Can Pyrolysis Oil Be Used as a Feedstock to Close the Gap in the Circular Economy of Polyolefins?
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- US Environmental Protection Agency. Advancing Sustainable Materials Management: 2014 Fact Sheet; United States Environmental Protection Agency, Office of Land and Emergency Management: Washington, DC, USA, 2016; p. 22. [Google Scholar]
- Hopewell, J.; Dvorak, R.; Kosior, E. Plastics Recycling: Challenges and Opportunities. Philos. Trans. R. Soc. Biol. Sci. 2009, 364, 2115–2126. [Google Scholar] [CrossRef] [PubMed]
- Rahimi, A.; García, J.M. Chemical Recycling of Waste Plastics for New Materials Production. Nat. Rev. Chem. 2017, 1, 0046. [Google Scholar] [CrossRef]
- Heller, M.C.; Mazor, M.H.; Keoleian, G.A. Plastics in the US: Toward a Material Flow Characterization of Production, Markets and End of Life. Environ. Res. Lett. 2020, 15, 094034. [Google Scholar] [CrossRef]
- Kunwar, B.; Cheng, H.N.; Chandrashekaran, S.R.; Sharma, B.K. Plastics to Fuel: A Review. Renew. Sustain. Energy Rev. 2016, 54, 421–428. [Google Scholar] [CrossRef]
- The Environmental Impact of Plastic Straws—Facts, Statistics and Infographic. Available online: https://get-green-now.com/environmental-impact-plastic-straws (accessed on 11 November 2022).
- Geyer, R.; Jambeck, J.R.; Law, K.L. Production, Use, and Fate of All Plastics Ever Made. Sci. Adv. 2017, 3, e1700782. [Google Scholar] [CrossRef] [PubMed]
- Celik, G.; Kennedy, R.M.; Hackler, R.A.; Ferrandon, M.; Tennakoon, A.; Patnaik, S.; Lapointe, A.M.; Ammal, S.C.; Heyden, A.; Perras, F.A.; et al. Upcycling Single-Use Polyethylene into High-Quality Liquid Products. ACS Cent. Sci 2019, 5, 1795–1803. [Google Scholar] [CrossRef]
- Ragaert, K.; Delva, L.; van Geem, K. Mechanical and Chemical Recycling of Solid Plastic Waste. Waste Manag. 2017, 69, 24–58. [Google Scholar] [CrossRef]
- Miandad, R.; Barakat, M.A.; Aburiazaiza, A.S.; Rehan, M.; Nizami, A.S. Catalytic Pyrolysis of Plastic Waste: A Review. Process Saf. Environ. Prot. 2016, 102, 822–838. [Google Scholar] [CrossRef]
- Hong, M.; Chen, E.Y.-X. Chemically Recyclable Polymers: A Circular Economy Approach to Sustainability. Green Chem. 2017, 19, 3692–3706. [Google Scholar] [CrossRef]
- Delferro, M.; Ferrandon, M.S.; Kennedy, R.M.; Celik, G.; Hackler, R.A.; Poeppelmeier, K.R.; Sadow, A.D. Catalytic Upcycling of Polymers. U.S. Patent 17/000,969, 4 March 2021. [Google Scholar]
- Hackler, R.A.; Vyavhare, K.; Kennedy, R.M.; Celik, G.; Kanbur, U.; Griffin, P.J.; Sadow, A.D.; Zang, G.; Elgowainy, A.; Sun, P.; et al. Synthetic Lubricants Derived from Plastic Waste and Their Tribological Performance. ChemSusChem 2021, 14, 4181–4189. [Google Scholar] [CrossRef]
- Celik, G.; Vyavhare, K.; Kennedy, R.M.; Hackler, R.A.; Erdemir, A.; Delferro, M. Catalytic Upcycling of Polyolefins into Lubricants. U.S. Patent 11,499,110, 15 November 2022. [Google Scholar]
- Karaba, A.; Dvořáková, V.; Patera, J.; Zámostný, P. Improving the Steam-Cracking Efficiency of Naphtha Feedstocks by Mixed/Separate Processing. J. Anal. Appl. Pyrolysis 2020, 146, 104768. [Google Scholar] [CrossRef]
- Rahimpour, M.R.; Jafari, M.; Iranshahi, D. Progress in Catalytic Naphtha Reforming Process: A Review. Appl. Energy 2013, 109, 79–93. [Google Scholar] [CrossRef]
- Ross, J.R.H. Catalysis in the Production of Energy Carriers from Oil. In Contemporary Catalysis: Fundamentals and Current Applications; Marinakis, K., Ed.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 233–249. [Google Scholar]
- Speight, J.G. 9—Gas Condensate. In Natural Gas: A Basic Handbook; Hammon, K., Ed.; Gulf Professional Publishing: Houston, TX, USA, 2019; pp. 325–358. [Google Scholar]
- Lange, J.-P. Managing Plastic Waste—Sorting, Recycling, Disposal and Product Redesign. ACS Sustain. Chem. Eng. 2021, 9, 15722–15738. [Google Scholar] [CrossRef]
- Das, P.; Tiwari, P. The Effect of Slow Pyrolysis on the Conversion of Packaging Waste Plastics (PE and PP) into Fuel. Waste Manag. 2018, 79, 615–624. [Google Scholar] [CrossRef]
- Nanda, S.; Berruti, F. Thermochemical Conversion of Plastic Waste to Fuels: A Review. Environ. Chem. Lett. 2021, 19, 123–148. [Google Scholar] [CrossRef]
- Dogu, O.; Pelucchi, M.; Van de Vijver, R.; Van Steenberge, P.H.M.; D’hooge, D.R.; Cuoci, A.; Mehl, M.; Frassoldati, A.; Faravelli, T.; Van Geem, K.M. The Chemistry of Chemical Recycling of Solid Plastic Waste via Pyrolysis and Gasification: State-of-the-Art, Challenges, and Future Directions. Prog. Energy Combust. Sci. 2021, 84, 100901. [Google Scholar] [CrossRef]
- Zhang, F.; Zhao, Y.; Wang, D.; Yan, M.; Zhang, J.; Zhang, P.; Ding, T.; Chen, L.; Chen, C. Current Technologies for Plastic Waste Treatment: A Review. J. Clean. Prod. 2021, 282, 124523. [Google Scholar] [CrossRef]
- Al-Salem, S.M.; Antelava, A.; Constantinou, A.; Manos, G.; Dutta, A. A Review on Thermal and Catalytic Pyrolysis of Plastic Solid Waste (PSW). J. Environ. Manag. 2017, 197, 177–198. [Google Scholar] [CrossRef]
- Lopez, G.; Artetxe, M.; Amutio, M.; Bilbao, J.; Olazar, M. Thermochemical Routes for the Valorization of Waste Polyolefinic Plastics to Produce Fuels and Chemicals. A Review. Renew. Sustain. Energy Rev. 2017, 73, 346–368. [Google Scholar] [CrossRef]
- Van Der Ree, T.; Weser, G. Method and System for Transferring Plastic Waste into a Fuel Having Properties of Diesel/Heating Oil. U.S. Patent 15/544,260, 11 January 2008. [Google Scholar]
- ASTM Standard D7169-05; Standard Test Method for Bromine Numbers of Petroleum Distillates and Commercial Aliphatic Olefins by Electrometric Titration. ASTM International: West Conshohocken, PA, USA, 2008; pp. 213–221. [CrossRef]
- Mangesh, V.L.; Padmanabhan, S.; Ganesan, S.; Prabhudevrahul, D.; Kumar Reddy, T.D. Prospects of Pyrolysis Oil from Plastic Waste as Fuel for Diesel Engines: A Review. In Proceedings of the IOP Conference Series: Materials Science and Engineering, Frontiers in Automobile and Mechanical Engineering, Sathyabama University, Chennai, India, 7–9 July 2016; IOP Publishing: Bristol, UK, 2017; Volume 197. [Google Scholar] [CrossRef]
- Lissitsyna, K.; Huertas, S.; Quintero, L.C.; Polo, L.M. PIONA Analysis of Kerosene by Comprehensive Two-Dimensional Gas Chromatography Coupled to Time of Flight Mass Spectrometry. Fuel 2014, 116, 716–722. [Google Scholar] [CrossRef]
- Kusenberg, M.; Zayoud, A.; Roosen, M.; Thi, H.D.; Abbas-Abadi, M.S.; Eschenbacher, A.; Kresovic, U.; de Meester, S.; van Geem, K.M. A Comprehensive Experimental Investigation of Plastic Waste Pyrolysis Oil Quality and Its Dependence on the Plastic Waste Composition. Fuel Process. Technol. 2022, 227, 107090. [Google Scholar] [CrossRef]
- Sojak, L.; Kubineca, R.; Jurdakovaa, H.; Hajekovab, E.; Bajus, M. GC-MS of Polyethylene and Polypropylene Thermal Cracking Products. Pet. Coal 2006, 48, 1–14. [Google Scholar]
- Kopinke, F.D.; Zimmermann, G.; Reyniers, G.C.; Froment, G.F. Relative Rates of Coke Formation from Hydrocarbons in Steam Cracking of Naphtha. 2. Paraffins, Naphthenes, Mono-, Di-, and Cycloolefins, and Acetylenes. Ind. Eng. Chem. Res. 1993, 32, 56–61. [Google Scholar] [CrossRef]
- Kopinke, F.D.; Zimmermann, G.; Reyniers, G.C.; Froment, G.F. Relative Rates of Coke Formation from Hydrocarbons in Steam Cracking of Naphtha. 3. Aromatic Hydrocarbons. Ind. Eng. Chem. Res. 1993, 32, 2620–2625. [Google Scholar] [CrossRef]
- Soják, L.; Kubinec, R.; Jurdáková, H.; Hájeková, E.; Bajus, M. High Resolution Gas Chromatographic–Mass Spectrometric Analysis of Polyethylene and Polypropylene Thermal Cracking Products. J. Anal. Appl. Pyrolysis 2007, 78, 387–399. [Google Scholar] [CrossRef]
- Kusenberg, M.; Faussone, G.C.; Thi, H.D.; Roosen, M.; Grilc, M.; Eschenbacher, A.; de Meester, S.; van Geem, K.M. Maximizing Olefin Production via Steam Cracking of Distilled Pyrolysis Oils from Difficult-to-Recycle Municipal Plastic Waste and Marine Litter. Sci. Total Environ. 2022, 838, 156092. [Google Scholar] [CrossRef] [PubMed]

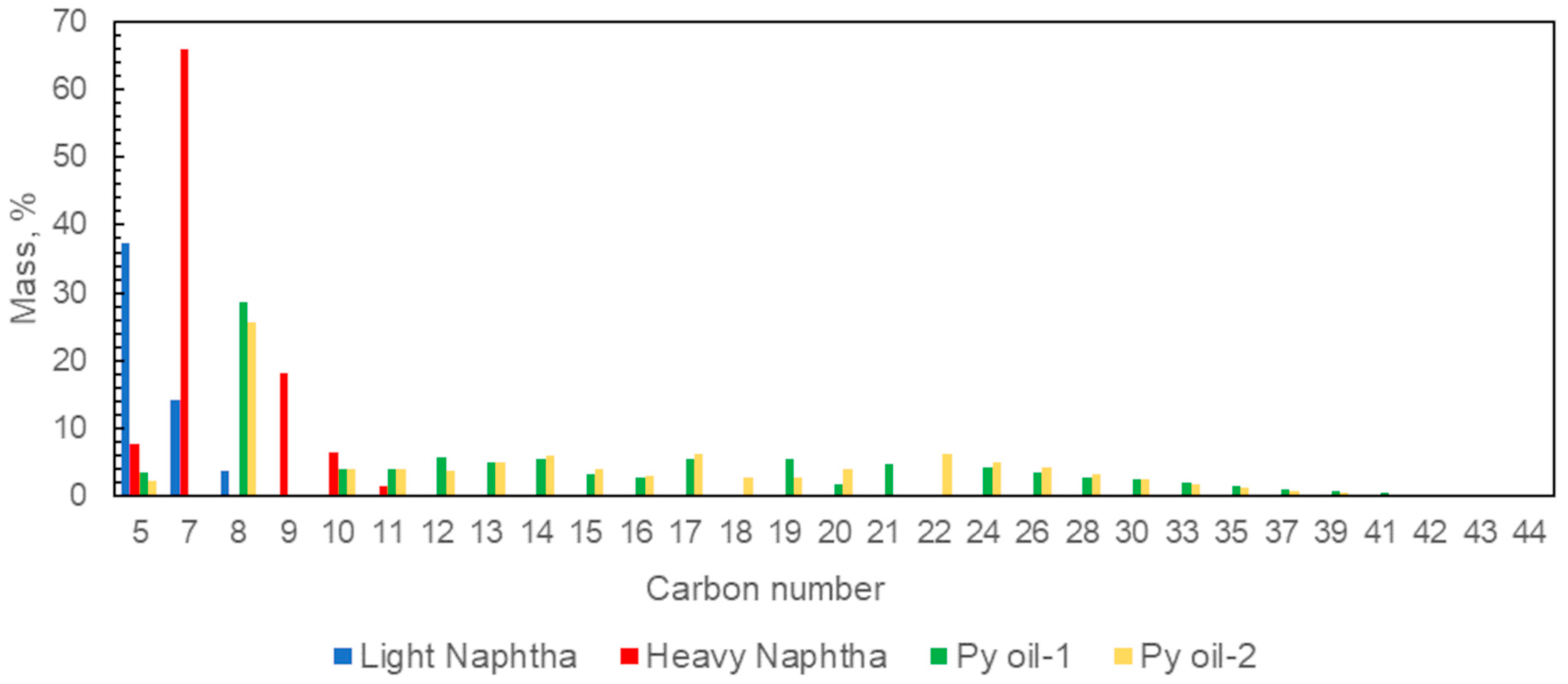
| Test | Unit | Light Naphtha | Heavy Naphtha | Py Oil-1 | Py Oil-2 | Method |
|---|---|---|---|---|---|---|
| Density, 15 °C | g/cm3 | Min. 0.67, max. 0.72 | ≥0.73 | 0.79 | 0.78 | ASTM D4052 |
| Total Sulfur | wt.% | Max. 0.060 | Max. 0.10 | 0.3 | 0.006 | ASTM D5453 |
| IBP * | °C | ≥33 | ≥50 | 60 | 55 | ASTM D1160 |
| 5% Fraction | °C | - | - | 97 | 97 | |
| 10% Fraction | °C | - | Min. 85 | 115 | 115 | |
| 20% Fraction | °C | - | - | 145 | 140 | |
| 30% Fraction | °C | - | Min. 105 | 155 | 165 | |
| 40% Fraction | °C | - | - | 195 | 210 | |
| 50% Fraction | °C | Min. 115 | Min. 120 | 232 | 242 | |
| 60% Fraction | °C | - | - | 272 | 280 | |
| 70% Fraction | °C | - | Min. 135 | 325 | 338 | |
| 80% Fraction | °C | - | - | 365 | 372 | |
| 90% Fraction | °C | Min. 170 | Min. 170 | 380 | 385 |
| Mass (%) | Boiling Point (°C) | |||
|---|---|---|---|---|
| Light Naphtha | Heavy Naphta | Py Oil-1 | Py Oil-2 | |
| 5 | 36.4 | 96.0 | 61.7 | 66.4 |
| 10 | 40.8 | 101.5 | 110.1 | 123.5 |
| 15 | - | 107.6 | 132.5 | 133.2 |
| 20 | 46.0 | 113.1 | 133.9 | 134.3 |
| 30 | 50.7 | 117.9 | 136.0 | 158.9 |
| 40 | 55.6 | 125.0 | 190.2 | 227.6 |
| 50 | 60.6 | 128.7 | 234.5 | 246.9 |
| 60 | 66.6 | 134.7 | 275.2 | 303.4 |
| 70 | 72.2 | 140.5 | 320.0 | 339.7 |
| 80 | 79.1 | 146.5 | 374.3 | 382.4 |
| 85 | - | 150.3 | 407.2 | 405.4 |
| 90 | 89.9 | 151.8 | 436.2 | 430.4 |
| 95 | 100.2 | 157.9 | 474.0 | 464.0 |
| 96 | - | 159.1 | 485.2 | 472.2 |
| 97 | - | 160.7 | 495.8 | 483.6 |
| 98 | - | 163.2 | 508.9 | 497.2 |
| 99 | - | 167.1 | 527.5 | 517.7 |
| FBP * | 117.9 | 171.4 | 541.6 | 537.9 |
| Sample | Bromine Number (g/100 g) |
|---|---|
| Light Naphtha | 1.2 |
| Heavy Naphtha | 0.3 |
| Py oil-1 | 85 |
| Py oil-2 | 304 |
| Py oil-1-F210 | 88 |
| Py oil-2-F210 | 216 |
| Component | Light Naphtha, % (w/w) | Heavy Naphtha, % (w/w) | Py Oil-1-F210, % (w/w) | Py Oil-2-F210, % (w/w) |
|---|---|---|---|---|
| Naphthenes | 15.7 | 36.3 | 9.3 | 10.1 |
| i-Paraffins | 41.7 | 28.8 | 19.1 | 19.1 |
| n-Paraffins | 40.3 | 25.9 | 7.8 | 5.5 |
| Cyclic Olefins | - | - | 45.3 | 43.4 |
| Olefins | 0.1 | 0.3 | 15.0 | 16.8 |
| Aromatics | 2.2 | 8.8 | 3.5 | 5.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Erkmen, B.; Ozdogan, A.; Ezdesir, A.; Celik, G. Can Pyrolysis Oil Be Used as a Feedstock to Close the Gap in the Circular Economy of Polyolefins? Polymers 2023, 15, 859. https://doi.org/10.3390/polym15040859
Erkmen B, Ozdogan A, Ezdesir A, Celik G. Can Pyrolysis Oil Be Used as a Feedstock to Close the Gap in the Circular Economy of Polyolefins? Polymers. 2023; 15(4):859. https://doi.org/10.3390/polym15040859
Chicago/Turabian StyleErkmen, Berrak, Adem Ozdogan, Ayhan Ezdesir, and Gokhan Celik. 2023. "Can Pyrolysis Oil Be Used as a Feedstock to Close the Gap in the Circular Economy of Polyolefins?" Polymers 15, no. 4: 859. https://doi.org/10.3390/polym15040859
APA StyleErkmen, B., Ozdogan, A., Ezdesir, A., & Celik, G. (2023). Can Pyrolysis Oil Be Used as a Feedstock to Close the Gap in the Circular Economy of Polyolefins? Polymers, 15(4), 859. https://doi.org/10.3390/polym15040859







