Polymer Composites with Quantum Dots as Potential Electrode Materials for Supercapacitors Application: A Review
Abstract
:1. Introduction
2. Importance of Various QDs and Polymers as Electrode Material in Supercapacitors
3. Synthesis of Quantum Dots, Polymer, and Nanocomposites
3.1. Electrochemical Process
3.2. Solvothermal/Hydrothermal Process
3.3. Microwave Synthesis
3.4. Direct Chemical Cutting Process
3.5. Hummers Method
4. Quantum Dots and Polymer Composites in Supercapacitor Applications
5. Electrolytes Used for Polymer Composite QDs Electrodes for Supercapacitor
6. Discussing Pros and Cons on Polymer Composite QDs as Electrode in Supercapacitors
7. Future Perspective of Polymer Composite QDs Electrodes in Supercapacitors
8. Applications of Biomass Derived Materials and Plastic Materials for Energy Storage Devices
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Balaji, T.E.; Das, H.T.; Maiyalagan, T. Recent Trends in Bimetallic Oxides and Their Composites as Electrode Materials for Supercapacitor Applications. ChemElectroChem 2021, 8, 1723–1746. [Google Scholar] [CrossRef]
- Uddin, S.; Das, H.T.; Maiyalagan, T.; Elumalai, P. Influence of designed electrode surfaces on double layer capacitance in aqueous electrolyte: Insights from standard models. Appl. Surf. Sci. 2018, 449, 445–453. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, L.; Su, H.; Huang, W.; Dong, X. Binary metal oxide: Advanced energy storage materials in supercapacitors. J. Mater. Chem. A 2014, 8, 43–59. [Google Scholar] [CrossRef]
- Das, H.T.; Mahendraprabhu, K.; Maiyalagan, T.; Elumalai, P. Performance of Solid-state Hybrid Energy-storage Device using Reduced Graphene-oxide Anchored Sol-gel Derived Ni/NiO Nanocomposite. Sci. Rep. 2017, 7, 15342. [Google Scholar] [CrossRef] [Green Version]
- Barai, H.R.; Banerjee, A.N.; Joo, S.W. Improved electrochemical properties of highly porous amorphous manganese oxide nanoparticles with crystalline edges for superior supercapacitors. J. Ind. Eng. Chem. 2017, 56, 212–224. [Google Scholar] [CrossRef]
- Kiran, S.K.; Padmini, M.; Das, H.T.; Elumalai, P. Performance of asymmetric supercapacitor using CoCr-layered double hydroxide and reduced graphene-oxide. J. Solid State Electrochem. 2017, 21, 927–938. [Google Scholar] [CrossRef]
- Duraisamy, E.; Das, H.T.; Sharma, A.S.; Elumalai, P. Supercapacitor and photocatalytic performances of hydrothermally-derived Co3O4/CoO@carbon nanocomposite. New J. Chem. 2018, 42, 6114–6124. [Google Scholar] [CrossRef]
- Al-Enizi, A.M.; Ubaidullah, M.; Ahmed, J.; Ahamad, T.; Ahmad, T.; Shaikh, S.F.; Naushad, M. Synthesis of NiOx@NPC composite for high-performance supercapacitor via waste PET plastic-derived Ni-MOF. Compos. Part B Eng. 2020, 183, 107655. [Google Scholar] [CrossRef]
- Saini, S.; Chand, P.; Joshi, A. Biomass derived carbon for supercapacitor applications: Review. J. Energy Storage 2021, 39, 102646. [Google Scholar] [CrossRef]
- Shaker, M.; Riahifar, R.; Li, Y. A review on the superb contribution of carbon and graphene quantum dots to electrochemical capacitors’ performance: Synthesis and application. Flat. Chem. 2020, 22, 100171. [Google Scholar] [CrossRef]
- Das, H.T.; Saravanya, S.; Elumalai, P. Disposed Dry Cells as Sustainable Source for Generation of Few Layers of Graphene and Manganese Oxide for Solid-State Symmetric and Asymmetric Supercapacitor Applications. ChemistrySelect 2018, 3, 13275–13283. [Google Scholar] [CrossRef]
- Panchu, S.J.; Raju, K.; Swart, H.C.; Chokkalingam, B.; Maaza, M.; Henini, M.; Moodley, M.K. Luminescent MoS2 Quantum Dots with Tunable Operating Potential for Energy-Enhanced Aqueous Supercapacitors. ACS Omega 2021, 6, 4542–4550. [Google Scholar] [CrossRef] [PubMed]
- Das, H.T.; Elango Balaji, T.; Mahendraprabhu, K.; Vinoth, S. Cost-Effective Nanomaterials Fabricated by Recycling Spent Batteries. In Topics in Mining, Metallurgy and Materials Engineering; Springer: Berlin/Heidelberg, Germany, 2021; pp. 147–174. [Google Scholar] [CrossRef]
- Suriyakumar, S.; Bhardwaj, P.; Grace, A.N.; Stephan, A.M. Role of Polymers in Enhancing the Performance of Electrochemical Supercapacitors: A Review. Batter. Supercaps 2021, 4, 571–584. [Google Scholar] [CrossRef]
- Shah, S.S.; Das, H.T.; Barai, H.R.; Aziz, A. Boosting the Electrochemical Performance of Polyaniline by One-Step Electrochemical Deposition on Nickel Foam for High-Performance Asymmetric Supercapacitor. Polymers 2022, 14, 270. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, V.; Prasad, K.H.; Das, H.T.; Ganapathy, K.; Nallani, S.; Maiyalagan, T. Novel Dispersion of 1D Nanofiber Fillers for Fast Ion-Conducting Nanocomposite Polymer Blend Quasi-Solid Electrolytes for Dye-Sensitized Solar Cells. ACS Omega 2022, 7, 1658–1670. [Google Scholar] [CrossRef]
- Barai, H.R.; Banerjee, A.N.; Hamnabard, N.; Joo, S.W. Synthesis of amorphous manganese oxide nanoparticles-to-crystalline nanorods through a simple wet-chemical technique using K+ ions as a ‘growth director’ and their morphology-controlled high performance supercapacitor applications. RSC Adv. 2016, 6, 78887–78908. [Google Scholar] [CrossRef]
- Kumar, S.; Saeed, G.; Zhu, L.; Hui, K.N.; Kim, N.H.; Lee, J.H. 0D to 3D carbon-based networks combined with pseudocapacitive electrode material for high energy density supercapacitor: A review. Chem. Eng. J. 2021, 403, 126352. [Google Scholar] [CrossRef]
- Aphale, A.; Maisuria, K.; Mahapatra, M.K.; Santiago, A.; Singh, P.; Patra, P. Hybrid Electrodes by In-Situ Integration of Graphene and Carbon-Nanotubes in Polypyrrole for Supercapacitors. Sci. Rep. 2015, 5, 14445. [Google Scholar] [CrossRef] [Green Version]
- Forouzandeh, P.; Kumaravel, V.; Pillai, S.C. Electrode Materials for Supercapacitors: A Review of Recent Advances. Catalysts 2020, 10, 969. [Google Scholar] [CrossRef]
- Barai, H.R.; Rahman, M.; Joo, S.W. Annealing-Free Synthesis of K-doped Mixed-Phase TiO2 Nanofibers on Ti Foil for Electrochemical Supercapacitor. Electrochim. Acta 2017, 253, 563–571. [Google Scholar] [CrossRef]
- Conway, B.E. Transition from “supercapacitor” to “battery” behavior in electrochemical energy storage. In Proceedings of the International Power Sources Symposium, Cherry Hill, NJ, USA, 25–28 June 1990; IEEE: Piscataway, NJ, USA, 1991; Volume 138, pp. 319–327. [Google Scholar] [CrossRef]
- Barai, H.R.; Banerjee, A.; Bai, F.; Joo, S.W. Surface modification of titania nanotube arrays with crystalline manganese-oxide nanostructures and fabrication of hybrid electrochemical electrode for high-performance supercapacitors. J. Ind. Eng. Chem. 2018, 62, 409–417. [Google Scholar] [CrossRef]
- Barai, H.R.; Lopa, N.S.; Ahmed, F.; Khan, N.A.; Ansari, S.A.; Joo, S.W.; Rahman, M. Synthesis of Cu-Doped Mn3O4@Mn-Doped CuO Nanostructured Electrode Materials by a Solution Process for High-Performance Electrochemical Pseudocapacitors. ACS Omega 2020, 5, 22356–22366. [Google Scholar] [CrossRef] [PubMed]
- Barai, H.R.; Barai, P.; Roy, M.; Joo, S.W. Solid-State Synthesis of Titanium-Doped Binary Strontium–Copper Oxide as a High-Performance Electrochemical Pseudocapacitive Electrode Nanomaterial. Energy Fuels 2021, 35, 16870–16881. [Google Scholar] [CrossRef]
- Barai, H.R.; Rahman, M.M.; Adeel, M.; Joo, S.W. MnSn(OH)6 derived Mn2SnO4@Mn2O3 composites as electrode materials for high-performance Supercapacitors. Mater. Res. Bull. 2022, 148, 111678. [Google Scholar] [CrossRef]
- Najib, S.; Erdem, E. Current progress achieved in novel materials for supercapacitor electrodes: Mini review. Nanoscale Adv. 2019, 1, 2817–2827. [Google Scholar] [CrossRef] [Green Version]
- Barai, H.R.; Rahman, M.; Joo, S.W. Template-free synthesis of two-dimensional titania/titanate nanosheets as electrodes for high-performance supercapacitor applications. J. Power Sources 2017, 372, 227–234. [Google Scholar] [CrossRef]
- Ujjain, S.K. Applications of Quantum Dots in Supercapacitors. Mater. Res. Found. 2021, 96, 169–190. [Google Scholar] [CrossRef]
- Li, M.; Chen, T.; Gooding, J.J.; Liu, J. Review of carbon and graphene quantum dots for sensing. ACS Sens. 2019, 4, 1732–1748. [Google Scholar] [CrossRef]
- Tian, P.; Tang, L.; Teng, K.; Lau, S. Graphene quantum dots from chemistry to applications. Mater. Today Chem. 2018, 10, 221–258. [Google Scholar] [CrossRef]
- Zvaigzne, M.; Domanina, I.; Il’gach, D.; Yakimansky, A.; Nabiev, I.; Samokhvalov, P. Quantum Dot–Polyfluorene Composites for White-Light-Emitting Quantum Dot-Based LEDs. Nanomaterials 2020, 10, 2487. [Google Scholar] [CrossRef]
- Watson, B.W.; Meng, L.; Fetrow, C.; Qin, Y. Core/Shell Conjugated Polymer/Quantum Dot Composite Nanofibers through Orthogonal Non-Covalent Interactions. Polymers 2016, 8, 408. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gordillo, H.; Suárez, I.; Abargues, R.; Rodríguez-Cantó, P.; Albert, S.; Martínez-Pastor, J.P. Polymer/QDs Nanocomposites for Waveguiding Applications. J. Nanomater. 2012, 2012, 1–9. [Google Scholar] [CrossRef]
- Hu, C.; Li, M.; Qiu, J.; Sun, Y.P. Design and fabrication of carbon dots for energy conversion and storage. Chem. Soc. Rev. 2019, 48, 2315–2337. [Google Scholar] [CrossRef]
- Mittal, S.S.; Ramadas, G.; Vasanthmurali, N.; Madaneshwar, V.S.; Kumar, M.S.; Kothurkar, N.K. Carbon Quantum Dot-Polypyrrole Nanocomposite for Supercapacitor Electrodes. IOP Conf. Ser. Mater. Sci. Eng. 2019, 577, 012194. [Google Scholar] [CrossRef]
- Arthisree, D.; Madhuri, W. Optically active polymer nanocomposite composed of polyaniline, polyacrylonitrile and green-synthesized graphene quantum dot for supercapacitor application. Int. J. Hydrogen Energy 2020, 45, 9317–9327. [Google Scholar] [CrossRef]
- Li, H.; He, X.; Kang, Z.; Huang, H.; Liu, Y.; Liu, J.; Lian, S.; Tsang, A.C.H.; Yang, X.; Lee, S.-T. Water-Soluble Fluorescent Carbon Quantum Dots and Photocatalyst Design. Angew. Chem. Int. Ed. 2010, 49, 4430–4434. [Google Scholar] [CrossRef]
- Li, Y.; Hu, Y.; Zhao, Y.; Shi, G.; Deng, L.; Hou, Y.; Qu, L. An Electrochemical Avenue to Green-Luminescent Graphene Quantum Dots as Potential Electron-Acceptors for Photovoltaics. Adv. Mater. 2011, 23, 776–780. [Google Scholar] [CrossRef]
- Li, X.; Rui, M.; Song, J.; Shen, Z.; Zeng, H. Carbon and Graphene Quantum Dots for Optoelectronic and Energy Devices: A Review. Adv. Funct. Mater. 2015, 25, 4929–4947. [Google Scholar] [CrossRef]
- Li, Y.; Zhao, Y.; Cheng, H.; Hu, Y.; Shi, G.; Dai, L.; Qu, L. Nitrogen-Doped Graphene Quantum Dots with Oxygen-Rich Functional Groups. J. Am. Chem. Soc. 2012, 134, 15–18. [Google Scholar] [CrossRef]
- Jing, M.; Wang, C.; Hou, H.; Wu, Z.; Zhu, Y.; Yang, Y.; Jia, X.; Zhang, Y.; Ji, X. Ultrafine nickel oxide quantum dots enbedded with few-layer exfoliative graphene for an asymmetric supercapacitor: Enhanced capacitances by alternating voltage. J. Power Sources 2015, 298, 241–248. [Google Scholar] [CrossRef]
- Yin, W.; Liu, X.; Zhang, X.; Gao, X.; Colvin, V.L.; Zhang, Y.; Yu, W.W. Synthesis of Tungsten Disulfide and Molybdenum Disulfide Quantum Dots and Their Applications. Chem. Mater. 2020, 32, 4409–4424. [Google Scholar] [CrossRef]
- Valappil, M.O.; Anil, A.; Shaijumon, M.; Pillai, V.K.; Alwarappan, S. A Single-Step Electrochemical Synthesis of Luminescent WS2 Quantum Dots. Chem. Eur. J. 2017, 23, 9144–9148. [Google Scholar] [CrossRef] [PubMed]
- Gopalakrishnan, D.; Damien, D.; Li, B.; Gullappalli, H.; Pillai, V.K.; Ajayan, P.M.; Shaijumon, M.M. Electrochemical synthesis of luminescent MoS2 quantum dots. Chem. Commun. 2015, 51, 6293–6296. [Google Scholar] [CrossRef] [PubMed]
- Anantharaj, S.; Valappil, M.O.; Karthick, K.; Pillai, V.K.; Alwarappan, S.; Kundu, S. Electrochemically chopped WS2 quantum dots as an efficient and stable electrocatalyst for water reduction. Catal. Sci. Technol. 2019, 9, 223–231. [Google Scholar] [CrossRef]
- Zhao, Z.; Xie, Y. Enhanced electrochemical performance of carbon quantum dots-polyaniline hybrid. J. Power Sources 2017, 337, 54–64. [Google Scholar] [CrossRef]
- Vandana, M.; Vijeth, H.; Ashokkumar, S.; Devendrappa, H. Hydrothermal synthesis of quantum dots dispersed on conjugated polymer as an efficient electrodes for highly stable hybrid supercapacitors. Inorg. Chem. Commun. 2020, 117, 107941. [Google Scholar] [CrossRef]
- Shi, X.; Yin, Z.-Z.; Xu, J.; Li, S.; Wang, C.; Wang, B.; Qin, Y.; Kong, Y. Preparation, characterization and the supercapacitive behaviors of electrochemically reduced graphene quantum dots/polypyrrole hybrids. Electrochim. Acta 2021, 385, 138435. [Google Scholar] [CrossRef]
- Li, L.; Li, M.; Liang, J.; Yang, X.; Luo, M.; Ji, L.; Guo, Y.; Zhang, H.; Tang, N.; Wang, X. Preparation of Core–Shell CQD@PANI Nanoparticles and Their Electrochemical Properties. ACS Appl. Mater. Interfaces 2019, 11, 22621–22627. [Google Scholar] [CrossRef]
- Alves, A.P.P.; Koizumi, R.; Samanta, A.; Machado, L.D.; Singh, A.K.; Galvao, D.S.; Silva, G.G.; Tiwary, C.S.; Ajayan, P.M. One-step electrodeposited 3D-ternary composite of zirconia nanoparticles, rGO and polypyrrole with enhanced supercapacitor performance. Nano Energy 2017, 31, 225–232. [Google Scholar] [CrossRef] [Green Version]
- Vasudevan, D.; Gaddam, R.R.; Trinchi, A.; Cole, I. Core–shell quantum dots: Properties and applications. J. Alloys Compd. 2015, 636, 395–404. [Google Scholar] [CrossRef]
- Ju, J.; Chen, W. Synthesis of highly fluorescent nitrogen-doped graphene quantum dots for sensitive, label-free detection of Fe (III) in aqueous media. Biosens. Bioelectron. 2014, 58, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarty, N.; Dey, A.; Krishnamurthy, S.; Chakraborty, A.K. CeO2/Ce2O3 quantum dot decorated reduced graphene oxide nanohybrid as electrode for supercapacitor. Appl. Surf. Sci. 2021, 536, 147960. [Google Scholar] [CrossRef]
- Duan, H.; Wang, T.; Wu, X.; Su, Z.; Zhuang, J.; Liu, S.; Zhu, R.; Chen, C.; Pang, H. CeO2 quantum dots doped Ni-Co hydroxide nanosheets for ultrahigh energy density asymmetric supercapacitors. Chin. Chem. Lett. 2020, 31, 2330–2332. [Google Scholar] [CrossRef]
- Oskueyan, G.; Lakouraj, M.M.; Mahyari, M. Nitrogen and sulfur Co-Doped graphene quantum dots decorated CeO2 nanoparticles/ polyaniline: As high efficient hybrid supercapacitor electrode materials. Electrochim. Acta 2019, 299, 125–131. [Google Scholar] [CrossRef]
- Ko, Y.; Shim, J.; Lee, C.-H.; Lee, K.S.; Cho, H.; Lee, K.-T.; Son, D.I. Synthesis and characterization of CuO/graphene (Core/shell) quantum dots for electrochemical applications. Mater. Lett. 2018, 217, 113–116. [Google Scholar] [CrossRef]
- Ghosh, K.; Srivastava, S.K. Enhanced Supercapacitor Performance and Electromagnetic Interference Shielding Effectiveness of CuS Quantum Dots Grown on Reduced Graphene Oxide Sheets. ACS Omega 2021, 6, 4582–4596. [Google Scholar] [CrossRef]
- Siwatch, P.; Sharma, K.; Tripathi, S. Facile synthesis of NiCo2O4 quantum dots for asymmetric supercapacitor. Electrochim. Acta 2020, 329, 135084. [Google Scholar] [CrossRef]
- Geng, J.; Ma, C.; Zhang, D.; Ning, X. Facile and fast synthesis of SnO2 quantum dots for high performance solid-state asymmetric supercapacitor. J. Alloys Compd. 2020, 825, 153850. [Google Scholar] [CrossRef]
- Guo, X.; Wang, Y.; Wu, F.; Ni, Y.; Kokot, S. The use of tungsten disulfide dots as highly selective, fluorescent probes for analysis of nitrofurazone. Talanta 2015, 144, 1036–1043. [Google Scholar] [CrossRef]
- Umrao, S.; Jang, M.-H.; Oh, J.-H.; Kim, G.; Sahoo, S.; Cho, Y.-H.; Srivastva, A.; Oh, I. Microwave bottom-up route for size-tunable and switchable photoluminescent graphene quantum dots using acetylacetone: New platform for enzyme-free detection of hydrogen peroxide. Carbon 2015, 81, 514–524. [Google Scholar] [CrossRef]
- Zheng, B.; Chen, Y.; Li, P.; Wang, Z.; Cao, B.; Bingqiang, C.; Liu, J.; Qiu, Z.; Zhang, W. Ultrafast ammonia-driven, microwave-assisted synthesis of nitrogen-doped graphene quantum dots and their optical properties. Nanophotonics 2017, 6, 259–267. [Google Scholar] [CrossRef]
- Dutta, S.; Jaiswal, K.K.; Verma, R.; Basavaraju, D.M.; Ramaswamy, A.P. Green synthesis of zinc oxide catalyst under microwave irradiation using banana (Musa spp.) corm (rhizome) extract for biodiesel synthesis from fish waste lipid. Biocatal. Agric. Biotechnol. 2019, 22, 101390. [Google Scholar] [CrossRef]
- Zhang, C.; Cui, Y.; Song, L.; Liu, X.; Hu, Z. Microwave assisted one-pot synthesis of graphene quantum dots as highly sensitive fluorescent probes for detection of iron ions and pH value. Talanta 2016, 150, 54–60. [Google Scholar] [CrossRef]
- Kumar, S.; Aziz, S.T.; Girshevitz, O.; Nessim, G.D. One-Step Synthesis of N-Doped Graphene Quantum Dots from Chitosan as a Sole Precursor Using Chemical Vapor Deposition. J. Phys. Chem. C 2018, 122, 2343–2349. [Google Scholar] [CrossRef]
- Zhuo, S.; Shao, M.; Lee, S.-T. Upconversion and Downconversion Fluorescent Graphene Quantum Dots: Ultrasonic Preparation and Photocatalysis. ACS Nano 2012, 6, 1059–1064. [Google Scholar] [CrossRef]
- Fresco-Cala, B.; Soriano, M.L.; Sciortino, A.; Cannas, M.; Messina, F.; Cardenas, S. One-pot synthesis of graphene quantum dots and simultaneous nanostructured self-assembly via a novel microwave-assisted method: Impact on triazine removal and efficiency monitoring. RSC Adv. 2018, 8, 29939–29946. [Google Scholar] [CrossRef] [Green Version]
- Cheng, C.; Shi, Y.; Li, M.; Xing, M.; Wu, Q. Carbon quantum dots from carbonized walnut shells: Structural evolution, fluorescence characteristics, and intracellular bioimaging. Mater. Sci. Eng. C 2017, 79, 473–480. [Google Scholar] [CrossRef]
- Zhou, J.; Liu, Y.; Tang, J.; Tang, W. Surface ligands engineering of semiconductor quantum dots for chemosensory and biological applications. Mater. Today 2017, 20, 360–376. [Google Scholar] [CrossRef]
- Kim, S.; Hwang, S.W.; Kim, M.-K.; Shin, D.Y.; Kim, C.O.; Yang, S.B.; Park, J.H.; Hwang, E.; Choi, S.-H.; Ko, G.; et al. Anomalous Behaviors of Visible Luminescence from Graphene Quantum Dots: Interplay between Size and Shape. ACS Nano 2012, 6, 8203–8208. [Google Scholar] [CrossRef]
- Shen, J.; Zhu, Y.; Yang, X.; Zong, J.; Zhang, J.; Li, C. One-pot hydrothermal synthesis of graphenequantum dots surface-passivated by polyethylene glycol and their photoelectric conversion under near-infrared light. New J. Chem. 2012, 36, 97–101. [Google Scholar] [CrossRef]
- Li, M.; Wu, W.; Ren, W.; Cheng, H.-M.; Tang, N.; Zhong, W.; Du, Y. Synthesis and upconversion luminescence of N-doped graphene quantum dots. Appl. Phys. Lett. 2012, 101, 103107. [Google Scholar] [CrossRef]
- Fei, H.; Ye, R.; Ye, G.; Gong, Y.; Peng, Z.; Fan, X.; Samuel, E.L.G.; Ajayan, P.M.; Tour, J.M. Boron- and Nitrogen-Doped Graphene Quantum Dots/Graphene Hybrid Nanoplatelets as Efficient Electrocatalysts for Oxygen Reduction. ACS Nano 2014, 8, 10837–10843. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wang, P.; Fernando, K.A.S.; Lecroy, G.E.; Maimaiti, H.; Harruff-Miller, B.A.; Lewis, W.K.; Bunker, C.E.; Hou, Z.L.; Sun, Y.P. Enhanced fluorescence properties of carbon dots in polymer films. J. Mater. Chem. C 2016, 4, 6967–6974. [Google Scholar] [CrossRef] [Green Version]
- Ouyang, Z.; Lei, Y.; Chen, Y.; Zhang, Z.; Jiang, Z.; Hu, J.; Lin, Y. Preparation and Specific Capacitance Properties of Sulfur, Nitrogen Co-Doped Graphene Quantum Dots. Nanoscale Res. Lett. 2019, 14, 219. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, J.; Liu, J.; Wu, J.; Chen, H.; Bi, H. Design and preparation of a ternary composite of graphene oxide/carbon dots/polypyrrole for supercapacitor application: Importance and unique role of carbon dots. Carbon 2017, 115, 134–146. [Google Scholar] [CrossRef]
- Kakaei, K.; Khodadoost, S.; Gholipour, M.; Shouraei, N. Core-shell polyaniline functionalized carbon quantum dots for supercapacitor. J. Phys. Chem. Solids 2021, 148, 109753. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, J.; Tian, Z.; Ye, Y.; Cai, Y.; Liang, C.; Terabe, K. A general strategy toward transition metal carbide/carbon core/shell nanospheres and their application for supercapacitor electrode. Carbon 2016, 100, 590–599. [Google Scholar] [CrossRef]
- Zhou, Y.; Sharma, S.K.; Peng, Z.; Leblanc, R.M. Polymers in Carbon Dots: A Review. Polymers 2017, 9, 67. [Google Scholar] [CrossRef] [Green Version]
- Ho, K.C.; Lin, L.Y. A review of electrode materials based on core–shell nanostructures for electrochemical supercapacitors. J. Mater. Chem. A 2019, 7, 3516–3530. [Google Scholar] [CrossRef]
- Devadas, B.; Imae, T. Effect of Carbon Dots on Conducting Polymers for Energy Storage Applications. ACS Sustain. Chem. Eng. 2017, 6, 127–134. [Google Scholar] [CrossRef]
- Abdunazar, A.; Zhang, Y.; Muslim, A.; Wang, L.; Lan, H. Preparation and electrochemical characterization of carbon dots/polyaniline composite materials. Polym. Bull. 2020, 77, 1067–1080. [Google Scholar] [CrossRef]
- Lian, Y.; Xu, Z.; Wang, D.; Bai, Y.; Ban, C.; Zhao, J.; Zhang, H. Nb2O5 quantum dots coated with biomass carbon for ultra-stable lithium-ion supercapacitors. J. Alloys Compd. 2021, 850, 156808. [Google Scholar] [CrossRef]
- Dinari, M.; Momeni, M.M.; Goudarzirad, M. Nanocomposite films of polyaniline/graphene quantum dots and its supercapacitor properties. Surface Eng. 2015, 32, 535–540. [Google Scholar] [CrossRef]
- Jian, X.; Yang, H.; Li, J.; Zhang, E.; Cao, L.; Liang, Z. Flexible all-solid-state high-performance supercapacitor based on electrochemically synthesized carbon quantum dots/polypyrrole composite electrode. Electrochim. Acta 2017, 228, 483–493. [Google Scholar] [CrossRef]
- Yasoda, Y.; Das, P.; Malik, S.; Kothurkar, N.K.; Batabyal, S.K. Urea-mediated synthesized carbon quantum dots to tune the electrochemical performance of polyaniline nanorods for supercapacitor device. J. Sci. Adv. Mater. Devices 2021, 7, 100403. [Google Scholar] [CrossRef]
- Shao, F.; Hu, N.; Su, Y.; Li, H.; Li, B.; Zou, C.; Li, G.; Yang, Z.; Zhang, Y. PANI/Graphene quantum dots/graphene co-coated compressed non-woven towel for wearable energy storage. Synth. Met. 2020, 270, 116571. [Google Scholar] [CrossRef]
- Qin, W.; Zhou, N.; Wu, C.; Xie, M.; Sun, H.; Guo, Y.; Pan, L. Mini-Review on the Redox Additives in Aqueous Electrolyte for High Performance Supercapacitors. ACS Omega 2020, 5, 3801–3808. [Google Scholar] [CrossRef] [Green Version]
- González, A.; Goikolea, E.; Barrena, J.A.; Mysyk, R. Review on supercapacitors: Technologies and materials. Renew. Sustain. Energy 2016, 58, 1189–1206. [Google Scholar] [CrossRef]
- Pal, B.; Yang, S.; Ramesh, S.; Thangadurai, V.; Jose, R. Electrolyte selection for supercapacitive devices: A critical review. Nanoscale Adv. 2019, 1, 3807–3835. [Google Scholar] [CrossRef] [Green Version]
- Luo, J.; Wang, J.; Liu, S.; Wu, W.; Jia, T.; Yang, Z.; Mu, S.; Huang, Y. Graphene quantum dots encapsulated tremella-like NiCo2O4 for advanced asymmetric supercapacitors. Carbon 2019, 146, 1–8. [Google Scholar] [CrossRef]
- Karthik, N.; Edison, T.N.J.I.; Atchudan, R.; Xiong, D.; Lee, Y.R. Electro-synthesis of sulfur doped nickel cobalt layered double hydroxide for electrocatalytic hydrogen evolution reaction and supercapacitor applications. J. Electroanal. Chem. 2019, 833, 105–112. [Google Scholar] [CrossRef]
- Ponnar, M.; Pushpanathan, K.; Santhi, R.; Ravichandran, S. Enhanced supercapacitor performance and ferromagnetic behavior of Ni-doped CeO2 quantum dots. J. Mater. Sci. Mater. Elec. 2020, 31, 12661–12677. [Google Scholar] [CrossRef]
- Xu, H.; Wang, H.; Lu, Y.; Zeng, Y.; Yang, Y.; Zhang, Z.; Wang, H.; Wang, X.; Li, L. CeO2 quantum dots for highly selective and ultrasensitive fluorescence detection of 4-nitrophenol via the fluorescence resonance energy transfer mechanism. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2021, 262, 120115. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Liu, C.; Qian, J.; Chen, Z.; Chen, F. Novel 3D porous graphene decorated with Co3O4/CeO2 for high performance supercapacitor power cell. J. Rare Earths 2017, 35, 995–1001. [Google Scholar] [CrossRef]
- Tian, X.; Zhu, Q.; Xu, B. “Water-in-Salt” Electrolytes for Supercapacitors: A Review. ChemSusChem 2021, 14, 2501–2515. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Zhou, J.; Cai, Z.; Fang, G.; Cai, Y.; Pan, A.; Liang, S. Nb2O5 quantum dots embedded in MOF derived nitrogen-doped porous carbon for advanced hybrid supercapacitor applications. J. Mater. Chem. A 2016, 4, 17838–17847. [Google Scholar] [CrossRef]
- Wei, J.S.; Ding, H.; Zhang, P.; Song, Y.F.; Chen, J.; Wang, Y.G.; Xiong, H.M. Carbon Dots/NiCo2O4 Nanocomposites with Various Morphologies for High Performance Supercapacitors. Small 2016, 12, 5927–5934. [Google Scholar] [CrossRef]
- Talluri, B.; Ghosh, S.; Rao, G.R.; Thomas, T. Nanocomposites of digestively ripened copper oxide quantum dots and graphene oxide as a binder free battery-like supercapacitor electrode material. Electrochim. Acta 2019, 321, 134709. [Google Scholar] [CrossRef]
- Bonu, V.; Gupta, B.; Chandra, S.; Das, A.; Dhara, S.; Tyagi, A.K. Electrochemical supercapacitor performance of SnO2 quantum dots. Electrochim. Acta 2016, 203, 230–237. [Google Scholar] [CrossRef] [Green Version]
- Luo, P.; Guan, X.; Yu, Y.; Li, X.; Yan, F. Hydrothermal Synthesis of Graphene Quantum Dots Supported on Three-Dimensional Graphene for Supercapacitors. Nanomaterials 2019, 9, 201. [Google Scholar] [CrossRef] [Green Version]
- Permatasari, F.A.; Irham, M.A.; Bisri, S.Z.; Iskandar, F. Carbon-Based Quantum Dots for Supercapacitors: Recent Advances and Future Challenges. Nanomaterials 2021, 11, 91. [Google Scholar] [CrossRef] [PubMed]
- Mehare, M.D.; Deshmukh, A.D.; Dhoble, S.J. Carbon Quantum Dots/Polyaniline Nanocomposite (S-CQD/PANI) for High Capacitive Asymmetric Supercapacitor Device. J. Nanosci. Nanotechnol. 2020, 20, 3785–3794. [Google Scholar] [CrossRef] [PubMed]
- Jian, X.; Li, J.-G.; Yang, H.-M.; Cao, L.-L.; Zhang, E.-H.; Liang, Z.-H. Carbon quantum dots reinforced polypyrrole nanowire via electrostatic self-assembly strategy for high-performance supercapacitors. Carbon 2017, 114, 533–543. [Google Scholar] [CrossRef]
- Ramadan, A.; Anas, M.; Ebrahim, S.; Soliman, M.; Abou-Aly, A. Effect of Co-doped graphene quantum dots to polyaniline ratio on performance of supercapacitor. J. Mater. Sci. Mater. Electron. 2020, 31, 7247–7259. [Google Scholar] [CrossRef]
- Liu, W.; Yan, X.; Chen, J.; Feng, Y.; Xue, Q. Novel and high-performance asymmetric micro-supercapacitors based on graphene quantum dots and polyaniline nanofibers. Nanoscale 2013, 5, 6053–6062. [Google Scholar] [CrossRef]
- Xie, F.; Zhou, M.; Wang, G.; Wang, Q.; Yan, M.; Bi, H. Morphology-dependent electrochemical performance of nitrogen-doped carbon dots@polyaniline hybrids for supercapacitors. Int. J. Energy Res. 2019, 43, 7529–7540. [Google Scholar] [CrossRef]
- Mondal, S.; Rana, U.; Malik, S. Graphene quantum dot-doped polyaniline nanofiber as high performance supercapacitor electrode materials. Chem. Commun. 2015, 51, 12365–12368. [Google Scholar] [CrossRef]
- Malik, R.; Lata, S.; Soni, U.; Rani, P.; Malik, R.S. Carbon quantum dots intercalated in polypyrrole (PPy) thin electrodes for accelerated energy storage. Electrochim. Acta 2020, 364, 137281. [Google Scholar] [CrossRef]
- Abidin, S.N.J.S.Z.; Mamat, S.; Rasyid, S.A.; Zainal, Z.; Sulaiman, Y. Fabrication of poly(vinyl alcohol)-graphene quantum dots coated with poly(3,4-ethylenedioxythiophene) for supercapacitor. J. Polym. Sci. Part A Polym. Chem. 2018, 56, 50–58. [Google Scholar] [CrossRef]
- Zhou, Y.; Xie, Y. Enhanced electrochemical stability of carbon quantum dots-incorporated and ferrous-coordinated polypyrrole for supercapacitor. J. Solid State Electrochem. 2018, 22, 2515–2529. [Google Scholar] [CrossRef]
- Kuzhandaivel, H.; Manickam, S.; Balasingam, S.K.; Franklin, M.C.; Kim, H.-J.; Nallathambi, K.S. Sulfur and nitrogen-doped graphene quantum dots/PANI nanocomposites for supercapacitors. New J. Chem. 2021, 45, 4101–4110. [Google Scholar] [CrossRef]
- Zhou, H.; Yan, Z.; Yang, X.; Lv, J.; Kang, L.; Liu, Z.H. RGO/MnO2/polypyrrole ternary film electrode for supercapacitor. Mater. Chem. Phys. 2016, 177, 40–47. [Google Scholar] [CrossRef]
- Kazemi, S.H.; Aghdam, S.A. High-Performance Asymmetric Supercapacitor Based on Ternary MnO2-Polyaniline-Reduced Graphene Oxide Quantum Dots Nanocomposite Electrode. J. Electron. Mater. 2019, 48, 5088–5098. [Google Scholar] [CrossRef]
- Kumar, A.; Bhattacharya, T.; Hasnain, S.M.; Nayak, A.K. Applications of biomass-derived materials for energy production, conversion, and storage. Mater. Sci. Energy Technol. 2020, 3, 905–920. [Google Scholar] [CrossRef]
- Wang, J.; Nie, P.; Ding, B.; Dong, S.; Hao, X.; Dou, H.; Zhang, X. Biomass derived carbon for energy storage devices. J. Mater. Chem. A 2017, 5, 2411–2428. [Google Scholar] [CrossRef]
- Liedel, C. Sustainable Battery Materials from Biomass. ChemSusChem 2020, 13, 2110–2141. [Google Scholar] [CrossRef]
- Mirjalili, A.; Dong, B.; Pena, P.; Ozkan, C.S.; Ozkan, M. Upcycling of polyethylene terephthalate plastic waste to microporous carbon structure for energy storage. Energy Storage 2020, 2, e201. [Google Scholar] [CrossRef]
- Peng, H.; Sun, X.; Weng, W.; Fang, X. Flexible Electronic Devices Based on Polymers. In Polymer Materials for Energy and Electronic Applications; Academic Press: Cambridge, MA, USA, 2017; pp. 325–354. [Google Scholar] [CrossRef]
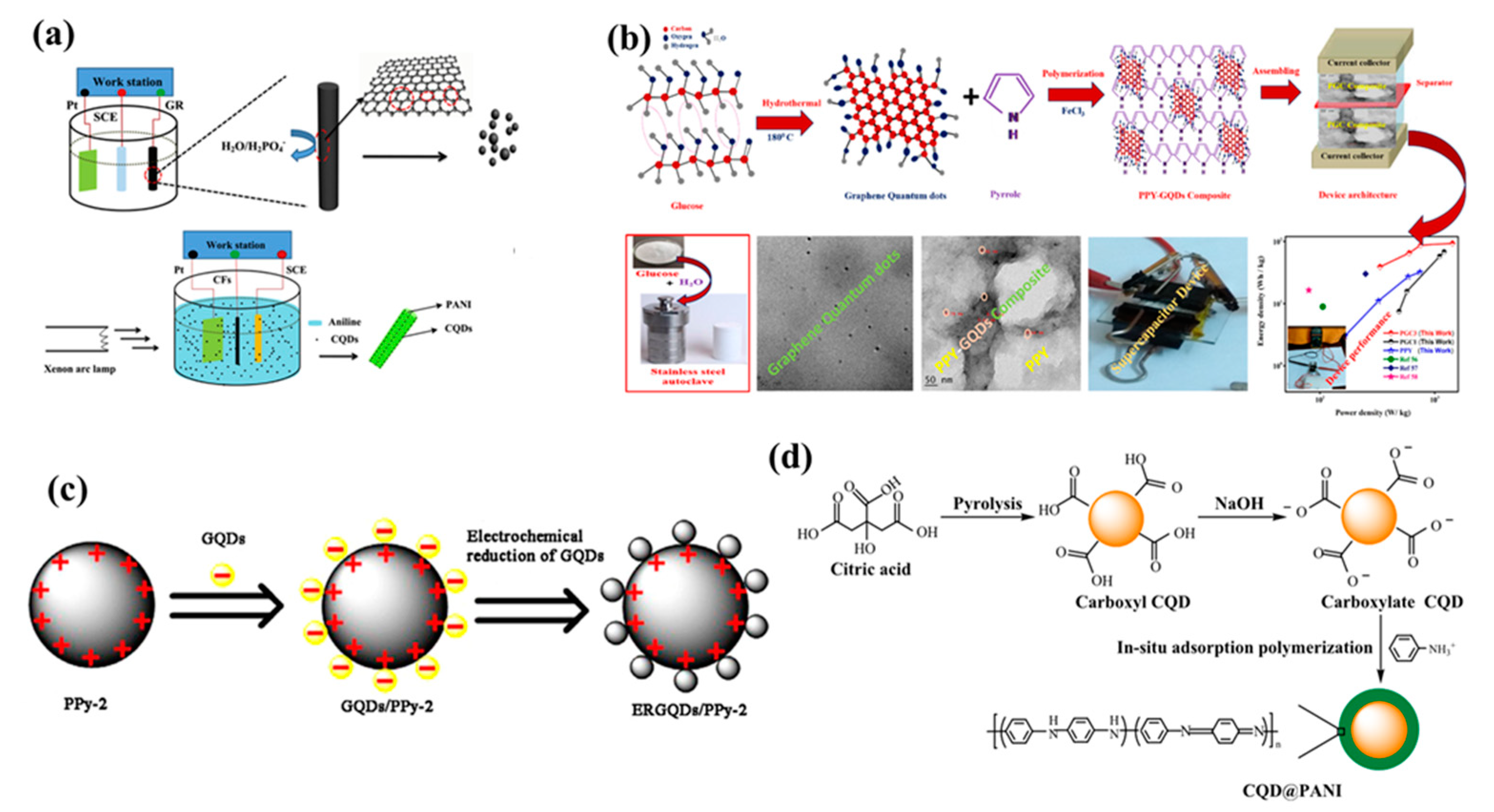

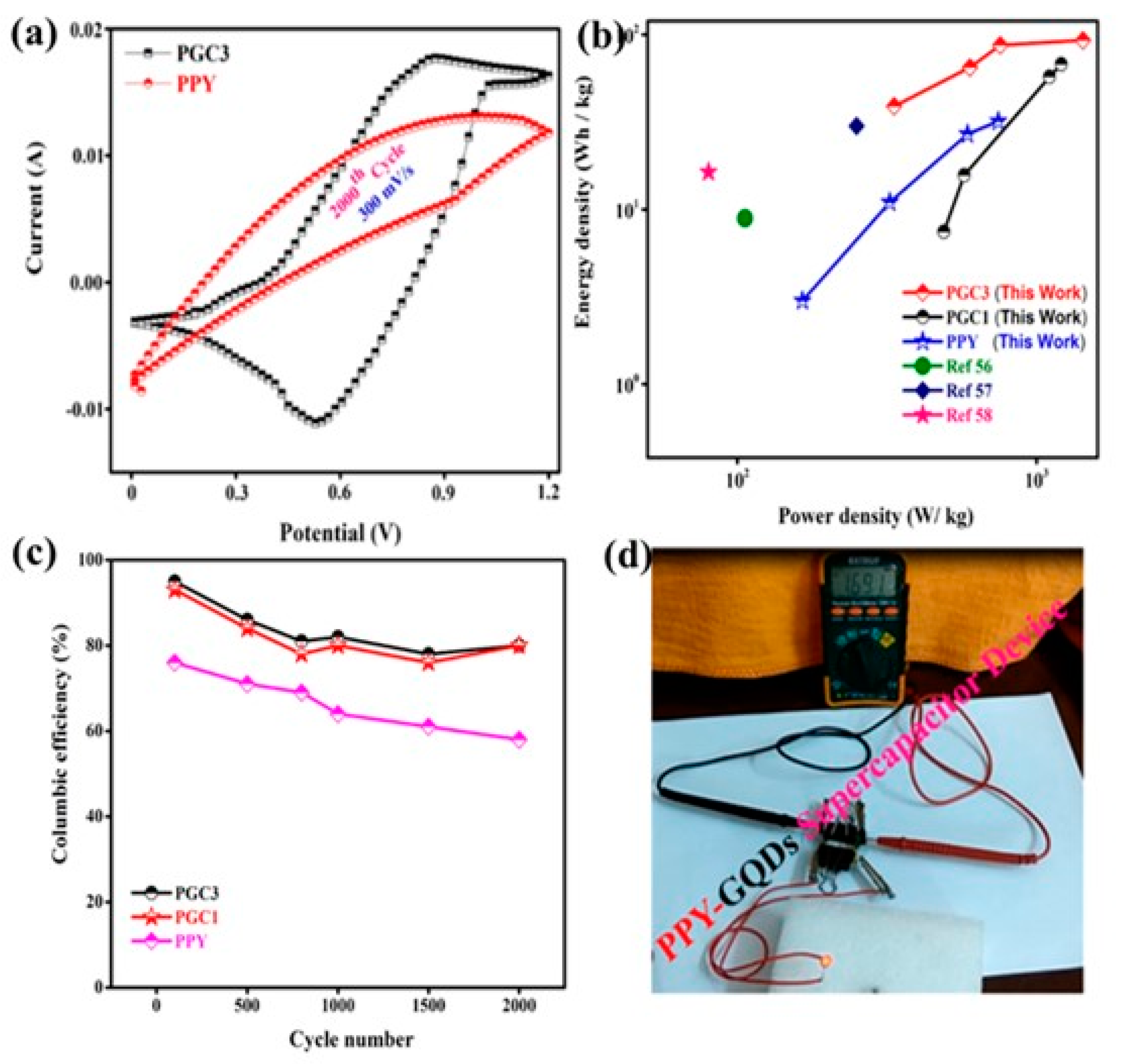
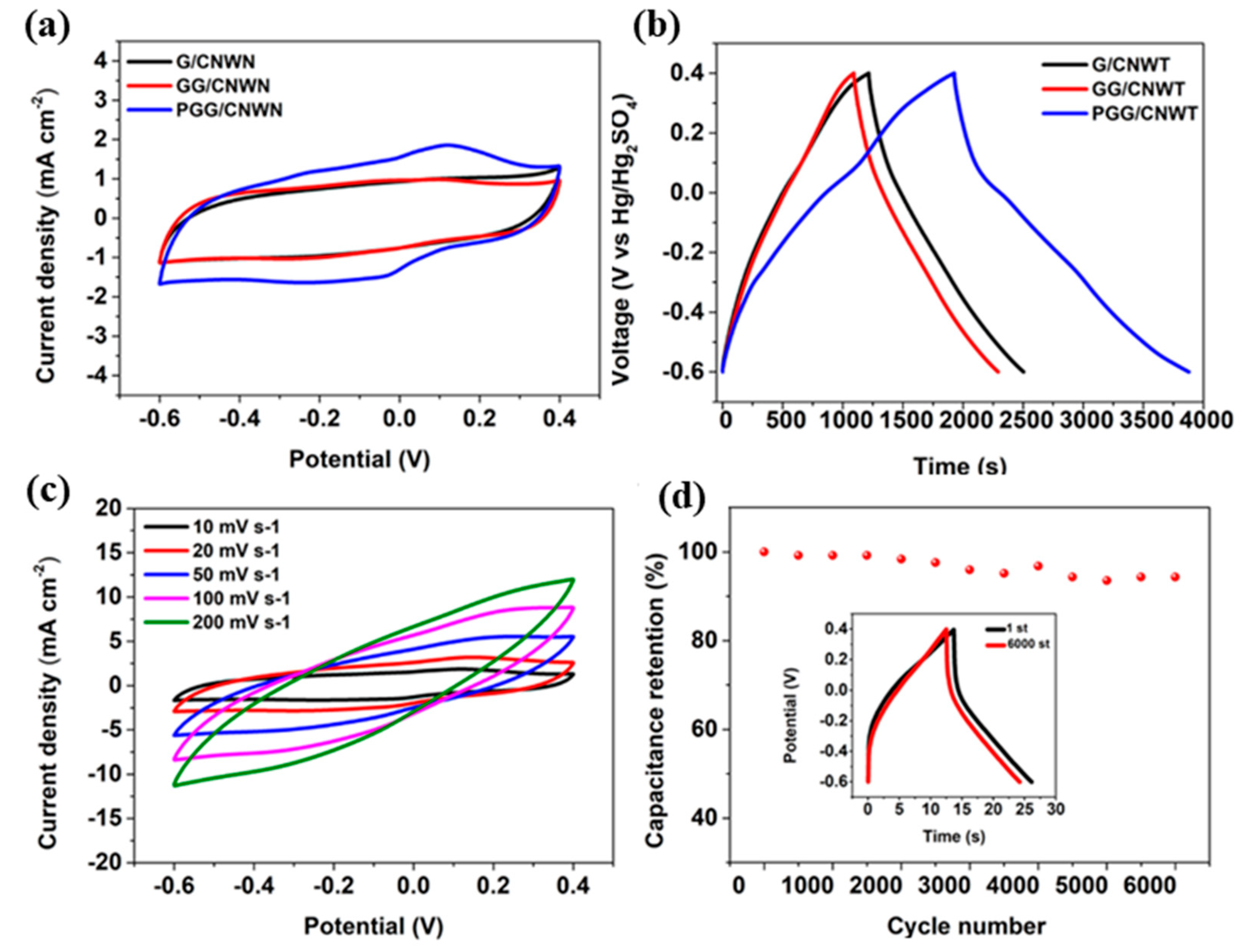
| Electrode | Electrolyte | Specific Capacitance | Retention Rate (Cycles) | Energy Density | Power Density | Ref. |
|---|---|---|---|---|---|---|
| CQDs/PPy PPY-GQDs | PVA-LiCl 1 M NaCl | 308 F g−1 647.54 F g−1 | 85.7 % (2000) 91.7% (2000 cycles) | NA 93 Wh kg−1 | NA 1430 W kg−1 | [86] [102] |
| GQDs/3DG | 1 M KOH | 242 F g−1 | 93% (10,000) | NA | NA | [103] |
| S-CQD/PANI | 1 M H2SO4 | 295 F g−1 | 80% (1000) | 40.86 Wh kg−1 | 2000 W kg−1 | [104] |
| CQDs/PPy-NW | 1.0 M KCl | 306 F g−1 | 85.2% (5000) | NA | NA | [105] |
| PANI/S,N:G QDs | 2 M KOH | 2524 F g−1 | 100% (1000) | 47.78 Wh kg−1 | 2250 W kg−1 | [106] |
| GQDs//PANI | H3PO4–PVA | 667.5 μF cm−2 | 85.6% (1500) | 0.093 μ Wh cm−2 | 7.52 μ W cm−2 | [107] |
| CQDs-PANI | H2SO4–PVA-EG | 738.3 F g−1 | 78.0% (1000) | 33.8 μ Wh cm−2 | 0.3 mW cm−2 | [108] |
| GQDP | 0.5 M H2SO4 | 1044 F g−1 | 80.1% (3000) | 117.45 Wh kg−1 | 448.8 W kg−1 | [109] |
| PPy/CQDs | 1M KCl | 1073 F g−1 | 62 % (2000) | 70.22 Wh kg−1 | 3060 W kg−1 | [110] |
| PVA-GQD/PEDO | 1 M H2SO4 | 291.86 F g−1 | 98& (1000) | 16.95 Wh kg−1 | 984.4 W kg−1 | [111] |
| CQDs/PPy-Fe | H2SO4– PVA | 317 F g−1 | 94.6% (2000) | 52 Wh kg−1 | 900 W kg−1 | [112] |
| S,N-GQD/PANI | 1 M H2SO4 | 645 F g−1 | 90% (1000) | 17.25 Wh kg−1 | 500 W kg−1 | [113] |
| ERGQDs/PPy | 1 M H2SO4 | 418 F g−1 | 86% (1000) | NA | NA | [114] |
| MnO2/PANI/rGO QDs | 1 M H2SO4 | 423 F g−1 | 85% (2000) | 34.47 Wh kg−1 | 640 W kg−1 | [115] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Das, H.T.; Barai, P.; Dutta, S.; Das, N.; Das, P.; Roy, M.; Alauddin, M.; Barai, H.R. Polymer Composites with Quantum Dots as Potential Electrode Materials for Supercapacitors Application: A Review. Polymers 2022, 14, 1053. https://doi.org/10.3390/polym14051053
Das HT, Barai P, Dutta S, Das N, Das P, Roy M, Alauddin M, Barai HR. Polymer Composites with Quantum Dots as Potential Electrode Materials for Supercapacitors Application: A Review. Polymers. 2022; 14(5):1053. https://doi.org/10.3390/polym14051053
Chicago/Turabian StyleDas, Himadri Tanaya, Paritosh Barai, Swapnamoy Dutta, Nigamananda Das, Payaswini Das, Madhusudan Roy, Md. Alauddin, and Hasi Rani Barai. 2022. "Polymer Composites with Quantum Dots as Potential Electrode Materials for Supercapacitors Application: A Review" Polymers 14, no. 5: 1053. https://doi.org/10.3390/polym14051053
APA StyleDas, H. T., Barai, P., Dutta, S., Das, N., Das, P., Roy, M., Alauddin, M., & Barai, H. R. (2022). Polymer Composites with Quantum Dots as Potential Electrode Materials for Supercapacitors Application: A Review. Polymers, 14(5), 1053. https://doi.org/10.3390/polym14051053







