Influence of Ionomer and Cyanuric Acid on Antistatic, Mechanical, Thermal, and Rheological Properties of Extruded Carbon Nanotube (CNT)/Polyoxymethylene (POM) Nanocomposites
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Extrusion and Injection Processing
2.3. Characterization
2.3.1. Scanning Electron Microscopy (SEM)
2.3.2. Differential scanning calorimetry (DSC)
2.3.3. Thermogravimetric Analysis (TGA)
2.3.4. Dynamic Mechanical Analysis (DMA)
2.3.5. Tensile Test
2.3.6. Izod Impact Strength Test
2.3.7. Melt Flow Index (MFI) Test
2.3.8. Rheological Properties
2.3.9. Transmission Electron Microscopy (TEM)
2.3.10. Fourier Transform Infrared (FTIR) Spectroscopy
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bashford, D. Polyoxymethylene; Polyacetals (POM). In Thermoplastics: Directory and Databook; Bashford, D., Ed.; Springer: Dordrecht, The Netherlands, 1997; pp. 255–267. ISBN 978-94-009-1531-2. [Google Scholar]
- Khalatbari, H.; Lazoglu, I. Friction Stir Incremental Forming of Polyoxymethylene: Process Outputs, Force and Temperature. Mater. Manuf. Process. 2021, 36, 94–105. [Google Scholar] [CrossRef]
- Lin, L.; Schlarb, A.K. Investigation on Morphology and Properties of Melt Compounded Polyoxymethylene/Carbon Nanotube Composites. J. Appl. Polym. Sci. 2015, 132, 42639. [Google Scholar] [CrossRef]
- Rajan, V.V.; Wäber, R.; Wieser, J. Online Monitoring of the Thermal Degradation of POM during Melt Extrusion. J. Appl. Polym. Sci. 2010, 115, 2394–2401. [Google Scholar] [CrossRef]
- Das, A.K.; Suin, S.; Shrivastava, N.K.; Maiti, S.; Mishra, J.K.; Khatua, B.B. Effect of Nanoclay on the Morphology and Properties of Acrylonitrile Butadiene Styrene Toughened Polyoxymethylene (POM)/Clay Nanocomposites. Polym. Compos. 2014, 35, 273–282. [Google Scholar] [CrossRef]
- Williams, J.L.; Stannett, V. The Diffusion of Gases and Water Vapor through Grafted Polyoxymethylene. J. Appl. Polym. Sci. 1970, 14, 1949–1959. [Google Scholar] [CrossRef]
- Odi-Owei, S.; Schipper, D.J. Tribological Behaviour of Unfilled and Composite Polyoxymethylene. Wear 1991, 148, 363–376. [Google Scholar] [CrossRef]
- Cho, M.H.; Park, S. Micro CNC Surface Texturing on Polyoxymethylene (POM) and Its Tribological Performance in Lubricated Sliding. Tribol. Int. 2011, 44, 859–867. [Google Scholar] [CrossRef]
- Lüftl, S.; Visakh, P.M.; Chandran, S. Polyoxymethylene Handbook: Structure, Properties, Applications and Their Nanocomposites; John Wiley & Sons: Hoboken, NJ, USA, 2014; ISBN 978-1-118-91442-7. [Google Scholar]
- Pielichowska, K. Polyoxymethylene-Homopolymer/Hydroxyapatite Nanocomposites for Biomedical Applications. J. Appl. Polym. Sci. 2012, 123, 2234–2243. [Google Scholar] [CrossRef]
- Dechet, M.A.; Baumeister, I.; Schmidt, J. Development of Polyoxymethylene Particles via the Solution-Dissolution Process and Application to the Powder Bed Fusion of Polymers. Materials 2020, 13, 1535. [Google Scholar] [CrossRef] [Green Version]
- Chen, C.; Zhao, X.; Ye, L. Polyoxymethylene/Carbon Nanotube Self-Assembly Networks with Improved Electrical Conductivity for Engineering Functional Structural Materials. ACS Appl. Nano Mater. 2021, 4, 9606–9615. [Google Scholar] [CrossRef]
- Jiao, Q.; Chen, Q.; Wang, L.; Chen, H.; Li, Y. Investigation on the Crystallization Behaviors of Polyoxymethylene with a Small Amount of Ionic Liquid. Nanomaterials 2019, 9, 206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Q.; Wang, J.; Zhang, B.-Y.; Guo, B.-H.; Yu, J.; Guo, Z.-X. Improved Electrical Conductivity of Polymer/Carbon Black Composites by Simultaneous Dispersion and Interaction-Induced Network Assembly. Compos. Sci. Technol. 2019, 179, 106–114. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhu, S.; Liu, Y.; Yang, B.; Wang, X. The Mechanical and Tribological Properties of Nitric Acid-Treated Carbon Fiber-Reinforced Polyoxymethylene Composites. J. Appl. Polym. Sci. 2015, 132, 41812. [Google Scholar] [CrossRef]
- Zhao, X.; Ye, L. Study on the Thermal Conductive Polyoxymethylene/Graphite Composites. J. Appl. Polym. Sci. 2009, 111, 759–767. [Google Scholar] [CrossRef]
- Mamunya, Y.P.; Muzychenko, Y.V.; Pissis, P.; Lebedev, E.V.; Shut, M.I. Processing, Structure, and Electrical Properties of Metal-Filled Polymers. J. Macromol. Sci. Part B 2001, 40, 591–602. [Google Scholar] [CrossRef]
- Iijima, S. Helical Microtubules of Graphitic Carbon. Nature 1991, 354, 56–58. [Google Scholar] [CrossRef]
- Iijima, S.; Ichihashi, T. Single-Shell Carbon Nanotubes of 1-Nm Diameter. Nature 1993, 363, 603–605. [Google Scholar] [CrossRef]
- Dai, H.; Wong, E.W.; Lieber, C.M. Probing Electrical Transport in Nanomaterials: Conductivity of Individual Carbon Nanotubes. Science 1996, 272, 523–526. [Google Scholar] [CrossRef]
- Takakura, A.; Beppu, K.; Nishihara, T.; Fukui, A.; Kozeki, T.; Namazu, T.; Miyauchi, Y.; Itami, K. Strength of Carbon Nanotubes Depends on Their Chemical Structures. Nat. Commun. 2019, 10, 3040. [Google Scholar] [CrossRef]
- Wang, Q.; Dai, J.; Li, W.; Wei, Z.; Jiang, J. The Effects of CNT Alignment on Electrical Conductivity and Mechanical Properties of SWNT/Epoxy Nanocomposites. Compos. Sci. Technol. 2008, 68, 1644–1648. [Google Scholar] [CrossRef]
- Wang, M.S.; Wang, J.Y.; Chen, Q.; Peng, L.-M. Fabrication and Electrical and Mechanical Properties of Carbon Nanotube Interconnections. Adv. Funct. Mater. 2005, 15, 1825–1831. [Google Scholar] [CrossRef]
- Ma, P.-C.; Siddiqui, N.A.; Marom, G.; Kim, J.-K. Dispersion and Functionalization of Carbon Nanotubes for Polymer-Based Nanocomposites: A Review. Compos. Part A Appl. Sci. Manuf. 2010, 41, 1345–1367. [Google Scholar] [CrossRef]
- Tanabi, H.; Erdal, M. Effect of CNTs Dispersion on Electrical, Mechanical and Strain Sensing Properties of CNT/Epoxy Nanocomposites. Results Phys. 2019, 12, 486–503. [Google Scholar] [CrossRef]
- Naito, M.; Yokoyama, T.; Hosokawa, K.; Nogi, K. Nanoparticle Technology Handbook; Elsevier: Amsterdam, The Netherlands, 2018; ISBN 978-0-444-64111-3. [Google Scholar]
- Zhao, X.; Ye, L. Structure and Mechanical Properties of Polyoxymethylene/Multi-Walled Carbon Nanotube Composites. Compos. Part B Eng. 2011, 42, 926–933. [Google Scholar] [CrossRef]
- Tasis, D.; Tagmatarchis, N.; Bianco, A.; Prato, M. Chemistry of Carbon Nanotubes. Chem. Rev. 2006, 106, 1105–1136. [Google Scholar] [CrossRef]
- Goriparthi, B.K.; Naga Eswar Naveen, P.; Ravi Sankar, H. Performance Evaluation of Composite Gears Composed of POM, CNTs, and PTFE. Polym. Compos. 2021, 42, 1123–1134. [Google Scholar] [CrossRef]
- Stanonis, D.J.; King, W.D.; Vail, S.L. Influence of Chain Length on the Rate of Hydrolysis of Polyoxymethylene Ethers. J. Appl. Polym. Sci. 1972, 16, 1447–1456. [Google Scholar] [CrossRef]
- Stanonis, D.J.; Vail, S.L.; King, W.D. The Preparation and Acid-Catalyzed Hydrolysis of Polyoxymethylene Isopropyl Methyl Ethers. J. Appl. Polym. Sci. 1973, 17, 3061–3067. [Google Scholar] [CrossRef]
- Berardinelli, F.M.; Dolce, T.J.; Walling, C. Degradation and Stabilization of Polyacetal Copolymers. J. Appl. Polym. Sci. 1965, 9, 1419–1429. [Google Scholar] [CrossRef]
- Archodoulaki, V.-M.; Lüftl, S.; Koch, T.; Seidler, S. Property Changes in Polyoxymethylene (POM) Resulting from Processing, Ageing and Recycling. Polym. Degrad. Stab. 2007, 92, 2181–2189. [Google Scholar] [CrossRef]
- Luo, W.; Ding, Q.; Li, Y.; Zhou, S.; Zou, H.; Liang, M. Effect of Shape Morphology on Mechanical, Rheological and Tribological Properties of Polyoxymethylene/Aramid Composites. Polym. Sci. Ser. A 2015, 57, 209–220. [Google Scholar] [CrossRef]
- Paasi, J. Assessment of ESD Threats to Electronic Components. J. Electrost. 2005, 63, 589–596. [Google Scholar] [CrossRef]
- Vinson, J.E.; Liou, J.J. Electrostatic Discharge in Semiconductor Devices: Protection Techniques. Proc. IEEE 2000, 88, 1878–1902. [Google Scholar] [CrossRef]
- de Souza Vieira, L.; dos Anjos, E.G.R.; Verginio, G.E.A.; Oyama, I.C.; Braga, N.F.; da Silva, T.F.; Montagna, L.S.; Rezende, M.C.; Passador, F.R. Carbon-Based Materials as Antistatic Agents for the Production of Antistatic Packaging: A Review. J. Mater. Sci. Mater. Electron. 2021, 32, 3929–3947. [Google Scholar] [CrossRef]
- Rosner, R.B. Conductive Materials for ESD Applications: An Overview. IEEE Trans. Device Mater. Reliab. 2001, 1, 9–16. [Google Scholar] [CrossRef]
- Lee, B.-Y.; Park, S.; Chung, D.-W.; Jang, K.-S. Incorporation of Alkyl-Functionalized Silica Nanoparticles into Hydrophilic Epoxy and Hydrophobic Polystyrene Matrices. J. Appl. Polym. Sci. 2022, 139, 51828. [Google Scholar] [CrossRef]
- Xu, W.; Qin, Z.; Yu, H.; Liu, Y.; Liu, N.; Zhou, Z.; Chen, L. Cellulose Nanocrystals as Organic Nanofillers for Transparent Polycarbonate Films. J. Nanoparticle Res. 2013, 15, 1562. [Google Scholar] [CrossRef]
- Jang, K.-S. Mechanics and Rheology of Basalt Fiber-Reinforced Polycarbonate Composites. Polymer 2018, 147, 133–141. [Google Scholar] [CrossRef]
- Jang, K.-S. Mineral Filler Effect on the Mechanics and Flame Retardancy of Polycarbonate Composites: Talc and Kaolin. e-Polymers 2016, 16, 379–386. [Google Scholar] [CrossRef]
- Jang, K.-S. Low-Density Polycarbonate Composites with Robust Hollow Glass Microspheres by Tailorable Processing Variables. Polym. Test. 2020, 84, 106408. [Google Scholar] [CrossRef]
- Cole, K.S.; Cole, R.H. Dispersion and Absorption in Dielectrics I. Alternating Current Characteristics. J. Chem. Phys. 1941, 9, 341–351. [Google Scholar] [CrossRef] [Green Version]
- Chuang, H.-K.; Han, C.D. Rheological Behavior of Polymer Blends. J. Appl. Polym. Sci. 1984, 29, 2205–2229. [Google Scholar] [CrossRef]
- Han, C.D.; Kim, J.; Kim, J.K. Determination of the Order-Disorder Transition Temperature of Block Copolymers. Available online: https://pubs.acs.org/doi/pdf/10.1021/ma00191a071 (accessed on 27 March 2022).
- Shaffer, M.S.P.; Fan, X.; Windle, A.H. Dispersion and Packing of Carbon Nanotubes. Carbon 1998, 36, 1603–1612. [Google Scholar] [CrossRef]
- Ramesh, S.; Ericson, L.M.; Davis, V.A.; Saini, R.K.; Kittrell, C.; Pasquali, M.; Billups, W.E.; Adams, W.W.; Hauge, R.H.; Smalley, R.E. Dissolution of Pristine Single Walled Carbon Nanotubes in Superacids by Direct Protonation. J. Phys. Chem. B 2004, 108, 8794–8798. [Google Scholar] [CrossRef]
- Thostenson, E.T.; Li, C.; Chou, T.-W. Nanocomposites in Context. Compos. Sci. Technol. 2005, 65, 491–516. [Google Scholar] [CrossRef]
- Coleman, J.N.; Khan, U.; Blau, W.J.; Gun’ko, Y.K. Small but Strong: A Review of the Mechanical Properties of Carbon Nanotube–Polymer Composites. Carbon 2006, 44, 1624–1652. [Google Scholar] [CrossRef]
- Skákalová, V.; Kaiser, A.B.; Dettlaff-Weglikowska, U.; Hrnčariková, K.; Roth, S. Effect of Chemical Treatment on Electrical Conductivity, Infrared Absorption, and Raman Spectra of Single-Walled Carbon Nanotubes. J. Phys. Chem. B 2005, 109, 7174–7181. [Google Scholar] [CrossRef]
- Fujigaya, T.; Nakashima, N. Non-Covalent Polymer Wrapping of Carbon Nanotubes and the Role of Wrapped Polymers as Functional Dispersants. Sci. Technol. Adv. Mater. 2015, 16, 024802. [Google Scholar] [CrossRef]
- Richard, C.; Balavoine, F.; Schultz, P.; Ebbesen, T.W.; Mioskowski, C. Supramolecular Self-Assembly of Lipid Derivatives on Carbon Nanotubes. Science 2003, 300, 775–778. [Google Scholar] [CrossRef]
- Islam, M.F.; Rojas, E.; Bergey, D.M.; Johnson, A.T.; Yodh, A.G. High Weight Fraction Surfactant Solubilization of Single-Wall Carbon Nanotubes in Water. Nano Lett. 2003, 3, 269–273. [Google Scholar] [CrossRef]
- Ishibashi, A.; Nakashima, N. Individual Dissolution of Single-Walled Carbon Nanotubes in Aqueous Solutions of Steroid or Sugar Compounds and Their Raman and Near-IR Spectral Properties. Chem.—Eur. J. 2006, 12, 7595–7602. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Hong, S.; Jung, S.; Strano, M.S.; Choi, J.; Baik, S. Dielectrophoresis of Surface Conductance Modulated Single-Walled Carbon Nanotubes Using Catanionic Surfactants. J. Phys. Chem. B 2006, 110, 1541–1545. [Google Scholar] [CrossRef] [PubMed]
- Romero, D.B.; Carrard, M.; De Heer, W.; Zuppiroli, L. A Carbon Nanotube/Organic Semiconducting Polymer Heterojunction. Adv. Mater. 1996, 8, 899–902. [Google Scholar] [CrossRef]
- Shigeta, M.; Komatsu, M.; Nakashima, N. Individual Solubilization of Single-Walled Carbon Nanotubes Using Totally Aromatic Polyimide. Chem. Phys. Lett. 2006, 418, 115–118. [Google Scholar] [CrossRef]
- Baskaran, D.; Mays, J.W.; Bratcher, M.S. Noncovalent and Nonspecific Molecular Interactions of Polymers with Multiwalled Carbon Nanotubes. Chem. Mater. 2005, 17, 3389–3397. [Google Scholar] [CrossRef] [Green Version]
- Yang, D.-Q.; Rochette, J.-F.; Sacher, E. Spectroscopic Evidence for Π−π Interaction between Poly(Diallyl Dimethylammonium) Chloride and Multiwalled Carbon Nanotubes. J. Phys. Chem. B 2005, 109, 4481–4484. [Google Scholar] [CrossRef]
- Sánchez-Soto, M.; Illescas, S.; Milliman, H.; Schiraldi, D.A.; Arostegui, A. Morphology and Thermomechanical Properties of Melt-Mixed Polyoxymethylene/Polyhedral Oligomeric Silsesquioxane Nanocomposites. Macromol. Mater. Eng. 2010, 295, 846–858. [Google Scholar] [CrossRef]
- Park, S.H.; Bandaru, P.R. Improved Mechanical Properties of Carbon Nanotube/Polymer Composites through the Use of Carboxyl-Epoxide Functional Group Linkages. Polymer 2010, 51, 5071–5077. [Google Scholar] [CrossRef]
- Cha, J.; Jun, G.H.; Park, J.K.; Kim, J.C.; Ryu, H.J.; Hong, S.H. Improvement of Modulus, Strength and Fracture Toughness of CNT/Epoxy Nanocomposites through the Functionalization of Carbon Nanotubes. Compos. Part B Eng. 2017, 129, 169–179. [Google Scholar] [CrossRef]
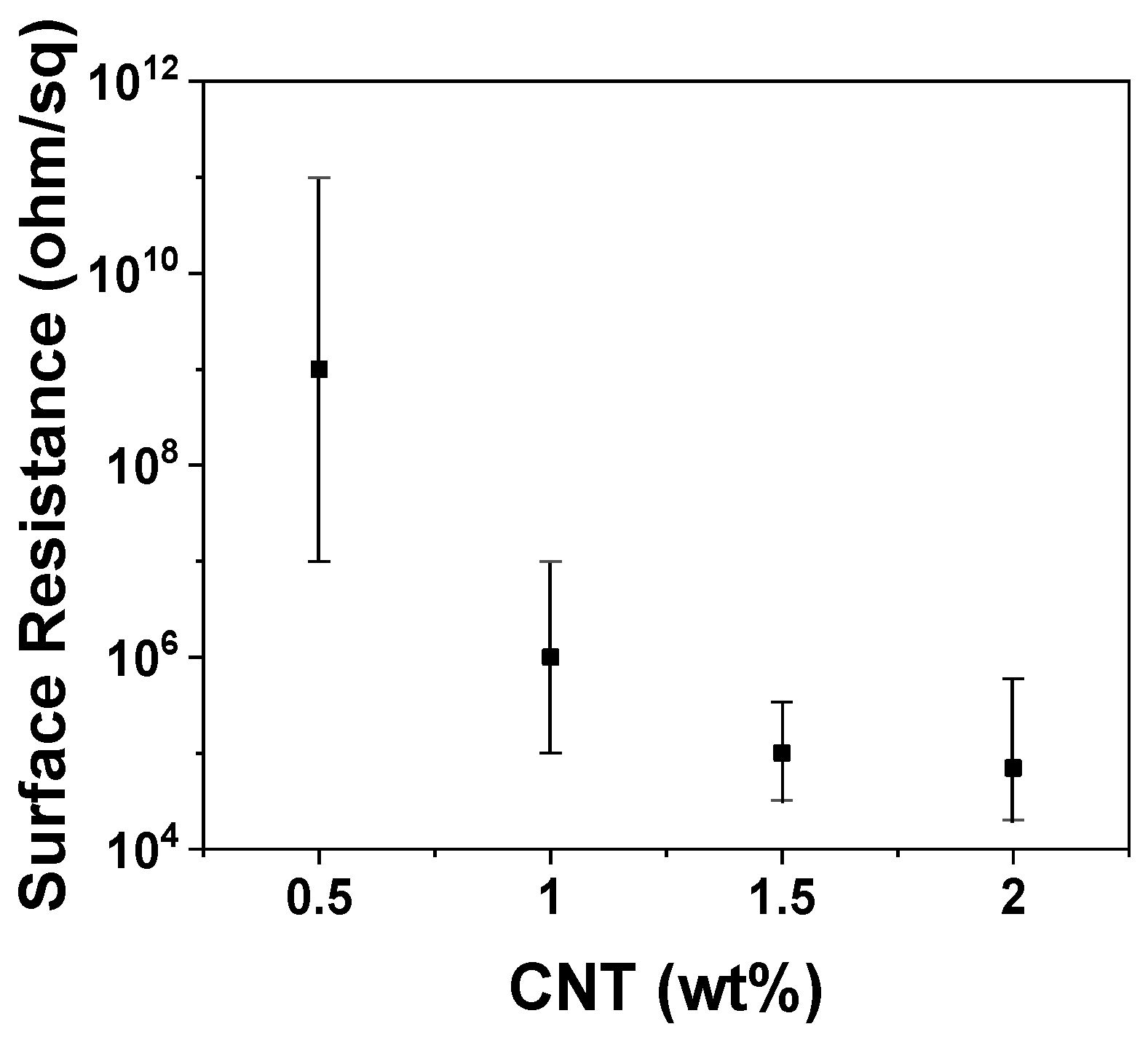













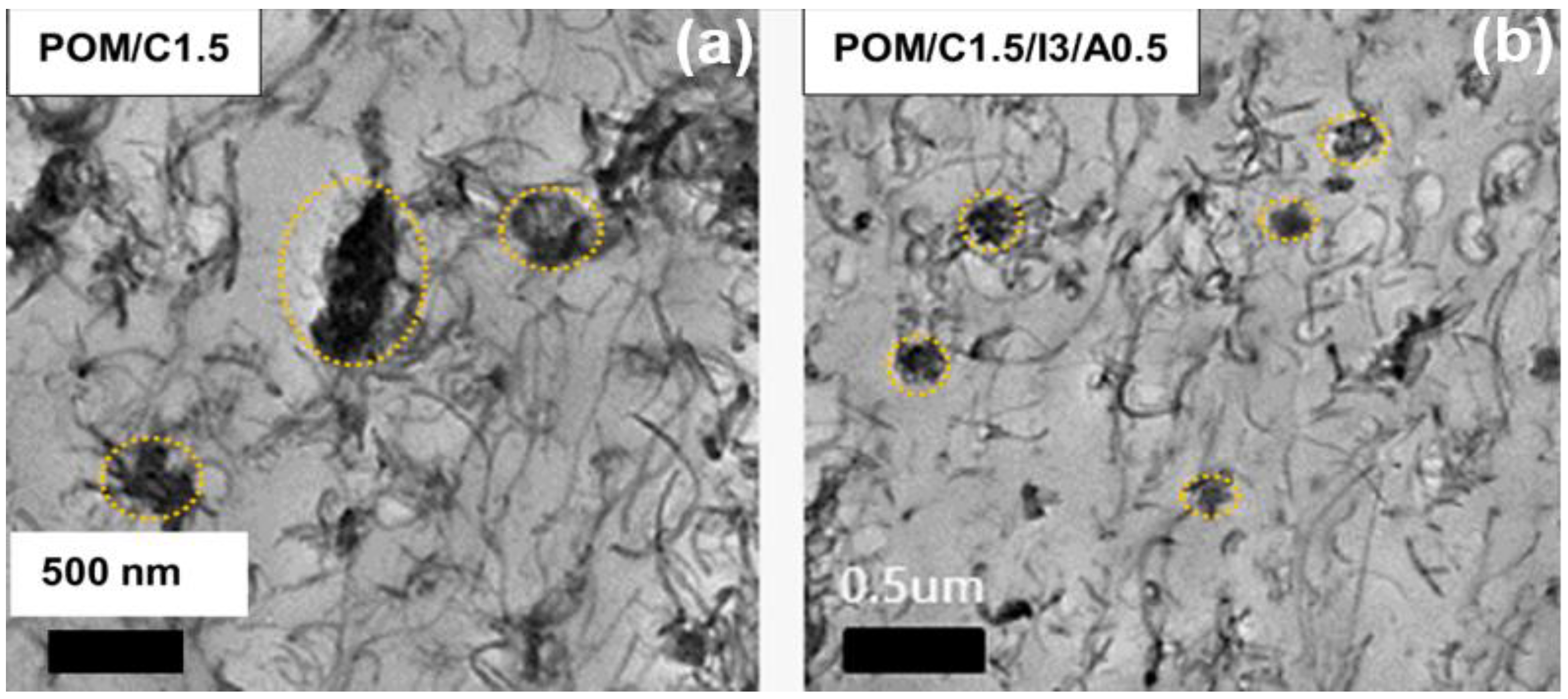
| CNT (wt%) | 0 | 0.5 | 1.0 | 1.5 | 2.0 |
|---|---|---|---|---|---|
| Tm (°C) | 165.4 | 167.0 | 165.7 | 167.8 | 168.6 |
| ΔHm (J/g) | 159.9 | 157.5 | 159.4 | 161.4 | 170.8 |
| Tc (°C) | 137.2 | 141.0 | 142.8 | 141.1 | 140.9 |
| ΔHc (J/g) | 153.9 | 153.6 | 148.2 | 153.0 | 159.6 |
| χc (%) * | 63.5 | 62.2 | 62.7 | 63.2 | 66.5 |
| Td (°C) | 287.3 | 287.2 | 287.8 | 267.8 | 266.1 |
| CNT (wt%) | 0.0 | 0.5 | 1.0 | 1.5 | 2.0 | |
|---|---|---|---|---|---|---|
| Tβ (°C) | G” | −30.6 | −4.3 | −30.1 | −22.1 | −22.5 |
| Tan δ | −11.7 | 1.5 | −9.6 | −8.5 | −13.2 | |
| Tα (°C) | G” | 93.3 | 97.8 | 100.5 | 99.9 | 98.3 |
| Tan δ | 114.8 | 120.8 | 123.1 | 125.7 | 125.1 | |
| Content (wt%) * | 0.0 | 0.5 | 1.0 | 2.0 | 3.0 | 5.0 | |
|---|---|---|---|---|---|---|---|
| Tm (°C) | Ionomer | 167.7 | 166.6 | 168.0 | 167.2 | 166.1 | 166.3 |
| Cyanuric acid | 169.3 | 167.8 | 167.0 | 167.6 | 168.1 | ||
| ∆Hm (J/g) | Ionomer | 161.4 | 168.7 | 181.3 | 164.2 | 159.7 | 181.0 |
| Cyanuric acid | 157.4 | 152.1 | 182.2 | 178.7 | 160.5 | ||
| Tc (°C) | Ionomer | 141.1 | 142.2 | 142.83 | 140.2 | 148.0 | 143.1 |
| Cyanuric acid | 136.6 | 139.3 | 137.2 | 139.7 | 133.6 | ||
| ∆Hc (J/g) | Ionomer | 153.0 | 155.9 | 175.5 | 152.6 | 143.5 | 163.4 |
| Cyanuric acid | 149.8 | 138.4 | 169.6 | 166.0 | 151,1 | ||
| χc (%) * | Ionomer | 63.2 | 65.6 | 70.2 | 62.9 | 60.6 | 67.2 |
| Cyanuric acid | 61.3 | 58.9 | 69.8 | 67.8 | 59.6 | ||
| Td (°C) | Ionomer | 267.8 | 285.7 | 280.1 | 279.9 | 275.7 | 284.2 |
| Cyanuric acid | 296.9 | 293.7 | 289.7 | 250.7 | 253.6 | ||
| Content (wt%) | None | C1.5 | C1.5 I3.0 | C1.5 A0.5 | C1.5 I3.0 A0.5 |
|---|---|---|---|---|---|
| Tm (°C) | 165.4 | 167.7 | 166.2 | 169.3 | 170.2 |
| ∆Hm (J/g) | 159.9 | 161.4 | 159.7 | 157.4 | 149.8 |
| Tc (℃) | 137.2 | 141.1 | 148.0 | 136.6 | 150.3 |
| ∆Hc (J/g) | 153.9 | 153.0 | 143.5 | 149.8 | 137.4 |
| χc (%) * | 63.5 | 63.2 | 60.6 | 61.3 | 56.5 |
| Td (°C) | 287.3 | 267.8 | 275.7 | 296.9 | 281.8 |
| Contents (wt%) | 0.0 | 0.5 | 1.0 | 2.0 | 3.0 | 5.0 | ||
|---|---|---|---|---|---|---|---|---|
| Tβ (°C) | G” | Ionomer | −22.1 | −21.3 | −33.5 | −10.9 | −14.3 | −28.5 |
| Cyanuric acid | −22.1 | −24.9 | −12.8 | −5.6 | −0.43 | 0.6 | ||
| Tan δ | Ionomer | −8.8 | −8.4 | −12.5 | −0.1 | 1.2 | −5.2 | |
| Cyanuric acid | −8.8 | −4.1 | 1.3 | 3.7 | 4.7 | 9.5 | ||
| Tα (°C) | G” | Ionomer | 99.9 | 100.0 | 100.7 | 97.9 | 97.3 | 95.5 |
| Cyanuric acid | 99.9 | 99.2 | 99.7 | 97.3 | 97.8 | 96.4 | ||
| Tan δ | Ionomer | 125.7 | 120.3 | 123.1 | 123.6 | 121.2 | 122.3 | |
| Cyanuric acid | 125.7 | 120.2 | 123.0 | 119.3 | 119.4 | 118.9 | ||
| Contents (wt%) | None | C1.5 | C1.5 I3.0 | C1.5 A0.5 | C1.5 I3.0 A0.5 | |
|---|---|---|---|---|---|---|
| Tβ (°C) | G” | −30.6 | −22.1 | −14.3 | −24.9 | −7.4 |
| Tan δ | −11.7 | −8.5 | 1.2 | −4.1 | −11.7 | |
| Tα (°C) | G” | 93.3 | 99.9 | 98.0 | 100.0 | 97.5 |
| Tan δ | 114.8 | 125.7 | 123.1 | 125.7 | 122.0 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yun, S.-S.; Shin, D.-h.; Jang, K.-S. Influence of Ionomer and Cyanuric Acid on Antistatic, Mechanical, Thermal, and Rheological Properties of Extruded Carbon Nanotube (CNT)/Polyoxymethylene (POM) Nanocomposites. Polymers 2022, 14, 1849. https://doi.org/10.3390/polym14091849
Yun S-S, Shin D-h, Jang K-S. Influence of Ionomer and Cyanuric Acid on Antistatic, Mechanical, Thermal, and Rheological Properties of Extruded Carbon Nanotube (CNT)/Polyoxymethylene (POM) Nanocomposites. Polymers. 2022; 14(9):1849. https://doi.org/10.3390/polym14091849
Chicago/Turabian StyleYun, Sang-Seok, Dong-hyeok Shin, and Keon-Soo Jang. 2022. "Influence of Ionomer and Cyanuric Acid on Antistatic, Mechanical, Thermal, and Rheological Properties of Extruded Carbon Nanotube (CNT)/Polyoxymethylene (POM) Nanocomposites" Polymers 14, no. 9: 1849. https://doi.org/10.3390/polym14091849
APA StyleYun, S.-S., Shin, D.-h., & Jang, K.-S. (2022). Influence of Ionomer and Cyanuric Acid on Antistatic, Mechanical, Thermal, and Rheological Properties of Extruded Carbon Nanotube (CNT)/Polyoxymethylene (POM) Nanocomposites. Polymers, 14(9), 1849. https://doi.org/10.3390/polym14091849







