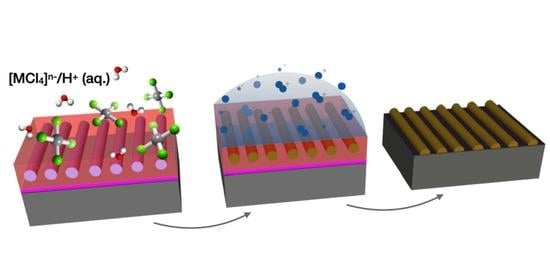
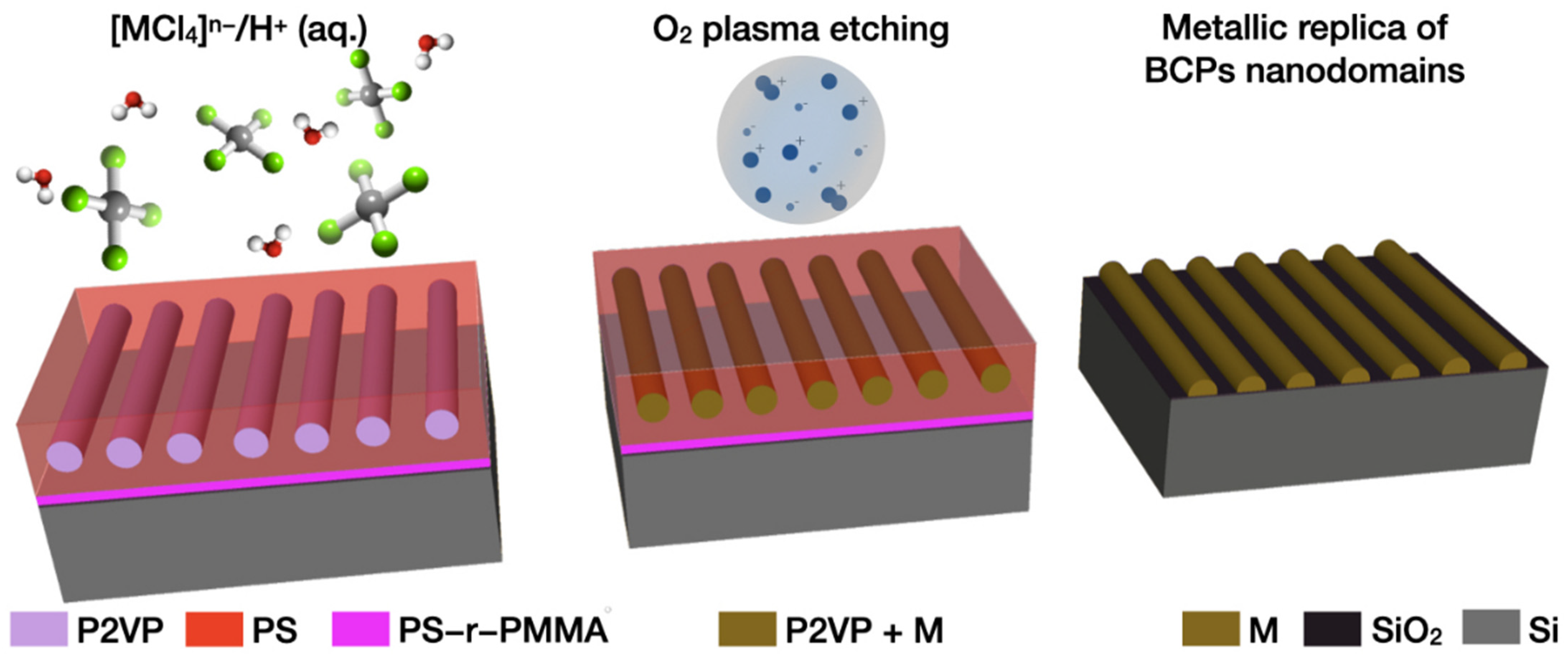
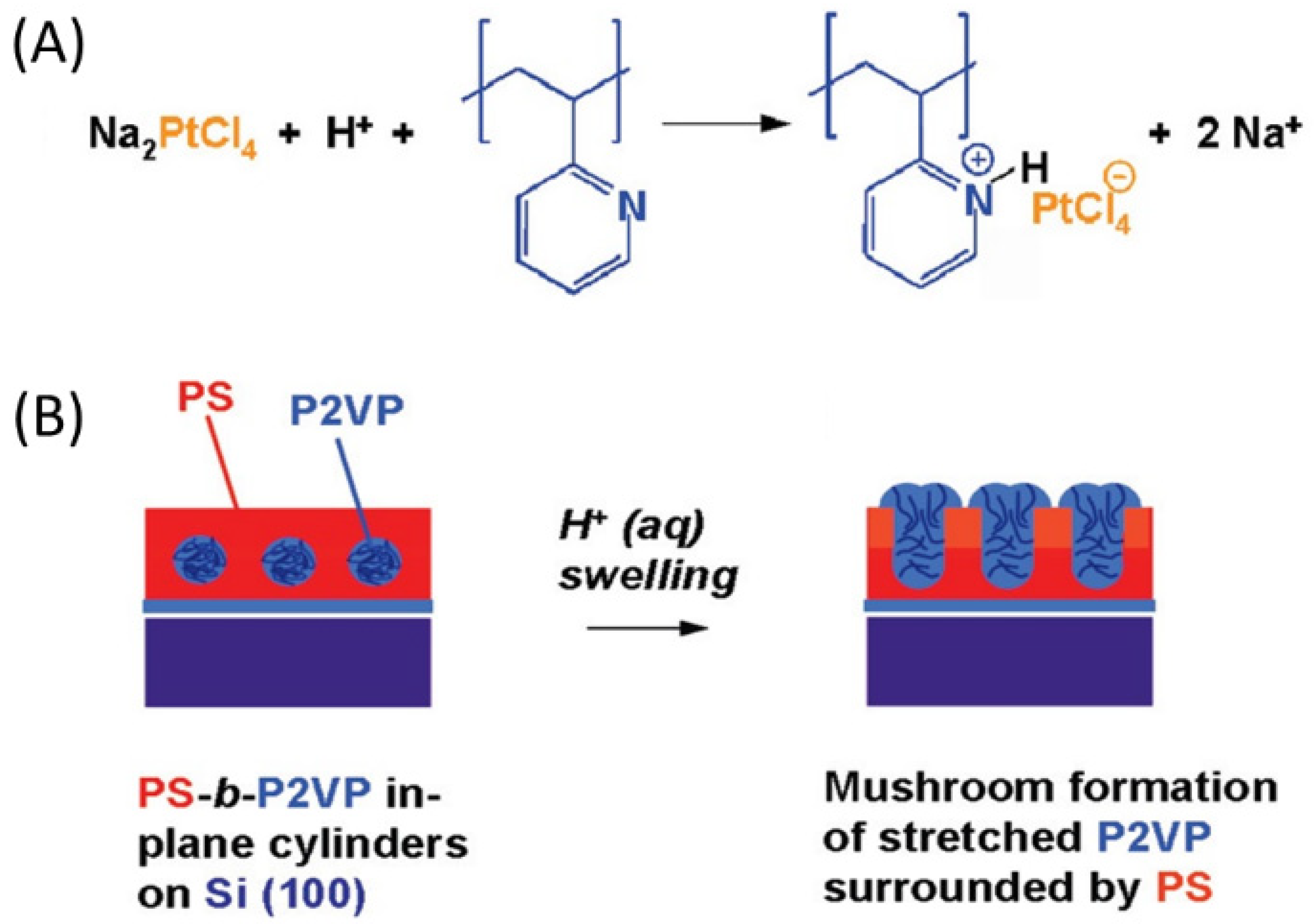
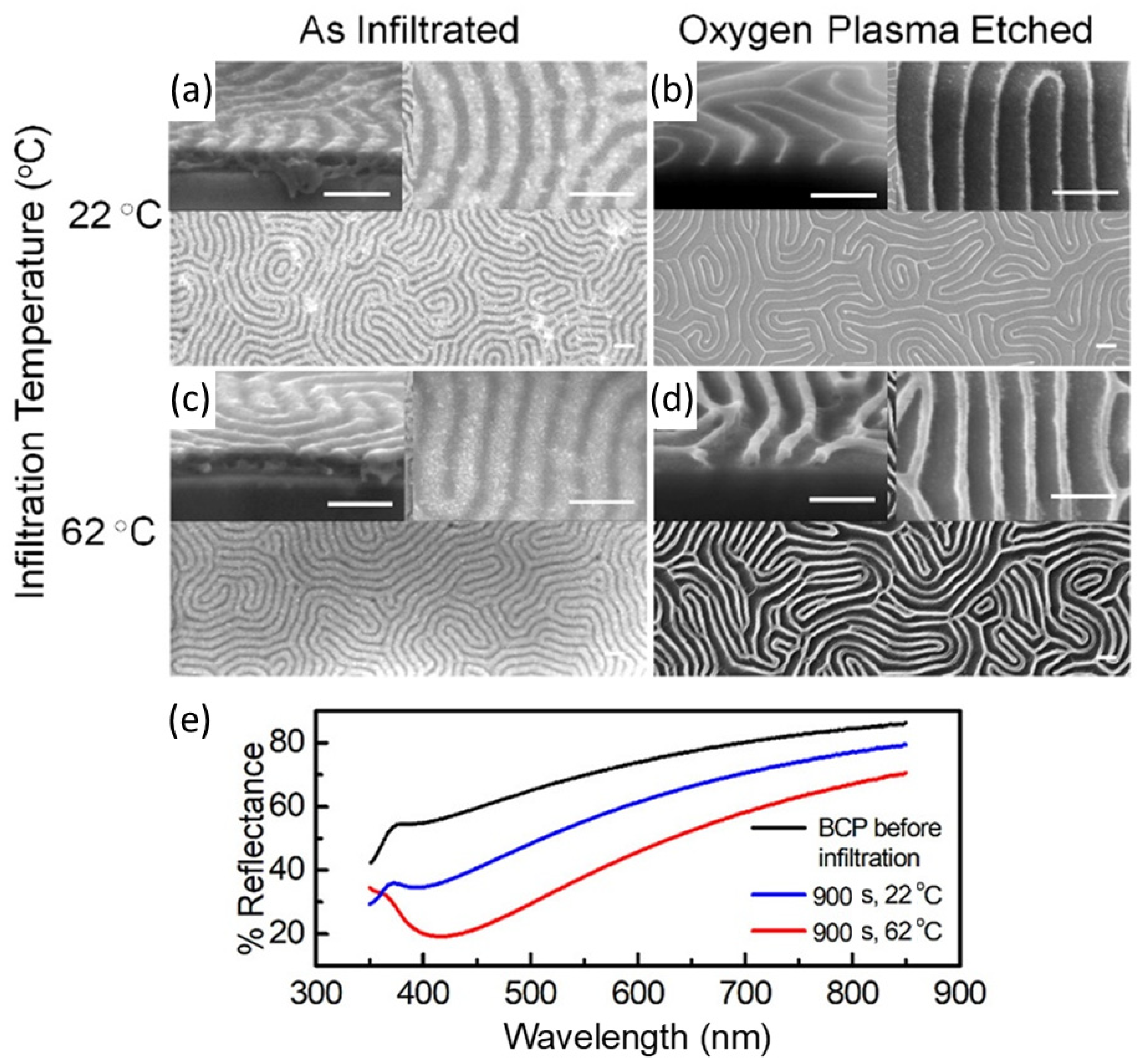
Liquid Phase Infiltration of Block Copolymers
Abstract
1. Introduction
2. LPI Processing and Mechanism
3. Metal Salt Diffusion and Reduction
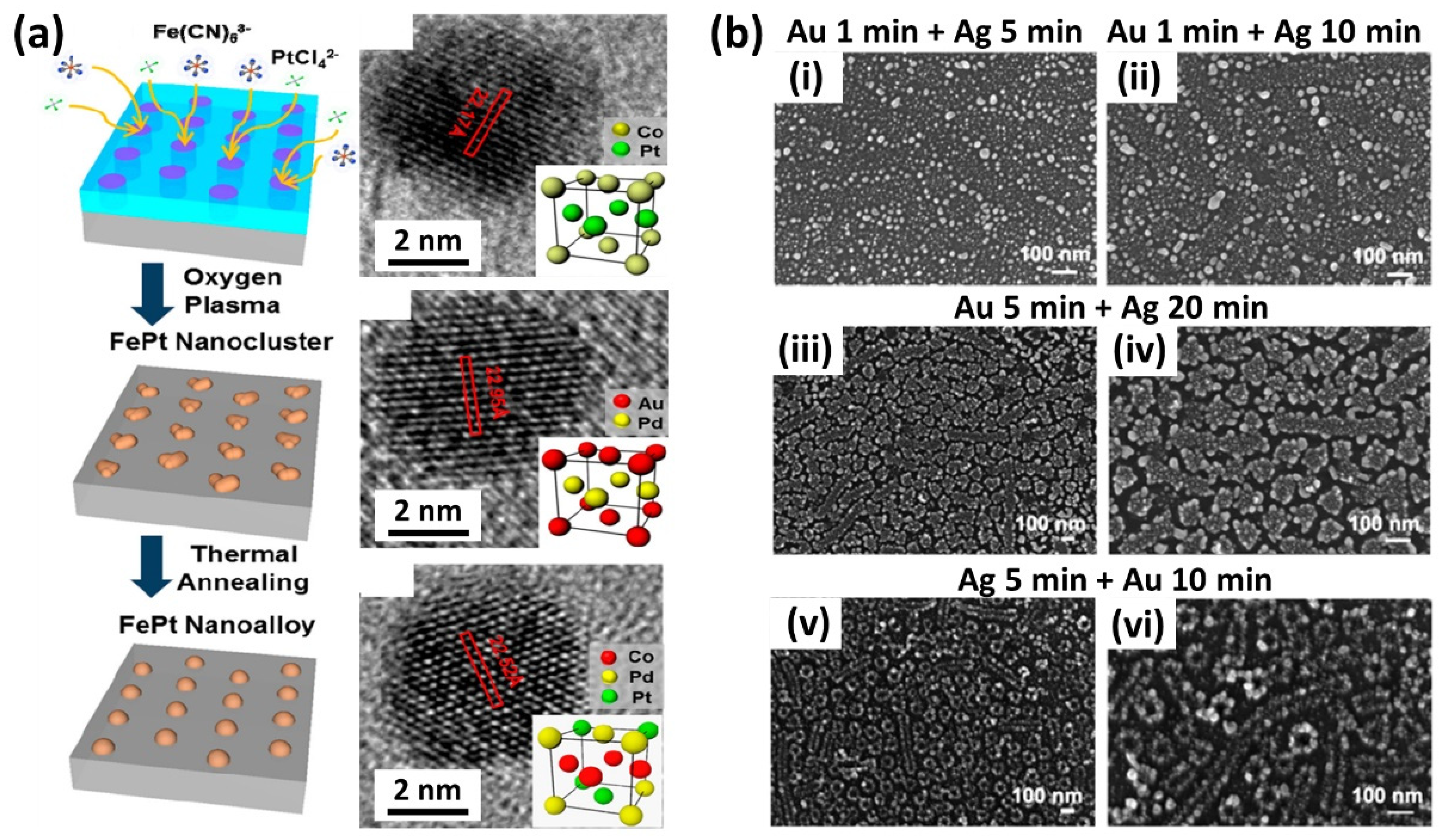
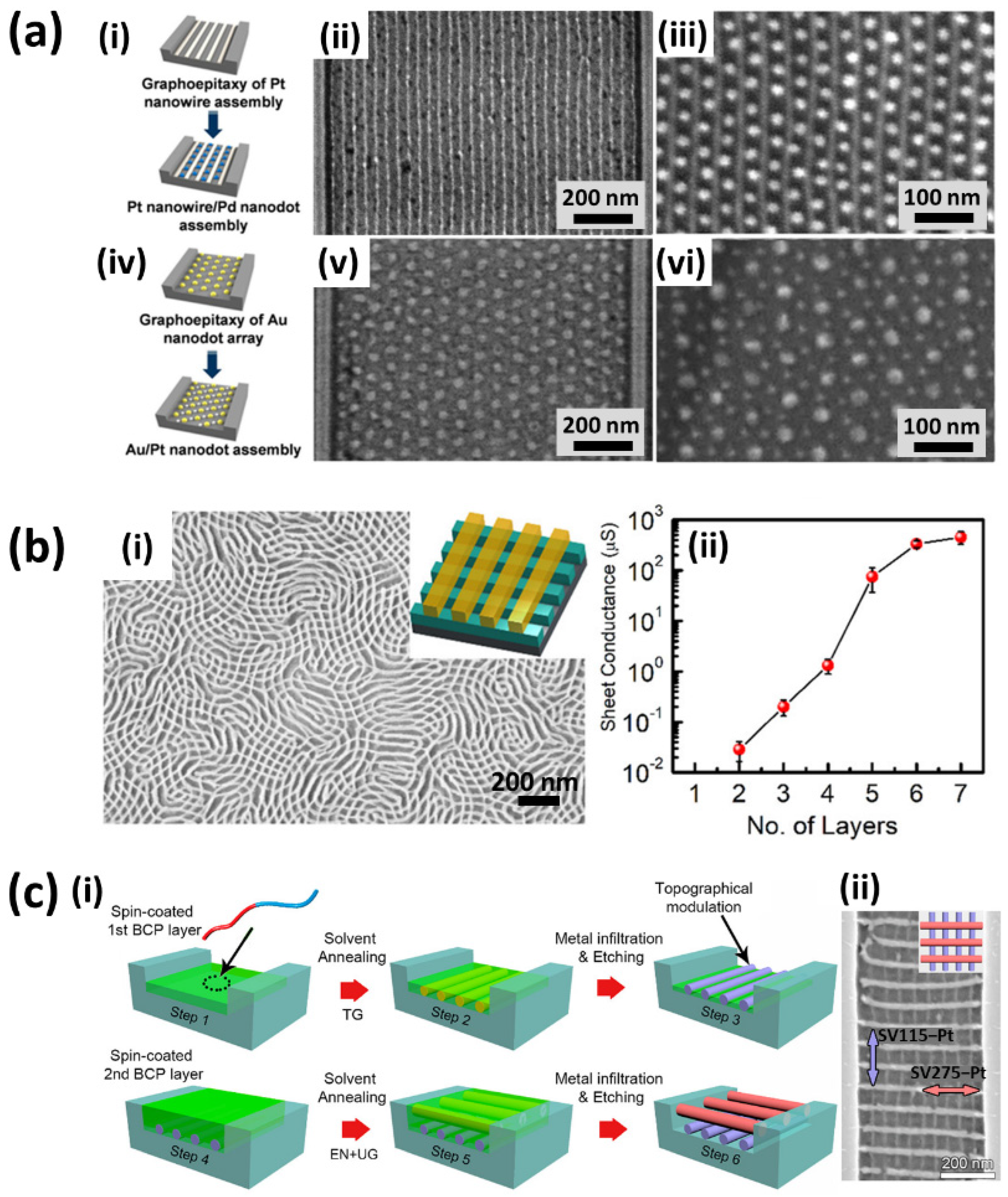
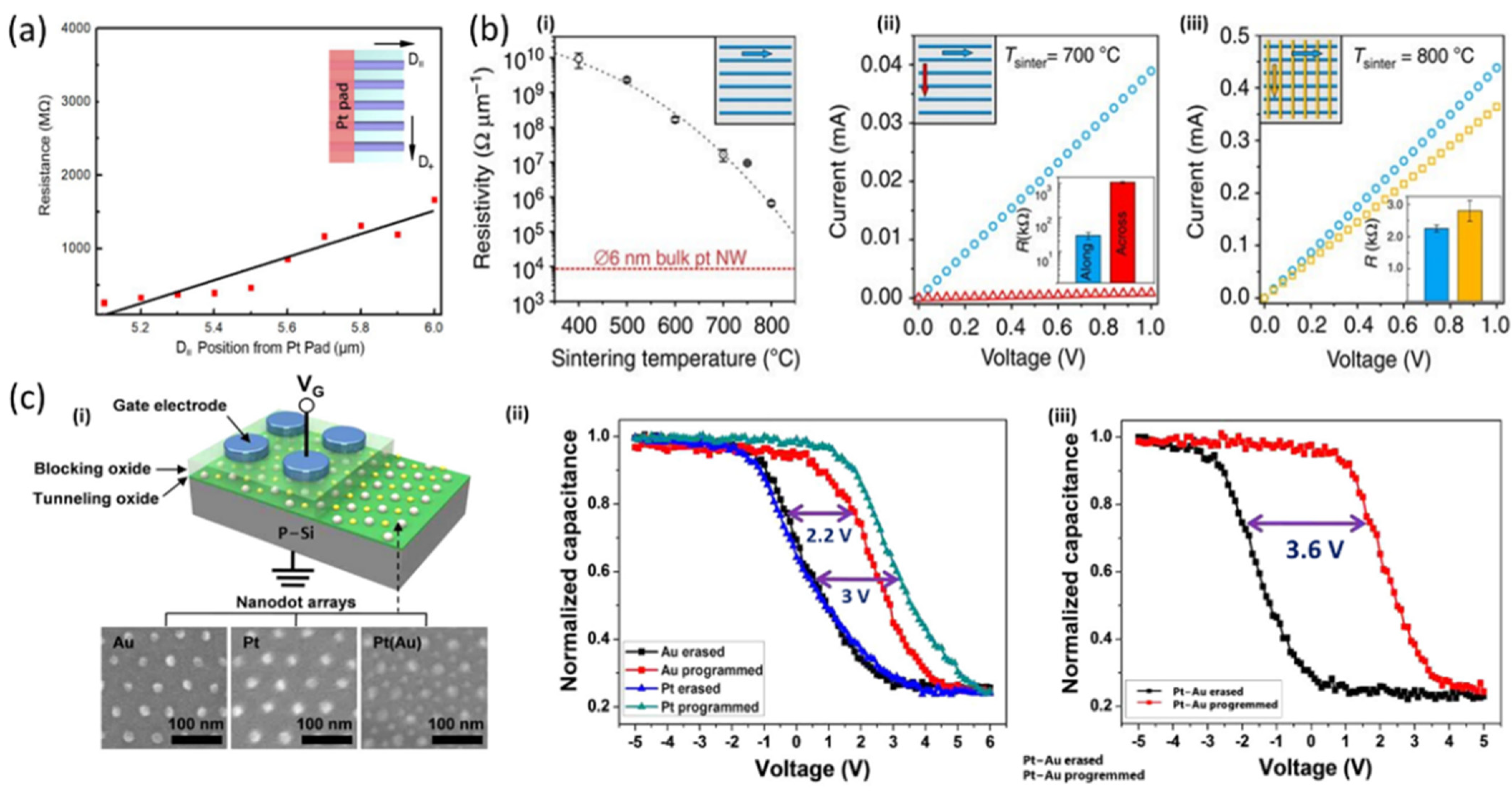
4. Multicomponent Materials and Complex Structures
5. Applications
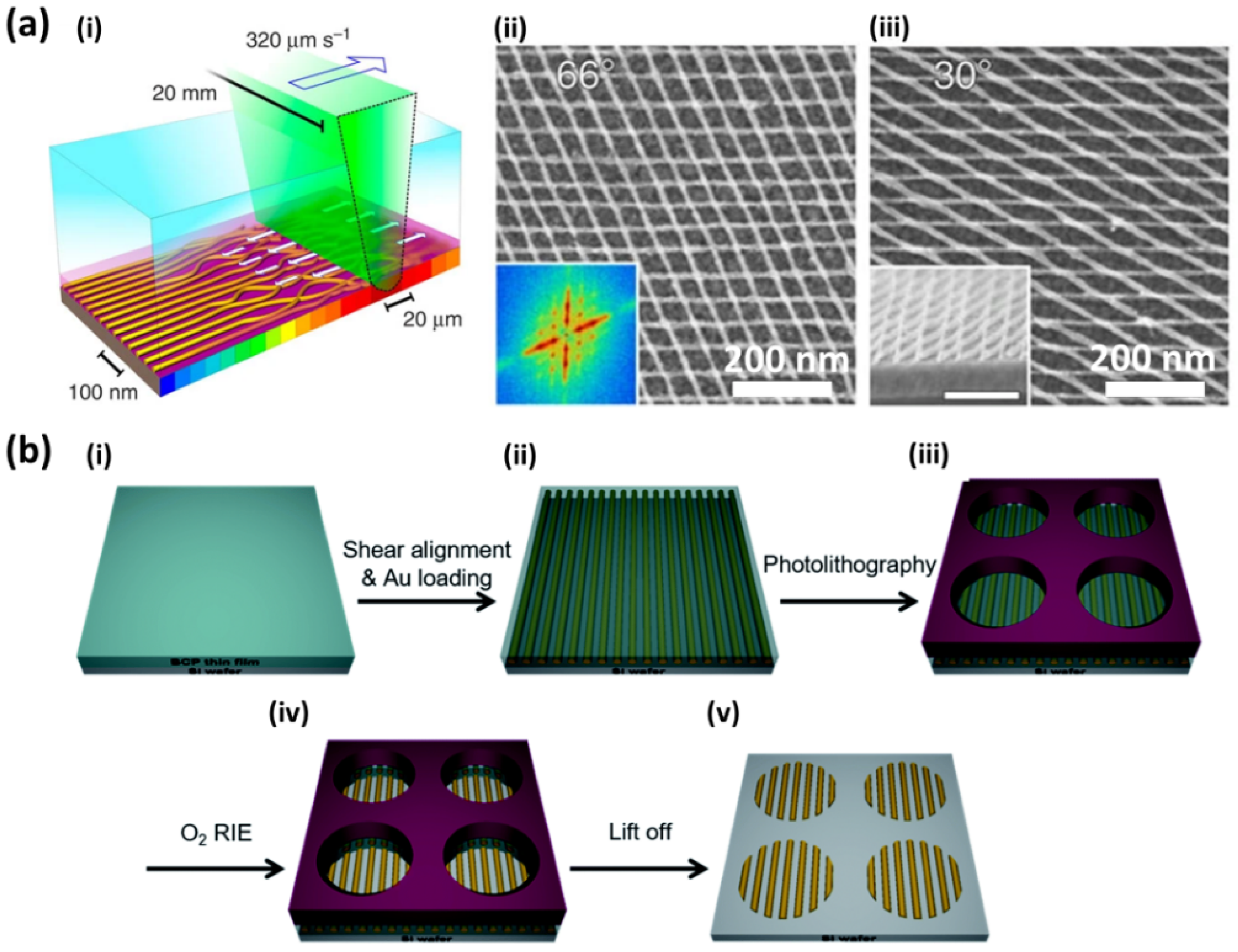
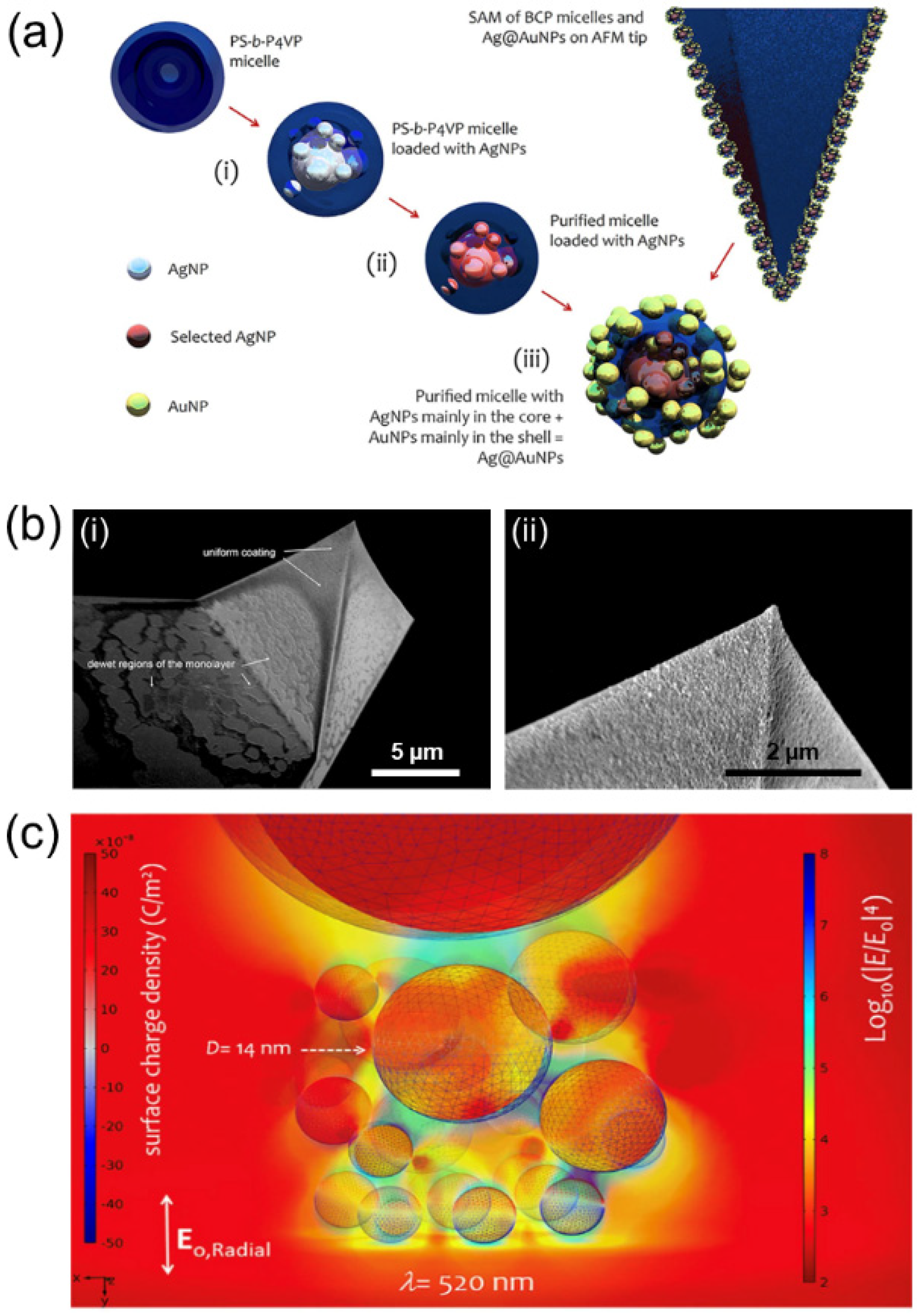
5.1. Photonics and Plasmonics
5.2. Electronics
5.3. Other Applications
6. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Liu, C.C.; Franke, E.; Mignot, Y.; Xie, R.; Yeung, C.W.; Zhang, J.; Chi, C.; Zhang, C.; Farrell, R.; Lai, K.; et al. Directed self-assembly of block copolymers for 7 nanometre FinFET technology and beyond. Nat. Electron. 2018, 1, 562–569. [Google Scholar] [CrossRef]
- Ferrarese Lupi, F.; Giammaria, T.J.; Seguini, G.; Vita, F.; Francescangeli, O.; Sparnacci, K.; Antonioli, D.; Gianotti, V.; Laus, M.; Perego, M. Fine tuning of lithographic masks through thin films of PS-b -PMMA with different molar mass by rapid thermal processing. ACS Appl. Mater. Interfaces 2014, 6, 7180–7188. [Google Scholar] [CrossRef] [PubMed]
- Ferrarese Lupi, F.; Murataj, I.; Celegato, F.; Angelini, A.; Frascella, F.; Chiarcos, R.; Antonioli, D.; Gianotti, V.; Tiberto, P.; Pirri, C.F.; et al. Tailored and Guided Dewetting of Block Copolymer/Homopolymer Blends. Macromolecules 2020, 53, 7207–7217. [Google Scholar] [CrossRef]
- Ferrarese Lupi, F.; Giammaria, T.J.; Miti, A.; Zuccheri, G.; Carignano, S.; Sparnacci, K.; Seguini, G.; De Leo, N.; Boarino, L.; Perego, M.; et al. Hierarchical Order in Dewetted Block Copolymer Thin Films on Chemically Patterned Surfaces. ACS Nano 2018, 12, 7076–7085. [Google Scholar] [CrossRef] [PubMed]
- Frascaroli, J.; Brivio, S.; Ferrarese Lupi, F.; Seguini, G.; Boarino, L.; Perego, M.; Spiga, S. Resistive switching in high-density nanodevices fabricated by block copolymer self-Assembly. ACS Nano 2015, 9, 2518–2529. [Google Scholar] [CrossRef]
- Murataj, I.; Channab, M.; Cara, E.; Pirri, C.F.; Boarino, L.; Angelini, A.; Ferrarese Lupi, F. Hyperbolic Metamaterials via Hierarchical Block Copolymer Nanostructures. Adv. Opt. Mater. 2021, 9, 2001933. [Google Scholar] [CrossRef]
- Guo, C.; Lin, Y.H.; Witman, M.D.; Smith, K.A.; Wang, C.; Hexemer, A.; Strzalka, J.; Gomez, E.D.; Verduzco, R. Conjugated block copolymer photovoltaics with near 3% efficiency through microphase separation. Nano Lett. 2013, 13, 2957–2963. [Google Scholar] [CrossRef] [PubMed]
- Malafronte, A.; Emendato, A.; Auriemma, F.; Sasso, C.; Laus, M.; Murataj, I.; Ferrarese Lupi, F.; De Rosa, C. Tailored inclusion of semiconductor nanoparticles in nanoporous polystyrene-block-polymethyl methacrylate thin films. Polymer 2020, 210, 122983. [Google Scholar] [CrossRef]
- Yang, H.C.; Waldman, R.Z.; Chen, Z.; Darling, S.B. Atomic layer deposition for membrane interface engineering. Nanoscale 2018, 10, 20505–20513. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Simon, A.; Abetz, C.; Held, M.; Höhme, A.L.; Schneider, E.S.; Segal-Peretz, T.; Abetz, V. Hybrid Organic–Inorganic–Organic Isoporous Membranes with Tunable Pore Sizes and Functionalities for Molecular Separation. Adv. Mater. 2021, 33, 2105251. [Google Scholar] [CrossRef] [PubMed]
- Cara, E.; Murataj, I.; Milano, G.; De Leo, N.; Boarino, L.; Ferrarese Lupi, F. Recent Advances in Sequential Infiltration Synthesis (SIS) of Block Copolymers (BCPs). Nanomaterials 2021, 11, 994. [Google Scholar] [CrossRef]
- Waldman, R.Z.; Mandia, D.J.; Yanguas-Gil, A.; Martinson, A.B.F.; Elam, J.W.; Darling, S.B. The chemical physics of sequential infiltration synthesis—A thermodynamic and kinetic perspective. J. Chem. Phys. 2019, 151, 190901. [Google Scholar] [CrossRef]
- Peng, Q.; Tseng, Y.C.; Darling, S.B.; Elam, J.W. A route to nanoscopic materials via sequential infiltration synthesis on block copolymer templates. ACS Nano 2011, 5, 4600–4606. [Google Scholar] [CrossRef] [PubMed]
- Peng, Q.; Tseng, Y.C.; Long, Y.; Mane, A.U.; DiDona, S.; Darling, S.B.; Elam, J.W. Effect of Nanostructured Domains in Self-Assembled Block Copolymer Films on Sequential Infiltration Synthesis. Langmuir 2017, 33, 13214–13223. [Google Scholar] [CrossRef] [PubMed]
- Chai, J.; Buriak, J.M. Using cylindrical domains of block copolymers to self-assemble and align metallic nanowires. ACS Nano 2008, 2, 489–501. [Google Scholar] [CrossRef]
- Paiva, A.E.; Vasquez, J.F.B.; Perova, T.S.; Borsagli, F.G.L.M.; Morris, M. Mechanism of liquid-phase metal infiltration into pyridine-containing polymeric thin films. Mater. Lett. 2022, 313, 131682. [Google Scholar] [CrossRef]
- Subramanian, A.; Tiwale, N.; Doerk, G.; Kisslinger, K.; Nam, C.Y. Enhanced Hybridisation and Nanopatterning via Heated Liquid-Phase Infiltration into Self-Assembled Block Copolymer Thin Films. ACS Appl. Mater. Interfaces 2020, 12, 1444–1453. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.G.; Choi, H.J.; Han, K.H.; Kim, J.H.; Lee, C.W.; Jung, E.I.; Jin, H.M.; Kim, S.O. Block Copolymer Nanopatterning for Nonsemiconductor Device Applications. ACS Appl. Mater. Interfaces 2022, 14, 12011–12037. [Google Scholar] [CrossRef]
- Cummins, C.; Ghoshal, T.; Holmes, J.D.; Morris, M.A. Strategies for Inorganic Incorporation using Neat Block Copolymer Thin Films for Etch Mask Function and Nanotechnological Application. Adv. Mater. 2016, 28, 5586–5618. [Google Scholar] [CrossRef]
- Bas, S.Z.; Cummins, C.; Borah, D.; Ozmen, M.; Morris, M.A. Electrochemical Sensing of Hydrogen Peroxide Using Block Copolymer Templated Iron Oxide Nanopatterns. Anal. Chem. 2018, 90, 1122–1128. [Google Scholar] [CrossRef] [PubMed]
- Han, H.; Oh, J.W.; Park, J.; Lee, H.; Park, C.; Lee, S.W.; Lee, K.; Jeon, S.; Kim, S.; Park, Y.; et al. Hierarchically Ordered Perovskites with High Photo-Electronic and Environmental Stability via Nanoimprinting Guided Block Copolymer Self-Assembly. Adv. Mater. Interfaces 2022, 9, 2200082. [Google Scholar] [CrossRef]
- Mun, J.H.; Cha, S.K.; Kim, H.; Moon, H.S.; Kim, J.Y.; Jin, H.M.; Choi, Y.J.; Baek, J.E.; Shin, J.; Kim, S.O. Nanodomain swelling block copolymer lithography for morphology tunable metal nanopatterning. Small 2014, 10, 3742–3749. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.; Lee, S.; Tang, A.S.; Kim, C.; Liu, R.; Im, K.; Jung, H.T.; Ross, C.A. Platinum Infiltration of a Block Copolymer for Interconnected Three-Dimensional Metal Nanostructures. ACS Appl. Nano Mater. 2021, 4, 793–801. [Google Scholar] [CrossRef]
- Mun, J.H.; Chang, Y.H.; Shin, D.O.; Yoon, J.M.; Choi, D.S.; Lee, K.M.; Kim, J.Y.; Cha, S.K.; Lee, J.Y.; Jeong, J.R.; et al. Monodisperse pattern nanoalloying for synergistic intermetallic catalysis. Nano Lett. 2013, 13, 5720–5726. [Google Scholar] [CrossRef] [PubMed]
- Mistark, P.A.; Park, S.; Yalcin, S.E.; Lee, D.H.; Yavuzcetin, O.; Tuominen, M.T.; Russell, T.P.; Achermann, M. Block-copolymer-based plasmonic nanostructures. ACS Nano 2009, 3, 3987–3992. [Google Scholar] [CrossRef] [PubMed]
- Menezes, W.G.; Zielasek, V.; Dzhardimalieva, G.I.; Pomogailo, S.I.; Thiel, K.; Wöhrle, D.; Hartwig, A.; Bäumer, M. Synthesis of stable AuAg bimetallic nanoparticles encapsulated by diblock copolymer micelles. Nanoscale 2012, 4, 1658–1664. [Google Scholar] [CrossRef] [PubMed]
- Shin, D.O.; Mun, J.H.; Hwang, G.T.; Yoon, J.M.; Kim, J.Y.; Yun, J.M.; Yang, Y.B.; Oh, Y.; Lee, J.Y.; Shin, J.; et al. Multicomponent nanopatterns by directed block copolymer self-assembly. ACS Nano 2013, 7, 8899–8907. [Google Scholar] [CrossRef] [PubMed]
- Yin, S.; Cao, W.; Ji, Q.; Cheng, Y.; Song, L.; Li, N.; Weindl, C.L.; Schwartzkopf, M.; Roth, S.V.; Müller-Buschbaum, P. Multidimensional Morphology Control for PS-b-P4VP Templated Mesoporous Iron (III) Oxide Thin Films. Adv. Mater. Interfaces 2021, 8, 2100141. [Google Scholar] [CrossRef]
- Cummins, C.; Borah, D.; Rasappa, S.; Chaudhari, A.; Ghoshal, T.; O’Driscoll, B.M.D.; Carolan, P.; Petkov, N.; Holmes, J.D.; Morris, M.A. Self-assembly of polystyrene-block-poly(4-vinylpyridine) block copolymer on molecularly functionalised silicon substrates: Fabrication of inorganic nanostructured etchmask for lithographic use. J. Mater. Chem. C 2013, 1, 7941–7951. [Google Scholar] [CrossRef]
- Glass, R.; Mller, M.; Spatz, J.P. Block Copolymer Micelle Nanolithography. Nanotechnology 2003, 14, 1153–1160. [Google Scholar] [CrossRef]
- Lohmüller, T.; Aydin, D.; Schwieder, M.; Morhard, C.; Louban, I.; Pacholski, C.; Spatz, J.P. Nanopatterning by Block Copolymer Micelle Nanolithography and Bioinspired Applications. Biointerphases 2011, 6, MR1–MR12. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Fernandez, A.; Aissou, K.; Pécastaings, G.; Hadziioannou, G.; Fleury, G.; Ponsinet, V. High refractive index in low metal content nanoplasmonic surfaces from self-assembled block copolymer thin films. Nanoscale Adv. 2019, 1, 849–857. [Google Scholar] [CrossRef] [PubMed]
- Majewski, P.W.; Rahman, A.; Black, C.T.; Yager, K.G. Arbitrary lattice symmetries via block copolymer nanomeshes. Nat. Commun. 2015, 6, 7448. [Google Scholar] [CrossRef] [PubMed]
- Chai, J.; Wang, D.; Fan, X.; Buriak, J.M. Assembly of aligned linear metallic patterns on silicon. Nat. Nanotechnol. 2007, 2, 500–506. [Google Scholar] [CrossRef]
- Aizawa, M.; Buriak, J.M. Nanoscale patterning of two metals on silicon surfaces using an ABC triblock copolymer template. J. Am. Chem. Soc. 2006, 128, 5877–5886. [Google Scholar] [CrossRef]
- Ghoshal, T.; Maity, T.; Godsell, J.F.; Roy, S.; Morris, M.A. Large scale monodisperse hexagonal arrays of superparamagnetic iron oxides nanodots: A facile block copolymer inclusion method. Adv. Mater. 2012, 24, 2390–2397. [Google Scholar] [CrossRef]
- Ghoshal, T.; Shaw, M.T.; Bolger, C.T.; Holmes, J.D.; Morris, M.A. A general method for controlled nanopatterning of oxide dots: A microphase separated block copolymer platform. J. Mater. Chem. 2012, 22, 12083–12089. [Google Scholar] [CrossRef]
- Lee, J.; Kwak, J.; Choi, C.; Han, S.H.; Kim, J.K. Phase behavior of poly (2-vinylpyridine)-block-poly (4-vinylpyridine) copolymers containing gold nanoparticles. Macromolecules 2017, 50, 9373–9379. [Google Scholar] [CrossRef]
- Lee, J.; Mishra, A.K.; Choi, C.; Kim, D.; Kim, E.Y.; Yong, K.; Kim, J.K. Three-Dimensional Nanoporous Metal Structures from Poly(2-vinylpyridine)-block-Poly(4-vinylpyridine) Copolymer Thin Film. ACS Appl. Mater. Interfaces 2020, 12, 15667–15674. [Google Scholar] [CrossRef]
- Mun, J.H.; Cha, S.K.; Kim, Y.C.; Yun, T.; Choi, Y.J.; Jin, H.M.; Lee, J.E.; Jeon, H.U.; Kim, S.Y.; Kim, S.O. Controlled Segmentation of Metal Nanowire Array by Block Copolymer Lithography and Reversible Ion Loading. Small 2017, 13, 1603939. [Google Scholar] [CrossRef]
- Shin, D.O.; Lee, D.H.; Moon, H.S.; Jeong, S.J.; Kim, J.Y.; Mun, J.H.; Cho, H.; Park, S.; Kim, S.O. Sub-nanometer level size tuning of a monodisperse nanoparticle array via block copolymer lithography. Adv. Funct. Mater. 2011, 21, 250–254. [Google Scholar] [CrossRef]
- Alvarez-Fernandez, A.; Nallet, F.; Fontaine, P.; Cummins, C.; Hadziioannou, G.; Barois, P.; Fleury, G.; Ponsinet, V. Large area Al2O3-Au raspberry-like nanoclusters from iterative block-copolymer self-assembly. RSC Adv. 2020, 10, 41088–41097. [Google Scholar] [CrossRef]
- Liu, R.; Huang, H.; Sun, Z.; Alexander-Katz, A.; Ross, C.A. Metallic nanomeshes fabricated by multimechanism directed self-assembly. ACS Nano 2021, 15, 16266–16276. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.C.; Kim, S.Y. Fabrication of gold nanowires in micropatterns using block copolymers. RSC Adv. 2018, 8, 19532–19538. [Google Scholar] [CrossRef] [PubMed]
- Langer, J.; Jimenez de Aberasturi, D.; Aizpurua, J.; Alvarez-Puebla, R.A.; Auguié, B.; Baumberg, J.J.; Bazan, G.C.; Bell, S.E.; Boisen, A.; Brolo, A.G.; et al. Present and future of surface-enhanced Raman scattering. ACS Nano 2019, 14, 28–117. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, T.; Harada, M.; Sakamoto, N. Incorporation of metal nanoparticles into block copolymer nanodomains via in-situ reduction of metal ions in microdomain space. Macromolecules 1999, 32, 6867–6870. [Google Scholar] [CrossRef]
- Hsu, S.W.; Rodarte, A.L.; Som, M.; Arya, G.; Tao, A.R. Colloidal plasmonic nanocomposites: From fabrication to optical function. Chem. Rev. 2018, 118, 3100–3120. [Google Scholar] [CrossRef] [PubMed]
- Zito, G.; Rusciano, G.; Vecchione, A.; Pesce, G.; Di Girolamo, R.; Malafronte, A.; Sasso, A. Nanometal skin of plasmonic heterostructures for highly efficient near-field scattering probes. Sci. Rep. 2016, 6, 31113. [Google Scholar] [CrossRef]
- Zhang, J.; Gao, Y.; Alvarez-Puebla, R.A.; Buriak, J.M.; Fenniri, H. Synthesis and SERS properties of nanocrystalline gold octahedra generated from thermal decomposition of HAuCl4 in block copolymers. Adv. Mater. 2016, 18, 3233–3237. [Google Scholar] [CrossRef]
- Glass, R.; Arnold, M.; Cavalcanti-Adam, E.A.; Blümmel, J.; Haferkemper, C.; Dodd, C.; Spatz, J.P. Block Copolymer Micelle Nanolithography on Non-Conductive Substrates. New J. Phys. 2004, 6, 101. [Google Scholar] [CrossRef]
- Sohn, B.H.; Seo, B.H. Fabrication of the multilayered nanostructure of alternating polymers and gold nanoparticles with thin films of self-assembling diblock copolymers. Chem. Mater. 2001, 13, 1752–1757. [Google Scholar] [CrossRef]
- Haridas, M.; Basu, J.K. Controlled photoluminescence from self-assembled semiconductor–metal quantum dot hybrid array films. Nanotechnology 2010, 21, 415202. [Google Scholar] [CrossRef]
- Jin, H.M.; Lee, S.E.; Kim, S.; Kim, J.Y.; Han, Y.S.; Kim, B.H. Directed high-χ block copolymer self-assembly by laser writing on silicon substrate. J. Appl. Polym. Sci. 2022, 139, 52291. [Google Scholar] [CrossRef]
- El-Nagar, G.A.; Muench, F.; Roth, C. Tailored Dendritic Platinum Nanostructures as a Robust and Efficient Direct Formic Acid Fuel Cell Anode. New J. Chem. 2019, 43, 4100–4105. [Google Scholar] [CrossRef]
- Berman, D.; Shevchenko, E. Design of Functional Composite and All-Inorganic Nanostructured Materials via Infiltration of Polymer Templates with Inorganic Precursors. J. Mater. Chem. C 2020, 8, 10604–10627. [Google Scholar] [CrossRef]
- Yang, H.; Xu, L.; Li, X.; Zhang, X. One Simple Route to Fabricate Gold Nanowire Network Films by Using P2VP-b-PEO Diblock Copolymers as Templates. Mater. Chem. Phys. 2009, 114, 525–529. [Google Scholar] [CrossRef]
- Mbenkum, B.N.; Schneider, A.S.; Schütz, G.; Xu, C.; Richter, G.; van Aken, P.A.; Majer, G.; Spatz, J.P. Low-Temperature Growth of Silicon Nanotubes and Nanowires on Amorphous Substrates. ACS Nano 2010, 4, 1805–1812. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Fernandez, A.; Cummins, C.; Saba, M.; Steiner, U.; Fleury, G.; Ponsinet, V.; Guldin, S. Block Copolymer Directed Metamaterials and Metasurfaces for Novel Optical Devices. Adv. Opt. Mat. 2021, 9, 2100175. [Google Scholar] [CrossRef]
- Robbins, S.W.; Beaucage, P.A.; Sai, H.; Tan, K.W.; Werner, J.G.; Sethna, J.P.; Disalvo, F.J.; Gruner, S.M.; Van Dover, R.B.; Wieser, U. Block copolymer self-assembly–directed synthesis of mesoporous gyroidal superconductors. Sci. Adv. 2016, 2, e1501119. [Google Scholar] [CrossRef] [PubMed]
- Kang, D.H.; Han, W.B.; Ryu, H.I.; Kim, N.H.; Kim, T.; Choi, N.; Kang, J.Y.; Yu, Y.G.; Kim, T.S. Tunable and scalable fabrication of block copolymer-based 3D polymorphic artificial cell membrane array. Nat. Commun. 2022, 13, 1261. [Google Scholar] [CrossRef]
- Kim, J.H.; Jeon, S.; In, J.H.; Nam, S.; Jin, H.M.; Han, K.H.; Yang, G.G.; Choi, H.J.; Kim, K.M.; Shin, J.; et al. Nanoscale Physical Unclonable Function Labels Based on Block Co-polymer Self-assembly. Nat. Electron. 2022, 5, 433–442. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Murataj, I.; Cara, E.; Baglieri, N.; Pirri, C.F.; De Leo, N.; Ferrarese Lupi, F. Liquid Phase Infiltration of Block Copolymers. Polymers 2022, 14, 4317. https://doi.org/10.3390/polym14204317
Murataj I, Cara E, Baglieri N, Pirri CF, De Leo N, Ferrarese Lupi F. Liquid Phase Infiltration of Block Copolymers. Polymers. 2022; 14(20):4317. https://doi.org/10.3390/polym14204317
Chicago/Turabian StyleMurataj, Irdi, Eleonora Cara, Nicoletta Baglieri, Candido Fabrizio Pirri, Natascia De Leo, and Federico Ferrarese Lupi. 2022. "Liquid Phase Infiltration of Block Copolymers" Polymers 14, no. 20: 4317. https://doi.org/10.3390/polym14204317
APA StyleMurataj, I., Cara, E., Baglieri, N., Pirri, C. F., De Leo, N., & Ferrarese Lupi, F. (2022). Liquid Phase Infiltration of Block Copolymers. Polymers, 14(20), 4317. https://doi.org/10.3390/polym14204317