Structural Characterisation and Assessment of the Novel Bacillus amyloliquefaciens RK3 Exopolysaccharide on the Improvement of Cognitive Function in Alzheimer’s Disease Mice
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Collection and Isolation
2.2. Biochemical and Molecular Identification of the Bacterial Strain
2.3. Production Extraction and Purification of EPS
2.4. Characterisation of the EPS
2.4.1. Estimation of Total Carbohydrates
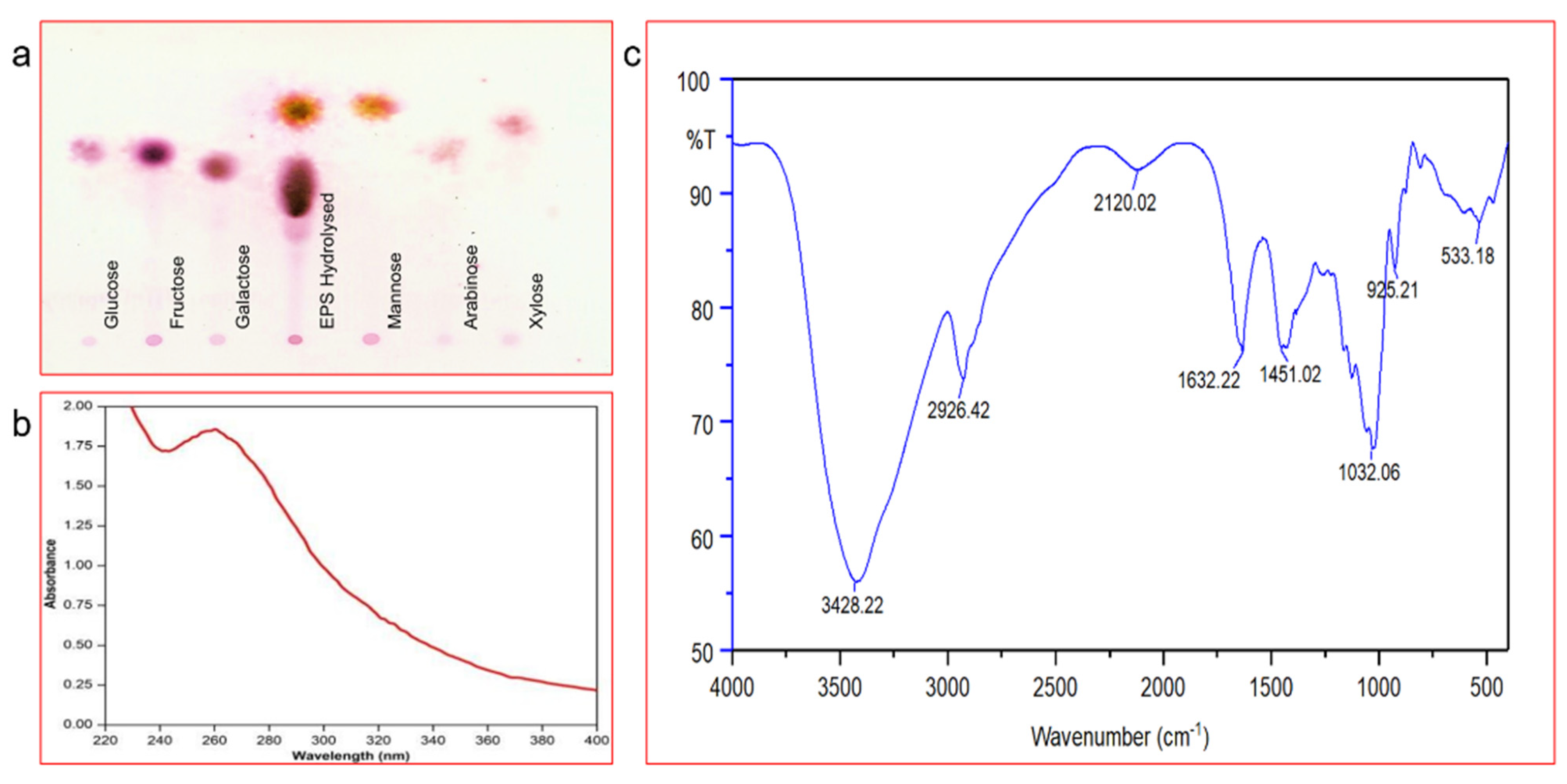
2.4.2. Thin-Layer Chromatography Analysis of EPS
2.4.3. UV-Visible Spectra, FT-IR and GC/MS Analysis of Purified EPS
2.4.4. NMR and SEM Analysis
2.5. Anti-Alzheimer Study of the EPS
2.5.1. Anti-Alzheimer Activity in Mice Models
2.5.2. Cognition and Behaviour Analysis of Mice Models
3. Results and Discussion
3.1. Isolation and Identification of the EPS Producing Strain
3.2. Production and Characterisation of the EPS
3.2.1. Yield and Carbohydrate Content
3.2.2. Estimation of Total Carbohydrates and Monosaccharide Composition
3.2.3. UV-Visible Spectra and FT-IR Spectra
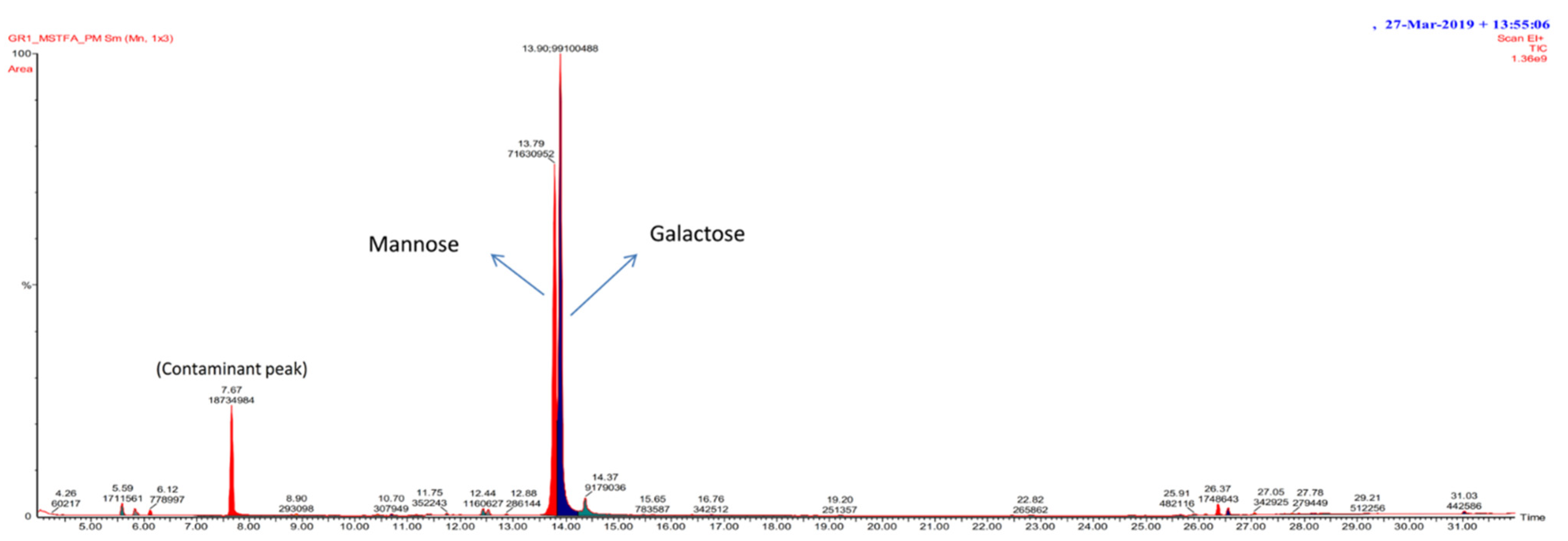
3.2.4. GC/MS Spectra of Exopolysaccharide
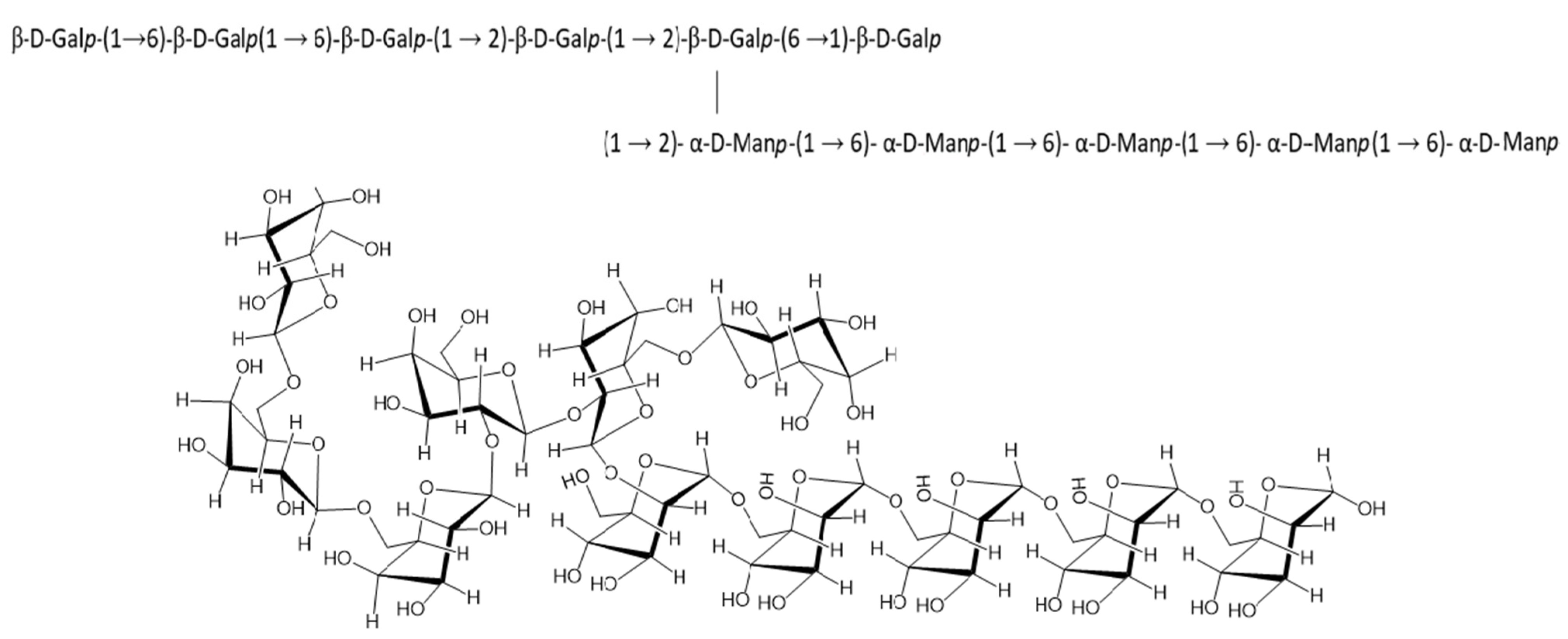
Linkage Analysis of GC/MS
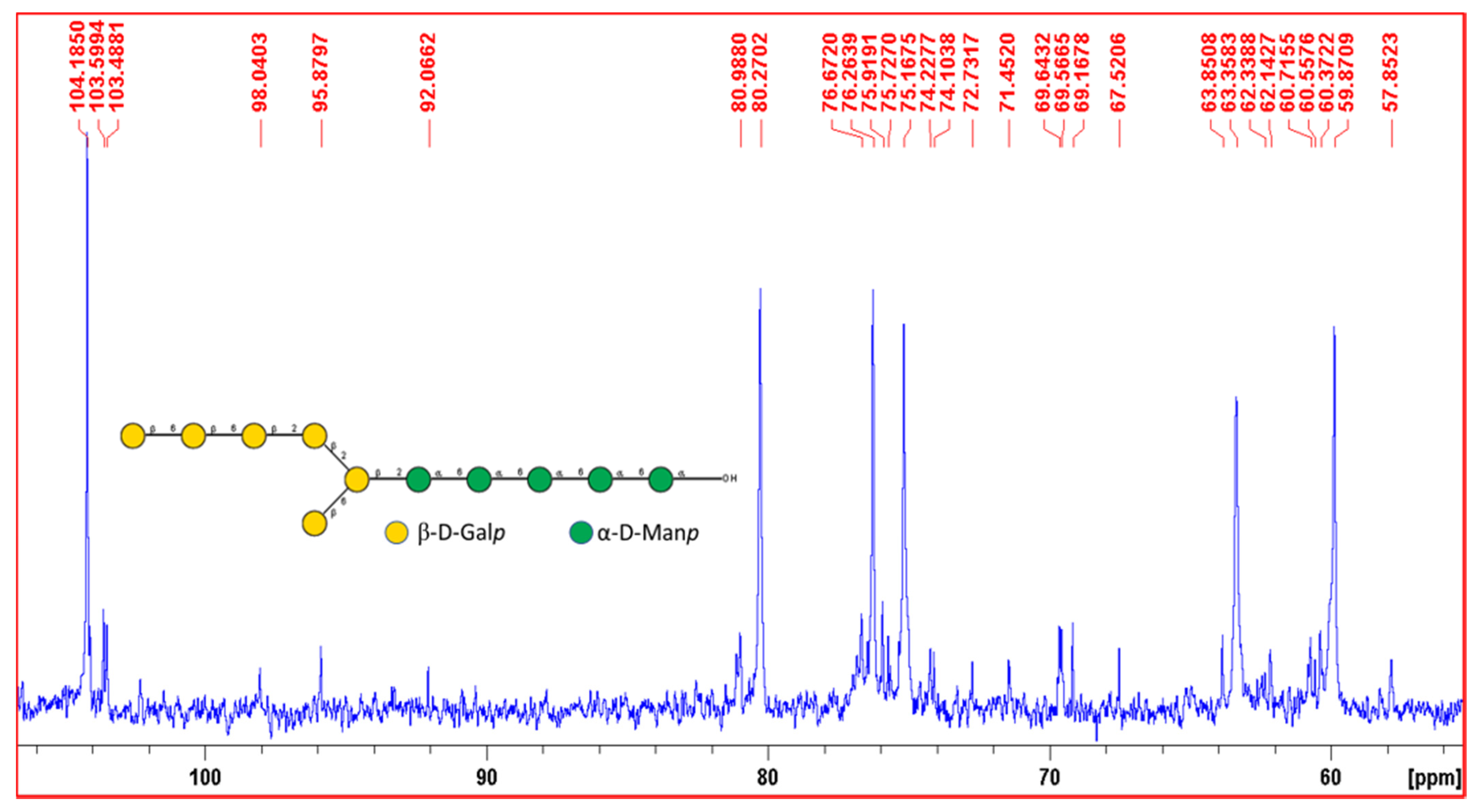
3.2.5. 1H and 13C NMR Spectra Interpretation of EPS
3.2.6. SEM Analysis of the EPS
3.3. Anti-Alzheimer Study
3.3.1. EPS Significantly Reduce the Convolutions of Alzheimer in Mice
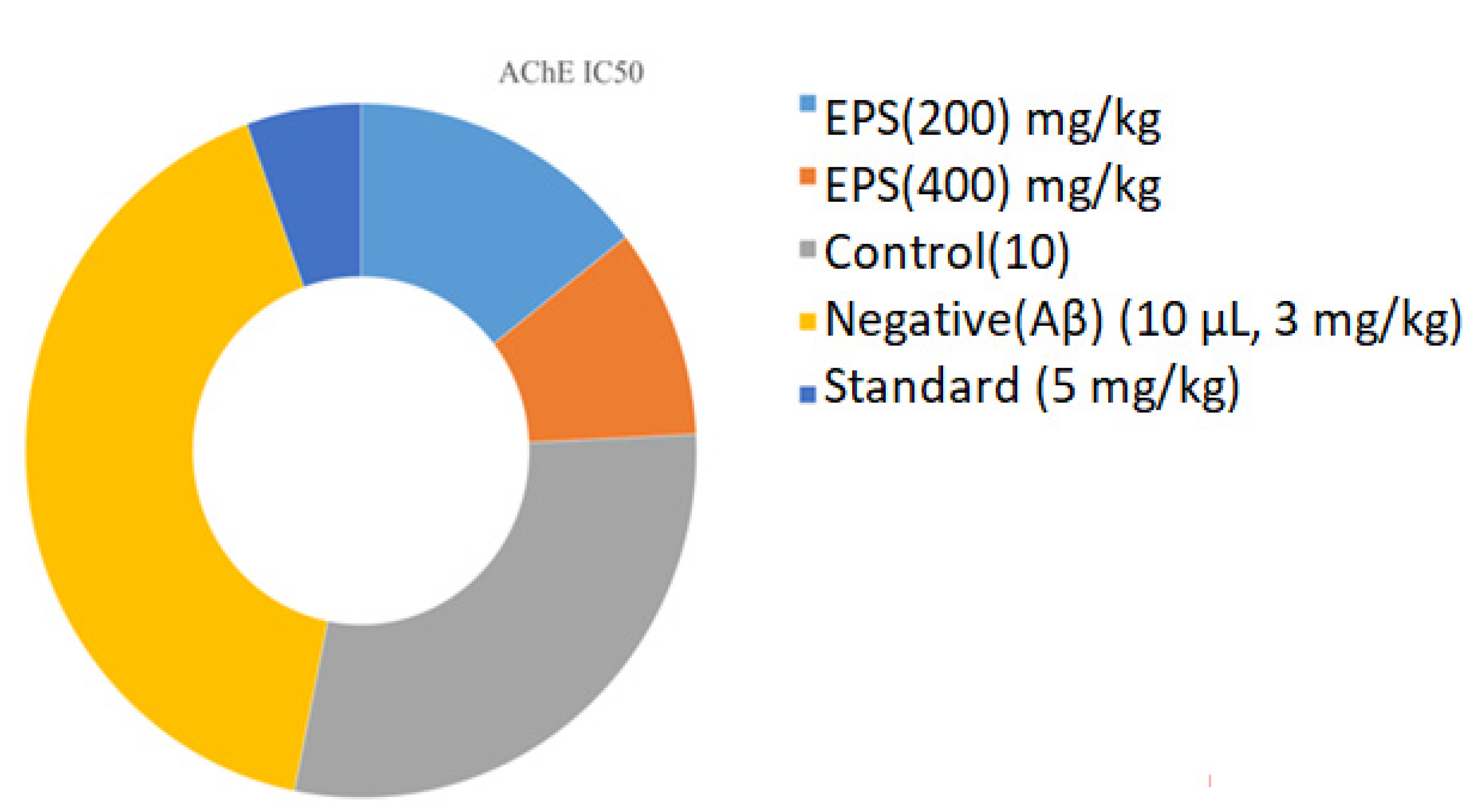
3.3.2. Acetylcholine Esterase Activity
3.4. Effects of EPS on Behavior and Cognition of Mice Model
3.4.1. Jumping Box Test
3.4.2. Rectangular Maze and Y Maze Results
3.5. Histopathology of Mice Brain
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, J.; Salem, D.R.; Sani, R.K. Extremophilic exopolysaccharides: A review and new perspectives on engineering strategies and applications. Carbohydr. Polym. 2019, 205, 8–26. [Google Scholar] [CrossRef]
- Decho, A.W.; Gutierrez, T. Microbial extracellular polymeric substances (EPSs) in ocean systems. Front. Microbiol. 2017, 8, 922. [Google Scholar] [CrossRef]
- Ates, O. Systems Biology of Microbial Exopolysaccharides Production. Front. Bioeng. Biotechnol. 2015, 3, 200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wold, C.W.; Kjeldsen, C.; Corthay, A.; Rise, F.; Christensen, B.E.; Duus, J.Ø.; Inngjerdingen, K.T. Structural characterization of bioactive heteropolysaccharides from the medicinal fungus Inonotus obliquus (Chaga). Carbohydr. Polym. 2018, 185, 27–40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nishimura, J. Preparation of exopolysaccharide synthesized by lactic acid bacteria. In Methods in Molecular Biology; Humana Press Inc.: New York, NY, USA, 2019; Volume 1887, pp. 85–94. [Google Scholar]
- Andrew, M.; Jayaraman, G. Structural features of microbial exopolysaccharides in relation to their antioxidant activity. Carbohydr. Res. 2020, 487, 107881. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.; Huang, L.; Li, K. Antioxidant activity changes of exopolysaccharides with different carbon sources from Lactobacillus plantarum LPC-1 and its metabolomic analysis. World J. Microbiol. Biotechnol. 2019, 35, 68. [Google Scholar] [CrossRef] [PubMed]
- Malick, A.; Khodaei, N.; Benkerroum, N.; Karboune, S. Production of exopolysaccharides by selected Bacillus strains: Optimization of media composition to maximize the yield and structural characterization. Int. J. Biol. Macromol. 2017, 102, 539–549. [Google Scholar] [CrossRef] [PubMed]
- Devarajan, T.; Jeyabalan, S. Industrial Biotechnology: Sustainable Production and Bioresource Utilization; CRC Press: Boca Raton, FL, USA, 2017; ISBN 9781771882620. [Google Scholar]
- Gupta, P.; Diwan, B. Bacterial Exopolysaccharide mediated heavy metal removal: A Review on biosynthesis, mechanism and remediation strategies. Biotechnol. Rep. 2017, 13, 58–71. [Google Scholar] [CrossRef]
- Yildiz, H.; Karatas, N. Microbial exopolysaccharides: Resources and bioactive properties. Process. Biochem. 2018, 72, 41–46. [Google Scholar] [CrossRef]
- Freitas, F.; Torres, C.A.V.; Araújo, D.; Farinha, I.; Pereira, J.R.; Concórdio-Reis, P.; Reis, M.A.M. Advanced Microbial Polysaccharides. In Biopolymers for Biomedical and Biotechnological Applications; Wiley: Hoboken, NJ, USA, 2021; pp. 19–62. [Google Scholar]
- Asgher, M.; Qamar, S.A.; Iqbal, H.M.N. Microbial exopolysaccharide-based nano-carriers with unique multi-functionalities for biomedical sectors. Biologia 2021, 76, 673–685. [Google Scholar] [CrossRef]
- Moscovici, M. Present and future medical applications of microbial exopolysaccharides. Front. Microbiol. 2015, 6, 1012. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hinchliffe, J.D.; Madappura, A.P.; Syed Mohamed, S.M.D.; Roy, I. Biomedical applications of bacteria-derived polymers. Polymers 2021, 13, 1081. [Google Scholar] [CrossRef] [PubMed]
- Pillay, V.; Choonara, Y.E.; Kumar, P. (Eds.) Unfolding the Biopolymer Landscape; Bentham Science Publishers: Sharjah, United Arab Emirates, 2016; Volume 2, ISBN 9781681081953. [Google Scholar]
- Michael, N. Biopolymers: Applications and Trends; William Andrew: Norwich, NY, USA, 2015; ISBN 9780323354332. [Google Scholar]
- Czaja, W.K.; Young, D.J.; Kawecki, M.; Brown, R.M. The future prospects of microbial cellulose in biomedical applications. Biomacromolecules 2007, 8, 1–12. [Google Scholar] [CrossRef]
- Wei, J.; Wang, B.; Li, Z.; Wu, Z.; Zhang, M.; Sheng, N.; Liang, Q.; Wang, H.; Chen, S. A 3D-printable TEMPO-oxidized bacterial cellulose/alginate hydrogel with enhanced stability via nanoclay incorporation. Carbohydr. Polym. 2020, 238, 116207. [Google Scholar] [CrossRef]
- Gupta, P.L.; Rajput, M.; Oza, T.; Trivedi, U.; Sanghvi, G. Eminence of Microbial Products in Cosmetic Industry. Nat. Prod. Bioprospect. 2019, 9, 267–278. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krishnaswamy, U.R.; Lakshmanaperumalsamy, P.; Achlesh, D. Microbial Exopolysaccharides as Biosurfactants in Environmental and Industrial Applications. In Advances in the Domain of Environmental Biotechnology; Springer: Singapore, 2021; pp. 81–111. [Google Scholar] [CrossRef]
- Xu, M.; Qin, M.; Cheng, Y.; Niu, X.; Kong, J.; Zhang, X.; Huang, D.; Wang, H. Alginate microgels as delivery vehicles for cell-based therapies in tissue engineering and regenerative medicine. Carbohydr. Polym. 2021, 266, 118128. [Google Scholar] [CrossRef] [PubMed]
- Suresh, S.; Singh, G.; Hanumanthaiah, K.; Venkatesh, K.; Kadeppagari, R.K. Alginate, Its Chemistry and Applications in Advanced Drug Delivery. In Polysaccharides in Advanced Drug Delivery; CRC Press: Boca Raton, FL, USA, 2020; pp. 131–146. [Google Scholar]
- Vaz, M.; Silvestre, S. Alzheimer’s disease: Recent treatment strategies. Eur. J. Pharmacol. 2020, 887, 173554. [Google Scholar] [CrossRef]
- Kumar, A.; Pintus, F.; Di Petrillo, A.; Medda, R.; Caria, P.; Matos, M.J.; Viña, D.; Pieroni, E.; Delogu, F.; Era, B.; et al. Novel 2-pheynlbenzofuran derivatives as selective butyrylcholinesterase inhibitors for Alzheimer’s disease. Sci. Rep. 2018, 8, 4424. [Google Scholar] [CrossRef] [Green Version]
- Chen, D.; Gao, H.; Peng, C.; Pei, S.; Dai, A.; Yu, X.; Zhou, P.; Wang, Y.; Cai, B. Quinones as preventive agents in Alzheimer’s diseases: Focus on NLRP3 inflammasomes. J. Pharm. Pharmacol. 2020, 72, 1481–1490. [Google Scholar] [CrossRef]
- Sivasankar, P. Marine Psychrophilic and Psychrotolerant Actinobacteria of the Polar Frontal Waters of the Southern Ocean, Their Diversity and Cryoprotective Potentials; Annamalai University: Chidambaram, India, 2015. [Google Scholar]
- Behera, H.T.; Mojumdar, A.; Mohapatra, C.; Ray, L. Microbial Polysaccharides with Potential Industrial Applications: Diversity, Synthesis, and Their Applications. In Microbial Polymers; Springer: Singapore, 2021; pp. 521–546. [Google Scholar]
- Selvaraj, C.; Singh, S.K. Eco-friendly Microbial Biopolymers: Recent Development, Biodegradation, and Applications. In Microbial Polymers; Springer: Singapore, 2021; pp. 547–577. [Google Scholar]
- Jing, X.; Sun, Y.; Ma, X.; Hu, H. Marine polysaccharides: Green and recyclable resources as wound dressings. Mater. Chem. Front. 2021. [Google Scholar] [CrossRef]
- Williams, P.A.; Phillips, G.O. Introduction to food hydrocolloids. In Handbook of Hydrocolloids; Elsevier, Woodhead Publishing: Sawston, UK, 2021; pp. 3–26. [Google Scholar]
- Jamróz, E.; Kulawik, P.; Kopel, P. The effect of nanofillers on the functional properties of biopolymer-based films: A review. Polymers 2019, 11, 675. [Google Scholar] [CrossRef] [Green Version]
- Costa, O.Y.A.; Raaijmakers, J.M.; Kuramae, E.E. Microbial extracellular polymeric substances: Ecological function and impact on soil aggregation. Front. Microbiol. 2018, 9, 1636. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ullah, M.W.; Ul-Islam, M.; Khan, T.; Park, J.K. Recent developments in the synthesis, properties, and applications of various microbial polysaccharides. In Handbook of Hydrocolloids; Elsevier, Woodhead Publishing: Sawston, UK, 2021; pp. 975–1015. [Google Scholar]
- Shanmugam, M.; Abirami, R.G. Microbial Polysaccharides—Chemistry and Applications. J. Biol. Act. Prod. Nat. 2019, 9, 73–78. [Google Scholar] [CrossRef]
- Jindal, N.; Singh Khattar, J. Microbial Polysaccharides in Food Industry. In Biopolymers for Food Design; Elsevier, Academic Press: Cambridge, MA, USA, 2018; pp. 95–123. ISBN 9780128115015. [Google Scholar]
- Glicksman, M. Dextran. In Food Hydrocolloids; CRC Press: Boca Raton, FL, USA, 2020; pp. 157–166. [Google Scholar]
- Stackebrandt, E. The Family Nocardiopsaceae. In The Prokaryotes: Actinobacteria; Rosenberg, E., DeLong, E.F., Lory, S., Stackebrandt, E., Thompson, F., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 695–700. ISBN 9783642301384. [Google Scholar]
- Sivasankar, P.; Manivasagan, P.; Vijayanand, P.; Sivakumar, K.; Sugesh, S.; Poongodi, S.; Maharani, V.; Vijayalakshmi, S.; Balasubramanian, T.; Palaniappan, S.; et al. Antibacterial and brine shrimp lethality effect of marine actinobacterium Streptomyces sp. CAS72 against human pathogenic bacteria. Asian Pac. J. Trop. Dis. 2013, 3, 286–293. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547. [Google Scholar] [CrossRef] [PubMed]
- Sivasankar, P.; Seedevi, P.; Poongodi, S.; Sivakumar, M.; Murugan, T.; Sivakumar, L.; Sivakumar, K.; Balasubramanian, T. Characterization, antimicrobial and antioxidant property of exopolysaccharide mediated silver nanoparticles synthesized by Streptomyces violaceus MM72. Carbohydr. Polym. 2018, 181, 752–759. [Google Scholar] [CrossRef] [PubMed]
- Sun, K.; Chen, Y.; Niu, Q.; Zhu, W.; Wang, B.; Li, P.; Ge, X. An exopolysaccharide isolated from a coral-associated fungus and its sulfated derivative activates macrophages. Int. J. Biol. Macromol. 2016, 82, 387–394. [Google Scholar] [CrossRef] [PubMed]
- DuBois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric Method for Determination of Sugars and Related Substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Kim, S.H.; Nam, S.H.; Yoo, S.J.; Yang, K.Y. Isolation and characterization of exopolysaccharide-producing Weissella cibaria JNU29 and its application for production of Korean rice cake ‘Sulgidduk’. Korean J. Food Preserv. 2020. [Google Scholar] [CrossRef]
- Sivasankar, P.; Poongodi, S.; Lobo, A.O.; Pugazhendhi, A. Characterization of a novel polymeric bioflocculant from marine actinobacterium Streptomyces sp. and its application in recovery of microalgae. Int. Biodeterior. Biodegrad. 2020, 148, 104883. [Google Scholar] [CrossRef]
- Sun, M.-L.; Zhao, F.; Shi, M.; Zhang, X.-Y.; Zhou, B.-C.; Zhang, Y.-Z.; Chen, X.-L. Characterization and Biotechnological Potential Analysis of a New Exopolysaccharide from the Arctic Marine Bacterium Polaribacter sp. SM1127. Sci. Rep. 2015, 5, 18435. [Google Scholar] [CrossRef] [Green Version]
- Singh, R.; Thota, S.; Bansal, R. Studies on 16,17-Pyrazoline Substituted Heterosteroids as Anti-Alzheimer and Anti-Parkinsonian Agents Using LPS Induced Neuroinflammation Models of Mice and Rats. ACS Chem. Neurosci. 2018, 9, 272–283. [Google Scholar] [CrossRef]
- Luo, W.; Wang, T.; Hong, C.; Yang, Y.C.; Chen, Y.; Cen, J.; Xie, S.Q.; Wang, C.J. Design, synthesis and evaluation of 4-dimethylamine flavonoid derivatives as potential multifunctional anti-Alzheimer agents. Eur. J. Med. Chem. 2016, 122, 17–26. [Google Scholar] [CrossRef]
- Yarube, I.; Ayo, J.; Magaji, R.; Umar, I. Insulin treatment increases brain nitric oxide and oxidative stress, but does not affect memory function in mice. Physiol. Behav. 2019, 211, 112640. [Google Scholar] [CrossRef]
- Alam, M.N.; Hossain, M.M.; Rahman, M.M.; Subhan, N.; Al Mamun, M.A.; Ulla, A.; Reza, H.M.; Alam, M.A. Astaxanthin Prevented Oxidative Stress in Heart and Kidneys of Isoproterenol-Administered Aged Rats. J. Diet. Suppl. 2018, 15, 42–54. [Google Scholar] [CrossRef] [PubMed]
- Tanga, N.; Kuboyama, K.; Kishimoto, A.; Kihara, M.; Kiyonari, H.; Watanabe, T.; Fujikawa, A.; Noda, M. Behavioral and neurological analyses of adult mice carrying null and distinct loss-of-receptor function mutations in protein tyrosine phosphatase receptor type Z (PTPRZ). PLoS ONE 2019, 14, e0217880. [Google Scholar] [CrossRef] [PubMed]
- Mazumder, M.K.; Paul, R.; Bhattacharya, P.; Borah, A. Neurological sequel of chronic kidney disease: From diminished Acetylcholinesterase activity to mitochondrial dysfunctions, oxidative stress and inflammation in mice brain. Sci. Rep. 2019, 9, 3097. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ibrahim, K.; Al-Mutary, M.; Bakhiet, A.; Khan, H. Histopathology of the Liver, Kidney, and Spleen of Mice Exposed to Gold Nanoparticles. Molecules 2018, 23, 1848. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vidhyalakshmi, R.; Valli Nachiyar, C.; Narendra Kumar, G.; Sunkar, S. Bacillus circulans exopolysaccharide: Production, characterization and bioactivities. Int. J. Biol. Macromol. 2016, 87, 405–414. [Google Scholar] [CrossRef] [PubMed]
- Bounaix, M.S.; Gabriel, V.; Morel, S.; Robert, H.; Rabier, P.; Remaud-Siméon, M.; Gabriel, B.; Fontagné-Faucher, C. Biodiversity of exopolysaccharides produced from sucrose by sourdough lactic acid bacteria. J. Agric. Food Chem. 2009, 57, 10889–10897. [Google Scholar] [CrossRef]
- Garrity, G.M.; Parker, C.T.; Tindall, B.J. International Code of Nomenclature of Prokaryotes. Int. J. Syst. Evol. Microbiol. 2015, 69, S1–S111. [Google Scholar] [CrossRef]
- Zhao, D.; Jiang, J.; Du, R.; Guo, S.; Ping, W.; Ling, H.; Ge, J. Purification and characterization of an exopolysaccharide from Leuconostoc lactis L2. Int. J. Biol. Macromol. 2019, 139, 1224–1231. [Google Scholar] [CrossRef] [PubMed]
- Palaniyandi, S.A.; Damodharan, K.; Suh, J.W.; Yang, S.H. Functional characterization of an exopolysaccharide produced by bacillus sonorensis MJM60135 isolated from ganjang. J. Microbiol. Biotechnol. 2018, 28, 663–670. [Google Scholar] [CrossRef]
- Bales, P.M.; Renke, E.M.; May, S.L.; Shen, Y.; Nelson, D.C. Purification and Characterization of Biofilm-Associated EPS Exopolysaccharides from ESKAPE Organisms and Other Pathogens. PLoS ONE 2013, 8, e67950. [Google Scholar] [CrossRef] [Green Version]
- Shingel, K.I. Determination of structural peculiarities of dexran, pullulan and γ-irradiated pullulan by Fourier-transform IR spectroscopy. Carbohydr. Res. 2002, 337, 1445–1451. [Google Scholar] [CrossRef]
- Sun, H.; Mu, T.; Xi, L.; Zhang, M.; Chen, J. Sweet potato (Ipomoea batatas L.) leaves as nutritional and functional foods. Food Chem. 2014, 156, 380–389. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Fan, Q.; Zhang, X.; Lu, X.; Xu, Y.; Zhu, W.; Zhang, J.; Hao, W.; Hao, L. Isolation, characterization, and pharmaceutical applications of an exopolysaccharide from aerococcus uriaeequi. Mar. Drugs 2018, 16, 337. [Google Scholar] [CrossRef] [Green Version]
- Ruiz-Matute, A.I.; Hernández-Hernández, O.; Rodríguez-Sánchez, S.; Sanz, M.L.; Martínez-Castro, I. Derivatization of carbohydrates for GC and GC-MS analyses. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2011, 879, 1226–1240. [Google Scholar] [CrossRef]
- Schauer, N.; Steinhauser, D.; Strelkov, S.; Schomburg, D.; Allison, G.; Moritz, T.; Lundgren, K.; Roessner-Tunali, U.; Forbes, M.G.; Willmitzer, L.; et al. GC-MS libraries for the rapid identification of metabolites in complex biological samples. FEBS Lett. 2005, 579, 1332–1337. [Google Scholar] [CrossRef]
- Wang, J.; Wu, T.; Fang, X.; Min, W.; Yang, Z. Characterization and immunomodulatory activity of an exopolysaccharide produced by Lactobacillus plantarum JLK0142 isolated from fermented dairy tofu. Int. J. Biol. Macromol. 2018, 115, 985–993. [Google Scholar] [CrossRef]
- Sran, K.S.; Sundharam, S.S.; Krishnamurthi, S.; Roy Choudhury, A. Production, characterization and bio-emulsifying activity of a novel thermostable exopolysaccharide produced by a marine strain of Rhodobacter johrii CDR-SL 7Cii. Int. J. Biol. Macromol. 2019, 127, 240–249. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Kumar, M.; Pandey, A.; Thakur, I.S. Genomic analysis of carbon dioxide sequestering bacterium for exopolysaccharides production. Sci. Rep. 2019, 9, 4270. [Google Scholar] [CrossRef] [PubMed]
- El-Newary, S.A.; Ibrahim, A.Y.; Asker, M.S.; Mahmoud, M.G.; El Awady, M.E. Production, characterization and biological activities of acidic exopolysaccharide from marine Bacillus amyloliquefaciens 3MS 2017. Asian Pac. J. Trop. Med. 2017, 10, 652–662. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Zhang, J.; Jiang, Y.Y.; Zhao, X.; Hao, X.N.; Li, L.; Yang, Z.N. Characterization and antioxidant activity of the exopolysaccharide produced by Bacillus amyloliquefaciens GSBa-1. J. Microbiol. Biotechnol. 2018, 28, 1282–1292. [Google Scholar] [CrossRef] [Green Version]
- Liu, C.F.; Tseng, K.C.; Chiang, S.S.; Lee, B.H.; Hsu, W.H.; Pan, T.M. Immunomodulatory and antioxidant potential of Lactobacillus exopolysaccharides. J. Sci. Food Agric. 2011, 91, 2284–2291. [Google Scholar] [CrossRef] [PubMed]
- Beynon, L.M.; Richards, J.C.; Perry, M.B. Identification of the Common Antigenic Determinant Shared by Streptococcus pneumoniae Serotypes 35A and 20 Capsular Polysaccharides—Structural Analysis of the Streptococcus pneumoniae Serotype 35A Capsular Polysaccharide. Eur. J. Biochem. 1997, 250, 163–167. [Google Scholar] [CrossRef] [Green Version]
- Beynon, L.M.; Cox, A.D.; Taylor, C.J.; Wilkinson, S.G.; Perry, M.B. Characterization of a lipopolysaccharide O antigen containing two different trisaccharide repeating units from Burkholderia cepacia serotype E (O2). Carbohydr. Res. 1995, 272, 231–239. [Google Scholar] [CrossRef]
- Beynon, L.M.; Richards, J.C.; Perry, M.B.; Kniskern, P.J. Characterization of the capsular antigen of Streptococcus pneumoniae serotype 35B. Can. J. Chem. 2011, 73, 41–48. [Google Scholar] [CrossRef]
- Oxley, D.; Wilkinson, S.G. Structure of a mannan isolated from the lipopolysaccharide of the reference strain (S3255) for a new serogroup of Serratia marcescens. Carbohydr. Res. 1991, 212, 213–217. [Google Scholar] [CrossRef]
- Eserstam, R.; Rajaguru, T.P.; Jansson, P.-E.; Weintraub, A.; Albert, M.J. The structure of the O-chain of the lipopolysaccharide of a prototypal diarrheagenic strain of Hafnia alvei that has characteristics of a new species under the genus Escherichia. Eur. J. Biochem. 2002, 269, 3289–3295. [Google Scholar] [CrossRef]
- Horton, D.; Mols, O.; Walaszek, Z.; Wernau, W.C. Structural and biosynthetic studies on xanthan by 13C-n.m.r. spectroscopy. Carbohydr. Res. 1985, 141, 340–346. [Google Scholar] [CrossRef]
- Brisson, J.-R.; Uhrinova, S.; Woods, R.J.; van der Zwan, M.; Jarrell, H.C.; Paoletti, L.C.; Kasper, D.L.; Jennings, H.J. NMR and Molecular Dynamics Studies of the Conformational Epitope of the Type III Group B Streptococcus Capsular Polysaccharide and Derivatives. Biochemistry 1997, 36, 3278–3292. [Google Scholar] [CrossRef] [PubMed]
- Xisto, M.I.D.S.; Bittencourt, V.C.B.; Liporagi-Lopes, L.C.; Haido, R.M.T.; Mendonça, M.S.A.; Sassaki, G.; Figueiredo, R.T.; Romanos, M.T.V.; Barreto-Bergter, E. O-Glycosylation in Cell Wall Proteins in Scedosporium prolificans Is Critical for Phagocytosis and Inflammatory Cytokines Production by Macrophages. PLoS ONE 2015, 10, e0123189. [Google Scholar] [CrossRef] [PubMed]
- Ikuta, K.; Shibata, N.; Blake, J.S.; Dahl, M.V.; Nelson, R.D.; Hisamichi, K.; Kobayashi, H.; Suzuki, S.; Okawa, Y. NMR study of the galactomannans of Trichophyton mentagrophytes and Trichophyton rubrum. Biochem. J. 1997, 323, 297–305. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kapaev, R.R.; Toukach, P. V GRASS: Semi-automated NMR-based structure elucidation of saccharides. Bioinformatics 2018, 34, 957–963. [Google Scholar] [CrossRef] [PubMed]
- Kavitake, D.; Devi, P.B.; Singh, S.P.; Shetty, P.H. Characterization of a novel galactan produced by Weissella confusa KR780676 from an acidic fermented food. Int. J. Biol. Macromol. 2016, 86, 681–689. [Google Scholar] [CrossRef] [PubMed]
- Badel, S.; Bernardi, T.; Michaud, P. New perspectives for Lactobacilli exopolysaccharides. Biotechnol. Adv. 2011, 29, 54–66. [Google Scholar] [CrossRef]
- Kanamarlapudi, S.L.R.K.; Muddada, S. Characterization of Exopolysaccharide Produced by Streptococcus thermophilus CC30. Biomed Res. Int. 2017, 2017, 4201809. [Google Scholar] [CrossRef] [Green Version]
- Pinto, T.; Lanctôt, K.L.; Herrmann, N. Revisiting the cholinergic hypothesis of behavioral and psychological symptoms in dementia of the Alzheimer’s type. Ageing Res. Rev. 2011, 10, 404–412. [Google Scholar] [CrossRef]
- Singh, M.; Kaur, M.; Chadha, N.; Silakari, O. Hybrids: A new paradigm to treat Alzheimer’s disease. Mol. Divers. 2016, 20, 271–297. [Google Scholar] [CrossRef] [PubMed]
- Farlow, M.R. Do cholinesterase inhibitors slow progression of Alzheimer’s disease? Int. J. Clin. Pract. Suppl. 2002, 127, 37–44. [Google Scholar]
- Hashimoto, M.; Kazui, H.; Matsumoto, K.; Nakano, Y.; Yasuda, M.; Mori, E. Does donepezil treatment slow the progression of hippocampal atrophy in patients with Alzheimer’s disease? Am. J. Psychiatry 2005, 162, 676–682. [Google Scholar] [CrossRef]
- Oh, M.H.; Houghton, P.J.; Whang, W.K.; Cho, J.H. Screening of Korean herbal medicines used to improve cognitive function for anti-cholinesterase activity. Phytomedicine 2004, 11, 544–548. [Google Scholar] [CrossRef] [PubMed]
- Schulz, V. Ginkgo extract or cholinesterase inhibitors in patients with dementia: What clinical trials and guidelines fail to consider. Phytomedicine 2003, 10, 74–79. [Google Scholar] [CrossRef] [PubMed]
- Morgan, D. Water Maze Tasks in Mice Special Reference to Alzheimer’s Transgenic Mice. In Methods of Behavior Analysis in Neuroscience; CRC Press: Boca Raton, FL, USA, 2008; ISBN 9781420052343. [Google Scholar]
- Roghani, M.; Joghataie, M.T.; Jalali, M.R.; Baluchnejadmojarad, T. Time course of changes in passive avoidance and Y-maze performance in male diabetic rats. Iran. Biomed. J. 2006, 10, 99–104. [Google Scholar]
- Nobakht, M.; Najafzadeh, N.; Shargh, B.K. Effects of tamoxifen on morphological and ultrastructural aspects of developing hippocampus of rat. Iran. Biomed. J. 2009, 13, 237–243. [Google Scholar]





| RT | Compound | Area | ~Relative % |
|---|---|---|---|
| 11.31 | 2-linked mannose | 1,715,051 | 7.84 |
| 11.39 | 2-linked galactose | 2,186,404 | 9.99 |
| 11.8 | T-Galactose | 2,327,302 | 10.64 |
| 12.79 | 6-linked mannose | 8,432,888 | 38.54 |
| 12.88 | 6-linked galactose | 5,788,062 | 26.45 |
| 14.3 | 2,6-linked galactose | 1,430,488 | 6.54 |
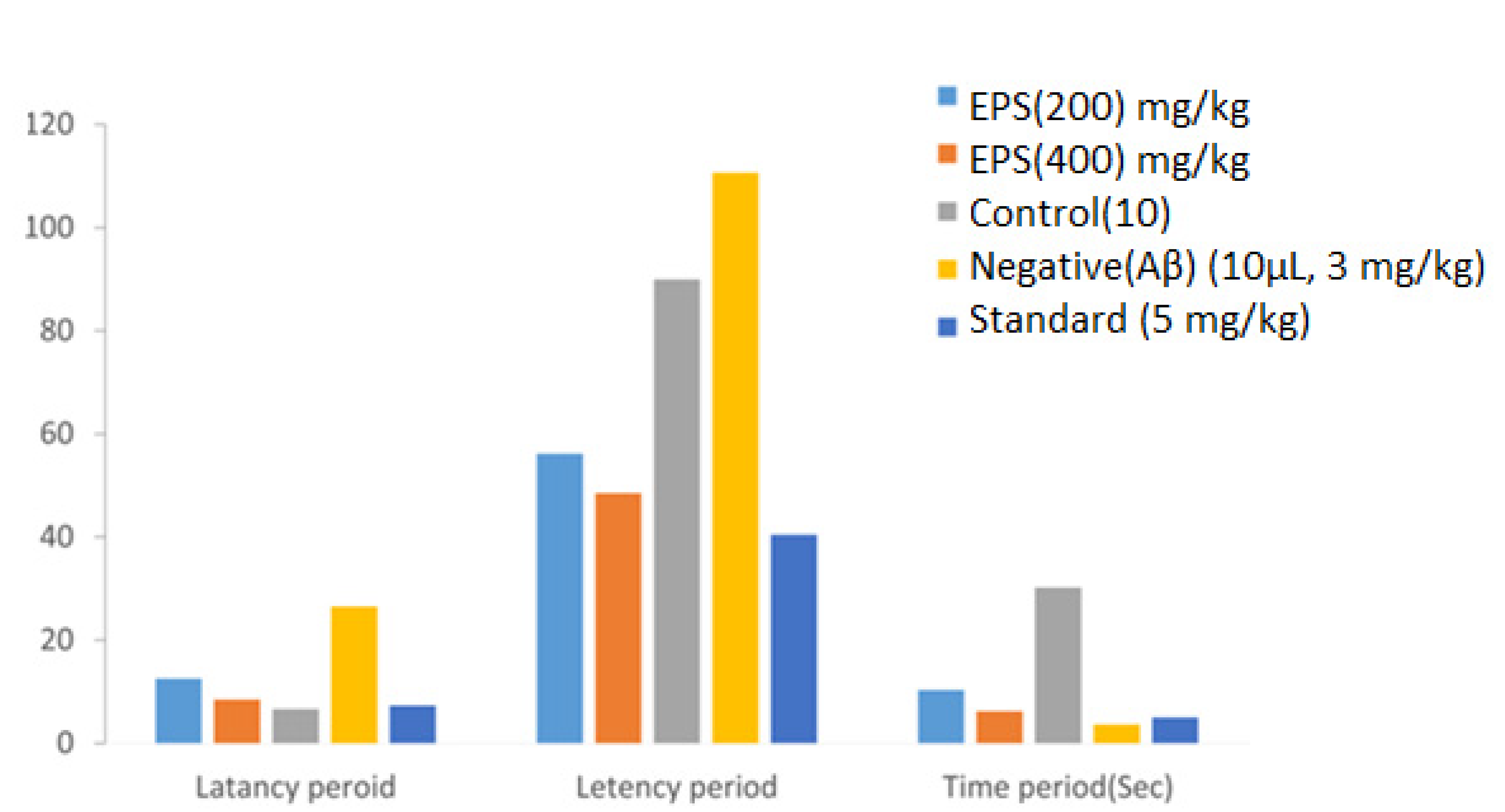
| Samples | Group (mg/kg) | Mean ± SEM (s) |
|---|---|---|
| EPS | 200 | 12.54 ± 0.60 |
| EPS | 400 | 08.48 ± 1.13 |
| Control | 10 | 6.62 ± 0.37 |
| Negative | (Aβ) (10 μL, 3 mg/kg) | 26.51 ± 0.84 |
| Standard | (5 mg/kg) | 7.3 ± 0.15 |
| Rectangular Maze Test | ||
|---|---|---|
| Samples | Group (mg/kg) | Mean ± SEM (s) |
| EPS | 200 | 56.15 ± 0.32 |
| EPS | 400 | 48.48 ± 1.25 |
| Control | 10 | 89.91 ± 0.67 |
| Negative | (Aβ) (10 μL, 3 mg/kg) | 110.6 ± 0.12 |
| Standard | (5 mg/kg) | 40.41 ± 0.83 |
| Y-Maze Test | ||
| Samples | Group (mg/kg) | Mean ± SEM (s) |
| EPS | 200 | 10.31 ± 0.24 |
| EPS | 400 | 6.18 ± 0.35 |
| Control | 10 | 30.21 ± 0.14 |
| Negative | (Aβ) (10 μL, 3 mg/kg) | 3.64 ± 0.24 |
| Standard | (5 mg/kg) | 5.01 ± 0.31 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gangalla, R.; Gattu, S.; Palaniappan, S.; Ahamed, M.; Macha, B.; Thampu, R.K.; Fais, A.; Cincotti, A.; Gatto, G.; Dama, M.; et al. Structural Characterisation and Assessment of the Novel Bacillus amyloliquefaciens RK3 Exopolysaccharide on the Improvement of Cognitive Function in Alzheimer’s Disease Mice. Polymers 2021, 13, 2842. https://doi.org/10.3390/polym13172842
Gangalla R, Gattu S, Palaniappan S, Ahamed M, Macha B, Thampu RK, Fais A, Cincotti A, Gatto G, Dama M, et al. Structural Characterisation and Assessment of the Novel Bacillus amyloliquefaciens RK3 Exopolysaccharide on the Improvement of Cognitive Function in Alzheimer’s Disease Mice. Polymers. 2021; 13(17):2842. https://doi.org/10.3390/polym13172842
Chicago/Turabian StyleGangalla, Ravi, Sampath Gattu, Sivasankar Palaniappan, Maqusood Ahamed, Baswaraju Macha, Raja Komuraiah Thampu, Antonella Fais, Alberto Cincotti, Gianluca Gatto, Murali Dama, and et al. 2021. "Structural Characterisation and Assessment of the Novel Bacillus amyloliquefaciens RK3 Exopolysaccharide on the Improvement of Cognitive Function in Alzheimer’s Disease Mice" Polymers 13, no. 17: 2842. https://doi.org/10.3390/polym13172842
APA StyleGangalla, R., Gattu, S., Palaniappan, S., Ahamed, M., Macha, B., Thampu, R. K., Fais, A., Cincotti, A., Gatto, G., Dama, M., & Kumar, A. (2021). Structural Characterisation and Assessment of the Novel Bacillus amyloliquefaciens RK3 Exopolysaccharide on the Improvement of Cognitive Function in Alzheimer’s Disease Mice. Polymers, 13(17), 2842. https://doi.org/10.3390/polym13172842









