Renovation of Agro-Waste for Sustainable Food Packaging: A Review
Abstract
1. Introduction
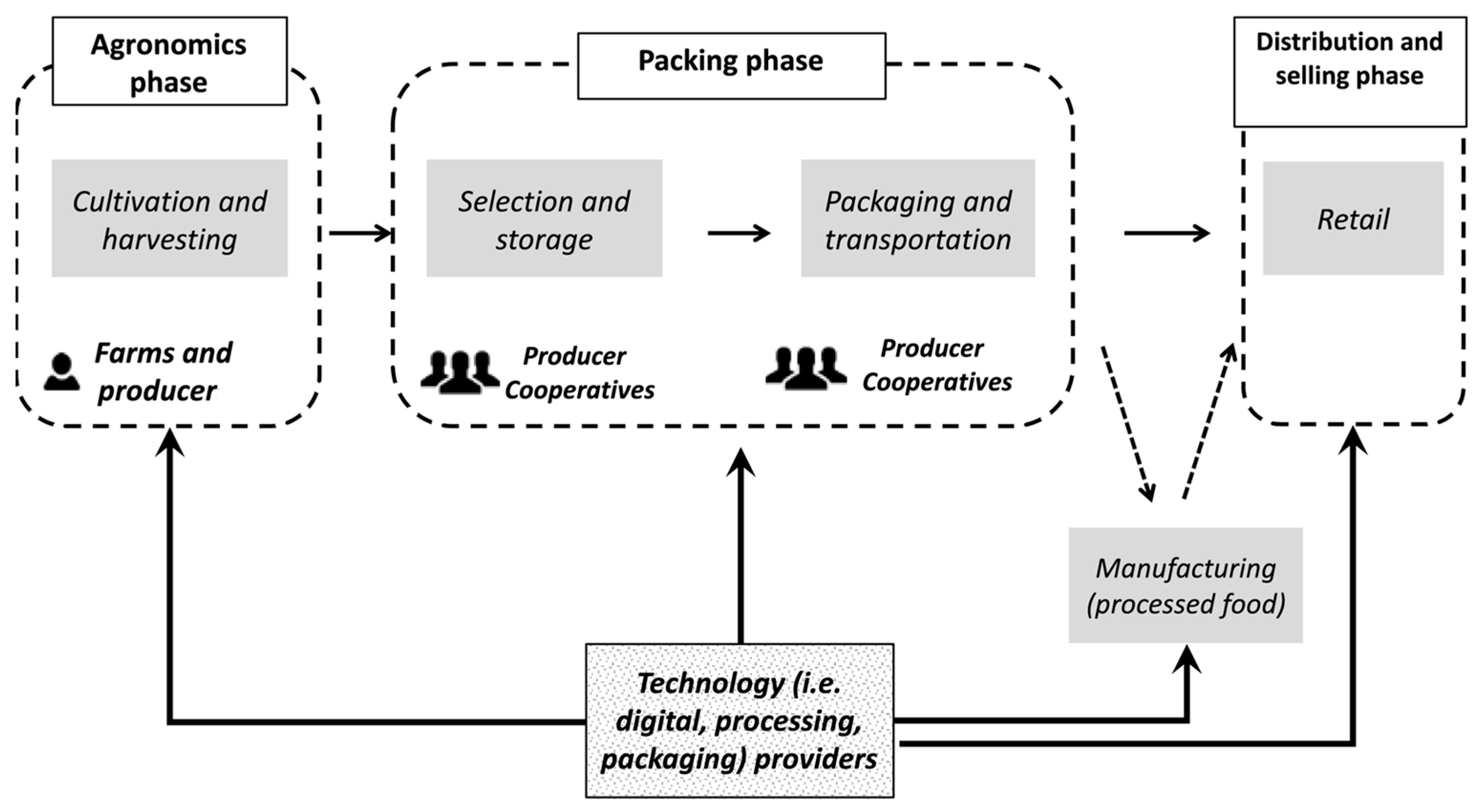
2. Industrial Agro-Waste
2.1. Agro-Waste Generation
2.2. Composition of Agricultural Waste Residues in Packaging
3. Packaging Developments Using Agro-Waste
3.1. Utilization of Crop Residues in Packaging
| Waste Source | Components | Extraction | Contents of Waste in Packaging | Polymer Blend | Role of Waste in Packaging | Packaging Form | Production Process | Major Findings | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| Pine needles | Cellulose | Sodium hydroxide (NaOH) and sodium sulfide (Na2S) | 65–90% | Microfibrillated cellulose (MFC) and halloysite nanotubes (Hal) | Matrix | Paper | Sheet former |
| [46] |
| Pine, eucalyptus, corn straw | Nanofiber | Sodium hydroxide extraction | >30% | Starch from rice flour | Filler | Film | Solution casting |
| [54] |
| Parsley and spinach stems | Powder | 2,2′-azinobis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt (ABTS) | 30–70 wt% | Poly (propylene carbonate) | Antioxidant | Film | Hot compression molding |
| [47] |
| Okra leaf stalk | Cellulose | Alkaline deep eutectic solvent | 70–100% | Mucilage polysaccharides | Matrix | Film | Solution casting |
| [48] |
| Mango seed | Fiber | Sodium hypochlorite extraction | 10–20% | PLA | Reinforcement | Sheet | Injection molded |
| [49] |
| Tamarind seed | Xyloglucan as powder | Microwave assisted extraction | 2 and 4% | Shallot stalk powder | Matrix | Film | Solution casting |
| [50] |
| Bay tree prunings | Carbohydrate | Water extraction | 10–40% | Chitosan | Filler | Film | Solution casting |
| [51] |
| Chinese chive root | Extract | Ethanol extraction | 1–5% | Chitosan | Active agent | Film | Solution casting |
| [55] |
| Rice straw | Cellulose fiber | Sodium hydroxide extraction | 25 and 35% | Chitosan | Reinforcement | Film | Solution casting |
| [44] |
| Rice straw | Cellulose | Naviglio extractor | 100% | - | Matrix | Film | Solution casting |
| [52] |
| Mandarin leaves and peel | Cellulose | Hexane and chloroform extraction | 100% | - | Matrix | Paper | Kraft paper |
| [53] |
3.2. Utilizations of Process Waste in Packaging
| Waste Source | Components | Extraction | Contents of Waste in Packaging | Polymer Blend | Role of Waste in Packaging | Packaging Form | Production Process | Major Findings | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| Cocoa pod husk | Cellulose | Alkaline treatment | 25–100% | Fiber extraction from sugarcane bagasse | Film-forming matrix | Film | Solution casting |
| [61] |
| Rice husk flakes | Rice husk flour | Mechanical grinding and sieving | 10% | Poly(3-hydroxybutyrate) (PHB) and poly(3-hydroxybutyrate-co-3-hydroxyvalerate) (PHBV) | Filler | Film | Thermo-compression |
| [56] |
| Soybean bran | Protein hydrolysates and soybean bran extract | Ethanol extraction | 2.5 g furcellaran and 5 g protein hydrolysates | Furcellaran | Antioxidant compounds | Single- and double-layer films | Solution casting |
| [59] |
| Raw wheat bran flakes | Lignocellulosic fillers | Milling | 10, 30 and 50% | Poly (butylene succinate) (PBS) | Filler | Pellets and moldings | Twin-screw extrusion and injection moldings |
| [62] |
| Beetroot bagasse | Betalain extract | Ultrafiltered extract | 1, 2, and 3% | Zein | Antioxidant compounds | Film | Solution casting |
| [61] |
| Cassava bagasse | Fiber | - | 3, 6, and 9% | Cornstarch | Hybridized agent and reinforcement | Film | Solution casting |
| [63] |
| Cassava bagasse | Lignocellulose nanofibers | Enzymatic pretreatments and colloidal mill | 0.65 and 1.3% | Cassava starch | Reinforcement | Film | Solution casting |
| [72] |
| Cassava bagasse | Dried cassava bagasse | Dehydration and grinding | 17, 33, 50, 66, and 100% | Cassava starch, gelatin, and Spirulina platensis biomass | Reinforcement | Film | Solution casting |
| [75] |
| Sugarcane bagasse and asparagus peel | Fiber | Heat drying and milling | 5, 10, 15, 20, 30, and 40% | Sweet potato starch | Reinforcement | Foam trays | Thermo-pressing |
| [57] |
| Sugarcane bagasse | Cellulose nanocrystals | Acid hydrolysis | 2, 5, and 8% | Whey protein isolate | Filler | Film | Solution casting |
| [60] |
| Woody residues from tomato, pepper, and eggplant | Lignocellulose nanofibers | Mechanical and TEMPO mediated oxidation pre-treatment | 2, 5, and 7% | Poly (vinyl alcohol) | Reinforcement | Film | Solution casting |
| [64] |
| Sweet potato residues | Starch and cellulose | Hot-air drying and ground into flour | 5, 10, 20, 30, and 40% | Poly(3-hydroxybutyrate-co-3-hydroxyvalerate) (PHBV) | Filler | Dog bones and film | Injectionmolding and compression molding |
| [58] |
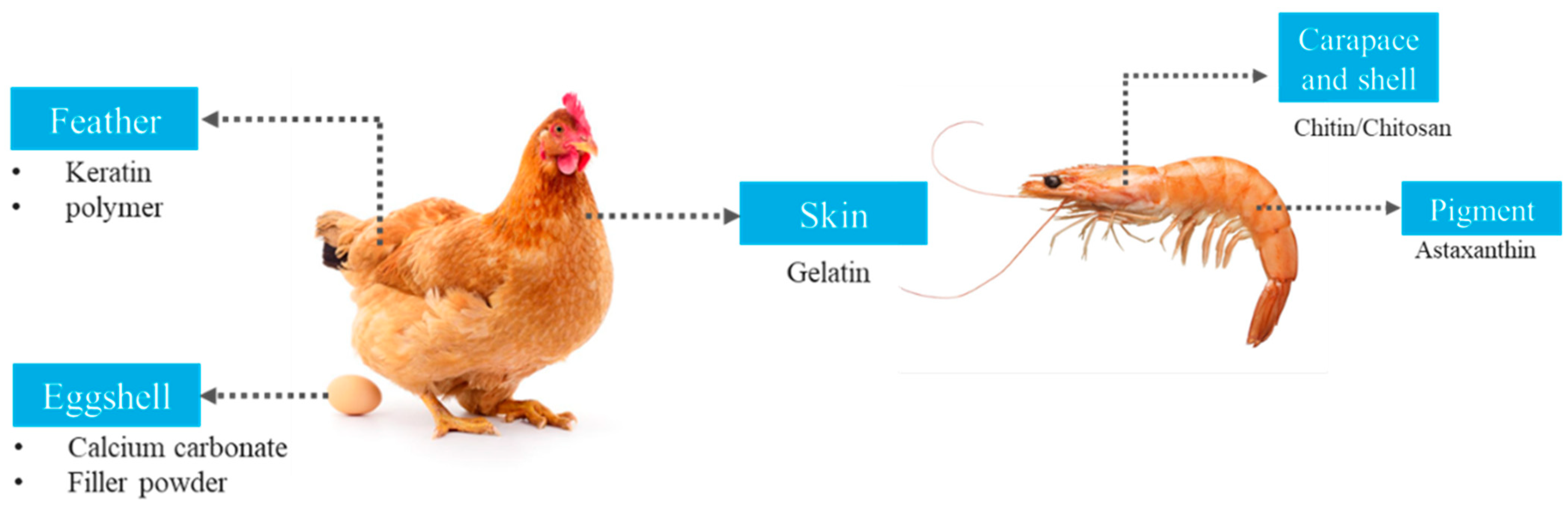
3.3. Use of Animal Waste in Packaging
| Waste Source | Components | Extraction | Contents of Waste in Packaging | Polymer Blend | Role of Waste in Packaging | Packaging Form | Production Process | Major Findings | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| Chicken skin | Gelatin | Chemical extraction using sodium hydroxide | 100 (w/w) | - | Matrix | Film | Solution casting |
| [81] |
| Poultry feathers | Powder | Milling and sieving | 625 g | - | Matrix | Film | Compression molding |
| [78] |
| Chicken feathers | Keratin | Chemical extraction using sodium hydroxide | 30:0, 30:1, 30:3 and 30:5 (starch:keratin v/v). | Turmeric starch | Filler | Film | Solution casting |
| [79] |
| Shrimp and crab shell | Protein hydrolysate | Enzymatic extraction (bromelain and alcalase) | 1.3- 6% | Chitosan and gelatin | Antioxidant and antimicrobial agent | Film | Casting solution |
| [95] |
| Bigeye tuna skin | Gelatin | Chemical extraction using NaOH | 5% | Spent coffee ground extract (SCGE) | Matrix | Film | Solvent casting |
| [82] |
| Common carp skin | Gelatin hydrolysate | Extraction using NaCl, NaOH, H2SO4, and C6H8O7. | 5 g | Furcellaran | Matrix | Film | Solution casting |
| [83] |
| Shrimp waste (head, shells of cephalothorax, and tails) | Astaxanthin | Extraction using ethanol | 15 μg/mL | Chitosan, gelatin | Antioxidant and antimicrobial compound | Film | Solution casting |
| [88] |
| Freshwater clam shells | Nanofiber | Thermal calcination process | 10 g in 200 mL dicumyl peroxide | PLA and fish gelatin | Antibacterial | Fiber | Electrospinning |
| [87] |
| Chicken egg shells | Powder | Grinding and sieving | 20 parts by weight | Polyurethane | Filler | Rigid foam | Compression molding |
| [86] |
| Chicken feathers | Fiber | Shredding | 1:10 | Polyethylene with a polypropylene core | Matrix | Non-woven feather fiber composite | Air-laid process |
| [96] |
| Eggshells | Silver-doped hydroxyapatite | Sonication | 0.5–4% | Polyurethane and algae oil | Antibacterial filler | Coating film | Coating bar applicator |
| [97] |
| Egg and shrimp shells | Powder | Grinding | 0–20 wt% | Native cassava starch, glycerol | Filler | Foam | Compression molding |
| [84] |
| Eggshell membranes | Gelatin | Extraction with alkali solution | 25–100% | Chitosan | Matrix | Film | Solution casting |
| [98] |
| Eggshells | Powder | Milling and sieving | 5, 10, 15, 20, 25 parts by weight | Polyurethane | Filler | Rigid foam | Compression molding |
| [99] |
| Shrimp cephalothoraxes and exoskeletons | Chitosan | Organic extraction using acetone and ethanol | 1:1 | Lactic acid and protein concentrate | Antioxidant and antimicrobial matrix | Film | Casting solution |
| [89] |
| White chicken feathers | Protein | Chemical extraction with NaOH | 5 g | Gelatin (0.5, 1, 1.5, 2 g), clove essential oil/cinnamaldehyde (0.5, 1, 1.5 g), | Matrix | Film | Solution casting |
| [77] |
| Feathers | Keratin | Ground and sieving | 50% | Glycerol:DI-H2O:Na2SO330:18.5:1.5 wt% | Matrix | Thin sheet | Extrusion |
| [80] |
| Chicken feet wasteLotus stem | GelatinStarch powder | NaOH and HCl | 4 g | Lotus steam starch | Matrix | Film | Coating |
| [93] |
4. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- Corrado, S.; Caldeira, C.; Eriksson, M.; Hanssen, O.J.; Hauser, H.E.; van Holsteijn, F.; Liu, G.; Östergren, K.; Parry, A.; Secondi, L.; et al. Food waste accounting methodologies: Challenges, opportunities, and further advancements. Glob. Food Secur. 2019, 20, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Shafiee-Jood, M.; Cai, X. Reducing Food Loss and Waste to Enhance Food Security and Environmental Sustainability. Environ. Sci. Technol. 2016, 50, 8432–8443. [Google Scholar] [CrossRef] [PubMed]
- Chaboud, G.; Daviron, B. Food losses and waste: Navigating the inconsistencies. Glob. Food Secur. 2017, 12, 1–7. [Google Scholar] [CrossRef]
- Ishangulyyev, R.; Kim, S.; Lee, S.H. Understanding Food Loss and Waste—Why Are We Losing and Wasting Food? Foods 2019, 8, 297. [Google Scholar] [CrossRef]
- Ciccullo, F.; Cagliano, R.; Bartezzaghi, G.; Perego, A. Implementing the circular economy paradigm in the agri-food supply chain: The role of food waste prevention technologies. Resour. Conserv. Recycl. 2020, 164, 105114. [Google Scholar] [CrossRef]
- Spang, E.S.; Moreno, L.C.; Pace, S.A.; Achmon, Y.; Donis-Gonzalez, I.; Gosliner, W.A.; Jablonski-Sheffield, M.P.; Momin, M.A.; Quested, T.E.; Winans, K.S.; et al. Food Loss and Waste: Measurement, Drivers, and Solutions. Annu. Rev. Environ. Resour. 2019, 44, 117–156. [Google Scholar] [CrossRef]
- Wohner, B.; Pauer, E.; Heinrich, V.; Tacker, M. Packaging-Related Food Losses and Waste: An Overview of Drivers and Issues. Sustainability 2019, 11, 264. [Google Scholar] [CrossRef]
- Laorenza, Y.; Chonhenchob, V.; Bumbudsanpharoke, N.; Jittanit, W.; Sae-Tan, S.; Rachtanapun, C.; Chanput, W.P.; Charoensiddhi, S.; Srisa, A.; Promhuad, K.; et al. Polymeric Packaging Applications for Seafood Products: Packaging-Deterioration Relevance, Technology and Trends. Polymers 2022, 14, 3706. [Google Scholar] [CrossRef]
- Promsorn, J.; Harnkarnsujarit, N. Pyrogallol loaded thermoplastic cassava starch based films as bio-based oxygen scavengers. Ind. Crop. Prod. 2022, 186, 115226. [Google Scholar] [CrossRef]
- Promhuad, K.; Bumbudsanpharoke, N.; Wadaugsorn, K.; Sonchaeng, U.; Harnkarnsujarit, N. Maltol-Incorporated Acetylated Cassava Starch Films for Shelf-Life-Extension Packaging of Bakery Products. Polymers 2022, 14, 5342. [Google Scholar] [CrossRef]
- San, H.; Laorenza, Y.; Behzadfar, E.; Sonchaeng, U.; Wadaugsorn, K.; Sodsai, J.; Kaewpetch, T.; Promhuad, K.; Srisa, A.; Wongphan, P.; et al. Functional Polymer and Packaging Technology for Bakery Products. Polymers 2022, 14, 3793. [Google Scholar] [CrossRef] [PubMed]
- Srisa, A.; Promhuad, K.; San, H.; Laorenza, Y.; Wongphan, P.; Wadaugsorn, K.; Sodsai, J.; Kaewpetch, T.; Tansin, K.; Harnkarnsujarit, N. Antibacterial, Antifungal and Antiviral Polymeric Food Packaging in Post-COVID-19 Era. Polymers 2022, 14, 4042. [Google Scholar] [CrossRef] [PubMed]
- Promhuad, K.; Srisa, A.; San, H.; Laorenza, Y.; Wongphan, P.; Sodsai, J.; Tansin, K.; Phromphen, P.; Chartvivatpornchai, N.; Ngoenchai, P.; et al. Applications of Hemp Polymers and Extracts in Food, Textile and Packaging: A Review. Polymers 2022, 14, 4274. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.; Ghoshal, G. Emerging trends in food packaging. Nutr. Food Sci. 2018, 48, 764–779. [Google Scholar] [CrossRef]
- Sonchaeng, U.; Promsorn, J.; Bumbudsanpharoke, N.; Chonhenchob, V.; Sablani, S.S.; Harnkarnsujarit, N. Polyesters Incorporating Gallic Acid as Oxygen Scavenger in Biodegradable Packaging. Polymers 2022, 14, 5296. [Google Scholar] [CrossRef]
- Tummala, R.R. Packaging: Past, present and future. In Proceedings of the 2005 6th International Conference on Electronic Packaging Technology, Shenzhen, China, 30 August–2 September 2005; pp. 3–7. [Google Scholar]
- Jain, R.; Tiwari, A. Biosynthesis of planet friendly bioplastics using renewable carbon source. J. Environ. Health Sci. Eng. 2015, 13, 11. [Google Scholar] [CrossRef]
- Kaewpetch, T.; Pratummang, A.; Suwarak, S.; Wongphan, P.; Promhuad, K.; Leelaphiwat, P.; Bumbudsanpharoke, N.; Lorenzo, J.M.; Harnkarnsujarit, N. Ylang-ylang (Cananga odorata) essential oils with flora odorants enhanced active function of biodegradable polyester films produced by extrusion. Food Biosci. 2023, 51, 102284. [Google Scholar] [CrossRef]
- Qian, M.; Liu, D.; Zhang, X.; Yin, Z.; Ismail, B.B.; Ye, X.; Guo, M. A review of active packaging in bakery products: Applications and future trends. Trends Food Sci. Technol. 2021, 114, 459–471. [Google Scholar] [CrossRef]
- Phothisarattana, D.; Harnkarnsujarit, N. Migration, aggregations and thermal degradation behaviors of TiO2 and ZnO incorporated PBAT/TPS nanocomposite blown films. Food Packag. Shelf Life 2022, 33, 100901. [Google Scholar] [CrossRef]
- Riaz, A.; Lei, S.; Akhtar, H.M.S.; Wan, P.; Chen, D.; Jabbar, S.; Abid, M.; Hashim, M.M.; Zeng, X. Preparation and characterization of chitosan-based antimicrobial active food packaging film incorporated with apple peel polyphenols. Int. J. Biol. Macromol. 2018, 114, 547–555. [Google Scholar] [CrossRef]
- Madurwar, M.V.; Ralegaonkar, R.V.; Mandavgane, S.A. Application of agro-waste for sustainable construction materials: A review. Constr. Build. Mater. 2013, 38, 872–878. [Google Scholar] [CrossRef]
- Yan, B.; Feng, L.; Zheng, J.; Zhang, Q.; Jiang, S.; Zhang, C.; Ding, Y.; Han, J.; Chen, W.; He, S. High performance supercapacitors based on wood-derived thick carbon electrodes synthesized via green activation process. Inorg. Chem. Front. 2022, 9, 6108–6123. [Google Scholar] [CrossRef]
- Feng, L.; Yan, B.; Zheng, J.; Chen, J.; Wei, R.; Jiang, S.; Yang, W.; Zhang, Q.; He, S. Soybean protein-derived N, O co-doped porous carbon sheets for supercapacitor applications. New J. Chem. 2022, 46, 10844–10853. [Google Scholar] [CrossRef]
- Obey, G.; Adelaide, M.; Ramaraj, R. Biochar derived from non-customized matamba fruit shell as an adsorbent for wastewater treatment. J. Bioresour. Bioprod. 2022, 7, 109–115. [Google Scholar] [CrossRef]
- Mitura, K.; Kornacka, J.; Kopczyńska, E.; Kalisz, J.; Czerwińska, E.; Affeltowicz, M.; Kaczorowski, W.; Kolesińska, B.; Frączyk, J.; Bakalova, T.; et al. Active Carbon-Based Nanomaterials in Food Packaging. Coatings 2021, 11, 161. [Google Scholar] [CrossRef]
- Dilucia, F.; LaCivita, V.; Conte, A.; Del Nobile, M.A. Sustainable Use of Fruit and Vegetable By-Products to Enhance Food Packaging Performance. Foods 2020, 9, 857. [Google Scholar] [CrossRef]
- Sharma, V.; Tsai, M.-L.; Nargotra, P.; Chen, C.-W.; Kuo, C.-H.; Sun, P.-P.; Dong, C.-D. Agro-Industrial Food Waste as a Low-Cost Substrate for Sustainable Production of Industrial Enzymes: A Critical Review. Catalysts 2022, 12, 1373. [Google Scholar] [CrossRef]
- Sadh, P.K.; Duhan, S.; Duhan, J.S. Agro-industrial wastes and their utilization using solid state fermentation: A review. Bioresour. Bioprocess. 2018, 5, 1. [Google Scholar] [CrossRef]
- Bhuimbar, M.V.; Bhagwat, P.K.; Dandge, P.B. Extraction and characterization of acid soluble collagen from fish waste: Development of collagen-chitosan blend as food packaging film. J. Environ. Chem. Eng. 2019, 7, 102983. [Google Scholar] [CrossRef]
- Hussain, C.; Singh, S.; Goswami, L. (Eds.) Emerging Trends to Approaching Zero Waste: Environmental and Social Perspectives; Elsevier: Amsterdam, The Netherlands, 2021. [Google Scholar]
- Resource Conservation and Recovery Act (RCRA). Code of Federal Regulations, Identification and listing of hazardous waste, 40 (1997): Section 261.4 (Exclusion, Materials which are not solid waste). 32. Regulatory Exclusions and Alternative Standards for the Recycling of Materials, Solid Wastes and Hazardous Wastes.” US EPA, 26 Jan. 2023. Available online: www.epa.gov/hw/regulatory-exclusions-and-alternative-standards-recycling-materials-solid-wastes-and-hazardous (accessed on 22 December 2022).
- Risdianto, H.; Sofianti, E.; Suhardi, S.H.; Setiadi, T. Optimisation of Laccase Production using White Rot Fungi and Agriculture Wastes in Solid State Fermentation. ITB J. Eng. Sci. 2012, 44, 93–105. [Google Scholar] [CrossRef]
- Salihu, A.; Abbas, O.; Sallau, A.B.; Alam, Z. Agricultural residues for cellulolytic enzyme production by Aspergillus niger: Effects of pretreatment. 3 Biotech 2015, 5, 1101–1106. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Euring, M.; Ostendorf, K.; Zhang, K. Biobased materials for food packaging. J. Bioresour. Bioprod. 2022, 7, 1–13. [Google Scholar] [CrossRef]
- Oyeoka, H.C.; Ewulonu, C.M.; Nwuzor, I.C.; Obele, C.M.; Nwabanne, J.T. Packaging and degradability properties of polyvinyl alcohol/gelatin nanocomposite films filled water hyacinth cellulose nanocrystals. J. Bioresour. Bioprod. 2021, 6, 168–185. [Google Scholar] [CrossRef]
- Kora, A.J. Leaves as dining plates, food wraps and food packing material: Importance of renewable resources in Indian culture. Bull. Natl. Res. Cent. 2019, 43, 205. [Google Scholar] [CrossRef]
- Manan, S.; Ullah, M.W.; Ul-Islam, M.; Atta, O.M.; Yang, G. Synthesis and applications of fungal mycelium-based advanced functional materials. J. Bioresour. Bioprod. 2021, 6, 1–10. [Google Scholar] [CrossRef]
- Dissanayake, D.G.K.; Weerasinghe, D.; Perera, T.D.R.; Bandara, M.M.A.L.; Thathsara, S.K.T.; Perera, S. A Sustainable Transparent Packaging Material from the Arecanut Leaf Sheath. Waste Biomass- Valorization 2021, 12, 5725–5742. [Google Scholar] [CrossRef]
- Kaya, M.; Khadem, S.; Cakmak, Y.S.; Mujtaba, M.; Ilk, S.; Akyuz, L.; Salaberria, A.M.; Labidi, J.; Abdulqadir, A.H.; Deligöz, E. Antioxidative and antimicrobial edible chitosan films blended with stem, leaf and seed extracts of Pistacia terebinthus for active food packaging. RSC Adv. 2018, 8, 3941–3950. [Google Scholar] [CrossRef]
- Campos, D.; Piccirillo, C.; Pullar, R.C.; Castro, P.M.; Pintado, M.M. Characterization and antimicrobial properties of food packaging methylcellulose films containing stem extract of Ginja cherry. J. Sci. Food Agric. 2014, 94, 2097–2103. [Google Scholar] [CrossRef]
- Fontes-Candia, C.; Erboz, E.; Martínez-Abad, A.; López-Rubio, A.; Martínez-Sanz, M. Superabsorbent food packaging bioactive cellulose-based aerogels from Arundo donax waste biomass. Food Hydrocoll. 2019, 96, 151–160. [Google Scholar] [CrossRef]
- Berthet, M.A.; Angellier-Coussy, H.; Chea, V.; Guillard, V.; Gastaldi, E.; Gontard, N. Sustainable food packaging: Valorising wheat straw fibres for tuning PHBV-based composites properties. Compos. Part A Appl. Sci. Manuf. 2015, 72, 139–147. [Google Scholar] [CrossRef]
- Elhussieny, A.; Faisal, M.; D’Angelo, G.; Aboulkhair, N.T.; Everitt, N.M.; Fahim, I.S. Valorisation of shrimp and rice straw waste into food packaging applications. Ain Shams Eng. J. 2020, 11, 1219–1226. [Google Scholar] [CrossRef]
- Benito-González, I.; López-Rubio, A.; Martínez-Sanz, M.M. High-performance starch biocomposites with cellulose from waste biomass: Film properties and retrogradation behaviour. Carbohydr. Polym. 2019, 216, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Ramakanth, D.; Akhila, K.; Gaikwad, K.K. Influence of halloysite nanotubes/microfibrillated cellulose on pine leaves waste based ethylene scavenging composite paper for food packaging applications. Appl. Clay Sci. 2023, 231, 106726. [Google Scholar] [CrossRef]
- Tran, T.N.; Lim, K.T.; Fiorentini, F.; Athanassiou, A. Antioxidant and Biocompatible CO 2 -Based Biocomposites from Vegetable Wastes for Active Food Packaging. Adv. Sustain. Syst. 2022, 6, 2100470. [Google Scholar] [CrossRef]
- Olawuyi, I.F.; Lee, W.Y. Development and Characterization of Biocomposite Films Based on Polysaccharides Derived from Okra Plant Waste for Food Packaging Application. Polymers 2022, 14, 4884. [Google Scholar] [CrossRef] [PubMed]
- Lima, E.M.B.; Middea, A.; Neumann, R.; Thiré, R.M.D.S.M.; Pereira, J.F.; de Freitas, S.C.; Penteado, M.S.; Lima, A.M.; Minguita, A.P.D.S.; Mattos, M.D.C.; et al. Biocomposites of PLA and Mango Seed Waste: Potential Material for Food Packaging and a Technological Alternative to Reduce Environmental Impact. Starch 2021, 73, 2000118. [Google Scholar] [CrossRef]
- Thivya, P.; Bhosale, Y.K.; Anandakumar, S.; Hema, V.; Sinija, V.R. Exploring the Effective Utilization of Shallot Stalk Waste and Tamarind Seed for Packaging Film Preparation. Waste Biomass- Valorization 2021, 12, 5779–5794. [Google Scholar] [CrossRef]
- Rincón, E.; Espinosa, E.; García-Domínguez, M.; Balu, A.; Vilaplana, F.; Serrano, L.; Jiménez-Quero, A. Bioactive pectic polysaccharides from bay tree pruning waste: Sequential subcritical water extraction and application in active food packaging. Carbohydr. Polym. 2021, 272, 118477. [Google Scholar] [CrossRef]
- Bilo, F.; Pandini, S.; Sartore, L.; Depero, L.E.; Gargiulo, G.; Bonassi, A.; Federici, S.; Bontempi, E. A sustainable bioplastic obtained from rice straw. J. Clean. Prod. 2018, 200, 357–368. [Google Scholar] [CrossRef]
- Kasaai, M.R.; Moosavi, A. Treatment of Kraft paper with citrus wastes for food packaging applications: Water and oxygen barrier properties improvement. Food Packag. Shelf Life 2017, 12, 59–65. [Google Scholar] [CrossRef]
- de Oliveira, A.L.M.; Bento, J.A.C.; Fidelis, M.C.; Dias, M.C.; de Barros, H.E.A.; Natarelli, C.V.L.; Lago, R.C.D.; Barbosa, J.W.; Ossani, P.C.; Caliari, M.; et al. Effect of pine, eucalyptus, and corn straw nanofibers on the structural properties of rice flour-based biodegradable films. Ind. Crop. Prod. 2023, 191, 115929. [Google Scholar] [CrossRef]
- Riaz, A.; Lagnika, C.; Luo, H.; Dai, Z.; Nie, M.; Hashim, M.M.; Liu, C.; Song, J.; Li, D. Chitosan-based biodegradable active food packaging film containing Chinese chive (Allium tuberosum) root extract for food application. Int. J. Biol. Macromol. 2020, 150, 595–604. [Google Scholar] [CrossRef] [PubMed]
- Melendez-Rodriguez, B.; Torres-Giner, S.; Aldureid, A.; Cabedo, L.; Lagaron, J.M. Reactive Melt Mixing of Poly(3-Hydroxybutyrate)/Rice Husk Flour Composites with Purified Biosustainably Produced Poly(3-Hydroxybutyrate-co-3-Hydroxyvalerate). Materials 2019, 12, 2152. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Tirado, J.P.; Vejarano, R.; Tapia-Blácido, D.R.; Angelats-Silva, L.M.; Siche, R. The addition of sugarcane bagasse and asparagus peel enhances the properties of sweet potato starch foams. Packag. Technol. Sci. 2019, 32, 227–237. [Google Scholar] [CrossRef]
- Vannini, M.; Marchese, P.; Sisti, L.; Saccani, A.; Mu, T.; Sun, H.; Celli, A. Integrated Efforts for the Valorization of Sweet Potato By-Products within a Circular Economy Concept: Biocomposites for Packaging Applications Close the Loop. Polymers 2021, 13, 1048. [Google Scholar] [CrossRef] [PubMed]
- Tkaczewska, J.; Jamróz, E.; Zając, M.; Guzik, P.; Gedif, H.D.; Turek, K.; Kopeć, M. Antioxidant edible double-layered film based on waste from soybean production as a vegan active packaging for perishable food products. Food Chem. 2023, 400, 134009. [Google Scholar] [CrossRef]
- Sukyai, P.; Anongjanya, P.; Bunyahwuthakul, N.; Kongsin, K.; Harnkarnsujarit, N.; Sukatta, U.; Sothornvit, R.; Chollakup, R. Effect of cellulose nanocrystals from sugarcane bagasse on whey protein isolate-based films. Food Res. Int. 2018, 107, 528–535. [Google Scholar] [CrossRef]
- Azmin, S.N.H.M.; Hayat, N.A.B.M.; Nor, M.S.M. Development and characterization of food packaging bioplastic film from cocoa pod husk cellulose incorporated with sugarcane bagasse fibre. J. Bioresour. Bioprod. 2020, 5, 248–255. [Google Scholar] [CrossRef]
- Sasimowski, E.; Majewski, Ł.; Grochowicz, M. Artificial Ageing, Chemical Resistance, and Biodegradation of Biocomposites from Poly(Butylene Succinate) and Wheat Bran. Materials 2021, 14, 7580. [Google Scholar] [CrossRef] [PubMed]
- Abotbina, W.; Sapuan, S.M.; Ilyas, R.A.; Sultan, M.T.H.; Alkbir, M.F.M. Preparation and Characterization of Black Seed/Cassava Bagasse Fiber-Reinforced Cornstarch-Based Hybrid Composites. Sustainability 2022, 14, 12042. [Google Scholar] [CrossRef]
- Bascón-Villegas, I.; Sánchez-Gutiérrez, M.; Pérez-Rodríguez, F.; Espinosa, E.; Rodríguez, A. Lignocellulose Nanofibre Obtained from Agricultural Wastes of Tomato, Pepper and Eggplants Improves the Performance of Films of Polyvinyl Alcohol (PVA) for Food Packaging. Foods 2021, 10, 3043. [Google Scholar] [CrossRef]
- Rodríguez-Félix, F.; Corte-Tarazón, J.A.; Rochín-Wong, S.; Fernández-Quiroz, J.D.; Garzón-García, A.M.; Santos-Sauceda, I.; Plascencia-Martínez, D.F.; Chan-Chan, L.H.; Vásquez-López, C.; Barreras-Urbina, C.G.; et al. Physicochemical, structural, mechanical and antioxidant properties of zein films incorporated with no-ultrafiltered and ultrafiltered betalains extract from the beetroot (Beta vulgaris) bagasse with potential application as active food packaging. J. Food Eng. 2022, 334. [Google Scholar] [CrossRef]
- Moustafa, H.; El-Wakil, A.E.-A.A.; Nour, M.T.; Youssef, A.M. Kenaf fibre treatment and its impact on the static, dynamic, hydrophobicity and barrier properties of sustainable polystyrene biocomposites. RSC Adv. 2020, 10, 29296–29305. [Google Scholar] [CrossRef]
- Bascón-Villegas, I.; Pereira, M.; Espinosa, E.; Sánchez-Gutiérrez, M.; Rodríguez, A.; Pérez-Rodríguez, F. A new eco-friendly packaging system incorporating lignocellulose nanofibres from agri-food residues applied to fresh-cut lettuce. J. Clean. Prod. 2022, 372, 133597. [Google Scholar] [CrossRef]
- Kumar, G.M.; Kumar, R.P. Characterization of pine needle ash particulates reinforced surface composite fabricated by friction stir process. Mater. Res. Express 2018, 6, 046539. [Google Scholar] [CrossRef]
- Menzel, C.; González-Martínez, C.; Vilaplana, F.; Diretto, G.; Chiralt, A. Incorporation of natural antioxidants from rice straw into renewable starch films. Int. J. Biol. Macromol. 2020, 146, 976–986. [Google Scholar] [CrossRef] [PubMed]
- Freitas, P.A.; González-Martínez, C.; Chiralt, A. Using rice straw fractions to develop reinforced, active PLA-starch bilayers for meat preservation. Food Chem. 2023, 405, 134990. [Google Scholar] [CrossRef] [PubMed]
- Ravindran, R.; Jaiswal, A.K. A comprehensive review on pre-treatment strategy for lignocellulosic food industry waste: Challenges and opportunities. Bioresour. Technol. 2016, 199, 92–102. [Google Scholar] [CrossRef]
- Travalini, A.P.; Lamsal, B.; Magalhães, W.L.E.; Demiate, I.M. Cassava starch films reinforced with lignocellulose nanofibers from cassava bagasse. Int. J. Biol. Macromol. 2019, 139, 1151–1161. [Google Scholar] [CrossRef]
- Ibrahim, M.I.J.; Sapuan, S.M.; Zainudin, E.S.; Zuhri, M.Y.M. Preparation and characterization of cornhusk/sugar palm fiber reinforced Cornstarch-based hybrid composites. J. Mater. Res. Technol. 2020, 9, 200–211. [Google Scholar] [CrossRef]
- Zhou, Y.; Fan, M.; Chen, L. Interface and bonding mechanisms of plant fibre composites: An overview. Compos. Part B: Eng. 2016, 101, 31–45. [Google Scholar] [CrossRef]
- Cardoso, T.; Esmerino, L.A.; Bolanho, B.C.; Demiate, I.M.; Danesi, E.D.G. Technological viability of biobased films formulated with cassava by-product and Spirulina platensis. J. Food Process. Eng. 2019, 42, 13136. [Google Scholar] [CrossRef]
- Jayathilakan, K.; Sultana, K.; Radhakrishna, K.; Bawa, A.S. Utilization of byproducts and waste materials from meat, poultry and fish processing industries: A review. J. Food Sci. Technol. 2012, 49, 278–293. [Google Scholar] [CrossRef] [PubMed]
- Song, N.-B.; Lee, J.-H.; Al Mijan, M.; Bin Song, K. Development of a chicken feather protein film containing clove oil and its application in smoked salmon packaging. Lwt 2014, 57, 453–460. [Google Scholar] [CrossRef]
- McGauran, T.; Harris, M.; Dunne, N.; Smyth, B.M.; Cunningham, E. Development and optimisation of extruded bio-based polymers from poultry feathers. Eur. Polym. J. 2021, 158, 110678. [Google Scholar] [CrossRef]
- Oluba, O.M.; Osayame, E.; Shoyombo, A.O. Production and characterization of keratin-starch bio-composite film from chicken feather waste and turmeric starch. Biocatal. Agric. Biotechnol. 2021, 33, 101996. [Google Scholar] [CrossRef]
- Barone, J.R.; Arikan, O. Composting and biodegradation of thermally processed feather keratin polymer. Polym. Degrad. Stab. 2007, 92, 859–867. [Google Scholar] [CrossRef]
- Jusoh, N.; Isa, M.; Sarbon, N. Physical, mechanical and antioxidant properties of chicken skin gelatin films incorporated with virgin coconut oil. Biocatal. Agric. Biotechnol. 2022, 45, 102525. [Google Scholar] [CrossRef]
- Getachew, A.T.; Ahmad, R.; Park, J.-S.; Chun, B.-S. Fish skin gelatin based packaging films functionalized by subcritical water extract from spent coffee ground. Food Packag. Shelf Life 2021, 29, 100735. [Google Scholar] [CrossRef]
- Tkaczewska, J.; Kulawik, P.; Jamróz, E.; Guzik, P.; Zając, M.; Szymkowiak, A.; Turek, K. One- and double-layered furcellaran/carp skin gelatin hydrolysate film system with antioxidant peptide as an innovative packaging for perishable foods products. Food Chem. 2021, 351, 129347. [Google Scholar] [CrossRef]
- Kaewtatip, K.; Chiarathanakrit, C.; Riyajan, S.-A. The effects of egg shell and shrimp shell on the properties of baked starch foam. Powder Technol. 2018, 335, 354–359. [Google Scholar] [CrossRef]
- Mousavi, S.M.; Hashemi, S.A.; Amani, A.M.; Saed, H.; Jahandideh, S.; Mojoudi, F. Polyethylene Terephthalate/Acryl Butadiene Styrene Copolymer Incorporated with Oak Shell, Potassium Sorbate and Egg Shell Nanoparticles for Food Packaging Applications: Control of Bacteria Growth, Physical and Mechanical Properties. Polym. Renew. Resour. 2017, 8, 177–196. [Google Scholar] [CrossRef]
- Leszczyńska, M.; Ryszkowska, J.; Szczepkowski, L.; Kurańska, M.; Prociak, A.; Leszczyński, M.K.; Gloc, M.; Antos-Bielska, M.; Mizera, K. Cooperative effect of rapeseed oil-based polyol and egg shells on the structure and properties of rigid polyurethane foams. Polym. Test. 2020, 90, 106696. [Google Scholar] [CrossRef]
- Wu, D.-Y.; Wang, S.-S.; Wu, C.-S. Antibacterial properties and cytocompatibility of biobased nanofibers of fish scale gelatine, modified polylactide, and freshwater clam shell. Int. J. Biol. Macromol. 2020, 165, 1219–1228. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Wei, R.; Jia, Z.; Song, R. Characteristics and bioactive functions of chitosan/gelatin-based film incorporated with ε-polylysine and astaxanthin extracts derived from by-products of shrimp (Litopenaeus vannamei). Food Hydrocoll. 2019, 100, 105436. [Google Scholar] [CrossRef]
- Arancibia, M.Y.; Alemán, A.; López-Caballero, M.E.; Gómez-Guillén, M.C.; Montero, P. Development of active films of chitosan isolated by mild extraction with added protein concentrate from shrimp waste. Food Hydrocoll. 2015, 43, 91–99. [Google Scholar] [CrossRef]
- Al-Ali, R.M.; Al-Hilifi, S.A.; Rashed, M.M. Fabrication, characterization, and anti-free radical performance of edible packaging-chitosan film synthesized from shrimp shell incorporated with ginger essential oil. J. Food Meas. Charact. 2021, 15, 2951–2962. [Google Scholar] [CrossRef]
- Dasumiati; Saridewi, N.; Malik, M. Food packaging development of bioplastic from basic waste of cassava peel (manihot uttilisima) and shrimp shell. IOP Conf. Ser. Mater. Sci. Eng. 2019, 602, 012053. [Google Scholar] [CrossRef]
- Qian, Z.-J.; Zhang, J.; Xu, W.-R.; Zhang, Y.-C. Development of active packaging films based on liquefied shrimp shell chitin and polyvinyl alcohol containing β-cyclodextrin/cinnamaldehyde inclusion. Int. J. Biol. Macromol. 2022, 214, 67–76. [Google Scholar] [CrossRef]
- Rather, J.A.; Makroo, H.A.; Showkat, Q.A.; Majid, D.; Dar, B. Recovery of gelatin from poultry waste: Characteristics of the gelatin and lotus starch-based coating material and its application in shelf-life enhancement of fresh cherry tomato. Food Packag. Shelf Life 2022, 31, 100775. [Google Scholar] [CrossRef]
- Chungsiriporn, J.; Khunthongkaew, P.; Wongnoipla, Y.; Sopajarn, A.; Karrila, S.; Iewkittayakorn, J. Fibrous packaging paper made of oil palm fiber with beeswax-chitosan solution to improve water resistance. Ind. Crop. Prod. 2022, 177, 114541. [Google Scholar] [CrossRef]
- Hajji, S.; Kchaou, H.; Bkhairia, I.; Salem, R.B.S.-B.; Boufi, S.; Debeaufort, F.; Nasri, M. Conception of active food packaging films based on crab chitosan and gelatin enriched with crustacean protein hydrolysates with improved functional and biological properties. Food Hydrocoll. 2021, 116, 106639. [Google Scholar] [CrossRef]
- Dieckmann, E.; Nagy, B.; Yiakoumetti, K.; Sheldrick, L.; Cheeseman, C. Thermal insulation packaging for cold-chain deliveries made from feathers. Food Packag. Shelf Life 2019, 21, 100360. [Google Scholar] [CrossRef]
- Patil, C.K.; Jirimali, H.D.; Paradeshi, J.S.; Chaudhari, B.L.; Gite, V.V. Functional antimicrobial and anticorrosive polyurethane composite coatings from algae oil and silver doped egg shell hydroxyapatite for sustainable development. Prog. Org. Coat. 2018, 128, 127–136. [Google Scholar] [CrossRef]
- Mohammadi, R.; Mohammadifar, M.A.; Rouhi, M.; Kariminejad, M.; Mortazavian, A.M.; Sadeghi, E.; Hasanvand, S. Physico-mechanical and structural properties of eggshell membrane gelatin- chitosan blend edible films. Int. J. Biol. Macromol. 2018, 107, 406–412. [Google Scholar] [CrossRef] [PubMed]
- Zieleniewska, M.; Leszczyński, M.K.; Szczepkowski, L.; Bryśkiewicz, A.; Krzyżowska, M.; Bień, K.; Ryszkowska, J. Development and applicational evaluation of the rigid polyurethane foam composites with egg shell waste. Polym. Degrad. Stab. 2016, 132, 78–86. [Google Scholar] [CrossRef]


| Agro-Waste | Lignin (%) | α Cellulose (%) | Hemicellulose (%) |
|---|---|---|---|
| Banana peel | 11 ± 1.12 | 9 ± 0.08 | 3 ± 0.55 |
| Cassava peel | 39 ± 0.34 | 8 ± 0.52 | 25 ± 0.41 |
| Cornstalks | 30 ± 1.19 | 15 ± 0.67 | 33 ± 2.63 |
| Empty fruit bunches | 25.79 | 19.30 | 25.55 |
| Finger millet hulls | 25 ± 0.73 | 32 ± 1.38 | 4 ± 1.6 |
| Groundnut husks | 36 ± 1. 41 | 4 20 ± 0.88 | 25 ± 1.03 |
| Rice | 31.97 | 31.10 | 18.35 |
| Rice husks | 18.82 | 41.05 | 17.63 |
| Rice straw | 35 ± 1.31 | 27 ± 1.97 | 16 ± 2.01 |
| Sawdust | 44 ± 1.77 | 17 ± 0.14 | 21 ± 1.92 |
| Shea nut cake | 26 ± 0.26 | 9 ± 0.33 | 21 ± 0.09 |
| Sorghum hulls | 39 ± 0.82 | 35± 0.07 | 4 ± 0.11 |
| Soybean hulls | 35 ± 0.12 | 16 ± 0.21 | 4 ± 0.19 |
| Sugarcane bagasse | 45 ± 0.52 | 26 ± 0.34 | 19 ± 0.13 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Varghese, S.A.; Pulikkalparambil, H.; Promhuad, K.; Srisa, A.; Laorenza, Y.; Jarupan, L.; Nampitch, T.; Chonhenchob, V.; Harnkarnsujarit, N. Renovation of Agro-Waste for Sustainable Food Packaging: A Review. Polymers 2023, 15, 648. https://doi.org/10.3390/polym15030648
Varghese SA, Pulikkalparambil H, Promhuad K, Srisa A, Laorenza Y, Jarupan L, Nampitch T, Chonhenchob V, Harnkarnsujarit N. Renovation of Agro-Waste for Sustainable Food Packaging: A Review. Polymers. 2023; 15(3):648. https://doi.org/10.3390/polym15030648
Chicago/Turabian StyleVarghese, Sandhya Alice, Harikrishnan Pulikkalparambil, Khwanchat Promhuad, Atcharawan Srisa, Yeyen Laorenza, Lerpong Jarupan, Tarinee Nampitch, Vanee Chonhenchob, and Nathdanai Harnkarnsujarit. 2023. "Renovation of Agro-Waste for Sustainable Food Packaging: A Review" Polymers 15, no. 3: 648. https://doi.org/10.3390/polym15030648
APA StyleVarghese, S. A., Pulikkalparambil, H., Promhuad, K., Srisa, A., Laorenza, Y., Jarupan, L., Nampitch, T., Chonhenchob, V., & Harnkarnsujarit, N. (2023). Renovation of Agro-Waste for Sustainable Food Packaging: A Review. Polymers, 15(3), 648. https://doi.org/10.3390/polym15030648








