Recent Biotechnological Applications of Polyhydroxyalkanoates (PHA) in the Biomedical Sector—A Review
Abstract
:1. Introduction
1.1. Polyhydroxyalkanoates
1.2. PHA Synthesis
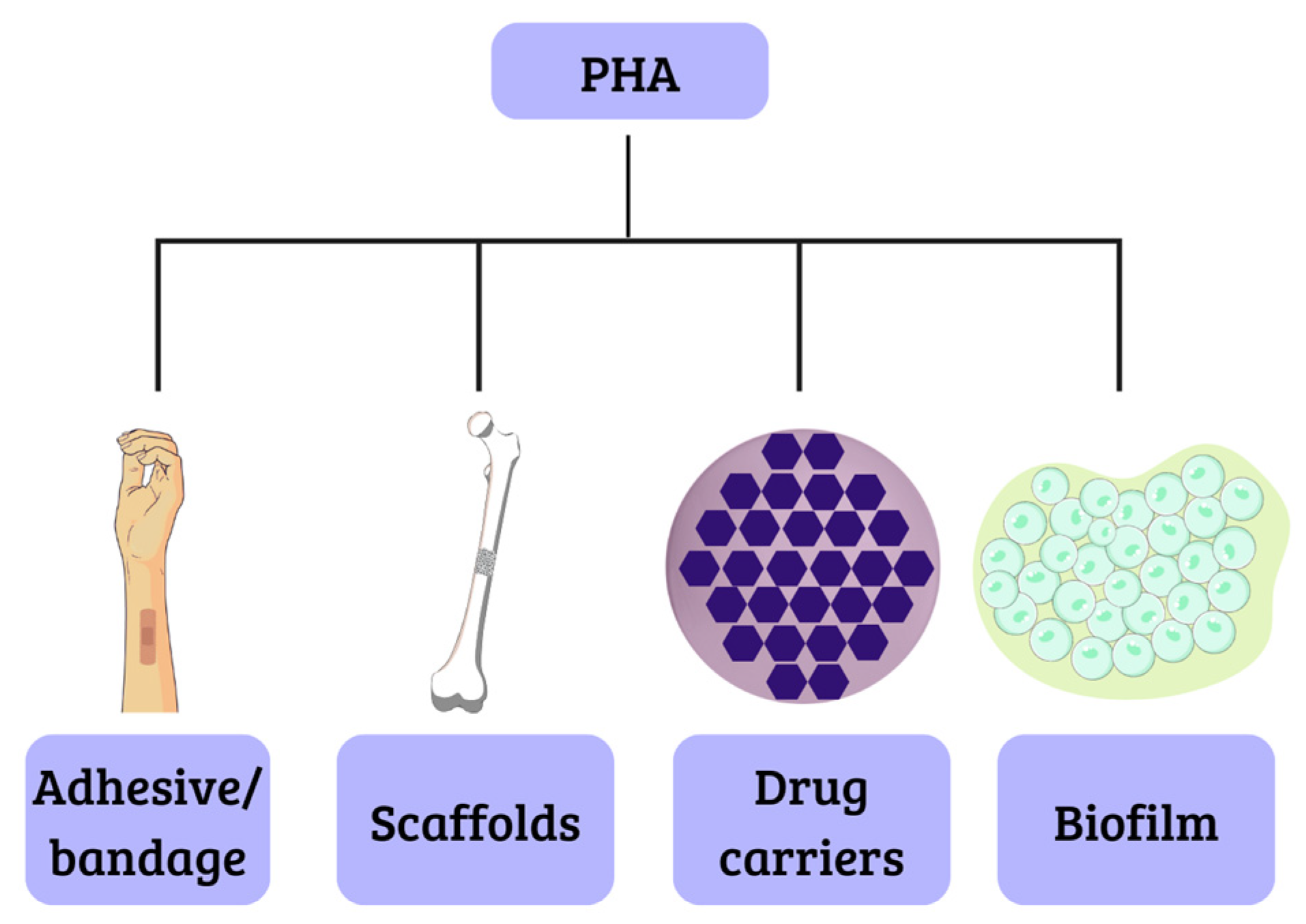
2. PHA in Biotechnology
2.1. Scaffolds in Fabric Engineering
2.2. Drug Carriers
| Polymer | Producing Body | Application | Author Year | Refs. |
|---|---|---|---|---|
| PHB | Stigeoclonium sp. B3 | Drug carrier/biomaterial | Mourão et al., 2020 | [36] |
| PHB | Stigeoclonium sp. B3 | Drug carrier/biomaterial | Mourão et al., 2021 | [40] |
| PHV | Bacillus cereus MCCB 281 | Drug carrier | Mohandas et al., 2018 | [49] |
| P(3HB)/P(3HB-co-3HV)/MCL-PHA | Cupriavidus necator DSM 428/Pseudomonas chlororaphis DSM 19603 | Scaffolds | Esmail et al., 2021 | [50] |
| PHB | Azotobacter vinelandii | Tissue engineering | Romo-Uribe et al., 2017 | [51] |
| PHB | Unspecified | Scaffolds | Codreanu et al., 2020 | [52] |
| PHB | Unspecified | Scaffolds | El-Shanshory et al., 2022 | [53] |
| PHB | Unspecified | Scaffolds | Pryadko et al., 2022 | [54] |
| P(3HO-co-3HHX) | Unspecified | Scaffolds | Ansari et al., 2017 | [55] |
| PHA | Unspecified | Scaffolds | Lizzarraga-Valderrama et al., 2020 | [57] |
| PHB | Unspecified (commercial) | Tissue engineering | Saska et al., 2018 | [58] |
| PHBVHHx | Unspecified | Scaffolds | Wei et al., 2018 | [59] |
| PHA | Pseudomonas aeruginosa ATCC 27853 | Tissue/carrier engineering | Impallomeni et al., 2018 | [62] |
| PHB | Bacillus Cereus VIT-SSR1 | Drug carrier | Evangeline et al., 2019 | [63] |
| PHB | Unspecified (commercial) | Drug carrier | Bini et al., 2017 | [64] |
| PHB | Pseudomonas aeruginosa SU-1 | Drug carrier | Senthilkumar et al., 2017 | [65] |
| PHBV | Unspecified | Drug carrier | Vardhan et al., 2017 | [66] |
| PHA | Unspecified | Drug carrier | Jiang et al., 2019 | [67] |
| PHB | Unspecified | Drug carrier | Chen et al., 2021 | [68] |
| PHA | Unspecified | Drug carrier | De Freitas E Castro et al., 2021 | [69] |
| PHA | Unspecified | Drug carrier | Huerta-Angeles et al., 2017 | [70] |
| PHA | Bacillus subtilis NCDC0671 | Drug carrier | Umesh et al., 2018 | [71] |
| P(3HO-co-3HD-co-3HDD) | Pseudomonas mendocin CH50 | Drug carrier | Owji et al., 2021 | [72] |
| poly(R-3-hydroxybutyrate-co-1,4-butylene adipate) | Unspecified | Drug carrier | Musumeci et al., 2019 | [73] |
| PHBHHx | Unspecified | Drug carrier | Fan et al., 2018 | [74] |
| P3HB | Unspecified | Drug carrier | Shershneva et al., 2018 | [75] |
| PHA | Unspecified | Drug carrier | Canãdas et al., 2021 | [76] |
| PHA | Unspecified | Drug carrier | Pavic et al., 2022 | [77] |
| PHB | Commercial of bacterial origin | Biomedical use | Salama et al., 2018 | [78] |
| P(3HV-co-3HB) | Halomonas sp. | Biomaterial | El-Malek et al., 2021 | [79] |
| PHO | Pseudomonas putida KT2440 | Wound treatment | Balcucho et al., 2023 | [80] |
| P(3HB)/P(3HB-co-3HD) | Pseudomonas mendocina CH50 and Bacillus subtilis OK2 | Wound treatment | Kalaoglu-Altan et al., 2021 | [81] |
| PHA | Pseudomonas chlororaphis subsp. aurantiaca | Adhesive | Pereira et al., 2018 | [82] |
| PHA | Pseudomonas putida KT2440 | Adhesive and biofilm | Urbina et al., 2018 | [83] |
| P(3HB)/P(3HB/3HV) | Unspecified | Herbicide carrier | Vijayamma et al., 2021 | [84] |
2.3. Other Applications in Medicine
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Johansen, M.R.; Christensen, T.B.; Ramos, T.M.; Syberg, K. A Review of the Plastic Value Chain from a Circular Economy Perspective. J. Environ. Manag. 2022, 302, 113975. [Google Scholar] [CrossRef] [PubMed]
- Vimal, K.E.K.; Mathiyazhagan, K.; Agarwal, V.; Luthra, S.; Kirupanandan, S. Analysis of Barriers That Impede the Elimination of Single-Use Plastic in Developing Economy Context. J. Clean. Prod. 2020, 272, 122629. [Google Scholar] [CrossRef]
- Ganesh, K.A.; Anjana, K.; Hinduja, M.; Sujitha, K.; Dharani, G. Review on Plastic Wastes in Marine Environment—Biodegradation and Biotechnological Solutions. Mar. Pollut. Bull. 2020, 150, 110733. [Google Scholar] [CrossRef]
- Bucci, K.; Tulio, M.; Rochman, C.M. What is Known and Unknown about the Effects of Plastic Pollution: A Meta-analysis and Systematic Review. Ecol. Appl. 2020, 30, e02044. [Google Scholar] [CrossRef]
- Okoffo, E.D.; Donner, E.; McGrath, S.P.; Tscharke, B.J.; O’Brien, J.W.; O’Brien, S.; Ribeiro, F.; Burrows, S.D.; Toapanta, T.; Rauert, C.; et al. Plastics in Biosolids from 1950 to 2016: A Function of Global Plastic Production and Consumption. Water Res. 2021, 201, 117367. [Google Scholar] [CrossRef]
- Ncube, L.K.; Ude, A.U.; Ogunmuyiwa, E.N.; Zulkifli, R.; Beas, I.N. An Overview of Plastic Waste Generation and Management in Food Packaging Industries. Recycling 2021, 6, 12. [Google Scholar] [CrossRef]
- Patrício Silva, A.L.; Prata, J.C.; Walker, T.R.; Duarte, A.C.; Ouyang, W.; Barcelò, D.; Rocha-Santos, T. Increased Plastic Pollution Due to COVID-19 Pandemic: Challenges and Recommendations. Chem. Eng. J. 2021, 405, 126683. [Google Scholar] [CrossRef]
- Drzyzga, O.; Prieto, A. Plastic Waste Management, a Matter for the ‘Community’. Microb. Biotechnol. 2019, 12, 66–68. [Google Scholar] [CrossRef]
- Objetivos de Desenvolvimento Sustentável|As Nações Unidas No Brasil. Available online: https://brasil.un.org/pt-br/sdgs (accessed on 23 August 2023).
- Udayakumar, G.P.; Muthusamy, S.; Selvaganesh, B.; Sivarajasekar, N.; Rambabu, K.; Sivamani, S.; Sivakumar, N.; Maran, J.P.; Hosseini-Bandegharaei, A. Ecofriendly Biopolymers and Composites: Preparation and Their Applications in Water-Treatment. Biotechnol. Adv. 2021, 52, 107815. [Google Scholar] [CrossRef]
- Sharma, V.; Sehgal, R.; Gupta, R. Polyhydroxyalkanoate (PHA): Properties and Modifications. Polymer 2021, 212, 123161. [Google Scholar] [CrossRef]
- Kalia, V.C.; Singh Patel, S.K.; Shanmugam, R.; Lee, J.-K. Polyhydroxyalkanoates: Trends and Advances toward Biotechnological Applications. Bioresour. Technol. 2021, 326, 124737. [Google Scholar] [CrossRef]
- Sabapathy, P.C.; Devaraj, S.; Meixner, K.; Anburajan, P.; Kathirvel, P.; Ravikumar, Y.; Zabed, H.M.; Qi, X. Recent Developments in Polyhydroxyalkanoates (PHAs) Production—A Review. Bioresour. Technol. 2020, 306, 123132. [Google Scholar] [CrossRef] [PubMed]
- Taguchi, S.; Matsumoto, K. Evolution of Polyhydroxyalkanoate Synthesizing Systems toward a Sustainable Plastic Industry. Polym. J. 2021, 53, 67–79. [Google Scholar] [CrossRef]
- Ishak, K.A.; Velayutham, T.S.; Annuar, M.S.M.; Sirajudeen, A.A.O. Structure-Property Interpretation of Biological Polyhydroxyalkanoates with Different Monomeric Composition: Dielectric Spectroscopy Investigation. Int. J. Biol. Macromol. 2021, 169, 311–320. [Google Scholar] [CrossRef] [PubMed]
- Raza, Z.A.; Riaz, S.; Banat, I.M. Polyhydroxyalkanoates: Properties and Chemical Modification Approaches for Their Functionalization. Biotechnol. Prog. 2018, 34, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Tan, G.-Y.; Chen, C.-L.; Li, L.; Ge, L.; Wang, L.; Razaad, I.; Li, Y.; Zhao, L.; Mo, Y.; Wang, J.-Y. Start a Research on Biopolymer Polyhydroxyalkanoate (PHA): A Review. Polymers 2014, 6, 706–754. [Google Scholar] [CrossRef]
- Balakrishna Pillai, A.; Jaya Kumar, A.; Thulasi, K.; Kumarapillai, H. Evaluation of Short-Chain-Length Polyhydroxyalkanoate Accumulation in Bacillus Aryabhattai. Braz. J. Microbiol. 2017, 48, 451–460. [Google Scholar] [CrossRef]
- Tarrahi, R.; Fathi, Z.; Seydibeyoğlu, M.Ö.; Doustkhah, E.; Khataee, A. Polyhydroxyalkanoates (PHA): From Production to Nanoarchitecture. Int. J. Biol. Macromol. 2020, 146, 596–619. [Google Scholar] [CrossRef]
- Goswami, M.; Rekhi, P.; Debnath, M.; Ramakrishna, S. Microbial Polyhydroxyalkanoates Granules: An Approach Targeting Biopolymer for Medical Applications and Developing Bone Scaffolds. Molecules 2021, 26, 860. [Google Scholar] [CrossRef]
- Sagong, H.-Y.; Son, H.F.; Choi, S.Y.; Lee, S.Y.; Kim, K.-J. Structural Insights into Polyhydroxyalkanoates Biosynthesis. Trends Biochem. Sci. 2018, 43, 790–805. [Google Scholar] [CrossRef]
- Muthuraj, R.; Valerio, O.; Mekonnen, T.H. Recent Developments in Short- and Medium-Chain- Length Polyhydroxyalkanoates: Production, Properties, and Applications. Int. J. Biol. Macromol. 2021, 187, 422–440. [Google Scholar] [CrossRef] [PubMed]
- Obruca, S.; Sedlacek, P.; Slaninova, E.; Fritz, I.; Daffert, C.; Meixner, K.; Sedrlova, Z.; Koller, M. Novel Unexpected Functions of PHA Granules. Appl. Microbiol. Biotechnol. 2020, 104, 4795–4810. [Google Scholar] [CrossRef]
- Obulisamy, P.K.; Mehariya, S. Polyhydroxyalkanoates from Extremophiles: A Review. Bioresour. Technol. 2021, 325, 124653. [Google Scholar] [CrossRef] [PubMed]
- Mitra, R.; Xu, T.; Chen, G.; Xiang, H.; Han, J. An Updated Overview on the Regulatory Circuits of Polyhydroxyalkanoates Synthesis. Microb. Biotechnol. 2022, 15, 1446–1470. [Google Scholar] [CrossRef]
- Verlinden RA, J.; Hill, D.J.; Kenward, M.A.; Williams, C.D.; Radecka, I. Bacterial Synthesis of Biodegradable Polyhydroxyalkanoates. J. Appl. Microbiol. 2007, 102, 1437–1449. [Google Scholar] [CrossRef] [PubMed]
- Ai, M.; Zhu, Y.; Jia, X. Recent Advances in Constructing Artificial Microbial Consortia for the Production of Medium-Chain-Length Polyhydroxyalkanoates. World J. Microbiol. Biotechnol. 2021, 37, 2. [Google Scholar] [CrossRef]
- Guleria, S.; Singh, H.; Sharma, V.; Bhardwaj, N.; Arya, S.K.; Puri, S.; Khatri, M. Polyhydroxyalkanoates Production from Domestic Waste Feedstock: A Sustainable Approach towards Bio-Economy. J. Clean. Prod. 2022, 340, 130661. [Google Scholar] [CrossRef]
- Możejko-Ciesielska, J.; Marciniak, P.; Szacherska, K. Polyhydroxyalkanoates Synthesized by Aeromonas Species: Trends and Challenges. Polymers 2019, 11, 1328. [Google Scholar] [CrossRef]
- Rehm, B.H.A.; Mitsky, T.A.; Steinbüchel, A. Role of Fatty Acid De Novo Biosynthesis in Polyhydroxyalkanoic Acid (PHA) and Rhamnolipid Synthesis by Pseudomonads: Establishment of the Transacylase (PhaG)-Mediated Pathway for PHA Biosynthesis in Escherichia coli. Appl. Environ. Microbiol. 2001, 67, 3102–3109. [Google Scholar] [CrossRef]
- Blunt, W.; Lagassé, A.; Jin, Z.; Dartiailh, C.; Sparling, R.; Gapes, D.J.; Levin, D.B.; Cicek, N. Efficacy of Medium Chain-Length Polyhydroxyalkanoate Biosynthesis from Different Biochemical Pathways under Oxygen-Limited Conditions Using Pseudomonas Putida LS46. Process Biochem. 2019, 82, 19–31. [Google Scholar] [CrossRef]
- Lu, J.; Tappel, R.C.; Nomura, C.T. Mini-Review: Biosynthesis of Poly(Hydroxyalkanoates). Polym. Rev. 2009, 49, 226–248. [Google Scholar] [CrossRef]
- Możejko-Ciesielska, J.; Serafim, L.S. Proteomic Response of Pseudomonas Putida KT2440 to Dual Carbon-Phosphorus Limitation during Mcl-PHAs Synthesis. Biomolecules 2019, 9, 796. [Google Scholar] [CrossRef] [PubMed]
- Urtuvia, V.; Villegas, P.; González, M.; Seeger, M. Bacterial Production of the Biodegradable Plastics Polyhydroxyalkanoates. Int. J. Biol. Macromol. 2014, 70, 208–213. [Google Scholar] [CrossRef] [PubMed]
- Valentino, F.; Karabegovic, L.; Majone, M.; Morgan-Sagastume, F.; Werker, A. Polyhydroxyalkanoate (PHA) Storage within a Mixed-Culture Biomass with Simultaneous Growth as a Function of Accumulation Substrate Nitrogen and Phosphorus Levels. Water Res. 2015, 77, 49–63. [Google Scholar] [CrossRef] [PubMed]
- Mourão, M.M.; Gradíssimo, D.G.; Santos, A.V.; Schneider, M.P.C.; Faustino, S.M.M.; Vasconcelos, V.; Xavier, L.P. Optimization of Polyhydroxybutyrate Production by Amazonian Microalga Stigeoclonium Sp. B23. Biomolecules 2020, 10, 1628. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.B.; Pereira, J.R.; Marreiros, B.C.; Reis, M.A.M.; Freitas, F. Microbial Production of Medium-Chain Length Polyhydroxyalkanoates. Process Biochem. 2021, 102, 393–407. [Google Scholar] [CrossRef]
- Muhammadi; Shabina; Afzal, M.; Hameed, S. Bacterial Polyhydroxyalkanoates-Eco-Friendly next Generation Plastic: Production, Biocompatibility, Biodegradation, Physical Properties and Applications. Green Chem. Lett. Rev. 2015, 8, 56–77. [Google Scholar] [CrossRef]
- Chen, G.-Q.; Jiang, X.-R. Engineering Bacteria for Enhanced Polyhydroxyalkanoates (PHA) Biosynthesis. Synth. Syst. Biotechnol. 2017, 2, 192–197. [Google Scholar] [CrossRef]
- Mourão, M.M.; Xavier, L.P.; Urbatzka, R.; Figueiroa, L.B.; da Costa, C.E.F.; Dias, C.G.B.T.; Schneider, M.P.C.; Vasconcelos, V.; Santos, A.V. Characterization and Biotechnological Potential of Intracellular Polyhydroxybutyrate by Stigeoclonium Sp. B23 Using Cassava Peel as Carbon Source. Polymers 2021, 13, 687. [Google Scholar] [CrossRef]
- Chen, G.-Q.; Chen, X.-Y.; Wu, F.-Q.; Chen, J.-C. Polyhydroxyalkanoates (PHA) toward Cost Competitiveness and Functionality. Adv. Ind. Eng. Polym. Res. 2020, 3, 1–7. [Google Scholar] [CrossRef]
- Gomes Gradíssimo, D.; Pereira Xavier, L.; Valadares Santos, A. Cyanobacterial Polyhydroxyalkanoates: A Sustainable Alternative in Circular Economy. Molecules 2020, 25, 4331. [Google Scholar] [CrossRef]
- Klimek, K.; Ginalska, G. Proteins and Peptides as Important Modifiers of the Polymer Scaffolds for Tissue Engineering Applications—A Review. Polymers 2020, 12, 844. [Google Scholar] [CrossRef]
- Jafari, M.; Paknejad, Z.; Rad, M.R.; Motamedian, S.R.; Eghbal, M.J.; Nadjmi, N.; Khojasteh, A. Polymeric Scaffolds in Tissue Engineering: A Literature Review: Polymeric Scaffolds in Tissue Engineering. J. Biomed. Mater. Res. 2017, 105, 431–459. [Google Scholar] [CrossRef] [PubMed]
- Qu, H.; Fu, H.; Han, Z.; Sun, Y. Biomaterials for Bone Tissue Engineering Scaffolds: A Review. RSC Adv. 2019, 9, 26252–26262. [Google Scholar] [CrossRef] [PubMed]
- Scott, L.; Jurewicz, I.; Jeevaratnam, K.; Lewis, R. Carbon Nanotube-Based Scaffolds for Cardiac Tissue Engineering—Systematic Review and Narrative Synthesis. Bioengineering 2021, 8, 80. [Google Scholar] [CrossRef] [PubMed]
- Mafi, P.; Hindocha, S.; Mafi, R.; Khan, W.S. Evaluation of Biological Protein-Based Collagen Scaffolds in Cartilage and Musculoskeletal Tissue Engineering—A Systematic Review of the Literature. CSCR 2012, 7, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Soleymani Eil Bakhtiari, S.; Karbasi, S.; Toloue, E.B. Modified Poly(3-Hydroxybutyrate)-Based Scaffolds in Tissue Engineering Applications: A Review. Int. J. Biol. Macromol. 2021, 166, 986–998. [Google Scholar] [CrossRef]
- Mohandas, S.P.; Balan, L.; Jayanath, G.; Anoop, B.S.; Philip, R.; Cubelio, S.S.; Bright Singh, I.S. Biosynthesis and Characterization of Polyhydroxyalkanoate from Marine Bacillus Cereus MCCB 281 Utilizing Glycerol as Carbon Source. Int. J. Biol. Macromol. 2018, 119, 380–392. [Google Scholar] [CrossRef] [PubMed]
- Esmail, A.; Pereira, J.R.; Zoio, P.; Silvestre, S.; Menda, U.D.; Sevrin, C.; Grandfils, C.; Fortunato, E.; Reis, M.A.M.; Henriques, C.; et al. Oxygen Plasma Treated-Electrospun Polyhydroxyalkanoate Scaffolds for Hydrophilicity Improvement and Cell Adhesion. Polymers 2021, 13, 1056. [Google Scholar] [CrossRef]
- Romo-Uribe, A.; Meneses-Acosta, A.; Domínguez-Díaz, M. Viability of HEK 293 Cells on Poly-β-Hydroxybutyrate (PHB) Biosynthesized from a Mutant Azotobacter Vinelandii Strain. Cast Film and Electrospun Scaffolds. Mater. Sci. Eng. C 2017, 81, 236–246. [Google Scholar] [CrossRef]
- Codreanu, A.; Balta, C.; Herman, H.; Cotoraci, C.; Mihali, C.V.; Zurbau, N.; Zaharia, C.; Rapa, M.; Stanescu, P.; Radu, I.-C.; et al. Bacterial Cellulose-Modified Polyhydroxyalkanoates Scaffolds Promotes Bone Formation in Critical Size Calvarial Defects in Mice. Materials 2020, 13, 1433. [Google Scholar] [CrossRef]
- El-Shanshory, A.A.; Agwa, M.M.; Abd-Elhamid, A.I.; Soliman, H.M.A.; Mo, X.; Kenawy, E.-R. Metronidazole Topically Immobilized Electrospun Nanofibrous Scaffold: Novel Secondary Intention Wound Healing Accelerator. Polymers 2022, 14, 454. [Google Scholar] [CrossRef] [PubMed]
- Pryadko, A.S.; Botvin, V.V.; Mukhortova, Y.R.; Pariy, I.; Wagner, D.V.; Laktionov, P.P.; Chernonosova, V.S.; Chelobanov, B.P.; Chernozem, R.V.; Surmeneva, M.A.; et al. Core-Shell Magnetoactive PHB/Gelatin/Magnetite Composite Electrospun Scaffolds for Biomedical Applications. Polymers 2022, 14, 529. [Google Scholar] [CrossRef] [PubMed]
- Ansari, N.F.; Annuar, M.S.M.; Murphy, B.P. A Porous Medium-chain-length Poly(3-hydroxyalkanoates)/Hydroxyapatite Composite as Scaffold for Bone Tissue Engineering. Eng. Life Sci. 2016, 17, 420–429. [Google Scholar] [CrossRef]
- Mukheem, A.; Muthoosamy, K.; Manickam, S.; Sudesh, K.; Shahabuddin, S.; Saidur, R.; Akbar, N.; Sridewi, N. Fabrication and Characterization of an Electrospun PHA/Graphene Silver Nanocomposite Scaffold for Antibacterial Applications. Materials 2018, 11, 1673. [Google Scholar] [CrossRef] [PubMed]
- Lizarraga-Valderrama, L.R.; Nigmatullin, R.; Ladino, B.; Taylor, C.S.; Boccaccini, A.R.; Knowles, J.C.; Claeyssens, F.; Haycock, J.W.; Roy, I. Modulation of Neuronal Cell Affinity of Composite Scaffolds Based on Polyhydroxyalkanoates and Bioactive Glasses. Biomed. Mater. 2020, 15, 045024. [Google Scholar] [CrossRef]
- Saska, S.; Pires, L.C.; Cominotte, M.A.; Mendes, L.S.; De Oliveira, M.F.; Maia, I.A.; Da Silva, J.V.L.; Ribeiro, S.J.L.; Cirelli, J.A. Three-Dimensional Printing and in Vitro Evaluation of Poly(3-Hydroxybutyrate) Scaffolds Functionalized with Osteogenic Growth Peptide for Tissue Engineering. Mater. Sci. Eng. C 2018, 89, 265–273. [Google Scholar] [CrossRef]
- Wei, D.-X.; Dao, J.-W.; Liu, H.-W.; Chen, G.-Q. Suspended Polyhydroxyalkanoate Microspheres as 3D Carriers for Mammalian Cell Growth. Artif. Cells Nanomed. Biotechnol. 2018, 46, 473–483. [Google Scholar] [CrossRef]
- Prakash, P.; Lee, W.-H.; Loo, C.-Y.; Wong, H.S.J.; Parumasivam, T. Advances in Polyhydroxyalkanoate Nanocarriers for Effective Drug Delivery: An Overview and Challenges. Nanomaterials 2022, 12, 175. [Google Scholar] [CrossRef]
- Koller, M. Biodegradable and Biocompatible Polyhydroxy-Alkanoates (PHA): Auspicious Microbial Macromolecules for Pharmaceutical and Therapeutic Applications. Molecules 2018, 23, 362. [Google Scholar] [CrossRef]
- Impallomeni, G.; Ballistreri, A.; Carnemolla, G.M.; Rizzo, M.G.; Nicolò, M.S.; Guglielmino, S.P.P. Biosynthesis and Structural Characterization of Polyhydroxyalkanoates Produced by Pseudomonas Aeruginosa ATCC 27853 from Long Odd-Chain Fatty Acids. Int. J. Biol. Macromol. 2018, 108, 608–614. [Google Scholar] [CrossRef]
- Evangeline, S.; Sridharan, T.B. Biosynthesis and Statistical Optimization of Polyhydroxyalkanoate (PHA) Produced by Bacillus Cereus VIT-SSR1 and Fabrication of Biopolymer Films for Sustained Drug Release. Int. J. Biol. Macromol. 2019, 135, 945–958. [Google Scholar] [CrossRef] [PubMed]
- Bini, R.A.; Silva, M.F.; Varanda, L.C.; Da Silva, M.A.; Dreiss, C.A. Soft Nanocomposites of Gelatin and Poly(3-Hydroxybutyrate) Nanoparticles for Dual Drug Release. Colloids Surf. B Biointerfaces 2017, 157, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Senthilkumar, P.; Dawn, S.S.; Sree Samanvitha, K.; Sanjay Kumar, S.; Narendra Kumar, G.; Samrot, A.V. Optimization and Characterization of Poly[R]Hydroxyalkanoate of Pseudomonas Aeruginosa SU-1 to Utilize in Nanoparticle Synthesis for Curcumin Delivery. Biocatal. Agric. Biotechnol. 2017, 12, 292–298. [Google Scholar] [CrossRef]
- Vardhan, H.; Mittal, P.; Adena, S.K.R.; Mishra, B. Long-Circulating Polyhydroxybutyrate-Co-Hydroxyvalerate Nanoparticles for Tumor Targeted Docetaxel Delivery: Formulation, Optimization and in Vitro Characterization. Eur. J. Pharm. Sci. 2017, 99, 85–94. [Google Scholar] [CrossRef]
- Jiang, L.; Luo, Z.; Loh, X.J.; Wu, Y.-L.; Li, Z. PHA-Based Thermogel as a Controlled Zero-Order Chemotherapeutic Delivery System for the Effective Treatment of Melanoma. ACS Appl. Bio. Mater. 2019, 2, 3591–3600. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Gao, J.; Yan, E.; Wang, Y.; Li, Y.; Lu, H.; Fan, L.; Wang, D.; An, Q. A Novel Porous Composite Membrane of PHA/PVA via Coupling of Electrospinning and Spin Coating for Antibacterial Applications. Mater. Lett. 2021, 301, 130279. [Google Scholar] [CrossRef]
- De Freitas E Castro, M.; Mendonça, T.T.; Da Silva, L.F.; Gomez, J.G.C.; Sanchez Rodriguez, R.J. Carriers Based on Poly-3-Hydroxyalkanoates Containing Nanomagnetite to Trigger Hormone Release. Int. J. Biol. Macromol. 2021, 166, 448–458. [Google Scholar] [CrossRef]
- Huerta-Angeles, G.; Brandejsová, M.; Nigmatullin, R.; Kopecká, K.; Vágnerová, H.; Šmejkalová, D.; Roy, I.; Velebný, V. Synthesis of Graft Copolymers Based on Hyaluronan and Poly(3-Hydroxyalkanoates). Carbohydr. Polym. 2017, 171, 220–228. [Google Scholar] [CrossRef]
- Umesh, M.; Priyanka, K.; Thazeem, B.; Preethi, K. Biogenic PHA Nanoparticle Synthesis and Characterization from Bacillus subtilis NCDC0671 Using Orange Peel Medium. Int. J. Polym. Mater. Polym. Biomater. 2018, 67, 996–1004. [Google Scholar] [CrossRef]
- Owji, N.; Mandakhbayar, N.; Gregory, D.A.; Marcello, E.; Kim, H.; Roy, I.; Knowles, J.C. Mussel Inspired Chemistry and Bacteria Derived Polymers for Oral Mucosal Adhesion and Drug Delivery. Front. Bioeng. Biotechnol. 2021, 9, 663764. [Google Scholar] [CrossRef] [PubMed]
- Musumeci, T.; Cupri, S.; Bonaccorso, A.; Impallomeni, G.; Ballistreri, A.; Puglisi, G.; Pignatello, R. Technology Assessment of New Biodegradable Poly(R-3-Hydroxybutyrate-Co-1,4-Butylene Adipate) Copolymers for Drug Delivery. J. Appl. Polym. Sci. 2019, 136, 47233. [Google Scholar] [CrossRef]
- Fan, F.; Wang, L.; Ouyang, Z.; Wen, Y.; Lu, X. Development and Optimization of a Tumor Targeting System Based on Microbial Synthesized PHA Biopolymers and PhaP Mediated Functional Modification. Appl. Microbiol. Biotechnol. 2018, 102, 3229–3241. [Google Scholar] [CrossRef]
- Shershneva, A.; Murueva, A.; Nikolaeva, E.; Shishatskaya, E.; Volova, T. Novel Spray-Dried PHA Microparticles for Antitumor Drug Release. Dry. Technol. 2018, 36, 1387–1398. [Google Scholar] [CrossRef]
- Cañadas, O.; García-García, A.; Prieto, M.; Pérez-Gil, J. Polyhydroxyalkanoate Nanoparticles for Pulmonary Drug Delivery: Interaction with Lung Surfactant. Nanomaterials 2021, 11, 1482. [Google Scholar] [CrossRef]
- Pavic, A.; Stojanovic, Z.; Pekmezovic, M.; Veljović, Đ.; O’Connor, K.; Malagurski, I.; Nikodinovic-Runic, J. Polyenes in Medium Chain Length Polyhydroxyalkanoate (Mcl-PHA) Biopolymer Microspheres with Reduced Toxicity and Improved Therapeutic Effect against Candida Infection in Zebrafish Model. Pharmaceutics 2022, 14, 696. [Google Scholar] [CrossRef]
- Salama, H.E.; Aziz, M.S.A.; Saad, G.R. Thermal Properties, Crystallization and Antimicrobial Activity of Chitosan Biguanidine Grafted Poly(3-Hydroxybutyrate) Containing Silver Nanoparticles. Int. J. Biol. Macromol. 2018, 111, 19–27. [Google Scholar] [CrossRef]
- El-malek, F.A.; Rofeal, M.; Farag, A.; Omar, S.; Khairy, H. Polyhydroxyalkanoate Nanoparticles Produced by Marine Bacteria Cultivated on Cost Effective Mediterranean Algal Hydrolysate Media. J. Biotechnol. 2021, 328, 95–105. [Google Scholar] [CrossRef]
- Balcucho, J.; Narváez, D.M.; Tarazona, N.A.; Castro-Mayorga, J.L. Microbially Synthesized Polymer-Metal Nanoparticles Composites as Promising Wound Dressings to Overcome Methicillin-Resistance Staphylococcus Aureus Infections. Polymers 2023, 15, 920. [Google Scholar] [CrossRef]
- Kalaoglu-Altan, O.I.; Baskan, H.; Meireman, T.; Basnett, P.; Azimi, B.; Fusco, A.; Funel, N.; Donnarumma, G.; Lazzeri, A.; Roy, I.; et al. Silver Nanoparticle-Coated Polyhydroxyalkanoate Based Electrospun Fibers for Wound Dressing Applications. Materials 2021, 14, 4907. [Google Scholar] [CrossRef]
- Pereira, J.R.; Araújo, D.; Marques, A.C.; Neves, L.A.; Grandfils, C.; Sevrin, C.; Alves, V.D.; Fortunato, E.; Reis, M.A.M.; Freitas, F. Demonstration of the Adhesive Properties of the Medium-Chain-Length Polyhydroxyalkanoate Produced by Pseudomonas Chlororaphis Subsp. Aurantiaca from Glycerol. Int. J. Biol. Macromol. 2019, 122, 1144–1151. [Google Scholar] [CrossRef]
- Urbina, L.; Wongsirichot, P.; Corcuera, M.Á.; Gabilondo, N.; Eceiza, A.; Winterburn, J.; Retegi, A. Application of Cider By-Products for Medium Chain Length Polyhydroxyalkanoate Production by Pseudomonas Putida KT2440. Eur. Polym. J. 2018, 108, 1–9. [Google Scholar] [CrossRef]
- Vijayamma, R.; Maria, H.J.; Thomas, S.; Shishatskaya, E.I.; Kiselev, E.G.; Nemtsev, I.V.; Sukhanova, A.A.; Volova, T.G. A Study of the Properties and Efficacy of Microparticles Based on P(3HB) and P(3HB/3HV) Loaded with Herbicides. J. Appl. Polym. Sci. 2022, 139, 51756. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Diniz, M.S.d.F.; Mourão, M.M.; Xavier, L.P.; Santos, A.V. Recent Biotechnological Applications of Polyhydroxyalkanoates (PHA) in the Biomedical Sector—A Review. Polymers 2023, 15, 4405. https://doi.org/10.3390/polym15224405
Diniz MSdF, Mourão MM, Xavier LP, Santos AV. Recent Biotechnological Applications of Polyhydroxyalkanoates (PHA) in the Biomedical Sector—A Review. Polymers. 2023; 15(22):4405. https://doi.org/10.3390/polym15224405
Chicago/Turabian StyleDiniz, Matheus Silva da Fonseca, Murilo Moraes Mourão, Luciana Pereira Xavier, and Agenor Valadares Santos. 2023. "Recent Biotechnological Applications of Polyhydroxyalkanoates (PHA) in the Biomedical Sector—A Review" Polymers 15, no. 22: 4405. https://doi.org/10.3390/polym15224405
APA StyleDiniz, M. S. d. F., Mourão, M. M., Xavier, L. P., & Santos, A. V. (2023). Recent Biotechnological Applications of Polyhydroxyalkanoates (PHA) in the Biomedical Sector—A Review. Polymers, 15(22), 4405. https://doi.org/10.3390/polym15224405







