzejda, R. Fatigue and impact properties of kenaf/glass-reinforced hybrid pultruded composites for structural applications. Materials 2024, 17, 302. [Google Scholar] [CrossRef]
- Liu, S.; Yang, J.; Yu, Y.; Liang, D.; Li, Y.; Si, X.; Song, S.; Meng, M.; Zhang, J.; Zhang, Y. Multifunctional flexible carbon fiber felt@nickel composite films with core–shell heterostructure: Exceptional Joule heating capability, thermal management, and electromagnetic interference shielding. Chem. Eng. J. 2024, 494, 153221. [Google Scholar] [CrossRef]
- Huang, Y.; Sultan, M.T.H.; Shahar, F.S.; Grzejda, R.; Łukaszewicz, A. Hybrid fiber-reinforced biocomposites for marine applications: A review. J. Compos. Sci. 2024, 8, 430. [Google Scholar] [CrossRef]
- Shahar, F.S.; Sultan, M.T.H.; Grzejda, R.; Łukaszewicz, A.; Oksiuta, Z.; Krishnamoorthy, R.R. Harnessing the potential of natural composites in biomedical 3D printing. Materials 2024, 17, 6045. [Google Scholar] [CrossRef]
- Ibrahim, N.I.; Sultan, M.T.H.; Łukaszewicz, A.; Shah, A.U.M.; Shahar, F.S.; Józwik, J.; Najeeb, M.I.; Grzejda, R. Characterization and isolation method of Gigantochloa scortechinii (Buluh Semantan) cellulose nanocrystals. Int. J. Biol. Macromol. 2024, 272, 132847. [Google Scholar] [CrossRef]
- Wang, T.C.; Su, Y.H.; Hung, Y.K.; Yeh, C.S.; Huang, L.W.; Gomulya, W.; Lai, L.H.; Loi, M.A.; Yang, J.S.; Wu, J.J. Charge collection enhancement by incorporation of gold–silica core–shell nanoparticles into P3HT:PCBM/ZnO nanorod array hybrid solar cells. Phys. Chem. Chem. Phys. 2015, 17, 19854–19861. [Google Scholar] [CrossRef]
- Gao, H.; Meng, J.; Sun, J.; Deng, J. Enhanced performance of polymer solar cells based on P3HT:PCBM via incorporating Au nanoparticles prepared by the micellar method. J. Mater. Sci. Mat. Electr. 2020, 31, 10760–10767. [Google Scholar] [CrossRef]
- Pillai, S.; Catchpole, K.R.; Trupke, T.; Green, M.A. Surface plasmon enhanced silicon solar cells. J. Appl. Phys. 2007, 101, 093105. [Google Scholar] [CrossRef]
- Sharifi, M.; Attar, F.; Saboury, A.A.; Akhtari, K.; Hooshmand, N.; Hasan, A.; El-Sayed, M.A.; Falahati, M. Plasmonic gold nanoparticles: Optical manipulation, imaging, drug delivery and therapy. J. Control. Release 2019, 311–312, 170–189. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Shankar, S.; Philips, D.S.; Ajayaghosh, A. Diketopyrrolopyrrole-based functional supramolecular polymers: Next-generation materials for optoelectronic applications. Mater. Today Chem. 2020, 16, 100242. [Google Scholar] [CrossRef]
- Kotowicz, S.; Małecki, J.G.; Pająk, A.K.; Siwy, M.; Łuszczyńska, B.; Selerowicz, A. Optical, electrochemical and thermal investigations with DFT calculations of new 1,8-naphthal-phthalimide compounds. J. Mol. Struct. 2025, 1332, 141711. [Google Scholar] [CrossRef]
- Yi, Z.; Sun, X.; Zhao, Y.; Guo, Y.; Chen, X.; Qin, J.; Yu, G.; Liu, Y. Diketopyrrolopyrrole-Based π Conjugated Copolymer Containing β Unsubstituted Quintetthiophene Unit: A Promising Material Exhibiting High Hole-Mobility for Organic Thin-Film Transistors. Chem. Mater. 2012, 24, 4350–4356. [Google Scholar] [CrossRef]
- Hajduk, B.; Jarka, P.; Bednarski, H.; Janeczek, H.; Kumari, P.; Farcas, A. Thermal Transitions and Structural Characteristics of Poly(3,4-ethylenedioxythiophene/cucurbit[7]uril) Polypseudorotaxane and Polyrotaxane Thin Films. Materials 2024, 17, 1318. [Google Scholar] [CrossRef]
- Hajduk, B.; Bednarski, H.; Jarząbek, B.; Nitschke, P.; Janeczek, H. Phase diagram of P3HT:PC70BM thin films based on variable-temperature spectroscopic ellipsometry. Polym. Test. 2020, 84, 106383. [Google Scholar] [CrossRef]
- Wang, T.; Pearson, A.J.; Dunbar, A.D.F.; Staniec, P.A.; Watters, D.C.; Coles, D.; Yi, H.; Iraqi, A.; Lidzey, D.G.; Jones, R.A.L. Competition between substrate-mediated π-π stacking and surface-mediated Tg depression in ultrathin conjugated polymer films. Eur. Phys. J. E 2012, 35, 129. [Google Scholar] [CrossRef]
- Kim, J.H.; Jang, J.; Zin, W.-C. Estimation of the thickness dependence of the glass transition temperature in various thin polymer films. Langmuir 2000, 16, 4064–4067. [Google Scholar] [CrossRef]
- Keddie, J.L.; Jones, R.A.; Cory, R.A. Size-dependent depression of the glass transition temperature in polymer films. Europhys. Eur. Lett. 1994, 27, 59–64. [Google Scholar] [CrossRef]
- Pratyusha, T.; Sivakumar, G.; Yella, A.; Gupta, D. Novel Ternary Blend of PCDTBT, PCPDTBT and PC70BM for the Fabrication of Bulk Heterojunction Organic Solar Cells. Mater. Today Proc. 2017, 4, 5067–5073. [Google Scholar] [CrossRef]
- Fischer, F.S.U.; Trefz, D.; Back, J.; Kayunkid, N.; Tornow, B.; Albrecht, S.; Yager, K.G.; Singh, G.; Karim, A.; Neher, D.; et al. Ludwigs, Highly Crystalline Films of PCPDTBT with Branched Side Chains by Solvent Vapor Crystallization: Influence on Opto-Electronic Properties. Adv. Mater. 2015, 27, 1223–1228. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, Z.; Touati, F.; Muhammad, F.F.; Najeeb, M.A.; Shakoor, R.A. Shakoor Effect of ambient temperature on the efficiency of the PCPDTBT:PC71BM BHJ solar cells. Appl. Phys. A 2017, 123, 486. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, F.; Wang, H.W.; Nordlund, D.; Sun, Z.; Ferdous, S.; Russell, T.P. Sequential Deposition: Optimization of Solvent Swelling for High-Performance Polymer Solar Cells. ACS Appl. Mater. Interfaces 2015, 7, 653–661. [Google Scholar] [CrossRef] [PubMed]
- Bijleveld, J.C.; Verstrijden, R.A.M.; Wienka, M.M.; Janssen, R.A.J. Copolymers of diketopyrrolopyrrole and thienothiophene for photovoltaic cells. J. Mater. Chem. 2011, 21, 9224–9231. [Google Scholar] [CrossRef]
- Notarianni, M.; Vernon, K.; Chou, A.; Aljada, M.; Liu, J.; Motta, N. Plasmonic effect of gold nanoparticles in organic solar cells. Sol. Energy 2014, 106, 23–37. [Google Scholar] [CrossRef]
- Vienna, H.A.; Klimpovuz, C.R.; Turchetti, D.A.; Rossi, A.L.; Oliveira, M.M.; Marletta, A.; Akcelrud, L.C. Polyfluorene-based conjugated nanocomposites with in-situ gold nanoparticles: Synthesis via rational chemical passivation and characterization of supramolecular fibrillar structures. J. Mol. Struct. 2025, 1322, 140431. [Google Scholar] [CrossRef]
- Li, Z.; Liu, C.; Guo, J.; Zhou, Y.; Shen, L.; Guo, W. Passivation effect of composite organic interlayer on polymer solar cells. Org. Electron. 2018, 63, 129–136. [Google Scholar] [CrossRef]
- Guo, C.; Hall, G.N.; Addison, J.B.; Yarger, J.L. Gold nanoparticle-doped silk film asbiocompatible, SERS substrate. RSC Adv. 2015, 5, 1937. [Google Scholar] [CrossRef]
- Long, N.V.; Ohtaki, M.; Yuasa, M.; Yoshida, S.; Kuragaki, T.; Thi, C.M.; Nogami, M. Synthesis and Self-Assembly of Gold Nanoparticles by Chemically Modified Polyol, Methods under Experimental Control. J. Nanomater. 2013, 2013, 93125. [Google Scholar] [CrossRef]
- Hajduk, B.; Jarka, P.; Tański, T.; Bednarski, H.; Janeczek, H.; Gnida, P.; Fijalkowski, M. An Investigation of the Thermal Transitions and Physical Properties of Semiconducting PDPP4T:PDBPyBT Blend Films. Materials 2022, 15, 8392. [Google Scholar] [CrossRef] [PubMed]
- Elashmawi, I.S.; Abdelghany, A.M.; Meikhail, M.S.; Aldhabi, A.A. Effect of gold and silver mixed nanofillers and gamma irradiation on the structure and physical properties of PVA/PVP nanocomposite. Radiat. Phys. Chem. 2025, 230, 112558. [Google Scholar] [CrossRef]
- Masirek, R.; Szkudlarek, E.; Piorkowska, E.; Slouf, M.; Kratochvil, J.; Baldrian, J. Nucleation of isotactic polypropylene crystallization by gold nanoparticles. J. Polym. Sci. B Polym. Phys. 2010, 48, 469–478. [Google Scholar] [CrossRef]
- Borgesi, A.; Tallarida, G.; Amore, G.; Cazzaniga, F.; Queirolo, F.; Alessandri, M.; Sassela, A. Influence of roughness and grain dimension on the optical functions of polycrystalline silicon films. Thin Solid Film. 1998, 313–314, 243–247. [Google Scholar] [CrossRef]
- Spangler, L.L.; Torkelson, J.M.; Royal, J.S. Influence of solvent and molecular weight on thickness and surface topography of spin-coated polymer films. Polym. Eng. Sci. 1990, 30, 644–653. [Google Scholar] [CrossRef]
- Fronk, S.L.; Wang, M.; Ford, M.; Coughlin, J.; Mai, C.-K. Effect of chiral 2-ethylhexyl side chains on chiroptical properties of the narrow bandgap conjugated polymers PCPDTBT and PCDTPT. Chem. Sci. 2016, 7, 5313–5321. [Google Scholar] [CrossRef]
- Wang, Y.; An, T.; Xue, J. Realizing high detectivity organic photodetectors in visible wavelength by doping highly ordered polymer PCPDTBT. Org. Electron. 2020, 82, 105700. [Google Scholar] [CrossRef]
- Wang, L.; Hu, M.; Zhang, Y.; Yuan, Z.; Hu, Y.; Zhao, X.; Chen, Y. High molecular weight polymeric acceptors based on semi-perfluoroalkylated perylene diimides for pseudo-planar heterojunction all-polymer organic solar cells. Polymer 2022, 255, 125114. [Google Scholar] [CrossRef]
- Nathanael, A.J.; Mangalaraj, D.; Ponpandian, N. Controlled growth and investigations on the morphology and mechanical properties of hydroxyapatite/titania nanocomposite thin films. Compos. Sci. Technol. 2010, 70, 1645–1651. [Google Scholar] [CrossRef]
- Predoi, D.; Talu, S.; Ciobanu, S.C.; Iconaru, S.L.; Matos, R.S.; da Fonseca Filho, H.D. Exploring the physicochemical traits, antifungal capabilities, and 3D spatial complexity of hydroxyapatite with Ag+–Mg2+ substitution in the biocomposite thin films. Micron 2024, 184, 103661. [Google Scholar] [CrossRef]
- Ha, J.S.; Kim, K.H.; Choi, D.H. 5-Bis(2-octyldodecyl)pyrrolo[3,4-c]pyrrole-1,4-(2H,5H)-dione-Based DonorAcceptor Alternating Copolymer Bearing 5,5′-Di (thiophen-2-yl)-2,20 -biselenophene Exhibiting 1.5 cm2V-1s-1Hole Mobility in Thin-Film Transistors. J. Am. Chem. Soc. 2011, 133, 10364–10367. [Google Scholar] [CrossRef] [PubMed]
- Hamad-Schifferli, K. Applications of Gold Nanoparticles in Plasmonic and Nanophotonic Biosensing. Adv. Biochem. Eng. Biotechnol. 2024, 187, 185–221. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Chikkaraddy, R.; Readman, C.; Hu, S.; Xiong, K.; Peng, J.; Lin, Q.; Baumberg, J.J. Metal to insulator transition for conducting polymers in plasmonic nanogaps. Light Sci. Appl. 2024, 13, 3. [Google Scholar] [CrossRef] [PubMed]
| Sample No | 1 | 2 | 3 | 4 | 5 |
|---|---|---|---|---|---|
| PCPDTBT [%] | 100 | 75 | 50 | 25 | 0 |
| PDPP4T [%] | 0 | 25 | 50 | 75 | 100 |
| Sample No | 6 | 7 | 8 | 9 | 10 |
|---|---|---|---|---|---|
| PCPDTBT [%] | 90 | 70 | 45 | 20 | 0 |
| PDPP4T [%] | 0 | 20 | 45 | 70 | 90 |
| Au [%] | 10 | 10 | 10 | 10 | 10 |
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | ||
|---|---|---|---|---|---|---|---|---|---|---|---|
| d [nm] | SiO2 | 111 | 113 | 114 | 93 | 110 | 90 | 100 | 116 | 111 | 97 |
| glass | 416 | 511 | 480 | 473 | 490 | 503 | 489 | 470 | 501 | 490 |
| PCPDTBT [%] | PDPP4T [%] | Eg [eV] | Eg [eV] (+Au) |
|---|---|---|---|
| 100 | 0 | 1.39 | 1.33 |
| 75 | 25 | 1.32 | 1.38 |
| 50 | 50 | 1.28 | 1.31 |
| 25 | 75 | 1.22 | 1.29 |
| 0 | 100 | 1.28 | 1.29 |
| Sample | Crystal Size (nm) |
|---|---|
| PCPDTBT | 16.1 |
| +Au | 47.3 |
| PDPP4T | 18.0 |
| +Au | 31.6 |
| BLEND | 21.5 |
| +Au | 60.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jarka, P.; Hajduk, B.; Kumari, P.; Janeczek, H.; Godzierz, M.; Tsekpo, Y.M.; Tański, T. Investigations on Thermal Transitions in PDPP4T/PCPDTBT/AuNPs Composite Films Using Variable Temperature Ellipsometry. Polymers 2025, 17, 704. https://doi.org/10.3390/polym17050704
Jarka P, Hajduk B, Kumari P, Janeczek H, Godzierz M, Tsekpo YM, Tański T. Investigations on Thermal Transitions in PDPP4T/PCPDTBT/AuNPs Composite Films Using Variable Temperature Ellipsometry. Polymers. 2025; 17(5):704. https://doi.org/10.3390/polym17050704
Chicago/Turabian StyleJarka, Paweł, Barbara Hajduk, Pallavi Kumari, Henryk Janeczek, Marcin Godzierz, Yao Mawuena Tsekpo, and Tomasz Tański. 2025. "Investigations on Thermal Transitions in PDPP4T/PCPDTBT/AuNPs Composite Films Using Variable Temperature Ellipsometry" Polymers 17, no. 5: 704. https://doi.org/10.3390/polym17050704
APA StyleJarka, P., Hajduk, B., Kumari, P., Janeczek, H., Godzierz, M., Tsekpo, Y. M., & Tański, T. (2025). Investigations on Thermal Transitions in PDPP4T/PCPDTBT/AuNPs Composite Films Using Variable Temperature Ellipsometry. Polymers, 17(5), 704. https://doi.org/10.3390/polym17050704










